The Urinary Exosomal miRNA Expression Profile is Predictive of Clinical Response in Lupus Nephritis
Abstract
1. Introduction
2. Results
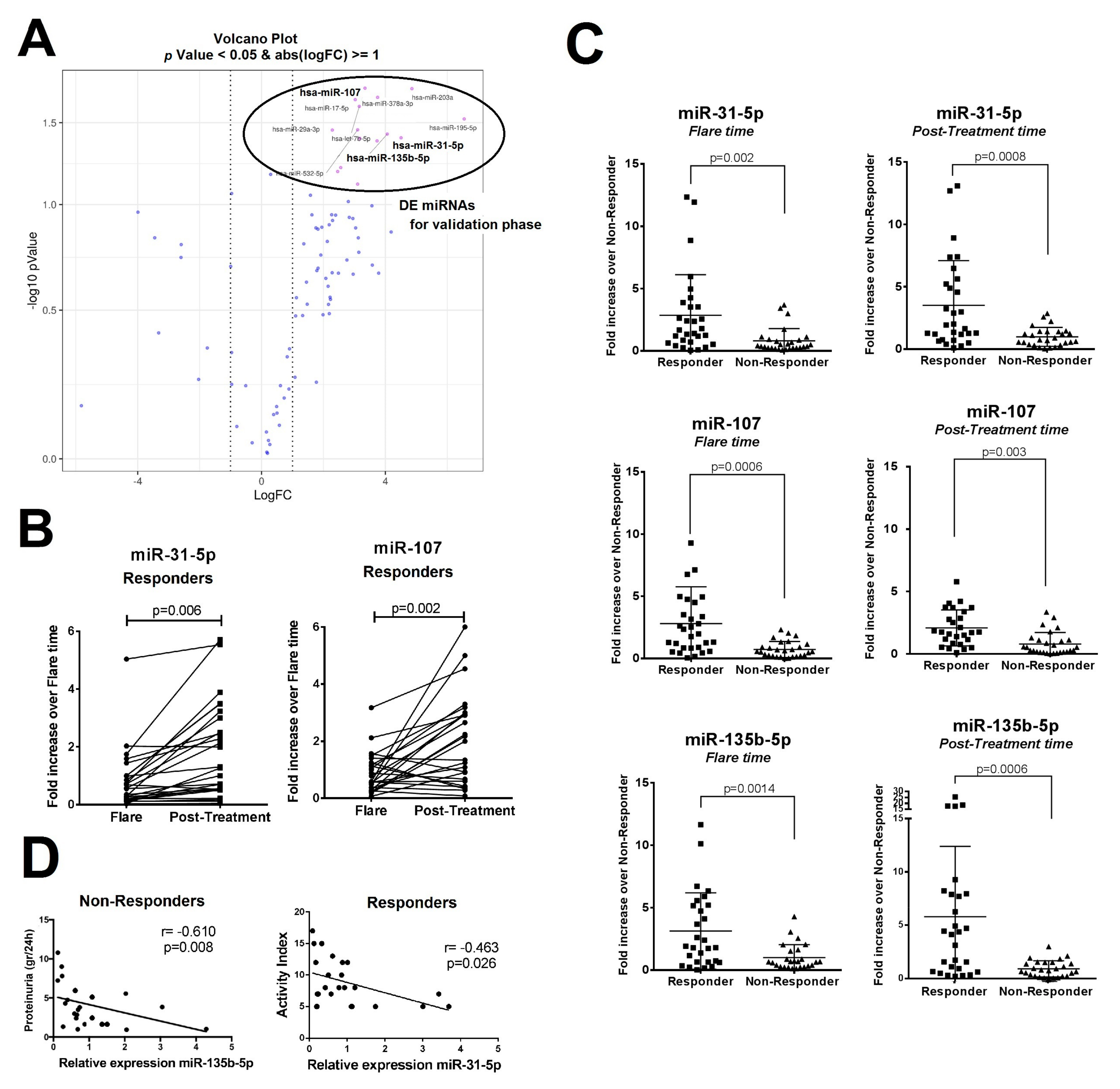
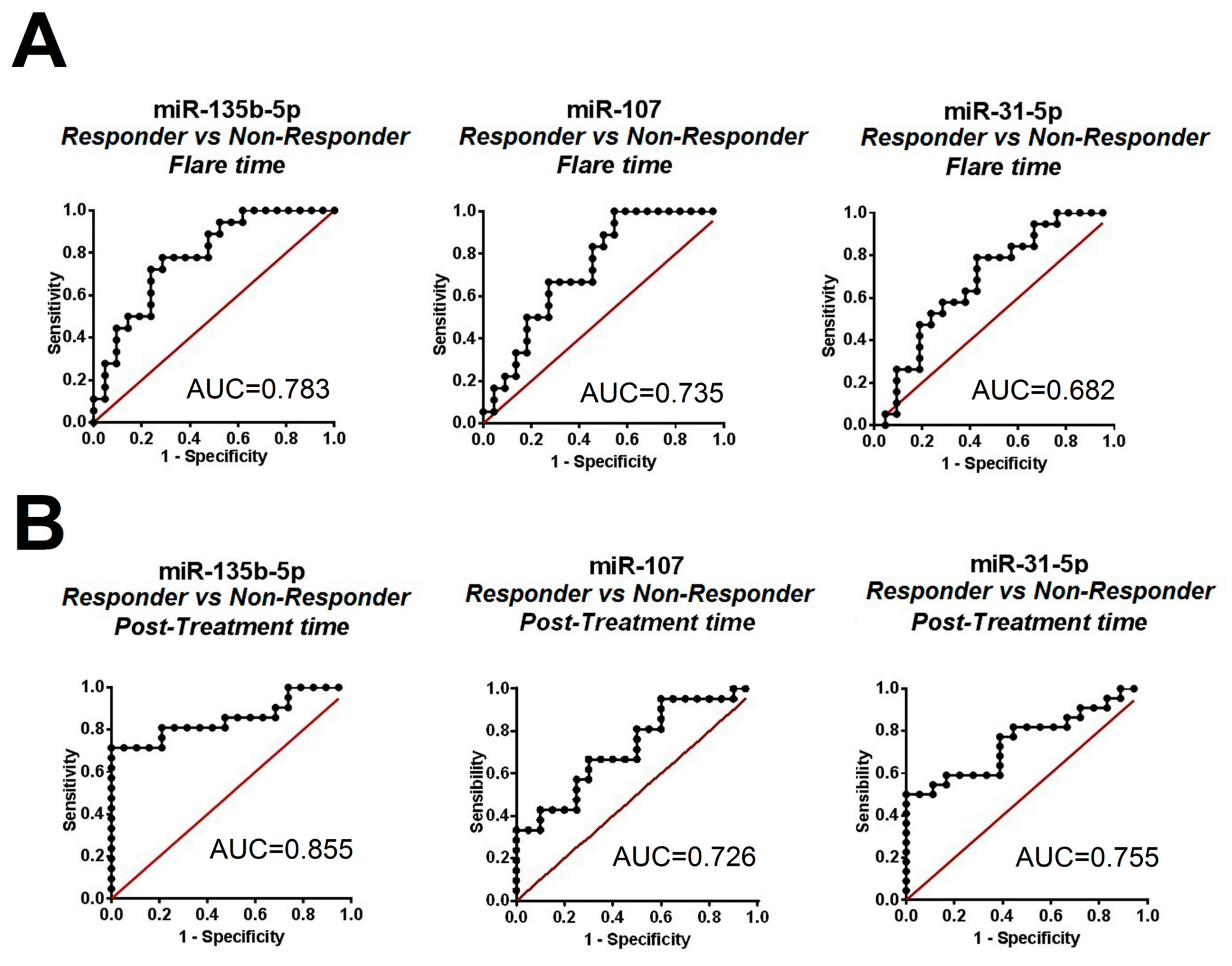
2.1. Differentially Expressed Urinary Exosomal miRNA Profile in Patients with LN According to Clinical Renal Outcome
