Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy
Abstract
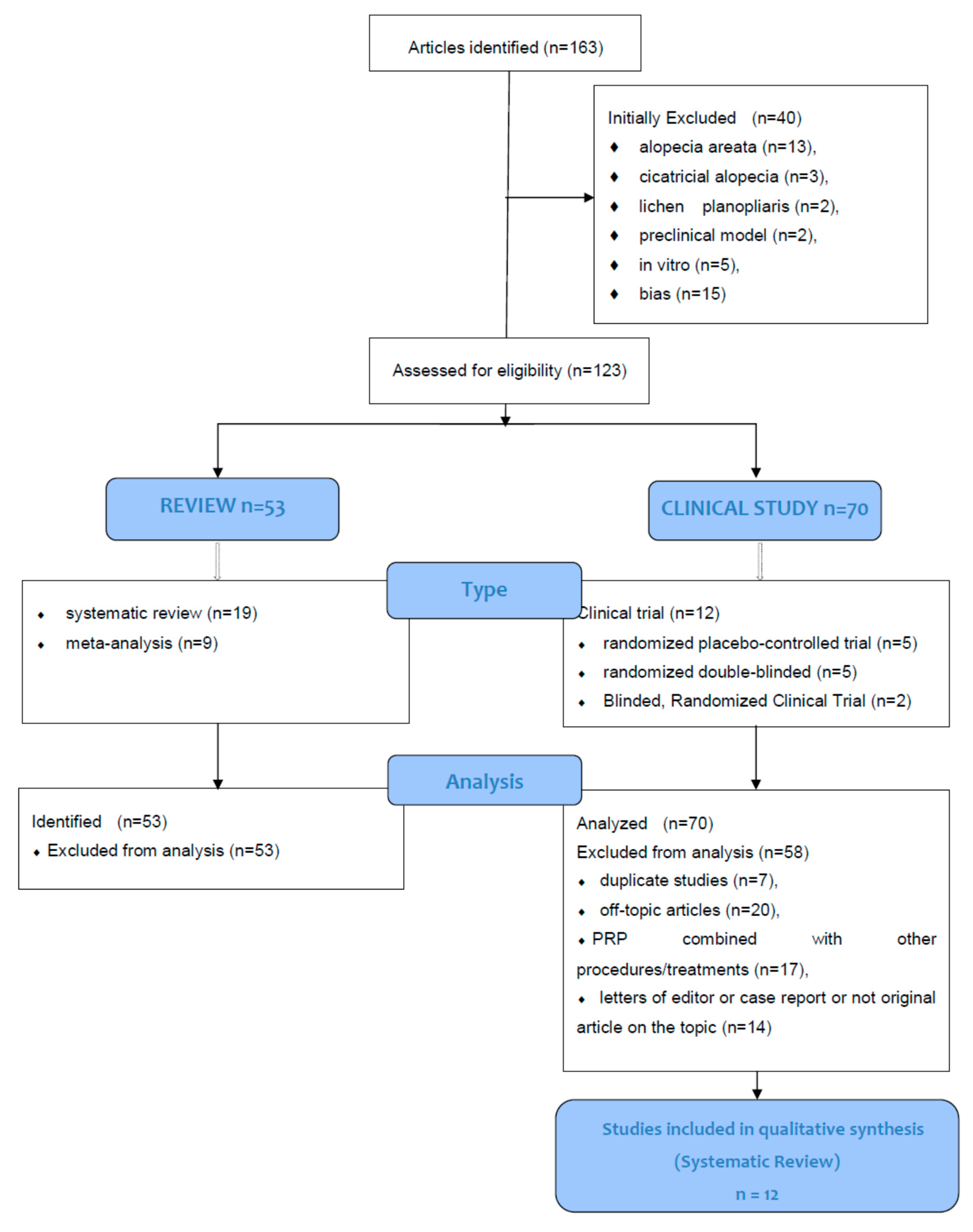
1. Introduction
2. Methods
2.1. Institutional Guidelines
- -
- Quantity of platelets to be obtained (1 × 106 µL ± 20%);
- -
- Exclusion criteria (platelets disorders, thrombocytopenia, anti-aggregating therapy, bone marrow aplasia, uncompensated diabetes, sepsis, and cancer);
- -
- Fields of application of the PRP only on the basis of available scientific evidence and guidelines of the national blood center;
- -
- Methods of preparation of the PRP (kits and procedure);
- -
- How to use the PRP (only topical or infiltrative);
- -
- Quality and sterility checks on the sample obtained;
- -
- Blood volume to withdrew (within 55 cc for each patient);
- -
- The volume of A-PRP and AA-PRP to be obtained (depending on the extension of the AGA area);
- -
- Labeling of each sample of PRP;
- -
- Informed consent;
- -
- Adverse reaction form; and
- -
- Data processing module.
2.2. Search Strategy
2.2.1. Study Assessment
2.2.2. Study Selection
2.2.3. Data Extraction
2.2.4. Outcome Measures
2.3. Brief History Analysis of PRP Use in Androgenetic Alopecia
2.3.1. A-PRP and AA-PRP Devices for Hair Regrowth
2.3.2. PRP and Growth Factors Assessment
- -
- Leukocyte-poor PRP (LP-PRP) or pure platelet-rich plasma (P-PRP). PRP without leukocytes and with a low-density fibrin network after activation;
- -
- Leukocyte-PRP (L-PRP). PRP with leukocytes and a low-density fibrin network after activation (most frequent);
- -
- Leukocyte-poor platelet-rich fibrin (LP-PRF) or pure platelet-rich fibrin (P-PRF). PRF without leukocytes and a high-density fibrin network.
- -
- Leukocytes platelet-rich fibrin (L-PRF). PRF with leukocytes and a high-density fibrin network.
