Dynamics of Salivary Gland AQP5 under Normal and Pathologic Conditions
Abstract
1. Introduction
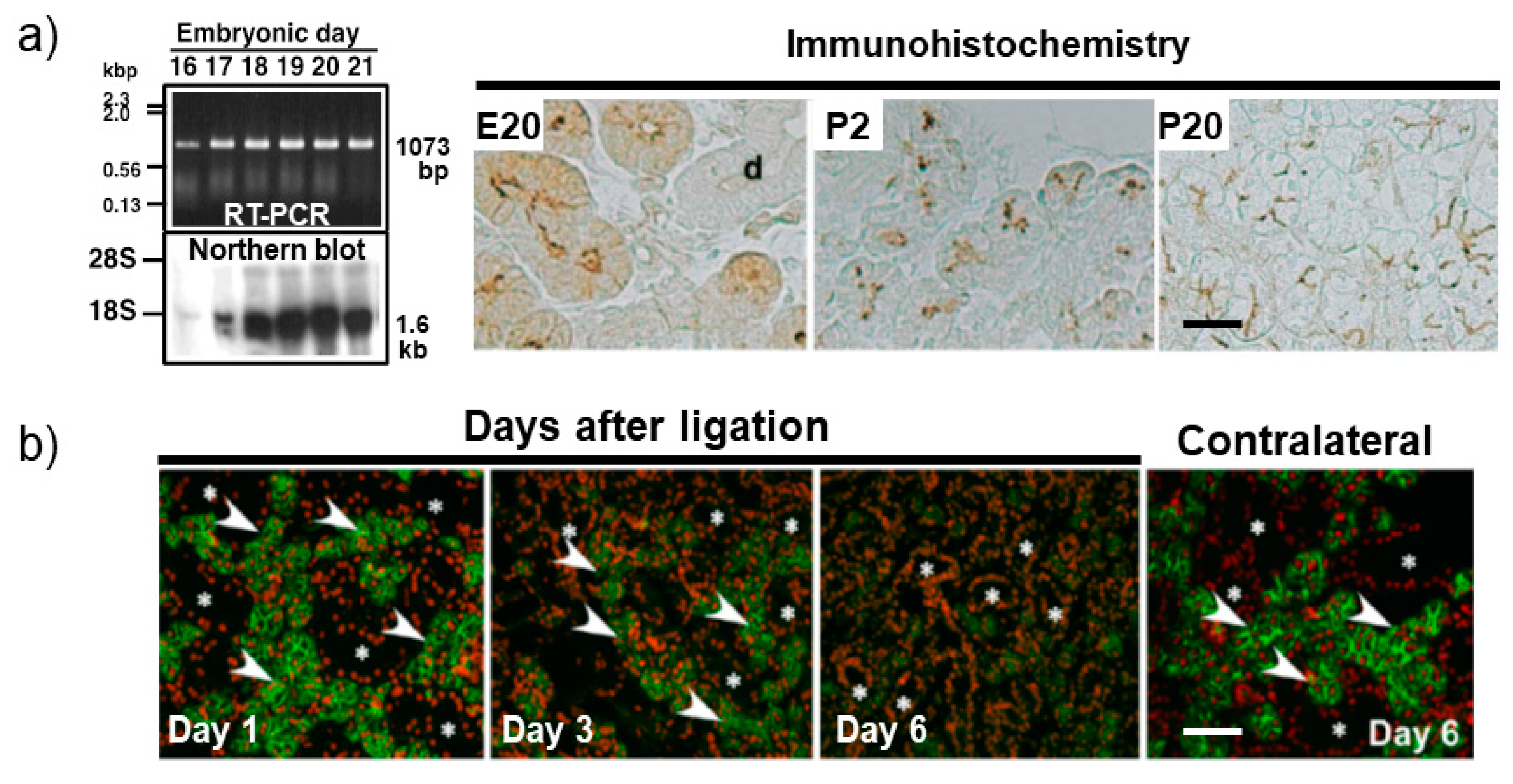
2. Expression and Localization of Salivary Gland Aquaporin 5 during Development, Differentiation, and Regeneration
3. Effects of the Autonomic Nervous System and Chemical Transmitters on Salivary Gland AQP5
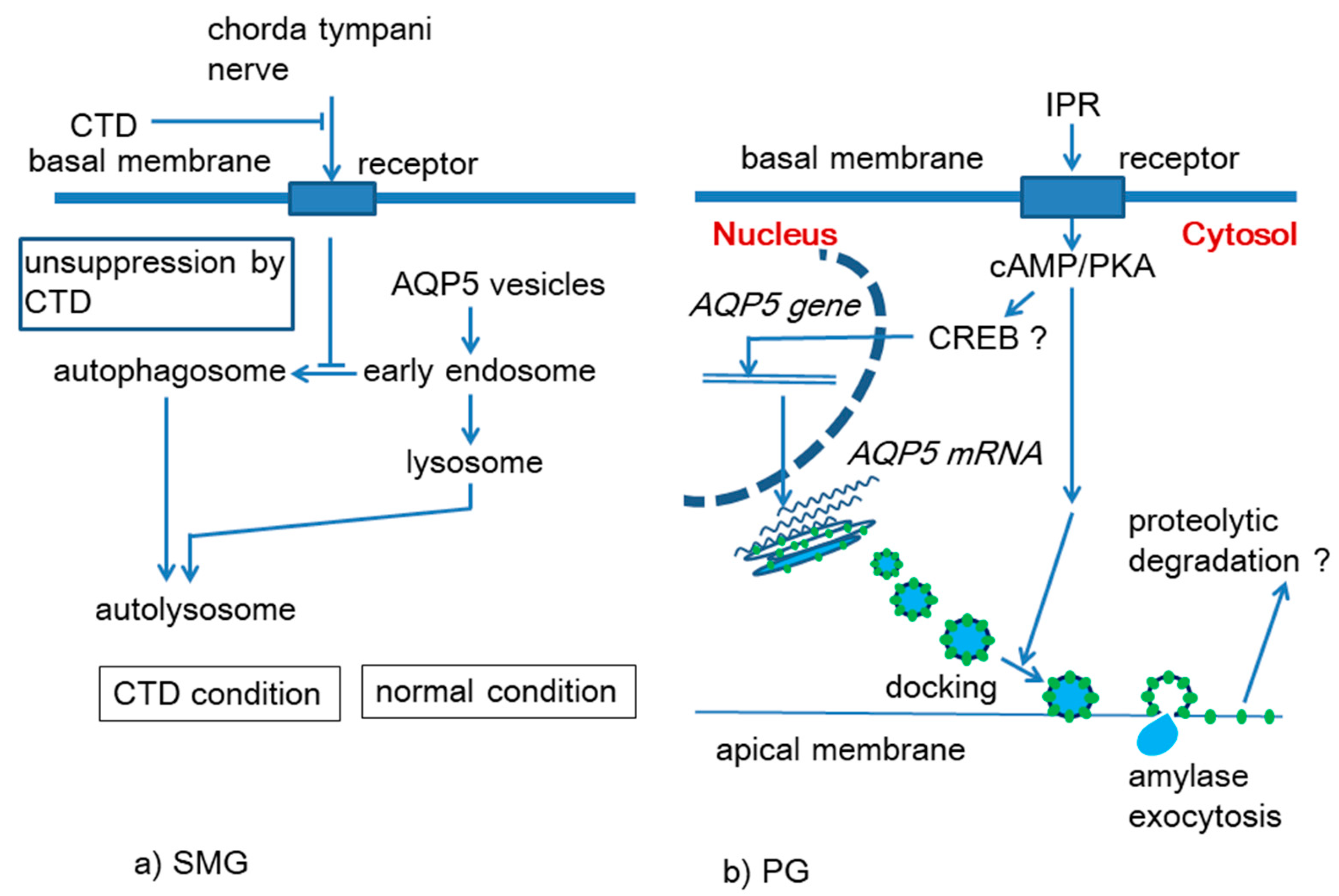
3.1. Effects of Chorda Tympani Denervation and Administration of M3 Receptor Agonist on AQP5 in the Submandibular Gland
3.2. Effects of Isoproterenol on AQP5 in the Mouse Parotid Gland
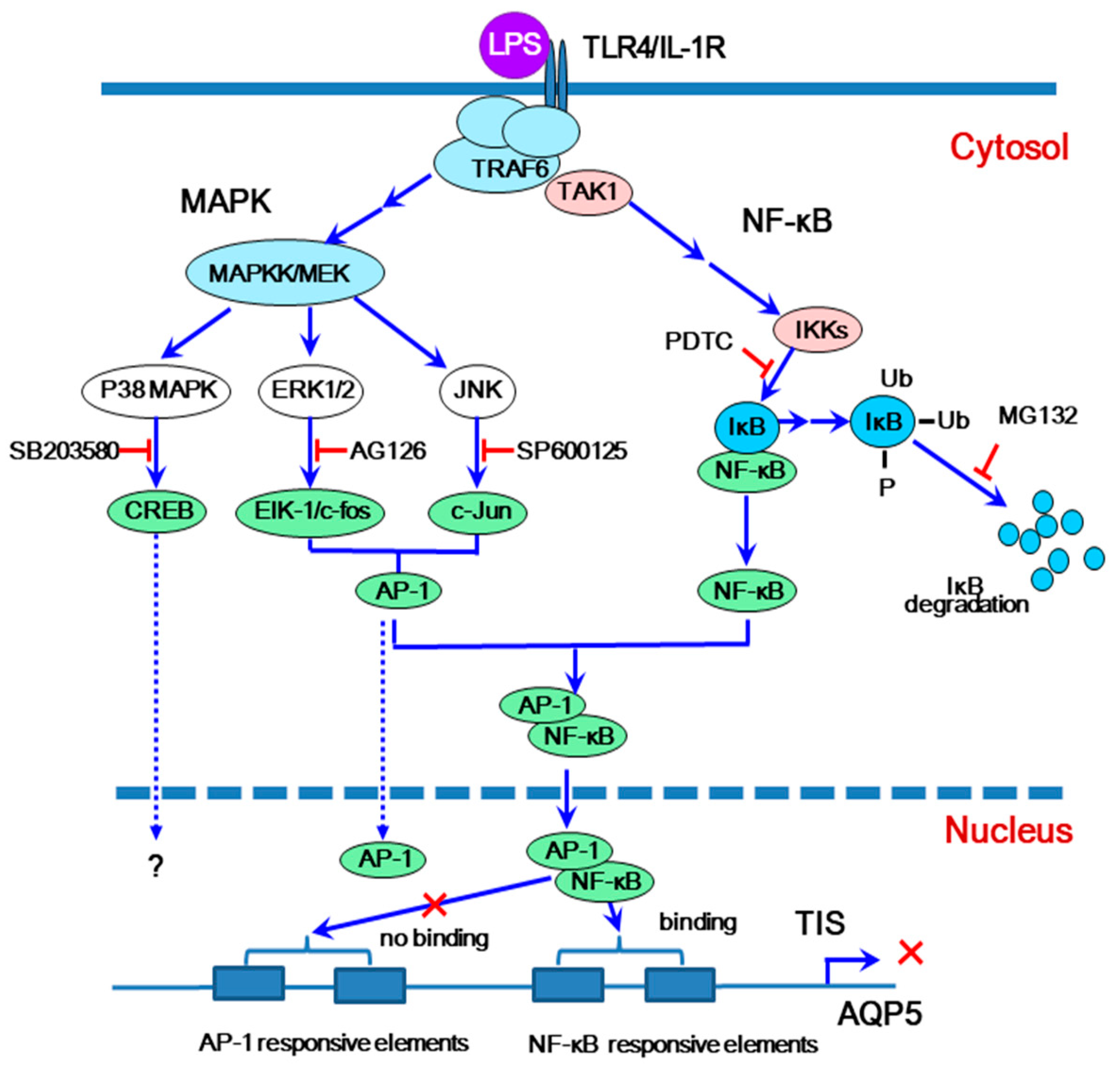
4. Downregulation of Salivary Gland AQP5 by Lipopolysaccharide via Cross-Coupling of Transcription Factors
5. Post-Translational Modifications of AQP5
5.1. Phosphorylation Sites of AQP5 and Related Signals
5.2. Roles of cAMP-Dependent AQP5 Phosphorylation
5.3. Possibility for Phosphorylation-Regulated AQP5 Gating
5.4. Ubiquitination of AQP5
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AP-1 | activator protein-1 |
| AQP | aquaporin |
| CTD | chorda tympani denervation |
| IPR | isoproterenol |
| LPS | lipopolysaccharide |
| MAPK | mitogen-activated protein kinase |
| NF-κB | nuclear factor-kappa B |
| PDTC | pyrrolidine dithiocarbamate |
| PG | parotid gland |
| PKA | protein kinase A |
| RT-PCR | reverse transcriptase-polymerase chain reaction |
| SMG | submandibular gland |
| TLR4 | Toll-like receptor 4 |
| TUNEL | TdT-mediated dUTP nick end labeling |
References
- Preston, G.M.; Carroll, T.P.; Guggino, W.B.; Agre, P. Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 1992, 256, 385–387. [Google Scholar] [CrossRef] [PubMed]
- Hosoi, K. Physiological role of aquaporin 5 in salivary glands. Pflügers Arch. Eur. J. Physiol. 2016, 468, 519–539. [Google Scholar] [CrossRef] [PubMed]
- Zardoya, R. Phylogeny and evolution of the major intrinsic protein family. Biol. Cell 2005, 97, 397–414. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, P.; Day, R.E.; Salman, M.M.; Conner, M.T.; Bill, R.M.; Conner, A.C. Beyond water homeostasis: Diverse functional roles of mammalian aquaporins. Biochim. Biophys. Acta 2015, 1850, 2410–2421. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, Z.; Song, Y.; Zhang, P.; Hu, J.; Bai, C. Expression of aquaporin 5 increases proliferation and metastasis potential of lung cancer. J. Pathol. 2010, 22, 210–220. [Google Scholar] [CrossRef]