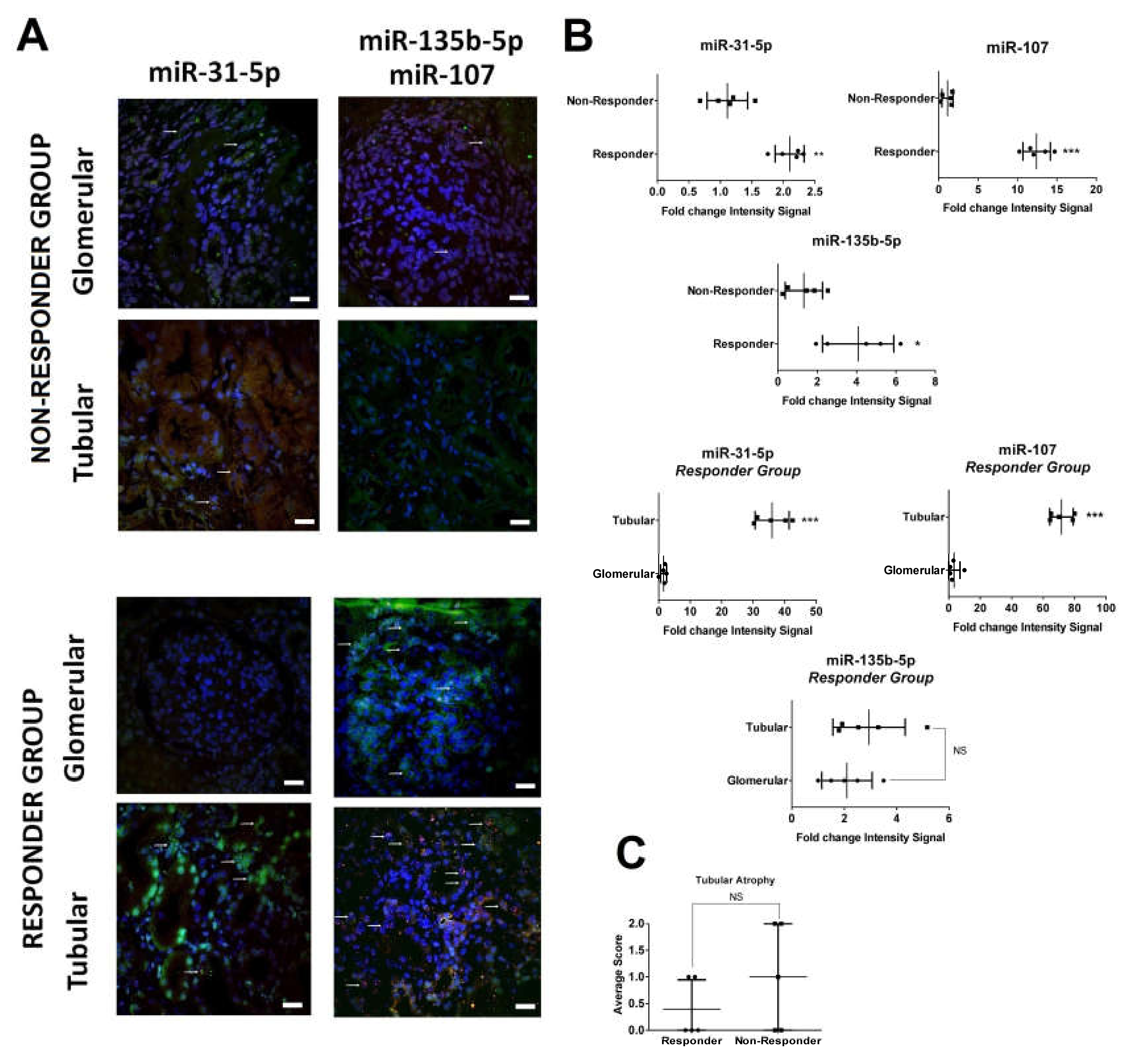
2.2. In Situ Renal Tissue Hybridization
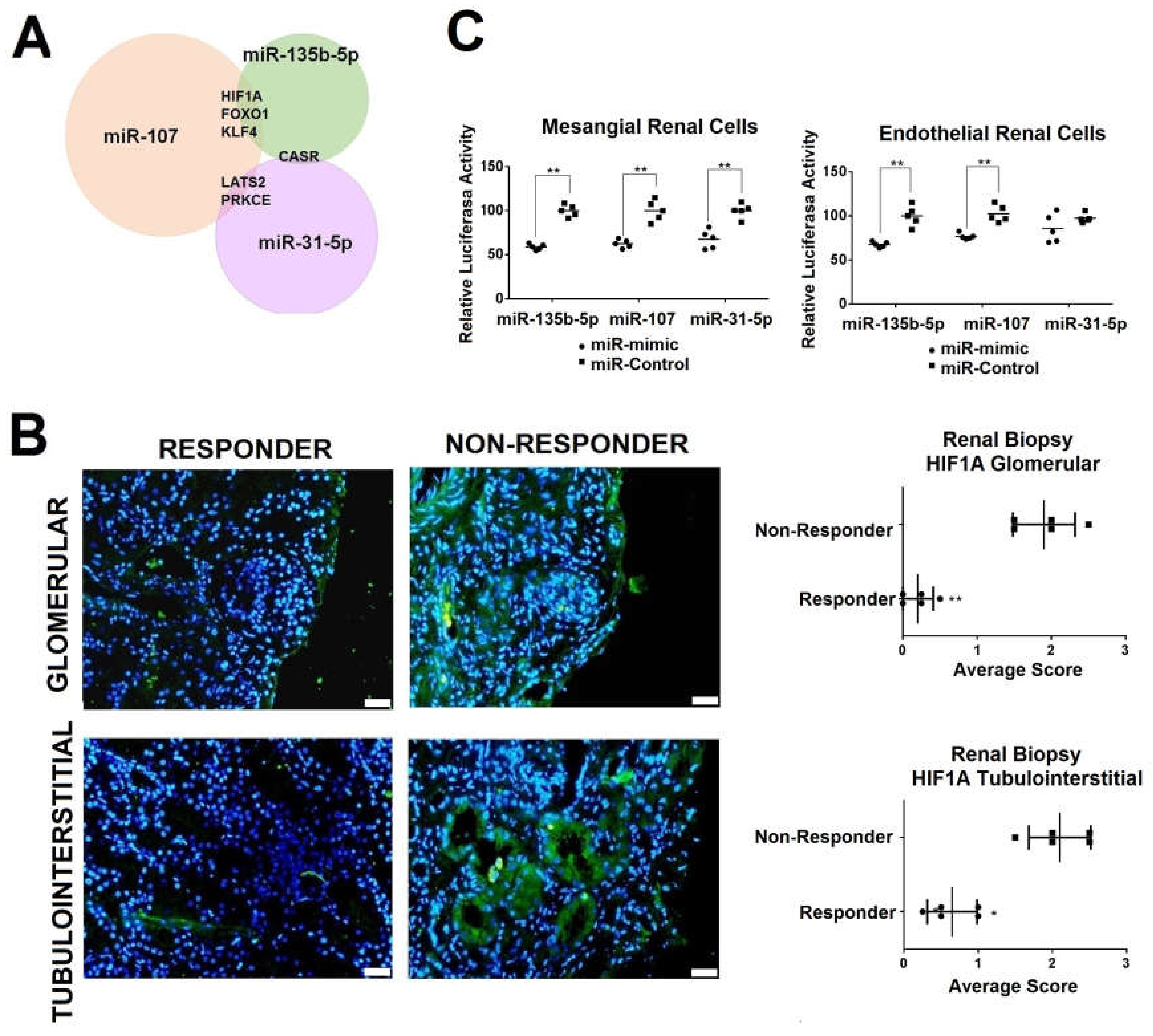
2.3. Urinary Exosome-Derived miR-31-5p, miR-107 and miR-135b-5p Are Secreted by Stimulated Tubular Cells and Internalized Mainly by Mesangial Cells
