MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs
Abstract
1. Introduction
2. Overview of MiRNA Biogenesis and Function
3. MiRNA Biogenesis at the Embryo-Maternal Interface
4. MiRNAs at the Embryo—Maternal Interface
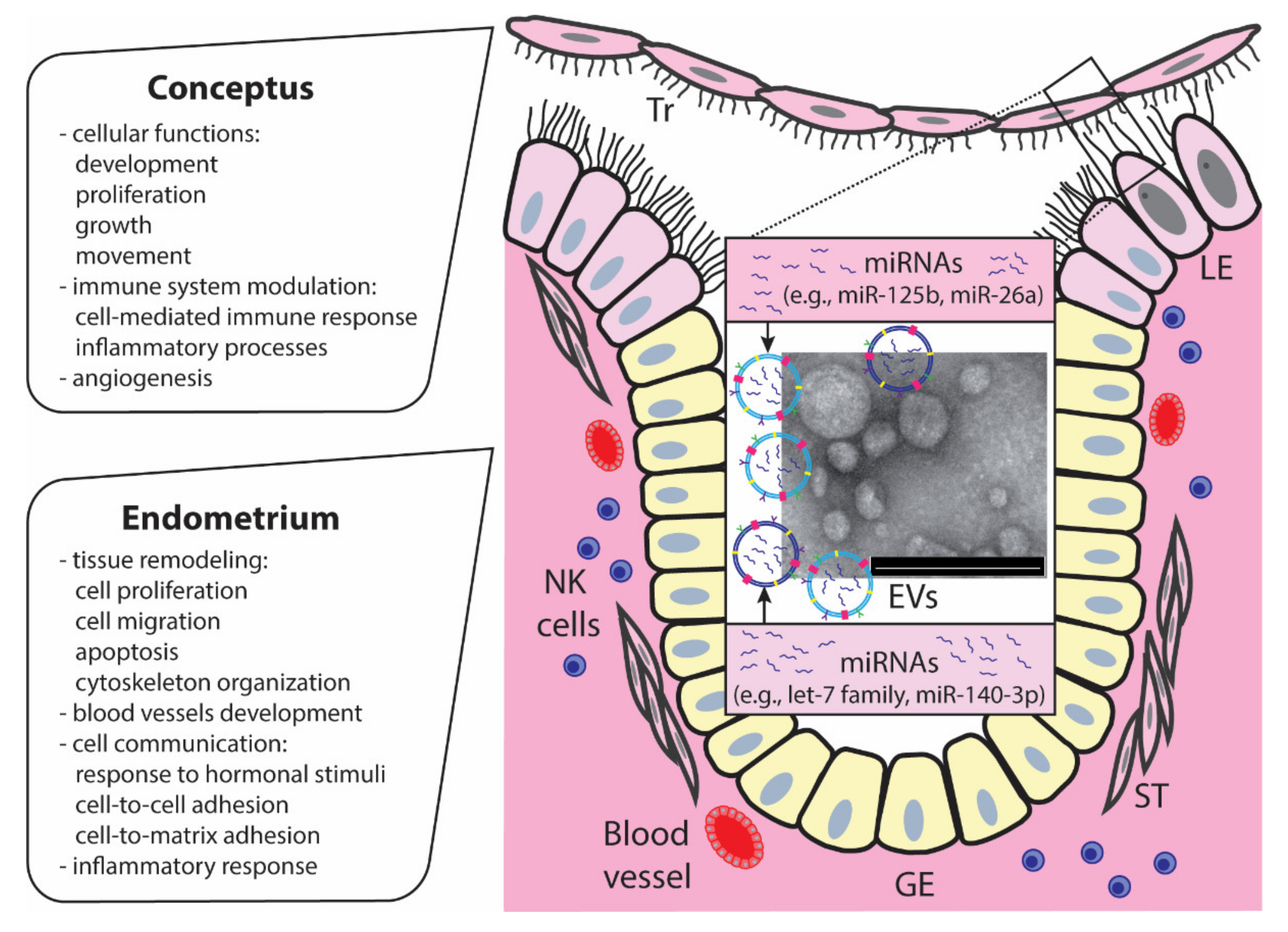
5. MiRNAs Carried by EVs at the Embryo–Maternal Interface
6. MiRNAs in Pregnant Corpus Luteum
7. Circulating MiRNAs during Early Pregnancy
8. Concluding Remarks and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 3′ UTR | Three prime untranslated region |
| 5′ UTR | Five prime untranslated region; |
| ACVR2B | Activin receptor type-2B |
| AGO1-4 | Argonaute RISC components 1-4 |
| AKR1C1 | Aldo-keto reductase family 1 member C1 |
| CDC42 | Cell division cycle 42 |
| CL | Corpus luteum |
| CREB | Cyclic AMP-responsive element-binding protein |
| DGCR8 | DiGeorge syndrome critical region gene 8 |
| DICER1 | Dicer 1, ribonuclease III |
| DROSHA | Drosha, ribonuclease III |
| E2 | Estradiol-17beta |
| EDN1 | Endothelin 1 |
| ESCRT | Endosomal sorting complex required for transport |
| ESR1 | Estrogen receptor 1 |
| ESR2 | Estrogen receptor 2 |
| EVs | Extracellular vesicles |
| FGF | Fibroblast growth factor |
| FGFR2 | Fibroblast growth factor receptor 2 |
| FOS | Fos proto-oncogene |
| IFNG | Interferon gamma |
| JUN | Jun proto-oncogene |
| KCNMA1 | Potassium calcium-activated channel subfamily M Alpha 1 |
| LIF | Leukemia inhibitory factor |
| LIFR | Leukemia inhibitory factor receptor |
| miRNA | MicroRNA |
| NR4A1 | Nuclear receptor subfamily 4 group A member 1 |
| OXTR | Oxytocin receptor |
| P4 | Progesterone |
| PGE2 | Prostaglandin E2 |
| PGR | Progesterone receptor |
| PTGER2 | Prostaglandin E receptor 2 |
| PTGS2 | Prostaglandin-endoperoxide synthase |
| RAB | RAS-related protein GTPases |
| RISC | RNA-Induced Silencing Complex |
| SIRT1 | Sirtuin 1 |
| SNP | Single nucleotide polymorphism |
| TARBP 2 | HIV-1 transactivation response (TAR), RNA- binding protein 2 |
| TGF-beta | Transforming growth factor beta |
| TGFBR1 | Transforming growth factor beta receptor 1 |
| TNRC6A | Trinucleotide repeat containing 6A |
| VEGFR1 | Vascular endothelial growth factor A receptor 1 |
| XPO5 | Exportin-5 |
References
- Ziecik, A.J.; Przygrodzka, E.; Jalali, B.M.; Kaczmarek, M.M. Regulation of the porcine corpus luteum during pregnancy. Reproduction 2018, 156, R57–R67. [Google Scholar] [CrossRef] [PubMed]
- Blitek, A.; Kaczmarek, M.M.; Waclawik, A.; Ziecik, A.J. Embryo-maternal relationships during the peri-implantation period- new and old players. In Control of Pig Reproduction IX.; Rodriguez-Martinez, H., Soede, N.M., Flowers, W.L., Eds.; Nottingham University Press: Nottingham, UK, 2013; pp. 61–70. [Google Scholar]
- Waclawik, A.; Kaczmarek, M.M.; Blitek, A.; Kaczynski, P.; Ziecik, A.J. Embryo-maternal dialogue during pregnancy establishment and implantation in the pig. Mol. Reprod. Dev. 2017, 84, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Kim, V.N. Processing of intronic microRNAs. EMBO J. 2007, 26, 775–783. [Google Scholar] [CrossRef] [PubMed]
- De Rie, D.; Abugessaisa, I.; Alam, T.; Arner, E.; Arner, P.; Ashoor, H.; Åström, G.; Babina, M.; Bertin, N.; Burroughs, A.M.; et al. An integrated expression atlas of miRNAs and their promoters in human and mouse. Nat. Biotechnol. 2017, 35, 872–878. [Google Scholar] [CrossRef]
