Perspectives on the Potential Benefits of Antihypertensive Peptides towards Metabolic Syndrome
Abstract
1. Introduction
2. Pathologies for Hypertension and Metabolic Syndrome
2.1. Systemic Renin Angiotensin System (RAS)
2.2. Endothelial Dysfunction
2.3. Oxidative Stress
2.4. Inflammation
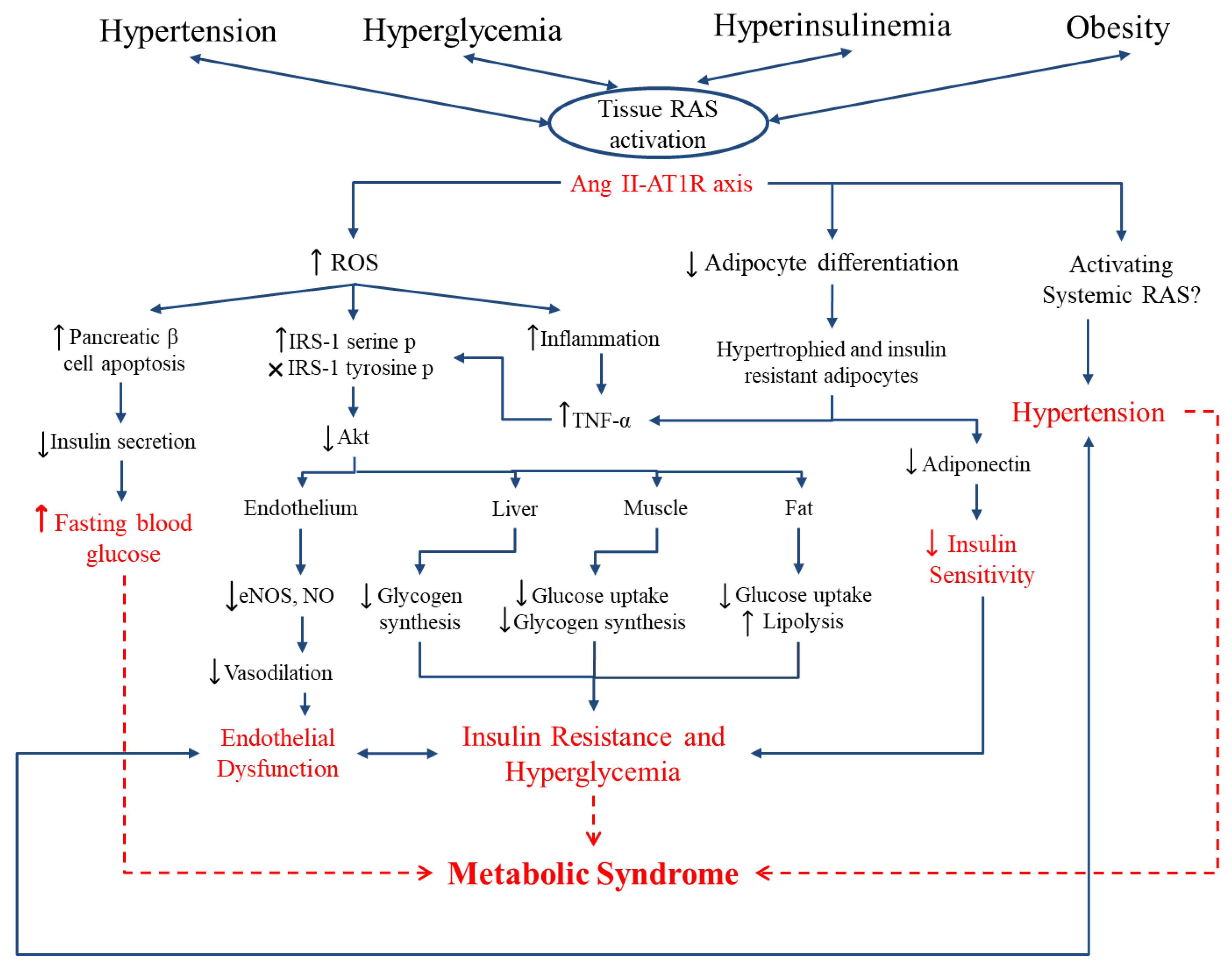
3. Renin-Angiotensin System (RAS), Oxidative Stress, and MetS
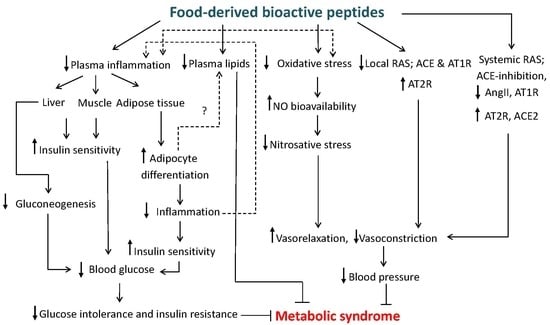
4. Food Protein Derived Bioactive Peptides; Peptides against RAS and MetS
4.1. Dairy-Derived Peptides
4.2. Egg-Derived Peptides
4.3. Marine-Derived Peptides
4.4. Peptides from Other Sources
| Treatment | Active Component | Model | Observed Effects | Ref |
|---|---|---|---|---|
| Milk fermented with Lb. case | VPP/IPP | In vitro | Antioxidant activities | [127] |
| Casein derived peptides | VPP/IPP | L-NAME-treated Wistar rats | Enhanced NO-bioavailability, reduced cardiac and renal damage | [126] |
| Casein derived peptides | VPP/IPP | In vitro (3T3-F442A preadipocytes) | Adipogenic differentiation, insulin mimetic, and anti-inflammatory effects | [133] |
| Fermented milk/casein hydrolysate | VPP/IPP | Apolipoprotein E–deficient mice | Reduced mRNA expression of inflammatory cytokines and oxidized LDL- receptor | [132] |
| Casein derived peptide | VPP | High-fat diet (HFD) fed C57BL/6J mice | Less inflammation in adipose tissue (reduced activated monocytes and pro-inflammatory macrophages, MCP-1 and IL-6 gene expression) | [125] |
| Egg white hydrolysate | WEKAFKDED, QAMPFRVTEQE, ERYPIL, VFKGL | In vitro (3T3 F442A preadipocytes) | Enhanced preadipocyte differentiation and showed insulin mimetic and sensitizing effects | [139,142] |
| Egg white hydrolysate | Mixture of peptides | Diet-induced insulin resistant SD rats | Improved glucose tolerance and insulin sensitivity, reduced adipocyte size and inflammation | [141] |
| Ovotransferrin-derived peptide | IRW | In vitro (HUVECs), SHRs | Reduced inflammatory gene expression, antioxidant and anti-inflammatory effects | [146,171,172] |
| Ovotransferrin-derived peptide | IRW | In vitro (Ang II-treated L6 cells) | Improved glucose uptake, and antioxidant effects (decreased Ang II-stimulated ROS formation and NADPH oxidase activation) | [149] |
| Ovotransferrin-derived peptide | IQW | In vitro (HUVECs) | Antioxidant and anti-inflammatory effects | [146] |
| Lysozyme hydrolysate | Mixture of peptides | Zucker diabetic fatty rats | Decreased oxidative stress, inflammation, and COX expression | [154] |
| Egg white hydrolysate | FRADHPFL, RADHPFL, YAEERYPIL, YRGGLEPINF, ESIINF, RDILNQ, IVF, YQIGL, SALAM, FSL | Obese Zucker rats | Antioxidant and anti-inflammatory effects, decreased epididymal fat mass, improved hepatic steatosis, and reduced plasma free fatty acids | [156] |
| Egg white hydrolysate | Mixture of peptides | High-fat/high-dextrose diet-fed Wistar rats | Reduced body weight, abdominal fat, and plasma glucose | [173] |
| Egg yolk protein hydrolysate | YINQMPQKSRE, YINQMPQKSREA, VTGRFAGHPAAQ, YIEAVNKVSPRAGQF | In vitro | Antioxidant, α-glucosidase and DPP-IV inhibitory activities | [157] |
| Pacific cod (Gadus macrocephalus) skin gelatin hydrolysate | TCSP, TGGGNV, LLMLDNDLPP | In vitro | Antioxidant | [161,162] |
| Skate (Okamejei kenojei) gelatin hydrolysate | MVGSAPGVL, LGPLGHQ | In vitro (human endothelial cells) | Antioxidant (radical scavenging activity, increased protein level and upregulated gene expression of antioxidant enzymes) | [163] |
| Sardine protein hydrolysate | Mixture of peptides | Stroke-prone SHRs | Improved glucose handling and insulin sensitivity | [164] |
| Sardine muscle hydrolysate | MY | In vitro (human endothelial cells) | Antioxidant activity (protecting endothelial cells from oxidative stress via induction of heme oxygenase-1 and ferritin) | [166] |
| Wild Chum Salmon protein hydrolysate | Oligopeptides with molecular weights of 130–3000 Da | High fat diet (HFD) fed SD rats | Reduced fasting blood glucose, reduced β-cells apoptosis, antioxidant and anti-inflammatory effects (reduced serum TNFα, IFNγ, and MDA, increased SOD and GSH) | [167] |
| Marine snail meat and visceral mass | YSQLENEFDR, YIAEDAER | In vitro, and zebrafish model | Antioxidant, α-amylase and α-glucosidase inhibitory activities | [168] |
| Hemp seed meal protein hydrolysate | Mixture of peptides | SHRs (young and adult) | Antioxidant effects (increased plasma SOD and CAT and decreased total peroxides) | [170] |
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACE | Angiotensin converting enzyme |
| Ang I | Angiotensin I |
| Ang II | Angiotensin II |
| AT1R | Angiotensin II type 1 receptor |
| AT2R | Angiotensin II type 2 receptor |
| CVDs | Cardiovascular diseases |
| eNOS | Endothelial nitric oxide synthase |
| ICAM-1 | Intercellular adhesion molecule 1 |
| IL | Interleukin |
| iNOS | Inducible nitric oxide synthase |
| IκB | Inhibitor kappaB |
| LDL | Low density lipoprotein |
| MCP-1 | Monocyte chemotactic protein-1 |
| MetS | Metabolic syndrome |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NF-κB | Nuclear factor kappaB |
| NO | Nitric oxide |
| NOS | Nitric oxide synthase |
| RAS | Renin angiotensin system |
| ROS | Reactive oxygen species |
| SHR | Spontaneously hypertensive rats |
| TNF | Tumor necrosis factor |
| VCAM-1 | Vascular adhesion molecule 1 |
| VSMC | Vascular smooth muscle cell |
| List of amino acids | |
| A (Ala) | Alanine |
| C (Cys) | Cysteine |
| D (Asp) | Aspartic acid |
| E (Glu) | Glutamic acid |
| F (Phe) | Phenylalanine |
| G (Gly) | Glycine |
| H (His) | Histidine |
| I (Ile) | Isoleucine |
| K (Lys) | Lysine |
| L (Leu) | Leucine |
| M (Met) | Methionine |
| N (Asn) | Asparagine |
| P (Pro) | Proline |
| Q (Glu) | Glutamine |
| R (Arg) | Arginine |
| S (Ser) | Serine |
| T (Thr) | Threonine |
| V (Val) | Valine |
| W (Trp) | Tryptophan |
| ROS | Reactive oxygen species |
| Y (Tyr) | Tyrosine |
References
- Han, T.S.; Lean, M.E. A clinical perspective of obesity, metabolic syndrome and cardiovascular disease. JRSM Cardiovasc. Dis. 2016, 5, 2048004016633371. [Google Scholar] [CrossRef] [PubMed]