2.3.3. Protocol: Manual Versus Mechanical and Controlled Hair Injection of A-PRP and AA-PRP
3. Results
3.1. Results Performing Literature Scans: PRP Studies with Hair Density and Hair Count Improvement
3.2. Results Performing Literature Scans: PRP Studies without Hair Density and Hair Count Improvement
3.3. Critical Assessment of Study Design
3.4. Side Effects
3.5. Considerations
4. Discussion
4.1. PRP Compared with Minoxidil® and Finasteride®
4.2. PRP Comparison with Autologous Adult Stem Cell-Based Therapy (ASCs-BT)
4.3. Evidence-Based Medicine’s Impact of PRP in AGA Treatment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AGA | Androgenic Alopecia |
| PRP | Platelet-Rich Plasma |
| PPP | Platelet-poor plasma |
| A-PRP | Autologous-Non-activated Platelet-Rich Plasma |
| AA-PRP | Autologous-Activated Platelet-Rich-Plasma |
| HG | Hair Growth |
| HRG | Hair re-growth |
| GFs | Growth Factors |
| VEGF | Vascular endothelial Growth factors |
| PDGF | Platelet Derived Growth factors |
| IGF-1 | Insulin like Growth factor-1 |
| TGF-ß | TGF-ß: transforming growth factor-beta |
| EGF | Epidermal growth factor |
| PRGF | Plasma rich in growth factor |
| DPCs | Dermal papilla cells |
| DP | Dermal papilla |
| HD | Hair density |
| HC | Hair count |
| HT | Hair thickness |
| H-C | Hair cycle |
| HCS | Hair cross-size |
| EC | European Committee |
| CAT | Committee for Advanced treatments; |
| GMP | Good Manufacturing Practices |
| GCP | Good Clinical Practices |
| GFs | Growth factors |
| HF-MSCs | Human follicle mesenchymal stem cells |
| HFSC | Human follicle stem cells |
| HF-ESCs | Hair Follicle Epithelial Stem Cells |
| KCs | Skin epidermal keratinocytes |
| ECM | Extracellular matrix |
| HF | Hair Follicle |
| HFs | Hair Follicles |
| SVFs | Stromal vascular Fraction Cells |
| AD-MSCs | Adipose-derived Mesenchymal Stem Cells |
| SCs | Stem Cells |
| HLs | Hair Loss |
| HD-AFSCs | Human Intra and Extra Dermal Adipose Tissue-Derived Hair Follicle Stem Cells |
| RPM | Right per minute |
| MPHL | Male pattern hair loss |
| FPHL | Female pattern hair loss |
| ASCs-BT | Adult stem cells based therapy |
| AT | Adipose tissue |
| BM | Bone marrow |
| HA | Hyaluronic acid |
| DHT | Dihydrotestosterone |
| D/P | Dalteparin |
| D/P MPs | Dalteparin and protamine micro-particles |
| AA | Alopecia Areata |
| CaCl-Ca2+ | Calcium chloride |
| PGE2 | Prostaglandin E2 |
| LIF | Leukemia-inhibiting factor |
| EBM | Evidence-based medicine |
| WKS | Weeks |
| MOS | Months. |
| b-FGF | Basic-Fibroblast Growth Factor |
| FGF-7 | Fibroblast Growth Factor-7. |
References
- Gentile, P.; Garcovich, S. Advances in Regenerative Stem Cell Therapy in Androgenic Alopecia and Hair Loss: Wnt pathway, Growth-Factor, and Mesenchymal Stem Cell Signaling Impact Analysis on Cell Growth and Hair Follicle Development. Cells 2019, 8, 466. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Scioli, M.G.; Bielli, A.; De Angelis, B.; De Sio, C.; De Fazio, D.; Ceccarelli, G.; Trivisonno, A.; Orlandi, A.; Cervelli, V.; et al. Platelet-Rich Plasma and Micrografts Enriched with Autologous Human Follicle Mesenchymal Stem Cells Improve Hair Re-Growth in Androgenetic Alopecia. Biomolecular Pathway Analysis and Clinical Evaluation. Biomedicines 2019, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S.; Bielli, A.; Scioli, M.G.; Orlandi, A.; Cervelli, V. The Effect of Platelet-Rich Plasma in Hair Regrowth: A Randomized Placebo-Controlled Trial. STEM CELLS Transl. Med. 2015, 4, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Cole, J.P.; Cole, M.A.; Garcovich, S.; Bielli, A.; Scioli, M.G.; Orlandi, A.; Insalaco, C.; Cervelli, V. Evaluation of Not-Activated and Activated PRP in Hair Loss Treatment: Role of Growth Factor and Cytokine Concentrations Obtained by Different Collection Systems. Int. J. Mol. Sci. 2017, 18, 408. [Google Scholar] [CrossRef]
- Metcalf, K.B.; Mandelbaum, B.R.; McIlwraith, C.W. Application of Platelet-Rich Plasma to Disorders of the Knee Joint. Cartil. 2013, 4, 295–312. [Google Scholar] [CrossRef]
- Dhurat, R.; Sukesh, M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. J. Cutan. Aesthetic Surg. 2014, 7, 189–197. [Google Scholar] [CrossRef]
- Cole, J.P.; Cole, M.A.; Insalaco, C.; Cervelli, V.; Gentile, P. Alopecia and platelet-derived therapies. Stem Cell Investig. 2017, 4, 88. [Google Scholar] [CrossRef]
- Cervelli, V.; Garcovich, S.; Bielli, A.; Cervelli, G.; Curcio, B.C.; Scioli, M.G.; Orlandi, A.; Gentile, P. The Effect of Autologous Activated Platelet Rich Plasma (AA-PRP) Injection on Pattern Hair Loss: Clinical and Histomorphometric Evaluation. BioMed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Mechanical and Controlled PRP Injections in Patients Affected by Androgenetic Alopecia. J. Vis. Exp. 2018, 27, 131. [Google Scholar] [CrossRef]
