Aquaporin 1, 3, and 5 Patterns in Salivary Gland Mucoepidermoid Carcinoma: Expression in Surgical Specimens and an In Vitro Pilot Study
Abstract
1. Introduction
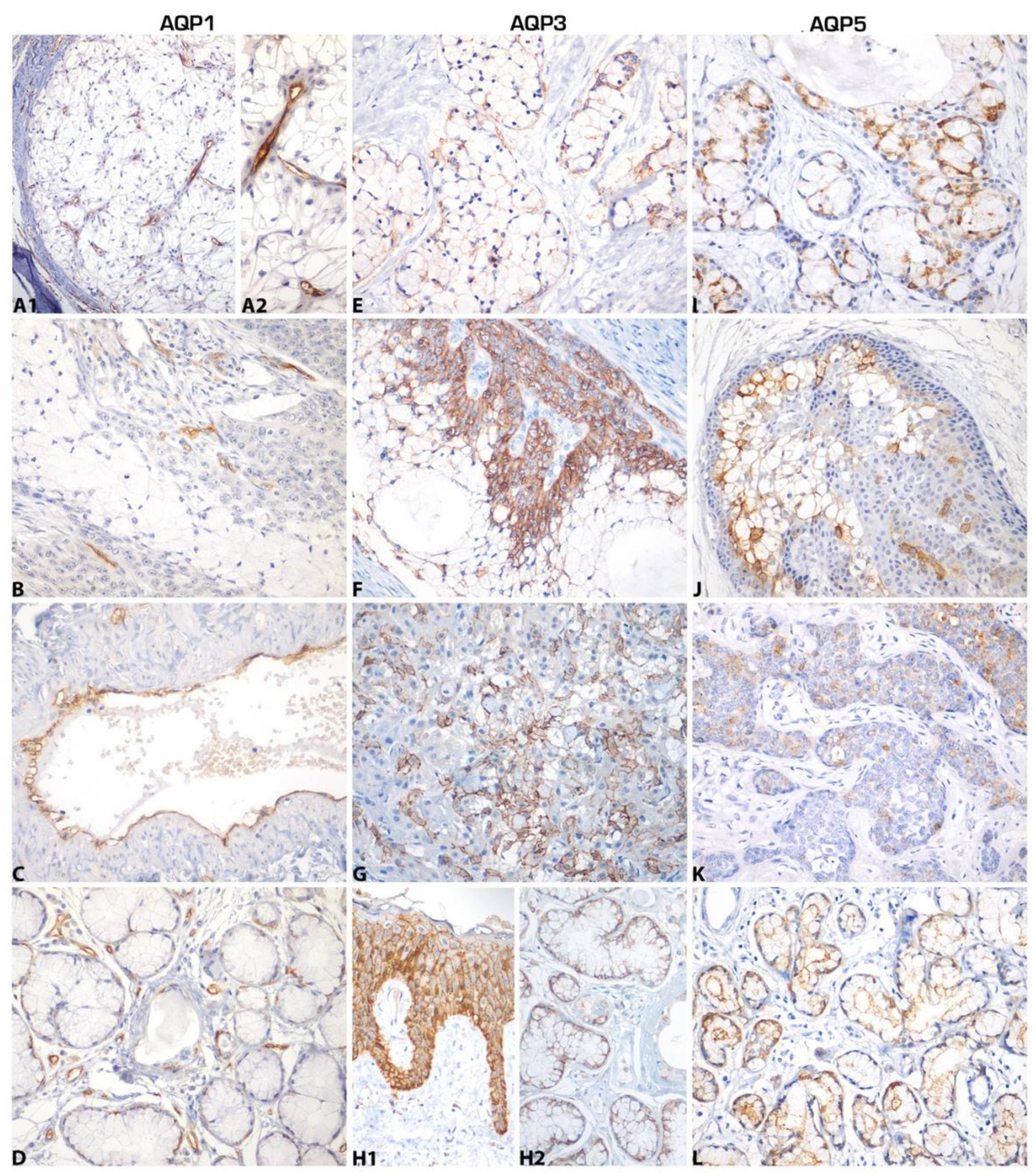
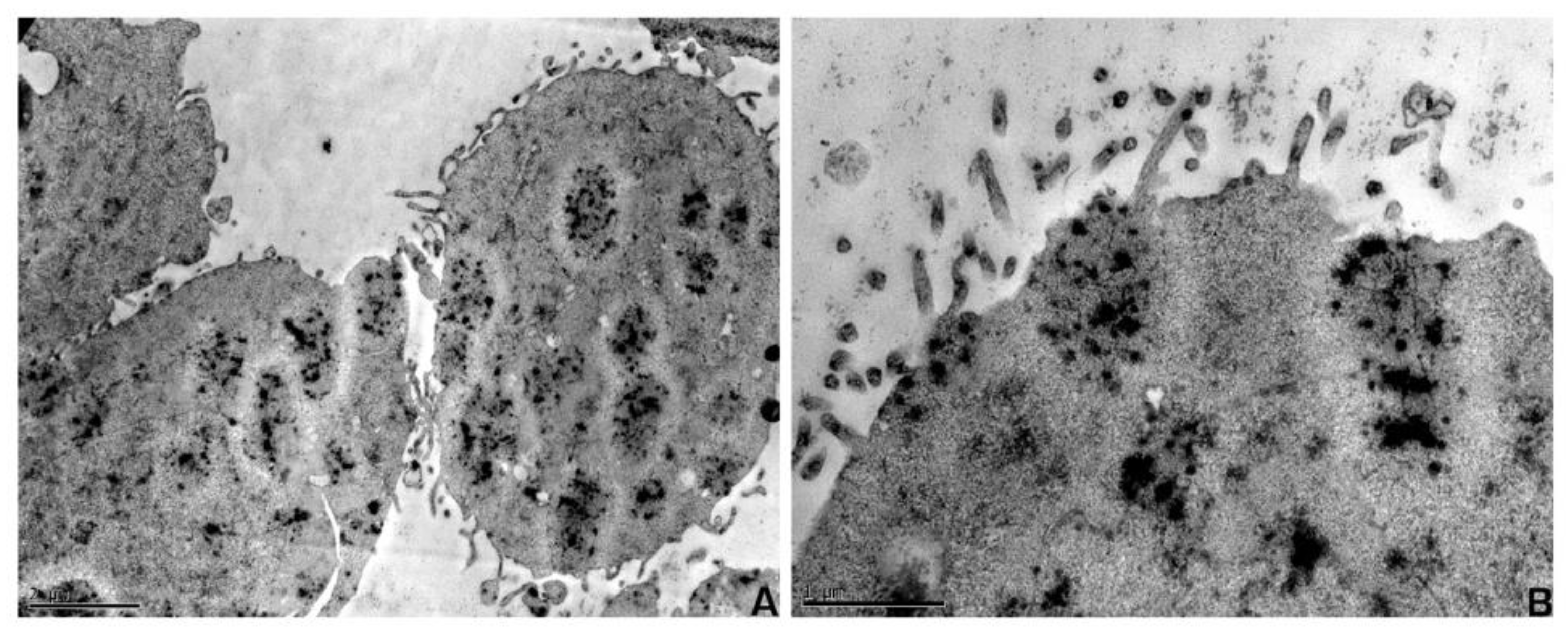
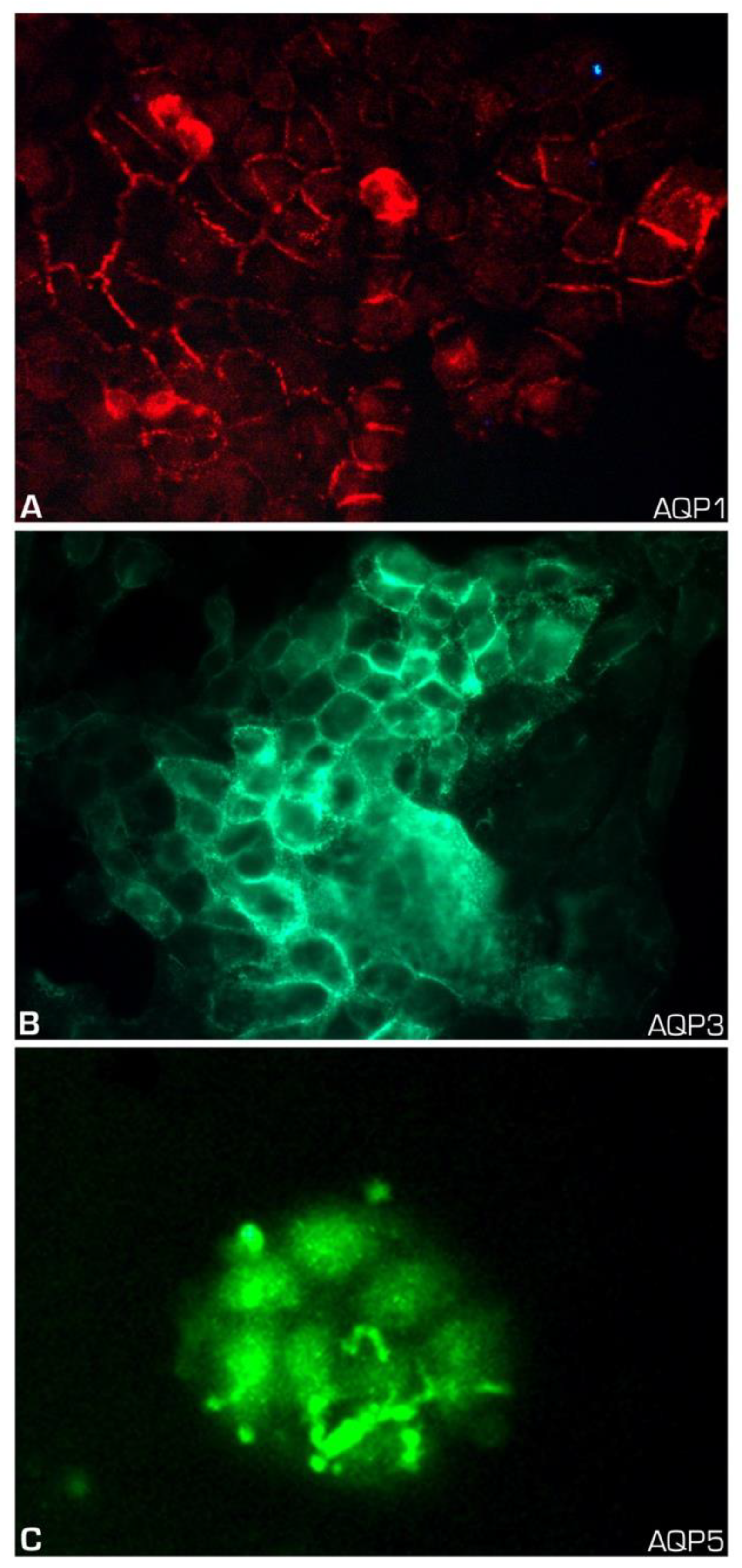
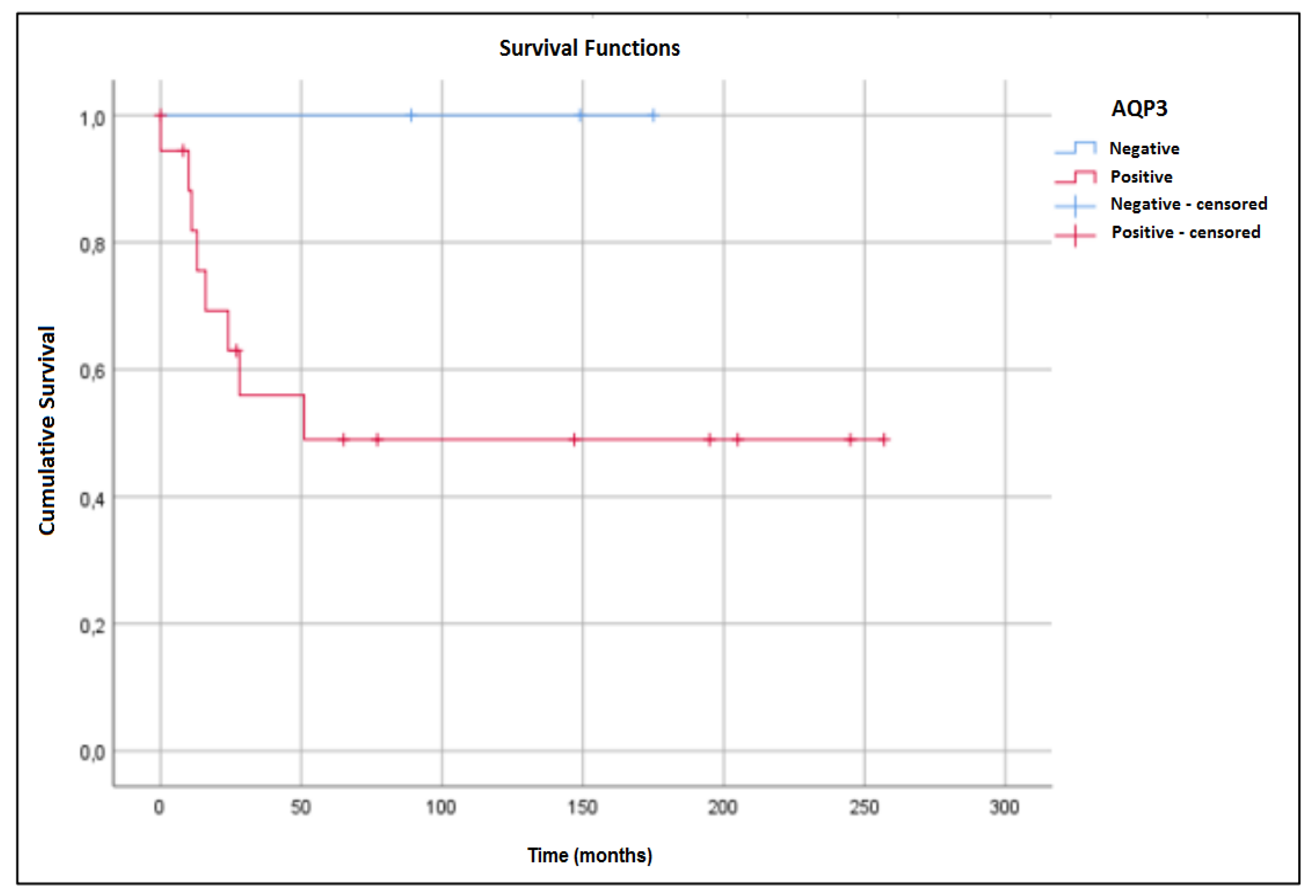
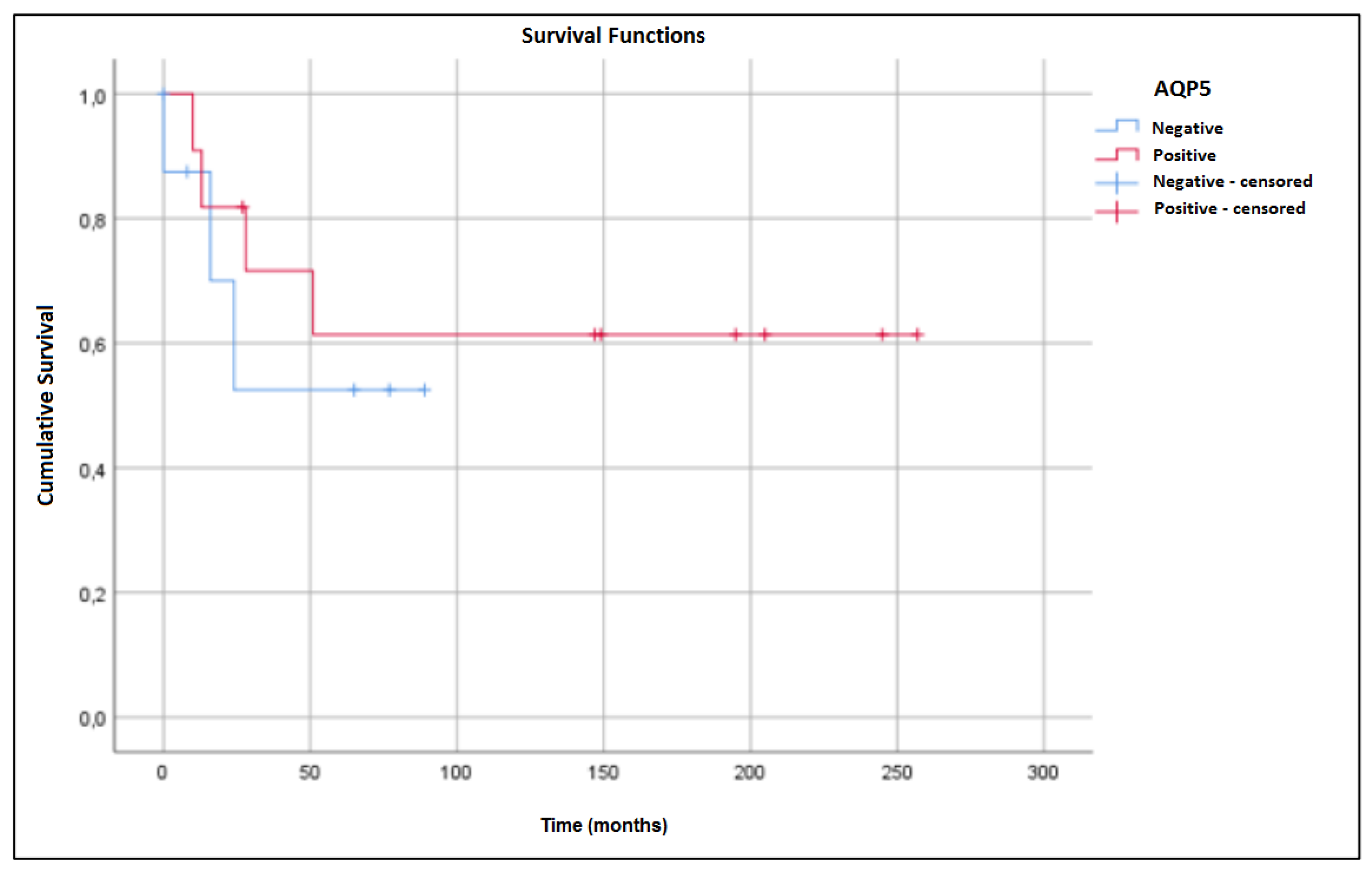
2. Results
3. Discussion
4. Materials and Methods
4.1. Tissue Samples
4.2. Immunohistochemistry
4.3. Cell Culture
4.4. Immunofluorescence
4.5. Electron Microscopy
4.6. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AQPs | Aquaporins |
| MEC | Mucoepidermoid carcinoma |
| ACC | Adenoid cystic carcinoma |
References
- Delporte, C.; Steinfeld, S. Distribution and roles of aquaporins in salivary glands. Biochim. Biophys. Acta BBA Biomembr. 2006, 1758, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, M.C.; Saadoun, S. Key roles of aquaporins in tumor biology. Biochim. Biophys. Acta BBA Biomembr. 2015, 1848, 2576–2583. [Google Scholar] [CrossRef] [PubMed]
- Delporte, C.; Bryla, A.; Perret, J. Aquaporins in Salivary Glands: From Basic Research to Clinical Applications. Int. J. Mol. Sci. 2016, 17, 166. [Google Scholar] [CrossRef] [PubMed]
- Aikman, B.; De Almeida, A.; Meier-Menches, S.M.; Casini, A.; Meier, S.M. Aquaporins in cancer development: Opportunities for bioinorganic chemistry to contribute novel chemical probes and therapeutic agents. Metallomics 2018, 10, 696–712. [Google Scholar] [CrossRef]
- Krane, C.M.; Melvin, J.E.; Nguyen, H.-V.; Richardson, L.; Towne, J.E.; Doetschman, T.; Menon, A.G. Salivary Acinar Cells from Aquaporin 5-deficient Mice Have Decreased Membrane Water Permeability and Altered Cell Volume Regulation. J. Boil. Chem. 2001, 276, 23413–23420. [Google Scholar] [CrossRef]
- Kozono, D.; Yasui, M.; King, L.S.; Agre, P. Aquaporin water channels: Atomic structure molecular dynamics meet clinical medicine. J. Clin. Investig. 2002, 109, 1395–1399. [Google Scholar] [CrossRef]
- Papadopoulos, M.C.; Saadoun, S.; Verkman, A.S. Aquaporins and cell migration. Pflugers Arch. Eur. J. Physiol. 2008, 456, 693–700. [Google Scholar] [CrossRef]
- Ribatti, D.; Ranieri, G.; Annese, T.; Nico, B. Aquaporins in cancer. Biochim. Biophys. Acta BBA Gen. Subj. 2014, 1840, 1550–1553. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Wu, X.; Yu, G. Aquaporins: New Targets for Cancer Therapy. Technol. Cancer Res. Treat. 2016, 15, 821–828. [Google Scholar] [CrossRef]
