Functional Analysis of Rice Long-Chain Acyl-CoA Synthetase 9 (OsLACS9) in the Chloroplast Envelope Membrane
Abstract
1. Introduction
2. Results and Discussion
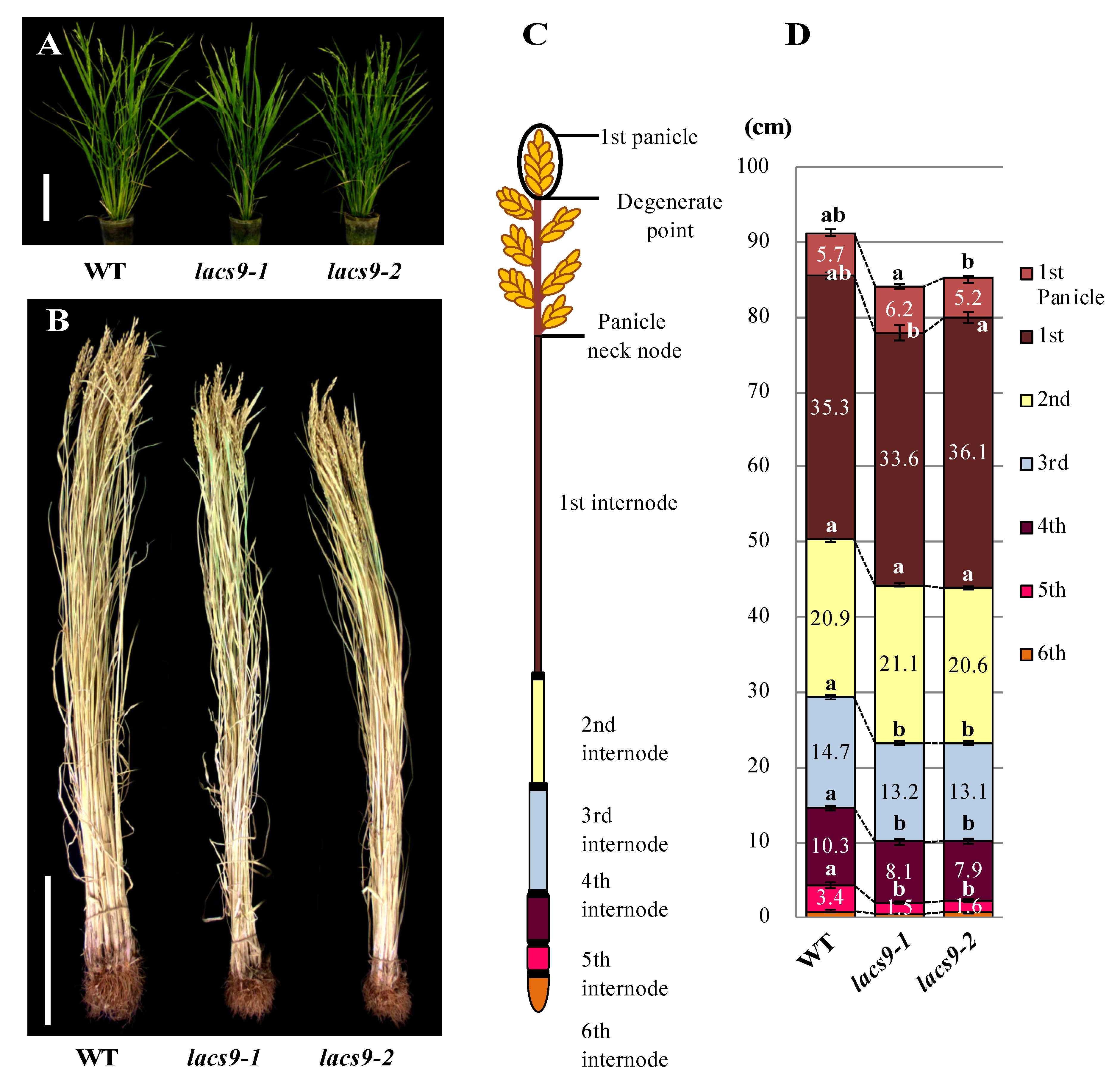
2.1. Loss-of-Function of OsLACS9 Affects Plant Growth
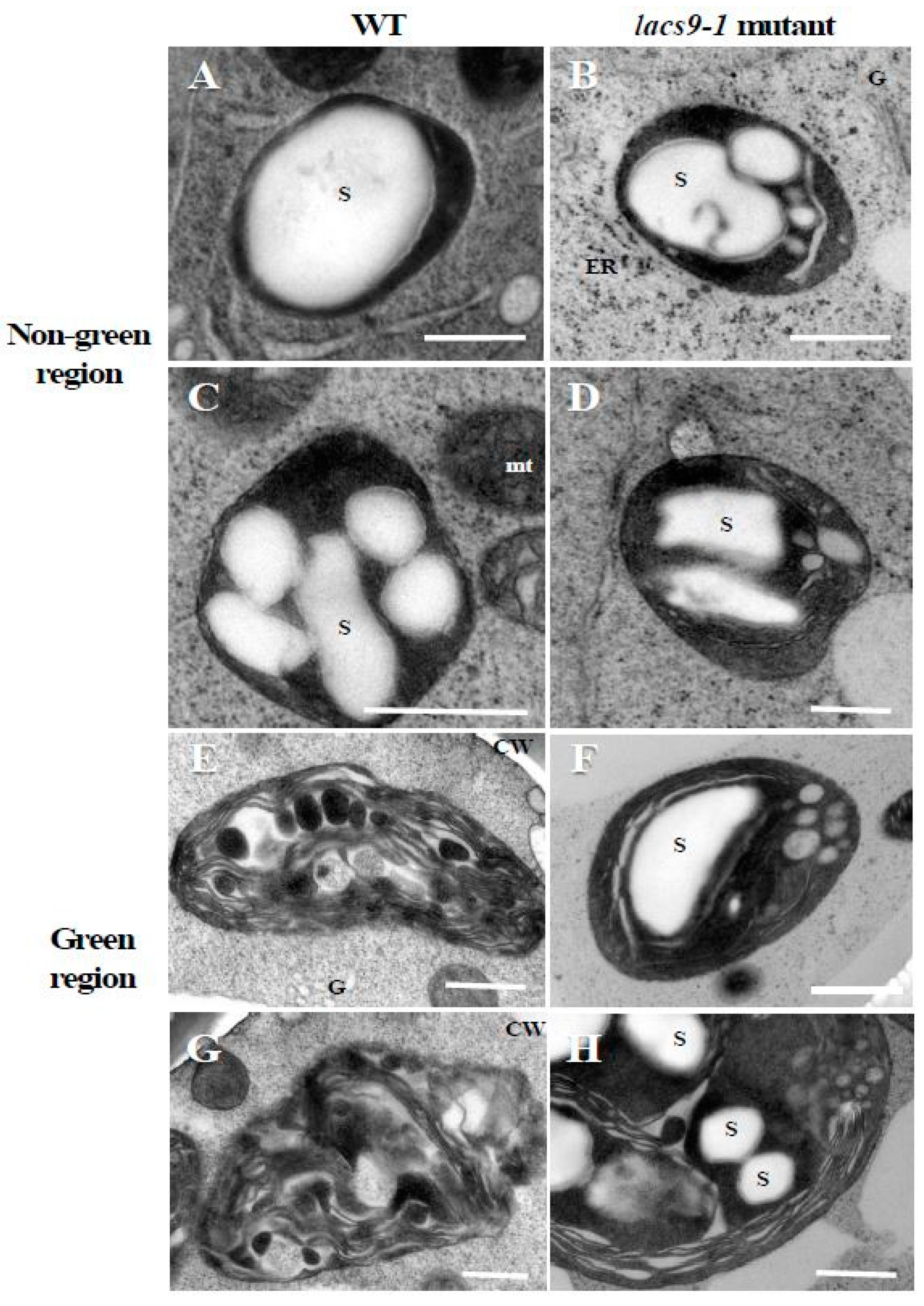
2.2. Chloroplasts of Mutants Lacking OsLACS9 Contain Large Starch Granules
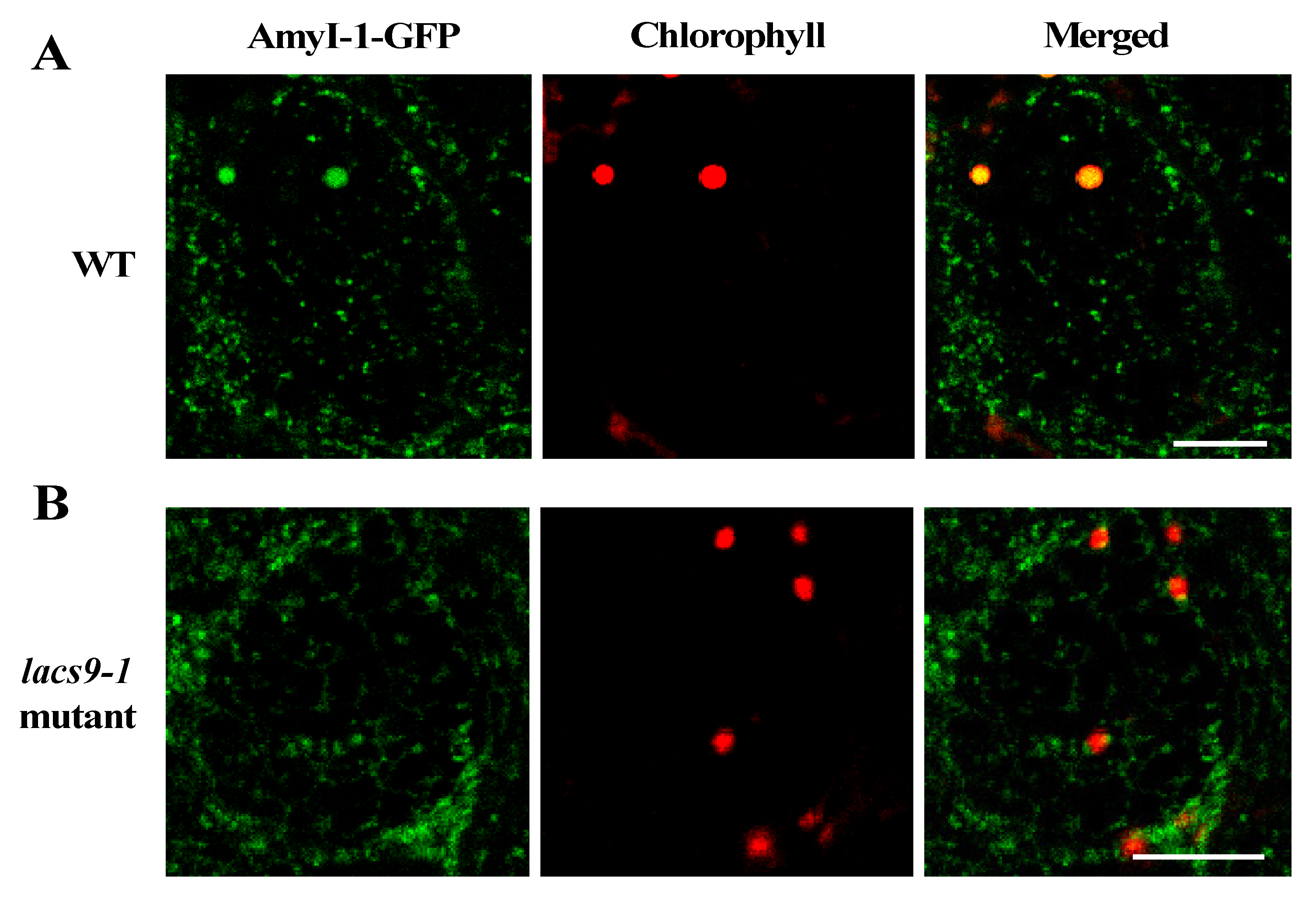
2.3. Prevention of Chloroplast Localization of AmyI-1 in lacs9 Mutant Cells
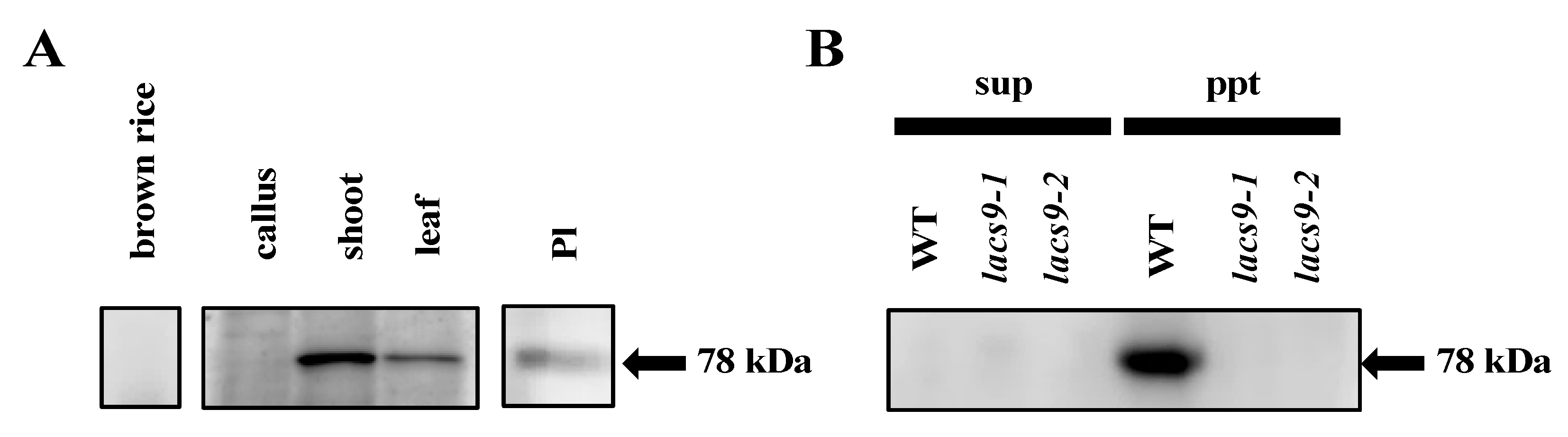
2.4. OsLACS9 is Localized to Both Plastids and The Membrane Fraction
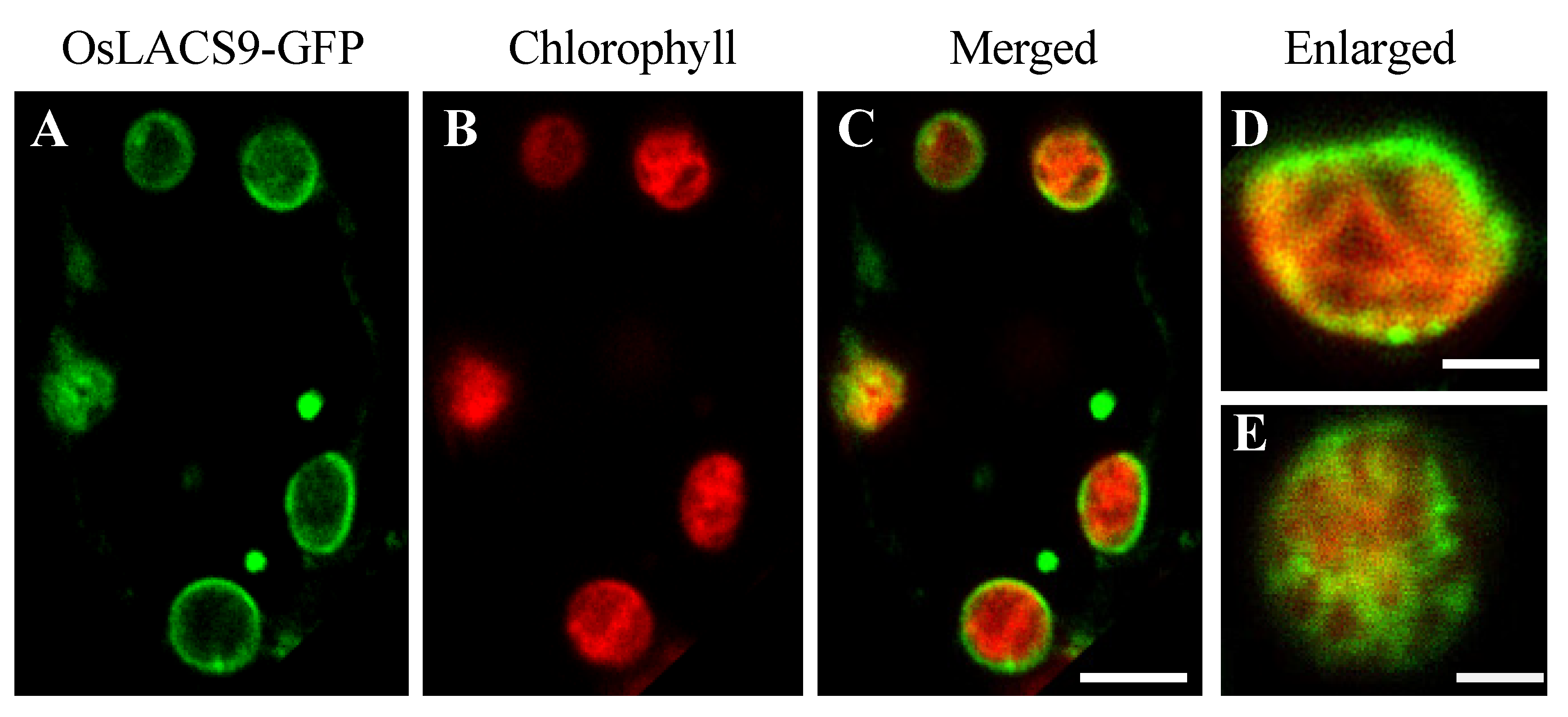
2.5. OsLACS9-GFP is Located in The Envelope Membrane of Rice Chloroplast
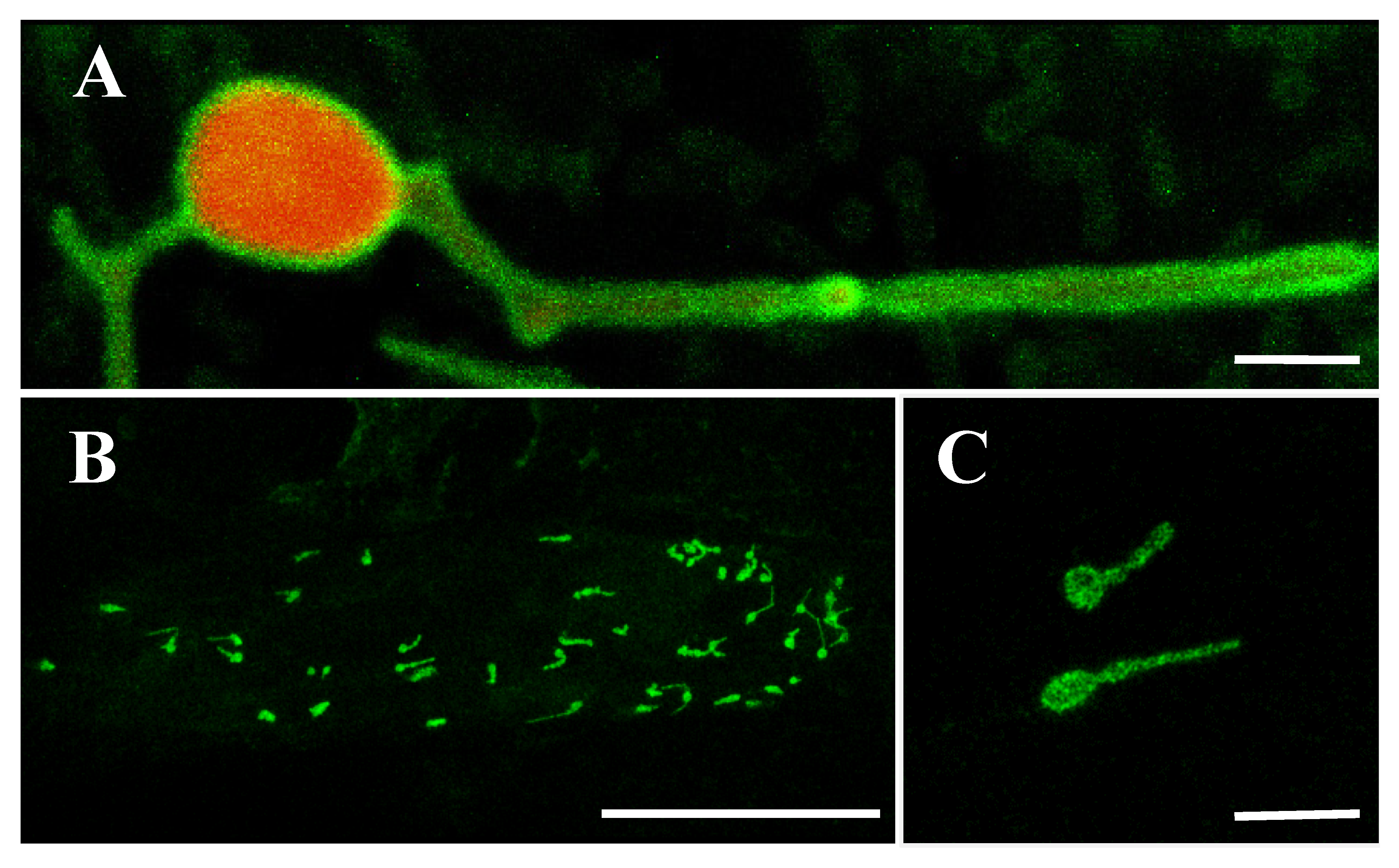
2.6. OsLACS9 Interacts with Each Other on Plastid Envelope Membrane
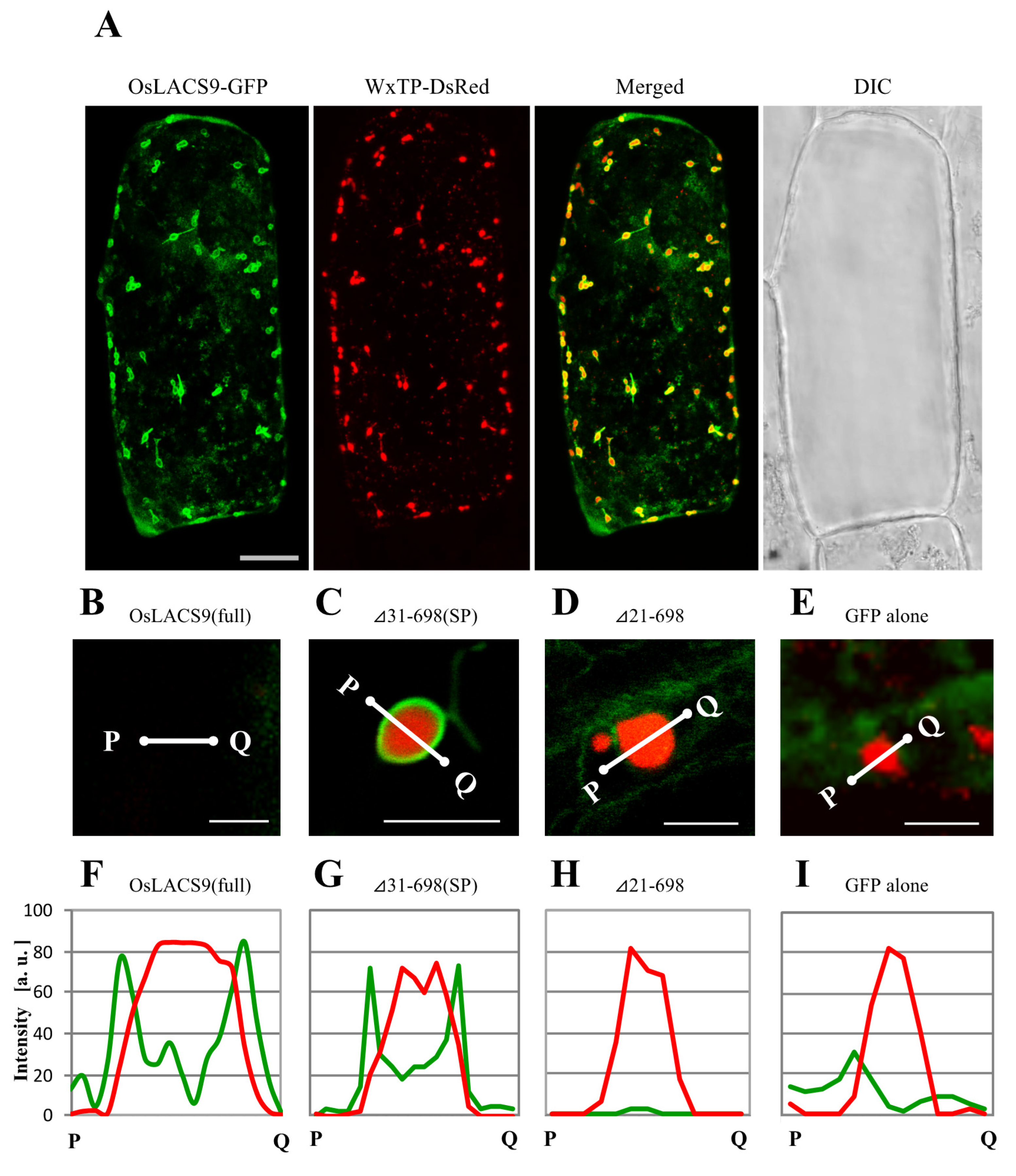
2.7. N-terminal Sequence of OsLACS9 is Necessary for Targeting to Chloroplasts in Onion Cells
3. Materials and Methods
3.1. Plant Materials and Growth Conditions
3.2. Plasmid Construction
3.3. Binary Vector Constructions and Plant Transformations
3.4. Subcellular Localization
3.5. Transient Expression Analysis
3.6. Microscopic Studies
3.6.1. Confocal Laser Scanning Microscope (CLSM)
3.6.2. Transmission Electron Microscope (TEM) Observation
3.7. Protein Extraction and Immunoblotting Analysis
3.8. Computer Analyses
3.9. Statistical Analyses
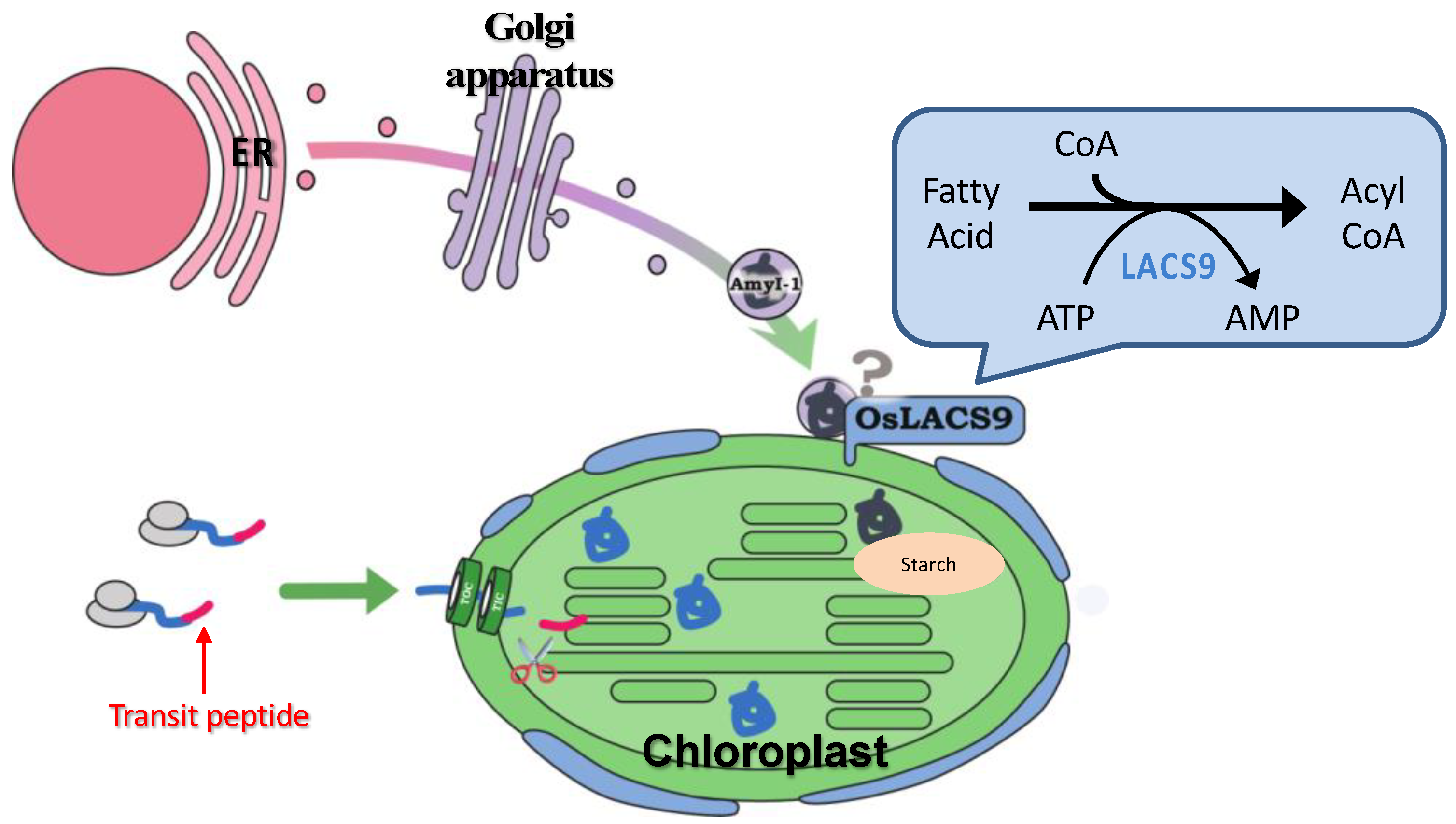
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Streb, S.; Zeeman, S.C. Starch metabolism in arabidopsis. Arab. Book 2012, 10, e0160. [Google Scholar] [CrossRef] [PubMed]
- Bahaji, A.; Li, J.; Sánchez-López, Á.M.; Baroja-Fernández, E.; Muñoz, F.J.; Ovecka, M.; Almagro, G.; Montero, M.; Ezquer, I.; Etxeberria, E.; et al. Starch biosynthesis, its regulation and biotechnological approaches to improve crop yields. Biotechnol. Adv. 2014, 32, 87–106. [Google Scholar] [CrossRef] [PubMed]