- Lai, X.; Vera, J. MicroRNA Clusters. In Encyclopedia of Systems Biology; Dubitzky, W., Wolkenhauer, O., Cho, K.H., Yokota, H., Eds.; Springer: New York, NY, USA, 2013; pp. 77–141. [Google Scholar]
- Khvorova, A.; Reynolds, A.; Jayasena, S.D. Functional siRNAs and miRNAs exhibit strand bias. Cell 2003, 115, 209–216. [Google Scholar] [CrossRef]
- Schwarz, D.S.; Hutvagner, G.; Du, T.; Xu, Z.; Aronin, N.; Zamore, P.D. Asymmetry in the assembly of the RNAi enzyme complex. Cell 2003, 115, 199–208. [Google Scholar] [CrossRef]
- Pillai, R.S. MicroRNA function: Multiple mechanisms for a tiny RNA? RNA 2005, 11, 1753–1761. [Google Scholar] [CrossRef]
- Valencia-Sanchez, M.A.; Liu, J.; Hannon, G.J.; Parker, R. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes. Dev. 2006, 20, 515–524. [Google Scholar] [CrossRef]
- Kim, Y.K.; Kim, B.; Kim, V.N. Re-evaluation of the roles of DROSHA, Export in 5, and DICER in microRNA biogenesis. Proc. Natl. Acad. Sci. USA 2016, 113, E1881–E1889. [Google Scholar] [CrossRef]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell. Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef]
- Broughton, J.P.; Lovci, M.T.; Huang, J.L.; Yeo, G.W.; Pasquinelli, A.E. Pairing beyond the seed supports microRNA targeting specificity. Mol. Cell. 2016, 64, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Sood, P.; Krek, A.; Zavolan, M.; Macino, G.; Rajewsky, N. Cell-type-specific signatures of microRNAs on target mRNA expression. Proc. Natl. Acad. Sci. USA 2006, 103, 2746–2751. [Google Scholar] [CrossRef] [PubMed]
- Morita, S.; Horii, T.; Kimura, M.; Goto, Y.; Ochiya, T.; Hatada, I. One Argonaute family member, AGO2 (Ago2), is essential for development and appears not to be involved in DNA methylation. Genomics 2007, 89, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Luense, L.J.; McGinnis, L.K.; Nothnick, W.B.; Christenson, L.K. Dicer1 is essential for female fertility and normal development of the female reproductive system. Endocrinol. 2008, 149, 6207–6712. [Google Scholar] [CrossRef]
- Stowe, H.M.; Curry, E.; Calcatera, S.M.; Krisher, R.L.; Paczkowski, M.; Pratt, S.L. Cloning and expression of porcine Dicer and the impact of developmental stage and culture conditions on MicroRNA expression in porcine embryos. Gene 2012, 501, 198–205. [Google Scholar] [CrossRef]
- Burrola-Barraza, M.E.; Hernández-Seáñez, R.; Barceló-Fimbres, M.; Rodríguez-Almeida, F.A.; González-Rodríguez, E.; García-Quiñónez, S.; Grado-Ahuir, J.A.; Moreno-Brito, V. Dicer gene expression during early bovine embryo development. Mol. Reprod. Dev. 2011, 78, 622. [Google Scholar] [CrossRef]
- Bernstein, E.; Kim, S.Y.; Carmell, M.A.; Murchison, E.P.; Alcorn, H.; Li, M.Z.; Mills, A.A.; Elledge, S.J.; Anderson, K.V.; Hannon, G.J. Dicer is essential for mouse development. Nat. Genet. 2003, 35, 215–217. [Google Scholar] [CrossRef]
- Gonzalez, G.; Behringer, R.R. Dicer is required for female reproductive tract development and fertility in the mouse. Mol. Reprod. Dev. 2009, 76, 678–688. [Google Scholar] [CrossRef]
- Nothnick, W.B.; Healy, C.; Hong, X. Steroidal regulation of uterine miRNAs is associated with modulation of the miRNA biogenesis components Exportin-5 and Dicer1. Endocrine 2010, 37, 265–273. [Google Scholar] [CrossRef]