- Reddy, M.G.S.; Dony, E. Role of aquaporins in oral cancer. J. Cancer Res. Ther. 2017, 13, 137. [Google Scholar] [CrossRef]
- Tomita, Y.; Dorward, H.; Yool, A.J.; Smith, E.; Townsend, A.R.; Price, T.J.; Hardingham, J.E. Role of Aquaporin 1 Signalling in Cancer Development and Progression. Int. J. Mol. Sci. 2017, 18, 299. [Google Scholar] [CrossRef] [PubMed]
- Dajani, S.; Saripalli, A.; Sharma-Walia, N. Water transport proteins–aquaporins (AQPs) in cancer biology. Oncotarget 2018, 9, 36392–36405. [Google Scholar] [CrossRef] [PubMed]
- Moosavi, M.-S.; Elham, Y. Aquaporins 1, 3 and 5 in Different Tumors, their Expression, Prognosis Value and Role as New Therapeutic Targets. Pathol. Oncol. Res. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bellezza, G.; Vannucci, J.; Bianconi, F.; Metro, G.; Del Sordo, R.; Andolfi, M.; Ferri, I.; Siccu, P.; Ludovini, V.; Puma, F.; et al. Prognostic implication of aquaporin 1 overexpression in resected lung adenocarcinoma. Interact. Cardiovasc. Thorac. Surg. 2017, 25, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, S.; Wada, K.; Usami, Y.; Tanaka, N.; Aikawa, T.; Okura, M.; Nakajima, A.; Kogo, M.; Kamisaki, Y. Differential expression of aquaporin 5 and aquaporin 3 in squamous cell carcinoma and adenoid cystic carcinoma. Int. J. Oncol. 2012, 41, 67–75. [Google Scholar] [CrossRef]
- Niu, D.; Kondo, T.; Nakazawa, T.; Yamane, T.; Mochizuki, K.; Kawasaki, T.; Matsuzaki, T.; Takata, K.; Katoh, R. Expression of aquaporin 3 in human neoplastic tissues. Histopathology 2012, 61, 543–551. [Google Scholar] [CrossRef]
- Kasa, P.; Farran, B.; Prasad, G.L.V.; Nagaraju, G.P. Aquaporins in female specific cancers. Gene 2019, 700, 60–64. [Google Scholar] [CrossRef]
- Zou, W.; Yang, Z.; Li, D.; Liu, Z.; Zou, Q.; Yuan, Y. AQP1 and AQP3 Expression are Associated with Severe Symptoms and Poor-prognosis of the Pancreatic Ductal Adenocarcinoma. Appl. Immunohistochem. Mol. Morphol. 2019, 27, 40–47. [Google Scholar] [CrossRef]
- Goode, R.K.; Auclair, P.L.; Ellis, G.L. Mucoepidermoid carcinoma of the major salivary glands: Clinical and histopathologic analysis of 234 cases with evaluation of grading criteria. Cancer 1998, 82, 1217–1224. [Google Scholar] [CrossRef]
