Aβ1-42 and Tau as Potential Biomarkers for Diagnosis and Prognosis of Amyotrophic Lateral Sclerosis
Abstract
1. Introduction
2. Results
2.1. Patients Characteristics
2.2. Aβ1-42, Total Tau, and Phospho-Tau as Diagnosis Biomarkers in ALS
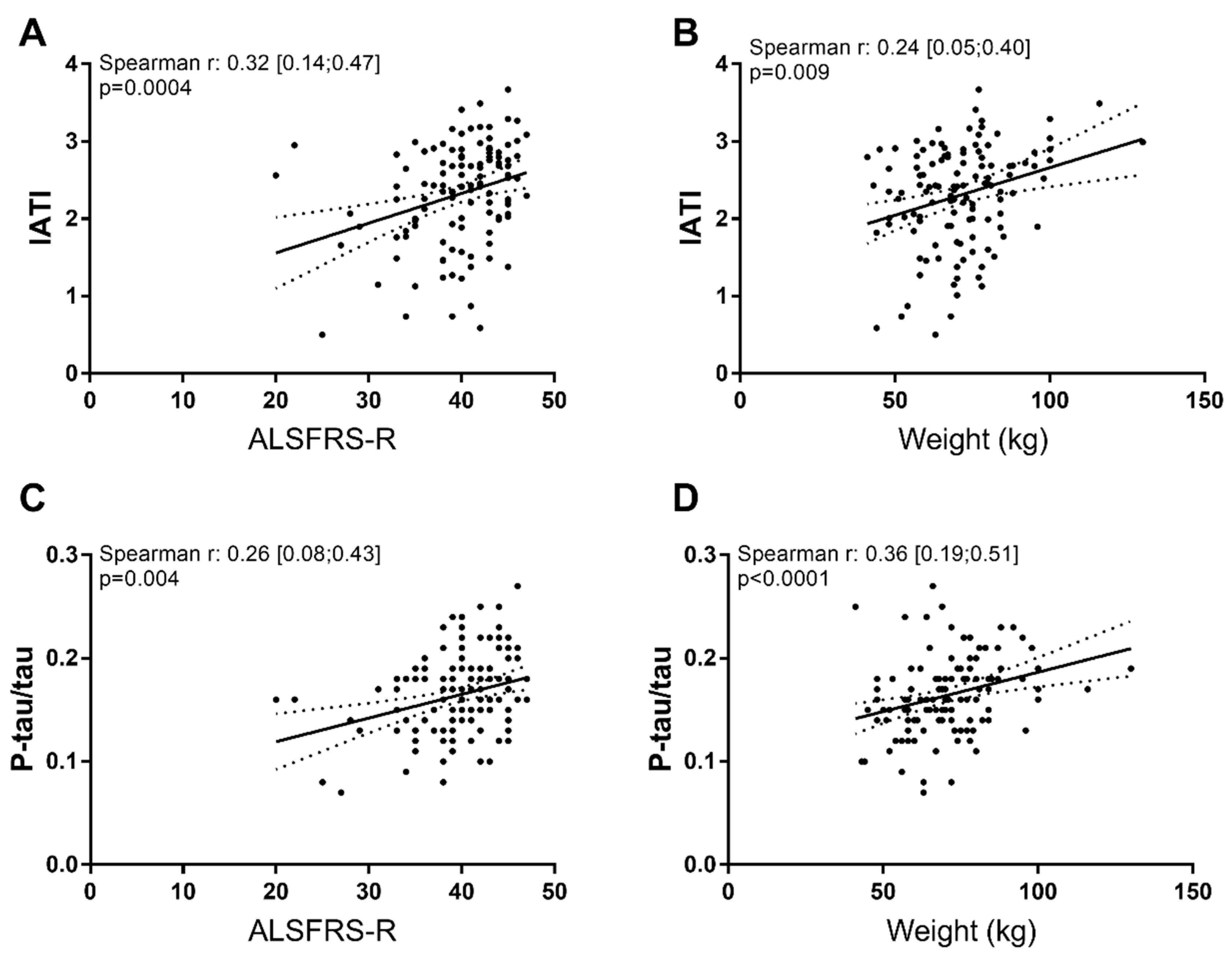
2.3. Correlation Between Biomarkers and Clinical Parameters at Baseline
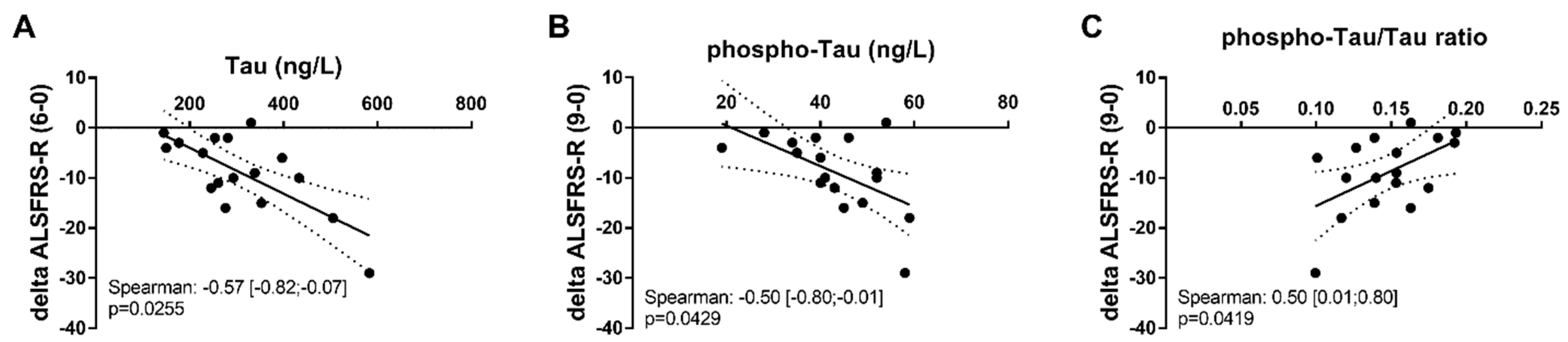
2.4. Biomarkers for ALS Prognosis
3. Discussion
3.1. Aβ1-42 Levels and Aβ1-42/Phospho-Tau Ratio as Diagnosis Biomarkers
3.2. Aβ1-42, Tau and Phospho-Tau Levels Were Related to Clinical Parameters at Baseline
3.3. Mild Prognosis Role of Phospho-Tau and Total Tau in ALS
4. Materials and Methods
4.1. Subjects
4.2. Measurements
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ALS | Amyotrophic lateral sclerosis |
| AD | Alzheimer’s disease |
| CSF | Cerebrospinal fluid |
| Aβ1-42 | Amyloidβ1-42 |
| IATI | Innotest Amyloid Tau Index |
References
- Hardiman, O.; Al-Chalabi, A.; Chio, A.; Corr, E.M.; Logroscino, G.; Robberecht, W.; Shaw, P.J.; Simmons, Z.; van den Berg, L.H. Amyotrophic lateral sclerosis. Nat. Rev. Dis. Primers 2017, 3, 17071. [Google Scholar] [CrossRef] [PubMed]
- Lanznaster, D.; de Assis, D.R.; Corcia, P.; Pradat, P.F.; Blasco, H. Metabolomics Biomarkers: A Strategy Toward Therapeutics Improvement in ALS. Front. Neurol. 2018, 9, 1126. [Google Scholar] [CrossRef] [PubMed]
- Poesen, K.; Van Damme, P. Diagnostic and Prognostic Performance of Neurofilaments in ALS. Front. Neurol. 2018, 9, 1167. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M. Tau protein and the neurofibrillary pathology of Alzheimer’s disease. Trends Neurosci. 1993, 16, 460–465. [Google Scholar] [CrossRef]
- Sunderland, T.; Linker, G.; Mirza, N.; Putnam, K.T.; Friedman, D.L.; Kimmel, L.H.; Bergeson, J.; Manetti, G.J.; Zimmermann, M.; Tang, B.; et al. Decreased beta-amyloid1-42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease. JAMA 2003, 289, 2094–2103. [Google Scholar] [CrossRef] [PubMed]
