Flotillins: At the Intersection of Protein S-Palmitoylation and Lipid-Mediated Signaling
Abstract
1. Introduction
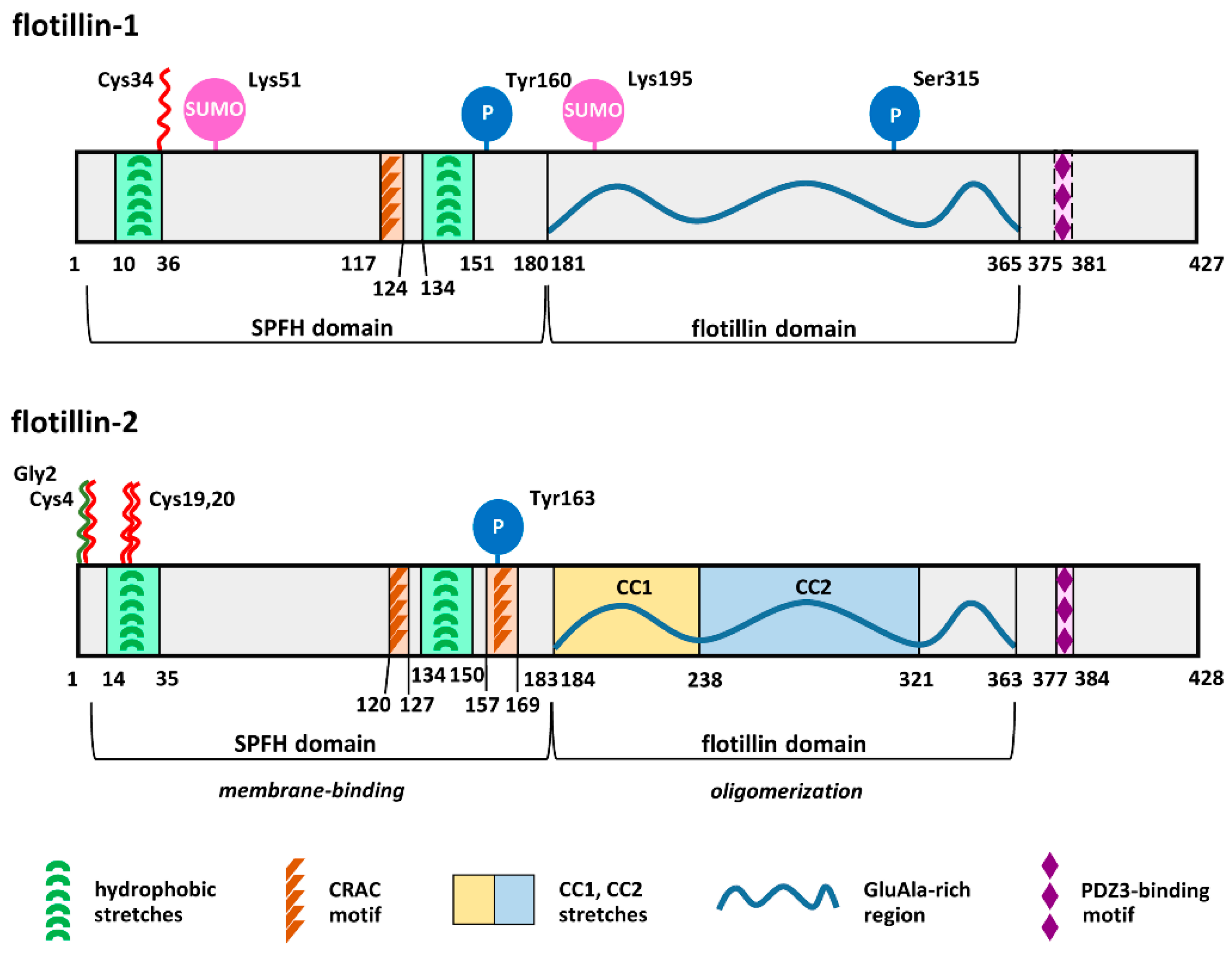
2. Acylation and Sphingosine Binding Affect Association of Flotillins with Membranes
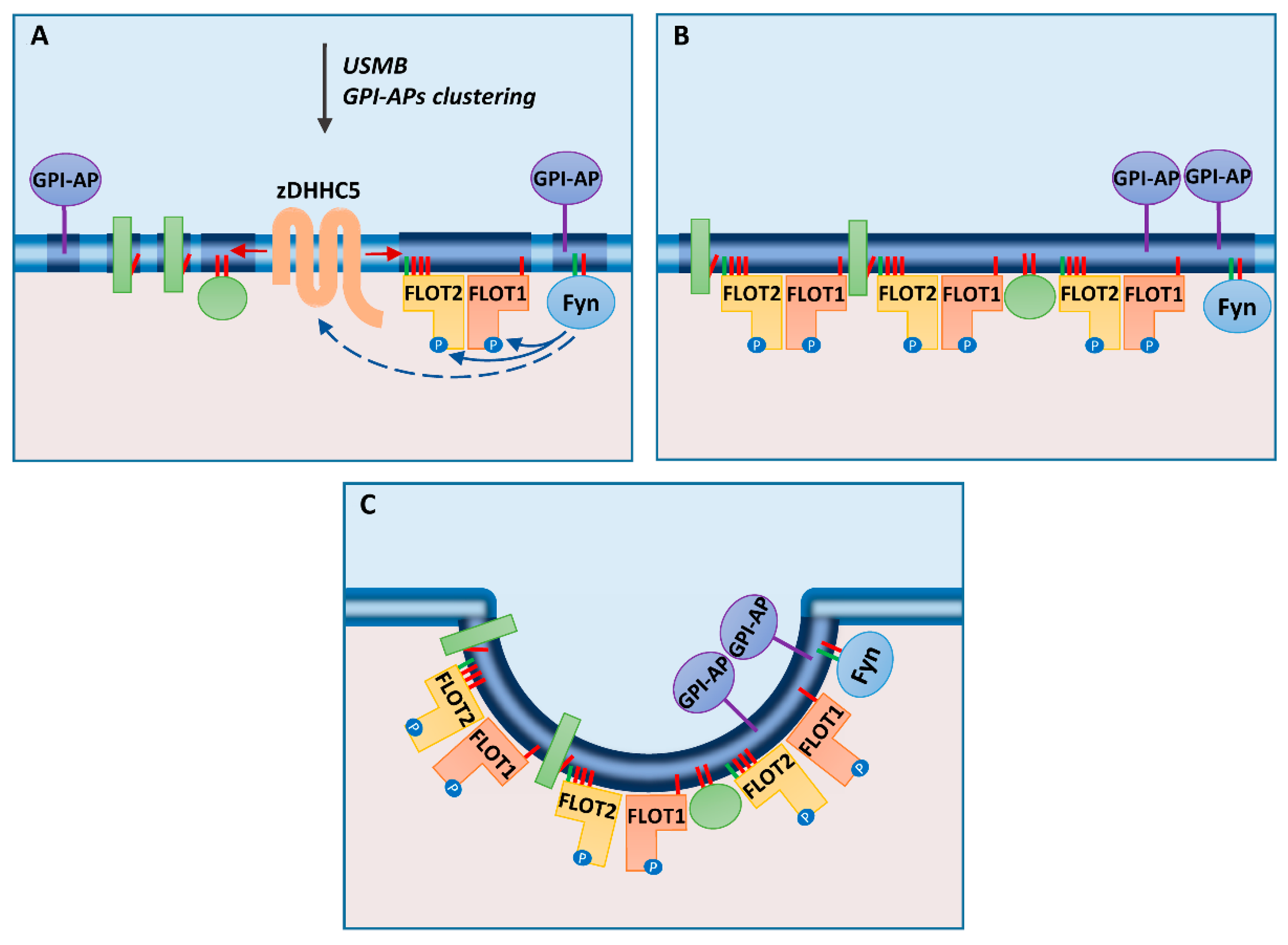
3. Flotillins Associate with Plasma Membrane Rafts
4. Association of Flotillins with Rafts Facilitates Endocytosis and Recycling of Certain Proteins
5. Flotillin-Mediated Endocytosis Requires Protein S-palmitoylation
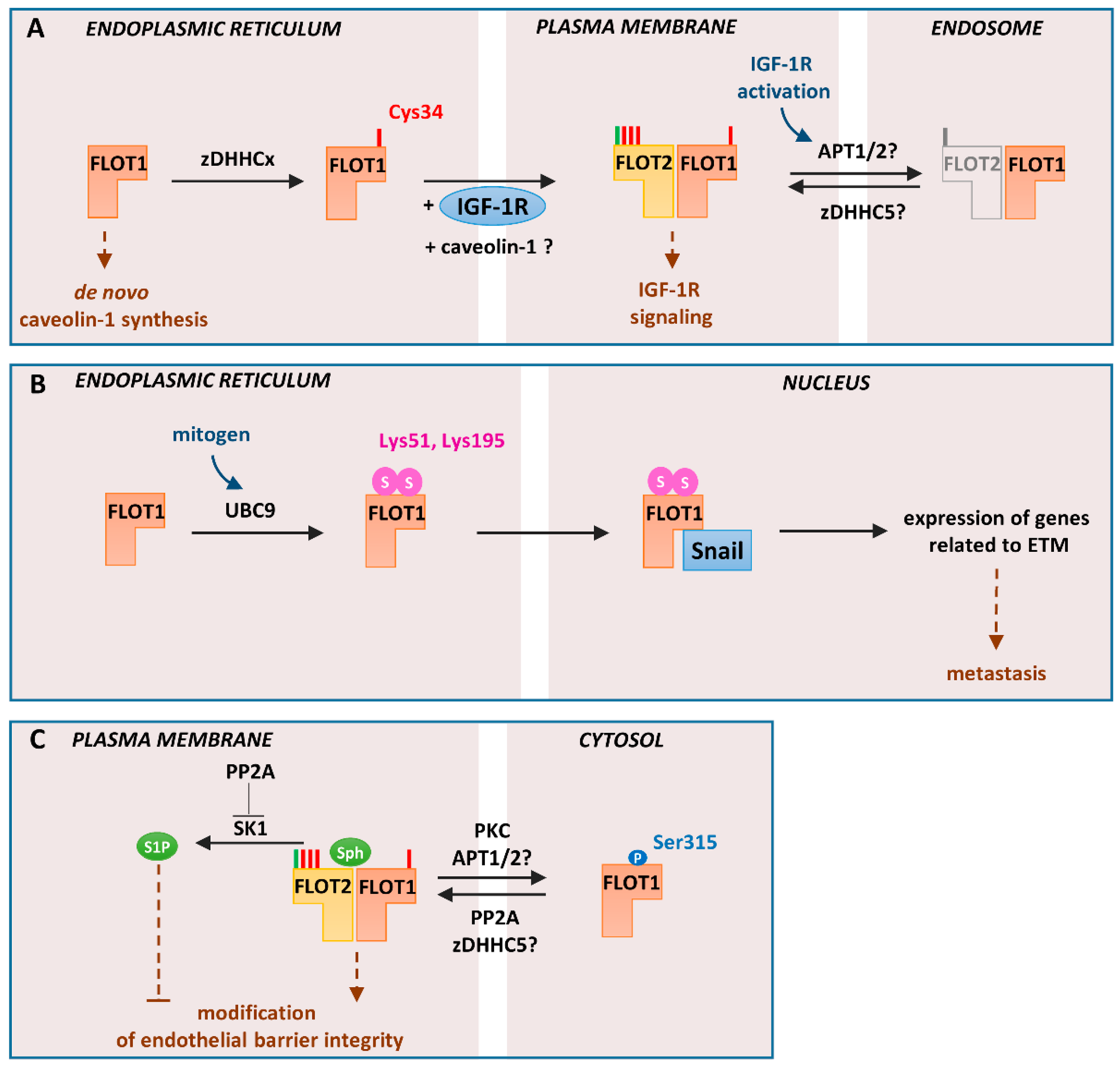
6. Some Unique Functions of Flotillin-1 Are Determined by Its S-palmitoylation
7. Flotillins Affect Sphingosine-1-phosphate Signaling and Are Indirectly Linked with PI(4,5)P2 Turnover
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor |
| APP | amyloid precursor protein |
| CC | coiled-coil |
| CRAC | cholesterol recognition/interaction amino acid motif |
| DAT | dopamine transporter |
| DRM | detergent resistant membranes |
| ESCRT | endosomal sorting complex required for transport |
| ETM | endothelial-to-mesenchymal |
| EGF | epidermal growth factor |
| EVs | extracellular vesicles |
| IGF-1 | insulin-like growth factor-1 |
| GPI | glycosylphosphatidylinositol |
| LPS | lipopolysaccharide |
| MAGUK | membrane-associated guanylate kinase |
| MEF | mouse embryonic fibroblasts |
| MEND | massive endocytosis |
| MPP1 | membrane palmitoylated protein 1 |
| PDZ | domain present in PSD-95, Dlg, and ZO-1/2 |
| PI(4,5)P2 | phosphatidylinositol 4,5-bisphosphate |
| PKC | protein kinase C |
| PPA2 | protein phosphatase 2A |
| S1P | sphingosine-1-phosphate |
| SK | sphingosine kinase |
| SPFH | domain present in stomatin, prohibitin, flotillin, and HflC/K |
| TCR | T cell receptor |
| TLR | Toll-like receptor |
| USMB | ultrasound, microbubbles and desipramine |
| zDHHC | zinc finger and Asp-His-His-Cys domain-containing |
References
- Otto, G.P.; Nichols, B.J. The roles of flotillin microdomains--endocytosis and beyond. J. Cell Sci. 2011, 124, 3933–3940. [Google Scholar] [CrossRef]
- Meister, M.; Tikkanen, R. Endocytic trafficking of membrane-bound cargo: A flotillin point of view. Membranes (Basel) 2014, 4, 356–371. [Google Scholar] [CrossRef]
- Liu, X.X.; Liu, W.D.; Wang, L.; Zhu, B.; Shi, X.; Peng, Z.X.; Zhu, H.C.; Liu, X.D.; Zhong, M.Z.; Xie, D.; et al. Roles of flotillins in tumors. J. Zhejiang Univ. Sci. B 2018, 19, 171–182. [Google Scholar] [CrossRef]
- Llanses Martinez, M.; Rainero, E. Membrane dynamics in cell migration. Essays Biochem. 2019, 63, 469–482. [Google Scholar]
- Schulte, T.; Paschke, K.A.; Laessing, U.; Lottspeich, F.; Stuermer, C.A. Reggie-1 and reggie-2, two cell surface proteins expressed by retinal ganglion cells during axon regeneration. Development 1997, 124, 577–587. [Google Scholar]
- Bickel, P.E.; Scherer, P.E.; Schnitzer, J.E.; Oh, P.; Lisanti, M.P.; Lodish, H.F. Flotillin and epidermal surface antigen define a new family of caveolae-associated integral membrane proteins. J. Biol. Chem. 1997, 272, 13793–13802. [Google Scholar] [CrossRef]
- Volonte, D.; Galbiati, F.; Li, S.; Nishiyama, K.; Okamoto, T.; Lisanti, M.P. Flotillins/cavatellins are differentially expressed in cells and tissues and form a hetero-oligomeric complex with caveolins in vivo. Characterization and epitope-mapping of a novel flotillin-1 monoclonal antibody probe. J. Biol. Chem. 1999, 274, 12702–12709. [Google Scholar] [CrossRef]
- Neumann-Giesen, C.; Fernow, I.; Amaddii, M.; Tikkanen, R. Role of EGF-induced tyrosine phosphorylation of reggie-1/flotillin-2 in cell spreading and signaling to the actin cytoskeleton. J. Cell Sci. 2007, 120, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Amaddii, M.; Meister, M.; Banning, A.; Tomasovic, A.; Mooz, J.; Rajalingam, K.; Tikkanen, R. Flotillin-1/reggie-2 protein plays dual role in activation of receptor-tyrosine kinase/mitogen-activated protein kinase signaling. J. Biol. Chem. 2012, 287, 7265–7278. [Google Scholar] [CrossRef] [PubMed]
- Kurrle, N.; Ockenga, W.; Meister, M.; Vollner, F.; Kuhne, S.; John, B.A.; Banning, A.; Tikkanen, R. Phosphatidylinositol 3-kinase dependent upregulation of the epidermal growth factor receptor upon flotillin-1 depletion in breast cancer cells. BMC Cancer 2013, 13, 575. [Google Scholar] [CrossRef] [PubMed]
- Koh, M.; Yong, H.Y.; Kim, E.S.; Son, H.; Jeon, Y.R.; Hwang, J.S.; Kim, M.O.; Cha, Y.; Choi, W.S.; Noh, D.Y.; et al. A novel role for flotillin-1 in H-Ras-regulated breast cancer aggressiveness. Int. J. Cancer 2016, 138, 1232–1245. [Google Scholar] [CrossRef] [PubMed]
- Meister, M.; Banfer, S.; Gartner, U.; Koskimies, J.; Amaddii, M.; Jacob, R.; Tikkanen, R. Regulation of cargo transfer between ESCRT-0 and ESCRT-I complexes by flotillin-1 during endosomal sorting of ubiquitinated cargo. Oncogenesis 2017, 6, e344. [Google Scholar] [CrossRef] [PubMed]
- Fork, C.; Hitzel, J.; Nichols, B.J.; Tikkanen, R.; Brandes, R.P. Flotillin-1 facilitates toll-like receptor 3 signaling in human endothelial cells. Basic Res. Cardiol. 2014, 109, 439. [Google Scholar] [CrossRef] [PubMed]
- Langhorst, M.F.; Reuter, A.; Luxenhofer, G.; Boneberg, E.M.; Legler, D.F.; Plattner, H.; Stuermer, C.A. Preformed reggie/flotillin caps: Stable priming platforms for macrodomain assembly in T cells. Faseb J. 2006, 20, 711–713. [Google Scholar] [CrossRef]
