The Role of Dorsal Raphe Serotonin Neurons in the Balance between Reward and Aversion
Abstract
1. Introduction
2. Results
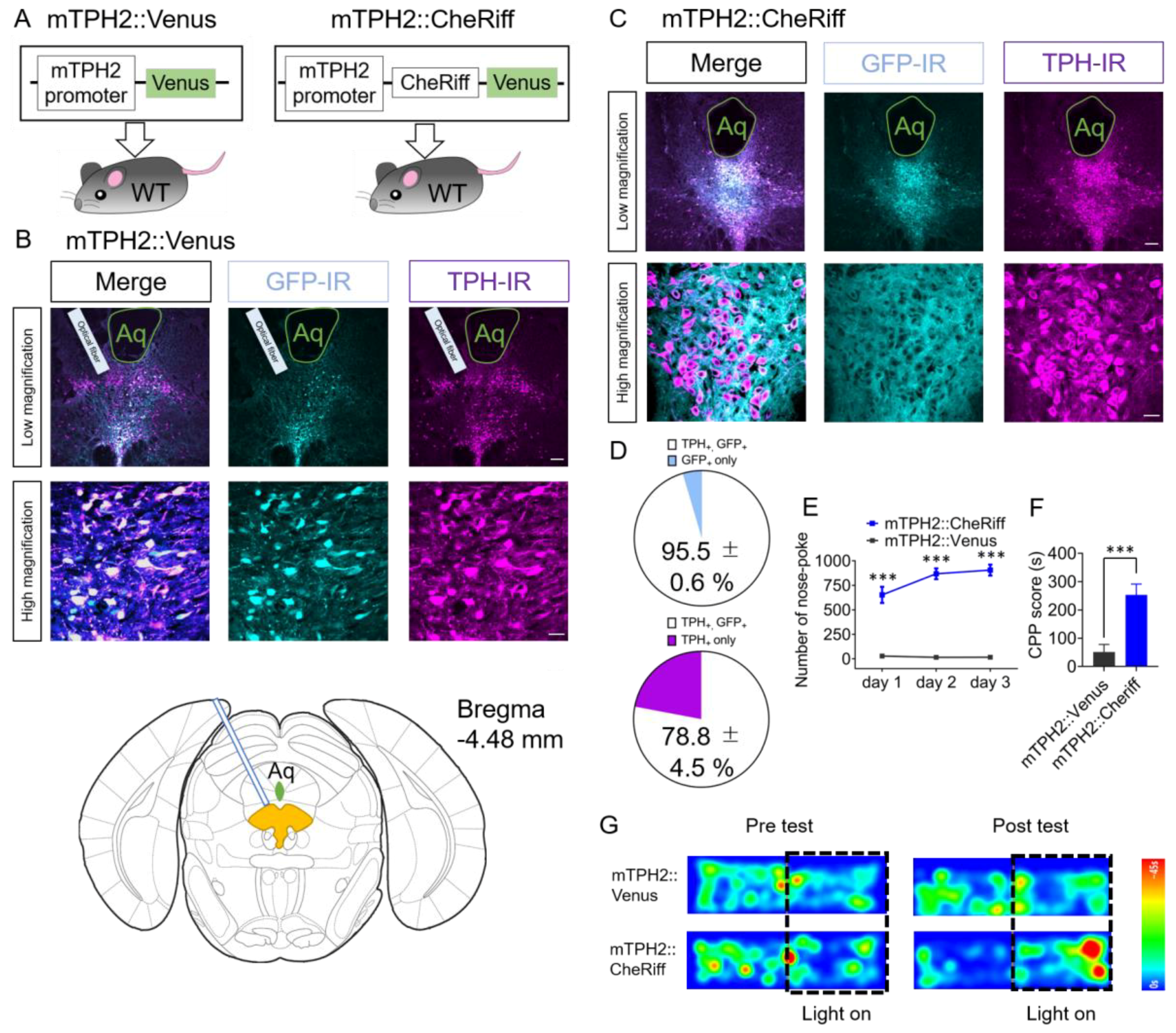
2.1. Effect of Optogenetic Activation of DRN 5-HT Neurons Transduced by AAV Bearing Mouse TPH2 Promoter in Nose-Poke Self-Stimulation Test and Conditioned Place Preference (CPP) Test
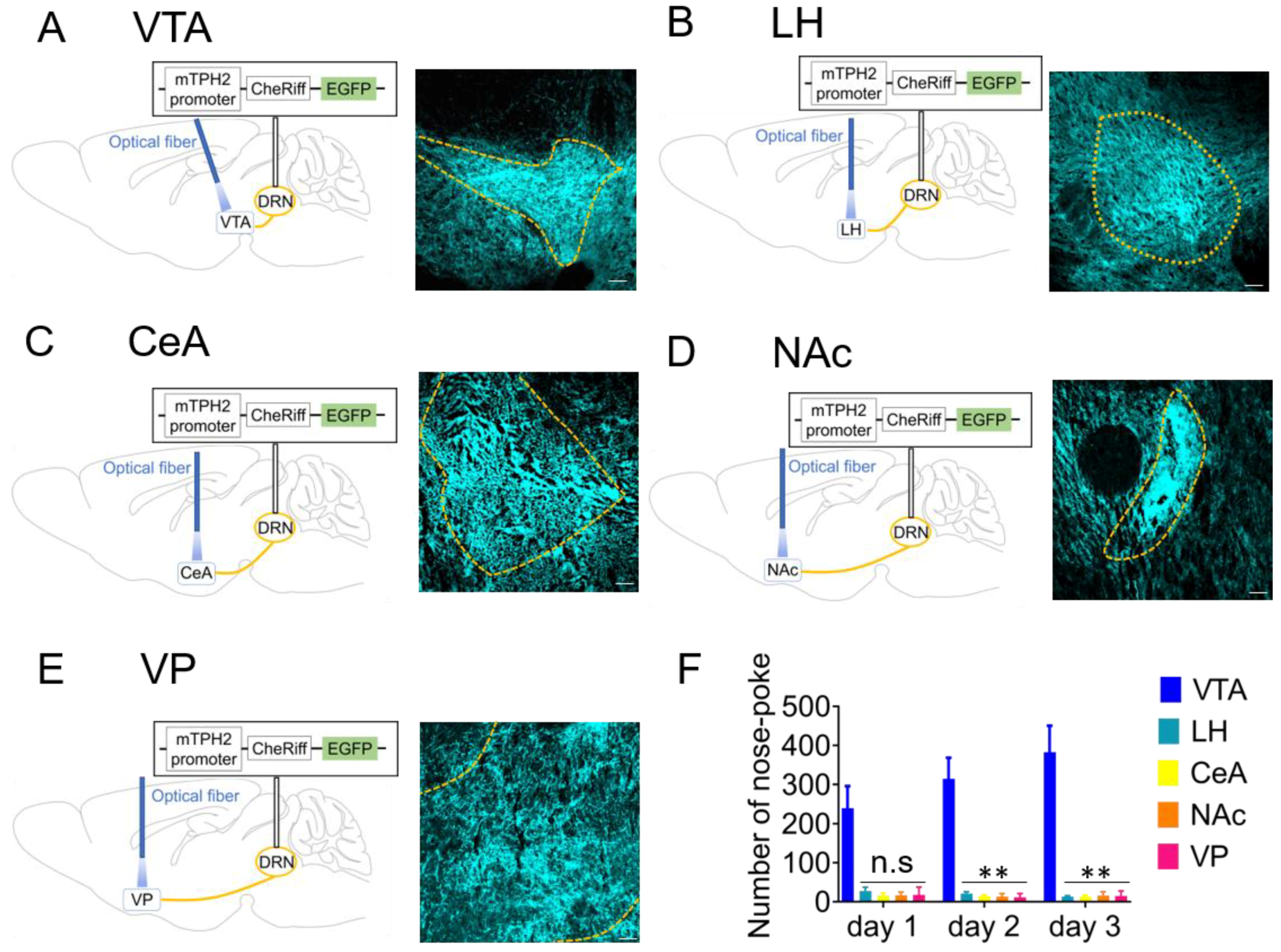
2.2. Optogenetic Activation of 5-HT Neuron Terminals in the VTA is Responsible for Reinforcement of Nose-Poke Behavior but not in the LH, CeA, NAc, or Ventral Pallidum (VP)
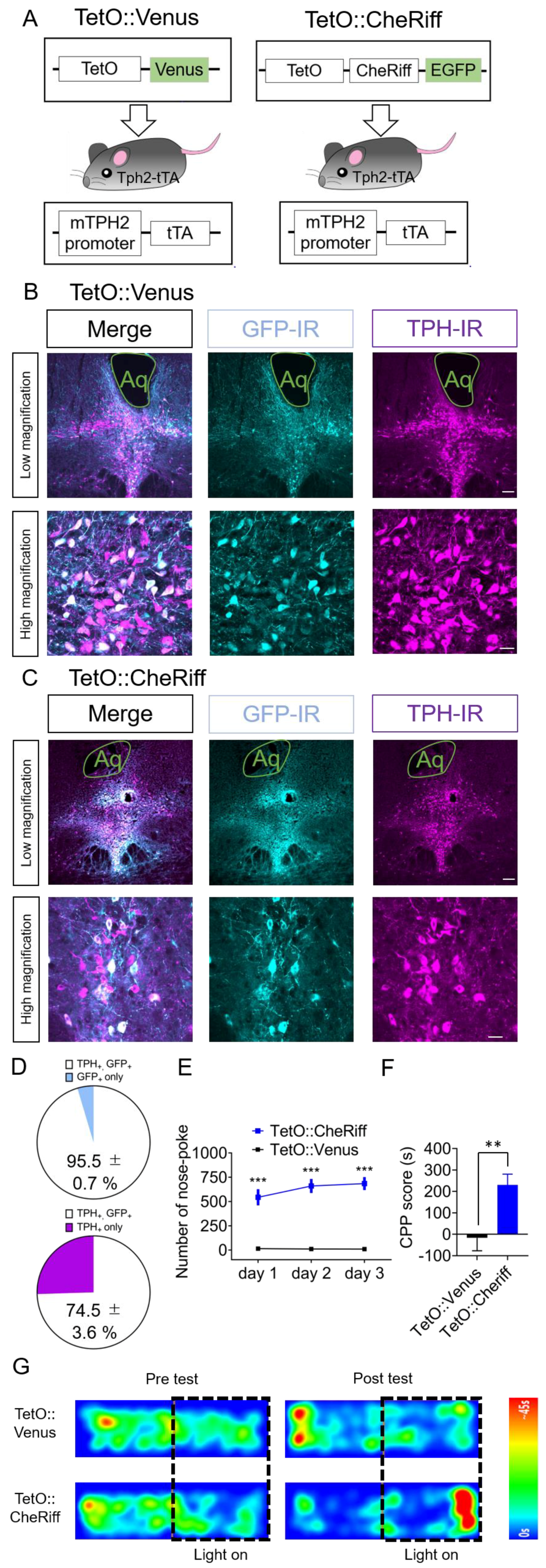
2.3. Effect of Optogenetic Activation of DRN 5-HT Neurons Targeted by Tph2-tTA Transgenic Mice in Nose-Poke Self-Stimulation Test and CPP Test
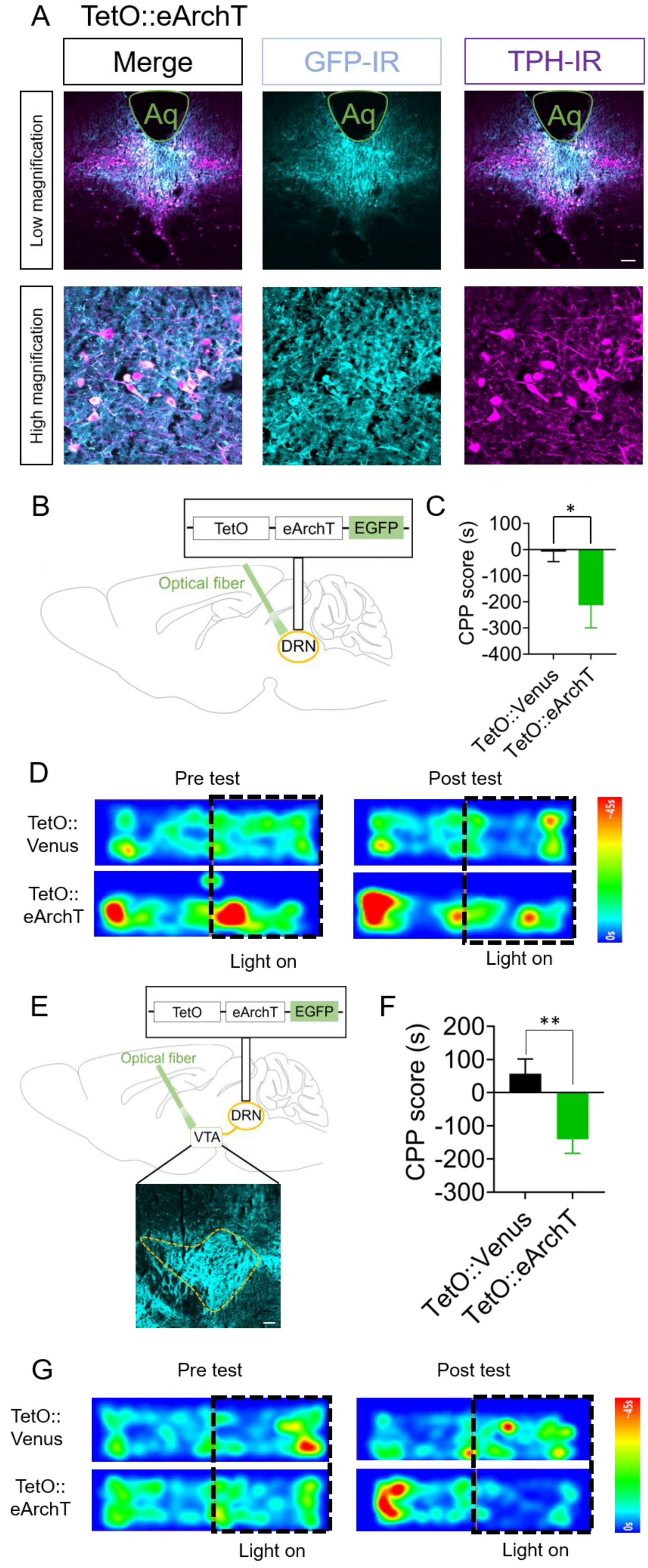
2.4. Optogenetic Inhibition of DRN 5-HT Neurons and Their Terminals in the VTA Elicits Conditioned Place Aversion
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Stereotaxic Surgeries
4.3. Vector Construction
4.4. Production and Purification of Adeno-Associated Virus (AAV) Vector
4.5. In Vivo Optogenetic Manipulation
4.6. Behavioral Tests
4.7. Self-Stimulation in an Operant Chamber
4.8. Conditioned Place Preference (CPP) Test
4.9. Conditioned Place Aversion (CPA) Test
4.10. Histology
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Costello, C.G. Depression: Loss of reinforcers of loss of reinforcer effectiveness? Behav. Ther. 1972, 3, 240–247. [Google Scholar] [CrossRef]
- Pizzagalli, D.; Iosifescu, D.; Hallett, L.; Ratner, K.; Maurizo, F. Evidence from a Probabilistic Reward Task. J. Psychiatr. Res. 2009, 43, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Der-Avakian, A.; Markou, A. The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci. 2012, 35, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Hall, G.B.C.; Milne, A.M.B.; MacQueen, G.M. An fMRI study of reward circuitry in patients with minimal or extensive history of major depression. Eur. Arch. Psychiatry Clin. Neurosci. 2014, 264, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Strauss, G.P.; Waltz, J.A.; Gold, J.M. A review of reward processing and motivational impairment in schizophrenia. Schizophr. Bull. 2014, 40, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Spanagel, R.; Weiss, F. The dopamine hypothesis of reward: Past and current status. Trends Neurosci. 1999, 22, 521–527. [Google Scholar] [CrossRef]