- Verkman, A.S.; Anderson, M.O.; Papadopoulos, M.C. Aquaporins: Important but elusive drug targets. Nat. Rev. Drug Discov. 2014, 13, 259–277. [Google Scholar] [CrossRef]
- Murdiastuti, K.; Purwanti, N.; Karabasil, M.R.; Li, X.; Yao, C.; Akamatsu, T.; Kanamori, N.; Hosoi, K. A naturally occurring point mutation in the rat aquaporin 5 gene, influencing its protein production by and secretion of water from salivary glands. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 291, G1081–G1088. [Google Scholar] [CrossRef]
- Ma, T.; Song, Y.; Gillespie, A.; Carlson, E.J.; Epstein, C.J.; Verkman, A.S. Defective secretion of saliva in transgenic mice lacking aquaporin-5 water channels. J. Biol. Chem. 1999, 274, 20071–20074. [Google Scholar] [CrossRef]
- Ichiyama, T.; Nakatani, E.; Tatsumi, K.; Hideshima, K.; Urano, T.; Nariai, Y.; Sekine, J. Expression of aquaporin 3 and 5 as a potential marker for distinguishing dry mouth from Sjögren’s syndrome. J. Oral Sci. 2018, 60, 212–220. [Google Scholar] [CrossRef]
- Steinfeld, S.; Cogan, E.; King, L.S.; Agre, P.; Kiss, R.; Delporte, C. Abnormal distribution of aquaporin-5 water channel protein in salivary glands from Sjögren’s syndrome patients. Lab. Investig. 2001, 81, 143–148. [Google Scholar] [CrossRef]
- Raina, S.; Preston, G.M.; Guggino, W.B.; Agre, P. Molecular cloning and characterization of an aquaporin cDNA from salivary, lacrimal, and respiratory tissues. J. Biol. Chem. 1995, 270, 1908–1912. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, T.; Suzuki, T.; Koyama, H.; Tanaka, S.; Takata, K. Aquaporin-5 (AQP5), a water channel protein, in the rat salivary and lacrimal glands: Immunolocalization and effect of secretory stimulation. Cell Tissue Res. 1999, 295, 513–521. [Google Scholar] [CrossRef]
- Azlina, A.; Javkhlan, P.; Hiroshima, Y.; Hasegawa, T.; Yao, C.; Akamatsu, T.; Hosoi, K. Roles of lysosomal proteolytic systems in AQP5 degradation in the submandibular gland of rats following chorda tympani parasympathetic denervation. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G1106–G1117. [Google Scholar] [CrossRef] [PubMed]
- King, L.S.; Nielsen, S.; Agre, P. Aquaporins in complex tissues. I. Developmental patterns in respiratory and glandular tissues of rat. Am. J. Physiol. Cell Physiol. 1997, 273, C1541–C1548. [Google Scholar] [CrossRef]