2.4. Renal HIF1A Expression is Reduced in Responder LN Patients via Exosomal miRNAs
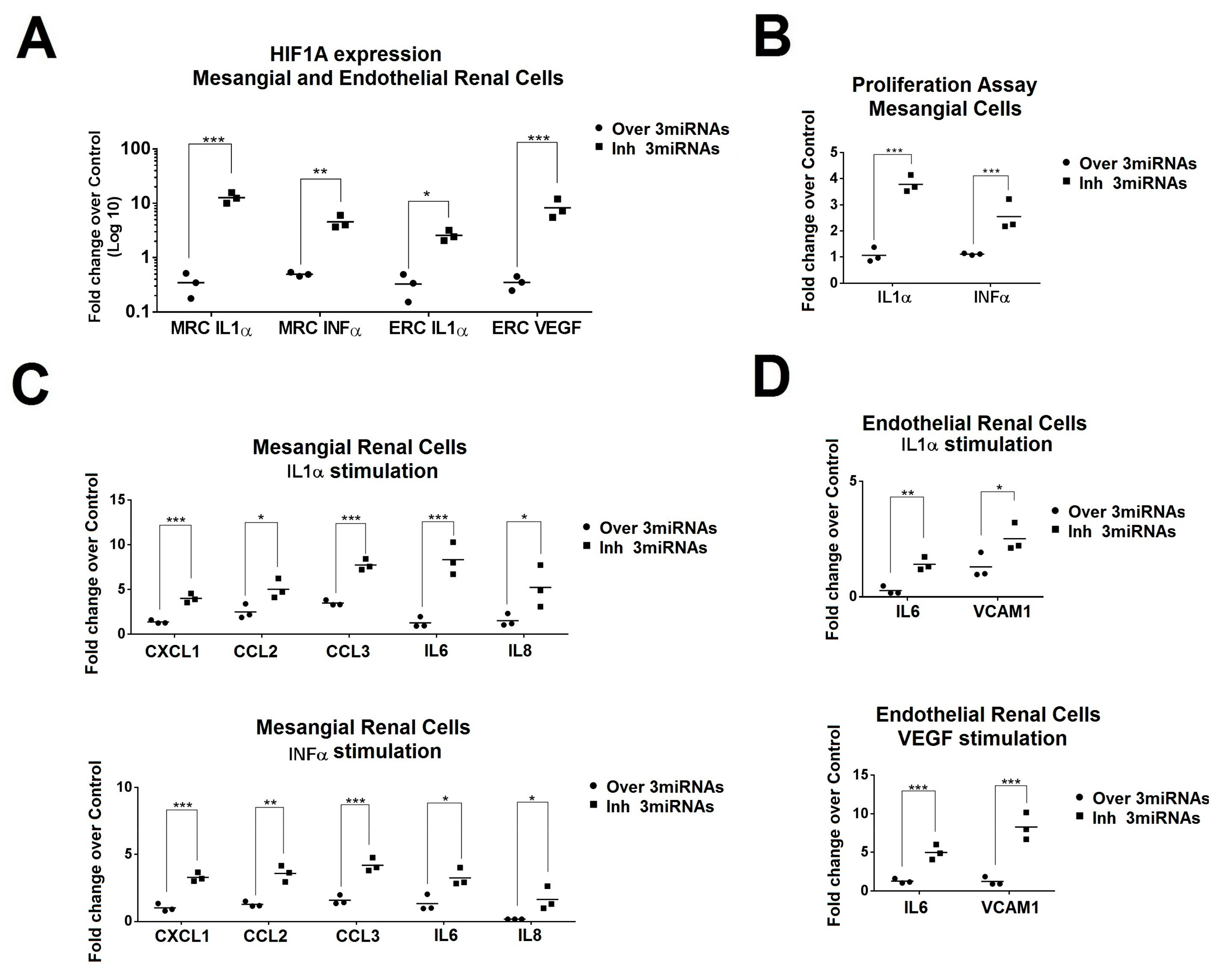
2.5. Simultaneous Modulation of Candidate miRNAs Induces a Superior HIF1A Inhibition in Primary Renal Cells and in the Production of Inflammatory Molecules.