- Slaughter, N.; Laux, I.; Tu, X.; Whitelegge, J.; Zhu, X.; Effros, R.; Bickel, P.; Nel, A. The flotillins are integral membrane proteins in lipid rafts that contain TCR-associated signaling components: Implications for T-cell activation. Clin. Immunol. 2003, 108, 138–151. [Google Scholar] [CrossRef]
- Compeer, E.B.; Kraus, F.; Ecker, M.; Redpath, G.; Amiezer, M.; Rother, N.; Nicovich, P.R.; Kapoor-Kaushik, N.; Deng, Q.; Samson, G.P.B.; et al. A mobile endocytic network connects clathrin-independent receptor endocytosis to recycling and promotes T cell activation. Nat. Commun. 2018, 9, 1597. [Google Scholar] [CrossRef]
- Redpath, G.M.I.; Ecker, M.; Kapoor-Kaushik, N.; Vartoukian, H.; Carnell, M.; Kempe, D.; Biro, M.; Ariotti, N.; Rossy, J. Flotillins promote T cell receptor sorting through a fast Rab5-Rab11 endocytic recycling axis. Nat. Commun. 2019, 10, 4392. [Google Scholar] [CrossRef]
- Stuermer, C.A.; Langhorst, M.F.; Wiechers, M.F.; Legler, D.F.; Von Hanwehr, S.H.; Guse, A.H.; Plattner, H. PrPc capping in T cells promotes its association with the lipid raft proteins reggie-1 and reggie-2 and leads to signal transduction. Faseb J. 2004, 18, 1731–1733. [Google Scholar] [CrossRef]
- Bodrikov, V.; Solis, G.P.; Stuermer, C.A. Prion protein promotes growth cone development through reggie/flotillin-dependent N-cadherin trafficking. J. Neurosci. 2011, 31, 18013–18025. [Google Scholar] [CrossRef]
- Kato, N.; Nakanishi, M.; Hirashima, N. Flotillin-1 regulates IgE receptor-mediated signaling in rat basophilic leukemia (RBL-2H3) cells. J. Immunol. 2006, 177, 147–154. [Google Scholar] [CrossRef]
- Fecchi, K.; Volonte, D.; Hezel, M.P.; Schmeck, K.; Galbiati, F. Spatial and temporal regulation of GLUT4 translocation by flotillin-1 and caveolin-3 in skeletal muscle cells. Faseb J. 2006, 20, 705–707. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, Y.; Nishii, H.; Takahashi, T.; Yamauchi, J.; Mizuno, N.; Tago, K.; Itoh, H. The lipid raft proteins flotillins/reggies interact with Gα and are involved in Gq-mediated p38 mitogen-activated protein kinase activation through tyrosine kinase. Cell. Signal. 2007, 19, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Munderloh, C.; Solis, G.P.; Bodrikov, V.; Jaeger, F.A.; Wiechers, M.; Malaga-Trillo, E.; Stuermer, C.A. Reggies/flotillins regulate retinal axon regeneration in the zebrafish optic nerve and differentiation of hippocampal and N2a neurons. J. Neurosci. 2009, 29, 6607–6615. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.; Kwon, H.; Jeong, K.; Lee, J.; Pak, Y. Essential role of flotillin-1 palmitoylation in the intracellular localization and signaling function of IGF-1 receptor. J. Cell Sci. 2015, 128, 2179–2190. [Google Scholar] [CrossRef] [PubMed]
- Banning, A.; Babuke, T.; Kurrle, N.; Meister, M.; Ruonala, M.O.; Tikkanen, R. Flotillins regulate focal adhesions by interacting with α-actinin and by influencing the activation of focal adhesion kinase. Cells 2018, 7, 28. [Google Scholar] [CrossRef]
- Neumann-Giesen, C.; Falkenbach, B.; Beicht, P.; Claasen, S.; Luers, G.; Stuermer, C.A.; Herzog, V.; Tikkanen, R. Membrane and raft association of reggie-1/flotillin-2: Role of myristoylation, palmitoylation and oligomerization and induction of filopodia by overexpression. Biochem. J. 2004, 378, 509–518. [Google Scholar] [CrossRef]
- Langhorst, M.F.; Solis, G.P.; Hannbeck, S.; Plattner, H.; Stuermer, C.A. Linking membrane microdomains to the cytoskeleton: Regulation of the lateral mobility of reggie-1/flotillin-2 by interaction with actin. FEBS Lett. 2007, 581, 4697–4703. [Google Scholar] [CrossRef]
- Ludwig, A.; Otto, G.P.; Riento, K.; Hams, E.; Fallon, P.G.; Nichols, B.J. Flotillin microdomains interact with the cortical cytoskeleton to control uropod formation and neutrophil recruitment. J. Cell Biol. 2010, 191, 771–781. [Google Scholar] [CrossRef]
- Affentranger, S.; Martinelli, S.; Hahn, J.; Rossy, J.; Niggli, V. Dynamic reorganization of flotillins in chemokine-stimulated human T-lymphocytes. BMC Cell Biol. 2011, 12, 28. [Google Scholar] [CrossRef]
- Martinelli, S.; Chen, E.J.; Clarke, F.; Lyck, R.; Affentranger, S.; Burkhardt, J.K.; Niggli, V. Ezrin/Radixin/Moesin proteins and flotillins cooperate to promote uropod formation in T cells. Front. Immunol. 2013, 4, 84. [Google Scholar] [CrossRef]
- Mathis, L.; Wernimont, S.; Affentranger, S.; Huttenlocher, A.; Niggli, V. Determinants of phosphatidylinositol-4-phosphate 5-kinase type Iγ90 uropod location in T-lymphocytes and its role in uropod formation. PeerJ. 2013, 1, e131. [Google Scholar] [CrossRef] [PubMed]
- Haglund, K.; Ivankovic-Dikic, I.; Shimokawa, N.; Kruh, G.D.; Dikic, I. Recruitment of Pyk2 and Cbl to lipid rafts mediates signals important for actin reorganization in growing neurites. J. Cell Sci. 2004, 117, 2557–2568. [Google Scholar] [CrossRef] [PubMed]
- Glebov, O.O.; Bright, N.A.; Nichols, B.J. Flotillin-1 defines a clathrin-independent endocytic pathway in mammalian cells. Nat. Cell Biol. 2006, 8, 46–54. [Google Scholar] [CrossRef]
- Fekri, F.; Abousawan, J.; Bautista, S.; Orofiamma, L.; Dayam, R.M.; Antonescu, C.N.; Karshafian, R. Targeted enhancement of flotillin-dependent endocytosis augments cellular uptake and impact of cytotoxic drugs. Sci. Rep. 2019, 9, 17768. [Google Scholar] [CrossRef] [PubMed]