- Wise, R.A. Dopamine, learning and motivation. Nat. Rev. Neurosci. 2004, 5, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Kirby, L.G.; Zeeb, F.D.; Winstanley, C.A. Contributions of serotonin in addiction vulnerability. Neuropharmacology 2011, 61, 421–432. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, J.; Li, Y.; Hu, F.; Lu, Y.; Ma, M.; Feng, Q.; Zhang, J.; Wang, D.; Zeng, J.; et al. Dorsal raphe neurons signal reward through 5-HT and glutamate. Neuron 2014, 81, 1360–1374. [Google Scholar] [CrossRef]
- Li, Y.; Zhong, W.; Wang, D.; Feng, Q.; Liu, Z.; Zhou, J.; Jia, C.; Hu, F.; Zeng, J.; Guo, Q.; et al. Serotonin neurons in the dorsal raphe nucleus encode reward signals. Nat. Commun. 2016, 7, 10503. [Google Scholar] [CrossRef]
- Wang, H.L.; Zhang, S.; Qi, J.; Wang, H.; Cachope, R.; Mejias-Aponte, C.A.; Gomez, J.A.; Mateo-Semidey, G.E.; Beaudoin, G.M.J.; Paladini, C.A.; et al. Dorsal Raphe Dual Serotonin-Glutamate Neurons Drive Reward by Establishing Excitatory Synapses on VTA Mesoaccumbens Dopamine Neurons. Cell Rep. 2019, 26, 1128–1142. [Google Scholar] [CrossRef] [PubMed]
- Rompre, P.P.; Miliaressis, E. Pontine and mesencephalic substrates of self-stimulation. Brain Res. 1985, 359, 246–259. [Google Scholar] [CrossRef]
- Rompré, P.P.; Boye, S. Localization of reward-relevant neurons in the pontine tegmentum: a moveable electrode mapping study. Brain Res. 1989, 496, 295–302. [Google Scholar] [CrossRef]
- Van Der Kooy, D.; Fibiger, H.C.; Phillips, A.G. An analysis of dorsal and median raphe self-stimulation: Effects of para-chlorophenylalanine. Pharm. Biochem. Behav. 1978, 8, 441–445. [Google Scholar] [CrossRef]
- Fakhoury, M.; Rompré, P.P.; Boye, S.M. Role of the dorsal diencephalic conduction system in the brain reward circuitry. Behav. Brain Res. 2016, 296, 431–441. [Google Scholar] [CrossRef]