- Akamatsu, T.; Parvin, M.N.; Murdiastuti, K.; Kosugi-Tanaka, C.; Yao, C.; Miki, O.; Kanamori, N.; Hosoi, K. Expression and localization of aquaporins, members of the water channel family, during development of the rat submandibular gland. Pflügers Arch. Eur. J. Physiol. 2003, 446, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Tada, J.; Sawa, T.; Yamanaka, N.; Shonn, M.; Akamatsu, T.; Tsumura, K.; Parvin, M.N.; Kanamori, N.; Hosoi, K. Involvement of vesicle-cytoskeleton interaction in AQP5 trafficking in AQP5-gene-transfected HSG cells. Biochem. Biophys. Res. Commun. 1999, 266, 443–447. [Google Scholar] [CrossRef]
- Akamatsu, T.; Azlina, A.; Purwanti, N.; Karabasil, M.R.; Hasegawa, T.; Yao, C.; Hosoi, K. Inhibition and transcriptional silencing of a subtilisin-like pro-protein convertase, PACE4/SPC4, reduces the branching morphogenesis of and AQP5 expression in rat embryonic submandibular gland. Dev. Biol. 2009, 325, 434–443. [Google Scholar] [CrossRef]
- Larsen, H.S.; Ruus, A.K.; Galtung, H.K. Aquaporin expression patterns in the developing mouse salivary gland. Eur. J. Oral Sci. 2009, 117, 655–662. [Google Scholar] [CrossRef]
- Aure, M.H.; Larsen, H.S.; Ruus, A.K.; Galtung, H.K. Aquaporin 5 distribution pattern during development of the mouse sublingual salivary gland. J. Mol. Histol. 2011, 42, 401–408. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, Y.; Zheng, C.; Zhang, X. Knockdown of AQP1 inhibits growth and invasion of human ovarian cancer cells. Mol. Med. Rep. 2017, 16, 5499–5504. [Google Scholar] [CrossRef]
- Luo, L.; Yang, R.; Zhao, S.; Chen, Y.; Hong, S.; Wang, K.; Wang, T.; Cheng, J.; Zhang, T.; Chen, D. Decreased miR-320 expression is associated with breast cancer progression, cell migration, and invasiveness via targeting Aquaporin 1. Acta Biochim. Biophys. Sin. (Shanghai) 2018, 50, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Alejandra, R.; Natalia, S.; Alicia, E.D. The blocking of aquaporin-3 (AQP3) impairs extravillous trophoblast cell migration. Biochem. Biophys. Res. Commun. 2018, 499, 227–232. [Google Scholar] [CrossRef]
- Guan, Y.; Chen, J.; Zhan, Y.; Lu, H. Effects of dexamethasone on C6 cell proliferation, migration and invasion through the upregulation of AQP1. Oncol. Lett. 2018, 15, 7595–7602. [Google Scholar] [CrossRef]
- Login, F.H.; Jensen, H.H.; Pedersen, G.A.; Koffman, J.S.; Kwon, T.H.; Parsons, M.; Nejsum, L.N. Aquaporins differentially regulate cell-cell adhesion in MDCK cells. FASEB J. 2019, 33, 6980–6994. [Google Scholar] [CrossRef]