- Gentile, P.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Stem cells from human hair follicles: First mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017, 4, 58. [Google Scholar] [CrossRef]
- Gentile, P. Autologous Cellular Method Using Micrografts of Human Adipose Tissue Derived Follicle Stem Cells in Androgenic Alopecia. Int. J. Mol. Sci. 2019, 20, 3446. [Google Scholar] [CrossRef] [PubMed]
- Schücklenk, U.; Ashcroft, R. International Research Ethics. Bioeth. 2000, 14, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Von Elm, E.; Altman, U.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiology 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J. The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Takikawa, M.; Nakamura, S.; Nakamura, S.; Ishirara, M.; Kishimoto, S.; Sasaki, K.; Yanagibayashi, S.; Azuma, R.; Yamamoto, N.; Kiyosawa, T. Enhanced Effect of Platelet-Rich Plasma Containing a New Carrier on Hair Growth. Dermatol. Surg. 2011, 37, 1721–1729. [Google Scholar] [CrossRef]
- Schiavone, G.; Raskovic, D.; Greco, J.; Abeni, D. Platelet-Rich Plasma for Androgenetic Alopecia. Dermatol. Surg. 2014, 40, 1010–1019. [Google Scholar] [CrossRef]
- Gkini, M.-A.; Kouskoukis, A.-E.; Tripsianis, G.; Rigopoulos, D.; Kouskoukis, K. Study of Platelet-Rich Plasma Injections in the Treatment of Androgenetic Alopecia Through an One-Year Period. J. Cutan. Aesthetic Surg. 2014, 7, 213–219. [Google Scholar] [CrossRef]
- Khatu, S.S.; E More, Y.; Gokhale, N.R.; Chavhan, D.C.; Bendsure, N. Platelet-Rich Plasma in Androgenic Alopecia: Myth or an Effective Tool. J. Cutan. Aesthetic Surg. 2014, 7, 107–110. [Google Scholar] [CrossRef]
- Singhal, P.; Agarwal, S.; Dhot, P.S.; Sayal, S.K. Efficacy of platelet-rich plasma in treatment of androgenic alopecia. Asian J. Transfus. Sci. 2015, 9, 159–162. [Google Scholar] [CrossRef]
- Alves, R.; Grimalt, R. Randomized Placebo-Controlled, Double-Blind, Half-Head Study to Assess the Efficacy of Platelet-Rich Plasma on the Treatment of Androgenetic Alopecia. Dermatol. Surg. 2016, 42, 491–497. [Google Scholar] [CrossRef]
- Puig, C.J.; Reese, R.; Peters, M. Double-Blind, Placebo-Controlled Pilot Study on the Use of Platelet-Rich Plasma in Women With Female Androgenetic Alopecia. Dermatol. Surg. 2016, 42, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Mapar, M.A.; Shahriari, S.; Haghighizadeh, M.H. Efficacy of platelet-rich plasma in the treatment of androgenetic (male-patterned) alopecia: A pilot randomized controlled trial. J. Cosmet. Laser Ther. 2016, 18, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Revathi, T.; Sacchidanand, S.; Nataraj, H. A study of the efficacy of platelet-rich plasma in the treatment of androgenetic alopecia in males. Indian J. Dermatol. Venereol. Leprol. 2017, 83, 412. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Pino, A.; Martinez, N.; Orive, G.; Berridi, D. The Effect of Plasma Rich in Growth Factors on Pattern Hair Loss. Dermatol. Surg. 2017, 43, 658–670. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D. The Newcastle-Ottawa Scale for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2014. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 31 January 2014).
- A Furukawa, T.; Barbui, C.; Cipriani, A.; Brambilla, P.; Watanabe, N. Imputing missing standard deviations in meta-analyses can provide accurate results. J. Clin. Epidemiology 2006, 59, 7–10. [Google Scholar] [CrossRef]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef]
- Kang, J.; Zheng, Z.; Choi, M.; Lee, S.-H.; Kim, D.Y.; Bin Cho, S. The effect of CD34+ cell-containing autologous platelet-rich plasma injection on pattern hair loss: A preliminary study. J. Eur. Acad. Dermatol. Venereol. 2012, 28, 72–79. [Google Scholar] [CrossRef]
- Okita, A.L.; Steiner, D.; Ferreira, S.B.; Ramos, P.M.; Ferreira, W.; Silveira, R.; Mussi, M.L.; Vaidergorn, R.E.; Ferreira, R.B. Treatment of Male-Pattern Alopecia with Platelet-Rich Plasma. Ski. Appendage Disord. 2019, 6, 97–101. [Google Scholar] [CrossRef]
- Hausauer, A.K.; Jones, D.H. Evaluating the Efficacy of Different Platelet-Rich Plasma Regimens for Management of Androgenetic Alopecia. Dermatol. Surg. 2018, 44, 1191–1200. [Google Scholar] [CrossRef]
- Schiavone, G.; Paradisi, A.; Ricci, F.; Abeni, D. Injectable Platelet-, Leukocyte-, and Fibrin-Rich Plasma (iL-PRF) in the Management of Androgenetic Alopecia. Dermatol. Surg. 2018, 44, 1183–1190. [Google Scholar] [CrossRef]