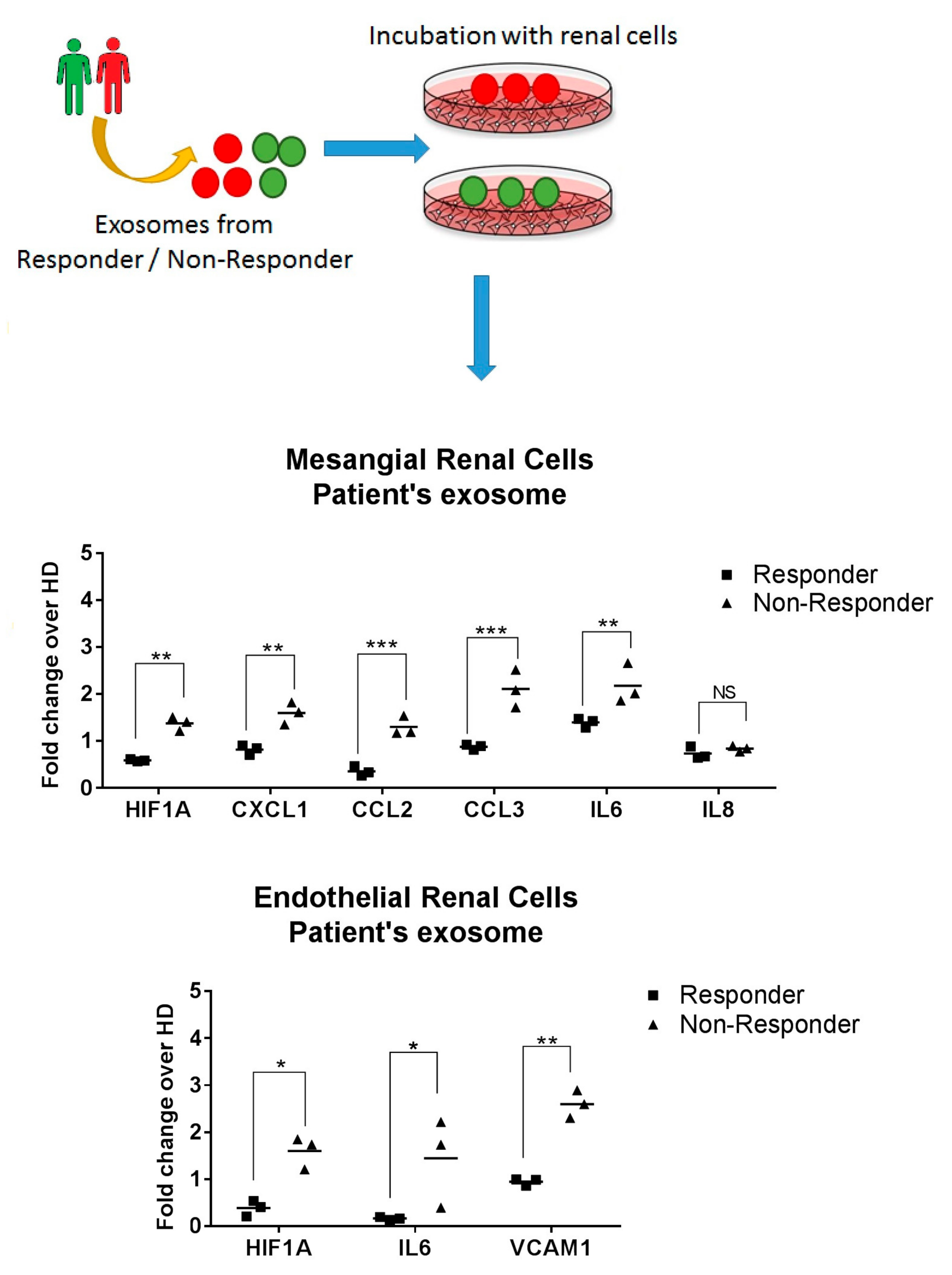
2.6. Exosomes from LN Responder Patients Reduces the Production of Inflammatory Cytokines
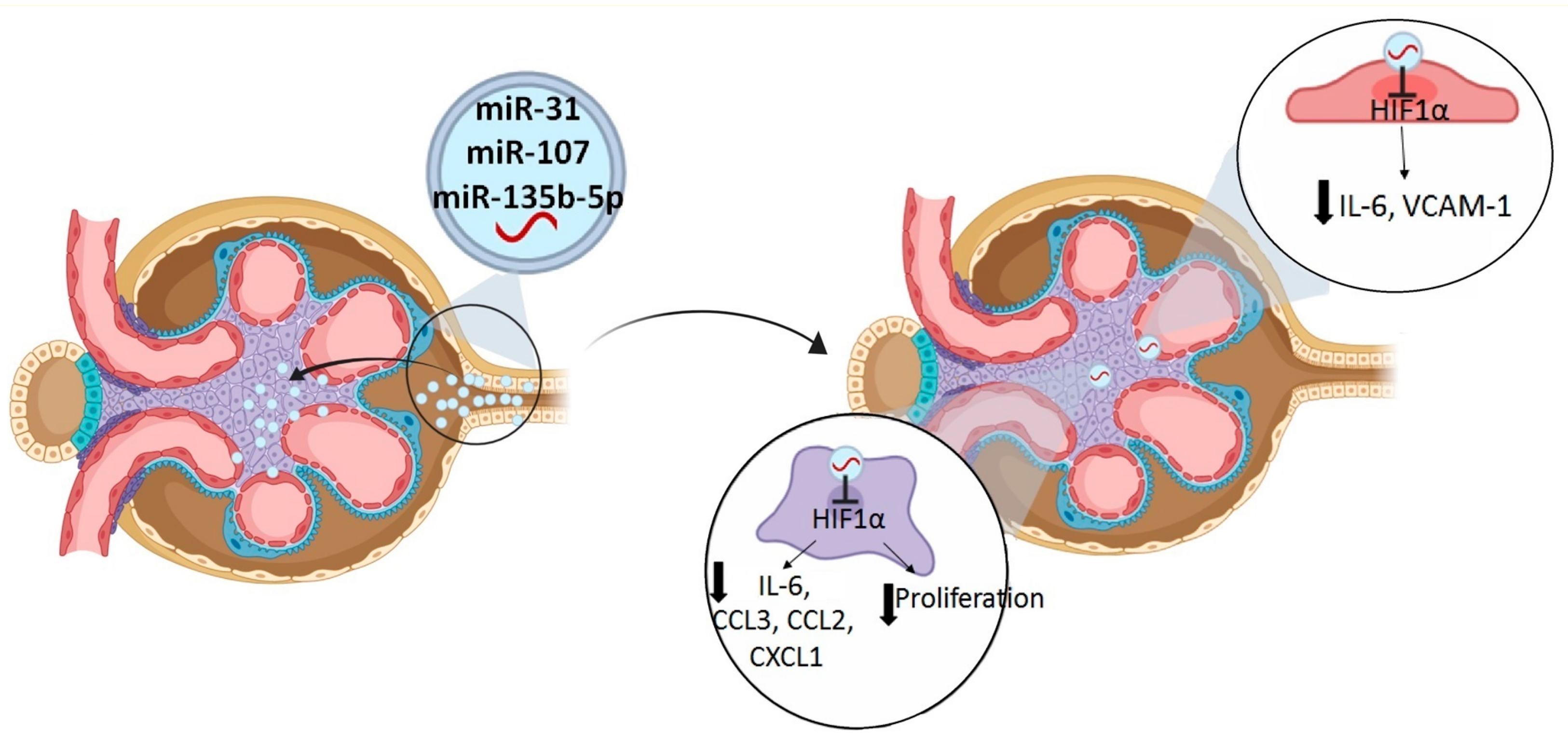
3. Discussion
4. Materials and Methods
4.1. Patients and Samples
4.2. Exosomal miRNA Extraction
4.3. MicroRNA qPCR-RT Arrays and Individual Assays
4.4. Double Fluorescent in situ hybridization (ISH) Detection and Immunofluorescence in Renal Biopsies
4.5. Production of the Study-Derived Exosomes by Primary Renal Cells
4.6. Fluorescently Labelled Urinary Exosome Internalization by Primary Renal Cells
4.7. Target Identification for Studied miRNAs
4.8. Luciferase Reporter Assay
4.9. MiR-135/miR-107/miR-31 Transfection in Primary Renal Cells
4.10. Gene Expression Analysis by Quantitative Reverse Transcription Polymerase Chain Reaction (qPCR-RT)
4.11. Patient’s Exosome Incubation into Primary Renal Cells
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tsokos, G.C.; Lo, M.S.; Reis, P.C.; Sullivan, K.E. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2016, 12, 716–730. [Google Scholar] [CrossRef] [PubMed]
- Rosner, S.; Ginzler, E.; Diamond, H. A multicentre study of outcome in systemic lupus erythematosus. II. Causes of death. Arthritis Rheum. 1982, 25, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Adler, M.; Chambers, S.; Edwards, C.; Neild, G.; Isenberg, D. An assessment of renal failure in an SLE cohort with special reference to ethnicity, over a 25-year period. Rheumatol. 2006, 45, 1144–1147. [Google Scholar] [CrossRef] [PubMed]
- Korbet, S.M.; Lewis, E.J.; Schwartz, M.M.; Reichlin, M.; Evans, J.; Rohde, R.D. Factors predictive of outcome in severe lupus nephritis. Am. J. Kidney Dis. 2000, 35, 904–914. [Google Scholar] [CrossRef]
- Reyes-Thomas, J.; Blanco, I.; Putterman, C. Urinary Biomarkers in Lupus Nephritis. Clin. Rev. Allergy Immunol. 2011, 40, 138–150. [Google Scholar] [CrossRef]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Boil. 2013, 200, 373–383. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Boldin, M.; Taganov, K.D.; Rao, D.; Yang, L.; Zhao, J.L.; Kalwani, M.; Garcia-Flores, Y.; Luong, M.; Devrekanli, A.; Xu, J.; et al. miR-146a is a significant brake on autoimmunity, myeloproliferation, and cancer in mice. J. Exp. Med. 2011, 208, 1189–1201. [Google Scholar] [CrossRef]
- Ramezani, A.; Devaney, J.M.; Cohen, S.; Wing, M.R.; Scott, R.; Knoblach, S.; Singhal, R.; Howard, L.; Kline, J.A.; Raj, D.S.; et al. Circulating and urinary microRNA profile in focal segmental glomerulosclerosis: A pilot study. Eur. J. Clin. Investig. 2015, 45, 394–404. [Google Scholar] [CrossRef]
- Delić, D.; Eisele, C.; Schmid, R.; Baum, P.; Wiech, F.; Gerl, M.; Zimdahl, H.; Pullen, S.S.; Urquhart, R. Urinary Exosomal miRNA Signature in Type II Diabetic Nephropathy Patients. PLoS ONE 2016, 11, e0150154. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, Y.; Zhou, J.; Zhang, Y. Urinary Exosomal miR-193a Can Be a Potential Biomarker for the Diagnosis of Primary Focal Segmental Glomerulosclerosis in Children. BioMed. Res. Int. 2017, 2017, 7298160. [Google Scholar] [CrossRef] [PubMed]