- Stuermer, C.A.; Lang, D.M.; Kirsch, F.; Wiechers, M.; Deininger, S.O.; Plattner, H. Glycosylphosphatidyl inositol-anchored proteins and fyn kinase assemble in noncaveolar plasma membrane microdomains defined by reggie-1 and -2. Mol. Biol. Cell 2001, 12, 3031–3045. [Google Scholar] [CrossRef] [PubMed]
- Riento, K.; Frick, M.; Schafer, I.; Nichols, B.J. Endocytosis of flotillin-1 and flotillin-2 is regulated by Fyn kinase. J. Cell Sci. 2009, 122, 912–918. [Google Scholar] [CrossRef]
- Ait-Slimane, T.; Galmes, R.; Trugnan, G.; Maurice, M. Basolateral internalization of GPI-anchored proteins occurs via a clathrin-independent flotillin-dependent pathway in polarized hepatic cells. Mol. Biol. Cell 2009, 20, 3792–3800. [Google Scholar] [CrossRef]
- Cremona, M.L.; Matthies, H.J.; Pau, K.; Bowton, E.; Speed, N.; Lute, B.J.; Anderson, M.; Sen, N.; Robertson, S.D.; Vaughan, R.A.; et al. Flotillin-1 is essential for PKC-triggered endocytosis and membrane microdomain localization of DAT. Nat. Neurosci. 2011, 14, 469–477. [Google Scholar] [CrossRef]
- Kobayashi, J.; Hasegawa, T.; Sugeno, N.; Yoshida, S.; Akiyama, T.; Fujimori, K.; Hatakeyama, H.; Miki, Y.; Tomiyama, A.; Kawata, Y.; et al. Extracellular alpha-synuclein enters dopaminergic cells by modulating flotillin-1-assisted dopamine transporter endocytosis. Faseb J. 2019, 33, 10240–10256. [Google Scholar] [CrossRef]
- Payne, C.K.; Jones, S.A.; Chen, C.; Zhuang, X. Internalization and trafficking of cell surface proteoglycans and proteoglycan-binding ligands. Traffic 2007, 8, 389–401. [Google Scholar] [CrossRef]
- Chen, K.; Wu, Q.; Hu, K.; Yang, C.; Wu, X.; Cheung, P.; Williams, K.J. Suppression of hepatic FLOT1 (flotillin-1) by type 2 diabetes mellitus impairs the disposal of remnant lipoproteins via syndecan-1. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Rajendran, L.; Honsho, M.; Gralle, M.; Donnert, G.; Wouters, F.; Hell, S.W.; Simons, M. Flotillin-dependent clustering of the amyloid precursor protein regulates its endocytosis and amyloidogenic processing in neurons. J. Neurosci. 2008, 28, 2874–2882. [Google Scholar] [CrossRef]
- Carcea, I.; Ma’ayan, A.; Mesias, R.; Sepulveda, B.; Salton, S.R.; Benson, D.L. Flotillin-mediated endocytic events dictate cell type-specific responses to semaphorin 3A. J. Neurosci. 2010, 30, 15317–15329. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.; Leme, A.F.; Kantovitz, K.R.; de Luciane Martins, E.N.; Sallum, E.A.; Casati, M.Z.; Nociti, F.H., Jr. Leucine-rich amelogenin peptide (LRAP) uptake by cementoblast requires flotillin-1 mediated endocytosis. J. Cell. Physiol. 2017, 232, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Hulsbusch, N.; Solis, G.P.; Katanaev, V.L.; Stuermer, C.A. Reggie-1/Flotillin-2 regulates integrin trafficking and focal adhesion turnover via Rab11a. Eur. J. Cell Biol. 2015, 94, 531–545. [Google Scholar] [CrossRef]
- Solis, G.P.; Hulsbusch, N.; Radon, Y.; Katanaev, V.L.; Plattner, H.; Stuermer, C.A. Reggies/flotillins interact with Rab11a and SNX4 at the tubulovesicular recycling compartment and function in transferrin receptor and E-cadherin trafficking. Mol. Biol. Cell 2013, 24, 2689–2702. [Google Scholar] [CrossRef]
- Bodrikov, V.; Pauschert, A.; Kochlamazashvili, G.; Stuermer, C.A.O. Reggie-1 and reggie-2 (flotillins) participate in Rab11a-dependent cargo trafficking, spine synapse formation and LTP-related AMPA receptor (GluA1) surface exposure in mouse hippocampal neurons. Exp. Neurol. 2017, 289, 31–45. [Google Scholar] [CrossRef]
- Planchon, D.; Rios Morris, E.; Genest, M.; Comunale, F.; Vacher, S.; Bieche, I.; Denisov, E.V.; Tashireva, L.A.; Perelmuter, V.M.; Linder, S.; et al. MT1-MMP targeting to endolysosomes is mediated by upregulation of flotillins. J. Cell Sci. 2018, 131, 1–17. [Google Scholar] [CrossRef]
- John, B.A.; Meister, M.; Banning, A.; Tikkanen, R. Flotillins bind to the dileucine sorting motif of beta-site amyloid precursor protein-cleaving enzyme 1 and influence its endosomal sorting. FEBS J. 2014, 281, 2074–2087. [Google Scholar] [CrossRef]
- Fan, W.; Guo, J.; Gao, B.; Zhang, W.; Ling, L.; Xu, T.; Pan, C.; Li, L.; Chen, S.; Wang, H.; et al. Flotillin-mediated endocytosis and ALIX-syntenin-1-mediated exocytosis protect the cell membrane from damage caused by necroptosis. Sci. Signal. 2019, 12, 583. [Google Scholar] [CrossRef]
- Rai, A.; Pathak, D.; Thakur, S.; Singh, S.; Dubey, A.K.; Mallik, R. Dynein clusters into lipid microdomains on phagosomes to drive rapid transport toward lysosomes. Cell 2016, 164, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Saslowsky, D.E.; Cho, J.A.; Chinnapen, H.; Massol, R.H.; Chinnapen, D.J.; Wagner, J.S.; De Luca, H.E.; Kam, W.; Paw, B.H.; Lencer, W.I. Intoxication of zebrafish and mammalian cells by cholera toxin depends on the flotillin/reggie proteins but not Derlin-1 or -2. J. Clin. Invest. 2010, 120, 4399–4409. [Google Scholar] [CrossRef] [PubMed]
- Pust, S.; Dyve, A.B.; Torgersen, M.L.; van Deurs, B.; Sandvig, K. Interplay between toxin transport and flotillin localization. PLoS ONE 2010, 5, e8844. [Google Scholar] [CrossRef] [PubMed]
- Strauss, K.; Goebel, C.; Runz, H.; Mobius, W.; Weiss, S.; Feussner, I.; Simons, M.; Schneider, A. Exosome secretion ameliorates lysosomal storage of cholesterol in Niemann-Pick type C disease. J. Biol. Chem. 2010, 285, 26279–26288. [Google Scholar] [CrossRef]