- Brandwein, M.S.; Ferlito, A.; Bradley, P.J.; Hille, J.J.; Rinaldo, A. Diagnosis and Classification of Salivary Neoplasms: Pathologic Challenges and Relevance to Clinical Outcomes. Acta Oto Laryngol. 2002, 122, 758–764. [Google Scholar] [CrossRef]
- Speight, P.M.; Barrett, A.W. Prognostic factors in malignant tumours of the salivary glands. Br. J. Oral Maxillofac. Surg. 2009, 47, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Rapidis, A.D.; Givalos, N.; Gakiopoulou, H.; Stavrianos, S.D.; Faratzis, G.; Lagogiannis, G.A.; Katsilieris, I.; Patsouris, E. Mucoepidermoid carcinoma of the salivary glands. Oral Oncol. 2007, 43, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.M.; Roman, S.A.; Sosa, J.A.; Judson, B.L. Histologic grade as prognostic indicator for mucoepidermoid carcinoma: A population-level analysis of 2400 patients. Head Neck 2014, 36, 158–163. [Google Scholar] [CrossRef] [PubMed]
- De Paula, F.; Teshima, T.H.N.; Hsieh, R.; Souza, M.M.; Coutinho-Camillo, C.M.; Nico, M.M.S.; Lourenco, S.V. The expression of water channel proteins during human salivary gland development: A topographic study of aquaporins 1, 3 and 5. J. Mol. Histol. 2017, 48, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Gresz, V.; Kwon, T.H.; Hurley, P.T.; Varga, G.; Zelles, T.; Nielsen, S.; Case, R.M.; Steward, M.C. Identification and localization of aquaporin water channels in human salivary glands. Am. J. Physiol. Liver Physiol. 2001, 281, G247–G254. [Google Scholar] [CrossRef] [PubMed]
- Beroukas, D.; Hiscock, J.; Gannon, B.J.; Jonsson, R.; Gordon, T.P.; A Waterman, S. Selective down-regulation of aquaporin-1 in salivary glands in primary Sjögren’s syndrome. Lab. Investig. 2002, 82, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Delporte, C. Aquaporins and Gland Secretion. In Advances in Experimental Medicine and Biology; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; Volume 969, pp. 63–79. [Google Scholar]
- Prevarskaya, N.; Skryma, R.; Shuba, Y. Ion channels and the hallmarks of cancer. Trends Mol. Med. 2010, 16, 107–121. [Google Scholar] [CrossRef]
- Becchetti, A. Ion channels and transporters in cancer. 1. Ion channels and cell proliferation in cancer. Am. J. Physiol. Physiol. 2011, 301, C255–C265. [Google Scholar] [CrossRef]