- Takahashi, S.; Nakamura, S.; Suzuki, R.; Islam, N.; Domon, T.; Yamamoto, T.; Wakita, M. Apoptosis and mitosis of parenchymal cells in the duct-ligated rat submandibular gland. Tissue Cell 2000, 32, 457–463. [Google Scholar] [CrossRef]
- Walker, N.I.; Gobé, G.C. Cell death and cell proliferation during atrophy of the rat parotid gland induced by duct obstruction. J. Pathol. 1987, 153, 333–344. [Google Scholar] [CrossRef]
- Purwanti, N.; Karabasil, M.R.; Matsuo, S.; Chen, G.; Javkhlan, P.; Azlina, A.; Hasegawa, T.; Yao, C.; Akamatsu, T.; Hosoi, K. Induction of Sca-1 via activation of STAT3 system in the duct cells of the mouse submandibular gland by ligation of main excretory duct. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G814–G824. [Google Scholar] [CrossRef]
- Yasumitsu, T.; Shimizu, O.; Shiratsuchi, H.; Miyake, Y.; Yonehara, Y. Distribution of aquaporin-5, transforming growth factor-β(1) and laminin during regeneration of atrophic rat submandibular glands after duct ligation. J. Oral Sci. 2018, 60, 595–600. [Google Scholar] [CrossRef]
- Aure, M.H.; Konieczny, S.F.; Ovitt, C.E. Salivary gland homeostasis is maintained through acinar cell self-duplication. Dev. Cell 2015, 33, 231–237. [Google Scholar] [CrossRef]
- Aure, M.H.; Arany, S.; Ovitt, C.E. Salivary Glands: Stem Cells, Self-duplication, or Both? J. Dent. Res. 2015, 94, 1502–1507. [Google Scholar] [CrossRef]
- Li, X.; Azlina, A.; Karabasil, M.R.; Purwanti, N.; Hasegawa, T.; Yao, C.; Akamatsu, T.; Hosoi, K. Degradation of submandibular gland AQP5 by parasympathetic denervation of chorda tympani and its recovery by cevimeline, an M3 muscarinic agonist. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G112–G123. [Google Scholar] [CrossRef] [PubMed]
- Endoh, T. Modulation of voltage-dependent calcium channels by neurotransmitters and neuropeptides in parasympathetic submandibular ganglion neurons. Arch. Oral Biol. 2004, 49, 539–557. [Google Scholar] [CrossRef] [PubMed]
- Proctor, G.B.; Carpenter, G.H. Regulation of salivary gland function by autonomic nerves. Auton. Neurosci. 2007, 133, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Nederfors, T.; Ericsson, T.; Twetman, S.; Dahlöf, C. Effects of the betaadrenoceptor antagonists atenolol and propranolol on human parotid and submandibular-sublingual salivary secretion. J. Dent. Res. 1994, 73, 5–10. [Google Scholar] [CrossRef]
- Baum, B.J. Principles of saliva secretion. Ann. N. Y. Acad. Sci. 1993, 694, 17–23. [Google Scholar] [CrossRef]