- Baslam, M.; Baroja-Fernaândez, E.; Ricarte-Bermejo, A.; Sánchez-López, A.M.; Aranjuelo, I.; Bahaji, A.; Muñoz, F.J.; Almagro, G.; Pujol, P.; Galarza, R.; et al. Genetic and isotope ratio mass spectrometric evidence for the occurrence of starch degradation and cycling in illuminated Arabidopsis leaves. PLoS ONE 2017, 12, e0171245. [Google Scholar] [CrossRef] [PubMed]
- Caspar, T.; Lin, T.P.; Kakefuda, G.; Benbow, L.; Preiss, J.; Somerville, C. Mutants of Arabidopsis with altered regulation of starch degradation. Plant Physiol. 1991, 95, 1181–1188. [Google Scholar] [CrossRef]
- Stitt, M.; Heldt, H.W. Simultaneous synthesis and degradation of starch in spinach chloroplasts in the light. Biochim. Biophys. Acta Bioenerg. 1981, 638, 1–11. [Google Scholar] [CrossRef]
- Szecowka, M.; Heise, R.; Tohge, T.; Nunes-Nesi, A.; Vosloh, D.; Huege, J.; Feil, R.; Lunn, J.; Nikoloski, Z.; Stitt, M.; et al. Metabolic fluxes in an illuminated arabidopsis rosette. Plant Cell 2013, 25, 694–714. [Google Scholar] [CrossRef]
- Kitajima, A.; Asatsuma, S.; Okada, H.; Hamada, Y.; Kaneko, K.; Nanjo, Y.; Kawagoe, Y.; Toyooka, K.; Matsuoka, K.; Takeuchi, M.; et al. The rice alpha-amylase glycoprotein is targeted from the Golgi apparatus through the secretory pathway to the plastids. Plant Cell 2009, 21, 2844–2858. [Google Scholar] [CrossRef]
- Asatsuma, S.; Sawada, C.; Itoh, K.; Okito, M.; Kitajima, A.; Mitsui, T. Involvement of alpha-amylase I-1 in starch degradation in rice chloroplasts. Plant Cell Physiol. 2005, 46, 858–869. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Suzuki, S.; Makino, A. Starch degradation by alpha-amylase in tobacco leaves during the curing process. Soil Sci. Plant Nutr. 2013, 59, 904–911. [Google Scholar] [CrossRef]
