Hormonal Effects on Hair Follicles
Abstract
1. Introduction
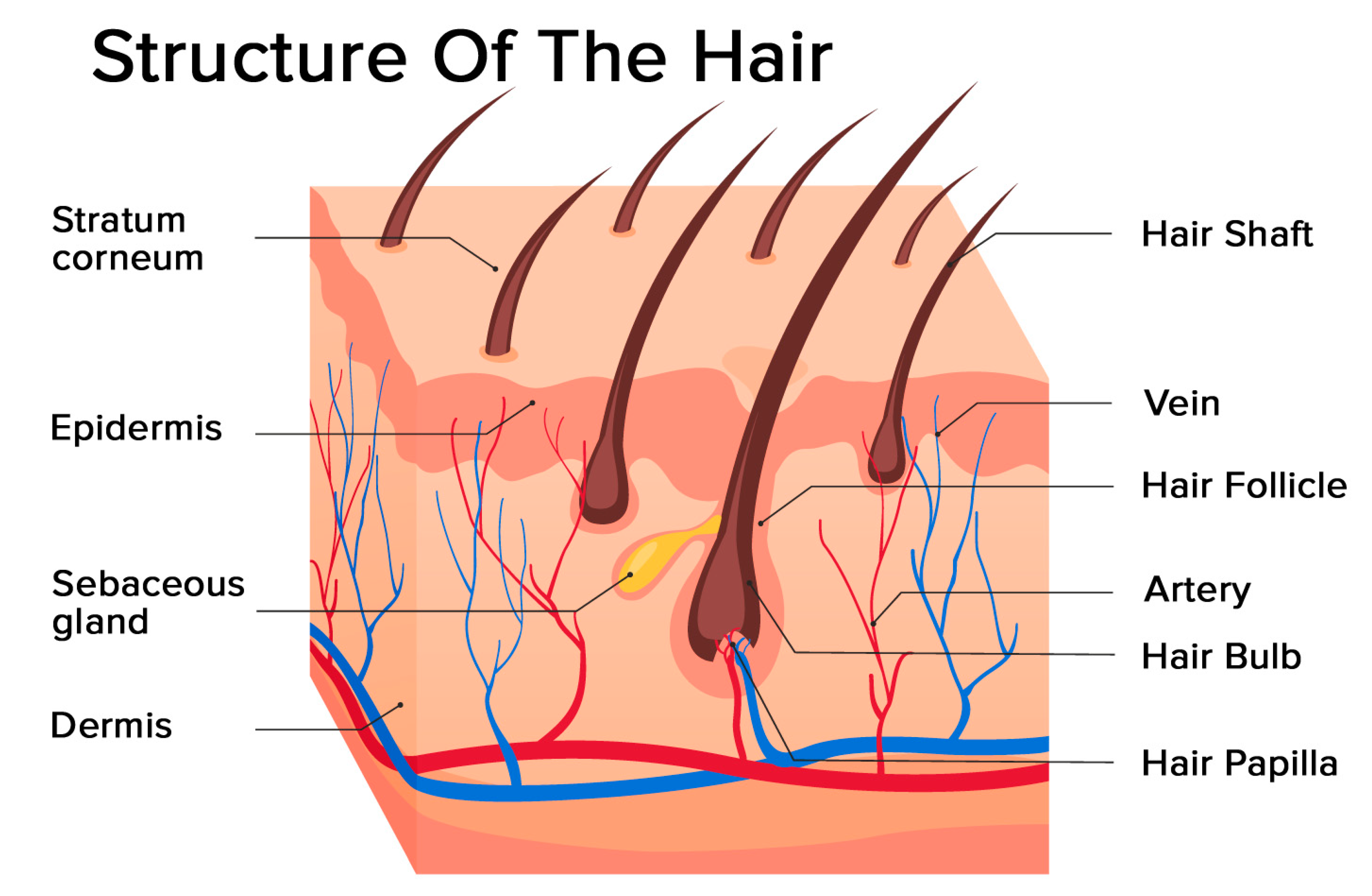
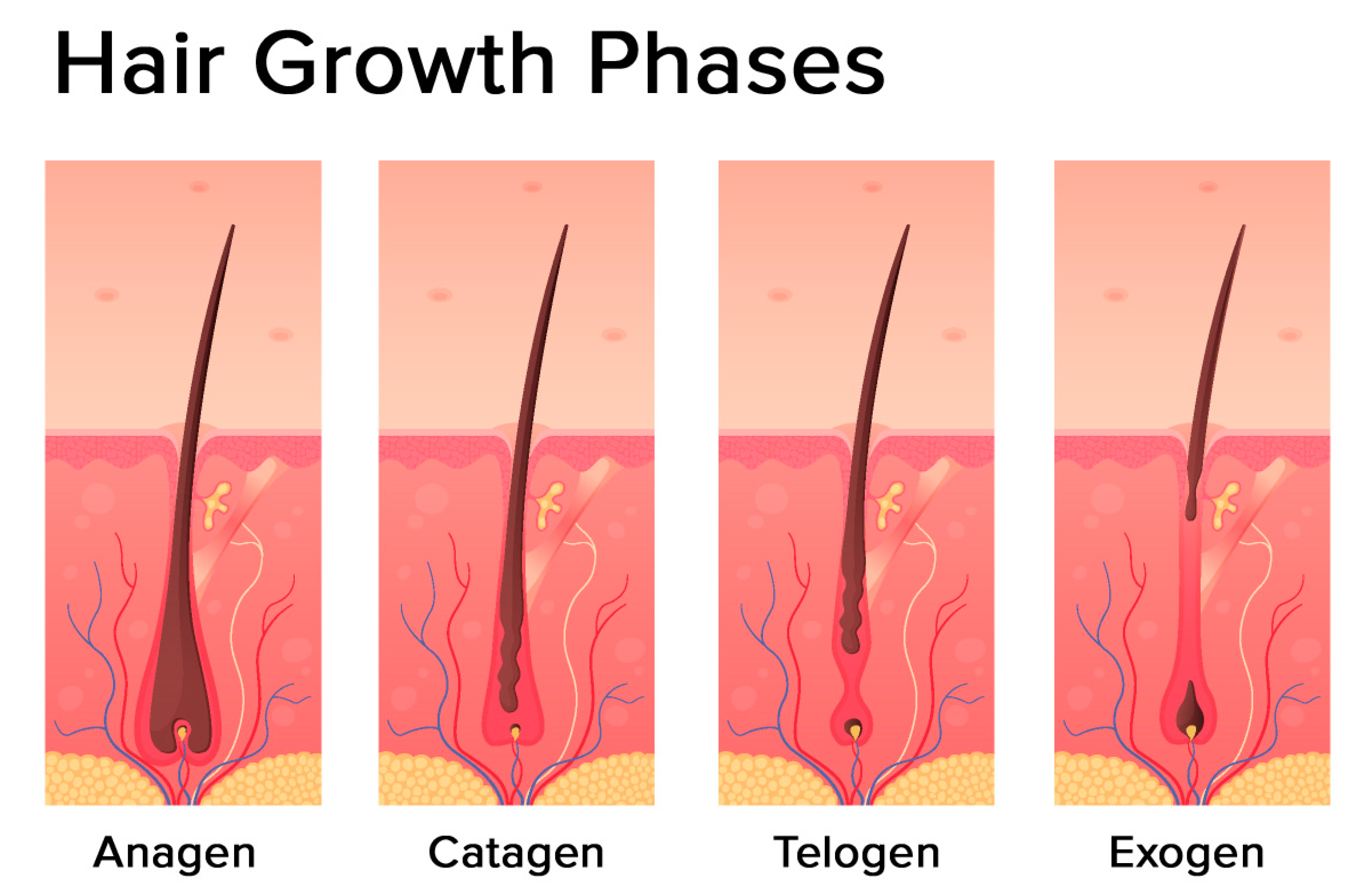
2. Hair Follicle Biology
2.1. Structure of the Hair Follicle
2.2. The Hair Follicle Cycle
3. Endocrine Regulations of the Hair Follicle
3.1. Androgens
3.1.1. General Information
3.1.2. Androgen Receptor
3.1.3. Androgen Action
3.2. Sex Hormones
3.2.1. Estradiol
3.2.2. Progesterone
3.3. Prolactin
3.4. Thyroid Gland Hormones
3.5. Melatonin
3.6. Other Hormones
3.6.1. CRH
3.6.2. ACTH
3.6.3. CORTISOL
3.6.4. TRH
3.6.5. GALANIN
4. Hormonal Effects on Hair at Different Stages of the Life of Women
4.1. Reproductive Age
4.2. Pregnancy
4.3. Menopause
5. Conclusions
Funding
Conflicts of Interest
Abbreviations
| T | Testosterone |
| DHT | Dihydrotestosterone |
| DHEA-S | Dehydroepiandrosterone sulfate |
| A | Androstendione |
| E2 | Estrogen |
| PRL | Prolactin |
| IGF-1 | Insulin-like growth factor |
| TGF-beta | Transforming growth factor-beta |
| 17 beta-HSD | 17β-Hydroxysteroid dehydrogenase |
| E2 | Estradiol |
| LH | Luteinizing hormone |
| PRL-R | Prolactin receptors |
| HFs | Human hair follicles |
| IFNγ | Interferon gamma |
| TNFα | Tumor necrosis factor alpha |
| TRH | Thyrotropin-releasing hormone |
| TSH | Thyroid-stimulating hormone |
| T4 | Thyroxine |
| T3 | Triiodothyronine |
| ATP | Adenosine triphosphate |
| HPT axis | Hypothalamic-pituitary-thyroid axis |
| CRH | Corticotropin-releasing hormone |
| HPA axis | Hypothalamic-pituitary-adrenal axis |
| POMC | Pro-opiomelanocortin |
| ACTH | Adrenocorticotropic hormone |
| HCC | Hair cortisol concentration |
| TGF-beta2 | Transforming growth factor-beta 2 |
| GAL | Galanin |
| PCOS | Polycystic ovary syndrome |
| FPHL | Female pattern hair loss |
| PPTE | Postpartum telogen effluvium |
| SHBG | Sex hormone binding globulin |
References
- Ceruti, J.M.; Leiros, G.J.; Balana, M.E. Androgens and androgen receptor action in skin and hair follicles. Mol. Cell. Endocrinol. 2018, 465, 122–133. [Google Scholar] [CrossRef]
- Inui, S.; Itami, S. Androgen actions on the human hair follicle: Perspectives. Exp. Dermatol. 2013, 22, 168–171. [Google Scholar] [CrossRef]
- Mason, K.A.; Schoelwer, M.J.; Rogol, A.D. Androgens during infancy, childhood, and adolescence: Physiology and use in clinical practice. Endocr. Rev. 2020, 41. [Google Scholar] [CrossRef]
- Schneider, M.R.; Schmidt-Ullrich, R.; Paus, R. The hair follicle as a dynamic miniorgan. Curr. Biol. 2009, 19, R132–R142. [Google Scholar] [CrossRef]
