Acute and Chronic Nicotine Exposures Differentially Affect Central Serotonin 2A Receptor Function: Focus on the Lateral Habenula
Abstract
1. Introduction
2. Results
2.1. Effect of Systemic Administration of Acute and Chronic Nicotine on 5-HT2AR Immunohistochemistry Expression in the LHb
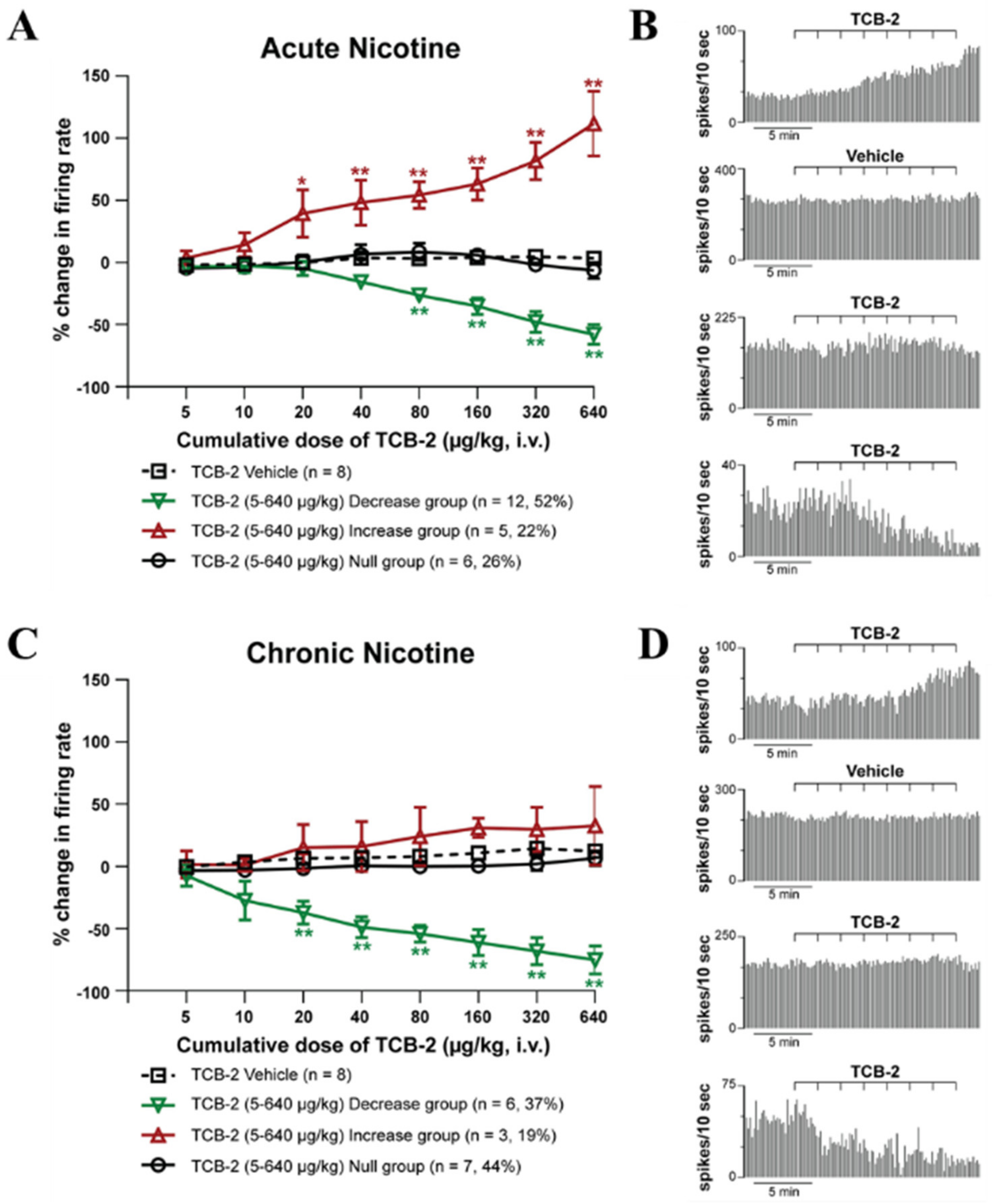
2.2. 5-HT2AR Electrophysiology Experiments
2.2.1. Electrophysiological Characteristics of Spontaneously Active the LHb Neurons
2.2.2. Effect of Systemic Administration of 5-HT2AR Agonist TCB-2 on the Firing Rate and Pattern of LHb Neurons of Acute and Chronic-Treated Rats
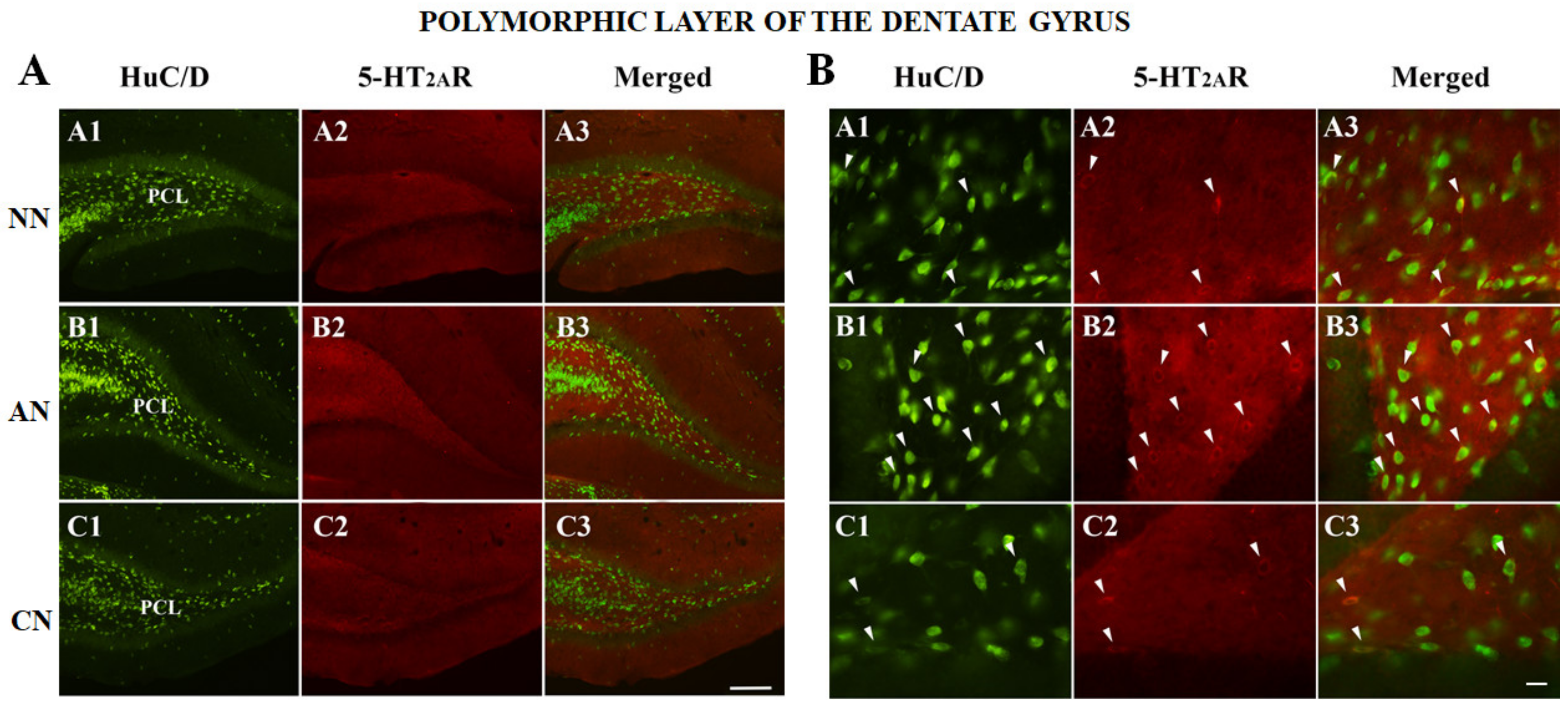
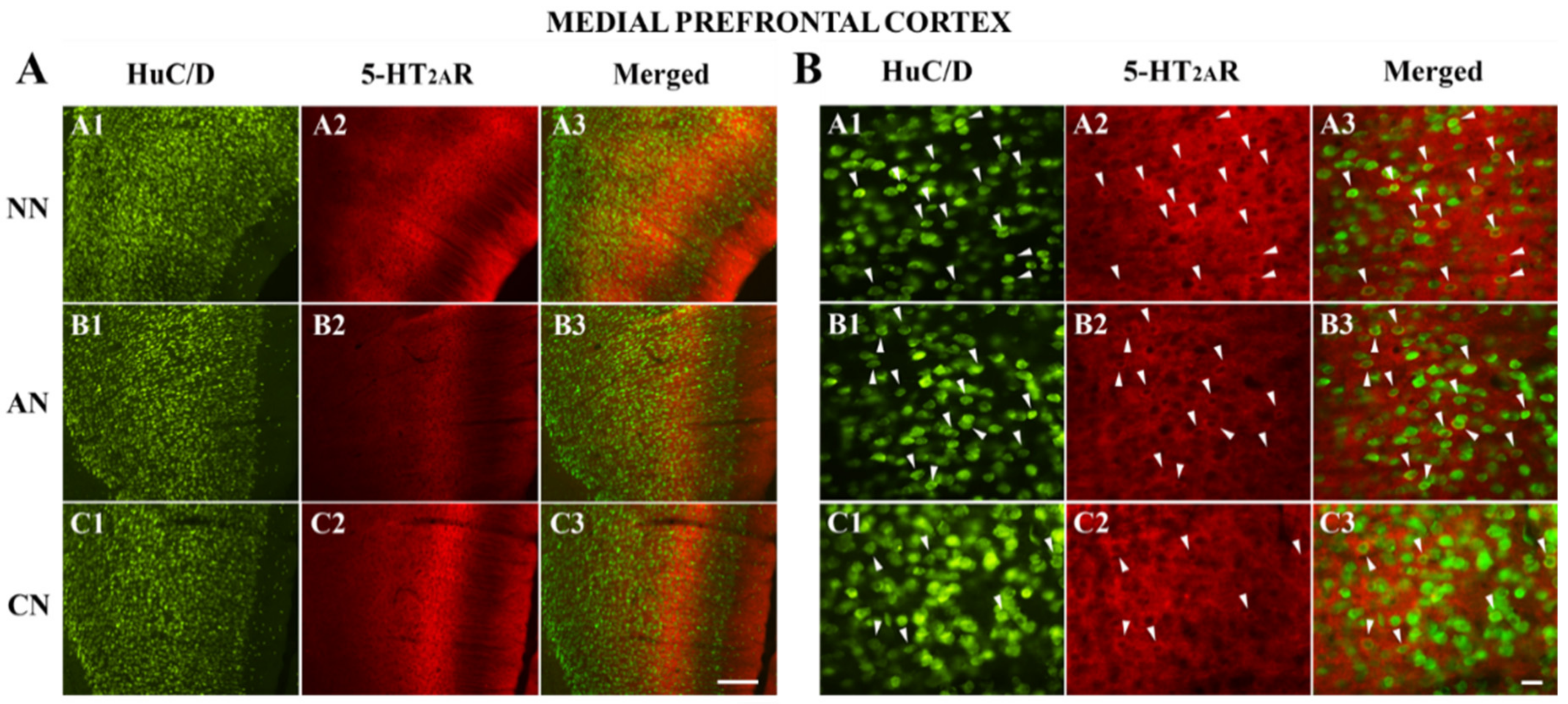
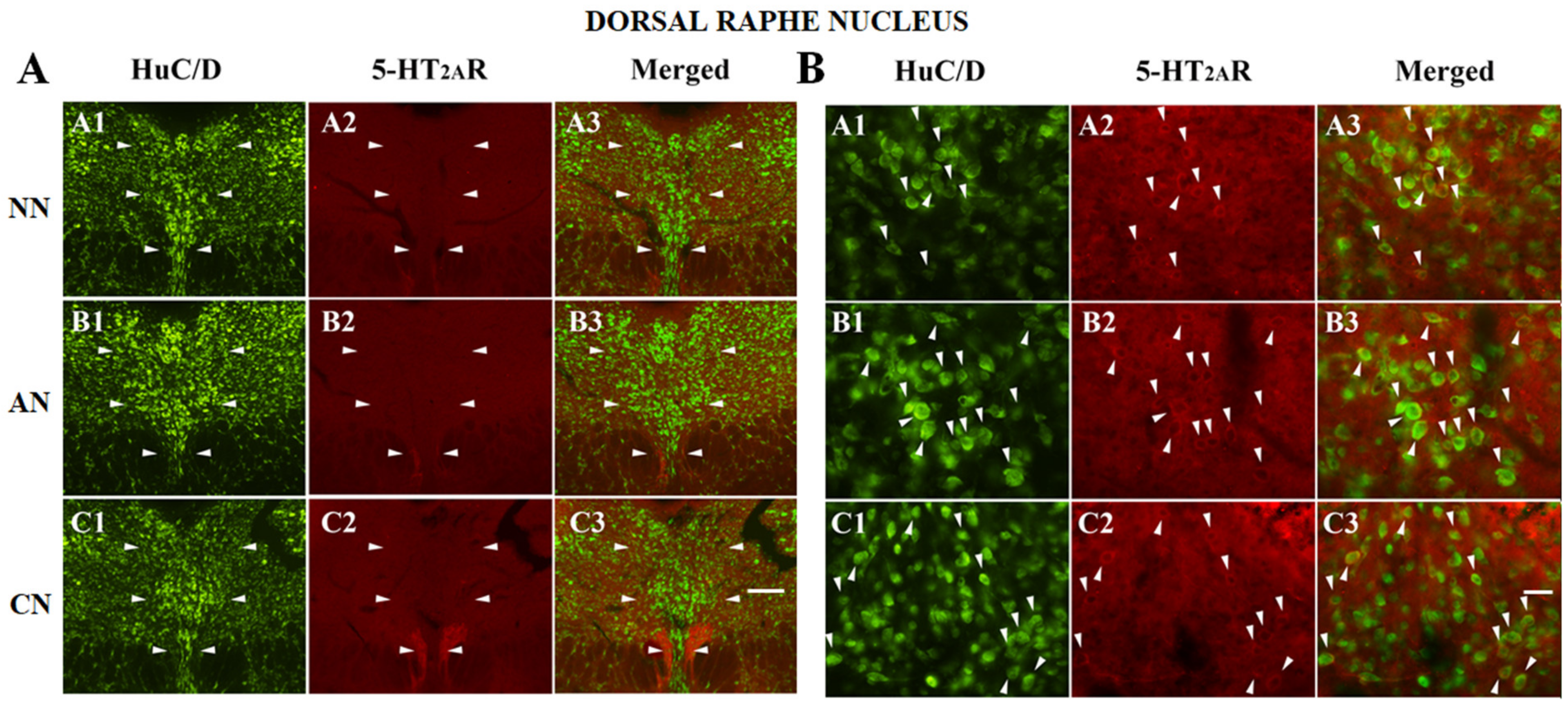
2.3. 5-HT2AR Colocalization Studies in the mPFC, DG, NAc, ST, VTA, SNc, and DRN
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Histological Procedures and Immunocytochemistry
