Functional Divergence of Microtubule-Associated TPX2 Family Members in Arabidopsis thaliana
Abstract
1. Introduction
2. Results
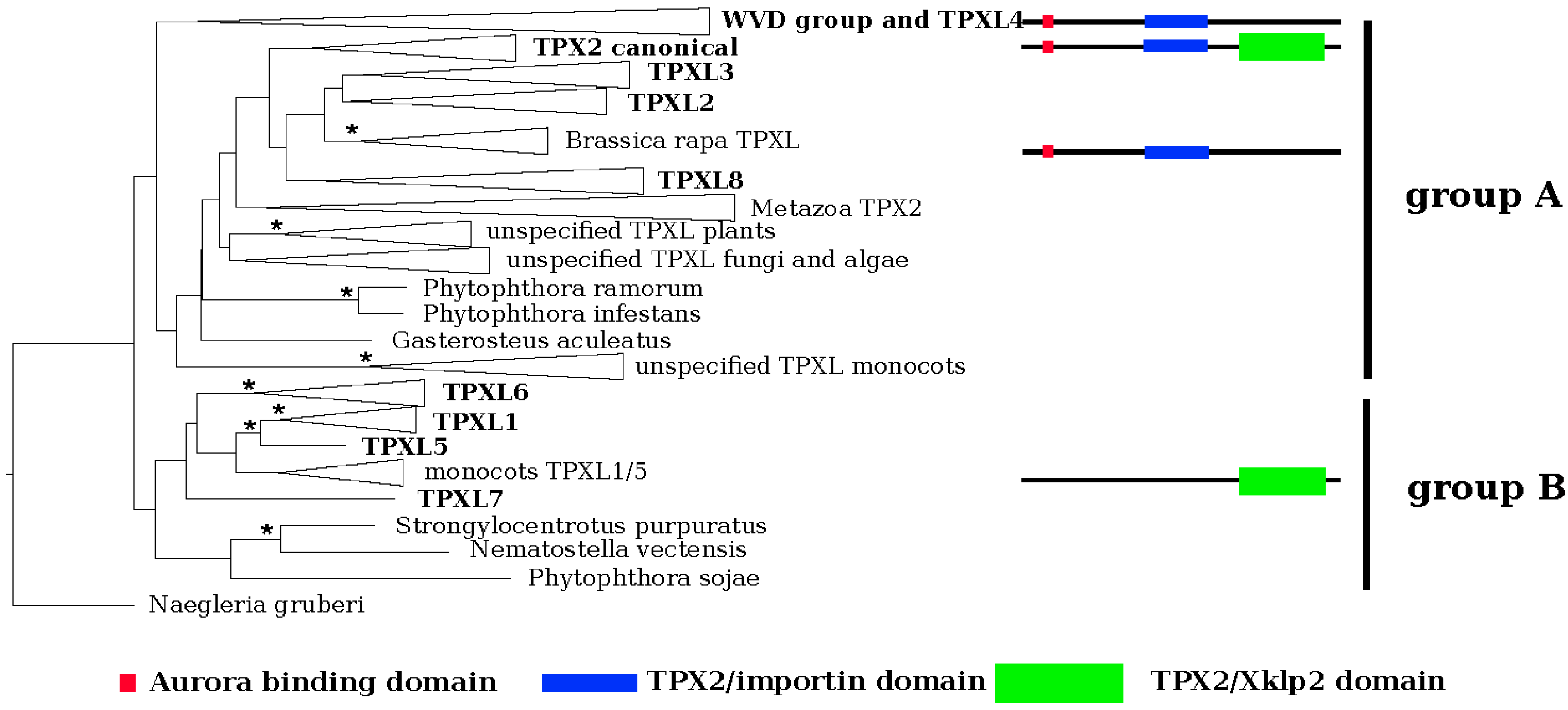
2.1. The A. thaliana Genome Possesses 20 Genes with Similarity to TPX2
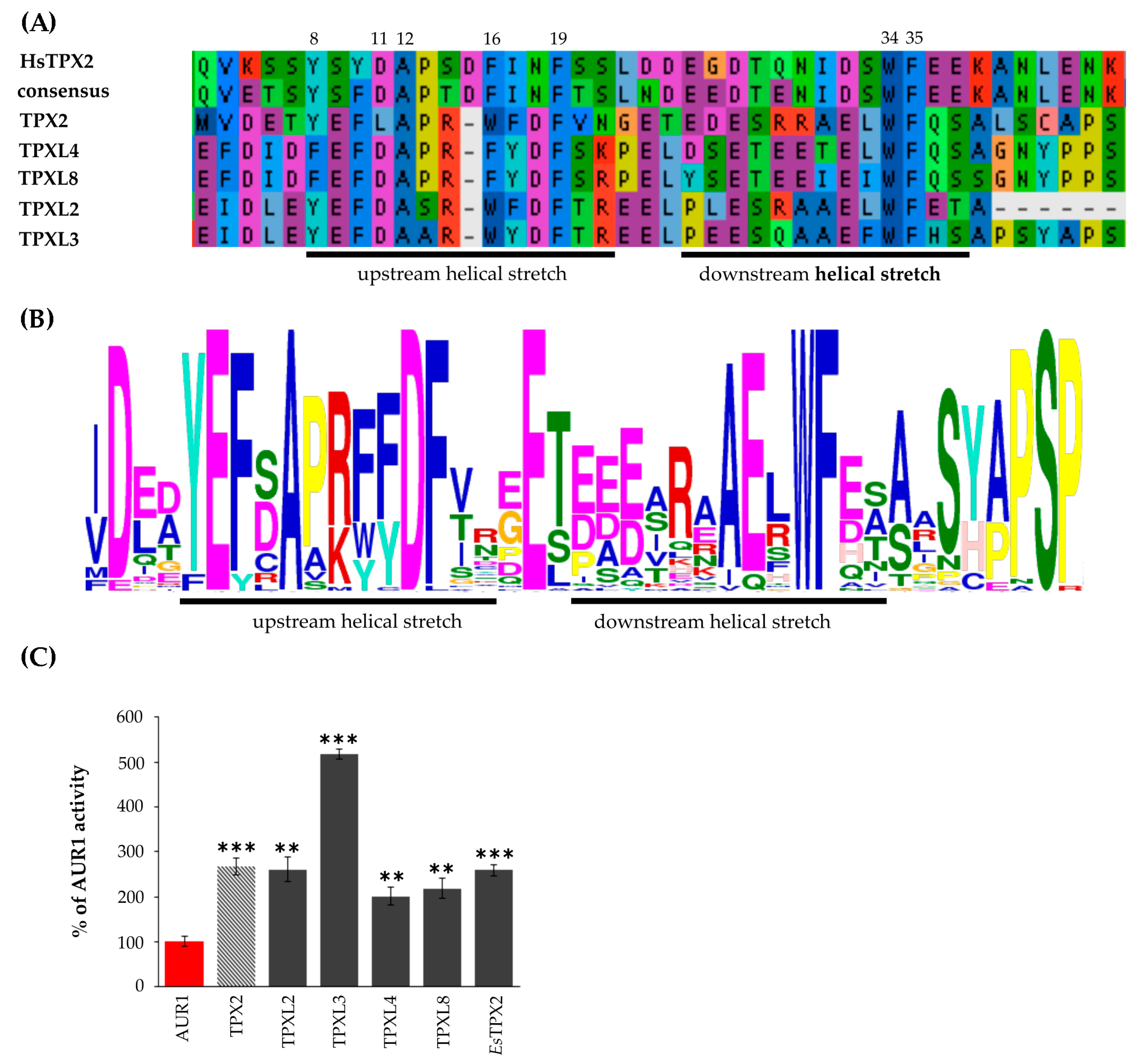
2.2. Functional Prediction of Arabidopsis Aurora Binding Domain
2.3. All Arabidopsis TPX2 Family Members Possessing An Aurora Kinase Binding Domain Activate AUR1 In Vitro
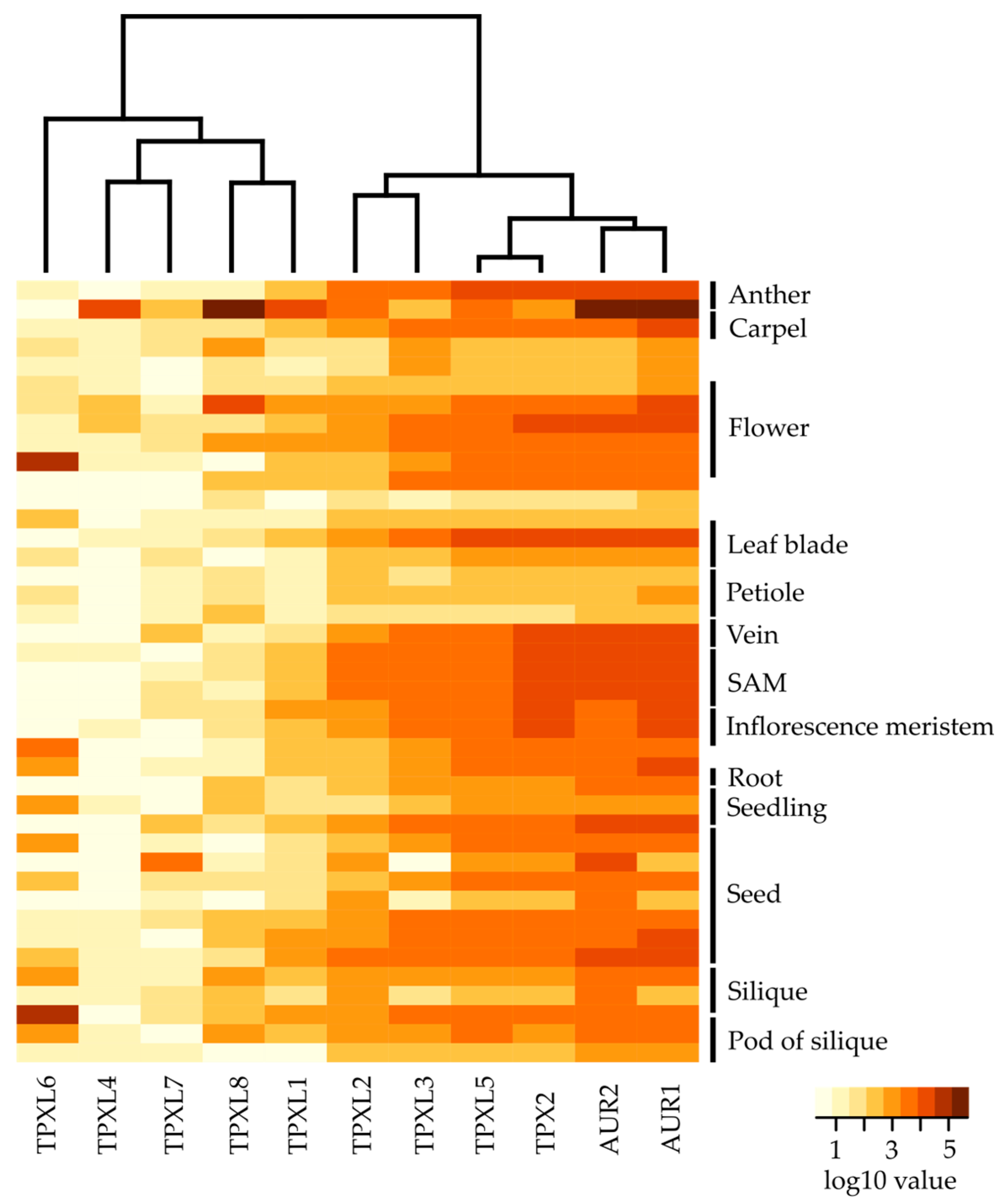
2.4. The Eight Closest Homologs of Canonical TPX2 are Differentially Expressed during Arabidopsis Development
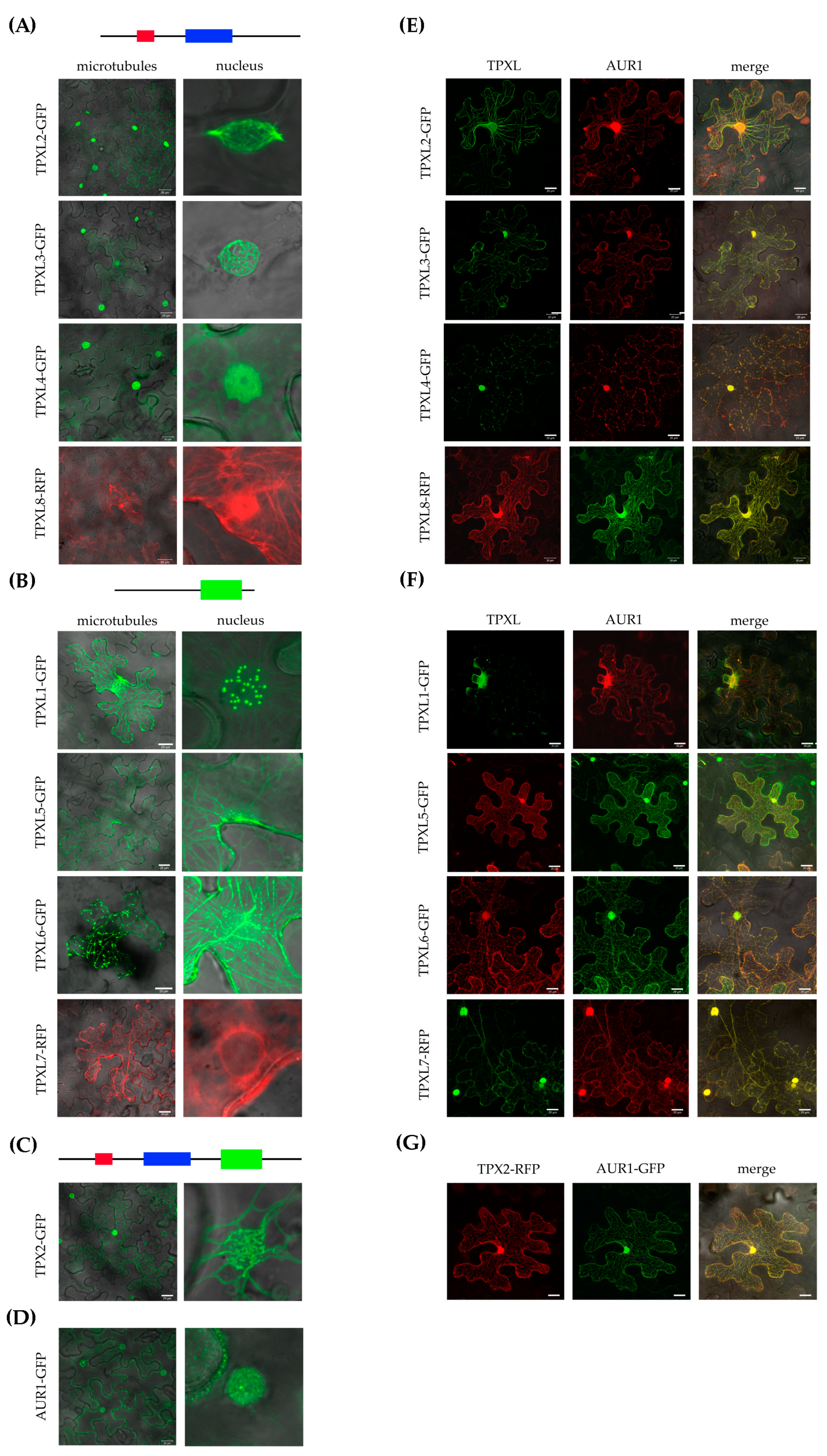
2.5. TPXL Proteins Mostly Localize on Microtubules
2.6. TPXL Proteins Re-localize Aurora1 Kinase by Loading It on Microtubular Arrays
3. Discussion
3.1. Diversity of TPX in Plants
3.2. Activation of Aurora Kinase by TPX2 Family Proteins Seems to Be Evolutionary Conserved
3.3. TPXL Members with Aurora Binding Domain are Strongly Expressed
3.4. The Localization of TPXL is Associated with Their Importance for Spindle Microtubules
4. Material and Methods
4.1. Identification of TPXL Proteins
4.2. Phylogenetic Analysis
4.3. Gene differential Expression Analyses
4.4. Plant Material
4.5. Cloning of TPXL Genes
4.6. Production of Recombinant Proteins
4.7. In Vitro Kinase Assay
4.8. Infiltration of N. benthamiana and Confocal Microscopy
4.9. Accession Numbers
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATP | Adenosine TriPhosphate |
| AUR1 | Aurora1 |
| AUR2 | Aurora2 |
| BLAST | Basic Local Alignment Search Tool |
| CENH3 | Centromeric Histone 3 |
| E. coli | Escherichia coli |
| eGFP | Enhanced Green Fluorescent Protein |
| GFP | Green Fluorescent Protein |