- Arendt, T.; Stieler, J.; Holzer, M. Brain hypometabolism triggers PHF-like phosphorylation of tau, a major hallmark of Alzheimer’s disease pathology. J. Neural. Transm. (Vienna) 2015, 122, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Bakota, L.; Brandt, R. Tau Biology and Tau-Directed Therapies for Alzheimer’s Disease. Drugs 2016, 76, 301–313. [Google Scholar] [CrossRef]
- Jimenez-Jimenez, F.J.; Hernanz, A.; Medina-Acebron, S.; de Bustos, F.; Zurdo, J.M.; Alonso, H.; Puertas, I.; Barcenilla, B.; Sayed, Y.; Cabrera-Valdivia, F. Tau protein concentrations in cerebrospinal fluid of patients with amyotrophic lateral sclerosis. Acta Neurol. Scand. 2005, 111, 114–117. [Google Scholar] [CrossRef]
- Brettschneider, J.; Petzold, A.; Sussmuth, S.D.; Ludolph, A.C.; Tumani, H. Axonal damage markers in cerebrospinal fluid are increased in ALS. Neurology 2006, 66, 852–856. [Google Scholar] [CrossRef]
- Paladino, P.; Valentino, F.; Piccoli, T.; Piccoli, F.; La Bella, V. Cerebrospinal fluid tau protein is not a biological marker in amyotrophic lateral sclerosis. Eur. J. Neurol. 2009, 16, 257–261. [Google Scholar] [CrossRef]
- Grossman, M.; Elman, L.; McCluskey, L.; McMillan, C.T.; Boller, A.; Powers, J.; Rascovsky, K.; Hu, W.; Shaw, L.; Irwin, D.J.; et al. Phosphorylated tau as a candidate biomarker for amyotrophic lateral sclerosis. JAMA Neurol. 2014, 71, 442–448. [Google Scholar] [CrossRef]
- Wilke, C.; Deuschle, C.; Rattay, T.W.; Maetzler, W.; Synofzik, M. Total tau is increased, but phosphorylated tau not decreased, in cerebrospinal fluid in amyotrophic lateral sclerosis. Neurobiol. Aging 2015, 36, 1072–1074. [Google Scholar] [CrossRef] [PubMed]
- Sussmuth, S.D.; Tumani, H.; Ecker, D.; Ludolph, A.C. Amyotrophic lateral sclerosis: Disease stage related changes of tau protein and S100 beta in cerebrospinal fluid and creatine kinase in serum. Neurosci. Lett. 2003, 353, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Levine, T.D.; Bowser, R.; Hank, N.; Saperstein, D. A pilot trial of memantine and riluzole in ALS: Correlation to CSF biomarkers. Amyotroph. Lateral Scler. Off. Publ. World Fed. Neurol. Res. Group Motor Neuron Dis. 2010, 11, 514–519. [Google Scholar] [CrossRef]