- Baslam, M.; Oikawa, K.; Kitajima-koga, A.; Kaneko, K.; Mitsui, T. Golgi-to-plastid traf fi cking of proteins through secretory pathway: Insights into vesicle-mediated import toward the plastids Golgi-to-plastid traf fi cking of proteins through secretory pathway: Insights into vesicle-mediated import toward the plasti. Plant Signal. Behav. 2016, 11, e1221558. [Google Scholar] [CrossRef]
- Burén, S. Targeting and Function of CAH1—Characterization of a Novel Protein Pathway to the Plant Cell Chloroplast; Institutionen för Fysiologisk Botanik, Umeå Universitet: Umeå, Sweden, 2010; ISBN 9789172649330. [Google Scholar]
- Gagat, P.; Bodył, A.; Mackiewicz, P. How protein targeting to primary plastids via the endomembrane system could have evolved? A new hypothesis based on phylogenetic studies. Biol. Direct 2013, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Villarejo, A.; Burén, S.; Larsson, S.; Déjardin, A.; Monné, M.; Rudhe, C.; Karlsson, J.; Jansson, S.; Lerouge, P.; Rolland, N.; et al. Evidence for a protein transported through the secretory pathway en route to the higher plant chloroplast. Nat. Cell Biol. 2005, 7, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Shiraya, T.; Mori, T.; Maruyama, T.; Sasaki, M.; Takamatsu, T.; Oikawa, K.; Itoh, K.; Kaneko, K.; Ichikawa, H.; Mitsui, T. Golgi/plastid-type manganese superoxide dismutase involved in heat-stress tolerance during grain filling of rice. Plant Biotechnol. J. 2015, 13, 1251–1263. [Google Scholar] [CrossRef] [PubMed]
- Nanjo, Y.; Oka, H.; Ikarashi, N.; Kaneko, K.; Kitajima, A.; Mitsui, T.; Muñoz, F.J.; Rodríguez-López, M.; Baroja-Fernández, E.; Pozueta-Romero, J. Rice plastidial N-glycosylated nucleotide pyrophosphatase/phosphodiesterase is transported from the ER-golgi to the chloroplast through the secretory pathway. Plant Cell 2006, 18, 2582–2592. [Google Scholar] [CrossRef]