- Nakamura, K.; Matsumoto, M.; Hikosaka, O. Reward-dependent modulation of neuronal activity in the primate dorsal raphe nucleus. J. Neurosci. 2008, 28, 5331–5343. [Google Scholar] [CrossRef]
- Zhong, W.; Li, Y.; Feng, Q.; Luo, M. Learning and stress shape the reward response patterns of serotonin neurons. J. Neurosci. 2017, 37, 8863–8875. [Google Scholar] [CrossRef]
- Harrison, A.A.; Liem, Y.T.B.; Markou, A. Fluoxetine combined with a serotonin-1A receptor antagonist reversed reward deficits observed during nicotine and amphetamine withdrawal in rats. Neuropsychopharmacology 2001, 25, 55–71. [Google Scholar] [CrossRef]
- Rocha, B.A.; Fumagalli, F.; Gainetdinov, R.R.; Jones, S.R.; Ator, R.; Giros, B.; Miller, G.W.; Caron, M.G. Cocaine self-administration in dopamine-transporter knockout mice. Nat. Neurosci. 1998, 1, 132–137. [Google Scholar] [CrossRef]
- Hnasko, T.S.; Sotak, B.N.; Palmiter, R.D. Cocaine-conditioned place preference by dopamine-deficient mice is mediated by serotonin. J. Neurosci. 2007, 27, 12484–12488. [Google Scholar] [CrossRef]
- Di Giovanni, G.; De Deurwaerdère, P. New therapeutic opportunities for 5-HT2C receptor ligands in neuropsychiatric disorders. Pharm. Ther. 2016, 157, 125–162. [Google Scholar] [CrossRef] [PubMed]
- De Deurwaerdère, P.; Di Giovanni, G. Serotonergic modulation of the activity of mesencephalic dopaminergic systems: Therapeutic implications. Prog. Neurobiol. 2017, 151, 175–236. [Google Scholar] [CrossRef] [PubMed]
- Azmitia, E.C.; Segal, M. An autoradiographic analysis of the differential ascending projections of the dorsal and median raphe nuclei in the rat. J. Comp. Neurol. 1978, 179, 641–667. [Google Scholar] [CrossRef] [PubMed]
- Ishimura, K.; Takeuchi, Y.; Fujiwara, K.; Tominaga, M.; Yoshioka, H.; Sawada, T. Quantitative analysis of the distribution of serotonin-immunoreactive cell bodies in the mouse brain. Neurosci. Lett. 1988, 91, 265–270. [Google Scholar] [CrossRef]