- Phuyal, S.; Hessvik, N.P.; Skotland, T.; Sandvig, K.; Llorente, A. Regulation of exosome release by glycosphingolipids and flotillins. FEBS J. 2014, 281, 2214–2227. [Google Scholar] [CrossRef]
- Jang, D.; Kwon, H.; Choi, M.; Lee, J.; Pak, Y. Sumoylation of flotillin-1 promotes EMT in metastatic prostate cancer by suppressing Snail degradation. Oncogene 2019, 38, 3248–3260. [Google Scholar] [CrossRef]
- Riento, K.; Zhang, Q.; Clark, J.; Begum, F.; Stephens, E.; Wakelam, M.J.; Nichols, B.J. Flotillin proteins recruit sphingosine to membranes and maintain cellular sphingosine-1-phosphate levels. PLoS ONE 2018, 13, e0197401. [Google Scholar] [CrossRef]
- Edgar, A.J.; Polak, J.M. Flotillin-1: Gene structure: cDNA cloning from human lung and the identification of alternative polyadenylation signals. Int. J. Biochem. Cell Biol. 2001, 33, 53–64. [Google Scholar] [CrossRef]
- Rivera-Milla, E.; Stuermer, C.A.; Malaga-Trillo, E. Ancient origin of reggie (flotillin), reggie-like, and other lipid-raft proteins: Convergent evolution of the SPFH domain. Cell. Mol. Life Sci. 2006, 63, 343–357. [Google Scholar] [CrossRef]
- Morrow, I.C.; Rea, S.; Martin, S.; Prior, I.A.; Prohaska, R.; Hancock, J.F.; James, D.E.; Parton, R.G. Flotillin-1/reggie-2 traffics to surface raft domains via a novel golgi-independent pathway. Identification of a novel membrane targeting domain and a role for palmitoylation. J. Biol. Chem. 2002, 277, 48834–48841. [Google Scholar] [CrossRef]
- Roitbak, T.; Surviladze, Z.; Tikkanen, R.; Wandinger-Ness, A. A polycystin multiprotein complex constitutes a cholesterol-containing signalling microdomain in human kidney epithelia. Biochem. J 2005, 392, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Solis, G.P.; Hoegg, M.; Munderloh, C.; Schrock, Y.; Malaga-Trillo, E.; Rivera-Milla, E.; Stuermer, C.A. Reggie/flotillin proteins are organized into stable tetramers in membrane microdomains. Biochem. J. 2007, 403, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Lopez, D.; Kolter, R. Functional microdomains in bacterial membranes. Genes Dev. 2010, 24, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Fernandez, E.; Koch, G.; Wagner, R.M.; Fekete, A.; Stengel, S.T.; Schneider, J.; Mielich-Suss, B.; Geibel, S.; Markert, S.M.; Stigloher, C.; et al. Membrane microdomain disassembly inhibits MRSA antibiotic resistance. Cell 2017, 171, 1354–1367.e20. [Google Scholar] [CrossRef]
- Wagner, R.M.; Kricks, L.; Lopez, D. Functional membrane microdomains organize signaling networks in bacteria. J. Membr. Biol. 2017, 250, 367–378. [Google Scholar] [CrossRef]
- Biernatowska, A.; Augoff, K.; Podkalicka, J.; Tabaczar, S.; Gajdzik-Nowak, W.; Czogalla, A.; Sikorski, A.F. MPP1 directly interacts with flotillins in erythrocyte membrane - Possible mechanism of raft domain formation. Biochim. Biophys. Acta Biomembr. 2017, 1859, 2203–2212. [Google Scholar] [CrossRef]
- Kurrle, N.; Vollner, F.; Eming, R.; Hertl, M.; Banning, A.; Tikkanen, R. Flotillins directly interact with γ-catenin and regulate epithelial cell-cell adhesion. PLoS ONE 2013, 8, e84393. [Google Scholar] [CrossRef]
- Guillaume, E.; Comunale, F.; Do Khoa, N.; Planchon, D.; Bodin, S.; Gauthier-Rouviere, C. Flotillin microdomains stabilize cadherins at cell-cell junctions. J. Cell Sci. 2013, 126, 5293–5304. [Google Scholar] [CrossRef]
- Liu, J.; Deyoung, S.M.; Zhang, M.; Dold, L.H.; Saltiel, A.R. The stomatin/prohibitin/flotillin/HflK/C domain of flotillin-1 contains distinct sequences that direct plasma membrane localization and protein interactions in 3T3-L1 adipocytes. J. Biol. Chem. 2005, 280, 16125–16134. [Google Scholar] [CrossRef]
- Baumann, C.A.; Ribon, V.; Kanzaki, M.; Thurmond, D.C.; Mora, S.; Shigematsu, S.; Bickel, P.E.; Pessin, J.E.; Saltiel, A.R. CAP defines a second signalling pathway required for insulin-stimulated glucose transport. Nature 2000, 407, 202–207. [Google Scholar] [CrossRef]
- Limpert, A.S.; Karlo, J.C.; Landreth, G.E. Nerve growth factor stimulates the concentration of TrkA within lipid rafts and extracellular signal-regulated kinase activation through c-Cbl-associated protein. Mol. Cell. Biol. 2007, 27, 5686–5698. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Qi, W.; Wang, L.J.; Miao, H.H.; Qu, Y.X.; Li, B.L.; Song, B.L. Flotillins play an essential role in Niemann-Pick C1-like 1-mediated cholesterol uptake. Proc. Natl. Acad. Sci. USA 2011, 108, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, L.H.; Shipston, M.J. The physiology of protein S-acylation. Physiol. Rev. 2015, 95, 341–376. [Google Scholar] [CrossRef]
- Zaballa, M.E.; van der Goot, F.G. The molecular era of protein S-acylation: Spotlight on structure, mechanisms, and dynamics. Crit. Rev. Biochem. Mol. Biol. 2018, 53, 420–451. [Google Scholar] [CrossRef] [PubMed]
- Sobocinska, J.; Roszczenko-Jasinska, P.; Ciesielska, A.; Kwiatkowska, K. Protein palmitoylation and its role in bacterial and viral infections. Front. Immunol. 2017, 8, 2003. [Google Scholar] [CrossRef] [PubMed]
- Babuke, T.; Ruonala, M.; Meister, M.; Amaddii, M.; Genzler, C.; Esposito, A.; Tikkanen, R. Hetero-oligomerization of reggie-1/flotillin-2 and reggie-2/flotillin-1 is required for their endocytosis. Cell. Signal. 2009, 21, 1287–1297. [Google Scholar] [CrossRef]