- Oda, E. Metabolic syndrome: Its history, mechanisms, and limitations. Acta Diabetol. 2012, 49, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Nerenberg, K.A.; Zarnke, K.B.; Leung, A.A.; Dasgupta, K.; Butalia, S.; McBrien, K.; Harris, K.C.; Nakhla, M.; Cloutier, L.; Gelfer, M.; et al. Hypertension Canada’s 2018 Guidelines for Diagnosis, Risk Assessment, Prevention, and Treatment of Hypertension in Adults and Children. Can. J. Cardiol. 2018, 34, 506–525. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2016, 26, 364–373. [Google Scholar] [CrossRef]
- Schillaci, G.; Pirro, M.; Vaudo, G.; Gemelli, F.; Marchesi, S.; Porcellati, C.; Mannarino, E. Prognostic value of the metabolic syndrome in essential hypertension. J. Am. Coll. Cardiol. 2004, 43, 1817–1822. [Google Scholar] [CrossRef]
- Mule, G.; Cottone, S.; Nardi, E.; Andronico, G.; Cerasola, G. Metabolic syndrome in subjects with essential hypertension: Relationships with subclinical cardiovascular and renal damage. Minerva Cardioangiol. 2006, 54, 173–194. [Google Scholar]
- Marcus, Y.; Shefer, G.; Stern, N. Adipose tissue renin-angiotensin-aldosterone system (RAAS) and progression of insulin resistance. Mol. Cell. Endocrinol. 2013, 378, 1–14. [Google Scholar] [CrossRef]
- Prasad, H.; Ryan, D.A.; Celzo, M.F.; Stapleton, D. Metabolic syndrome: Definition and therapeutic implications. Postgrad. Med. 2012, 124, 21–30. [Google Scholar] [CrossRef]
- Povel, C.M.; Beulens, J.W.; van der Schouw, Y.T.; Dolle, M.E.; Spijkerman, A.M.; Verschuren, W.M.; Feskens, E.J.; Boer, J.M. Metabolic syndrome model definitions predicting type 2 diabetes and cardiovascular disease. Diabetes Care 2013, 36, 362–368. [Google Scholar] [CrossRef]
- Maeda, A.; Tamura, K.; Wakui, H.; Ohsawa, M.; Azushima, K.; Uneda, K.; Kanaoka, T.; Kobayashi, R.; Ohki, K.; Matsuda, M.; et al. Effects of the Angiotensin receptor blocker olmesartan on adipocyte hypertrophy and function in mice with metabolic disorders. Biomed. Res. Int. 2014, 2014, 946492. [Google Scholar] [CrossRef]
- Goossens, G.H.; Moors, C.C.; van der Zijl, N.J.; Venteclef, N.; Alili, R.; Jocken, J.W.; Essers, Y.; Cleutjens, J.P.; Clement, K.; Diamant, M.; et al. Valsartan improves adipose tissue function in humans with impaired glucose metabolism: A randomized placebo-controlled double-blind trial. PLoS ONE 2012, 7, e39930. [Google Scholar] [CrossRef] [PubMed]
- Nevelsteen, I.; Bito, V.; Van der Mieren, G.; Vanderper, A.; Van den Bergh, A.; Sipido, K.R.; Mubagwa, K.; Herijgers, P. ACE-inhibition, but not weight reduction restores cardiomyocyte response to beta-adrenergic stimulation in the metabolic syndrome. BMC Cardiovasc. Disord. 2013, 13, 51. [Google Scholar] [CrossRef] [PubMed]
- Van der Mieren, G.; Nevelsteen, I.; Vanderper, A.; Oosterlinck, W.; Flameng, W.; Herijgers, P. Angiotensin-converting enzyme inhibition and food restriction restore delayed preconditioning in diabetic mice. Cardiovasc. Diabetol. 2013, 12, 36. [Google Scholar] [CrossRef] [PubMed]
- Bitkin, E.C.; Boyraz, M.; Taskin, N.; Akcay, A.; Ulucan, K.; Akyol, M.B.; Akcay, T. Effects of ACE inhibitors on insulin resistance and lipid profile in children with metabolic syndrome. J. Clin. Res. Pediatr. Endocrinol. 2013, 5, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Yakubova, A.; Thorrez, L.; Svetlichnyy, D.; Zwarts, L.; Vulsteke, V.; Laenen, G.; Oosterlinck, W.; Moreau, Y.; Dehaspe, L.; Van Houdt, J.; et al. ACE-inhibition induces a cardioprotective transcriptional response in the metabolic syndrome heart. Sci. Rep. 2018, 8, 16169. [Google Scholar] [CrossRef]
- Cicero, A.F.; Colletti, A. Role of phytochemicals in the management of metabolic syndrome. Phytomed. Int. J. Phytother. Phytopharm. 2016, 23, 1134–1144. [Google Scholar] [CrossRef]
- Erdmann, K.; Cheung, B.W.Y.; Schroder, H. The possible roles of food-derived bioactive peptides in reducing the risk of cardiovascular disease. J. Nutr. Biochem. 2008, 19, 643–654. [Google Scholar] [CrossRef]
- Udenigwe, C.C.; Aluko, R.E. Food Protein-Derived Bioactive Peptides: Production, Processing, and Potential Health Benefits. J. Food Sci. 2012, 77, R11–R24. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Jahandideh, F.; Wu, J.P. Food-Derived Bioactive Peptides on Inflammation and Oxidative Stress. Biomed. Res. Int. 2014. [Google Scholar] [CrossRef]
- Majumder, K.; Wu, J.P. Molecular Targets of Antihypertensive Peptides: Understanding the Mechanisms of Action Based on the Pathophysiology of Hypertension. Int. J. Mol. Sci. 2015, 16, 256–283. [Google Scholar] [CrossRef]
- Daskaya-Dikmen, C.; Yucetepe, A.; Karbancioglu-Guler, F.; Daskaya, H.; Ozcelik, B. Angiotensin-I-Converting Enzyme (ACE)-Inhibitory Peptides from Plants. Nutrients 2017, 9, 316. [Google Scholar] [CrossRef] [PubMed]
- Mendizabal, Y.; Llorens, S.; Nava, E. Hypertension in metabolic syndrome: Vascular pathophysiology. Int. J. Hypertens. 2013, 2013, 230868. [Google Scholar] [CrossRef] [PubMed]
- Schulz, E.; Gori, T.; Munzel, T. Oxidative stress and endothelial dysfunction in hypertension. Hypertens. Res. 2011, 34, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Perazella, M.A.; Setaro, J.F. Renin-angiotensin-aldosterone system: Fundamental aspects and clinical implications in renal and cardiovascular disorders. J. Nucl. Cardiol. Off. Publ. Am. Soc. Nucl. Cardiol. 2003, 10, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, J.L.; Sigmund, C.D. Minireview: Overview of the renin-angiotensin system—An endocrine and paracrine system. Endocrinology 2003, 144, 2179–2183. [Google Scholar] [CrossRef] [PubMed]
- Oparil, S.; Zaman, M.A.; Calhoun, D.A. Pathogenesis of hypertension. Ann. Intern. Med. 2003, 139, 761–776. [Google Scholar] [CrossRef]
- Wassmann, S.; Nickenig, G. Pathophysiological regulation of the AT1-receptor and implications for vascular disease. J. Hypertens. Suppl. Off. J. Int. Soc. Hypertens. 2006, 24, S15–S21. [Google Scholar] [CrossRef]