- Luo, M.; Zhou, J.; Liu, Z. Reward processing by the dorsal raphe nucleus: 5-HT and beyond. Learn. Mem. 2015, 22, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Byerley, W.F.; Reimherr, F.W.; Wood, D.R.; Grosser, B.I. Fluoxetine, a selective serotonin uptake inhibitor, for the treatment of outpatients with major depression. J. Clin. Psychopharmacol. 1988, 8, 112–115. [Google Scholar] [CrossRef]
- Muijen, M.; Silverstone, T.; Mehmet, A.; Christie, M. A comparative clinical trial of and placebo in depressed outpatients. Acta Psychiatr Scand 1988, 78, 384–390. [Google Scholar] [CrossRef]
- Feighner, J.P.; Overõ, K. Multicenter, placebo-controlled, fixed-dose study of citalopram in moderate-to-severe depression. J. Clin. Psychiatry 1999, 60, 824–830. [Google Scholar] [CrossRef]
- Ressler, K.J.; Nemeroff, C.B. Role of serotonergic and noradrenergic systems in the pathophysiology of depression and anxiety disorders. Depress. Anxiety 2000, 12, 2–19. [Google Scholar] [CrossRef]
- Teissier, A.; Chemiakine, A.; Inbar, B.; Bagchi, S.; Ray, R.S.; Palmiter, R.D.; Dymecki, S.M.; Moore, H.; Ansorge, M.S. Activity of Raphé Serotonergic Neurons Controls Emotional Behaviors. Cell Rep. 2015, 13, 1965–1976. [Google Scholar] [CrossRef]
- Nishitani, N.; Nagayasu, K.; Asaoka, N.; Yamashiro, M.; Andoh, C.; Nagai, Y.; Kinoshita, H.; Kawai, H.; Shibui, N.; Liu, B.; et al. Manipulation of dorsal raphe serotonergic neurons modulates active coping to inescapable stress and anxiety-related behaviors in mice and rats. Neuropsychopharmacology 2019, 44, 721–732. [Google Scholar] [CrossRef] [PubMed]
- McDevitt, R.A.; Tiran-Cappello, A.; Shen, H.; Balderas, I.; Britt, J.P.; Marino, R.A.M.; Chung, S.L.; Richie, C.T.; Harvey, B.K.; Bonci, A. Serotonergic versus nonserotonergic dorsal raphe projection neurons: Differential participation in reward circuitry. Cell Rep. 2014, 8, 1857–1869. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.W.; Miyazaki, K.; Tanaka, K.F.; Yamanaka, A.; Takahashi, A.; Tabuchi, S.; Doya, K. Optogenetic activation of dorsal raphe serotonin neurons enhances patience for future rewards. Curr. Biol. 2014, 24, 2033–2040. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, M.S.; Murakami, M.; Mainen, Z.F. Activation of dorsal raphe serotonergic neurons promotes waiting but is not reinforcing. Curr. Biol. 2015, 25, 306–315. [Google Scholar] [CrossRef]