- Elderdfi, M.; Sikorski, A.F. Interaction of membrane palmitoylated protein-1 with model lipid membranes. Gen. Physiol. Biophys. 2018, 37, 603–617. [Google Scholar] [CrossRef]
- Kumar, A.; Saba, J.D. Sphingosine-1-phosphate. In Encyclopedia of Signaling Molecules; Choi, S., Ed.; Springer New York: New York, NY, USA, 2012; pp. 1771–1778. [Google Scholar]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef]
- Resh, M.D. Fatty acylation of proteins: The long and the short of it. Prog. Lipid Res. 2016, 63, 120–131. [Google Scholar] [CrossRef]
- Lu, D.; Sun, H.Q.; Wang, H.; Barylko, B.; Fukata, Y.; Fukata, M.; Albanesi, J.P.; Yin, H.L. Phosphatidylinositol 4-kinase IIα is palmitoylated by Golgi-localized palmitoyltransferases in cholesterol-dependent manner. J. Biol. Chem. 2012, 287, 21856–21865. [Google Scholar] [CrossRef]
- Biernatowska, A.; Podkalicka, J.; Majkowski, M.; Hryniewicz-Jankowska, A.; Augoff, K.; Kozak, K.; Korzeniewski, J.; Sikorski, A.F. The role of MPP1/p55 and its palmitoylation in resting state raft organization in HEL cells. Biochim. Biophys. Acta 2013, 1833, 1876–1884. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Martin, B.R.; Cravatt, B.F.; Hofmann, S.L. DHHC5 protein palmitoylates flotillin-2 and is rapidly degraded on induction of neuronal differentiation in cultured cells. J. Biol. Chem. 2012, 287, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Frick, M.; Bright, N.A.; Riento, K.; Bray, A.; Merrified, C.; Nichols, B.J. Coassembly of flotillins induces formation of membrane microdomains, membrane curvature, and vesicle budding. Curr. Biol. 2007, 17, 1151–1156. [Google Scholar] [CrossRef]
- Baumann, T.; Affentranger, S.; Niggli, V. Evidence for chemokine-mediated coalescence of preformed flotillin hetero-oligomers in human T-cells. J. Biol. Chem. 2012, 287, 39664–39672. [Google Scholar] [CrossRef]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Zurzolo, C.; Simons, K. Glycosylphosphatidylinositol-anchored proteins: Membrane organization and transport. Biochim. Biophys. Acta 2016, 1858, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Kusumi, A.; Fujiwara, T.K.; Morone, N.; Yoshida, K.J.; Chadda, R.; Xie, M.; Kasai, R.S.; Suzuki, K.G. Membrane mechanisms for signal transduction: The coupling of the meso-scale raft domains to membrane-skeleton-induced compartments and dynamic protein complexes. Semin. Cell Dev. Biol. 2012, 23, 126–144. [Google Scholar] [CrossRef]
- Horejsi, V.; Hrdinka, M. Membrane microdomains in immunoreceptor signaling. FEBS Lett. 2014, 588, 2392–2397. [Google Scholar] [CrossRef]
- Plociennikowska, A.; Hromada-Judycka, A.; Borzecka, K.; Kwiatkowska, K. Co-operation of TLR4 and raft proteins in LPS-induced pro-inflammatory signaling. Cell. Mol. Life Sci. 2015, 72, 557–581. [Google Scholar] [CrossRef]
- Raghupathy, R.; Anilkumar, A.A.; Polley, A.; Singh, P.P.; Yadav, M.; Johnson, C.; Suryawanshi, S.; Saikam, V.; Sawant, S.D.; Panda, A.; et al. Transbilayer lipid interactions mediate nanoclustering of lipid-anchored proteins. Cell 2015, 161, 581–594. [Google Scholar] [CrossRef]
- Stone, M.B.; Shelby, S.A.; Nunez, M.F.; Wisser, K.; Veatch, S.L. Protein sorting by lipid phase-like domains supports emergent signaling function in B lymphocyte plasma membranes. Elife 2017, 6, e19891. [Google Scholar] [CrossRef] [PubMed]
- Lang, D.M.; Lommel, S.; Jung, M.; Ankerhold, R.; Petrausch, B.; Laessing, U.; Wiechers, M.F.; Plattner, H.; Stuermer, C.A. Identification of reggie-1 and reggie-2 as plasmamembrane-associated proteins which cocluster with activated GPI-anchored cell adhesion molecules in non-caveolar micropatches in neurons. J. Neurobiol. 1998, 37, 502–523. [Google Scholar] [CrossRef]
- Otahal, P.; Angelisova, P.; Hrdinka, M.; Brdicka, T.; Novak, P.; Drbal, K.; Horejsi, V. A new type of membrane raft-like microdomains and their possible involvement in TCR signaling. J. Immunol. 2010, 184, 3689–3696. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, D.; Ries, J.; Schwille, P.; Simons, K. Plasma membranes are poised for activation of raft phase coalescence at physiological temperature. Proc. Natl. Acad. Sci. USA 2008, 105, 10005–10010. [Google Scholar] [CrossRef] [PubMed]
- Levental, I.; Lingwood, D.; Grzybek, M.; Coskun, U.; Simons, K. Palmitoylation regulates raft affinity for the majority of integral raft proteins. Proc. Natl. Acad. Sci. USA 2010, 107, 22050–22054. [Google Scholar] [CrossRef] [PubMed]
- Levental, I.; Grzybek, M.; Simons, K. Raft domains of variable properties and compositions in plasma membrane vesicles. Proc. Natl. Acad. Sci. USA 2011, 108, 11411–11416. [Google Scholar] [CrossRef]
- Plociennikowska, A.; Hromada-Judycka, A.; Dembinska, J.; Roszczenko, P.; Ciesielska, A.; Kwiatkowska, K. Contribution of CD14 and TLR4 to changes of the PI(4,5)P2 level in LPS-stimulated cells. J. Leukoc. Biol. 2016, 100, 1363–1373. [Google Scholar] [CrossRef]
- Lorent, J.H.; Diaz-Rohrer, B.; Lin, X.; Spring, K.; Gorfe, A.A.; Levental, K.R.; Levental, I. Structural determinants and functional consequences of protein affinity for membrane rafts. Nat. Commun. 2017, 8, 1219. [Google Scholar] [CrossRef]