Animals and Fixation
4.3. Immunoperoxidase Experiments
4.4. Double Immunofluorescence Experiments
4.5. Specificity of Antibodies
4.6. Thionin Staining
4.7. Analysis of Sections
4.7.1. Immunoperoxidase Experiments
4.7.2. Double Immunofluorescence Experiments
4.8. Electrophysiological Recordings
4.8.1. Extracellular Single-Unit Recordings
4.8.2. Drugs and Pharmacological Treatments
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| (±)-DOI | 2,5-dimethoxy-4-iodoamphetamine |
| 5-HT | 5-hydroxytryptamine |
| 5-HT2CRs | 5-HT2C receptors |
| 5-HTARs | 5-HTA receptors |
| AN | Acute nicotine |
| CN | Chronic nicotine |
| DA | Dopamine |
| DG | dentate gyrus |
| DRN | dorsal raphe nucleus |
| GABA | Gamma-aminobutyric acid |
| GLU | Glutamate |
| HT2A | 5-HTA receptor gene |
| IHC | immunohistochemistry |
| mPFC | Medial prefrontal cortex |
| NAc | nucleus accumbens |
| nAChRs | Nicotinic cholinergic receptors |
| NN | Naïve nicotine |
| PBS | phosphate-buffered saline |
| SNc | substantia nigra pars compacta |
| ST | Striatum |
| TCB-2 | (4-Bromo-3,6-dimethoxybenzocyclobuten-1-yl)methylamine hydrobromide |
| VTA | Ventral tegmental area |
References
- WHO. WHO Report on the Global Tobacco Epidemic 2017. 2017, WHO/NMH/PND/17.4. Available online: https://www.who.int/tobacco/global_report/2017/en/ (accessed on 6 March 2020).
- Leone, A.; Landini, L.; Leone, A. What is Tobacco Smoke? Sociocultural Dimensions of the Association with Cardiovascular Risk. Curr. Pharm. Des. 2010, 16, 2510–2517. [Google Scholar] [CrossRef]
- Office on Smoking and Health. Surgeon General’s Report: The Health Consequences of Smoking—50 Years of Progress. Available online: https://www.cdc.gov/tobacco/data_statistics/sgr/50th-anniversary/index.htm (accessed on 20 May 2019).