- Sole, C.; Cortes-Hernández, J.; Felip, M.L.; Vidal, M.; Ordi-Ros, J. miR-29c in urinary exosomes as predictor of early renal fibrosis in lupus nephritis. Nephrol. Dial. Transplant. 2015, 30, 1488–1496. [Google Scholar] [CrossRef] [PubMed]
- Solé, C.; Moliné, T.; Vidal, M.; Ordi-Ros, J.; Cortés-Hernández, J. An Exosomal Urinary miRNA Signature for Early Diagnosis of Renal Fibrosis in Lupus Nephritis. Cells 2019, 8, 773. [Google Scholar] [CrossRef] [PubMed]
- Ichii, O.; Otsuka-Kanazawa, S.; Horino, T.; Kimura, J.; Nakamura, T.; Matsumoto, M.; Toi, M.; Kon, Y. Decreased miR-26a Expression Correlates with the Progression of Podocyte Injury in Autoimmune Glomerulonephritis. PLoS ONE 2014, 9, e110383. [Google Scholar] [CrossRef]
- Quiroz, E.N.; Pacheco-Lugo, L.; Navarro-Quiroz, R.; Lorenzi, H.; España-Puccini, P.; Díaz-Olmos, Y.; Almendrales, L.; Olave, V.; Torres, H.G.; Diaz-Perez, A.; et al. Profiling analysis of circulating microRNA in peripheral blood of patients with class IV lupus nephritis. PLoS ONE 2017, 12, e0187973. [Google Scholar]
- Li, Y.; Xu, X.; Tang, X.; Bian, X.; Shen, B.; Zhao, H.; Luo, S.; Chen, Z.; Zhang, K. MicroRNA expression profile of urinary exosomes in Type IV lupus nephritis complicated by cellular crescent. J. Boil. Res. 2018, 25, 16. [Google Scholar] [CrossRef]
- Perez-Hernandez, J.; Forner, M.J.; Pinto, C.; Chaves, F.J.; Cortes, R.; Redon, J. Increased Urinary Exosomal MicroRNAs in Patients with Systemic Lupus Erythematosus. PLoS ONE 2015, 10, e0138618. [Google Scholar] [CrossRef]
- Korbet, S.M.; Schwartz, M.M.; Evans, J.; Lewis, E.J. Collaborative Study Group. Severe lupus nephritis: Racial differences in presentation and outcome. J. Am. Soc. Nephrol. 2007, 18, 244–254. [Google Scholar] [CrossRef]
- Gildea, J.J.; Seaton, J.E.; Victor, K.; Reyes, C.M.; Wang, R.B.; Pettigrew, A.C.; Courtner, C.E.; Shah, N.; Tran, H.T.; Van Sciver, R.; et al. Exosomal transfer from human renal proximal tubule cells to distal tubule and collecting duct cells. Clin. Biochem. 2014, 47, 89–94. [Google Scholar] [CrossRef]
- Pisitkun, T.; Shen, R.-F.; Knepper, M.A. Identification and proteomic profiling of exosomes in human urine. Proc. Natl. Acad. Sci. USA 2004, 101, 13368–13373. [Google Scholar] [CrossRef]
- Costa, V.; Carina, V.; Conigliaro, A.; Raimondi, L.; De Luca, A.; Bellavia, D.; Salamanna, F.; Setti, S.; Alessandro, R.; Fini, M.M.; et al. miR-31-5p Is a LIPUS-Mechanosensitive MicroRNA that Targets HIF-1α Signaling and Cytoskeletal Proteins. Int. J. Mol. Sci. 2019, 20, 1569. [Google Scholar] [CrossRef]
- Yamakuchi, M.; Lotterman, C.D.; Bao, C.; Hruban, R.H.; Karim, B.; Mendell, J.T.; Huso, D.; Lowenstein, C.J. P53-induced microRNA-107 inhibits HIF-1 and tumor angiogenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 6334–6339. [Google Scholar] [CrossRef]
- Yang, S.; Yin, J.; Hou, X. Inhibition of miR-135b by SP-1 promotes hypoxia-induced vascular endothelial cell injury via HIF-1α. Exp. Cell Res. 2018, 370, 31–38. [Google Scholar] [CrossRef]
- Deng, W.; Ren, Y.; Feng, X.; Yao, G.; Chen, W.; Sun, Y.; Wang, H.; Gao, X.; Sun, L. Hypoxia Inducible Factor-1 Alpha Promotes Mesangial Cell Proliferation in Lupus Nephritis. Am. J. Nephrol. 2014, 40, 507–515. [Google Scholar] [CrossRef]