- Krawczynski, K.; Najmula, J.; Bauersachs, S.; Kaczmarek, M.M. MicroRNAome of porcine conceptuses and trophoblasts: Expression profile of microRNAs and their potential to regulate genes crucial for establishment of pregnancy. Biol. Reprod. 2015, 92, 21. [Google Scholar] [CrossRef]
- Krawczynski, K.; Bauersachs, S.; Reliszko, Z.P.; Graf, A.; Kaczmarek, M.M. Expression of microRNAs and isomiRs in the porcine endometrium: Implications for gene regulation at the maternal-conceptus interface. BMC Genom. 2015, 16, 906. [Google Scholar] [CrossRef] [PubMed]
- Geisert, R.D.; Renegar, R.H.; Thatcher, W.W.; Roberts, R.M.; Bazer, F.W. Establishment of pregnancy in the pig: I. Interrelationships between preimplantation development of the pig blastocyst and uterine endometrial secretions. Biol. Reprod. 1982, 27, 925–939. [Google Scholar] [CrossRef] [PubMed]
- Geisert, R.D.; Brookbank, J.W.; Roberts, R.M.; Bazer, F.W. Establishment of pregnancy in the pig: II. Cellular remodeling of the porcine blastocyst during elongation on day 12 of pregnancy. Biol. Reprod. 1982, 27, 941–955. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, V. Electron microscopy of the initial stages of placentation in the pig. Anat. Embryol. 1985, 172, 281–293. [Google Scholar] [CrossRef]
- Ross, J.W.; Ashworth, M.D.; Stein, D.R.; Couture, O.P.; Tuggle, C.K.; Geisert, R.D. Identification of differential gene expression during porcine conceptus rapid trophoblastic elongation and attachment to uterine luminal epithelium. Physiol. Genom. 2009, 36, 140–148. [Google Scholar] [CrossRef]
- Østrup, E.; Bauersachs, S.; Blum, H.; Wolf, E.; Hyttel, P. Differential endometrial gene expression in pregnant and nonpregnant sows. Biol. Reprod. 2010, 83, 277–285. [Google Scholar] [CrossRef]
- Franczak, A.; Wojciechowicz, B.; Kotwica, G. Transcriptomic analysis of the porcine endometrium during early pregnancy and the estrous cycle. Reprod. Biol. 2013, 13, 229–237. [Google Scholar] [CrossRef]
- Kiewisz, J.; Krawczynski, K.; Lisowski, P.; Blitek, A.; Zwierzchowski, L.; Ziecik, A.J.; Kaczmarek, M.M. Global gene expression profiling of porcine endometria on Days 12 and 16 of the estrous cycle and pregnancy. Theriogenology 2014, 82, 897–909. [Google Scholar] [CrossRef]
- Yang, C.-X.; Du, Z.-Q.; Wright, E.C.; Rothschild, M.F.; Prather, R.S.; Ross, J.W. Small RNA profile of the cumulus-oocyte complex and early embryos in the pig. Biol. Reprod. 2012, 87, 117. [Google Scholar] [CrossRef]
- Bidarimath, M.; Edwards, A.K.; Wessels, J.M.; Khalaj, K.; Kridli, R.T.; Tayade, C. Distinct microRNA expression in endometrial lymphocytes, endometrium, and trophoblast during spontaneous porcine fetal loss. J. Reprod. Immunol. 2015, 107, 64–79. [Google Scholar] [CrossRef]
- Wessels, J.M.; Edwards, A.K.; Khalaj, K.; Kridli, R.T.; Bidarimath, M.; Tayade, C. The microRNAome of pregnancy: Deciphering miRNA networks at the maternal-fetal interface. PLoS ONE 2013, 8, e72264. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.C. MicroRNA expression and function during porcine oocyte maturation and early embryonic development. PhD Thesis, Iowa State University, Ames, IA, USA, 2012. Available online: http://lib.dr.iastate.edu/cgi/viewcontent.cgi?article=3529&context=etd (accessed on 31 October 2019).