- Di Giovanni, G. Nicotine Addiction: Prevention, Health Effects and Treatment Options; Nova Science Publishers: New York, NT, USA, 2012; pp. 1–283. [Google Scholar]
- Rose, J.E. Nicotine and nonnicotine factors in cigarette addiction. Psychophamacology (Berl.) 2006, 184, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Stolerman, I.P.; Jarvis, M.J. The scientific case that nicotine is addictive. Psychopharmacology (Berl.) 1995, 117, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Di Matteo, V.; Pierucci, M.; Di Giovanni, G.; Benigno, A.; Esposito, E. The neurobiological bases for the pharmacotherapy of nicotine addiction. Curr. Pharm. Des. 2007, 13, 1269–1284. [Google Scholar] [CrossRef] [PubMed]
- Corrigall, W.A.; Franklin, K.B.; Coen, K.M.; Clarke, P.B. The mesolimbic dopaminergic system is implicated in the reinforcing effects of nicotine. Psychopharmacology 1992, 107, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Di Chiara, G. Role of dopamine in the behavioural actions of nicotine related to addiction. Eur. J. Pharmacol. 2000, 393, 295–314. [Google Scholar] [CrossRef]
- Alasmari, F.; Al-Rejaie, S.S.; AlSharari, S.D.; Sari, Y. Targeting glutamate homeostasis for potential treatment of nicotine dependence. Brain Res. Bull. 2016, 121, 1–8. [Google Scholar] [CrossRef]
- Di Matteo, V.; Pierucci, M.; Benigno, A.; Esposito, E.; Crescimanno, G.; Di Giovanni, G. Critical role of nitric oxide on nicotine-induced hyperactivation of dopaminergic nigrostriatal system: Electrophysiological and neurochemical evidence in rats. CNS Neurosci. Ther. 2010, 16, 127–136. [Google Scholar] [CrossRef]
- D’Souza, M.S.; Markou, A. The "stop" and "go" of nicotine dependence: role of GABA and glutamate. Cold Spring Harb. Perspect. Med. 2013, 3. [Google Scholar] [CrossRef]
- Lester, R.J. Nicotinic Receptors; Springer International Publishing: New York, NY, USA, 2014; pp. 1–461. [Google Scholar]
- Pierucci, M.; Di Matteo, V.; Esposito, E. Stimulation of Serotonin2C Receptors Blocks the Hyperactivation of Midbrain Dopamine Neurons Induced by Nicotine Administration. J. Pharmacol. Exp. Ther. 2004, 309, 109–118. [Google Scholar] [CrossRef]
- Grottick, A.J.; Corrigall, W.A.; Higgins, G.A. Activation of 5-HT(2C) receptors reduces the locomotor and rewarding effects of nicotine. Psychopharmacology 2001, 157, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Higgins, G.A.; Fletcher, P.J. Therapeutic Potential of 5-HT2C Receptor Agonists for Addictive Disorders. ACS Chem. Neurosci. 2015, 6, 1071–1088. [Google Scholar] [CrossRef] [PubMed]
- Pierucci, M.; Chambers, S.; Partridge, L.; De Deurwaerdère, P.; Di Giovanni, G. Role of central serotonin receptors in nicotine addiction. In Nicotinic Receptors; Springer International Publishing: New York, NY, USA, 2014; pp. 279–305. [Google Scholar]
- De Deurwaerdere, P.; Di Giovanni, G. Serotonergic modulation of the activity of mesencephalic dopaminergic systems: Therapeutic implications. Prog. Neurobiol. 2017, 151, 175–236. [Google Scholar] [CrossRef] [PubMed]
- Di Giovanni, G.; De Deurwaerdere, P.; Di Mascio, M.; Di Matteo, V.; Esposito, E.; Spampinato, U. Selective blockade of serotonin-2C/2B receptors enhances mesolimbic and mesostriatal dopaminergic function: a combined in vivo electrophysiological and microdialysis study. Neuroscience 1999, 91, 587–597. [Google Scholar] [CrossRef]
- Fletcher, P.J.; Rizos, Z.; Noble, K.; Soko, A.D.; Silenieks, L.B.; Lê, A.D.; Higgins, G.A. Effects of the 5-HT2C receptor agonist Ro60-0175 and the 5-HT2A receptor antagonist M100907 on nicotine self-administration and reinstatement. Neuropharmacology 2012, 62, 2288–2298. [Google Scholar] [CrossRef]
- Zaniewska, M.; McCreary, A.C.; Wydra, K.; Filip, M. Effects of serotonin (5-HT) 2 receptor ligands on depression-like behavior during nicotine withdrawal. Neuropharmacology 2010, 58, 1140–1146. [Google Scholar] [CrossRef]
- Rose, J.E.; Behm, F.M.; Salley, A.N.; Bates, J.E.; Coleman, R.E.; Hawk, T.C.; Turkington, T.G. Regional brain activity correlates of nicotine dependence. Neuropsychopharmacology 2007, 32, 2441. [Google Scholar] [CrossRef]