- Clapp, C.; De La Escalera, G.M. Aquaporin-1: A novel promoter of tumor angiogenesis. Trends Endocrinol. Metab. 2006, 17, 1–2. [Google Scholar] [CrossRef]
- Nico, B.; Ribatti, M. Aquaporins in tumor growth and angiogenesis. Cancer Lett. 2010, 294, 135–138. [Google Scholar] [CrossRef]
- Qin, F.; Zhang, H.; Shao, Y.; Liu, X.; Yang, L.; Huang, Y.; Fu, L.; Gu, F.; Ma, Y. Expression of aquaporin 1, a water channel protein, in cytoplasm is negatively correlated with prognosis of breast cancer patients. Oncotarget 2016, 7, 8143–8154. [Google Scholar] [CrossRef]
- Wei, X.; Dong, J. Aquaporin 1 promotes the proliferation and migration of lung cancer cell in vitro. Oncol. Rep. 2015, 34, 1440–1448. [Google Scholar] [CrossRef]
- Yun, S.; Sun, P.-L.; Jin, Y.; Kim, H.; Park, E.; Park, S.Y.; Lee, K.; Lee, K.; Chung, J.-H. Aquaporin 1 Is an Independent Marker of Poor Prognosis in Lung Adenocarcinoma. J. Pathol. Transl. Med. 2016, 50, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Hojo, S.; Sekine, S.; Sawada, S.; Okumura, T.; Nagata, T.; Shimada, Y.; Tsukada, K. Expression of aquaporin-1 is a poor prognostic factor for stage II and III colon cancer. Mol. Clin. Oncol. 2013, 1, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Yoon, G.S. Overexpression of Aquaporin-1 is a Prognostic Factor for Biochemical Recurrence in Prostate Adenocarcinoma. Pathol. Oncol. Res. 2017, 23, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Shao, C.; Bishop, J.A.; Feng, Z.; Trock, B.J.; Westra, W.H.; Ha, P.K. Aquaporin-1 promoter hypermethylation is associated with improved prognosis in salivary gland adenoid cystic carcinoma. Otolaryngol. Neck Surg. 2014, 150, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Sun, W.; Tan, M.; Glazer, C.A.; Bhan, S.; Zhong, X.; Fakhry, C.; Sharma, R.; Westra, W.H.; Hoque, M.O.; et al. Integrated, genome-wide screening for hypomethylated oncogenes in salivary gland adenoid cystic carcinoma. Clin. Cancer Res. 2011, 17, 4320–4330. [Google Scholar] [CrossRef]
- Wang, W.; Hart, P.; Piesco, N.; Lu, X.; Gorry, M.; Hart, T. Aquaporin Expression in Developing Human Teeth and Selected Orofacial Tissues. Calcif. Tissue Int. 2003, 72, 222–227. [Google Scholar] [CrossRef]
- Delporte, C. Aquaporins in salivary glands and pancreas. Biochim. Biophys. Acta BBA Gen. Subj. 2014, 1840, 1524–1532. [Google Scholar] [CrossRef]