- Yang, F.; Kawedia, J.D.; Menon, A.G. Cyclic AMP regulates aquaporin 5 expression at both transcriptional and post-transcriptional levels through a protein kinase A pathway. J. Biol. Chem. 2003, 278, 32173–32180. [Google Scholar] [CrossRef]
- Chen, G.; Yao, C.; Hasegawa, T.; Akamatsu, T.; Yoshimura, H.; Hosoi, K. Effects of isoproterenol on aquaporin 5 levels in the parotid gland of mice in vivo. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E100–E108. [Google Scholar] [CrossRef]
- Matsuki, M.; Hashimoto, S.; Shimono, M.; Murakami, M.; Fujita-Yoshigaki, J.; Furuyama, S.; Sugiya, H. Involvement of aquaporin-5 water channel in osmoregulation in parotid secretory granules. J. Membr. Biol. 2005, 203, 119–126. [Google Scholar] [CrossRef]
- Beutler, B.; Rietschel, E.T. Innate immune sensing and its roots: The story of endotoxin. Nat. Rev. Immunol. 2003, 3, 169–176. [Google Scholar] [CrossRef]
- Salman, M.M.; Sheilabi, M.A.; Bhattacharyya, D.; Kitchen, P.; Conne, A.C.; Bill, R.M.; Woodroofe, M.N.; Conner, M.T.; Princivalle, A.P. Transcriptome analysis suggests a role for the differential expression of cerebral aquaporins and the MAPK signalling pathway in human temporal lobe epilepsy. Eur. J. Neurosci. 2017, 46, 2121–2132. [Google Scholar] [CrossRef]
- Yang, M.; Gao, F.; Liu, H.; Yu, W.H.; Zhuo, F.; Qiu, G.P.; Ran, J.H.; Sun, S.Q. Hyperosmotic induction of aquaporin expression in rat astrocytes through a different MAPK pathway. J. Cell Biochem. 2013, 114, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Stambe, C.; Nikolic-Paterson, D.J.; Hill, P.A.; Dowling, J.; Atkins, R.C. p38 Mitogen-activated protein kinase activation and cell localization in human glomerulonephritis: Correlation with renal injury. J. Am. Soc. Nephrol. 2004, 15, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Li, X.; Kwartarini, M.; Kosugi-Tanaka, C.; Akamatsu, T.; Kanamori, N.; Hosoi, K. Lipopolysaccharide-induced elevation and secretion of interleukin-1beta in the submandibular gland of male mice. Immunology 2005, 116, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Karabasil, M.R.; Purwanti, N.; Li, X.; Akamatsu, T.; Kanamori, N.; Hosoi, K. Tissue kallikrein mK13 is a candidate processing enzyme for the precursor of interleukin-1beta in the submandibular gland of mice. J. Biol. Chem. 2006, 281, 7968–7976. [Google Scholar] [CrossRef]