- Bombardi, C. Brain Distribution. In 5-HT2A Receptors in the Central Nervous System; Guiard, B.P., Di Giovanni, G., Eds.; Springer International Publishing: New York, NY, USA, 2018; pp. 67–84. [Google Scholar]
- Zaniewska, M.; McCreary, A.C.; Wydra, K.; Faron-Górecka, A.; Filip, M. Context-controlled nicotine-induced changes in the labeling of serotonin (5-HT)2A and 5-HT2C receptors in the rat brain. Pharmacol. Rep. 2015, 67, 451–459. [Google Scholar] [CrossRef]
- Bianco, I.H.; Wilson, S.W. The habenular nuclei: a conserved asymmetric relay station in the vertebrate brain. Philos. Trans. R. Soc. Lond. 2009, 364, 1005–1020. [Google Scholar] [CrossRef]
- Pierucci, M.; Pitruzzella, A.; Valentino, M.; Zammit, C.; Muscat, R.; Benigno, A.; Di Giovanni, G. Lateral Habenula contribution in nicotine addiction: Focus on dopamine, GABA and serotonin interactions. Malta Medical Journal 2011, 23, 28–32. [Google Scholar]
- Dao, D.Q.; Perez, E.E.; Teng, Y.; Dani, J.A.; De Biasi, M. Nicotine enhances excitability of medial habenular neurons via facilitation of neurokinin signaling. J. Neurosci. 2014, 34, 4273–4284. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, K.M.; Molfese, D.L.; Salas, R. The role of the habenula in drug addiction. Front. Hum. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Casarrubea, M.; Davies, C.; Faulisi, F.; Pierucci, M.; Colangeli, R.; Partridge, L.; Chambers, S.; Cassar, D.; Valentino, M.; Muscat, R.; et al. Acute nicotine induces anxiety and disrupts temporal pattern organization of rat exploratory behavior in hole-board: A potential role for the lateral habenula. Front. Cell. Neurosci. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Metzger, M.; Bueno, D.; Lima, L.B. The lateral habenula and the serotonergic system. Pharmacol. Biochem. Behav. 2017, 162, 22–28. [Google Scholar] [CrossRef]
- Tchenio, A.; Valentinova, K.; Mameli, M. Can the Lateral Habenula Crack the Serotonin Code? Front. Synaptic Neurosci. 2016, 8, 34. [Google Scholar] [CrossRef]
- Di Giovanni, G.; De Deurwaerdere, P. TCB-2 [(7R)-3-bromo-2, 5-dimethoxy-bicyclo[4.2.0]octa-1,3,5-trien-7-yl]methanamine]: A hallucinogenic drug, a selective 5-HT2A receptor pharmacological tool, or none of the above? Neuropharmacology 2017. [Google Scholar] [CrossRef]
- McLean, T.H.; Parrish, J.C.; Braden, M.R.; Marona-Lewicka, D.; Gallardo-Godoy, A.; Nichols, D.E. 1-Aminomethylbenzocycloalkanes: conformationally restricted hallucinogenic phenethylamine analogues as functionally selective 5-HT2A receptor agonists. J. Med. Chem. 2006, 49, 5794–5803. [Google Scholar] [CrossRef]
- Delicata, F.; Bombardi, C.; Pierucci, M.; Di Maio, R.; De Deurwaerdere, P.; Di Giovanni, G. Preferential modulation of the lateral habenula activity by serotonin-2A rather than -2C receptors: Electrophysiological and neuroanatomical evidence. CNS Neurosci.Ther. 2018, 24, 721–733. [Google Scholar] [CrossRef]
- Fletcher, P.J.; Le, A.D.; Higgins, G.A. Serotonin receptors as potential targets for modulation of nicotine use and dependence. Prog. Brain Res. 2008, 172, 361–383. [Google Scholar] [CrossRef]
- Hernandez-Lopez, S.; Garduno, J.; Mihailescu, S. Nicotinic modulation of serotonergic activity in the dorsal raphe nucleus. Rev. Neurosci. 2013, 24, 455–469. [Google Scholar] [CrossRef]
- Wagner, F.; Bernard, R.; Derst, C.; French, L.; Veh, R.W. Microarray analysis of transcripts with elevated expressions in the rat medial or lateral habenula suggest fast GABAergic excitation in the medial habenula and habenular involvement in the regulation of feeding and energy balance. Brain Struct. Funct. 2016, 221, 4663–4689. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, K.; Czachura, J.F. Nicotine withdrawal leads to increased sensitivity of serotonergic neurons to the 5-HT1A agonist 8-OH-DPAT. Psychopharmacology (Berl) 1997, 133, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Sperling, R.; Commons, K.G. Shifting topographic activation and 5-HT1A receptor-mediated inhibition of dorsal raphe serotonin neurons produced by nicotine exposure and withdrawal. J. Neurosci. 2011, 33, 1866–1875. [Google Scholar] [CrossRef] [PubMed]