- Lepselter, J.; Elman, M. Biological and clinical aspects in laser hair removal. J. Dermatol. Treat. 2004, 15, 72–83. [Google Scholar] [CrossRef]
- Hernandez, I.; Alam, M.; Platt, C.; Hardman, J.; Smart, E.; Poblet, E.; Bertolini, M.; Paus, R.; Jimenez, F. A technique for more precise distinction between catagen and telogen human hair follicles ex vivo. J. Am. Acad. Dermatol. 2018, 79, 558–559. [Google Scholar] [CrossRef]
- Walters, K.A.; Rodriguez Paris, V.; Aflatounian, A.; Handelsman, D.J. Androgens and ovarian function: Translation from basic discovery research to clinical impact. J. Endocrinol. 2019, 242, R23–R50. [Google Scholar] [CrossRef]
- Hoffmann, R.; Rot, A.; Niiyama, S.; Billich, A. Steroid sulfatase in the human hair follicle concentrates in the dermal papilla. J. Investig. Dermatol. 2001, 117, 1342–1348. [Google Scholar] [CrossRef][Green Version]
- Chaturvedi, A.P.; Dehm, S.M. Androgen receptor dependence. Adv. Exp. Med. Biol 2019, 1210, 333–350. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Moriya, T.; Suzuki, T.; Chiba, M.; Ishida, K.; Takeyama, J.; Endoh, M.; Watanabe, M.; Sasano, H. Sex steroid hormone receptors in human skin appendage and its neoplasms. Endocr. J. 2005, 52, 317–325. [Google Scholar] [CrossRef]
- Brown, T.M.; Krishnamurthy, K. Histology, Hair and Follicle. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2020. [Google Scholar]
- Ohnemus, U.; Uenalan, M.; Inzunza, J.; Gustafsson, J.A.; Paus, R. The hair follicle as an estrogen target and source. Endocr. Rev. 2006, 27, 677–706. [Google Scholar] [CrossRef]
- Randall, V.A. Hormonal regulation of hair follicles exhibits a biological paradox. Semin. Cell Dev. Biol. 2007, 18, 274–285. [Google Scholar] [CrossRef]
- Truong, A.T.; Duez, C.; Belayew, A.; Renard, A.; Pictet, R.; Bell, G.I.; Martial, J.A. Isolation and characterization of the human prolactin gene. EMBO J. 1984, 3, 429–437. [Google Scholar] [CrossRef]
- Goffin, V.; Binart, N.; Touraine, P.; Kelly, P.A. Prolactin: The new biology of an old hormone. Annu. Rev. Physiol. 2002, 64, 47–67. [Google Scholar] [CrossRef]
- Castle-Miller, J.; Bates, D.O.; Tortonese, D.J. Mechanisms regulating angiogenesis underlie seasonal control of pituitary function. Proc. Natl. Acad. Sci. USA 2017, 114, E2514–E2523. [Google Scholar] [CrossRef]
- Ignacak, A.; Kasztelnik, M.; Sliwa, T.; Korbut, R.A.; Rajda, K.; Guzik, T.J. Prolactin—Not only lactotrophin. A “new” view of the “old” hormone. J. Physiol. Pharmacol. 2012, 63, 435–443. [Google Scholar]