- Scarafino, A.; D’Errico, E.; Introna, A.; Fraddosio, A.; Distaso, E.; Tempesta, I.; Morea, A.; Mastronardi, A.; Leante, R.; Ruggieri, M.; et al. Diagnostic and prognostic power of CSF Tau in amyotrophic lateral sclerosis. J. Neurol. 2018, 265, 2353–2362. [Google Scholar] [CrossRef] [PubMed]
- Calingasan, N.Y.; Chen, J.; Kiaei, M.; Beal, M.F. Beta-amyloid 42 accumulation in the lumbar spinal cord motor neurons of amyotrophic lateral sclerosis patients. Neurobiol. Dis. 2005, 19, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Steinacker, P.; Hendrich, C.; Sperfeld, A.D.; Jesse, S.; Lehnert, S.; Pabst, A.; von Arnim, C.A.; Mottaghy, F.M.; Uttner, I.; Tumani, H.; et al. Concentrations of beta-amyloid precursor protein processing products in cerebrospinal fluid of patients with amyotrophic lateral sclerosis and frontotemporal lobar degeneration. J. Neural Transm. (Vienna) 2009, 116, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Kamalainen, A.; Herukka, S.K.; Hartikainen, P.; Helisalmi, S.; Moilanen, V.; Knuuttila, A.; Jansson, L.; Tienari, P.J.; Remes, A.M. Cerebrospinal fluid biomarkers for Alzheimer’s disease in patients with frontotemporal lobar degeneration and amyotrophic lateral sclerosis with the C9ORF72 repeat expansion. Dement. Geriatr. Cogn. Disord. 2015, 39, 287–293. [Google Scholar] [CrossRef]
- Burkle, A.; Moreno-Villanueva, M.; Bernhard, J.; Blasco, M.; Zondag, G.; Hoeijmakers, J.H.; Toussaint, O.; Grubeck-Loebenstein, B.; Mocchegiani, E.; Collino, S.; et al. MARK-AGE biomarkers of ageing. Mech. Ageing Dev. 2015, 151, 2–12. [Google Scholar] [CrossRef]
- Abu-Rumeileh, S.; Mometto, N.; Bartoletti-Stella, A.; Polischi, B.; Oppi, F.; Poda, R.; Stanzani-Maserati, M.; Cortelli, P.; Liguori, R.; Capellari, S.; et al. Cerebrospinal Fluid Biomarkers in Patients with Frontotemporal Dementia Spectrum: A Single-Center Study. J. Alzheimers Dis. 2018, 66, 551–563. [Google Scholar] [CrossRef]
- Paterson, R.W.; Slattery, C.F.; Poole, T.; Nicholas, J.M.; Magdalinou, N.K.; Toombs, J.; Chapman, M.D.; Lunn, M.P.; Heslegrave, A.J.; Foiani, M.S.; et al. Cerebrospinal fluid in the differential diagnosis of Alzheimer’s disease: Clinical utility of an extended panel of biomarkers in a specialist cognitive clinic. Alzheimers Res. Ther. 2018, 10, 32. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.Q.; Li, X.Y.; Zhang, Y.B.; Cheng, H.R.; Ma, Y.; Chen, D.F.; Tao, Q.Q.; Li, H.L.; Wu, Z.Y. The discriminative capacity of CSF beta-amyloid 42 and Tau in neurodegenerative diseases in the Chinese population. J. Neurol. Sci. 2020, 412, 116756. [Google Scholar] [CrossRef] [PubMed]