- Su, L.; Zhao, S.; Zhu, M.; Yu, M. Differential expression of microRNAs in porcine placentas on Days 30 and 90 of gestation. Reprod. Fertil. Dev. 2010, 22, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wang, M.; Su, L.; Li, X.; Zhao, S.; Yu, M. The expression pattern of microRNAs and the associated pathways Involved in the development of porcine placental folds that contribute to the expansion of the exchange surface area. Biol. Reprod. 2015, 93, 62. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Liu, R.; Cheng, W.; Zhu, M.; Li, X.; Zhao, S.; Yu, M. Expression patterns of microRNAs in porcine endometrium and their potential roles in embryo implantation and placentation. PLoS ONE 2014, 9, e87867. [Google Scholar] [CrossRef]
- Bick, J.T.; Flöter, V.L.; Robinson, M.D.; Bauersachs, S.; Ulbrich, S.E. Small RNA-seq analysis of single porcine blastocysts revealed that maternal estradiol-17beta exposure does not affect miRNA isoform (isomiR) expression. BMC Genomics 2018, 19, 590. [Google Scholar] [CrossRef]
- Bidarimath, M.; Khalaj, K.; Kridli, R.T.; Kan, F.W.; Koti, M.; Tayade, C. Extracellular vesicle mediated intercellular communication at the porcine maternal-fetal interface: A new paradigm for conceptus-endometrial cross-talk. Sci. Rep. 2017, 7, 40476. [Google Scholar] [CrossRef]
- Przygrodzka, E.; Sokołowska, G.; Myszczynski, K.; Krawczynski, K.; Kaczmarek, M.M. Clustered microRNAs: The molecular mechanism supporting the maintenance of luteal function during early pregnancy. FASEB J. 2020. [Google Scholar] [CrossRef]
- Reliszko, Z.P.; Gajewski, Z.; Kaczmarek, M.M. Signs of embryo-maternal communication: miRNAs in the maternal serum of pregnant pigs. Reproduction 2017, 154, 217–228. [Google Scholar] [CrossRef]
- Reliszko, Z.P.; Myszczynski, K.; Kaczmarek, M.M. Differential expression of circulating miRNAs in maternal serum during early pregnancy in the pig. In Proceedings of the 10th International Conference on Pig Reproduction, Columbia, MO, USA, 11–14 June 2017. [Google Scholar]
- Cloonan, N.; Wani, S.; Xu, Q.; Gu, J.; Lea, K.; Heater, S.; Barbacioru, C.; Steptoe, A.L.; Martin, H.C.; Nourbakhsh, E.; et al. MicroRNAs and their isomiRs function cooperatively to target common biological pathways. Genome Biol. 2011, 12, R126. [Google Scholar] [CrossRef]
- Geisert, R.D.; Thatcher, W.W.; Roberts, R.M.; Bazer, F.W. Establishment of Pregnancy in the Pig: III. Endometrial secretory response to estradiol valerate administered on Day 11 of the estrous cycle. Biol. Reprod. 1982, 27, 957–965. [Google Scholar] [CrossRef]
- Geisert, R.D.; Morgan, G.L.; Zavy, M.T.; Blair, R.M.; Gries, L.K.; Cox, A.; Yellin, T. Effect of asynchronous transfer and oestrogen administration on survival and development of porcine embryos. J. Reprod. Fertil. 1991, 93, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Pope, W.F.; Lawyer, M.S.; Butler, W.R.; Foote, R.H.; First, N.L. Dose-response shift in the ability of gilts to remain pregnant following exogenous estradiol-17 beta exposure. J. Anim. Sci. 1986, 63, 1208–1210. [Google Scholar] [CrossRef] [PubMed]
- Flöter, V.L.; Lorenz, A.K.; Kirchner, B.; Pfaffl, M.W.; Bauersachs, S.; Ulbrich, S.E. Impact of preimplantational oral low-dose estradiol-17β exposure on the endometrium: The role of miRNA. Mol. Reprod. Dev. 2018, 85, 417–426. [Google Scholar] [CrossRef]