- Jha, A.K.; Udayan, U.K.; Roy, P.K.; Amar, A.K.J.; Chaudhary, R.K.P. Original article: Platelet-rich plasma with microneedling in androgenetic alopecia along with dermoscopic pre- and post-treatment evaluation. J. Cosmet. Dermatol. 2017, 17, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Tawfik, A.A.; Osman, M.A. The effect of autologous activated platelet-rich plasma injection on female pattern hair loss: A randomized placebo-controlled study. J. Cosmet. Dermatol. 2017, 17, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.J.; Lawrence, C.; Cserhalmi-Friedman, P.B.; Christiano, A.M.; Jahoda, C.A. Trans-gender induction of hair follicles. Nat. 1999, 402, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Talavera-Adame, D.; Newman, D.; Newman, N. Conventional and novel stem cell based therapies for androgenic alopecia. Stem Cells Cloning: Adv. Appl. 2017, 10, 11–19. [Google Scholar] [CrossRef]
- Semalty, M.; Semalty, A.; Joshi, G.P.; Rawat, M.S.M. Hair growth and rejuvenation: An overview. J. Dermatol. Treat. 2010, 22, 123–132. [Google Scholar] [CrossRef]
- Han, J.H.; Kwon, O.S.; Chung, J.-H.; Cho, K.H.; Eun, H.C.; Kim, K.H. Effect of minoxidil on proliferation and apoptosis in dermal papilla cells of human hair follicle. J. Dermatol. Sci. 2004, 34, 91–98. [Google Scholar] [CrossRef]
- Tosti, A.; Piraccini, B.M. Finasteride and the hair cycle. J. Am. Acad. Dermatol. 2000, 42, 848–849. [Google Scholar] [CrossRef]
- Sawaya, M.E.; Blume-Peytavi, U.; Mullins, D.L. Effects of finasteride on apoptosis and regulation of the human hair cycle. J. Cutan. Med. Surg. 2002, 6, 1–9. [Google Scholar] [CrossRef]
- Vaccari, J.P.D.R.; Sawaya, M.E.; Brand, F.; Nusbaum, B.P.; Bauman, A.J.; Bramlett, H.M.; Dietrich, D.W.; Keane, R.W. Caspase-1 Level Is Higher in the Scalp in Androgenetic Alopecia. Dermatol. Surg. 2012, 38, 1033–1039. [Google Scholar] [CrossRef]
- Adil, A.; Godwin, M. The effectiveness of treatments for androgenetic alopecia: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2017, 77, 136–141. [Google Scholar] [CrossRef]
- Van Neste, D.; Fuh, V.; Sanchez-Pedreno, P.; Lopez-Bran, E.; Wolff, H.; Whiting, D.; Roberts, J.; Kopera, D.; Stene, J.-J.; Calvieri, S.; et al. Finasteride increases anagen hair in men with androgenetic alopecia. Br. J. Dermatol. 2000, 143, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Gong, L.; Guo, M.; Liu, T.; Shi, A.; Zong, H.; Xu, X.; Chen, H.; Gao, X.; Li, Y.-H. Randomized trial of electrodynamic microneedle combined with 5% minoxidil topical solution for the treatment of Chinese male Androgenetic alopecia. J. Cosmet. Laser Ther. 2020, 22, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Trink, A.; Sorbellini, E.; Bezzola, P.; Rodella, L.F.; Ramot, Y.; Rezzani, R.; Rinaldi, F. A randomized, double-blind, placebo- and active-controlled, half-head study to evaluate the effects of platelet-rich plasma on alopecia areata. Br. J. Dermatol. 2013, 169, 690–694. [Google Scholar] [CrossRef] [PubMed]
- Saczonek, A.O.; Krajewska-Włodarczyk, M.; Kruszewska, A.; Banasiak, Ł.; Placek, W.; Maksymowicz, W.; Wojtkiewicz, J. Therapeutic Potential of Stem Cells in Follicle Regeneration. Stem Cells Int. 2018, 2018, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Jalili, R.B.; Kilani, R.T.; Li, Y.; Khosravi-Maharlooie, M.; Nabai, L.; Wang, E.H.C.; McElwee, K.J.; Ghahary, A. Fibroblast cell-based therapy prevents induction of alopecia areata in an experimental model. Cell Transplant. 2018, 27, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Stoll, S.; Dietlin, C.; Nett-Mettler, C.S. Microneedling as a successful treatment for alopecia X in two Pomeranian siblings. Veter- Dermatol. 2015, 26, 387. [Google Scholar] [CrossRef]
- Fertig, R.M.; Gamret, A.C.; Cervantes, J.; Tosti, A. Microneedling for the treatment of hair loss? J. Eur. Acad. Dermatol. Venereol. 2017, 32, 564–569. [Google Scholar] [CrossRef]
- Garza, L.A.; Yang, C.; Zhao, T.; Blatt, H.B.; Lee, M.; He, H.; Stanton, D.C.; Carrasco, L.; Spiegel, J.H.; Tobias, J.W.; et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Investig. 2011, 121, 613–622. [Google Scholar] [CrossRef]
- Balañá, M.E.; Charreau, H.E.; Leirós, G.J. Epidermal stem cells and skin tissue engineering in hair follicle regeneration. World J. Stem Cells 2015, 7, 711–727. [Google Scholar] [CrossRef]
- Yu, H.; Fang, N.; Kumar, S.M.; Li, L.; Nguyen, T.K.; Acs, G.; Herlyn, M.; Xu, X. Isolation of a Novel Population of Multipotent Adult Stem Cells from Human Hair Follicles. Am. J. Pathol. 2006, 168, 1879–1888. [Google Scholar] [CrossRef]
- Cotsarelis, G.; Sun, T.-T.; Lavker, R.M. Label-retaining cells reside in the bulge area of pilosebaceous unit: Implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 1990, 61, 1329–1337. [Google Scholar] [CrossRef]
- Tumbar, T.; Guasch, G.; Greco, V.; Blanpain, C.; Lowry, W.E.; Rendl, M.; Fuchs, E. Defining the Epithelial Stem Cell Niche in Skin. Sci. 2004, 303, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.J.; Liu, Y.; Marles, L.; Yang, Z.; Trempus, C.; Li, S.; Lin, J.; A Sawicki, J.; Cotsarelis, G. Capturing and profiling adult hair follicle stem cells. Nat. Biotechnol. 2004, 22, 411–417. [Google Scholar] [CrossRef]