- Sjogren, M.; Davidsson, P.; Wallin, A.; Granerus, A.K.; Grundstrom, E.; Askmark, H.; Vanmechelen, E.; Blennow, K. Decreased CSF-beta-amyloid 42 in Alzheimer’s disease and amyotrophic lateral sclerosis may reflect mismetabolism of beta-amyloid induced by disparate mechanisms. Dement. Geriatr. Cogn. Disord. 2002, 13, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Stanga, S.; Brambilla, L.; Tasiaux, B.; Dang, A.H.; Ivanoiu, A.; Octave, J.N.; Rossi, D.; van Pesch, V.; Kienlen-Campard, P. A Role for GDNF and Soluble APP as Biomarkers of Amyotrophic Lateral Sclerosis Pathophysiology. Front. Neurol. 2018, 9, 384. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.M.; Khandelwal, P.J.; Stanczyk, B.B.; Rebeck, G.W.; Moussa, C.E. beta-amyloid triggers ALS-associated TDP-43 pathology in AD models. Brain Res. 2011, 1386, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Rusina, R.; Ridzon, P.; Kulist’ak, P.; Keller, O.; Bartos, A.; Buncova, M.; Fialova, L.; Koukolik, F.; Matej, R. Relationship between ALS and the degree of cognitive impairment, markers of neurodegeneration and predictors for poor outcome. A prospective study. Eur. J. Neurol. 2010, 17, 23–30. [Google Scholar] [CrossRef]
- Pijnenburg, Y.A.; Verwey, N.A.; van der Flier, W.M.; Scheltens, P.; Teunissen, C.E. Discriminative and prognostic potential of cerebrospinal fluid phosphoTau/tau ratio and neurofilaments for frontotemporal dementia subtypes. Alzheimers Dement. (Amst.) 2015, 1, 505–512. [Google Scholar] [CrossRef]
- Serio, A.; Bilican, B.; Barmada, S.J.; Ando, D.M.; Zhao, C.; Siller, R.; Burr, K.; Haghi, G.; Story, D.; Nishimura, A.L.; et al. Astrocyte pathology and the absence of non-cell autonomy in an induced pluripotent stem cell model of TDP-43 proteinopathy. Proc. Natl. Acad. Sci. USA 2013, 110, 4697–4702. [Google Scholar] [CrossRef]