- Prymas, K.; Swiatkowska, A.; Traczyk, G.; Ziemlinska, E.; Dziewulska, A.; Ciesielska, A.; Kwiatkowska, K. Sphingomyelin synthase activity affects TRIF-dependent signaling of Toll-like receptor 4 in cells stimulated with lipopolysaccharide. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158549. [Google Scholar] [CrossRef]
- Rajendran, L.; Masilamani, M.; Solomon, S.; Tikkanen, R.; Stuermer, C.A.; Plattner, H.; Illges, H. Asymmetric localization of flotillins/reggies in preassembled platforms confers inherent polarity to hematopoietic cells. Proc. Natl. Acad. Sci. USA 2003, 100, 8241–8246. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, L.; Beckmann, J.; Magenau, A.; Boneberg, E.M.; Gaus, K.; Viola, A.; Giebel, B.; Illges, H. Flotillins are involved in the polarization of primitive and mature hematopoietic cells. PLoS ONE 2009, 4, e8290. [Google Scholar] [CrossRef] [PubMed]
- Gorgens, A.; Beckmann, J.; Ludwig, A.K.; Mollmann, M.; Durig, J.; Horn, P.A.; Rajendran, L.; Giebel, B. Lipid raft redistribution and morphological cell polarization are separable processes providing a basis for hematopoietic stem and progenitor cell migration. Int. J. Biochem. Cell Biol. 2012, 44, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Bodin, S.; Planchon, D.; Rios Morris, E.; Comunale, F.; Gauthier-Rouviere, C. Flotillins in intercellular adhesion - from cellular physiology to human diseases. J. Cell Sci. 2014, 127, 5139–5147. [Google Scholar] [CrossRef] [PubMed]
- Pust, S.; Klokk, T.I.; Musa, N.; Jenstad, M.; Risberg, B.; Erikstein, B.; Tcatchoff, L.; Liestol, K.; Danielsen, H.E.; van Deurs, B.; et al. Flotillins as regulators of ErbB2 levels in breast cancer. Oncogene 2013, 32, 3443–3451. [Google Scholar] [CrossRef]
- Salzer, U.; Prohaska, R. Stomatin, flotillin-1, and flotillin-2 are major integral proteins of erythrocyte lipid rafts. Blood 2001, 97, 1141–1143. [Google Scholar] [CrossRef]
- Langhorst, M.F.; Reuter, A.; Jaeger, F.A.; Wippich, F.M.; Luxenhofer, G.; Plattner, H.; Stuermer, C.A. Trafficking of the microdomain scaffolding protein reggie-1/flotillin-2. Eur. J. Cell Biol. 2008, 87, 211–226. [Google Scholar] [CrossRef]
- Sorkina, T.; Caltagarone, J.; Sorkin, A. Flotillins regulate membrane mobility of the dopamine transporter but are not required for its protein kinase C dependent endocytosis. Traffic 2013, 14, 709–724. [Google Scholar] [CrossRef]
- Bitsikas, V.; Riento, K.; Howe, J.D.; Barry, N.P.; Nichols, B.J. The role of flotillins in regulating abeta production, investigated using flotillin 1-/-, flotillin 2-/- double knockout mice. PLoS ONE 2014, 9, e85217. [Google Scholar] [CrossRef]
- Howie, J.; Reilly, L.; Fraser, N.J.; Vlachaki Walker, J.M.; Wypijewski, K.J.; Ashford, M.L.; Calaghan, S.C.; McClafferty, H.; Tian, L.; Shipston, M.J.; et al. Substrate recognition by the cell surface palmitoyl transferase DHHC5. Proc. Natl. Acad. Sci. USA 2014, 111, 17534–17539. [Google Scholar] [CrossRef]
- He, M.; Abdi, K.M.; Bennett, V. Ankyrin-G palmitoylation and βII-spectrin binding to phosphoinositide lipids drive lateral membrane assembly. J. Cell Biol. 2014, 206, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hao, J.W.; Wang, X.; Guo, H.; Sun, H.H.; Lai, X.Y.; Liu, L.Y.; Zhu, M.; Wang, H.Y.; Li, Y.F.; et al. DHHC4 and DHHC5 facilitate fatty acid uptake by palmitoylating and targeting CD36 to the plasma membrane. Cell Rep. 2019, 26, 209–221.e5. [Google Scholar] [CrossRef] [PubMed]
- Hilgemann, D.W.; Lin, M.J.; Fine, M.; Deisl, C. On the existence of endocytosis driven by membrane phase separations. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183007. [Google Scholar] [CrossRef] [PubMed]
- Breusegem, S.Y.; Seaman, M.N.J. Genome-wide RNAi screen reveals a role for multipass membrane proteins in endosome-to-golgi retrieval. Cell Rep. 2014, 9, 1931–1945. [Google Scholar] [CrossRef]
- Sergeeva, O.A.; van der Goot, F.G. Anthrax toxin requires ZDHHC5-mediated palmitoylation of its surface-processing host enzymes. Proc. Natl. Acad. Sci. USA 2019, 116, 1279–1288. [Google Scholar] [CrossRef]
- Brigidi, G.S.; Santyr, B.; Shimell, J.; Jovellar, B.; Bamji, S.X. Activity-regulated trafficking of the palmitoyl-acyl transferase DHHC5. Nat. Commun. 2015, 6, 8200. [Google Scholar] [CrossRef]
- Swanwick, C.C.; Shapiro, M.E.; Vicini, S.; Wenthold, R.J. Flotillin-1 promotes formation of glutamatergic synapses in hippocampal neurons. Dev. Neurobiol. 2010, 70, 875–883. [Google Scholar] [CrossRef]
- Raemaekers, T.; Peric, A.; Baatsen, P.; Sannerud, R.; Declerck, I.; Baert, V.; Michiels, C.; Annaert, W. ARF6-mediated endosomal transport of Telencephalin affects dendritic filopodia-to-spine maturation. Embo J. 2012, 31, 3252–3269. [Google Scholar] [CrossRef]
- Kaiser, H.J.; Lingwood, D.; Levental, I.; Sampaio, J.L.; Kalvodova, L.; Rajendran, L.; Simons, K. Order of lipid phases in model and plasma membranes. Proc. Natl. Acad. Sci. USA 2009, 106, 16645–166650. [Google Scholar] [CrossRef]
- Podkalicka, J.; Biernatowska, A.; Majkowski, M.; Grzybek, M.; Sikorski, A.F. MPP1 as a factor regulating phase separation in giant plasma membrane-derived vesicles. Biophys. J. 2015, 108, 2201–2211. [Google Scholar] [CrossRef]