- Li, Y.-Z.; Zhao, J.-Y.; Wu, S.-M.; Fan, X.-W.; Luo, X.-L.; Chen, B.-S.; Anderson, J.V.; Ceballos, H.; Jansson, C.; Westerbergh, A.; et al. Characters related to higher starch accumulation in cassava storage roots. Sci. Rep. 2016, 6, 19823. [Google Scholar] [CrossRef]
- Li, H.; Gidley, M.J.; Dhital, S. High-amylose starches to bridge the “Fiber Gap”: development, structure, and nutritional functionality. Compr. Rev. Food Sci. Food Saf. 2019, 18, 362–379. [Google Scholar] [CrossRef]
- Morrison, W.R.; Milligan, T.P.; Azudin, M.N. A relationship between the amylose and lipid contents of starches from diploid cereals. J. Cereal Sci. 1984, 2, 257–271. [Google Scholar] [CrossRef]
- Pérez, S.; Bertoft, E. The molecular structures of starch components and their contribution to the architecture of starch granules: A comprehensive review. Starch Stärke 2010, 62, 389–420. [Google Scholar] [CrossRef]
- Li, N.; Gügel, I.L.; Giavalisco, P.; Zeisler, V.; Schreiber, L.; Soll, J.; Philippar, K. FAX1, a novel membrane protein mediating plastid fatty acid export. PLoS Biol. 2015, 13, e1002053. [Google Scholar] [CrossRef]
- Schnurr, J.A.; Shockey, J.M.; De Boer, G.-J.; Browse, J.A. Fatty acid export from the chloroplast. Molecular characterization of a major plastidial acyl-coenzyme a synthetase from arabidopsis. Plant Physiol. 2002, 129, 1700–1709. [Google Scholar] [CrossRef]
- Xiao, S.; Chye, M.L. New roles for acyl-CoA-binding proteins (ACBPs) in plant development, stress responses and lipid metabolism. Prog. Lipid Res. 2011, 50, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Chapman, K.D.; Ohlrogge, J.B. Compartmentation of triacylglycerol accumulation in plants. J. Biol. Chem. 2012, 287, 2288–2294. [Google Scholar] [CrossRef] [PubMed]