- Bindom, S.M.; Hans, C.P.; Xia, H.; Boulares, A.H.; Lazartigues, E. Angiotensin I-converting enzyme type 2 (ACE2) gene therapy improves glycemic control in diabetic mice. Diabetes 2010, 59, 2540–2548. [Google Scholar] [CrossRef]
- Fernandes, L.; Fortes, Z.B.; Nigro, D.; Tostes, R.C.; Santos, R.A.; Catelli De Carvalho, M.H. Potentiation of bradykinin by angiotensin-(1-7) on arterioles of spontaneously hypertensive rats studied in vivo. Hypertension 2001, 37, 703–709. [Google Scholar] [CrossRef]
- Ocaranza, M.P.; Jalil, J.E. On Endogenous Angiotensin II Antagonism in Hypertension: The Role of Dipeptidyl Peptidase III. Hypertension 2016, 68, 552–554. [Google Scholar] [CrossRef]
- Lautner, R.Q.; Villela, D.C.; Fraga-Silva, R.A.; Silva, N.; Verano-Braga, T.; Costa-Fraga, F.; Jankowski, J.; Jankowski, V.; Sousa, F.; Alzamora, A.; et al. Discovery and characterization of alamandine: A novel component of the renin-angiotensin system. Circ. Res. 2013, 112, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Hrenak, J.; Paulis, L.; Simko, F. Angiotensin A/Alamandine/MrgD Axis: Another Clue to Understanding Cardiovascular Pathophysiology. Int. J. Mol. Sci. 2016, 17, 1098. [Google Scholar] [CrossRef] [PubMed]
- Keidar, S.; Kaplan, M.; Hoffman, A.; Aviram, M. Angiotensin II stimulates macrophage-mediated oxidation of low density lipoproteins. Atherosclerosis 1995, 115, 201–215. [Google Scholar] [CrossRef]
- Li, D.; Saldeen, T.; Romeo, F.; Mehta, J.L. Oxidized LDL upregulates angiotensin II type 1 receptor expression in cultured human coronary artery endothelial cells: The potential role of transcription factor NF-kappaB. Circulation 2000, 102, 1970–1976. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.D.; Schaefer, R.; Mehta, J.L. Cross-talk between dyslipidemia and renin-angiotensin system and the role of LOX-1 and MAPK in atherogenesis studies with the combined use of rosuvastatin and candesartan. Atheroscler. Suppl. 2006, 184, 295–301. [Google Scholar] [CrossRef]
- Deanfield, J.E.; Halcox, J.P.; Rabelink, T.J. Endothelial function and dysfunction: Testing and clinical relevance. Circulation 2007, 115, 1285–1295. [Google Scholar] [CrossRef]
- Rajendran, P.; Rengarajan, T.; Thangavel, J.; Nishigaki, Y.; Sakthisekaran, D.; Sethi, G.; Nishigaki, I. The vascular endothelium and human diseases. Int. J. Biol. Sci. 2013, 9, 1057–1069. [Google Scholar] [CrossRef]
- Forstermann, U.; Munzel, T. Endothelial nitric oxide synthase in vascular disease: From marvel to menace. Circulation 2006, 113, 1708–1714. [Google Scholar] [CrossRef]
- Somlyo, A.P.; Somlyo, A.V. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: Modulated by G proteins, kinases, and myosin phosphatase. Physiol. Rev. 2003, 83, 1325–1358. [Google Scholar] [CrossRef]
- Kim, J.A.; Montagnani, M.; Koh, K.K.; Quon, M.J. Reciprocal relationships between insulin resistance and endothelial dysfunction—Molecular and pathophysiological mechanisms. Circulation 2006, 113, 1888–1904. [Google Scholar] [CrossRef]
- Xu, J.; Zou, M.H. Molecular insights and therapeutic targets for diabetic endothelial dysfunction. Circulation 2009, 120, 1266–1286. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.M.; Lin, P.H.; Yao, Q.; Chen, C. Chemical and molecular mechanisms of antioxidants: Experimental approaches and model systems. J. Cell. Mol. Med. 2010, 14, 840–860. [Google Scholar] [CrossRef] [PubMed]
- Janssen-Heininger, Y.M.W.; Mossman, B.T.; Heintz, N.H.; Forman, H.J.; Kalyanaraman, B.; Finkel, T.; Stamler, J.S.; Rhee, S.G.; van der Vliet, A. Redox-based regulation of signal transduction: Principles, pitfalls, and promises. Free Radic. Biol. Med. 2008, 45, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Poli, G.; Leonarduzzi, G.; Biasi, F.; Chiarpotto, E. Oxidative stress and cell signalling. Curr. Med. Chem. 2004, 11, 1163–1182. [Google Scholar] [CrossRef]
- Brieger, K.; Schiavone, S.; Miller, F.J., Jr.; Krause, K.H. Reactive oxygen species: From health to disease. Swiss Med. Wkly. 2012, 142, w13659. [Google Scholar] [CrossRef]
- Higuchi, S.; Ohtsu, H.; Suzuki, H.; Shirai, H.; Frank, G.D.; Eguchi, S. Angiotensin II signal transduction through the AT(I) receptor: Novel insig hts into mechanisms and pathophysiology. Clin. Sci. 2007, 112, 417–428. [Google Scholar] [CrossRef]
- Wassmann, S.; Wassmann, K.; Nickenig, G. Modulation of oxidant and antioxidant enzyme expression and function in vascular cells. Hypertension 2004, 44, 381–386. [Google Scholar] [CrossRef]
- Szabo, C. Role of nitrosative stress in the pathogenesis of diabetic vascular dysfunction. Br. J. Pharm. 2009, 156, 713–727. [Google Scholar] [CrossRef]
- Vona, R.; Gambardella, L.; Cittadini, C.; Straface, E.; Pietraforte, D. Biomarkers of Oxidative Stress in Metabolic Syndrome and Associated Diseases. Oxid. Med. Cell. Longev. 2019. [Google Scholar] [CrossRef]
- Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity: Implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes. Res. Clin. Pract. 2013, 7, E330–E341. [Google Scholar] [CrossRef] [PubMed]
- Otani, H. Oxidative Stress as Pathogenesis of Cardiovascular Risk Associated with Metabolic Syndrome. Antioxid. Redox Signal. 2011, 15, 1911–1926. [Google Scholar] [CrossRef] [PubMed]
- Kayama, Y.; Raaz, U.; Jagger, A.; Adam, M.; Schellinger, I.N.; Sakamoto, M.; Suzuki, H.; Toyama, K.; Spin, J.M.; Tsao, P.S. Diabetic Cardiovascular Disease Induced by Oxidative Stress. Int. J. Mol. Sci. 2015, 16, 25234–25263. [Google Scholar] [CrossRef] [PubMed]
- Spahis, S.; Borys, J.M.; Levy, E. Metabolic Syndrome as a Multifaceted Risk Factor for Oxidative Stress. Antioxid. Redox Signal. 2017, 26, 445–461. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, M.; Khemka, V.K.; Chatterjee, G.; Ganguly, A.; Mukhopadhyay, S.; Chakrabarti, S. Enhanced ROS production and oxidative damage in subcutaneous white adipose tissue mitochondria in obese and type 2 diabetes subjects. Mol. Cell. Biochem. 2015, 399, 95–103. [Google Scholar] [CrossRef]
- Anderson, E.J.; Lustig, M.E.; Boyle, K.E.; Woodlief, T.L.; Kane, D.A.; Lin, C.T.; Price, J.W.; Kang, L.; Rabinovitch, P.S.; Szeto, H.H.; et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J. Clin. Investig. 2009, 119, 573–581. [Google Scholar] [CrossRef]