| GST | Glutathione S-Transferase |
| H3 | Histone 3 |
| KLEEK | Short motif of amino acids lysine, leucine, glutamic acid, glutamic acid, lysineS |
| MAP65 | Microtubule-Associated Protein 65 |
| N. benhamiana | Nicotiana benthamiana |
| OD | Optical Density |
| PCR | Polymerase Chain Reaction |
| RFP | Red Fluorescent Protein |
| TPX2 | Targeting Protein for Xklp2 |
| TPXL (1–8) | Targeting Protein for Xklp2-Like (1–8) |
| WVD | Wave-Dampened |
| WDL | Wave-Dampened Like |
References
- Willems, E.; Dedobbeleer, M.; Digregorio, M.; Lombard, A.; Lumapat, P.N.; Rogister, B. The functional diversity of Aurora kinases: A comprehensive review. Cell Div. 2018, 13, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Weimer, A.K.; Demidov, D.; Lermontova, I.; Beeckman, T.; Van Damme, D. Aurora kinases throughout plant development. Trends Plant Sci. 2016, 21, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Demidov, D. Identification and dynamics of two classes of aurora-like kinases in arabidopsis and other plants. Plant Cell 2005, 17, 836–848. [Google Scholar] [CrossRef] [PubMed]
- Kawabe, A.; Matsunaga, S.; Nakagawa, K.; Kurihara, D.; Yoneda, A.; Hasezawa, S.; Uchiyama, S.; Fukui, K. Characterization of plant Aurora kinases during mitosis. Plant Mol. Biol. 2005, 58, 1–13. [Google Scholar] [CrossRef]
- Van Damme, D.; De Rybel, B.; Gudesblat, G.; Demidov, D.; Grunewald, W.; De Smet, I.; Houben, A.; Beeckman, T.; Russinova, E. Arabidopsis α Aurora kinases function in formative cell division plane orientation. Plant Cell 2011, 23, 4013–4024. [Google Scholar] [CrossRef]
- Bayliss, R.; Sardon, T.; Vernos, I.; Conti, E. Structural basis of Aurora-A activation by TPX2 at the mitotic spindle. Mol. Cell 2003, 12, 851–862. [Google Scholar] [CrossRef]