- Ichiyama, T.; Nakatani, E.; Tatsumi, K.; Hideshima, K.; Urano, T.; Nariai, Y.; Sekine, J. Expression of aquaporin 3 and 5 as a potential marker for distinguishing dry mouth from Sjögren’s syndrome. J. Oral Sci. 2018, 60, 212–220. [Google Scholar] [CrossRef]
- Marlar, S.; Jensen, H.H.; Login, F.H.; Nejsum, L.N. Aquaporin-3 in Cancer. Int. J. Mol. Sci. 2017, 18, 2106. [Google Scholar] [CrossRef] [PubMed]
- Hara-Chikuma, M.; Verkman, A.S. Prevention of Skin Tumorigenesis and Impairment of Epidermal Cell Proliferation by Targeted Aquaporin-3 Gene Disruption. Mol. Cell. Biol. 2008, 28, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Huang, L.; Shao, M. Aquaporin 3 facilitates tumor growth in pancreatic cancer by modulating mTOR signaling. Biochem. Biophys. Res. Commun. 2017, 486, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- Kusayama, M.; Wada, K.; Nagata, M.; Ishimoto, S.; Takahashi, H.; Yoneda, M.; Nakajima, A.; Okura, M.; Kogo, M.; Kamisaki, Y. Critical role of aquaporin 3 on growth of human esophageal and oral squamous cell carcinoma. Cancer Sci. 2011, 102, 1128–1136. [Google Scholar] [CrossRef]
- Matsuo, K.; Kawano, K. Immunohistochemical distribution and morphometric analysis of aquaporin-3 in oral squamous cell carcinoma. Int. J. Oral Maxillofac. Surg. 2014, 43, 13–21. [Google Scholar] [CrossRef]
- Breyer, J.; Otto, W.; Burger, M.; Hartmann, A.; Rubenwolf, P.C. Aquaporin 3 Expression Loss in Urothelial Carcinoma: Association with Tumor Invasion Depth, but not with Grading? Bladder Cancer 2017, 3, 31–34. [Google Scholar] [CrossRef]
- Nakamura, M.; Saga, T.; Watanabe, K.; Takahashi, N.; Tabira, Y.; Kusukawa, J.; Yamaki, K.-I. An Immunohistochemistry-Based Study on Aquaporin (AQP)-1, 3, 4, 5 and 8 in the Parotid Glands, Submandibular Glands and Sublingual Glands Sjögren’s Syndrome Mouse Models Chronically Administered Cevimeline. Kurume Med. J. 2013, 60, 7–19. [Google Scholar] [CrossRef]
- Sapmaz, E.; Uysal, M.; Tumer, M.K.; Sapmaz, H.I.; Somuk, B.T.; Arici, A.; Taş, U. Investigation of age-related changes in the expression of aquaporin-1 and aquaporin-5 in the salivary glands of mice. Acta Oto-Laryngol. 2016, 136, 1–7. [Google Scholar] [CrossRef]
- Direito, I.; Madeira, A.; Brito, M.A.; Soveral, G. Aquaporin-5: From structure to function and dysfunction in cancer. Cell. Mol. Life Sci. 2016, 73, 1623–1640. [Google Scholar] [CrossRef]