- Hulstaert, F.; Blennow, K.; Ivanoiu, A.; Schoonderwaldt, H.C.; Riemenschneider, M.; De Deyn, P.P.; Bancher, C.; Cras, P.; Wiltfang, J.; Mehta, P.D.; et al. Improved discrimination of AD patients using beta-amyloid(1–42) and tau levels in CSF. Neurology 1999, 52, 1555–1562. [Google Scholar] [CrossRef]
- De Riva, V.; Galloni, E.; Marcon, M.; Di Dionisio, L.; Deluca, C.; Meligrana, L.; Bolner, A.; Perini, F. Analysis of combined CSF biomarkers in AD diagnosis. Clin. Lab. 2014, 60, 629–634. [Google Scholar] [CrossRef]
- Bourbouli, M.; Rentzos, M.; Bougea, A.; Zouvelou, V.; Constantinides, V.C.; Zaganas, I.; Evdokimidis, I.; Kapaki, E.; Paraskevas, G.P. Cerebrospinal Fluid TAR DNA-Binding Protein 43 Combined with Tau Proteins as a Candidate Biomarker for Amyotrophic Lateral Sclerosis and Frontotemporal Dementia Spectrum Disorders. Dement. Geriatr. Cogn. Disord. 2017, 44, 144–152. [Google Scholar] [CrossRef] [PubMed]
| ALS | Control | p-Value | |
|---|---|---|---|
| Number | 123 | 90 | |
| Gender (male) | 63.41% | 56.7% | 0.33 |
| Age (years) | 66.06 | 67.45 | 0.35 |
| Site of onset [19] | 71% | ||
| Disease duration (from onset; years) | 3.29 ± 2.03 | ||
| Weigth at baseline (kg) | 74.1 ± 1.4 | ||
| ALSFRS-R | 39.77 ± 0.43 |
| Parameters | Controls | ALS | p |
|---|---|---|---|
| Aβ1-42 (ng/L) | 992.9 ± 358.3 | 12,77.0 ± 296.6 | <0.0001 |
| Total Tau (ng/L) | 485.3 ± 519.5 | 291.6 ± 140.6 | 0.18 |
| Phospho-Tau (ng/L) | 49.8 ± 27.9 | 44.5 ± 15.5 | 0.78 |
| IATI | 1.69 ± 0.62 | 2.32 ± 0.66 | <0.0001 |
| Ratio Aβ1-42/phospho-Tau | 24.3 ± 11.6 | 31.2 ± 9.8 | <0.0001 |
| Ratio phospho-Tau/total Tau | 0.15 ± 0.06 | 0.16 ± 0.38 | 0.48 |
| Biomarker/Ratios | Basal ALSFRS-R | Basal Weight |
|---|---|---|
| Aβ1-42 | 0.30 [0.12; 0.46] p = 0.001 | −0.47 [−0.23; 0.14] p = 0.61 |
| Phospho-tau | 0.05 [−0.14; 0.23] p = 0.59 | −0.21 [−0.38; −0.03] p = 0.02 |
| Total tau | −0.15 [−0.33; 0.03] p = 0.09 | −0.34 [−0.49; −0.17] p = 0.0001 |
| IATI | 0.32 [0.14; 0.47] p = 0.0004 | 0.24 [0.05; 0.40] p = 0.009 |
| Ratio Aβ1-42/P-tau | 0.09 [−0.09; 0.28] p = 0.29 | 0.14 [−0.04; 0.32] p = 0.12 |
| Ratio p-Tau/total Tau | 0.26 [0.08; 0.43] p = 0.004 | 0.36 [0.19; 0.51] p < 0.0001 |