- Fu, J.; Bian, M.; Liu, J.; Jiang, Q.; Zhang, C. A single amino acid change converts Aurora-A into Aurora-B-like kinase in terms of partner specificity and cellular function. Proc. Natl. Acad. Sci. USA 2009, 106, 6939–6944. [Google Scholar] [CrossRef]
- Petrovská, B.; Cenklová, V.; Pochylová, Ž.; Kourová, H.; Doskočilová, A.; Plíhal, O.; Binarová, L.; Binarová, P. Plant Aurora kinases play a role in maintenance of primary meristems and control of endoreduplication. New Phytol. 2012, 193, 590–604. [Google Scholar] [CrossRef]
- Demidov, D.; Lermontova, I.; Weiss, O.; Fuchs, J.; Rutten, T.; Kumke, K.; Sharbel, T.F.; Van Damme, D.; De Storme, N.; Geelen, D.; et al. Altered expression of Aurora kinases in Arabidopsis results in aneu- and polyploidization. Plant J. 2014, 80, 449–461. [Google Scholar] [CrossRef]
- Boruc, J.; Weimer, A.K.; Stoppin-Mellet, V.; Mylle, E.; Kosetsu, K.; Cedeño, C.; Jaquinod, M.; Njo, M.; De Milde, L.; Tompa, P.; et al. Phosphorylation of MAP65-1 by Arabidopsis Aurora kinases is required for efficient cell cycle progression. Plant Physiol. 2017, 173, 582–599. [Google Scholar] [CrossRef]
- Smertenko, A.P.; Chang, H.-Y.; Sonobe, S.; Fenyk, S.I.; Weingartner, M.; Bögre, L.; Hussey, P.J. Control of the AtMAP65-1 interaction with microtubules through the cell cycle. J. Cell Sci. 2006, 119, 3227–3237. [Google Scholar] [CrossRef] [PubMed]
- Gruss, O.J.; Wittmann, M.; Yokoyama, H.; Pepperkok, R.; Kufer, T.; Silljé, H.; Karsenti, E.; Mattaj, I.W.; Vernos, I. Chromosome-induced microtubule assembly mediated by TPX2 is required for spindle formation in HeLa cells. Nat. Cell Biol. 2002, 4, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Gruss, O.J.; Vernos, I. The mechanism of spindle assembly: Functions of Ran and its target TPX2. J. Cell Biol. 2004, 166, 949–955. [Google Scholar] [CrossRef] [PubMed]