- Browne, C.J.; Abela, A.R.; Chu, D.; Li, Z.; Ji, X.; Lambe, E.K.; Fletcher, P.J. Dorsal raphe serotonin neurons inhibit operant responding for reward via inputs to the ventral tegmental area but not the nucleus accumbens: evidence from studies combining optogenetic stimulation and serotonin reuptake inhibition. Neuropsychopharmacology 2019, 44, 793–804. [Google Scholar] [CrossRef]
- Ohmura, Y.; Tanaka, K.F.; Tsunematsu, T.; Yamanaka, A.; Yoshioka, M. Optogenetic activation of serotonergic neurons enhances anxiety-like behaviour in mice. Int. J. Neuropsychopharmacol. 2014, 17, 1777–1783. [Google Scholar] [CrossRef]
- Benzekhroufa, K.; Liu, B.-H.; Teschemacher, A.G.; Kasparov, S. Targeting central serotonergic neurons with lentiviral vectors based on a transcriptional amplification strategy. Gene Ther. 2009, 16, 681–688. [Google Scholar] [CrossRef]
- Hochbaum, D.R.; Zhao, Y.; Farhi, S.L.; Klapoetke, N.; Werley, C.A.; Kapoor, V.; Zou, P.; Kralj, J.M.; Maclaurin, D.; Smedemark-Margulies, N.; et al. All-optical electrophysiology in mammalian neurons using engineered microbial rhodopsins. Nat. Methods 2014, 11, 825–833. [Google Scholar] [CrossRef]
- Vertes, R.P. A PHA-L analysis of ascending projections of the dorsal raphe nucleus in the rat. J. Comp. Neurol. 1991, 313, 643–668. [Google Scholar] [CrossRef]
- Muzerelle, A.; Scotto-Lomassese, S.; Bernard, J.F.; Soiza-Reilly, M.; Gaspar, P. Conditional anterograde tracing reveals distinct targeting of individual serotonin cell groups (B5–B9) to the forebrain and brainstem. Brain Struct. Funct. 2016, 221, 535–561. [Google Scholar] [CrossRef]
- Miyazaki, K.; Miyazaki, K.W.; Yamanaka, A.; Tokuda, T.; Tanaka, K.F.; Doya, K. Reward probability and timing uncertainty alter the effect of dorsal raphe serotonin neurons on patience. Nat. Commun. 2018, 9, 2048. [Google Scholar] [CrossRef] [PubMed]