- Taylor, G.; Lehrer, M.S.; Jensen, P.J.; Sun, T.-T.; Lavker, R.M. Involvement of Follicular Stem Cells in Forming Not Only the Follicle but Also the Epidermis. Cell 2000, 102, 451–461. [Google Scholar] [CrossRef]
- Pesce, M.; Schöler, H.R. Oct-4: Gatekeeper in the Beginnings of Mammalian Development. STEM CELLS 2001, 19, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Alonso, L. The hair cycle. J. Cell Sci. 2006, 119, 391–393. [Google Scholar] [CrossRef]
- Greco, V.; Chen, T.; Rendl, M.; Schöber, M.; Pasolli, H.A.; Stokes, N.; Cruz-Racelis, J.D.; Fuchs, E. A Two-Step Mechanism for Stem Cell Activation during Hair Regeneration. Cell Stem Cell 2009, 4, 464. [Google Scholar] [CrossRef][Green Version]
- Blanpain, C.; Lowry, W.E.; Geoghegan, A.; Polak, L.; Fuchs, E. Self-Renewal, Multipotency, and the Existence of Two Cell Populations within an Epithelial Stem Cell Niche. Cell 2004, 118, 635–648. [Google Scholar] [CrossRef]
- Botchkarev, V.; Kishimoto, J. Molecular Control of Epithelial–Mesenchymal Interactions During Hair Follicle Cycling. J. Investig. Dermatol. Symp. Proc. 2003, 8, 46–55. [Google Scholar] [CrossRef]
- Roh, C.; Tao, Q.; Lyle, S. Dermal papilla-induced hair differentiation of adult epithelial stem cells from human skin. Physiol. Genom. 2004, 19, 207–217. [Google Scholar] [CrossRef]
- Hsu, Y.-C.; Pasolli, H.A.; Fuchs, E. Dynamics between Stem Cells, Niche, and Progeny in the Hair Follicle. Cell 2011, 144, 92–105. [Google Scholar] [CrossRef] [PubMed]
- A Jahoda, C. Induction of follicle formation and hair growth by vibrissa dermal papillae implanted into rat ear wounds: Vibrissa-type fibres are specified. Dev. 1992, 115, 1103–1109. [Google Scholar]
- Kalabusheva, E.; Terskikh, V.; Vorotelyak, E. Hair Germ Model In Vitro via Human Postnatal Keratinocyte-Dermal Papilla Interactions: Impact of Hyaluronic Acid. Stem Cells Int. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.-Y.; Sennett, R.; Rezza, A.; Clavel, C.; Grisanti, L.; Zemla, R.; Najam, S.; Rendl, M. Wnt/β-catenin signaling in dermal condensates is required for hair follicle formation. Dev. Boil. 2013, 385, 179–188. [Google Scholar] [CrossRef]
- Huelsken, J.; Vogel, R.; Erdmann, B.; Cotsarelis, G.; Birchmeier, W. beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell 2001, 105, 533–545. [Google Scholar] [CrossRef]
- Festa, E.; Fretz, J.; Berry, R.; Schmidt, B.; Rodeheffer, M.; Horowitz, M.; Horsley, V. Adipocyte Lineage Cells Contribute to the Skin Stem Cell Niche to Drive Hair Cycling. Cell 2011, 146, 761–771. [Google Scholar] [CrossRef]
- Perez-Meza, D.; Ziering, C.; Sforza, M.; Krishnan, G.; Ball, E.; Daniels, E. Hair follicle growth by stromal vascular fraction-enhanced adipose transplantation in baldness. Stem Cells Cloning: Adv. Appl. 2017, 10, 1–10. [Google Scholar] [CrossRef]
- Fukuoka, H.; Suga, H. Hair Regeneration Treatment Using Adipose-Derived Stem Cell Conditioned Medium: Follow-up With Trichograms. Eplasty 2015, 15, 10. [Google Scholar]
- Cervelli, V.; Lucarini, L.; Spallone, D.; Palla, L.; Colicchia, G.M.; Gentile, P.; De Angelis, B. Use of Platelet-Rich Plasma and Hyaluronic Acid in the Loss of Substance with Bone Exposure. Adv. Ski. Wound Care 2011, 24, 176–181. [Google Scholar] [CrossRef]
- Scioli, M.G.; Bielli, A.; Gentile, P.; Cervelli, V.; Orlandi, A. Combined treatment with platelet-rich plasma and insulin favours chondrogenic and osteogenic differentiation of human adipose-derived stem cells in three-dimensional collagen scaffolds. J. Tissue Eng. Regen. Med. 2016, 11, 2398–2410. [Google Scholar] [CrossRef]
- Nicoli, F.; Balzani, A.; Lazzeri, D.; Gentile, P.; Chilgar, R.M.; Di Pasquali, C.; Nicoli, F.; Bocchini, I.; Agovino, A.; Cervelli, V. Severe hidradenitis suppurativa treatment using platelet-rich plasma gel and Hyalomatrix. Int. Wound J. 2013, 12, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Bottini, D.J.; Spallone, D.; Curcio, B.C.; Cervelli, V. Application of Platelet-Rich Plasma in Maxillofacial Surgery. J. Craniofacial Surg. 2010, 21, 900–904. [Google Scholar] [CrossRef] [PubMed]
- Hausauer, A.K.; Humphrey, S. The Physician’s Guide to Platelet-Rich Plasma in Dermatologic Surgery Part I: Definitions, Mechanisms of Action, and Technical Specifications. Dermatol Surg. 2020, 46, 348–357. [Google Scholar] [CrossRef] [PubMed]
| Authors | Study Type | Characteristics of Enrolled Subjects (Completed Study) | Objective Measures | Objective Assessment of Hair Growth | Subjective Assessment of Hair Growth | Year | Ref | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Randomized | Controlled | Blinded | Half-head | |||||||