- Ma, C.; Wei, J.; Zhan, F.; Wang, R.; Fu, K.; Wan, X.; Li, Z. Urinary Hypoxia-Inducible Factor-1alpha Levels Are Associated with Histologic Chronicity Changes and Renal Function in Patients with Lupus Nephritis. Yonsei Med. J. 2012, 53, 587–592. [Google Scholar] [CrossRef]
- Thomas, J.L.; Pham, H.; Li, Y.; Hall, E.; Perkins, G.A.; Ali, S.S.; Patel, H.H.; Singh, P. Hypoxia-inducible factor-1α activation improves renal oxygenation and mitochondrial function in early chronic kidney disease. Am. J. Physiol. Physiol. 2017, 313, F282–F290. [Google Scholar] [CrossRef]
- Kojima, H.; Gu, H.; Nomura, S.; Caldwell, C.C.; Kobata, T.; Carmeliet, P.; Semenza, G.L.; Sitkovsky, M.V. Abnormal B lymphocyte development and autoimmunity in hypoxia-inducible factor 1alpha-deficient chimeric mice. Proc. Natl. Acad. Sci. USA 2002, 99, 2170–2174. [Google Scholar] [CrossRef]
- Shi, L.Z.; Wang, R.; Huang, G.; Vogel, P.; Neale, G.; Green, D.R.; Chi, H. HIF1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J. Exp. Med. 2011, 208, 1367–1376. [Google Scholar] [CrossRef]
- Dang, E.V.; Barbi, J.; Yang, H.Y.; Jinasena, D.; Yu, H.; Zheng, Y.; Bordman, Z.; Fu, J.; Kim, Y.; Yen, H.R.; et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell 2011, 146, 772–784. [Google Scholar] [CrossRef]
- Zinkernagel, A.S.; Johnson, R.S.; Nizet, V. Hypoxia inducible factor (HIF) function in innate immunity and infection. J. Mol. Med. 2007, 85, 1339–1346. [Google Scholar] [CrossRef]
- Hochberg, M.C. Updating the American college of rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef]
- Gladman, D.D.; Ibañez, M.; Urowitz, M. Systemic lupus erythematosus disease activity index 2000. J. Rheumatol. 2002, 29, 288–291. [Google Scholar]
- Gladman, D.; Ginzler, E.; Goldsmith, C.; Fortin, P.; Liang, M.; Sánchez-Guerrero, J.; Urowitz, M.; Bacon, P.; Bombardieri, S.; Hanly, J.; et al. The development and initial validation of the systemic lupus international collaborating clinics/American college of rheumatology damage index for systemic lupus erythematosus. Arthritis Rheum. 1996, 39, 363–369. [Google Scholar] [CrossRef]
- Yu, F.; Haas, M.; Glassock, R.; Zhao, M.-H. Redefining lupus nephritis: Clinical implications of pathophysiologic subtypes. Nat. Rev. Nephrol. 2017, 13, 483–495. [Google Scholar] [CrossRef]
- Austin, H.A.; Muenz, L.R.; Joyce, K.M.; Antonovych, T.A.; Kullick, M.E.; Klippel, J.H.; Decker, J.L.; Balow, J.E. Prognostic factors in lupus nephritis. Am. J. Med. 1983, 75, 382–391. [Google Scholar] [CrossRef]
- Renal Disease Subcommittee of the American College of Rheumatology Ad Hoc Committee on Systemic Lupus Erythematosus Response Criteria; Liang, M.H.; Schur, P.H.; Fortin, P.; St.Clair, E.W.; Balow, J.E.; Costenbader, K.; Crofford, L.J.; De Pablo, P.; Dooley, M.A.; et al. The American college of rheumatology response criteria for proliferative and membranous renal disease in systemic lupus erythematosus clinical trials. Arthritis Rheum. 2006, 54, 421–432. [Google Scholar]