- Foitzik, K.; Krause, K.; Conrad, F.; Nakamura, M.; Funk, W.; Paus, R. Human scalp hair follicles are both a target and a source of prolactin, which serves as an autocrine and/or paracrine promoter of apoptosis-driven hair follicle regression. Am. J. Pathol. 2006, 168, 748–756. [Google Scholar] [CrossRef]
- Martinet, L.; Allain, D.; Weiner, C. Role of prolactin in the photoperiodic control of moulting in the mink (Mustela vison). J. Endocrinol. 1984, 103, 9–15. [Google Scholar] [CrossRef]
- Duncan, M.J.; Goldman, B.D. Hormonal regulation of the annual pelage color cycle in the Djungarian hamster, Phodopus sungorus. II. Role of prolactin. J. Exp. Zool. 1984, 230, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Craven, A.J.; Ormandy, C.J.; Robertson, F.G.; Wilkins, R.J.; Kelly, P.A.; Nixon, A.J.; Pearson, A.J. Prolactin signaling influences the timing mechanism of the hair follicle: Analysis of hair growth cycles in prolactin receptor knockout mice. Endocrinology 2001, 142, 2533–2539. [Google Scholar] [CrossRef]
- Langan, E.A.; Vidali, S.; Pigat, N.; Funk, W.; Lisztes, E.; Bíró, T.; Goffin, V.; Griffiths, C.E.M.; Paus, R. Tumour necrosis factor alpha, interferon gamma and substance P are novel modulators of extrapituitary prolactin expression in human skin. PLoS ONE 2013, 8, e60819. [Google Scholar] [CrossRef]
- Slominski, A.; Malarkey, W.B.; Wortsman, J.; Asa, S.L.; Carlson, A. Human skin expresses growth hormone but not the prolactin gene. J. Lab. Clin. Med. 2000, 136, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Glickman, S.P.; Rosenfield, R.L.; Bergenstal, R.M.; Helke, J. Multiple androgenic abnormalities, including elevated free testosterone, in hyperprolactinemic women. J. Clin. Endocrinol. Metab. 1982, 55, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Schiebinger, R.J.; Chrousos, G.P.; Culter, G.B.J.; Loriaux, D.L. The effect of serum prolactin on plasma adrenal androgens and the production and metabolic clearance rate of dehydroepiandrosterone sulfate in normal and hyperprolactinemic subjects. J. Clin. Endocrinol. Metab. 1986, 62, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Serafini, P.; Lobo, R.A. Prolactin modulates peripheral androgen metabolism. Fertil. Steril. 1986, 45, 41–46. [Google Scholar] [CrossRef]