- Yao, C.; Purwanti, N.; Karabasil, M.R.; Azlina, A.; Javkhlan, P.; Hasegawa, T.; Akamatsu, T.; Hosoi, T.; Ozawa, K.; Hosoi, K. Potential down-regulation of salivary gland AQP5 by LPS via cross-coupling of NF-kappaB and p-c-Jun/c-Fos. Am. J. Pathol. 2010, 177, 724–734. [Google Scholar] [CrossRef]
- Schüle, R.; Umesono, K.; Mangelsdorf, D.J.; Bolado, J.; Pike, J.W.; Evans, R.M. Jun-Fos and receptors for vitamins A and D recognize a common response element in the human osteocalcin gene. Cell 1990, 61, 497–504. [Google Scholar] [CrossRef]
- Schüle, R.; Rangarajan, P.; Yang, N.; Kliewer, S.; Ransone, L.J.; Bolado, J.; Verma, I.M.; Evans, R.M. Retinoic acid is a negative regulator of AP-1-responsive genes. Proc. Natl. Acad. Sci. USA 1991, 88, 6092–6096. [Google Scholar] [CrossRef]
- Stein, B.; Baldwin, A.S., Jr.; Ballard, D.W.; Greene, W.C.; Angel, P.; Herrlich, P. Cross-coupling of the NF-kappa B p65 and Fos/Jun transcription factors produces potentiated biological function. EMBO J. 1993, 12, 3879–3891. [Google Scholar] [CrossRef]
- Shyu, Y.J.; Suarez, C.D.; Hu, C.D. Visualization of AP-1 NF-κB ternary complexes in living cells by using a BiFC-based FRET. Proc. Natl. Acad. Sci. USA 2008, 105, 151–156. [Google Scholar] [CrossRef]
- Conner, M.T.; Conner, A.C.; Bland, C.E.; Taylor, L.H.J.; Brown, J.E.P.; Parri, H.R.; Bill, R.M. Rapid aquaporin translocation regulates cellular water flow: Mechanism of hypotonicity-induced subcellular localization of aquaporin 1 water channel. J. Biol. Chem. 2012, 287, 11516–11525. [Google Scholar] [CrossRef]
- Kitchen, P.; Day, R.E.; Taylor, L.H.J.; Salman, M.M.; Bill, R.M.; Conner, M.T.; Conner, A.C. Identification and molecular mechanisms of the rapid tonicity-induced relocalization of the aquaporin 4 channel. J. Biol. Chem. 2015, 290, 16873–16881. [Google Scholar] [CrossRef] [PubMed]
- Fushimi, K.; Uchida, S.; Hara, Y.; Hirata, Y.; Marumo, F.; Sasaki, S. Cloning and expression of apical membrane water channel of rat kidney collecting tubule. Nature 1993, 361, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Kamsteeg, E.-J.; Hendriks, G.; Boone, M.; Konings, I.B.M.; Oorschot, V.; van der Sluijs, P.; Klumperman, J.; Deen, P.M.T. Short-chain ubiquitination mediates the regulated endocytosis of the aquaporin-2 water channel. Proc. Natl. Acad. Sci. USA 2006, 103, 18344–18349. [Google Scholar] [CrossRef]