- Mattis, J.; Tye, K.M.; Ferenczi, E.A.; Ramakrishnan, C.; O’Shea, D.J.; Prakash, R.; Gunaydin, L.A.; Hyun, M.; Fenno, L.E.; Gradinaru, V.; et al. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat. Methods 2011, 9, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Friedmann, D.; Xiong, J.; Liu, C.D.; Ferguson, B.R.; Weerakkody, T.; DeLoach, K.E.; Ran, C.; Pun, A.; Sun, Y.; et al. Anatomically Defined and Functionally Distinct Dorsal Raphe Serotonin Sub-systems. Cell 2018, 175, 472–487. [Google Scholar] [CrossRef] [PubMed]
- Nagai, Y.; Nishitani, N.; Yasuda, M.; Ueda, Y.; Fukui, Y.; Andoh, C.; Shirakawa, H.; Nakagawa, T.; Inoue, K.; Nagayasu, K.; et al. Identification of neuron-type specific promoters in monkey genome and their functional validation in mice. Biochem. Biophys. Res. Commun. 2019, 518, 619–624. [Google Scholar] [CrossRef]
- Tanaka, K.F.; Matsui, K.; Sasaki, T.; Sano, H.; Sugio, S.; Fan, K.; Hen, R.; Nakai, J.; Yanagawa, Y.; Hasuwa, H.; et al. Expanding the Repertoire of Optogenetically Targeted Cells with an Enhanced Gene Expression System. Cell Rep. 2012, 2, 397–406. [Google Scholar] [CrossRef]
- Grimm, D.; Lee, J.S.; Wang, L.; Desai, T.; Akache, B.; Storm, T.A.; Kay, M.A. In Vitro and In Vivo Gene Therapy Vector Evolution via Multispecies Interbreeding and Retargeting of Adeno-Associated Viruses. J. Virol. 2008, 82, 5887–5911. [Google Scholar] [CrossRef]
- Jacobs, B.L.; Azmitia, E.C. Structure and function of the brain serotonin system. Physiol. Rev. 1992, 72, 165–230. [Google Scholar] [CrossRef]
- Charara, A.; Parent, A. Chemoarchitecture of the primate dorsal raphe nucleus. J. Chem. Neuroanat. 1998, 15, 111–127. [Google Scholar] [CrossRef]
- Gras, C.; Herzog, E.; Bellenchi, G.C.; Bernard, V.; Ravassard, P.; Pohl, M.; Gasnier, B.; Giros, B.; El Mestikawy, S. A third vesicular glutamate transporter expressed by cholinergic and serotoninergic neurons. J. Neurosci. 2002, 22, 5442–5451. [Google Scholar] [CrossRef]
- Hioki, H.; Nakamura, H.; Ma, Y.F.; Konno, M.; Hayakawa, T.; Nakamura, K.C.; Fujiyama, F.; Kaneko, T. Vesicular glutamate transporter 3-expressing nonserotonergic projection neurons constitute a subregion in the rat midbrain raphe nuclei. J. Comp. Neurol. 2010, 518, 668–686. [Google Scholar] [CrossRef]