- Li, W.; Xi, Y.; Xue, S.; Wang, Y.; Wu, L.; Liu, H.; Lei, M. Sequence analysis of microRNAs during pre-implantation between Meishan and Yorkshire pigs. Gene 2018, 646, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Ge, Q.; Zhu, Y.; Li, H.; Tian, F.; Xie, X.; Bai, Y. Differential expression of circulating miRNAs in maternal plasma in pregnancies with fetal macrosomia. Int. J. Mol. Med. 2015, 35, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.M.; Han, T.; Sargent, I.L.; Yin, G.W.; Yao, Y.Q. Differential expression profile of microRNAs in human placentas from preeclamptic pregnancies vs normal pregnancies. Am. J. Obstet. Gynecol. 2009, 200, 661.e1–661.e7. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Deng, D.; Liu, X.; Xiao, Y.; Huang, J.; Wang, F.; Li, X.; Yu, M. A miR-18a binding-site polymorphism in CDC42 3′UTR affects CDC42 mRNA expression in placentas and is associated with litter size in pigs. Mamm. Genome 2019, 30, 34–41. [Google Scholar] [CrossRef]
- Lei, B.; Gao, S.; Luo, L.F.; Xia, X.Y.; Jiang, S.W.; Deng, C.Y.; Xiong, Y.Z.; Li, F.E. A SNP in the miR-27a gene is associated with litter size in pigs. Mol. Biol. Rep. 2011, 38, 3725–3729. [Google Scholar] [CrossRef]
- Bischof, R.J.; Brandon, M.R.; Lee, C.S. Cellular immune responses in the pig uterus during pregnancy. J. Reprod. Immunol. 1995, 29, 161–178. [Google Scholar] [CrossRef]
- Keys, J.L.; King, G.J. Microscopic examination of porcine conceptus-maternal interface between days 10 and 19 of pregnancy. Am. J. Anat. 1990, 188, 221–238. [Google Scholar] [CrossRef]
- Barranco, I.; Padilla, L.; Parrilla, I.; Álvarez-Barrientos, A.; Pérez-Patiño, C.; Peña, F.J.; Martínez, E.A.; Rodriguez-Martínez, H.; Roca, J. Extracellular vesicles isolated from porcine seminal plasma exhibit different tetraspanin expression profiles. Sci. Rep. 2019, 9, 11584. [Google Scholar] [CrossRef]
- Guzewska, M.M.; Heifetz, Y.; Kaczmarek, M.M. Secretion patterns of extracellular vesicles during early pregnancy in the pig – in situ transmission electron microscopy study. In Proceedings of the 4th Winter Workshop of the Society for Biology of Reproduction, Zakopane, Poland, 13–15 February 2019. [Google Scholar]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell. Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell. Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef]
- Hurley, J.H. The ESCRT complexes. Crit. Rev. Biochem. Mol. Biol. 2010, 45, 463–487. [Google Scholar] [CrossRef]
- Saftig, P.; Klumperman, J. Lysosome biogenesis and lysosomal membrane proteins: Trafficking meets function. Nat. Rev. Mol. Cell. Biol. 2009, 10, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Yáñez-Mó, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles. 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- Guzewska, M.M.; Heifetz, Y.; Kaczmarek, M.M. Expression and localization of extracellular vesicles biogenesis pathway elements in the porcine endometrium and conceptuses. In Proceedings of the 51st Annual Meeting of Society for the Study of Reproduction, New Orleans, LA, USA, 10–13 July 2018. [Google Scholar]
- McMahon, H.T.; Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell. Biol. 2011, 12, 517–533. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell. Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Burns, G.W.; Brooks, K.E.; Spencer, T.E. Extracellular vesicles originate from the conceptus and uterus during early pregnancy in sheep. Biol. Reprod. 2016, 94, 56. [Google Scholar] [CrossRef]