- Shockey, J.; Browse, J. Genome-level and biochemical diversity of the acyl-activating enzyme superfamily in plants. Plant J. 2011, 66, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Groot, P.H.; Scholte, H.R.; Hülsmann, W.C. Fatty acid activation: Specificity, localization, and function. Adv. Lipid Res. 1976, 14, 75–126. [Google Scholar] [PubMed]
- Shockey, J.M.; Fulda, M.S.; Browse, J.A. Arabidopsis contains nine long-chain acyl-coenzyme a synthetase genes that participate in fatty acid and glycerolipid metabolism. Plant Physiol. 2002, 129, 1710–1722. [Google Scholar] [CrossRef]
- Breuers, F.K.H.; Bräutigam, A.; Geimer, S.; Welzel, U.Y.; Stefano, G.; Renna, L.; Brandizzi, F.; Weber, A.P.M. Dynamic remodeling of the plastid envelope membranes—A tool for chloroplast envelope in vivo localizations. Front. Plant Sci. 2012, 3, 7. [Google Scholar] [CrossRef]
- Andrews, J.; Keegstra, K. Acyl-CoA synthetase is located in the outer membrane and Acyl-CoA thioesterase in the inner membrane of pea chloroplast envelopes. Plant Physiol. 1983, 72, 735–740. [Google Scholar] [CrossRef]
- Pongdontri, P.; Hills, M. Characterization of a novel plant acyl-coA synthetase that is expressed in lipogenic tissues of Brassica napus L. Plant Mol. Biol. 2001, 47, 717–726. [Google Scholar] [CrossRef]
- Aznar-Moreno, J.A.; Venegas Calerón, M.; Martínez-Force, E.; Garcés, R.; Mullen, R.; Gidda, S.K.; Salas, J.J. Sunflower (Helianthus annuus ) long-chain acyl-coenzyme A synthetases expressed at high levels in developing seeds. Physiol. Plant. 2014, 150, 363–373. [Google Scholar] [CrossRef]
- Fulda, M.; Shockey, J.; Werber, M.; Wolter, F.P.; Heinz, E. Two long-chain acyl-CoA synthetases from Arabidopsis thaliana involved in peroxisomal fatty acid β-oxidation. Plant J. 2002, 32, 93–103. [Google Scholar] [CrossRef]