- Chowdhury, S.K.R.; Sangle, G.V.; Xie, X.P.; Stelmack, G.L.; Halayko, A.J.; Shen, G.X. Effects of extensively oxidized low-density lipoprotein on mitochondrial function and reactive oxygen species in porcine aortic endothelial cells. Am. J. Physiol. Endocrinol. Matab. 2010, 298, E89–E98. [Google Scholar] [CrossRef]
- Holvoet, P.; Kritchevsky, S.B.; Tracy, R.P.; Mertens, A.; Rubin, S.M.; Butler, J.; Goodpaster, B.; Harris, T.B. The metabolic syndrome, circulating oxidized LDL, and risk of myocardial infarction in well-functioning elderly people in the health, aging, and body composition cohort. Diabetes 2004, 53, 1068–1073. [Google Scholar] [CrossRef]
- Rodrigo, R.; Prat, H.; Passalacqua, W.; Araya, J.; Bachler, J.P. Decrease in oxidative stress through supplementation of vitamins C and E is associated with a reduction in blood pressure in patients with essential hypertension. Clin. Sci. 2008, 114, 625–634. [Google Scholar] [CrossRef]
- Virdis, A.; Neves, M.F.; Amiri, F.; Touyz, R.M.; Schiffrin, E.L. Role of NAD(P)H oxidase on vascular alterations in angiotensin II-infused mice. J. Hypertens. 2004, 22, 535–542. [Google Scholar] [CrossRef]
- Ardalan, M.R.; Rafieian-Kopaei, M. Antioxidant supplementation in hypertension. J. Ren. Inj. Prev. 2014, 3, 39–40. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Hosseini, M.S.; Khalili, N.; Naimi, E.; Majeed, M.; Sahebkar, A. Antioxidant and anti-inflammatory effects of curcuminoid-piperine combination in subjects with metabolic syndrome: A randomized controlled trial and an updated meta-analysis. Clin. Nutr. 2015, 34, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Stull, A.J.; Cash, K.C.; Champagne, C.M.; Gupta, A.K.; Boston, R.; Beyl, R.A.; Johnson, W.D.; Cefalu, W.T. Blueberries improve endothelial function, but not blood pressure, in adults with metabolic syndrome: A randomized, double-blind, placebo-controlled clinical trial. Nutrients 2015, 7, 4107–4123. [Google Scholar] [CrossRef]
- Silveira, J.Q.; Dourado, G.K.; Cesar, T.B. Red-fleshed sweet orange juice improves the risk factors for metabolic syndrome. Int. J. Food Sci. Nutr. 2015, 66, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Huerta, O.D.; Aguilera, C.M.; Martin, M.V.; Soto, M.J.; Rico, M.C.; Vallejo, F.; Tomas-Barberan, F.; Perez-de-la-Cruz, A.J.; Gil, A.; Mesa, M.D. Normal or High Polyphenol Concentration in Orange Juice Affects Antioxidant Activity, Blood Pressure, and Body Weight in Obese or Overweight Adults. J. Nutr. 2015, 145, 1808–1816. [Google Scholar] [CrossRef]
- Urquiaga, I.; D’Acuna, S.; Perez, D.; Dicenta, S.; Echeverria, G.; Rigotti, A.; Leighton, F. Wine grape pomace flour improves blood pressure, fasting glucose and protein damage in humans: A randomized controlled trial. Biol. Res. 2015, 48, 49. [Google Scholar] [CrossRef]
- Sprague, A.H.; Khalil, R.A. Inflammatory cytokines in vascular dysfunction and vascular disease. Biochem. Pharm. 2009, 78, 539–552. [Google Scholar] [CrossRef]
- Pober, J.S.; Sessa, W.C. Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol. 2007, 7, 803–815. [Google Scholar] [CrossRef]
- Fiorucci, S.; Distrutti, E.; Mencarelli, A.; Rizzo, G.; Di Lorenzo, A.R.; Baldoni, M.; del Soldato, P.; Morelli, A.; Wallace, J.L. Cooperation between aspirin-triggered lipoxin and nitric oxide (NO) mediates antiadhesive properties of 2-(acetyloxy)benzoic acid 3-(nitrooxymethyl)phenyl ester (NCX-4016) (NO-aspirin) on neutrophil-endothelial cell adherence (vol 309, pg 1174, 2008). J. Pharmacol. Exp. Ther. 2009, 329, 848. [Google Scholar]
- Stefano, G.B.; Challenger, S.; Kream, R. Hyperglycemia-associated alterations in cellular signaling and dysregulated mitochondrial bioenergetics in human metabolic disorders. Eur. J. Nutr. 2016, 55, 2339–2345. [Google Scholar] [CrossRef]
- Akoumianakis, I.; Antoniades, C. Impaired Vascular Redox Signaling in the Vascular Complications of Obesity and Diabetes Mellitus. Antioxid. Redox Signal. 2019, 30, 333–353. [Google Scholar] [CrossRef] [PubMed]
- Lassegue, B.; Griendling, K.K. NADPH Oxidases: Functions and Pathologies in the Vasculature. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Marfella, R.; Ciotola, M.; Di Palo, C.; Giugliano, F.; Giugliano, G.; D’Armiento, M.; D’Andrea, F.; Giugliano, D. Effect of a Mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome—A randomized trial. JAMA J. Am. Med. Assoc. 2004, 292, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Han, T.S.; Sattar, N.; Williams, K.; Gonzalez-Villalpando, C.; Lean, M.E.; Haffner, S.M. Prospective study of C-reactive protein in relation to the development of diabetes and metabolic syndrome in the Mexico City Diabetes Study. Diabetes Care 2002, 25, 2016–2021. [Google Scholar] [CrossRef] [PubMed]
- Mirhafez, S.R.; Ebrahimi, M.; Karimian, M.S.; Avan, A.; Tayefi, M.; Heidari-Bakavoli, A.; Parizadeh, M.R.; Moohebati, M.; Azarpazhooh, M.R.; Esmaily, H.; et al. Serum high-sensitivity C-reactive protein as a biomarker in patients with metabolic syndrome: Evidence-based study with 7284 subjects. Eur. J. Clin. Nutr. 2016, 70, 1298–1304. [Google Scholar] [CrossRef]
- Mazidi, M.; Toth, P.P.; Banach, M. C-reactive Protein Is Associated With Prevalence of the Metabolic Syndrome, Hypertension, and Diabetes Mellitus in US Adults. Angiology 2018, 69, 438–442. [Google Scholar] [CrossRef]
- Li, Y.W.; Zhong, X.M.; Cheng, G.C.; Zhao, C.H.; Zhang, L.; Hong, Y.; Wan, Q.L.; He, R.L.; Wang, Z.Z. Hs-CRP and all-cause, cardiovascular, and cancer mortality risk: A meta-analysis. Atherosclerosis 2017, 259, 75–82. [Google Scholar] [CrossRef]
- Ferrannini, E.; Buzzigoli, G.; Bonadonna, R.; Giorico, M.A.; Oleggini, M.; Graziadei, L.; Pedrinelli, R.; Brandi, L.; Bevilacqua, S. Insulin Resistance in Essential-Hypertension. N. Engl. J. Med. 1987, 317, 350–357. [Google Scholar] [CrossRef]
- Park, S.E.; Rhee, E.J.; Park, C.Y.; Oh, K.W.; Park, S.W.; Kim, S.W.; Lee, W.Y. Impact of hyperinsulinemia on the development of hypertension in normotensive, nondiabetic adults: A 4-year follow-up study. Metab. Clin. Exp. 2013, 62, 532–538. [Google Scholar] [CrossRef]