- Van Beek, N.; Bodo, E.; Kromminga, A.; Gaspar, E.; Meyer, K.; Zmijewski, M.A.; Slominski, A.; Wenzel, B.E.; Paus, R. Thyroid hormones directly alter human hair follicle functions: Anagen prolongation and stimulation of both hair matrix keratinocyte proliferation and hair pigmentation. J. Clin. Endocrinol. Metab. 2008, 93, 4381–4388. [Google Scholar] [CrossRef]
- Bodo, E.; Kromminga, A.; Biro, T.; Borbiro, I.; Gaspar, E.; Zmijewski, M.A.; van Beek, N.; Langbein, L.; Slominski, A.T.; Paus, R. Human female hair follicles are a direct, nonclassical target for thyroid-stimulating hormone. J. Investig. Dermatol. 2009, 129, 1126–1139. [Google Scholar] [CrossRef]
- Wallace, D.C.; Fan, W.; Procaccio, V. Mitochondrial energetics and therapeutics. Annu. Rev. Pathol. 2010, 5, 297–348. [Google Scholar] [CrossRef]
- Vidali, S.; Knuever, J.; Lerchner, J.; Giesen, M.; Biro, T.; Klinger, M.; Kofler, B.; Funk, W.; Poeggeler, B.; Paus, R. Hypothalamic-Pituitary-Thyroid axis hormones stimulate mitochondrial function and biogenesis in human hair follicles. J. Investig. Dermatol. 2014, 134, 33–42. [Google Scholar] [CrossRef]
- Knuever, J.; Poeggeler, B.; Gaspar, E.; Klinger, M.; Hellwig-Burgel, T.; Hardenbicker, C.; Toth, B.I.; Biro, T.; Paus, R. Thyrotropin-Releasing hormone controls mitochondrial biology in human epidermis. J. Clin. Endocrinol. Metab. 2012, 97, 978–986. [Google Scholar] [CrossRef]
- Slominski, A.; Tobin, D.J.; Zmijewski, M.A.; Wortsman, J.; Paus, R. Melatonin in the skin: Synthesis, metabolism and functions. Trends Endocrinol. Metab. 2008, 19, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Kromminga, A.; Dunlop, T.W.; Tychsen, B.; Conrad, F.; Suzuki, N.; Memezawa, A.; Bettermann, A.; Aiba, S.; Carlberg, C.; et al. A role of melatonin in neuroectodermal-mesodermal interactions: The hair follicle synthesizes melatonin and expresses functional melatonin receptors. FASEB J. 2005, 19, 1710–1712. [Google Scholar] [CrossRef] [PubMed]
- Janjetovic, Z.; Jarrett, S.G.; Lee, E.F.; Duprey, C.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites protect human melanocytes against UVB-induced damage: Involvement of NRF2-mediated pathways. Sci. Rep. 2017, 7, 1274. [Google Scholar] [CrossRef]
- Haslam, I.S.; Jadkauskaite, L.; Szabo, I.L.; Staege, S.; Hesebeck-Brinckmann, J.; Jenkins, G.; Bhogal, R.K.; Lim, F.L.; Farjo, N.; Farjo, B.; et al. Oxidative damage control in a human (mini-) organ: Nrf2 Activation protects against oxidative stress-induced hair growth inhibition. J. Investig. Dermatol. 2017, 137, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Ermak, G.; Hwang, J.; Chakraborty, A.; Mazurkiewicz, J.E.; Mihm, M. Proopiomelanocortin, corticotropin releasing hormone and corticotropin releasing hormone receptor genes are expressed in human skin. FEBS Lett. 1995, 374, 113–116. [Google Scholar] [CrossRef]