- Jessen, D.; Olbrich, A.; Knüfer, J.; Krüger, A.; Hoppert, M.; Polle, A.; Fulda, M. Combined activity of LACS1 and LACS4 is required for proper pollen coat formation in Arabidopsis. Plant J. 2011, 68, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Weng, H.; Molina, I.; Shockey, J.; Browse, J. Organ fusion and defective cuticle function in a lacs1 lacs2 double mutant of Arabidopsis. Planta 2010, 231, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- Schnurr, J.; Shockey, J.; Browse, J. The Acyl-CoA synthetase encoded by LACS2 is essential for normal cuticle development in arabidopsis. Plant Cell 2004, 16, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Lü, S.; Song, T.; Kosma, D.K.; Parsons, E.P.; Rowland, O.; Jenks, M.A. Arabidopsis CER8 encodes LONG-CHAIN ACYL-COA SYNTHETASE 1 (LACS1) that has overlapping functions with LACS2 in plant wax and cutin synthesis. Plant J. 2009, 59, 553–564. [Google Scholar] [CrossRef]
- Fulda, M.; Schnurr, J.; Abbadi, A.; Heinz, E.; Browse, J. Peroxisomal Acyl-CoA Synthetase Activity Is Essential for Seedling Development in Arabidopsis thaliana. Plant Cell 2004, 16, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Katavic, V.; Li, F.; Haughn, G.W.; Kunst, L. Insertional mutant analysis reveals that long-chain acyl-CoA synthetase 1 (LACS1), but not LACS8, functionally overlaps with LACS9 in Arabidopsis seed oil biosynthesis. Plant J. 2010, 64, 1048–1058. [Google Scholar] [CrossRef]
- Ritzenthaler, C.; Nebenführ, A.; Movafeghi, A.; Stussi-Garaud, C.; Behnia, L.; Pimpl, P.; Staehelin, L.A.; Robinson, D.G. Reevaluation of the effects of brefeldin a on plant cells using tobacco bright yellow 2 cells expressing golgi-targeted green fluorescent protein and copi antisera. Plant Cell 2002, 14, 237–261. [Google Scholar] [CrossRef]