- Vos, J.W.; Pieuchot, L.; Evrard, J.-L.; Janski, N.; Bergdoll, M.; de Ronde, D.; Perez, L.H.; Sardon, T.; Vernos, I.; Schmit, A.C. The plant TPX2 protein regulates prospindle assembly before nuclear envelope breakdown. Plant Cell 2008, 20, 2783–2797. [Google Scholar] [CrossRef]
- Tomaštíková, E.; Demidov, D.; Jeřábková, H.; Binarová, P.; Houben, A.; Doležel, J.; Petrovská, B. TPX2 protein of Arabidopsis activates Aurora kinase 1, but not Aurora kinase 3 in vitro. Plant Mol. Biol. Report. 2015, 33. [Google Scholar] [CrossRef]
- Petrovská, B.; Jeřábková, H.; Kohoutová, L.; Cenklová, V.; Pochylová, Ž.; Gelová, Z.; Kočárová, G.; Váchová, L.; Kurejová, M.; Tomaštíková, E.; et al. Overexpressed TPX2 causes ectopic formation of microtubular arrays in the nuclei of acentrosomal plant cells. J. Exp. Bot. 2013, 64. [Google Scholar] [CrossRef]
- Evrard, J.; Pieuchot, L.; Vos, J.W.; Vernos, I.; Schmit, A. Plant TPX2 and related proteins. Plant Signal Behav. 2009, 4, 69–72. [Google Scholar] [CrossRef]
- Boruc, J.; Deng, X.-G.; Mylle, E.; Besbrugge, N.; Van Durme, M.; Demidov, D.; Tomaštíková, E.D.; Tan, T.R.C.; Vandorpe, M.; Eeckhout, D.; et al. TPX2-LIKE PROTEIN 3 is the primary activator of α Aurora kinases and is essential for embryogenesis. Plant Physiol. 2019, 180, 1389–1405. [Google Scholar] [CrossRef]
- Du, P.; Kumar, M.; Yao, Y.; Xie, Q.; Wang, J.; Zhang, B.; Gan, S.; Wang, Y.; Wu, A.M. Genome-wide analysis of the TPX2 family proteins in Eucalyptus grandis. BMC Genom. 2016, 17, 967. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Szklarczyk, D.; Forslund, K.; Cook, H.; Heller, D.; Walter, M.C.; Rattei, T.; Mende, D.R.; Sunagawa, S.; Kuhn, M.; et al. EGGNOG 4.5: A hierarchical orthology framework with improved functional annotations for eukaryotic, prokaryotic and viral sequences. Nucleic Acids Res. 2016, 44, 286–293. [Google Scholar] [CrossRef]
- Liu, X.; Qin, T.; Ma, Q.; Sun, J.; Liu, Z.; Yuan, M.; Mao, T. Light-Regulated Hypocotyl Elongation Involves Proteasome-Dependent Degradation of the Microtubule Regulatory Protein WDL3 in Arabidopsis. Plant Cell 2013, 25, 1740–1755. [Google Scholar] [CrossRef]
- Yuen, C.Y.L.; Pearlman, R.S.; Silo-Suh, L.; Hilson, P.; Carroll, K.L.; Masson, P.H. WVD2 and WDL1 modulate helical organ growth and anisotropic cell expansion in Arabidopsis. Plant Physiol. 2014, 131, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Perrin, R.M.; Wang, Y.; Yuen, C.Y.L.; Will, J.; Masson, P.H. WVD2 is a novel microtubule-associated protein in Arabidopsis thaliana. Plant J. 2007, 49, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.L.; Boden, M.; Buske, F.A.; Frith, M.; Grant, C.E.; Clementi, L.; Ren, J.; Li, W.W.; Noble, W.S. MEME Suite: Tools for motif discovery and searching. Nucleic Acids Res. 2009, 37, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Demidov, D.; Hesse, S.; Tewes, A.; Rutten, T.; Fuchs, J.; Karimi Ashtiyani, R.; Lein, S.; Fischer, A.; Reuter, G.; Houben, A. Aurora1 phosphorylation activity on histone H3 and its cross-talk with other post-translational histone modifications in Arabidopsis. Plant J. 2009, 59, 221–230. [Google Scholar] [CrossRef]
- Klepikova, A.V.; Kasianov, A.S.; Gerasimov, E.S.; Logacheva, M.D.; Penin, A.A. A high resolution map of the arabidopsis thaliana developmental transcriptome based on RNA-seq profiling. Plant J. 2016, 88, 1058–1070. [Google Scholar] [CrossRef]