- Paul, M.; Poyan Mehr, A.; Kreutz, R. Physiology of local renin-angiotensin systems. Physiol. Rev. 2006, 86, 747–803. [Google Scholar] [CrossRef] [PubMed]
- Olivares-Reyes, J.A.; Arellano-Plancarte, A.; Castillo-Hernandez, J.R. Angiotensin II and the development of insulin resistance: Implications for diabetes. Mol. Cell. Endocrinol. 2009, 302, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.A.; Whaley-Connell, A.; Habibi, J.; Wei, Y.Z.; Lastra, G.; Manrique, C.; Stas, S.; Sowers, J.R. Renin-angiotensin-aldosterone system and oxidative stress in cardiovascular insulin resistance. Am. J. Physiol. Heart C 2007, 293, H2009–H2023. [Google Scholar] [CrossRef] [PubMed]
- Samuelsson, A.M.; Bollano, E.; Mobini, R.; Larsson, B.M.; Omerovic, E.; Fu, M.; Waagstein, F.; Holmang, A. Hyperinsulinemia: Effect on cardiac mass/function, angiotensin II receptor expression, and insulin signaling pathways. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H787–H796. [Google Scholar] [CrossRef]
- Shinozaki, K.; Ayajiki, K.; Nishio, Y.; Sugaya, T.; Kashiwagi, A.; Okamura, T. Evidence for a causal role of the renin-angiotensin system in vascular dysfunction associated with insulin resistance. Hypertension 2004, 43, 255–262. [Google Scholar] [CrossRef]
- Hsueh, W.A.; Wyne, K. Renin-Angiotensin-aldosterone system in diabetes and hypertension. J. Clin. Hypertens. (Greenwich) 2011, 13, 224–237. [Google Scholar] [CrossRef]
- Santos, S.H.; Braga, J.F.; Mario, E.G.; Porto, L.C.; Rodrigues-Machado Mda, G.; Murari, A.; Botion, L.M.; Alenina, N.; Bader, M.; Santos, R.A. Improved lipid and glucose metabolism in transgenic rats with increased circulating angiotensin-(1-7). Arterioscler. Thromb. Vasc. Biol. 2010, 30, 953–961. [Google Scholar] [CrossRef]
- Liu, C.; Lv, X.H.; Li, H.X.; Cao, X.; Zhang, F.; Wang, L.; Yu, M.; Yang, J.K. Angiotensin-(1-7) suppresses oxidative stress and improves glucose uptake via Mas receptor in adipocytes. Acta Diabetol. 2012, 49, 291–299. [Google Scholar] [CrossRef]
- Giani, J.F.; Mayer, M.A.; Munoz, M.C.; Silberman, E.A.; Hocht, C.; Taira, C.A.; Gironacci, M.M.; Turyn, D.; Dominici, F.P. Chronic infusion of angiotensin-(1-7) improves insulin resistance and hypertension induced by a high-fructose diet in rats. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E262–E271. [Google Scholar] [CrossRef]
- Chou, C.L.; Pang, C.Y.; Lee, T.J.; Fang, T.C. Direct renin inhibitor prevents and ameliorates insulin resistance, aortic endothelial dysfunction and vascular remodeling in fructose-fed hypertensive rats. Hypertens. Res. 2013, 36, 123–128. [Google Scholar] [CrossRef]
- Chou, C.L.; Lai, Y.H.; Lin, T.Y.; Lee, T.J.; Fang, T.C. Aliskiren prevents and ameliorates metabolic syndrome in fructose-fed rats. Arch. Med. Sci. AMS 2011, 7, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Fogari, R.; Zoppi, A.; Mugellini, A.; Lazzari, P.; Derosa, G. Different effects of aliskiren and losartan on fibrinolysis and insulin sensitivity in hypertensive patients with metabolic syndrome. Horm. Metab. Res. 2010, 42, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, N.M. Management of hypertension in patients with type 2 diabetes mellitus: Guidelines based on current evidence. Ann. Intern. Med. 2001, 135, 1079–1083. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J. Renin-angiotensin system inhibition prevents type 2 diabetes mellitus—Part 2. Overview of physiological and biochemical mechanisms. Diabetes Metab. 2004, 30, 498–505. [Google Scholar] [CrossRef]
- Sowers, J.R. Insulin resistance and hypertension. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H1597–H1602. [Google Scholar] [CrossRef] [PubMed]
- Engeli, S.; Boschmann, M.; Frings, P.; Beck, L.; Janke, J.; Titze, J.; Luft, F.C.; Heer, M.; Jordan, J. Influence of salt intake on renin-angiotensin and natriuretic peptide system genes in human adipose tissue. Hypertension 2006, 48, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J. Prevention of type 2 diabetes mellitus through inhibition of the Renin-Angiotensin system. Drugs 2004, 64, 2537–2565. [Google Scholar] [CrossRef]
- Lindholm, L.H.; Ibsen, H.; Borch-Johnsen, K.; Olsen, M.H.; Wachtell, K.; Dahlof, B.; Devereux, R.B.; Beevers, G.; de Faire, U.; Fyhrquist, F.; et al. Risk of new-onset diabetes in the Losartan Intervention For Endpoint reduction in hypertension study. J. Hypertens. 2002, 20, 1879–1886. [Google Scholar] [CrossRef]
- Hansson, L.; Lindholm, L.H.; Niskanen, L.; Lanke, J.; Hedner, T.; Niklason, A.; Luomanmaki, K.; Dahlof, B.; de Faire, U.; Morlin, C.; et al. Effect of angiotensin-converting-enzyme inhibition compared with conventional therapy on cardiovascular morbidity and mortality in hypertension: The Captopril Prevention Project (CAPPP) randomised trial. Lancet 1999, 353, 611–616. [Google Scholar] [CrossRef]
- Henriksen, E.J. Improvement of insulin sensitivity by antagonism of the renin-angiotensin system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R974–R980. [Google Scholar] [CrossRef]
- Zappe, D.H.; Sowers, J.R.; Hsueh, W.A.; Haffner, S.M.; Deedwania, P.C.; Fonseca, V.A.; Keeling, L.; Sica, D.A. Metabolic and antihypertensive effects of combined angiotensin receptor blocker and diuretic therapy in prediabetic hypertensive patients with the cardiometabolic syndrome. J. Clin. Hypertens. (Greenwich) 2008, 10, 894–903. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.E. The role of the renin-angiotensin-aldosterone system in diabetes and its vascular complications. Am. J. Hypertens. 2004, 17, 16S–20S, quiz A12-14. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Deedwania, P.C. The renin angiotensin system as a therapeutic target to prevent diabetes and its complications. Cardiol. Clin. 2005, 23, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Segura, J.; Ruilope, L.M. Obesity, essential hypertension and renin-angiotensin system. Public Health Nutr. 2007, 10, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.M.; Engeli, S. The role of renin-angiotensin system blockade in the management of hypertension associated with the cardiometabolic syndrome. J. Cardiometab. Syndr. 2006, 1, 29–35. [Google Scholar] [CrossRef]