- Buzas, E.I.; Gyorgy, B.; Nagy, G.; Falus, A.; Gay, S. Emerging role of extracellular vesicles in inflammatory diseases. Nat. Rev. Rheumatol. 2014, 10, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Thery, C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc. Natl. Acad. Sci. USA 2016, 113, E968–E977. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Ikezu, T. Emerging roles of extracellular vesicles in neurodegenerative disorders. Neurobiol. Dis. 2019, 130, 104512. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Skotland, T.; Berge, V.; Sandvig, K.; Llorente, A. Exosomal proteins as prostate cancer biomarkers in urine: From mass spectrometry discovery to immunoassay-based validation. Eur. J. Pharm. Sci. 2017, 98, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, M.; Kimura, N.; Akatsu, H.; Hashizume, Y.; Ferdous, T.; Tachita, T.; Iida, S.; Zou, K.; Matsubara, E.; Michikawa, M. Flotillin is a novel diagnostic blood marker of Alzheimer’s disease. J. Alzheimers Dis. 2019, 72, 1165–1176. [Google Scholar] [CrossRef]
- Hoehne, M.; de Couet, H.G.; Stuermer, C.A.; Fischbach, K.F. Loss- and gain-of-function analysis of the lipid raft proteins Reggie/Flotillin in Drosophila: They are posttranslationally regulated, and misexpression interferes with wing and eye development. Mol. Cell. Neurosci. 2005, 30, 326–338. [Google Scholar] [CrossRef]
- Berger, T.; Ueda, T.; Arpaia, E.; Chio, I.I.; Shirdel, E.A.; Jurisica, I.; Hamada, K.; You-Ten, A.; Haight, J.; Wakeham, A.; et al. Flotillin-2 deficiency leads to reduced lung metastases in a mouse breast cancer model. Oncogene 2013, 32, 4989–4994. [Google Scholar] [CrossRef]
- Sobocinska, J.; Roszczenko-Jasinska, P.; Zareba-Koziol, M.; Hromada-Judycka, A.; Matveichuk, O.V.; Traczyk, G.; Lukasiuk, K.; Kwiatkowska, K. Lipopolysaccharide upregulates palmitoylated enzymes of the hhosphatidylinositol cycle: An insight from proteomic studies. Mol. Cell. Proteom. 2018, 17, 233–254. [Google Scholar] [CrossRef]
- Thalwieser, Z.; Kiraly, N.; Fonodi, M.; Csortos, C.; Boratko, A. Protein phosphatase 2A-mediated flotillin-1 dephosphorylation up-regulates endothelial cell migration and angiogenesis regulation. J. Biol. Chem. 2019, 294, 20196–20206. [Google Scholar] [CrossRef]
- Zhou, H.; Di Palma, S.; Preisinger, C.; Peng, M.; Polat, A.N.; Heck, A.J.; Mohammed, S. Toward a comprehensive characterization of a human cancer cell phosphoproteome. J. Proteome Res. 2013, 12, 260–271. [Google Scholar] [CrossRef]
- Bian, Y.; Song, C.; Cheng, K.; Dong, M.; Wang, F.; Huang, J.; Sun, D.; Wang, L.; Ye, M.; Zou, H. An enzyme assisted RP-RPLC approach for in-depth analysis of human liver phosphoproteome. J. Proteomics 2014, 96, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Giordano, F.; Wu, Y.; Chan, J.; Zhu, C.; Milosevic, I.; Wu, X.; Yao, K.; Chen, B.; Baumgart, T.; et al. Coupling between endocytosis and sphingosine kinase 1 recruitment. Nat. Cell Biol. 2014, 16, 652–662. [Google Scholar] [CrossRef] [PubMed]
- Lima, S.; Milstien, S.; Spiegel, S. Sphingosine and Sphingosine Kinase 1 Involvement in Endocytic Membrane Trafficking. J. Biol. Chem. 2017, 292, 3074–3088. [Google Scholar] [CrossRef] [PubMed]
- Botelho, R.J.; Grinstein, S. Phagocytosis. Curr. Biol. 2011, 21, R533–R538. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, T.; Takahashi, T.; Nakamori, M.; Yamazaki, Y.; Kurashige, T.; Nagano, Y.; Nishida, Y.; Izumi, Y.; Matsumoto, M. Phosphatidylinositol-4,5-bisphosphate is enriched in granulovacuolar degeneration bodies and neurofibrillary tangles. Neuropathol. Appl. Neurobiol. 2014, 40, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, W.; Ren, C.; Wen, Q.; Liu, W.; Yang, X.; Wang, L.; Zhu, B.; Zeng, L.; Feng, X.; et al. Flotillin-2 promotes metastasis of nasopharyngeal carcinoma by activating NF-κB and PI3K/Akt3 signaling pathways. Sci. Rep. 2015, 5, 11614. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Li, J.; Chen, J.; Tang, X.; Wu, S.; Liao, C. Knockdown of flotillin-2 impairs the proliferation of breast cancer cells through modulation of Akt/FOXO signaling. Oncol. Rep. 2015, 33, 2285–2290. [Google Scholar] [CrossRef]
- Kwiatkowska, K. One lipid, multiple functions: How various pools of PI(4,5)P2 are created in the plasma membrane. Cell. Mol. Life Sci. 2010, 67, 3927–3946. [Google Scholar] [CrossRef]
| SIGNALING | ||
| Receptor | Affected events | Ref. |
| EGF receptor | signaling leading to cell adhesion | [8] |
| EGF receptor clustering and phosphorylation, ERK1/2 and Akt phosphorylation; flotillins as MAP kinase scaffolding proteins | [9] | |
| EGF receptor expression in breast cancer cells | [10] | |
| activation of H-Ras in breast cancer cells | [11] | |
| EGF receptor sorting and lysosomal degradation | [12] | |
| TLR3 | ligand internalization | [13] |
| TCR | raft association and recycling | [14,15,16,17] |
| PrP | [Ca+2] increase, Fyn and ERK1/2 activation, N-cadherin trafficking | [18,19] |
| IgE receptor | [Ca+2] increase, phosphorylation of IgE receptor γ chain and ERK1/2 | [20] |
| insulin receptor | insulin-induced glucose uptake via Glut4 transporter | [21] |
| Gq protein- coupled receptors | p38 phosphorylation | [22] |
| not determined | signaling leading to axon regeneration: Rho GTPase activation, formation of N-WASP-Arp3/cortactin complexes, p38, ERK1/2 and FAK kinase phosphorylation | [23] |
| IGF-1 receptor* | IGF-1 receptor transport from endoplasmic reticulum to plasma membrane | [24] |
| integrins | ERK2 and FAK kinase phosphorylation | [25] |
| ACTIN CYTOSKELETON REMODELING | ||
| Affected processes | ||