- Schatz, C.; Santarella, R.; Hoenger, A.; Karsenti, E.; Mattaj, I.W.; Gruss, O.J.; Carazo--Salas, R.E. Importin alpha-regulated nucleation of microtubules by TPX2. EMBO J. 2003, 22, 2060–2070. [Google Scholar] [CrossRef]
- Neumayer, G.; Nguyen, M.D. TPX2 impacts acetylation of histone H4 at lysine16: Implications for DNA damage response. PLoS ONE. 2014, 9, 1–9. [Google Scholar] [CrossRef]
- Neumayer, G.; Belzil, C.; Gruss, O.J.; Nguyen, M.D. TPX2: Of spindle assembly, DNA damage response, and cancer. Cell Mol. Life Sci. 2014, 71, 3027–3047. [Google Scholar] [CrossRef]
- Yamada, M.; Goshima, G. Mitotic spindle assembly in land plants: Molecules and mechanisms. Biology 2017, 6, 6. [Google Scholar] [CrossRef]
- Gicking, A.M.; Swentowsky, K.W.; Dawe, R.K.; Qiu, W. Functional diversification of the kinesin-14 family in land plants. FEBS Lett. 2018, 592, 1918–1928. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Dixit, R. Functions of the Arabidopsis kinesin superfamily of microtubule-based motor proteins. Protoplasma 2012, 249, 887–899. [Google Scholar] [CrossRef] [PubMed]
- Takagi, M.; Sakamoto, T.; Suzuki, R.; Nemoto, K.; Obayashi, T.; Hirakawa, T.; Matsunaga, T.M.; Kurihara, D.; Nariai, Y.; Urano, T.; et al. Plant Aurora kinases interact with and phosphorylate transcription factors. J. Plant Res. 2016, 129, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- Demidov, D.; Heckmann, S.; Weiss, O.; Rutten, T.; Dvořák Tomaštíková, E.; Kuhlmann, M.; Patrick Scholl, P.; Municio, C.M.; Lermontova, I.; Houben, A. Deregulated Phosphorylation of CENH3 at Ser65 Affects the Development of Floral Meristems in Arabidopsis thaliana. Front Plant Sci. 2019, 10, 928. [Google Scholar] [CrossRef] [PubMed]