| Takikawa et al. | - | Yes | No | No | 26 (26) 16 M, 10 F, aged 28–59, thin hair in the frontal or parietal areas | 1. Mean number of hairs (digital and dermoscopic imaging) 2. Mean HCS of hairs (digital and dermoscopic imaging) 3. Epidermal thickness, collagen and blood vessel density around hair follicles (4-mm punch biopsy) | 1. Yes* 2. Yes (p < 0.01) 3. Yes* | Patients reported less depilation when shampooing, greater bounce/resilience of hair, maintenance of healthy hairs | 2011 | [15] |
| Schiavone et al. | - | No | No | No | 64 (64) 42 M, mean age 28, stage II–V; 22 F, mean age 32, Stage I–II | 1. Hair count and hair thickness using Jaeschke 15-point scale rating of clinical change (macrophotographs examined by 2 independent evaluators) | 1. Yes (mean change in clinical rating of 3.2 and 3.9)* | N.a | 2014 | [16] |
| Gkini et al. | No | No | No | No | 22 (20) 18 M, aged 24–72, stage II-5a; 2 F, aged 58–72, Stage I | 1. Hair pull test 2. Hair density and quality (dermoscopic photomicrographs and macroscopic photographs) | 1. Yes* 2. Yes, p < 0.001; overall improvement in hair density and quality per photographs | Patient self-assessment questionnaire: mean result rating of 7.1 on a 1–10 scale; 85% reported improvement in hair quality and thickness; 65% reported increases in hair density | 2014 | [17] |
| Khatu et al. | No | No | No | No | 11 (11) 11 M, aged 20–40, stage II–IV | 1. Hair pull test 2. Hair count (Trichoscan) 3. Hair loss (clinical examination, macroscopic photos) | 1. Yes (81.81% achieved a negative pull test at 12 wks.) 2. Yes (average mean gain of 22.09 follicular units/cm2)a 3. Yes (moderate improvement in hair volume and coverage with reduction in hair loss) | Patient satisfaction questionnaire: mean overall satisfaction rating of 7 out of 10 | 2014 | [18] |
| Cervelli et al. | Yes | Yes | Yes | Yes | 10 (10) 10 M, aged 20–52 stage IIa–IV | #1–4: Computerized phototrichogram and global photography: 1. Hair count 2. Hair density 3. Terminal hair density 4. Epidermal thickness and hair follicle density (3-mm punch biopsy) 5. Percentage of Ki67+ keratinocytes & blood vessel density (immunohistochemistry) | 1. Yes (p < 0.0001) at 3 mos 2. Yes (p < 0.0001) at 3 mos 3. Yes (p = 0.0003) at 3 mos 4. Yes (p < 0.05) at 3 mos 5. Yes (p < 0.05) at 14 wks. | Physician and patient global assessment scale—results not reported | 2014 | [8] |
| Gentile et al. | Yes | Yes | Yes | Yes | 23 (20) 20 M, aged 19–63 stage IIa–IV | #1–3: Computerized phototrichogram and global photography: 1. Hair count and total hair density 2. Terminal hair density 3. Epidermal thickness and hair follicle density (3-mm punch biopsy) 4. Keratinocyte proliferation and small blood vessel proliferation around hair follicles (immunohistochemistry) 5. Relapse of AGA | 1. Yes (p < 0.0001) 2. Yes (p = 0.0003) 3. Yes (p < 0.05) 4. Yes (p < 0.05) 5. Four patients reported progressive hair loss at 12–16 mos | Physician and patient global assessment scale)—results not reported | 2015 | [3] |
| Singhal et al. | No | Yes | No | No |
20 (20) 16 M, aged 25–32 4 F aged 32–35 |
1. Hair count (hair pull test) 2. Hair growth, hair volume, hair quality, fullness (global photographs) | 1. Yes, pulled hair count was reduced by 65% (vs. 0% in controls)* 2. Yes, hair growth noted in 6 patients after 7 days but in 4 patients after 15 days; yet, all patients (10) had good hair growth after 3 mos* | N.a | 2015 | [19] |
| Alves and Grimalt | Yes | Yes | Yes | Yes | 25 (24) 11 M, aged 18–65, stage II–V; 11 F, aged 18–86, Stage I–II | #1–6: Phototrichogram and global photography 1. Anagen hair (%) 2. Telogen hair (%) 3. Anagen: telogen ratio 4. Hair density 5. Terminal hair density 6. Hair count | PRP vs. placebo: 1–3, 5, 6. No (p > 0.05) 4. Yes, at 3 and 6 mos (p < 0.05) PRP vs. baseline: 1–5. Yes (p < 0.05) 6. No (p > 0.05) | N.a | 2016 | [20] |
| Puig et al. | Yes | Yes | Yes | No | 26 (26) 26 F, stage II | 1. Hair count (photography) 2. Hair mass index (Cohen HairCheck® system | 1. No (p = 0.503) 2. No (p = 0.220) | 13.3% of treatment group vs. 0% of control group reported substantial improvement in hair loss, rate of hair loss, hair thickness, and ease of managing/styling hair; 26.7% of treatment group vs. 18.3% of control group reported feeling coarser/heavier hair | 2016 | [21] |
| Mapar et al. | Yes | Yes | Yes | Yes |
19 (17) 17 M, aged 24–45, stage IV–VI |
1. Terminal hair count (magnifying glass) 2. Vellus hair count (magnifying glass) | 1. No (p = 0.25 at 6 mos) 2. No (p = 0.23 at 6 mos) | n.a. | 2016 | [22] |
| Gupta et al. | - | No | No | No |
30 (30) 30 M, aged 25– 35 stage III–VII |