- Mitsui, T.; Ochiai, A.; Yamakawa, H.; Kaneko, K.; Kitajima-Koga, A.; Baslam, M. Novel molecular and cell biological insights into function of rice α-amylase. Amylase 2018, 2, 30–38. [Google Scholar] [CrossRef]
- Radif, Y.; Ndiaye, H.; Kalantzi, V.; Jacobs, R.; Hall, A.; Minogue, S.; Waugh, M.G. The endogenous subcellular localisations of the long chain fatty acid-activating enzymes ACSL3 and ACSL4 in sarcoma and breast cancer cells. Mol. Cell. Biochem. 2018, 448, 275–286. [Google Scholar] [CrossRef]
- Hiei, Y.; Ohta, S.; Komari, T.; Kumashiro, T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J. 1994, 6, 271–282. [Google Scholar] [CrossRef]
- Fukuoka, H.; Ogawa, T.; Mitsuhara, I.; Iwai, T.; Isuzugawa, K.; Nishizawa, Y.; Gotoh, Y.; Nishizawa, Y.; Tagiri, A.; Ugaki, M.; et al. Agrobacterium-mediated transformation of monocot and dicot plants using the NCR promoter derived from soybean chlorotic mottle virus. Plant Cell Rep. 2000, 19, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, T.; Yamaguchi, J.; Akazawa, T. Physicochemical and serological characterization of rice α-amylase isoforms and identification of their corresponding genes. Plant Physiol. 1996, 110, 1395–1404. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, K.; Takamatsu, T.; Inomata, T.; Oikawa, K.; Itoh, K.; Hirose, K.; Amano, M.; Nishimura, S.-I.; Toyooka, K.; Matsuoka, K.; et al. N-Glycomic and microscopic subcellular localization analyses of NPP1, 2 and 6 strongly indicate that trans-Golgi compartments participate in the Golgi to plastid traffic of nucleotide pyrophosphatase/phosphodiesterases in rice. Plant Cell Physiol. 2016, 57, 1610–1628. [Google Scholar] [CrossRef]
- Takamatsu, T.; Baslam, M.; Inomata, T.; Oikawa, K.; Itoh, K.; Ohnishi, T.; Kinoshita, T.; Mitsui, T. Optimized method of extracting rice chloroplast DNA for high-quality plastome resequencing and de novo assembly. Front. Plant Sci. 2018, 9, 266. [Google Scholar] [CrossRef]