- Yim, H.E.; Yoo, K.H. Renin-Angiotensin system—Considerations for hypertension and kidney. Electrolyte Blood Press. 2008, 6, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Giacchetti, G.; Faloia, E.; Mariniello, B.; Sardu, C.; Gatti, C.; Camilloni, M.A.; Guerrieri, M.; Mantero, F. Overexpression of the renin-angiotensin system in human visceral adipose tissue in normal and overweight subjects. Am. J. Hypertens. 2002, 15, 381–388. [Google Scholar] [CrossRef]
- Yasue, S.; Masuzaki, H.; Okada, S.; Ishii, T.; Kozuka, C.; Tanaka, T.; Fujikura, J.; Ebihara, K.; Hosoda, K.; Katsurada, A.; et al. Adipose tissue-specific regulation of angiotensinogen in obese humans and mice: Impact of nutritional status and adipocyte hypertrophy. Am. J. Hypertens. 2010, 23, 425–431. [Google Scholar] [CrossRef]
- Underwood, P.C.; Adler, G.K. The renin angiotensin aldosterone system and insulin resistance in humans. Curr. Hypertens. Rep. 2013, 15, 59–70. [Google Scholar] [CrossRef]
- Sarzani, R.; Marcucci, P.; Salvi, F.; Bordicchia, M.; Espinosa, E.; Mucci, L.; Lorenzetti, B.; Minardi, D.; Muzzonigro, G.; Dessi-Fulgheri, P.; et al. Angiotensin II stimulates and atrial natriuretic peptide inhibits human visceral adipocyte growth. Int. J. Obes. 2008, 32, 259–267. [Google Scholar] [CrossRef][Green Version]
- Lee, M.H.; Song, H.K.; Ko, G.J.; Kang, Y.S.; Han, S.Y.; Han, K.H.; Kim, H.K.; Han, J.Y.; Cha, D.R. Angiotensin receptor blockers improve insulin resistance in type 2 diabetic rats by modulating adipose tissue. Kidney Int. 2008, 74, 890–900. [Google Scholar] [CrossRef]
- Henriksen, E.J.; Prasannarong, M. The role of the renin-angiotensin system in the development of insulin resistance in skeletal muscle. Mol. Cell. Endocrinol. 2013, 378, 15–22. [Google Scholar] [CrossRef]
- Cavazos, A.; de Mejia, E.G. Identification of Bioactive Peptides from Cereal Storage Proteins and Their Potential Role in Prevention of Chronic Diseases. Compr. Rev. Food Sci. F 2013, 12, 364–380. [Google Scholar] [CrossRef]
- Putnam, K.; Shoemaker, R.; Yiannikouris, F.; Cassis, L.A. The renin-angiotensin system: A target of and contributor to dyslipidemias, altered glucose homeostasis, and hypertension of the metabolic syndrome. Am. J. Physiol. Heart C 2012, 302, H1219–H1230. [Google Scholar] [CrossRef]
- Marcone, S.; Belton, O.; Fitzgerald, D.J. Milk-derived bioactive peptides and their health promoting effects: A potential role in atherosclerosis. Br. J. Clin. Pharm. 2017, 83, 152–162. [Google Scholar] [CrossRef]
- Wu, J.; Jahandideh, F.; Yu, W.; Majumder, K. Bioactive Peptides from Meat Proteins as Functional Food Components. In Functional Polymers in Food Science; Cirillo, G., Gianfranco Spizzirri, U., Lemma, F., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 181–208. [Google Scholar]
- De Campos Zani, S.; Wu, J.; Chan, B.C. Egg and Soy-Derived Peptides and Hydrolysates: A Review of Their Physiological Actions against Diabetes and Obesity. Nutrients 2018, 10, 549. [Google Scholar] [CrossRef]
- Korhonen, H.; Pihlanto, A. Bioactive peptides: Production and functionality. Int. Dairy J. 2006, 16, 945–960. [Google Scholar] [CrossRef]
- Dia, V.P.; de Mejia, E.G. Mode of administration affected the capability of soybean-derived peptide lunasin to prevent metastasis of human colon cancer cells in a mouse model. FASEB J. 2013, 27, 863. [Google Scholar]
- Liu, J.; Jia, S.H.; Kirberger, M.; Chen, N. Lunasin as a promising health-beneficial peptide. Eur. Rev. Med. Pharm. 2014, 18, 2070–2075. [Google Scholar]
- Singh, B.P.; Vij, S.; Hati, S. Functional significance of bioactive peptides derived from soybean. Peptides 2014, 54, 171–179. [Google Scholar] [CrossRef]
- Guang, C.; Phillips, R.D. Plant Food-Derived Angiotensin I Converting Enzyme Inhibitory Peptides. J. Agric. Food Chem. 2009, 57, 5113–5120. [Google Scholar] [CrossRef]
- Iwaniak, A.; Minkiewicz, P.; Darewicz, M. Food-Originating ACE Inhibitors, Including Antihypertensive Peptides, as Preventive Food Components in Blood Pressure Reduction. Compr. Rev. Food Sci. F 2014, 13, 114–134. [Google Scholar] [CrossRef]
- Kim, S.M.; Kim, J.Y.; Choue, R.W. Effects of IPP and VPP Supplementation on Blood Pressure, ACE Activity and NO Production in Spontaneously Hypertensive Rats. FASEB J. 2008, 22, 1112. [Google Scholar]
- Aihara, K.; Osaka, M.; Yoshida, M. Oral administration of the milk casein-derived tripeptide Val-Pro-Pro attenuates high-fat diet-induced adipose tissue inflammation in mice. Br. J. Nutr. 2014, 112, 513–519. [Google Scholar] [CrossRef]
- Nonaka, A.; Nakamura, T.; Hirota, T.; Matsushita, A.; Asakura, M.; Ohki, K.; Kitakaze, M. The milk-derived peptides Val-Pro-Pro and Ile-Pro-Pro attenuate arterial dysfunction in L-NAME-treated rats. Hypertens. Res. 2014, 37, 703–707. [Google Scholar] [CrossRef]
- Solieri, L.; Rutella, G.S.; Tagliazucchi, D. Impact of non-starter lactobacilli on release of peptides with angiotensin-converting enzyme inhibitory and antioxidant activities during bovine milk fermentation. Food Microbiol. 2015, 51, 108–116. [Google Scholar] [CrossRef]
- Aihara, K.; Kajimoto, O.; Hirata, H.; Takahashi, R.; Nakamura, Y. Effect of powdered fermented milk with Lactobacillus helveticus on subjects with high-normal blood pressure or mild hypertension. J. Am. Coll. Nutr. 2005, 24, 257–265. [Google Scholar] [CrossRef]
- Engberink, M.F.; Schouten, E.G.; Kok, F.J.; van Mierlo, L.A.J.; Brouwer, I.A.; Geleijnse, J.M. Lactotripeptides show no effect on human blood pressure—Results from a double-blind randomized controlled trial. Hypertension 2008, 51, 399–405. [Google Scholar] [CrossRef]
- Boelsma, E.; Kloek, J. IPP-rich milk protein hydrolysate lowers blood pressure in subjects with stage 1 hypertension, a randomized controlled trial. Nutr. J. 2010, 9, 52. [Google Scholar] [CrossRef]