- Ito, N.; Ito, T.; Kromminga, A.; Bettermann, A.; Takigawa, M.; Kees, F.; Straub, R.H.; Paus, R. Human hair follicles display a functional equivalent of the hypothalamic-pituitary-adrenal axis and synthesize cortisol. FASEB J. 2005, 19, 1332–1334. [Google Scholar] [CrossRef]
- Paus, R.; Maurer, M.; Slominski, A.; Czarnetzki, B.M. Mast cell involvement in murine hair growth. Dev. Biol. 1994, 163, 230–240. [Google Scholar] [CrossRef]
- Rose, J. Adrenocorticotropic Hormone (ACTH) but not alpha-melanocyte stimulating hormone (a-MSH) as a mediator of adrenalectomy induced hair growth in mink. J. Investig. Dermatol. 1998, 110, 456–457. [Google Scholar] [CrossRef]
- Cieszynski, L.; Jendrzejewski, J.; Wisniewski, P.; Owczarzak, A.; Sworczak, K. Hair cortisol concentration in a population without hypothalamic-pituitary-adrenal axis disorders. Adv. Clin. Exp. Med. 2019, 28, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Sunesara, I.; Rehm, K.E.; Marshall, G.D., Jr. Hair cortisol concentrations are associated with hair growth rate. Neuroimmunomodulation 2016, 23, 287–294. [Google Scholar] [CrossRef]
- Thom, E. Stress and the hair growth cycle: Cortisol-Induced hair growth disruption. J. Drugs Dermatol. JDD 2016, 15, 1001–1004. [Google Scholar] [PubMed]
- Gáspár, E.; Hardenbicker, C.; Bodó, E.; Wenzel, B.; Ramot, Y.; Funk, W.; Kromminga, A.; Paus, R. Thyrotropin releasing hormone (TRH): A new player in human hair-growth control. FASEB J. 2009, 24, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Holub, B.S.; Kloepper, J.E.; Tóth, B.I.; Bíro, T.; Kofler, B.; Paus, R. The neuropeptide galanin is a novel inhibitor of human hair growth. Br. J. Dermatol. 2012, 167, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, R.L. Clinical Practice. Hirsutism. N. Engl. J. Med. 2005, 353, 2578–2588. [Google Scholar] [CrossRef]
- Kini, S.; Ramalingam, M. Hirsutism. Obstet. Gynaecol. Reprod. Med. 2018, 28, 129–135. [Google Scholar] [CrossRef]
- Hatch, R.; Rosenfield, R.L.; Kim, M.H.; Tredway, D. Hirsutism: Implications, etiology, and management. Am. J. Obstet. Gynecol. 1981, 140, 815–830. [Google Scholar] [CrossRef]