- Jung, H.J.; Park, J.-Y.; Jeon, H.-S.; Kwon, T.-H. Aquaporin-5: A Marker Protein for Proliferation and Migration of Human Breast Cancer Cells. PLoS ONE 2011, 6, e28492. [Google Scholar] [CrossRef]
- Jensen, H.H.; Login, F.H.; Park, J.-Y.; Kwon, T.-H.; Nejsum, L.N. Immunohistochemical evalulation of activated Ras and Rac1 as potential downstream effectors of aquaporin-5 in breast cancer in vivo. Biochem. Biophys. Res. Commun. 2017, 493, 1210–1216. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, Z.; Song, Y.; Zhang, P.; Hu, J.; Bai, C. Expression of aquaporin 5 increases proliferation and metastasis potential of lung cancer. J. Pathol. 2010, 221, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhao, C.; Chen, D.; Zhou, Z. Overexpression of AQP5 in cervical cancer: Correlation with clinicopathological features and prognosis. Med. Oncol. 2012, 29, 1998–2004. [Google Scholar] [CrossRef]
- Yang, J.H.; Yu, Y.Q.; Yan, C.X. Localisation and expression of aquaporin subtypes in epithelial ovarian tumours. Histol. Histopathol. 2011, 26, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zhu, Y.; Zhang, X.; Chen, X.; Zheng, W.; Yang, J. Down-regulated aquaporin 5 inhibits proliferation and migration of human epithelial ovarian cancer 3AO cells. J. Ovarian Res. 2014, 7, 78. [Google Scholar] [CrossRef]
- He, Z.; Dong, W.; Hu, J.; Ren, X. AQP5 promotes hepatocellular carcinoma metastasis via NF-κB-regulated epithelial-mesenchymal transition. Biochem. Biophys. Res. Commun. 2017, 490, 343–348. [Google Scholar] [CrossRef]
- Kang, S.K.; Chae, Y.K.; Woo, J.; Kim, M.S.; Park, J.C.; Lee, J.; Soria, J.C.; Jang, S.J.; Sidransky, D.; Moon, C. Role of Human Aquaporin 5 In Colorectal Carcinogenesis. Am. J. Pathol. 2008, 173, 518–525. [Google Scholar] [CrossRef]
- Woo, J.; Lee, J.; Kim, M.S.; Jang, S.J.; Sidransky, D.; Moon, C. The effect of aquaporin 5 overexpression on the Ras signaling pathway. Biochem. Biophys. Res. Commun. 2008, 367, 291–298. [Google Scholar] [CrossRef]
- Wang, W.; Li, Q.; Yang, T.; Bai, G.; Li, D.; Li, Q.; Sun, H. Expression of AQP5 and AQP8 in human colorectal carcinoma and their clinical significance. World J. Surg. Oncol. 2012, 10, 242. [Google Scholar] [CrossRef]