- Chanson-Rolle, A.; Aubin, F.; Braesco, V.; Hamasaki, T.; Kitakaze, M. Influence of the Lactotripeptides Isoleucine-Proline-Proline and Valine-Proline-Proline on Systolic Blood Pressure in Japanese Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2015, 10, e0142235. [Google Scholar] [CrossRef]
- Nakamura, T.; Hirota, T.; Mizushima, K.; Ohki, K.; Naito, Y.; Yamamoto, N.; Yoshikawa, T. Milk-Derived Peptides, Val-Pro-Pro and Ile-Pro-Pro, Attenuate Atherosclerosis Development in Apolipoprotein E-Deficient Mice: A Preliminary Study. J. Med. Food 2013, 16, 396–403. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Wu, J.P. Milk-Derived Tripeptides IPP (Ile-Pro-Pro) and VPP (Val-Pro-Pro) Promote Adipocyte Differentiation and Inhibit Inflammation in 3T3-F442A Cells. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Miguel, M.; Aleixandre, A. Antihypertensive peptides derived from egg proteins. J. Nutr. 2006, 136, 1457–1460. [Google Scholar] [CrossRef]
- Miguel, M.; Alonso, M.J.; Salaices, M.; Aleixandre, A.; Lopez-Fandino, R. Antihypertensive, ACE-inhibitory and vasodilator properties of an egg white hydrolysate: Effect of a simulated intestinal digestion. Food Chem. 2007, 104, 163–168. [Google Scholar] [CrossRef]
- Jahandideh, F.; Majumder, K.; Chakrabarti, S.; Morton, J.S.; Panahi, S.; Kaufman, S.; Davidge, S.T.; Wu, J.P. Beneficial Effects of Simulated Gastro-Intestinal Digests of Fried Egg and Its Fractions on Blood Pressure, Plasma Lipids and Oxidative Stress in Spontaneously Hypertensive Rats. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Jahandideh, F.; Chakrabarti, S.; Majumder, K.; Li, Q.; Panahi, S.; Morton, J.S.; Davidge, S.T.; Wu, J. Egg white protein hydrolysate reduces blood pressure, improves vascular relaxation and modifies aortic angiotensin II receptors expression in spontaneously hypertensive rats. J. Funct. Foods 2016, 27, 667–673. [Google Scholar] [CrossRef]
- Yu, Z.P.; Zhao, W.Z.; Ding, L.; Wang, Y.Q.; Chen, F.; Liu, J.B. Short- and long-term antihypertensive effect of egg protein-derived peptide QIGLF. J. Sci. Food Agric. 2017, 97, 551–555. [Google Scholar] [CrossRef]
- Jahandideh, F.; Chakrabarti, S.; Davidge, S.T.; Wu, J. Egg white hydrolysate shows insulin mimetic and sensitizing effects in 3T3-F442A pre-adipocytes. PLoS ONE 2017, 12, e0185653. [Google Scholar] [CrossRef]
- Son, M.; Wu, J. Egg white hydrolysate and peptide reverse insulin resistance associated with tumor necrosis factor-alpha (TNF-alpha) stimulated mitogen-activated protein kinase (MAPK) pathway in skeletal muscle cells. Eur. J. Nutr. 2019, 58, 1961–1969. [Google Scholar] [CrossRef]
- Jahandideh, F.; de Campos Zani, S.; Son, M.; Proctor, S.D.; Davidge, S.T.; Chan, C.B.; Wu, J. Egg white hydrolysate enhances insulin sensitivity in high-fat diet-induced insulin-resistant rats via Akt activation. Br. J. Nutr. 2019, 122, 14–24. [Google Scholar] [CrossRef]
- Jahandideh, F.; Liu, P.; Wu, J. Purification and identification of adipogenic-differentiating peptides from egg white hydrolysate. Food Chem. 2018, 259, 25–30. [Google Scholar] [CrossRef]
- Majumder, K.; Wu, J.P. Purification and characterisation of angiotensin I converting enzyme (ACE) inhibitory peptides derived from enzymatic hydrolysate of ovotransferrin. Food Chem. 2011, 126, 1614–1619. [Google Scholar] [CrossRef]
- Majumder, K.; Chakrabarti, S.; Morton, J.S.; Panahi, S.; Kaufman, S.; Davidge, S.T.; Wu, J.P. Egg-Derived Tri-Peptide IRW Exerts Antihypertensive Effects in Spontaneously Hypertensive Rats. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Majumder, K.; Chakrabarti, S.; Morton, J.S.; Panahi, S.; Kaufman, S.; Davidge, S.T.; Wu, J.P. Egg-derived ACE-inhibitory peptides IQW and LKP reduce blood pressure in spontaneously hypertensive rats. J. Funct. Foods 2015, 13, 50–60. [Google Scholar] [CrossRef]
- Majumder, K.; Chakrabarti, S.; Davidge, S.T.; Wu, J.P. Structure and Activity Study of Egg Protein Ovotransferrin Derived Peptides (IRW and IQW) on Endothelial Inflammatory Response and Oxidative Stress. J. Agric. Food Chem. 2013, 61, 2120–2129. [Google Scholar] [CrossRef]
- Huang, W.Y.; Majumder, K.; Wu, J. Oxygen radical absorbance capacity of peptides from egg white protein ovotransferrin and their interaction with phytochemicals. Food Chem. 2010, 123, 635–641. [Google Scholar] [CrossRef]
- Liao, W.; Bhullar, K.S.; Chakrabarti, S.; Davidge, S.T.; Wu, J. Egg White-Derived Tripeptide IRW (Ile-Arg-Trp) Is an Activator of Angiotensin Converting Enzyme 2. J. Agric. Food Chem. 2018, 66, 11330–11336. [Google Scholar] [CrossRef]
- Son, M.; Chan, C.B.; Wu, J. Egg White Ovotransferrin-Derived ACE Inhibitory Peptide Ameliorates Angiotensin II-Stimulated Insulin Resistance in Skeletal Muscle Cells. Mol. Nutr. Food Res. 2018, 62. [Google Scholar] [CrossRef]
- Jahandideh, F.; Chakrabarti, S.; Davidge, S.T.; Wu, J.P. Antioxidant Peptides Identified from Ovotransferrin by the ORAC Method Did Not Show Anti-Inflammatory and Antioxidant Activities in Endothelial Cells. J. Agric. Food Chem. 2016, 64, 113–119. [Google Scholar] [CrossRef]
- Park, K.H.; Nan, Y.H.; Park, Y.; Kim, J.I.; Park, I.S.; Hahm, K.S.; Shin, S.Y. Cell specificity, anti-inflammatory activity, and plausible bactericidal mechanism of designed Trp-rich model antimicrobial peptides. BBA-Biomembr. 2009, 1788, 1193–1203. [Google Scholar] [CrossRef]
- Ruiz-Ramirez, A.; Ortiz-Balderas, E.; Gardozo-Saldana, G.; Diaz-Diaz, E.; El-Hafidi, M. Glycine restores glutathione and protects against oxidative stress in vascular tissue from sucrose-fed rats. Clin. Sci. 2014, 126, 19–29. [Google Scholar] [CrossRef]
- Jaworek, J.; Leja-Szpak, A.; Bonior, J.; Nawrot, K.; Tomaszewska, R.; Stachura, J.; Sendur, R.; Pawlik, W.; Brzozowski, T.; Konturek, S.J. Protective effect of melatonin and its precursor L-tryptophan on acute pancreatitis induced by caerulein overstimulation or ischemia/reperfusion. J. Pineal Res. 2003, 34, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.M.; Landheer, S.; van Gilst, W.H.; van Amerongen, A.; Hammes, H.P.; Henning, R.H.; Deelman, L.E.; Buikema, H. Attenuation of Renovascular Damage in Zucker Diabetic Fatty Rat by NWT-03, an Egg Protein Hydrolysate with ACE- and DPP4-Inhibitory Activity. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Garces-Rimon, M.; Lopez-Exposito, I.; Lopez-Fandino, R.; Miguel, M. Egg white hydrolysates with in vitro biological multiactivities to control complications associated with the metabolic syndrome. Eur. Food Res. Technol. 2016, 242, 61–69. [Google Scholar] [CrossRef]