| Parameter | Month 6 (n = 18) | Month 9 (n = 17) | Month 12 (n = 17) |
|---|---|---|---|
| Aβ1-42 | −0.19 [−0.61; 0.32] p = 0.44 | 0.30 [−0.22; 0.69] p = 0.23 | −0.36 [−0.72; 0.16] p = 0.16 |
| Phospho-Tau | −0.46 [−0.77; 0.03] p = 0.06 | −0.50 [−0.80; 0.01] p = 0.04 | −0.17 [−0.61; 0.35] p = 0.50 |
| Total Tau | −0.54 [−0.82; −0.07] p = 0.02 | −0.24 [−0.65; 0.29] p = 0.36 | −0.23 [−0.61; 0.23] p = 0.31 |
| IATI | 0.20 [−0.31; 0.62] p = 0.43 | 0.37 [−0.15; 0.73] p = 0.14 | 0.01 [−0.48; 0.50] p = 0.96 |
| Ratio Aβ1-42/p-Tau | 0.28 [−0.23; 0.67] p = 0.27 | 0.43 [−0.07; 0.76] p = 0.08 | −0.05 [−0.45; 0.53] p = 0.85 |
| p-Tau/total Tau | 0.44 [−0.05; 0.76] p = 0.07 | 0.50 [0.01; 0.80] p = 0.04 | 0.22 [−0.31; 0.64] p = 0.40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanznaster, D.; Hergesheimer, R.C.; Bakkouche, S.E.; Beltran, S.; Vourc’h, P.; Andres, C.R.; Dufour-Rainfray, D.; Corcia, P.; Blasco, H. Aβ1-42 and Tau as Potential Biomarkers for Diagnosis and Prognosis of Amyotrophic Lateral Sclerosis. Int. J. Mol. Sci. 2020, 21, 2911. https://doi.org/10.3390/ijms21082911
Lanznaster D, Hergesheimer RC, Bakkouche SE, Beltran S, Vourc’h P, Andres CR, Dufour-Rainfray D, Corcia P, Blasco H. Aβ1-42 and Tau as Potential Biomarkers for Diagnosis and Prognosis of Amyotrophic Lateral Sclerosis. International Journal of Molecular Sciences. 2020; 21(8):2911. https://doi.org/10.3390/ijms21082911
Chicago/Turabian StyleLanznaster, Débora, Rudolf C. Hergesheimer, Salah Eddine Bakkouche, Stephane Beltran, Patrick Vourc’h, Christian R. Andres, Diane Dufour-Rainfray, Philippe Corcia, and Hélène Blasco. 2020. "Aβ1-42 and Tau as Potential Biomarkers for Diagnosis and Prognosis of Amyotrophic Lateral Sclerosis" International Journal of Molecular Sciences 21, no. 8: 2911. https://doi.org/10.3390/ijms21082911
APA StyleLanznaster, D., Hergesheimer, R. C., Bakkouche, S. E., Beltran, S., Vourc’h, P., Andres, C. R., Dufour-Rainfray, D., Corcia, P., & Blasco, H. (2020). Aβ1-42 and Tau as Potential Biomarkers for Diagnosis and Prognosis of Amyotrophic Lateral Sclerosis. International Journal of Molecular Sciences, 21(8), 2911. https://doi.org/10.3390/ijms21082911