- Ng, Y.H.; Rome, S.; Jalabert, A.; Forterre, A.; Singh, H.; Hincks, C.L.; Salamonsen, L.A. Endometrial exosomes/microvesicles in the uterine microenvironment: A new paradigm for embryo-endometrial cross talk at implantation. PLoS ONE 2013, 8, e58502. [Google Scholar] [CrossRef]
- Tang, F.; Kaneda, M.; O’Carroll, D.; Hajkova, P.; Barton, S.C.; Sun, Y.A.; Lee, C.; Tarakhovsky, A.; Lao, K.; Surani, M.A. Maternal microRNAs are essential for mouse zygotic development. Genes Dev. 2007, 21, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, A.K.; Andreu-Vieyra, C.; Franco, H.L.; Ma, L.; Chen, R.; Han, D.Y.; Zhu, H.; Agno, J.E.; Gunaratne, P.H.; DeMayo, F.J.; et al. Deletion of Dicer in somatic cells of the female reproductive tract causes sterility. Mol. Endocrinol. 2008, 22, 2336–2352. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, S.D.; Carletti, M.Z.; Hong, X.; Christenson, L.K. Hormonal regulation of MicroRNA expression in periovulatory mouse mural granulosa cells. Biol. Reprod. 2008, 79, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- McBride, D.; Carré, W.; Sontakke, S.D.; Hogg, C.O.; Law, A.; Donadeu, F.X.; Clinton, M. Identification of miRNAs associated with the follicular- luteal transition in the ruminant ovary. Reproduction 2012, 144, 221–233. [Google Scholar] [CrossRef]
- Ma, T.; Jiang, H.; Gao, Y.; Zhao, Y.; Dai, L.; Xiong, Q.; Xu, Y.; Zhao, Z.; Zhang, J. Microarray analysis of differentially expressed microRNAs in non-regressed and regressed bovine corpus luteum tissue, microRNA-378 may suppress luteal cell apoptosis by targeting the interferon gamma receptor 1 gene. J. Appl. Gen. 2011, 52, 481–486. [Google Scholar] [CrossRef]
- Maalouf, S.W.; Liu, W.S.; Albert, I.; Pate, J.L. Regulating life or death: Potential role of microRNA in rescue of the corpus luteum. Mol. Cell. Endocrinol. 2014, 398, 78–88. [Google Scholar] [CrossRef]
- Gecaj, R.M.; Schanzenbach, C.I.; Kirchner, B.; Pfaffl, M.W.; Riedmaier, I.; Tweedie-Cullen, R.Y.; Berisha, B. The dynamics of microRNA transcriptome in bovine corpus luteum during its formation, function, and regression. Front. Genet. 2017, 8, 213. [Google Scholar] [CrossRef]
- Mohammed, B.T.; Sontakke, S.D.; Ioannidis, J.; Duncan, W.C.; Donadeu, F.X. The adequate corpus luteum: miR-96 promotes luteal cell survival and progesterone production. J. Clin. Endocrinol. Metab. 2017, 102, 2188–2198. [Google Scholar] [CrossRef]
- Diaz, F.J.; Wiltbank, M.C. Acquisition of luteolytic capacity involves differential regulation by prostaglandin F2a of genes involved in progesterone biosynthesis in the porcine corpus luteum. Domest. Anim. Endocrinol. 2005, 28, 172–189. [Google Scholar] [CrossRef]
- Diaz, F.J.; Luo, W.; Wiltbank, M.C. Effect of decreasing intraluteal progesterone on sensitivity of the early porcine corpus luteum to the luteolytic actions of prostaglandin F2alpha. Biol. Reprod. 2011, 841, 26–33. [Google Scholar] [CrossRef]
- Przygrodzka, E.; Witek, K.J.; Kaczmarek, M.M.; Andronowska, A.; Ziecik, A.J. Expression of factors associated with apoptosis in the porcine corpus luteum throughout the luteal phase of the estrous cycle and early pregnancy: Their possible involvement in acquisition of luteolytic sensitivity. Theriogenology 2015, 83, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Przygrodzka, E.; Kaczmarek, M.M.; Kaczynski, P.; Ziecik, A.J. Steroid hormones, prostanoids, and angiogenic systems during rescue of the corpus luteum in pigs. Reproduction 2016, 15, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, S.W.; Smith, C.L.; Pate, J.L. Changes in MicroRNA expression during maturation of the bovine corpus luteum: Regulation of luteal cell proliferation and function by microRNA-34a. Biol. Reprod. 2016, 94, 71. [Google Scholar] [CrossRef] [PubMed]