- Kufer, T.A.; Silljé, H.H.W.; Körner, R.; Gruss, O.J.; Meraldi, P.; Nigg, E.A. Human TPX2 is required for targeting aurora-a kinase to the spindle. J. Cell Biol. 2002, 158, 617–623. [Google Scholar] [CrossRef]
- Bird, A.W.; Hyman, A.A. Building a spindle of the correct length in human cells requires the interaction between TPX2 and Aurora A. J. Cell Biol. 2008, 182, 289–300. [Google Scholar] [CrossRef]
- Iyer, J.; Tsai, M.Y. A novel role for TPX2 as a scaffold and co-activator protein of the Chromosomal Passenger Complex. Cell Signal. 2012, 24, 1677–1689. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Finn, R.D.; Tate, J.; Mistry, J.; Coggill, P.C.; Sammut, S.J.; Hotz, H.-R. The Pfam protein families database. Nucleic Acids Res. 2008, 36, 281–288. [Google Scholar] [CrossRef]
- Letunic, I.; Doerks, T.; Bork, P. SMART 7: Recent updates to the protein domain annotation resource. Nucleic Acids Res. 2011, 1–4. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A. Creating the CIPRES Science gateway. In Proceedings of the 2010 Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; pp. 1–7. [Google Scholar]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Molecular biology and evolution. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Conesa, A.; Madrigal, P.; Tarazona, S.; Gomez-Cabrero, D.; Cervera, A.; McPherson, A.; Szcześniak, M.W.; Gaffney, D.J.; Elo, L.L.; Zhang, X.; et al. A survey of best practices for RNA-seq data analysis. Genome Biol. 2016, 17. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org (accessed on 9 February 2019).