- Törnroth-Horsefield, S.; Wang, Y.; Hedfalk, K.; Johanson, U.; Karlsson, M.; Tajkhorshid, E.; Neutze, R.; Kjellbom, P. Structural mechanism of plant aquaporin gating. Nature 2006, 439, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Fischer, G.; Kosinska-Eriksson, U.; Aponte-Santamaría, C.; Palmgren, M.; Geijer, C.; Hedfalk, K.; Hohmann, S.; De Groot, B.L.; Neutze, R.; Lindkvist-Petersson, K. Crystal structure of a yeast aquaporin at 1.15 Å reveals a novel gating mechanism. PLoS Biol. 2009, 7, e1000130. [Google Scholar] [CrossRef] [PubMed]
- Kalman, K.; Németh-Cahalan, K.L.; Froger, A.; Hall, J.E. Phosphorylation determines the calmodulin-mediated Ca2+ response and water permeability of AQP0. J. Biol. Chem. 2008, 283, 21278–21283. [Google Scholar] [CrossRef] [PubMed]
- Fenton, R.A.; Moeller, H.B.; Hoffert, J.D.; Yu, M.J.; Nielsen, S.; Knepper, M.A. Acute regulation of aquaporin-2 phosphorylation at Ser-264 by vasopressin. Proc. Natl. Acad. Sci. USA 2008, 105, 3134–3139. [Google Scholar] [CrossRef] [PubMed]
- Hoffert, J.D.; Fenton, R.A.; Moeller, H.B.; Simons, B.; Tchapyjnikov, D.; McDill, B.W.; Yu, M.J.; Pisitkun, T.; Chen, F.; Knepper, M.A. Vasopressin-stimulated increase in phosphorylation at Ser269 potentiates plasma membrane retention of aquaporin-2. J. Biol. Chem. 2008, 283, 24617–24627. [Google Scholar] [CrossRef]
- Hoffert, J.D.; Nielsen, J.; Yu, M.J.; Pisitkun, T.; Schleicher, S.M.; Nielsen, S.; Knepper, M.A. Dynamics of aquaporin-2 serine-261 phosphorylation in response to short-term vasopressin treatment in collecting duct. Am. J. Physiol. Ren. Physiol. 2007, 292, F691–F700. [Google Scholar] [CrossRef]
- Nishimoto, G.; Zelenina, M.; Li, D.; Yasui, M.; Aperia, A.; Nielsen, S.; Nairn, A.C. Arginine vasopressin stimulates phosphorylation of aquaporin-2 in rat renal tissue. Am. J. Physiol. Ren. Physiol. 1999, 276, F254–F259. [Google Scholar] [CrossRef]
- Hasegawa, T.; Azlina, A.; Javkhlan, P.; Yao, C.; Akamatsu, T.; Hosoi, K. Novel phosphorylation of aquaporin-5 at its threonine 259 through cAMP signaling in salivary gland cells. Am. J. Physiol. Cell Physiol. 2011, 301, C667–C678. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.; Chae, Y.K.; Jang, S.J.; Kim, M.S.; Baek, J.H.; Park, J.C.; Trink, B.; Ratovitski, E.; Lee, T.; Park, B.; et al. Membrane trafficking of AQP5 and cAMP dependent phosphorylation in bronchial epithelium. Biochem. Biophys. Res. Commun. 2008, 366, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Kosugi-Tanaka, C.; Li, X.; Yao, C.; Akamatsu, T.; Kanamori, N.; Hosoi, K. Protein kinase A-regulated membrane trafficking of a green fluorescent protein-aquaporin 5 chimera in MDCK cells. Biochim. Biophys. Acta Mol. Cell Res. 2006, 1763, 337–344. [Google Scholar] [CrossRef]