- Ren, J.; Isakova, A.; Friedmann, D.; Zeng, J.; Grutzner, S.M.; Pun, A.; Zhao, G.Q.; Kolluru, S.S.; Wang, R.; Lin, R.; et al. Single-cell transcriptomes and whole-brain projections of serotonin neurons in the mouse dorsal and median raphe nuclei. eLife 2019, 8, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Tzschentke, T.M.; Schmidt, W.J. Functional heterogeneity of the rat medial prefrontal cortex: Effects of discrete subarea-specific lesions on drug-induced conditioned place preference and behavioural sensitization. Eur. J. Neurosci. 1999, 11, 4099–4109. [Google Scholar] [CrossRef] [PubMed]
- Otis, J.M.; Dashew, K.B.; Mueller, D. Neurobiological dissociation of retrieval and reconsolidation of cocaine-associated memory. J. Neurosci. 2013, 33, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yanagida, J.; Kamii, H.; Wada, S.; Domoto, M.; Sasase, H.; Deyama, S.; Takarada, T.; Hinoi, E.; Sakimura, K.; et al. Glutamatergic neurons in the medial prefrontal cortex mediate the formation and retrieval of cocaine-associated memories in mice. Addict. Biol. 2020, 25, e12723. [Google Scholar] [CrossRef]
- Tian, M.K.; Schmidt, E.F.; Lambe, E.K. Serotonergic Suppression of Mouse Prefrontal Circuits Implicated in Task Attention. eNeuro 2016, 3. [Google Scholar] [CrossRef]
- Hayashi, K.; Nakao, K.; Nakamura, K. Appetitive and Aversive Information Coding in the Primate Dorsal Raphe Nucleus. J. Neurosci. 2015, 35, 6195–6208. [Google Scholar] [CrossRef]
- Tan, K.R.; Yvon, C.; Turiault, M.; Mirzabekov, J.J.; Doehner, J.; Labouèbe, G.; Deisseroth, K.; Tye, K.M.; Lüscher, C. GABA Neurons of the VTA Drive Conditioned Place Aversion. Neuron 2012, 73, 1173–1183. [Google Scholar] [CrossRef]
- Solecki, W.B.; Wilczkowski, M.; Pradel, K.; Karwowska, K.; Kielbinski, M.; Drwięga, G.; Zajda, K.; Blasiak, T.; Soltys, Z.; Rajfur, Z.; et al. Effects of brief inhibition of the ventral tegmental area dopamine neurons on the cocaine seeking during abstinence. Addict. Biol. 2019. [Google Scholar] [CrossRef]
- Franklin, B.J.K.; Paxinos, G. The Mouse Brain in Stereotaxic Coordinates, 3rd ed.; Academic Press: New York, NY, USA, 2007. [Google Scholar]