- Tomaštíková, E.; Cenklová, V.; Kohoutová, L.; Petrovská, B.; Váchová, L.; Halada, P.; Kočárová, G.; Binarová, P. Interactions of an Arabidopsis RanBPM homologue with LisH-CTLH domain proteins revealed high conservation of CTLH complexes in eukaryotes. BMC Plant Biol. 2012, 12. [Google Scholar] [CrossRef]
- Phan, H.T.; Conrad, U. Vaccine Technologies for Veterinary Viral Diseases; Humana Press: New York, NY, USA, 2016; Volume 1349, pp. 35–47. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dvořák Tomaštíková, E.; Rutten, T.; Dvořák, P.; Tugai, A.; Ptošková, K.; Petrovská, B.; van Damme, D.; Houben, A.; Doležel, J.; Demidov, D. Functional Divergence of Microtubule-Associated TPX2 Family Members in Arabidopsis thaliana. Int. J. Mol. Sci. 2020, 21, 2183. https://doi.org/10.3390/ijms21062183
Dvořák Tomaštíková E, Rutten T, Dvořák P, Tugai A, Ptošková K, Petrovská B, van Damme D, Houben A, Doležel J, Demidov D. Functional Divergence of Microtubule-Associated TPX2 Family Members in Arabidopsis thaliana. International Journal of Molecular Sciences. 2020; 21(6):2183. https://doi.org/10.3390/ijms21062183
Chicago/Turabian StyleDvořák Tomaštíková, Eva, Twan Rutten, Petr Dvořák, Alisa Tugai, Klara Ptošková, Beáta Petrovská, Daniel van Damme, Andreas Houben, Jaroslav Doležel, and Dmitri Demidov. 2020. "Functional Divergence of Microtubule-Associated TPX2 Family Members in Arabidopsis thaliana" International Journal of Molecular Sciences 21, no. 6: 2183. https://doi.org/10.3390/ijms21062183
APA StyleDvořák Tomaštíková, E., Rutten, T., Dvořák, P., Tugai, A., Ptošková, K., Petrovská, B., van Damme, D., Houben, A., Doležel, J., & Demidov, D. (2020). Functional Divergence of Microtubule-Associated TPX2 Family Members in Arabidopsis thaliana. International Journal of Molecular Sciences, 21(6), 2183. https://doi.org/10.3390/ijms21062183