- Sekine, S.; Shimada, Y.; Nagata, T.; Moriyama, M.; Omura, T.; Watanabe, T.; Hori, R.; Yoshioka, I.; Okumura, T.; Sawada, S.; et al. Prognostic significance of aquaporins in human biliary tract carcinoma. Oncol. Rep. 2012, 27, 1741–1747. [Google Scholar] [CrossRef][Green Version]
- Machida, Y.; Ueda, Y.; Shimasaki, M.; Sato, K.; Sagawa, M.; Katsuda, S.; Sakuma, T. Relationship of aquaporin 1, 3, and 5 expression in lung cancer cells to cellular differentiation, invasive growth, and metastasis potential. Hum. Pathol. 2011, 42, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, S.; Jiang, H.; Yang, Y.; Jiang, Y. Co-expression of AQP3 and AQP5 in esophageal squamous cell carcinoma correlates with aggressive tumor progression and poor prognosis. Med. Oncol. 2013, 30, 636. [Google Scholar] [CrossRef] [PubMed]
- Shimasaki, M.; Kanazawa, Y.; Sato, K.; Tsuchiya, H.; Ueda, Y. Aquaporin-1 and -5 are involved in the invasion and proliferation of soft tissue sarcomas. Pathol. Res. Pr. 2018, 214, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Hara-Chikuma, M.; Verkman, A.S. Aquaporin-3 facilitates epidermal cell migration and proliferation during wound healing. J. Mol. Med. 2008, 86, 221–231. [Google Scholar] [CrossRef]
- Warner, K.A.; Adams, A.; Bernardi, L.; Nör, C.; Finkel, K.A.; Zhang, Z.; McLean, S.A.; Helman, J.; Wolf, G.T.; Divi, V.; et al. Characterization of tumorigenic cell lines from the recurrence and lymph node metastasis of a human salivary mucoepidermoid carcinoma. Oral Oncol. 2013, 49, 1059–1066. [Google Scholar] [CrossRef]
| Protein | Positive | Negative | Number of Cases (%) * |
|---|---|---|---|
| AQP1 ** | 0 (0.0) | 21 (100.0) | 21 (100.0) |
| AQP3 | 18 (85.7) | 3 (14.3) | 21 (100.0) |
| AQP5 | 12 (60.0) | 8 (40.0) | 20 (100.0) |
| Characteristic | Category | Mucoepidermoid Carcinoma (%) |
|---|---|---|
| Gender | Male | 13 (54.0) |
| Female | 11 (46.0) | |
| Age | ≤40 years | 08 (33.3) |
| >40 years | 16 (66.7) | |
| Race | White | 22 (91.7) |
| Non-white | 02 (8.3) | |
| Histological grade | Low | 12 (50.0) |
| Intermediate | 04 (16.6) | |
| High | 07 (29.2) | |
| n/a * | 01 (4.2) | |
| Tumor site | Parotid | 09 (37.5) |
| Sublingual | 01 (4.2) | |
| Minor Salivary Glands | 14 (58.3) | |
| Vascular invasion | Yes | 03 (12.5) |
| No | 15 (62.5) | |
| n/a | 06 (25.0) | |
| Perineural infiltration | Yes | 06 (25.0) |