- Zuo, W.; Xiao, C.; Gao, M.; Hopf, F.W.; Krnjevic, K.; McIntosh, J.M.; Fu, R.; Wu, J.; Bekker, A.; Ye, J.H. Nicotine regulates activity of lateral habenula neurons via presynaptic and postsynaptic mechanisms. Sci. Rep. 2016, 6, 32937. [Google Scholar] [CrossRef]
- Xie, G.; Zuo, W.; Wu, L.; Li, W.; Wu, W.; Bekker, A.; Ye, J.-H. Serotonin modulates glutamatergic transmission to neurons in the lateral habenula. Sci. Rep. 2016, 6, 23798. [Google Scholar] [CrossRef]
- Zuo, W.; Zhang, Y.; Xie, G.; Gregor, D.; Bekker, A.; Ye, J.H. Serotonin stimulates lateral habenula via activation of the post-synaptic serotonin 2/3 receptors and transient receptor potential channels. Neuropharmacology 2016, 101, 449–459. [Google Scholar] [CrossRef]
- Singer, S.; Rossi, S.; Verzosa, S.; Hashim, A.; Lonow, R.; Cooper, T.; Sershen, H.; Lajtha, A. Nicotine-induced changes in neurotransmitter levels in brain areas associated with cognitive function. Neurochem.Res. 2004, 29, 1779–1792. [Google Scholar] [CrossRef]
- Cornea-Hebert, V.; Riad, M.; Wu, C.; Singh, S.K.; Descarries, L. Cellular and subcellular distribution of the serotonin 5-HT2A receptor in the central nervous system of adult rat. J. Comp. Neurol. 1999, 409, 187–209. [Google Scholar] [CrossRef]
- Leysen, J.E.; Eens, A.; Gommeren, W.; van Gompel, P.; Wynants, J.; Janssen, P.A. Identification of nonserotonergic [3H]ketanserin binding sites associated with nerve terminals in rat brain and with platelets; relation with release of biogenic amine metabolites induced by ketanserin- and tetrabenazine-like drugs. J. Pharmacol. Exp. Ther. 1988, 244, 310–321. [Google Scholar]
- Miguelez, C.; Morera-Herreras, T.; De Deurwaerdère, P. 5-HT2A receptors in the Basal Ganglia. In 5-HT2A Receptors in the Central Nervous System; Guiard, B., Di Giovanni, G., Eds.; Springer International Publishing: New York, NY, USA, 2018; Volume 32, pp. 273–310. [Google Scholar]
- Graziane, N.M.; Neumann, P.A.; Dong, Y. A Focus on Reward Prediction and the Lateral Habenula: Functional Alterations and the Behavioral Outcomes Induced by Drugs of Abuse. Front. Synaptic Neurosci 2018, 10. [Google Scholar] [CrossRef]
- Lecca, S.; Melis, M.; Luchicchi, A.; Ennas, M.G.; Castelli, M.P.; Muntoni, A.L.; Pistis, M. Effects of drugs of abuse on putative rostromedial tegmental neurons, inhibitory afferents to midbrain dopamine cells. Neuropsychopharmacology 2011, 36, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.D.; Kenny, P.J. Nicotine aversion: Neurobiological mechanisms and relevance to tobacco dependence vulnerability. Neuropharmacology 2014, 76, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Olausson, P.; Akesson, P.; Engel, J.A.; Soderpalm, B. Effects of 5-HT1A and 5-HT2 receptor agonists on the behavioral and neurochemical consequences of repeated nicotine treatment. Eur. J. Pharmacol. 2001, 420, 45–54. [Google Scholar] [CrossRef]
- Batman, A.M.; Munzar, P.; Beardsley, P.M. Attenuation of nicotine’s discriminative stimulus effects in rats and its locomotor activity effects in mice by serotonergic 5-HT2A/2C receptor agonists. Psychopharmacology (Berl) 2005, 179, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Zaniewska, M.; McCreary, A.C.; Przegalinski, E.; Filip, M. Effects of the serotonin 5-HT2A and 5-HT2C receptor ligands on the discriminative stimulus effects of nicotine in rats. Eur. J. Pharmacol. 2007, 571, 156–165. [Google Scholar] [CrossRef]
- Levin, E.; Icenogle, L.; Farzad, A. Ketanserin attenuates nicotine-induced working memory improvement in rats. Pharmacol. Biochem. Behav. 2005, 82, 289–292. [Google Scholar] [CrossRef]
- Rezvani, A.H.; Caldwell, D.P.; Levin, E.D. Nicotinic-serotonergic drug interactions and attentional performance in rats. Psychopharmacology 2005, 179, 521–528. [Google Scholar] [CrossRef]