- Stocco, C.O.; Zhong, L.; Sugimoto, Y.; Ichikawa, A.; Lau, L.F.; Gibori, G. Prostaglandin F2alpha-induced expression of 20alpha-hydroxysteroid dehydrogenase involves the transcription factor NUR77. J. Biol. Chem. 2000, 275, 37202–37211. [Google Scholar] [CrossRef]
- Stocco, C.O.; Lau, L.F.; Gibori, G. A calcium/calmodulin-dependent activation of ERK1/2 614 mediates JunD phosphorylation and induction of nur77 and 20alpha-hsd genes by prostaglandin F2alpha in 615 ovarian cells. J. Biol. Chem. 2002, 277, 3293–3302. [Google Scholar] [CrossRef]
- Atli, M.O.; Bender, R.W.; Mehta, V.; Bastos, M.R.; Luo, W.; Vezina, C.M.; Wiltbank, M.C. Patterns of gene expression in the bovine corpus luteum following repeated intrauterine infusions of low doses of prostaglandin F2alpha. Biol. Reprod. 2012, 86, 130. [Google Scholar] [CrossRef]
- Weber, J.A.; Baxter, D.H.; Zhang, S.; Huang, D.Y.; Huang, K.H.; Lee, M.J.; Galas, D.J.; Wang, K. The microRNA spectrum in 12 body fluids. Clin. Chem. 2010, 56, 1733–1741. [Google Scholar] [CrossRef]
- Wang, K.; Yuan, Y.; Cho, J.H.; McClarty, S.; Baxter, D.; Galas, D.J. Comparing the microRNA spectrum between serum and plasma. PLoS ONE 2012, 7, e41561. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Arroyo, J.D.; Chevillet, J.R.; Kroh, E.M.; Ruf, I.K.; Pritchard, C.C.; Gibson, D.F.; Mitchell, P.S.; Bennett, C.F.; Pogosova-Agadjanyan, E.L.; Stirewalt, D.L.; et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 2011, 108, 5003–5008. [Google Scholar] [CrossRef]
- Vickers, K.C.; Palmisano, B.T.; Shoucri, B.M.; Shamburek, R.D.; Remaley, A.T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat. Cell. Biol. 2011, 13, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.S.; Ishibashi, O.; Ishikawa, G.; Ishikawa, T.; Katayama, A.; Mishima, T.; Takizawa, T.; Shigihara, T.; Goto, T.; Izumi, A.; et al. Human villous trophoblasts express and secrete placenta-specific microRNAs into maternal circulation via exosomes. Biol. Reprod. 2009, 81, 717–729. [Google Scholar] [CrossRef] [PubMed]
- Mouillet, J.F.; Ouyang, Y.; Coyne, C.B.; Sadovsky, Y. MicroRNAs in placental health and disease. Am. J. Obstet. Gynecol. 2015, 213, S163–S172. [Google Scholar] [CrossRef]
- Ioannidis, J.; Donadeu, F.X. Circulating miRNA signatures of early pregnancy in cattle. BMC Genom. 2016, 17, 184. [Google Scholar] [CrossRef] [PubMed]
- Guelfi, G.; Stefanetti, V.; De Luca, S.; Giontella, A.; Barile, V.L.; Barbato, O. Serum microRNAs in buffalo cows: Potential biomarkers of pregnancy. Res. Vet. Sci. 2017, 115, 294–300. [Google Scholar] [CrossRef]
- Ioannidis, J.; Donadeu, F.X. Changes in circulating microRNA levels can be identified as early as day 8 of pregnancy in cattle. PLoS ONE 2017, 12, e0174892. [Google Scholar] [CrossRef]
- Pohler, K.G.; Green, J.A.; Moley, L.A.; Gunewardena, S.; Hung, W.T.; Payton, R.R.; Hong, X.; Christenson, L.K.; Geary, T.W.; Smith, M.F. Circulating microRNA as candidates for early embryonic viability in cattle. Mol. Reprod. Dev. 2017, 84, 731–743. [Google Scholar] [CrossRef]