1. Hair density (CapilliCare trichoscan) 2. Hair diameter (CapilliCare trichoscan) 3. Independent observer clinical evaluation (global macrophotographs) | 1. Yes (39.7 ± 16.5% increase compared to baseline)* 2. Yes (39.8 ± 17.2% increase compared to baseline)* 3. Average improvement = 30.2 ± 12.2% | Patient self-assessment questionnaire: treatment group reported 30 ± 13.1% mean improvement (range 10–70%); 93.3% reported complete cessation of hair fall by 2 mos; 66.7% reported increase in hair growth; 36.7% reported improvement in hair texture | 2017 | [23] |
| Anitua et al. | No | No | Yes | No |
19 (19) 12 M, aged 27–60, stage III–VI; 2 F, aged 32–60, stage II-frontal |
#1–4 Computerized phototrichogram 1. Hair density 2. Hair diameter 3. Terminal/vellus-like hair ratio 4. Thin/regular/thick hair shafts among terminal follicles 5. Independent observer clinical evaluation (mean improvement score using global macro-photographs) 6. Epidermal thickness perivascular inflammatory infiltrate, rete ride number, terminal/miniaturized hair ratio, and collagen, reticular fiber and elastic fiber mesh quantity (3 mm punch biopsies) 7. Proliferative epidermal/follicular cells, newly formed blood vessels, and presence of bulge stem cell niches (immunohistochemistry) | 1. Yes (p < 0.05) 2. Yes (p < 0.05) 3. Yes (p < 0.05) 4. Yes (p < 0.05) 5. Yes; 0.75/1* 6. Yes (p < 0.05 for most) 7. Yes (p < 0.05 for most) |
Patient self-satisfaction score following a Likert scale: 7 = very satisfied, 6 = satisfied, 5 = indifferent, 1 = unsatisfied, and 0 = very unsatisfied; most patients (15/19) declared noticeable hair loss decrease, 13/19 declared noticeable improvement in hair quality and appearance, and 11/19 stated they would continue with PRGF treatment | 2017 | [24] |
| Authors | PRP n° Treat | Int | Max F-up | Type of PRP Injections | Protocol | Activators | RPM orG | Centrif. Time | Blood Volume | PRP Volume |
|---|---|---|---|---|---|---|---|---|---|---|
| Takikawa et al. [15] | 5 | 2–3 wks | 12 wks | Subcutaneous injection (3 mL) into selected 1 × 1 cm areas measured from the nasal tip and upper part of the auricular base | Manual Double Spi | - |
a. 1700 rpm b. 3000 rpm |
a. 15 min b. 5 min | 15 mL | 3 mL |
| Schiavone et al. [16] | 2 | 3 mos | 6 mos | After local anesthesia (xylocaine 1%, with adrenaline 1:100,000) was administered, cutaneous inflammation was induced via application of gentle pressure using 1.0-mm-deep Scalp-roller to favor activation of injected platelets; then, superficial injections were administered 1 cm apart |
GPS III Platelet Separation System a. Single spin at baseline b. Double spin at 3 months | No (Scalp roller used to favor– platelet activation | - | - | a. 60 mL b. 40 mL |
a. 6–8 mL PRP + 3–4 mL of plasmatic protein concentrate = 9–12 mL; 0.2–0.3 mL per injection b. Same as above |
| Gkini et al. [17] | 3 (+1 booster) | 21 days (booster 6 mos after onset) | 1 year | Injections (0.05–0.1 mL/cm2) were performed using nappage technique in affected areas to a depth of 1.5–2.5 mm; a specific area was checked at all times by defining a “V” (Kang’s point) |
RegenA-PRPCentri (Regenlab) Single spin method |
Calcium gluconate (0.1 mL per 0.9 mL of PRP; 1:9 ratio) | 1500× g | 5 min | 16 mL | 6 mL (0.05–0.1 mL/cm2) |
| Khatu et al. [18] | 4 | 2 wks | 12 wks | Nappage technique injections (2–3 mL) into a prefixed 1 × 1 cm squared area over the right parietal area; anesthetic cream was applied before each treatment after cleaning the skin with cetavlon, spirit, and povidoneiodine | Manual Double Spin | Calcium chloride (1:9 ratio) | a. 1500 rpm b. 2500 rpm | a. 6 min b. 15 min | 20 mL | 2–3 mL |
| Cervelli et al. [8] | 3 | 4 wks |
1 year (at baseline and 14 wks, 6 mos, and 12 mos after initial treatment) | Intradermal injections (0.1 mL/cm2) into 2 of the 4 selected halves (e.g., frontal or parietal) (placebo was injected into the other 2 halves) after the scalp was cleansed with 70% alcohol; local anesthesia was not used | Cascade-Selphyl-Esforax Kit | Ca2+ | 1100× g | 10 min | 18 mL | 9 mL |
| Gentile et al. [3] | 3 | 4 wks | 2 years (at baseline and 2, 6, 12, 16, and 23 mos after initial treatment) | Interfollicular injections of PRP (0.1 mL/cm2) within 2 of the 4 selected areas of the scalp (physiologic solution into the other 2 areas), after cleaning skin with 70% alcohol; target areas were marked with semi-permanent tattoos for subsequent treatment and evaluation; local anesthesia was not used |
a. Cascade-Selphyl- Esforax system b. PRL platelet-rich lipotransfert system c. C-punt system | a. Ca2+ b. Nothing c. Nothing |
a. 1100× g b. 1200 rpm c 1200 rpm + double spin 1900 rpm |
a. 10 min b. 10 min c. 10 min +10 min |
a. 18 mL b. 55 mL c. 55 mL |
a. 9 mL b. 20 mL c. 20 mL |
| Singhal et al. [19] | 4 | 2–3 wks | 3 mos (at 1-wk intervals) | Injections using nappage technique (multiple small injections in linear pattern 1 cm apart) after area was cleansed with spirit and povidone-iodine | Double spin method | Calcium chloride (9:1 ratio) |
a. 1500 rpm b. 2500 rpm |