- Garces-Rimon, M.; Gonzalez, C.; Uranga, J.A.; Lopez-Miranda, V.; Lopez-Fandino, R.; Miguel, M. Pepsin Egg White Hydrolysate Ameliorates Obesity-Related Oxidative Stress, Inflammation and Steatosis in Zucker Fatty Rats. PLoS ONE 2016, 11. [Google Scholar] [CrossRef]
- Zambrowicz, A.; Eckert, E.; Pokora, M.; Bobak, L.; Dabrowska, A.; Szoltysik, M.; Trziszka, T.; Chrzanowska, J. Antioxidant and antidiabetic activities of peptides isolated from a hydrolysate of an egg-yolk protein by-product prepared with a proteinase from Asian pumpkin (Cucurbita ficifolia). RSC Adv. 2015, 5, 10460–10467. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Ng, T.B.; Wong, J.H. Marine Peptides: Bioactivities and Applications. Mar. Drugs 2015, 13, 4006–4043. [Google Scholar] [CrossRef]
- Manikkam, V.; Vasiljevic, T.; Donkor, O.N.; Mathai, M.L. A Review of Potential Marine-derived Hypotensive and Anti-obesity Peptides. Crit. Rev. Food Sci. Nutr. 2016, 56, 92–112. [Google Scholar] [CrossRef]
- Ovchinnikova, T.V. Structure, Function, and Therapeutic Potential of Marine Bioactive Peptides. Mar. Drugs 2019, 17, 505. [Google Scholar] [CrossRef]
- Ngo, D.H.; Ryu, B.; Vo, T.S.; Himaya, S.W.A.; Wijesekara, I.; Kim, S.K. Free radical scavenging and angiotensin-I converting enzyme inhibitory peptides from Pacific cod (Gadus macrocephalus) skin gelatin. Int. J. Biol. Macromol. 2011, 49, 1110–1116. [Google Scholar] [CrossRef]
- Himaya, S.W.A.; Ngo, D.H.; Ryu, B.; Kim, S.K. An active peptide purified from gastrointestinal enzyme hydrolysate of Pacific cod skin gelatin attenuates angiotensin-1 converting enzyme (ACE) activity and cellular oxidative stress. Food Chem. 2012, 132, 1872–1882. [Google Scholar] [CrossRef]
- Ngo, D.H.; Ryu, B.; Kim, S.K. Active peptides from skate (Okamejei kenojei) skin gelatin diminish angiotensin-I converting enzyme activity and intracellular free radical-mediated oxidation. Food Chem. 2014, 143, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Otani, L.; Ninomiya, T.; Murakami, M.; Osajima, K.; Kato, H.; Murakami, T. Sardine peptide with angiotensin I-converting enzyme inhibitory activity improves glucose tolerance in stroke-prone spontaneously hypertensive rats. Biosci. Biotechnol. Biochem. 2009, 73, 2203–2209. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Seki, E.; Osajima, K.; Yoshida, M.; Asada, K.; Matsui, T.; Osajima, Y. Antihypertensive effect of valyl-tyrosine, a short chain peptide derived from sardine muscle hydrolyzate, on mild hypertensive subjects. J. Hum. Hypertens. 2000, 14, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Erdmann, K.; Grosser, N.; Schipporeit, K.; Schroder, H. The ACE inhibitory dipeptide Met-Tyr diminishes free radical formation in human endothelial cells via induction of heme oxygenase-1 and ferritin. J. Nutr. 2006, 136, 2148–2152. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.F.; Peng, H.B.; Liu, G.Q.; Zhang, F.; Li, Y. Beneficial effects of oligopeptides from marine salmon skin in a rat model of type 2 diabetes. Nutrition 2010, 26, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.S.; Han, L.W.; Shi, Y.P.; Li, X.B.; Zhang, X.M.; Hou, H.R.; Lin, H.W.; Liu, K.C. Two Novel Multi-Functional Peptides from Meat and Visceral Mass of Marine Snail Neptunea arthritica cumingii and Their Activities In Vitro and In Vivo. Mar. Drugs 2018, 16, 473. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gong, F.; Chen, M.F.; Li, C.; Hong, P.; Sun, S.; Zhou, C.; Qian, Z.J. In Vitro Vascular-Protective Effects of a Tilapia By-Product Oligopeptide on Angiotensin II-Induced Hypertensive Endothelial Injury in HUVEC by Nrf2/NF-kappaB Pathways. Mar. Drugs 2019, 17, 431. [Google Scholar] [CrossRef]
- Girgih, A.T.; Alashi, A.M.; He, R.; Malomo, S.A.; Raj, P.; Netticadan, T.; Aluko, R.E. A Novel Hemp Seed Meal Protein Hydrolysate Reduces Oxidative Stress Factors in Spontaneously Hypertensive Rats. Nutrients 2014, 6, 5652–5666. [Google Scholar] [CrossRef]
- Majumder, K.; Liang, G.X.; Chen, Y.H.; Guan, L.L.; Davidge, S.T.; Wu, J.P. Egg ovotransferrin-derived ACE inhibitory peptide IRW increases ACE2 but decreases proinflammatory genes expression in mesenteric artery of spontaneously hypertensive rats. Mol. Nutr. Food Res. 2015, 59, 1735–1744. [Google Scholar] [CrossRef]
- Huang, W.Y.; Chakrabarti, S.; Majumder, K.; Jiang, Y.Y.; Davidge, S.T.; Wu, J.P. Egg-Derived Peptide IRW Inhibits TNF-alpha-Induced Inflammatory Response and Oxidative Stress in Endothelial Cells. J. Agric. Food Chem. 2010, 58, 10840–10846. [Google Scholar] [CrossRef]
- Moreno-Fernandez, S.; Garces-Rimon, M.; Gonzalez, C.; Uranga, J.A.; Lopez-Miranda, V.; Vera, G.; Miguel, M. Pepsin egg white hydrolysate ameliorates metabolic syndrome in high-fat/high-dextrose fed rats. Food Funct. 2018, 9, 78–86. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jahandideh, F.; Wu, J. Perspectives on the Potential Benefits of Antihypertensive Peptides towards Metabolic Syndrome. Int. J. Mol. Sci. 2020, 21, 2192. https://doi.org/10.3390/ijms21062192
Jahandideh F, Wu J. Perspectives on the Potential Benefits of Antihypertensive Peptides towards Metabolic Syndrome. International Journal of Molecular Sciences. 2020; 21(6):2192. https://doi.org/10.3390/ijms21062192
Chicago/Turabian StyleJahandideh, Forough, and Jianping Wu. 2020. "Perspectives on the Potential Benefits of Antihypertensive Peptides towards Metabolic Syndrome" International Journal of Molecular Sciences 21, no. 6: 2192. https://doi.org/10.3390/ijms21062192
APA StyleJahandideh, F., & Wu, J. (2020). Perspectives on the Potential Benefits of Antihypertensive Peptides towards Metabolic Syndrome. International Journal of Molecular Sciences, 21(6), 2192. https://doi.org/10.3390/ijms21062192