- Inomata, T.; Baslam, M.; Masui, T.; Koshu, T.; Takamatsu, T.; Kaneko, K.; Pozueta-Romero, J.; Mitsui, T. Proteomics analysis reveals non-controlled activation of photosynthesis and protein synthesis in a rice npp1 mutant under high temperature and elevated CO2 conditions. Int. J. Mol. Sci. 2018, 19, 2655. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kitajima-Koga, A.; Baslam, M.; Hamada, Y.; Ito, N.; Taniuchi, T.; Takamatsu, T.; Oikawa, K.; Kaneko, K.; Mitsui, T. Functional Analysis of Rice Long-Chain Acyl-CoA Synthetase 9 (OsLACS9) in the Chloroplast Envelope Membrane. Int. J. Mol. Sci. 2020, 21, 2223. https://doi.org/10.3390/ijms21062223
Kitajima-Koga A, Baslam M, Hamada Y, Ito N, Taniuchi T, Takamatsu T, Oikawa K, Kaneko K, Mitsui T. Functional Analysis of Rice Long-Chain Acyl-CoA Synthetase 9 (OsLACS9) in the Chloroplast Envelope Membrane. International Journal of Molecular Sciences. 2020; 21(6):2223. https://doi.org/10.3390/ijms21062223
Chicago/Turabian StyleKitajima-Koga, Aya, Marouane Baslam, Yuuki Hamada, Namiko Ito, Tomoko Taniuchi, Takeshi Takamatsu, Kazusato Oikawa, Kentaro Kaneko, and Toshiaki Mitsui. 2020. "Functional Analysis of Rice Long-Chain Acyl-CoA Synthetase 9 (OsLACS9) in the Chloroplast Envelope Membrane" International Journal of Molecular Sciences 21, no. 6: 2223. https://doi.org/10.3390/ijms21062223
APA StyleKitajima-Koga, A., Baslam, M., Hamada, Y., Ito, N., Taniuchi, T., Takamatsu, T., Oikawa, K., Kaneko, K., & Mitsui, T. (2020). Functional Analysis of Rice Long-Chain Acyl-CoA Synthetase 9 (OsLACS9) in the Chloroplast Envelope Membrane. International Journal of Molecular Sciences, 21(6), 2223. https://doi.org/10.3390/ijms21062223