- Ferriman, D.; Gallwey, J.D. Clinical assessment of body hair growth in women. J. Clin. Endocrinol. Metab. 1961, 21, 1440–1447. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Carmina, E.; Dewailly, D.; Gambineri, A.; Kelestimur, F.; Moghetti, P.; Pugeat, M.; Qiao, J.; Wijeyaratne, C.N.; Witchel, S.F.; et al. Epidemiology, diagnosis and management of hirsutism: A consensus statement by the Androgen Excess and Polycystic Ovary Syndrome Society. Hum. Reprod. Update 2012, 18, 146–170. [Google Scholar] [CrossRef]
- Ehrmann, D.A.; Rosenfield, R.L.; Barnes, R.B.; Brigell, D.F.; Sheikh, Z. Detection of functional ovarian hyperandrogenism in women with androgen excess. N. Engl. J. Med. 1992, 327, 157–162. [Google Scholar] [CrossRef]
- Martin, K.A.; Anderson, R.R.; Chang, R.J.; Ehrmann, D.A.; Lobo, R.A.; Murad, M.H.; Pugeat, M.M.; Rosenfield, R.L. Evaluation and treatment of hirsutism in premenopausal women: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1233–1257. [Google Scholar] [CrossRef]
- Carmina, E.; Azziz, R.; Bergfeld, W.; Escobar-Morreale, H.F.; Futterweit, W.; Huddleston, H.; Lobo, R.; Olsen, E. Female Pattern hair loss and androgen excess: A report from the multidisciplinary androgen excess and PCOS committee. J. Clin. Endocrinol. Metab. 2019, 104, 2875–2891. [Google Scholar] [CrossRef] [PubMed]
- Olsen, E.A.; Hordinsky, M.; Roberts, J.L.; Whiting, D.A. Female pattern hair loss. J. Am. Acad. Dermatol. 2002, 47, 795. [Google Scholar] [CrossRef] [PubMed]
- Birch, M.P.; Lalla, S.C.; Messenger, A.G. Female pattern hair loss. Clin. Exp. Dermatol. 2002, 27, 383–388. [Google Scholar] [CrossRef]
- Cousen, P.; Messenger, A. Female pattern hair loss in complete androgen insensitivity syndrome. Br. J. Dermatol. 2010, 162, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Yip, L.; Zaloumis, S.; Irwin, D.; Severi, G.; Hopper, J.; Giles, G.; Harrap, S.; Sinclair, R.; Ellis, J. Gene-Wide association study between the aromatase gene (CYP19A1) and female pattern hair loss. Br. J. Dermatol. 2009, 161, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Ramos, P.M.; Brianezi, G.; Martins, A.C.; da Silva, M.G.; Marques, M.E.; Miot, H.A. Apoptosis in follicles of individuals with female pattern hair loss is associated with perifollicular microinflammation. Int. J. Cosmet. Sci. 2016, 38, 651–654. [Google Scholar] [CrossRef]
- Pekmezci, E.; Turkoglu, M.; Gokalp, H.; Kutlubay, Z. Minoxidil downregulates Interleukin-1 alpha gene expression in HaCaT Cells. Int. J. Trichol. 2018, 10, 108–112. [Google Scholar] [CrossRef]