- Zaniewska, M.; McCreary, A.C.; Wydra, K.; Filip, M. Differential effects of serotonin (5-HT)2 receptor-targeting ligands on locomotor responses to nicotine-repeated treatment. Synapse (New York, N.Y.) 2010, 64, 511–519. [Google Scholar] [CrossRef]
- Kutlu, M.G.; Gould, T.J. Nicotinic modulation of hippocampal cell signaling and associated effects on learning and memory. Physiol. Behav. 2016, 155, 162–171. [Google Scholar] [CrossRef]
- Meneses, A. 5-HT systems: emergent targets for memory formation and memory alterations. Rev. Neurosci. 2013, 24, 629–664. [Google Scholar] [CrossRef]
- Bombardi, C.; Di Giovanni, G. Functional anatomy of 5-HT2A receptors in the amygdala and hippocampal complex: relevance to memory functions. Exp. Brain Res. 2013, 230, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Afshar, S.; Shahidi, S.; Rohani, A.H.; Komaki, A.; Asl, S.S. The effect of NAD-299 and TCB-2 on learning and memory, hippocampal BDNF levels and amyloid plaques in Streptozotocin-induced memory deficits in male rats. Psychopharmacology 2018, 235, 2809–2822. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Asgeirsdottir, H.N.; Cohen, S.J.; Munchow, A.H.; Barrera, M.P.; Stackman, R.W., Jr. Stimulation of serotonin 2A receptors facilitates consolidation and extinction of fear memory in C57BL/6J mice. Neuropharmacology 2013, 64, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Bekinschtein, P.; Renner, M.C.; Gonzalez, M.C.; Weisstaub, N. Role of medial prefrontal cortex serotonin 2A receptors in the control of retrieval of recognition memory in rats. J. Neurosci. 2013, 33, 15716–15725. [Google Scholar] [CrossRef] [PubMed]
- Malin, D.; Henceroth, M.; Rao, G.S.; Campbell, J.; Ma, J.-N.; Tsai, P.-H.; Kishbaugh, J.C.; Burstein, E.S. Inverse agonists of the 5-HT2A receptor reduce nicotine withdrawal signs in rats. Neurosci. Lett. 2019, 713, 134524. [Google Scholar] [CrossRef]
- Zaniewska, M.; Alenina, N.; Wydra, K.; Frohler, S.; Kusmider, M.; McCreary, A.C.; Chen, W.; Bader, M.; Filip, M. Discovering the mechanisms underlying serotonin (5-HT)2A and 5-HT2C receptor regulation following nicotine withdrawal in rats. J. Neurochem. 2015, 134, 704–716. [Google Scholar] [CrossRef]
- Lewis, D.A.; Campbell, M.J.; Morrison, J.H. An immunohistochemical characterization of somatostatin-28 and somatostatin-281-12 in monkey prefrontal cortex. J. Comp. Neurol. 1986, 248, 1–18. [Google Scholar] [CrossRef]
- Weber, E.T.; Andrade, R. Htr2a Gene and 5-HT(2A) Receptor Expression in the Cerebral Cortex Studied Using Genetically Modified Mice. Front. Neurosci. 2010, 4, 36. [Google Scholar] [CrossRef]
- Nocjar, C.; Alex, K.D.; Sonneborn, A.; Abbas, A.I.; Roth, B.L.; Pehek, E.A. Serotonin-2C and -2a receptor co-expression on cells in the rat medial prefrontal cortex. Neuroscience 2015, 297, 22–37. [Google Scholar] [CrossRef]
- Marusich, M.F.; Furneaux, H.M.; Henion, P.D.; Weston, J.A. Hu neuronal proteins are expressed in proliferating neurogenic cells. J. Neurobiol. 1994, 25, 143–155. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 6th ed.; Academic Press: New York, NY, USA, 2007; p. 456. [Google Scholar]
- Casarrubea, M.; Pierucci, M.; Aiello, S.; Cassar, D.; Deidda, G.; Crescimanno, G.; Di Giovanni, G. Effects of chronic nicotine on the temporal structure of anxiety-related behavior in rats tested in hole-board. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019. [Google Scholar] [CrossRef] [PubMed]
- Di Giovanni, G.; De Deurwaerdere, P. New therapeutic opportunities for 5-HT2C receptor ligands in neuropsychiatric disorders. Pharmacol. Ther. 2016, 157, 125–162. [Google Scholar] [CrossRef] [PubMed]
- Venzi, M.; David, F.; Bellet, J.; Cavaccini, A.; Bombardi, C.; Crunelli, V.; Di Giovanni, G. Role for serotonin2A (5-HT2A) and 2C (5-HT2C) receptors in experimental absence seizures. Neuropharmacology 2016, 108, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, R.W.; Pierucci, M.; Calcagno, E.; Di Giovanni, G.; Di Matteo, V.; Benigno, A.; Esposito, E. Selective activation of 5-HT(2C) receptors stimulates GABA-ergic function in the rat substantia nigra pars reticulata: A combined in vivo electrophysiological and neurochemical study. Neuroscience 2007, 144, 1523–1535. [Google Scholar] [CrossRef] [PubMed]