| No | 13 (54.2) | |
| n/a | 05 (20.8) | |
| Lymph node metastasis | Yes | 10 (41.7) |
| No | 06 (25.0) | |
| n/a | 08 (33.3) | |
| Local recurrence | Yes | 06 (25.0) |
| No | 13 (54.2) | |
| n/a | 05 (20.8) |
| Primary Serum | Clone | Source | Working Titer | Antigen Retrieval |
|---|---|---|---|---|
| AQP1 | 1/22 | Abcam | 1:1000 | Pressure cooker, citrate pH 6.0 |
| AQP3 | Polyclonal | Abcam | 1:1000 | Pressure cooker, citrate pH 6.0 |
| AQP5 | EPR3747 | Abcam | 1:1000 | Pressure cooker, citrate pH 6.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stamboni, M.B.; Gomes, Á.N.d.M.; Souza, M.M.d.; Oliveira, K.K.; Arruda, C.F.J.; de Paula, F.; Bettim, B.B.; Marques, M.M.; Kowalski, L.P.; Pinto, C.A.L.; et al. Aquaporin 1, 3, and 5 Patterns in Salivary Gland Mucoepidermoid Carcinoma: Expression in Surgical Specimens and an In Vitro Pilot Study. Int. J. Mol. Sci. 2020, 21, 1287. https://doi.org/10.3390/ijms21041287
Stamboni MB, Gomes ÁNdM, Souza MMd, Oliveira KK, Arruda CFJ, de Paula F, Bettim BB, Marques MM, Kowalski LP, Pinto CAL, et al. Aquaporin 1, 3, and 5 Patterns in Salivary Gland Mucoepidermoid Carcinoma: Expression in Surgical Specimens and an In Vitro Pilot Study. International Journal of Molecular Sciences. 2020; 21(4):1287. https://doi.org/10.3390/ijms21041287
Chicago/Turabian StyleStamboni, Mérin Barbara, Ágatha Nagli de Mello Gomes, Milena Monteiro de Souza, Katia Klug Oliveira, Claudia Fabiana Joca Arruda, Fernanda de Paula, Barbara Beltrame Bettim, Márcia Martins Marques, Luiz Paulo Kowalski, Clóvis Antônio Lopes Pinto, and et al. 2020. "Aquaporin 1, 3, and 5 Patterns in Salivary Gland Mucoepidermoid Carcinoma: Expression in Surgical Specimens and an In Vitro Pilot Study" International Journal of Molecular Sciences 21, no. 4: 1287. https://doi.org/10.3390/ijms21041287
APA StyleStamboni, M. B., Gomes, Á. N. d. M., Souza, M. M. d., Oliveira, K. K., Arruda, C. F. J., de Paula, F., Bettim, B. B., Marques, M. M., Kowalski, L. P., Pinto, C. A. L., Arana-Chavez, V. E., Lourenço, S. V., & Coutinho-Camillo, C. M. (2020). Aquaporin 1, 3, and 5 Patterns in Salivary Gland Mucoepidermoid Carcinoma: Expression in Surgical Specimens and an In Vitro Pilot Study. International Journal of Molecular Sciences, 21(4), 1287. https://doi.org/10.3390/ijms21041287