- Silahtaroglu, A.; Nolting, R.; Dyrskjøt, L.; Berezikov, E.; Møller, M.; Tommerup, N.; Kauppinen, S. Detection of microRNAs in frozen tissue sections by fluorescence in situ hybridization using locked nucleic acid probes and tyramide signal amplification. Nat. Protoc. 2007, 2, 2520–2528. [Google Scholar] [CrossRef]
- Lourenco, E.; Cava, A. Cytokines in Systemic Lupus Erythematosus. Curr. Mol. Med. 2009, 9, 242–254. [Google Scholar] [CrossRef]
- Iwata, Y.; Furuichi, K.; Kaneko, S.; Wada, T. The Role of Cytokine in the Lupus Nephritis. J. Biomed. Biotechnol. 2011, 2011, 594809. [Google Scholar] [CrossRef]
- Bustin, S.; Benes, V.; Garson, J.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]

| Responders (n = 7) | Non-Responders (n = 7) | p-Value | |
|---|---|---|---|
| Demographic | |||
| Age (years), median (IQR) | 36.9 (22.5–41.2) | 27.9 (22.9–37.5) | 0.330 |
| Duration of SLE (years), median (IQR) | 3.1 (0.1–13.3) | 7.2 (5.9–9.2) | 0.981 |
| Sex, male/female | 2/5 | 6/1 | 0.031 |
| Race/ethnicity, n (%) | |||
| Caucasian | 6 (85.7%) | 5 (72.1%) | 0.700 |
| Hispanic | 1 (14.3%) | 2 (28.6%) | 0.700 |
| Laboratory parameters, median (IQR) | |||
| Serum creatinine, mg/dL | 1.1 (1.0–1.3) | 1.1 (0.7–1.5) | 0.551 |
| eGFR (mL/min/1.73 m2) | 66 (48–98) | 65 (30–101) | 0.901 |
| Anti-dsDNA Abs, IU/mL | 79 (14–292) | 73 (46–580) | 0.372 |
| Serum C3, mg/dL | 61 (24–85) | 69 (53–90) | 0.309 |
| Serum C4, mg/dL Hemoglobin, g/dL Leucocytes, ×10−9/L Lymphocytes, ×10−9/L ESR, mm/h Protein, g/dL Albumin, g/dL | 8 (7–11) 11.6 (10.4–12.8) 5.7 (5.4–11.0) 1.3 (0.8–2.0) 33 (17–78) 6.5 (5.5–7.3) 3.3 (2.9–3.9) | 11 (6–16) 11.6 (9.1–14.5) 7.4 (4.9–9.3) 0.9 (0.7–1.6) 51 (11–89) 5.1 (4.7–6.3) 2.8 (2.3–3.3) | 0.635 0.983 0.893 0.208 0.746 0.114 0.100 |
| Proteinuria, g/24 h Leucocituria Hematuria Cast | 4.9 (1.4–6.8) 62 (29–241) 85 (17–300) 1 (1–7) | 3.5 (1.6–6.5) 85 (30–174) 148 (42–470) 2 (0–15) | 0.756 0.672 0.496 0.609 |
| Disease Activity, median (IQR) | |||
| SLEDAI-2K global score | 20 (14–23) | 18 (7–26) | 0.409 |
| Complete remission, n (%) | 7 (100) | 0 (0) | 0.409 |
| Time to remission (days), mean (SEM) | 149 (25) | n.a. | n.a. |
| Renal Biopsy, n (%) | |||
| Class III | 2 (28.6%) | 3 (42.9%) | 0.999 |
| Class IV | 5 (71.4%) | 4 (57.1%) | 0.999 |
| Activity Index, median (IQR) | 10 (8–12) | 7 (5–12) | 0.607 |
| Glomerular endocapillary proliferation | 2 (1–2) | 1 (1–2) | 0.999 |
| Glomerular neutrophilic infiltration | 1 (1–2) | 1 (0–2) | 0.662 |
| Wire-loop deposits and hyaline thrombi | 1 (0–2) | 1 (1–1) | 0.999 |
| Glomerular fibrinoid necrosis and karyorrhexis | 2 (1–4) | 2 (1–3) | 0.858 |
| Cellular crescents | 2 (1–2) | 2 (2–4) | 0.312 |
| Interstitial inflammation | 1 (0–1) | 1 (1–1) | 0.442 |
| Chronicity Index, median (IQR) | 2 (0–4) | 3 (1–4) | 0.524 |
| Glomerular sclerosis | 0 (0–2) | 1 (0–1) | 0.877 |
| Fibrous crescents | 0 (0–1) | 0 (0–1) | 0.867 |
| Tubular atrophy | 0 (0–1) | 0 (0–2) | 0.505 |
| Interstitial fibrosis | 0 (0–1) | 1 (0–1) | 0.218 |
| Characteristics | Responders (n = 22) | Non-Responders (n = 21) | p Value |
|---|---|---|---|
| Demographic | |||
| Age (years), mean | 34.3 (2.3) | 34.3 (2.3) | 0.981 |
| Duration of SLE (years), mean | 7.1 (1.6) | 5.8 (1.6) | 0.571 |
| Sex, male/female | 2/20 | 8/13 | 0.034 |
| Race/ethnicity, n (%) | |||