- Klohonatz, K.M.; Cameron, A.D.; Hergenreder, J.R.; da Silveira, J.C.; Belk, A.D.; Veeramachaneni, D.N.; Bouma, G.J.; Bruemmer, J.E. Circulating miRNAs as potential alternative cell signaling associated with maternal recognition of pregnancy in the mare. Biol. Reprod. 2016, 95, 124. [Google Scholar] [CrossRef]
- Cortez, M.A.; Bueso-Ramos, C.; Ferdin, J.; Lopez-Berestein, G.; Sood, A.; Calin, G.A. MicroRNAs in body fluids—the mix of hormones and biomarkers. Nat. Rev. Clin. Oncol. 2011, 8, 467–477. [Google Scholar] [CrossRef]
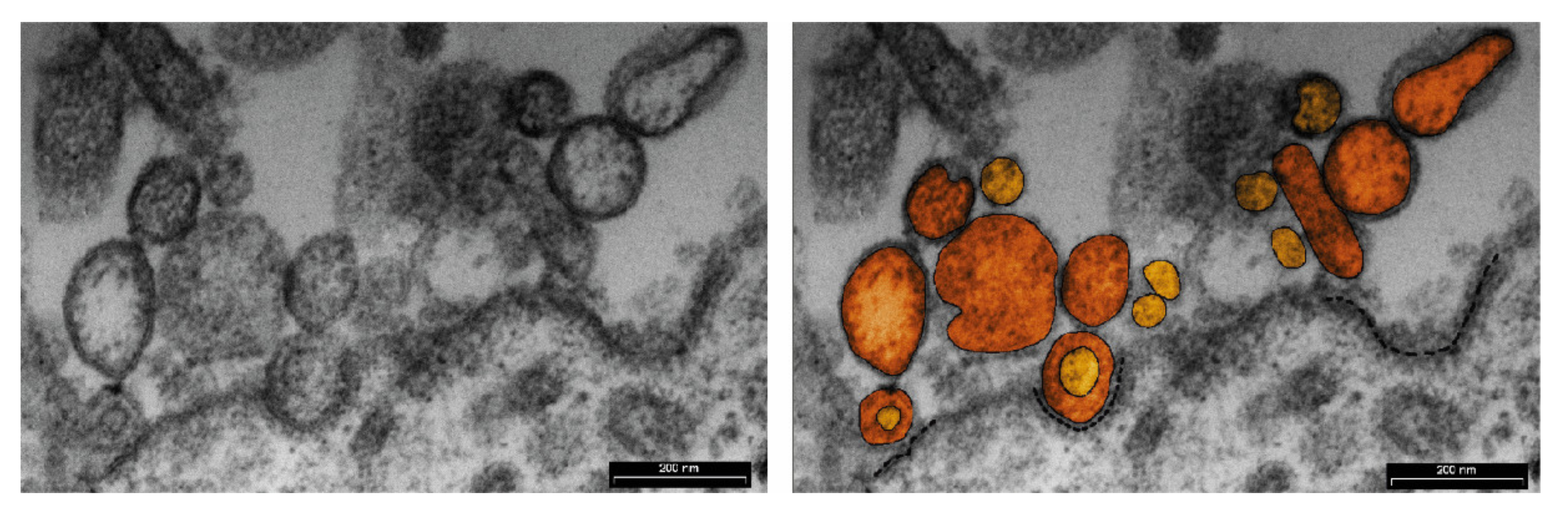

| Source of MiRNAs | Examples of Detected MiRNAs | Ref. | |
|---|---|---|---|
| Embryo | 8 cell | miR-17, -125a-5p, -125b, -128, -205 | [17] |
| blastocyst | miR -92a, -129-5p, -205, -210, -302a | [17] | |
| day 10 | miR-371-3p, -455, -449a, -634, -940, -1193-3p | [22] | |
| miR-7, -302b, -302d, -371-5p, -378 | [38] | ||
| day 12 | miR-96, -138, -152, -483-5p, -765 | [22] | |
| miR-301, -467, -4057 | [34] | ||
| day 14 | miR-10a, -21, -23b, -200a, -301a, -574 | [34] | |
| day 16 (trophoblast) | miR-26a, -27a, -148a | [22] | |
| day 20 (trophoblast) | miR-125b, -199a-3p, -199a-5p | [22] | |
| miR-15b-5p, -18a, -20a-5p, -126-5p, -155-5p, -221-5p | [32] | ||
| day 25 (placenta) | miR-17, -18a, -19a, -20a, -92a | [36] | |
| day 30 (placenta) | miR-17, -106a, -107, -345-5p, -615-3p | [35] | |
| day 50 (trophoblast/placenta) | let-7f-5p, miR-150, -221-5p,-222, | [32] | |
| let-7a/b/c/d/e/f/i, -23a, -24, -27a/b, -29a, -30a/e, -141, -205 | [36] | ||
| Endometrium | day 12 | let-7f/g/i, miR-10b/d, -143-3p | [38] |
| day 15 | let-7b/c/i, miR-125a-3p, -135a*, -140-3p, -149, -181c/d, -200c, -361, -494, -542-5p | [37] | |
| day 16 | miR-23b, -127, -411, -449a | [23] | |
| day 20 | miR-1, -10b, -27a, -30d -101-1, -126, -143, -146b, -193, -193b-3p, -205, -574 | [23] | |
| miR-21, -22-3p, -27a -29a, -30b-5p, -30d, -30e-5p, -149, -183, -191, -205, -296, -323, -362, -432-3p,-503, -574, -4335, -4339 | [33] | ||
| day 26 | miR-15b, -17-5p, -18a, 20a, 30a-3p/5p, -92b, -106a, -126, -132, -221, -222 | [37] | |
| day 50 | miR-30c/e, -215, -411, -487b | [37] | |
| EVs1 | ULFs2 from gestational day 14 and 16 | miR-26a, -125b | [22] |
| CM3 of porcine trophectoderm cells | miR-15b, -16, -17-5p, -20a, -126-5p, -150, -155-5p, -221-5p | [39] | |
| Ovary | CL from gestational day 14 | miR-21a-3p, -345-3p, -371-5p, -4334-5p, -9788-3p, -9840-3p, -9850-5p | [40] |
| Serum | gestational day 16 and 20 | let-7a/b/c/f, miR-10a, -23b, -26a, -30b-5p, -125b, -143-3p | [41,42] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaczmarek, M.M.; Najmula, J.; Guzewska, M.M.; Przygrodzka, E. MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs. Int. J. Mol. Sci. 2020, 21, 2229. https://doi.org/10.3390/ijms21062229
Kaczmarek MM, Najmula J, Guzewska MM, Przygrodzka E. MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs. International Journal of Molecular Sciences. 2020; 21(6):2229. https://doi.org/10.3390/ijms21062229
Chicago/Turabian StyleKaczmarek, Monika M., Joanna Najmula, Maria M. Guzewska, and Emilia Przygrodzka. 2020. "MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs" International Journal of Molecular Sciences 21, no. 6: 2229. https://doi.org/10.3390/ijms21062229
APA StyleKaczmarek, M. M., Najmula, J., Guzewska, M. M., & Przygrodzka, E. (2020). MiRNAs in the Peri-Implantation Period: Contribution to Embryo–Maternal Communication in Pigs. International Journal of Molecular Sciences, 21(6), 2229. https://doi.org/10.3390/ijms21062229