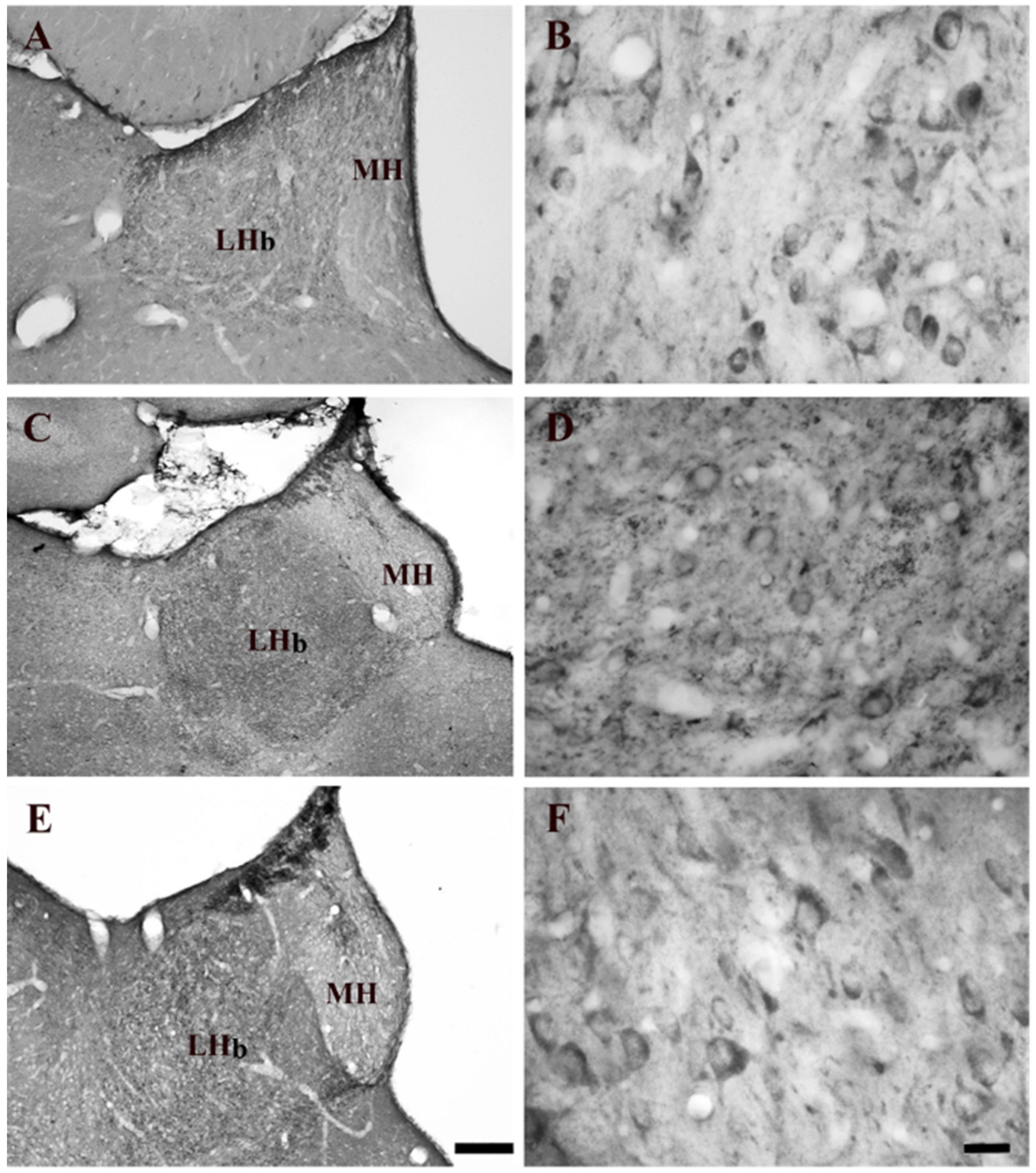

| Lateral Habenula (LHb) | Naïve Nicotine (NN) | Acute Nicotine (AN) | Chronic Nicotine (CN) |
|---|---|---|---|
| Density 5-HT2AR-IR neurons | 57.1 ± 12.3 | 52.2 ± 13.3 | 51.4 ± 13.9 |
| HuC/D-IR neurons | 600 | 608 | 664 |
| HuC/D/5-HT2AR-IR | 64 | 56 | 56 |
| % of 5-HT2AR-IR | 9.6% (64/664) | 8.4% (56/664) | 7.8% (56/720) |
| % area covered by 5-HT2AR-IR | 53.5 ± 8.8 | 67.7 ± 15.8 * | 58.4 ± 11.6 |
| Medial Prefrontal Cortex (mPFC) | Naïve Nicotine (NN) | Acute Nicotine (AN) | Chronic Nicotine (CN) |
|---|---|---|---|
| HuC/D-IR neurons | 242 | 189 | 317 |
| HuC/D/5-HT2AR-IR | 454 | 379 | 443 |
| % of 5-HT2AR-IR | 65.2% (454/696) | 66.7% (379/568) | 58.3% (443/760) |
| % area covered by 5-HT2AR-IR | 75.9 ± 14.1 | 63.3 ± 14.1 * | 76.1 ± 13.4 ^ |
| Dentate Gyrus (DG) | NN | AN | CN |
| HuC/D-IR neurons | 192 | 138 | 172 |
| HuC/D/5-HT2AR-IR | 44 | 99 | 36 |
| % of 5-HT2AR-IR | 18.6% (44/236) | 41.8% (99/237) * | 17.3% (36/208) |
| % area covered by 5-HT2AR-IR | 57.6 ± 12.9 | 69.6 ± 19.7 | 52.1 ± 15.8 ^ |
| Nucleus Accumbens (NAc) | NN | AN | CN |
| HuC/D-IR neurons | 33 | 31 | 36 |
| HuC/D/5-HT2AR-IR | 59 | 49 | 52 |
| % of 5-HT2AR-IR | 64.1% (59/92) | 61.3% (49/80) | 59.1% (52/88) |
| % area covered by 5-HT2AR-IR | 27.4 ± 8.9 | 33.6 ± 11.9 | 46.7 ± 25.4 * |
| Striatum (ST) | NN | AN | CN |
| HuC/D-IR neurons | 298 | 302 | 256 |
| HuC/D/5-HT2AR-IR | 302 | 319 | 296 |
| % of 5-HT2AR-IR | 50.3% (302/600) | 51.4% (319/621) | 53.6% (296/552) |
| % area covered by 5-HT2AR-IR | 71.8 ± 13.5 | 77.9 ± 14.1 | 67.1 ± 12.4 ^ |
| Ventral Tegmental Area (VTA) | NN | AN | CN |
| HuC/D-IR neurons | 88 | 97 | 104 |
| HuC/D/5-HT2AR-IR | 24 | 47 | 32 |
| % of 5-HT2AR-IR | 21.4% (24/112) | 32.6% (47/144) | 23.5% (32/136) |
| % area covered by 5-HT2AR-IR | 45.4 ± 12.2 | 59.2 ± 14.9 * | 54.3 ± 16.2 |
| Substantia Nigra Pars Compacta (SNc) | NN | AN | CN |
| HuC/D-IR neurons | 184 | 205 | 232 |
| HuC/D/5-HT2AR-IR | 112 | 91 | 108 |
| % of 5-HT2AR-IR | 37.8% (112/296) | 30.7% (91/296) | 31.8% (108/340) |
| % area covered by 5-HT2AR-IR | 42.8 ± 12.4 | 49.9 ± 13.1 | 49.1 ± 14.5 |
| Dorsal raphe Nucleus (DRN) | NN | AN | CN |
| HuC/D-IR neurons | 368 | 284 | 264 |
| HuC/D/5-HT2AR-IR | 92 | 104 | 132 |
| % of 5-HT2AR-IR | 20% (92/460) | 26.8% (104/388) | 33.3% (132/396) |
| % area covered by 5-HT2AR-IR | 19.4 ± 5.2 | 20.1 ± 5.9 | 19.1 ± 4.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bombardi, C.; Delicata, F.; Tagliavia, C.; Pierucci, M.; Deidda, G.; Casarrubea, M.; De Deurwaerdère, P.; Di Giovanni, G. Acute and Chronic Nicotine Exposures Differentially Affect Central Serotonin 2A Receptor Function: Focus on the Lateral Habenula. Int. J. Mol. Sci. 2020, 21, 1873. https://doi.org/10.3390/ijms21051873
Bombardi C, Delicata F, Tagliavia C, Pierucci M, Deidda G, Casarrubea M, De Deurwaerdère P, Di Giovanni G. Acute and Chronic Nicotine Exposures Differentially Affect Central Serotonin 2A Receptor Function: Focus on the Lateral Habenula. International Journal of Molecular Sciences. 2020; 21(5):1873. https://doi.org/10.3390/ijms21051873
Chicago/Turabian StyleBombardi, Cristiano, Francis Delicata, Claudio Tagliavia, Massimo Pierucci, Gabriele Deidda, Maurizio Casarrubea, Philippe De Deurwaerdère, and Giuseppe Di Giovanni. 2020. "Acute and Chronic Nicotine Exposures Differentially Affect Central Serotonin 2A Receptor Function: Focus on the Lateral Habenula" International Journal of Molecular Sciences 21, no. 5: 1873. https://doi.org/10.3390/ijms21051873
APA StyleBombardi, C., Delicata, F., Tagliavia, C., Pierucci, M., Deidda, G., Casarrubea, M., De Deurwaerdère, P., & Di Giovanni, G. (2020). Acute and Chronic Nicotine Exposures Differentially Affect Central Serotonin 2A Receptor Function: Focus on the Lateral Habenula. International Journal of Molecular Sciences, 21(5), 1873. https://doi.org/10.3390/ijms21051873