| Caucasian | 21 (95.5%) | 18 (85.7%) | 0.630 |
| Hispanic | 1 (4.5%) | 3 (14.3%) | 0.630 |
| Laboratory parameters, mean | |||
| Serum creatinine, mg/dL | 1.10 (0.10) | 1.3 (0.2) | 0.387 |
| eGFR (mL/min/1.73 m2) | 73 (10) | 68 (12) | 0.710 |
| Anti-dsDNA Abs, IU/mL | 234 (54) | 255 (68) | 0.812 |
| Serum C3, mg/dL | 60 (6) | 68 (4) | 0.265 |
| Serum C4, mg/dL Hemoglobin, g/dL Leucocytes, ×10−9/L Lymphocytes, ×10−9/L ESR, mm/h Protein, g/dL Albumin, g/dL | 9 (1) 10.8 (0.5) 6.8 (0.6) 1.7 (0.4) 63 (7) 6.3 (0.4) 3.2 (0.2) | 11 (1.4) 11.2 (0.5) 7.1 (0.8) 1.5 (0.3) 47 (8) 6.5 (0.3) 3.1 (0.2) | 0.274 0.515 0.787 0.072 0.140 0.072 0.108 |
| Proteinuria, g/24 h Leucocituria Hematuria Cast | 3590 (736) 173 (49) 553 (286) 3 (2) | 4589 (671) 149 (55) 308 (164) 4 (1) | 0.322 0.750 0.472 0.416 |
| Disease Activity, mean | |||
| SLEDAI-2K global score | 17.5 (2.5) | 16.5 (1.5) | 0.122 |
| Complete remission, n (%) | 22 (100) | 0 (0) | 0.122 |
| Time to remission (days), mean (SEM) | 214 (48) | n.a. | n.a. |
| Renal Biopsy, n (%) | |||
| Class III | 7 (31.8%) | 8 (38.8%) | 0.999 |
| Class IV | 15 (68.2%) | 13 (61.2%) | 0.999 |
| Activity Index, mean | 8.6 (1.1) | 7.6 (1.2) | 0.625 |
| Glomerular endocapillary proliferation | 1.6 (0.2) | 1.6 (0.3) | 0.913 |
| Glomerular neutrophilic infiltration | 1.0 (0.2) | 0.9 (0.3) | 0.849 |
| Wire-loop deposits and hyaline thrombi | 0.8 (0.4) | 1.0 (0.2) | 0.715 |
| Glomerular fibrinoid necrosis and karyorrhexis | 1.8 (0.6) | 1.6 (0.5) | 0.792 |
| Cellular crescents | 1.8 (0.5) | 1.8 (0.5) | 0.991 |
| Interstitial inflammation | 0.7 (0.3) | 0.9 (0.2) | 0.575 |
| Chronicity Index, mean | 1.8 (0.6) | 2.4 (0.6) | 0.487 |
| Glomerular sclerosis | 0.6 (0.3) | 0.6 (0.2) | 0.801 |
| Fibrous crescents | 0.2 (0.1) | 0.2 (0.1) | 0.700 |
| Tubular atrophy | 0.7 (0.2) | 0.9 (0.2) | 0.598 |
| Interstitial fibrosis | 0.6 (0.2) | 0.8 (0.2) | 0.594 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Vives, E.; Solé, C.; Moliné, T.; Vidal, M.; Agraz, I.; Ordi-Ros, J.; Cortés-Hernández, J. The Urinary Exosomal miRNA Expression Profile is Predictive of Clinical Response in Lupus Nephritis. Int. J. Mol. Sci. 2020, 21, 1372. https://doi.org/10.3390/ijms21041372
Garcia-Vives E, Solé C, Moliné T, Vidal M, Agraz I, Ordi-Ros J, Cortés-Hernández J. The Urinary Exosomal miRNA Expression Profile is Predictive of Clinical Response in Lupus Nephritis. International Journal of Molecular Sciences. 2020; 21(4):1372. https://doi.org/10.3390/ijms21041372
Chicago/Turabian StyleGarcia-Vives, Eloi, Cristina Solé, Teresa Moliné, Marta Vidal, Irene Agraz, Josep Ordi-Ros, and Josefina Cortés-Hernández. 2020. "The Urinary Exosomal miRNA Expression Profile is Predictive of Clinical Response in Lupus Nephritis" International Journal of Molecular Sciences 21, no. 4: 1372. https://doi.org/10.3390/ijms21041372
APA StyleGarcia-Vives, E., Solé, C., Moliné, T., Vidal, M., Agraz, I., Ordi-Ros, J., & Cortés-Hernández, J. (2020). The Urinary Exosomal miRNA Expression Profile is Predictive of Clinical Response in Lupus Nephritis. International Journal of Molecular Sciences, 21(4), 1372. https://doi.org/10.3390/ijms21041372