a. 6 min b. 15 min | 20 mL | 8–12 mL |
| Alves and Grimalt [20] | 3 | 4 wks | 6 mos (at 3-mo intervals) | Injections (0.15 mL/cm2) within four 1 × 1 cm selected circular areas of the frontal and occipital scalp (marked with a dot tattoo) depending on the treatment-designated side of the scalp (vs. control side of the scalp received placebo (normal saline); no local anesthesia was used | Single spin method | Calcium chloride (10%, 0.15 mL) | 460× g | 8 min | 18 mL | 3 mL |
| Puig et al. [21] | 1 | N.a | 26 wks (at 4-wk intervals) | Single subcutaneous injection within the 4 cm2 area in the central scalp (termed the “hair check data box”), after anesthesia (2% lidocaine and 0.5% bupivacaine) was administered | Angel PRP system (Cytomedix) | Nothing | - | - | 60 mL | 10 mL |
| Mapar et al. [22] | 2 | 4 wks | 6 mos (at 1, 3, and 6 mos after initial treatment) | Injections (1.5 mL of PRP) within one of two 2.5 × 2.5 cm square regions, at least 3 cm apart, in the scalp randomly assigned to be a case square (control square received 1.5 mL of normal saline); randomization of case and control squares was performed using a random number table; iron oxide- and titanium dioxide-containing substances were used to tattoo the corners of the squares | Double spin method using Tubex PRP tube (Moohan Enterprise) |
Calcium gluconate (0.1 mL per mL of PRP) | a. 3000 rpm b. 3300 rpm |
a. 6 min b. 3 min | 9 mL | 1.5 mL |
| Gupta et al. [23] | 6 | 2 wks | 6 mos | Scalp was activated by micro-needling; then, PRP was massaged into the vertex of the scalp (10 cm from the glabella) | Double spin method | - | - | - | - | - |
| Anitua et al. [24] | 5 |
1 mo for first 4 sessions; final session 7 mos after start point | 1 year | Intradermal injections of PRGF into hair-depleted areas | Single spin method | PRGF activator (BTI Biotechnology Institute) | 580 rpm | 8 min | 18 mL | 3–4 mL |
| Growth Factors | Bio-Molecular Pathway in Hair Re-Growth |
|---|---|
| VEGF | Improves perifollicular angiogenesis; Elevated expression in dermal papilla cells during anagen phase; Endothelial cell-specific mitogen; Micro-vascular permeability and perifollicular vascularization; |
| EGF | Improves the activity and growth of follicle outer-root sheath cells by activation of Wnt/β-catenin signaling; Cell growth modulator during follicular differentiation; Proliferation and migration of follicular outer root sheath cells; |
| FGF | Improves the advancement of hair follicles; Anagen phase induction via B-catenin expression; Angiogenesis; Dermal fibroblast and hair follicle mitogen; |
| PDGF | Up-regulate the genes associated with HF separation, induction, and control of anagen; Angiogenesis and vascularization; Hair follicle dermal stem cell proliferation; Mesenchymal stem cell mitogen; |
| IGF-1 | Improves the migration, survival, and proliferation of HF cells; Hair follicle proliferation during development; Increase hair density and inhibit apoptosis; |
| HGF | Enhance the proliferation of follicular epithelial cells Hair follicle elongation; Inhibits catagen phase induction; |
| TGF-ß | Stimulates the signaling pathways that manage the Hair cycle; Extracellular matrix synthesis; Fibroblast and mesenchymal stem cell proliferation; Hair folliculogenesis and maturation; |
| IL-6 | Involved in WIHN through STAT3 enactment |
| IGFBP-1 to -6 | Manages the IGF-1 effect and its connection with extracellular matrix proteins at the Hair follicle level |
| BMP | Maintains the DPC phenotype (fundamental for stimulation of HFSCs) |
| BMPR1 | Maintains the proper identity of the DPCs (basic for explicit DPC work) |
| M-CSF | Involved in wound-induced hair growth |
| M-CSFR | Involved in wound-induced hair growth |
| Wnt3a | Involved in HF advancement through β-catenin signaling |
| PGE2 | Stimulates anagen in HF |
| PGF2α | Enhance change from telogen to anagen |
| BIO | GSK-3 inhibitor |
| PGD2 | Enhances follicle regeneration |
| Iron and l-lysine95 | Still under examination |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gentile, P.; Garcovich, S. Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy. Int. J. Mol. Sci. 2020, 21, 2702. https://doi.org/10.3390/ijms21082702
Gentile P, Garcovich S. Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy. International Journal of Molecular Sciences. 2020; 21(8):2702. https://doi.org/10.3390/ijms21082702
Chicago/Turabian StyleGentile, Pietro, and Simone Garcovich. 2020. "Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy" International Journal of Molecular Sciences 21, no. 8: 2702. https://doi.org/10.3390/ijms21082702
APA StyleGentile, P., & Garcovich, S. (2020). Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy. International Journal of Molecular Sciences, 21(8), 2702. https://doi.org/10.3390/ijms21082702