- Futterweit, W.; Dunaif, A.; Yeh, H.C.; Kingsley, P. The prevalence of hyperandrogenism in 109 consecutive female patients with diffuse alopecia. J. Am. Acad. Dermatol. 1988, 19, 831–836. [Google Scholar] [CrossRef]
- Heymann, W.R. Cutaneous manifestations of thyroid disease. J. Am. Acad. Dermatol. 1992, 26, 885–902. [Google Scholar] [CrossRef]
- Puri, N. A study on cutaneous manifestations of thyroid disease. Indian J. Dermatol. 2012, 57, 247–248. [Google Scholar] [CrossRef]
- Nissimov, J.; Elchalal, U. Scalp hair diameter increases during pregnancy. Clin. Exp. Dermatol. 2003, 28, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Mirmirani, P. Hormonal changes in menopause: Do they contribute to a ’midlife hair crisis’ in women? Br. J. Dermatol. 2011, 165 (Suppl. 3), 7–11. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.Y.; Yoon, J.S.; Jo, S.J.; Shin, C.Y.; Shin, J.Y.; Kim, J.I.; Kwon, O.; Kim, K.H. A role of placental growth factor in hair growth. J. Dermatol. Sci. 2014, 74, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Mirallas, O.; Grimalt, R. The postpartum telogen effluvium fallacy. Skin Appendage Disord. 2016, 1, 198–201. [Google Scholar] [CrossRef]
- Yang, Y.; Han, Y.; Wang, W.; Du, T.; Li, Y.; Zhang, J.; Yang, D.; Zhao, X. Assessing new terminal body and facial hair growth during pregnancy: Toward developing a simplified visual scoring system for hirsutism. Fertil. Steril. 2016, 105, 494–500. [Google Scholar] [CrossRef][Green Version]
- Nassi, R.; Ladu, C.; Vezzosi, C.; Mannelli, M. Cushing’s syndrome in pregnancy. Gynecol. Endocrinol. 2015, 31, 102–104. [Google Scholar] [CrossRef]
- Verma, V.; Paul, S.; Chahal, K.; Singh, J. Pregnancy luteoma: A rare case report. Int. J. Appl. Basic Med. Res. 2016, 6, 282–283. [Google Scholar] [CrossRef]
- Papantoniou, N.; Belitsos, P.; Hatzipapas, I.; Rodolakis, A.; Papaspyrou, I.; Antsaklis, A. Excessive hirsutism in pregnancy because of Krukenberg tumor. J. Matern. Fetal Neonatal Med. 2011, 25, 869–871. [Google Scholar] [CrossRef]
- Piérard-Franchimont, C.; Piérard, G.E. Alterations in hair follicle dynamics in women. BioMed Res. Int. 2013, 2013, 957432. [Google Scholar] [CrossRef]
- Mirmirani, P. Managing hair loss in midlife women. Maturitas 2013, 74, 119–122. [Google Scholar] [CrossRef]