- Nagai, T.; Ibata, K.; Park, E.S.; Kubota, M.; Mikoshiba, K.; Miyawaki, A. A variant of YFP with fast and efficient maturation for cell-biological applications. Nature 2002, 20, 1585–1588. [Google Scholar]
- Zhang, T.; Deyama, S.; Domoto, M.; Wada, S.; Yanagida, J.; Sasase, H.; Hinoi, E.; Nishitani, N.; Nagayasu, K.; Kaneko, S.; et al. Activation of GABAergic neurons in the nucleus accumbens mediates the expression of cocaine-associated memory. Biol. Pharm. Bull. 2018, 41, 1084–1088. [Google Scholar] [CrossRef]
- Tzschentke, T.M. Measuring reward with the conditioned place preference paradigm: A comprehensive review of drug effects, recent progress and new issues. Prog. Neurobiol. 1998, 56, 613–672. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagai, Y.; Takayama, K.; Nishitani, N.; Andoh, C.; Koda, M.; Shirakawa, H.; Nakagawa, T.; Nagayasu, K.; Yamanaka, A.; Kaneko, S. The Role of Dorsal Raphe Serotonin Neurons in the Balance between Reward and Aversion. Int. J. Mol. Sci. 2020, 21, 2160. https://doi.org/10.3390/ijms21062160
Nagai Y, Takayama K, Nishitani N, Andoh C, Koda M, Shirakawa H, Nakagawa T, Nagayasu K, Yamanaka A, Kaneko S. The Role of Dorsal Raphe Serotonin Neurons in the Balance between Reward and Aversion. International Journal of Molecular Sciences. 2020; 21(6):2160. https://doi.org/10.3390/ijms21062160
Chicago/Turabian StyleNagai, Yuma, Kaito Takayama, Naoya Nishitani, Chihiro Andoh, Masashi Koda, Hisashi Shirakawa, Takayuki Nakagawa, Kazuki Nagayasu, Akihiro Yamanaka, and Shuji Kaneko. 2020. "The Role of Dorsal Raphe Serotonin Neurons in the Balance between Reward and Aversion" International Journal of Molecular Sciences 21, no. 6: 2160. https://doi.org/10.3390/ijms21062160
APA StyleNagai, Y., Takayama, K., Nishitani, N., Andoh, C., Koda, M., Shirakawa, H., Nakagawa, T., Nagayasu, K., Yamanaka, A., & Kaneko, S. (2020). The Role of Dorsal Raphe Serotonin Neurons in the Balance between Reward and Aversion. International Journal of Molecular Sciences, 21(6), 2160. https://doi.org/10.3390/ijms21062160