| filopodia formation | [26] | |
| cell spreading | [8] | |
| proper localization of Vav during T cell spreading | [14] | |
| F-actin binding | [27] | |
| axon regeneration | [23] | |
| uropod formation in neutrophils | [28] | |
| uropod formation in T cells, activation of ezrin/radix/moesin, localization of PIP5KIγ to uropod | [29,30,31] | |
| formation of lamellipodia at the growth cone of neurons | [32] | |
| cell adhesion and migration via α-actinin binding | [25] | |
| ENDOCYTOSIS | ||
| Cargo | ||
| fluid phase (magnetic dextran, AF488-dextran) | [33,34] | |
| GPI-anchored proteins | [18,33,35,36,37] | |
| DAT | [38,39] | |
| proteoglycans, e.g., syndecan-1 | [40,41] | |
| APP | [42] | |
| semaphorin 3A receptor | [43] | |
| leucine-rich amelogenin peptide | [44] | |
| PROTEIN TRAFFICKING | ||
| Protein/receptor | ||
| recycling | ||
| DAT | [39] | |
| TCR | [16,17] | |
| α-5 and β-1 integrin | [45] | |
| E-cadherin | [46] | |
| AMPA receptor (GluA1, GluN1 subunits) | [47] | |
| MT1-MMP** | [48] | |
| trafficking to lysosomes | ||
| EGF receptor | [12] | |
| BACE1 | [49] | |
| pseudokinase MLKL | [50] | |
| proteoglycans and bound ligands, including VLDL | [40,41] | |
| late phagosomes | [51] | |
| from endoplasmic reticulum to plasma membrane | ||
| IGF-1 receptor* | [24] | |
| caveolin-1* | [13] | |
| retrograde transport | ||
| cholera toxin | [52] | |
| Shiga toxin | [53] | |
| RELEASE OF EXTRACELLULAR VESICLES | ||
| Affected processes | ||
| regulation of cholesterol content in exosomes | [54] | |
| regulation of caveolin-1 and annexin-2 content in exosomes*** | [55] | |
| GENE EXPRESSION | ||
| Gene | ||
| encoding proteins of ETM transition* | [56] | |
| encoding interferon-stimulated protein 5* | [57] |
| Protein | Method Used for Identification of the Binding to Flotillin-1 and/or Flotillin-2 | Ref. |
|---|---|---|
| F-actin | flotillin-2 (SPFH domain), direct binding, in vitro binding assay | [27] |
| MPP1 | flotilllin-1 and -2, direct binding, overlay assay | [66] |
| Lyn | flotillin-1, direct binding, mammalian two-hybrid system | [20] |
| flotillin-1, co-immunoprecipitation | ||
| Gαq | flotillin-1 and -2, direct binding, pull-down assay; flotillin-1, direct binding, yeast two-hybrid system | [22] |
| flotillin-1 and -2 (38 and 43 N-terminal a. a.), co-immunoprecipitation | ||
| SNX4 | flotillin-2 (SPFH domain), direct binding, in vitro pull-down assay | [46] |
| flotillin-2, co-immunoprecipitation | ||
| Rab11 | flotillin-2 (SPFH domain), direct binding, in vitro pull-down assay | [46] |
| flotillin-2, co-immunoprecipitation | ||
| Hrs | flotillin-1, direct binding, in vitro pull-down assay | [12] |
| flotillin-1 and -2, pull-down from cell lysates, co-immunoprecipitation | ||
| Tsg101 | flotillin-1 and -2, pull-down from cell lysates | [12] |
| BACE1 | flotillin-1, direct binding, in vitro pull-down assay | [49] |
| flotillin-1 and -2, pull-down from cell lysates; flotillin-2, co-immunoprecipitation | ||
| EGF receptor | flotillin-1 and -2, co-immunoprecipitation | [9] |
| cRAF, MEK1, ERK2, cRAF, MEK1/2, ERK1/2, KSR1 | flotillin-1, direct binding, in vitro pull-down assay | [9] |
| flotillin-1, pull-down from cell lysates | [9] | |
| γ-catenin | flotillin-1 and -2, direct binding, in vitro binding assay | [67] |
| flotillin-1and -2, pull-down from cell lysates, co-immunoprecipitation | ||
| polycystin-1, β-catenin, E-cadherin | flotillin-2, co-immunoprecipitation | [61] |
| N-cadherin, E-cadherin | flotillin-1 and -2, co-immunoprecipitation | [68] |
| Exo70, Fyn, ERK1/2, N-cadherin | flotillin-2, co-immunoprecipitation | [19] |
| CAP, ArgBP2, ArgBP2 | flotillin-1, pull-down from cell lysates | [32] |
| CAP, Cbl | flotillin-1 (and not specified), pull-down from cell lysates, co-immunoprecipitation | [69,70,71] |
| PrP, Thy-1, Fyn, Lck | flotillin-1 and -2, co-immunoprecipitation | [18] |
| NPC1L1 | flotillin- 1 and -2, co-immunoprecipitation | [72] |
| α-actinin | flotillin-1 and -2, pull-down from cell lysates, co-immunoprecipitation | [25] |
| Vav | flotillin-2, co-immunoprecipitation | [14] |
| LRAP | flotillin-1, pull-down from cell lysates, co-immunoprecipitation | [44] |
| syndecan-1 | flotillin-1 (10-36 a.a), co-immunoprecipitation | [41] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwiatkowska, K.; Matveichuk, O.V.; Fronk, J.; Ciesielska, A. Flotillins: At the Intersection of Protein S-Palmitoylation and Lipid-Mediated Signaling. Int. J. Mol. Sci. 2020, 21, 2283. https://doi.org/10.3390/ijms21072283
Kwiatkowska K, Matveichuk OV, Fronk J, Ciesielska A. Flotillins: At the Intersection of Protein S-Palmitoylation and Lipid-Mediated Signaling. International Journal of Molecular Sciences. 2020; 21(7):2283. https://doi.org/10.3390/ijms21072283
Chicago/Turabian StyleKwiatkowska, Katarzyna, Orest V. Matveichuk, Jan Fronk, and Anna Ciesielska. 2020. "Flotillins: At the Intersection of Protein S-Palmitoylation and Lipid-Mediated Signaling" International Journal of Molecular Sciences 21, no. 7: 2283. https://doi.org/10.3390/ijms21072283
APA StyleKwiatkowska, K., Matveichuk, O. V., Fronk, J., & Ciesielska, A. (2020). Flotillins: At the Intersection of Protein S-Palmitoylation and Lipid-Mediated Signaling. International Journal of Molecular Sciences, 21(7), 2283. https://doi.org/10.3390/ijms21072283