- Dolinko, A.V.; Ginsburg, E.S. Hyperandrogenism in menopause: A case report and literature review. Fertil. Res. Pract. 2015, 1, 7. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Wojnarowska, F. Physiological changes in scalp, facial and body hair after the menopause: A cross-sectional population-based study of subjective changes. Br. J. Dermatol. 2011, 164, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Zouboulis, C.C. Hormones and the pilosebaceus unit. Dermatoendocrinology 2009, 1, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Tsai, S.-J.; Sheu, H.-M.; Tsai, J.-C.; Zouboulis, C.C. Testosterone synthesized in cultured human SZ95 sebocytes derives mainly from from dehydroepiandrosterone. Exp. Dermatol. 2010, 19, 470–472. [Google Scholar] [CrossRef]
| Stage of Life | Main Hormones Involved | Effect |
|---|---|---|
| Puberty | Androgens | Transformation of vellus hair into terminal hair in the pubic and axillary regions |
| Reproductive age | Androgen excess (e.g PCOs, NCAH, Cushing’s syndrome, hyperprolactinemia) | Hirsutism |
| Unknown/unexplained role of sex hormones | Female pattern hair loss | |
| Hyperthyreosis | Alopecia areata | |
| Hypothyreosis | Diffuse alopecia | |
| Pregnancy | High levels of estrogen, progesterone, prolactin, and growth factors | Increases in the hair growth, in the hair diameter, and in the anagen/telogen ratio |
| Puerperium | Decrease in estrogen and progesterone | Postpartum telogen effluvium |
| Menopause | Estrogen depletion | Female pattern hair loss Facial hirsutism |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grymowicz, M.; Rudnicka, E.; Podfigurna, A.; Napierala, P.; Smolarczyk, R.; Smolarczyk, K.; Meczekalski, B. Hormonal Effects on Hair Follicles. Int. J. Mol. Sci. 2020, 21, 5342. https://doi.org/10.3390/ijms21155342
Grymowicz M, Rudnicka E, Podfigurna A, Napierala P, Smolarczyk R, Smolarczyk K, Meczekalski B. Hormonal Effects on Hair Follicles. International Journal of Molecular Sciences. 2020; 21(15):5342. https://doi.org/10.3390/ijms21155342
Chicago/Turabian StyleGrymowicz, Monika, Ewa Rudnicka, Agnieszka Podfigurna, Paulina Napierala, Roman Smolarczyk, Katarzyna Smolarczyk, and Blazej Meczekalski. 2020. "Hormonal Effects on Hair Follicles" International Journal of Molecular Sciences 21, no. 15: 5342. https://doi.org/10.3390/ijms21155342
APA StyleGrymowicz, M., Rudnicka, E., Podfigurna, A., Napierala, P., Smolarczyk, R., Smolarczyk, K., & Meczekalski, B. (2020). Hormonal Effects on Hair Follicles. International Journal of Molecular Sciences, 21(15), 5342. https://doi.org/10.3390/ijms21155342