- Kitchen, P.; Öberg, F.; Sjöhamn, J.; Hedfalk, K.; Bill, R.M.; Conner, A.C.; Conner, M.T.; Törnroth-Horsefield, S. Plasma membrane abundance of human aquaporin 5 is dynamically regulated by multiple pathways. PLoS ONE 2015, 10, e0143027. [Google Scholar] [CrossRef]
- Koffman, J.S.; Arnspang, E.C.; Marlar, S.; Nejsum, L.N. Opposing effects of cAMP and T259 phosphorylation on plasma membrane diffusion of the water channel aquaporin-5 in madin-darby canine kidney cells. PLoS ONE 2015, 10, e0133324. [Google Scholar] [CrossRef]
- Biswas, R.; Ahn, J.C.; Moon, J.H.; Kim, J.; Choi, Y.H.; Park, S.Y.; Chung, P.S. Low-level laser therapy with 850nm recovers salivary function via membrane redistribution of aquaporin 5 by reducing intracellular Ca(2+) overload and ER stress during hyperglycemia. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 1770–1780. [Google Scholar] [CrossRef]
- Moon, C.; Sidransky, D.; Gong, G.; Woo, J.; Soria, J.C.; Lee, J.; Kim, M.-J.; Kim, M.S.; Jang, S.J.; Lee, S.K.; et al. Expression of aquaporin 5 (AQP5) promotes tumor invasion in human non small cell lung cancer. PLoS ONE 2008, 3, e2162. [Google Scholar]
- Woo, J.; Lee, J.; Kim, M.S.; Jang, S.J.; Sidransky, D.; Moon, C. The effect of aquaporin 5 overexpression on the Ras signaling pathway. Biochem. Biophys. Res. Commun. 2008, 367, 291–298. [Google Scholar] [CrossRef]
- Woo, J.; Lee, J.; Chae, Y.K.; Kim, M.S.; Baek, J.H.; Park, J.C.; Park, M.J.; Smith, I.M.; Trink, B.; Ratovitski, E.; et al. Overexpression of AQP5, a putative oncogene, promotes cell growth and transformation. Cancer Lett. 2008, 264, 54–62. [Google Scholar] [CrossRef]
- Jung, H.J.; Park, J.Y.; Jeon, H.S.; Kwon, T.H. Aquaporin-5: A marker protein for proliferation and migration of human breast cancer cells. PLoS ONE 2011, 6, e28492. [Google Scholar] [CrossRef]
- Guo, X.; Sun, T.; Yang, M.; Li, Z.; Li, Z.; Gao, Y. Prognostic value of combined aquaporin 3 and aquaporin 5 overexpression in hepatocellular carcinoma. BioMed Res. Int. 2013, 2013, 206525. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Fujii, T.; Oya, T.; Horikawa, N.; Tabuchi, Y.; Takahashi, Y.; Morii, M.; Takeguchi, N.; Tsukada, K.; Sakai, H. Involvement of aquaporin-5 in differentiation of human gastric cancer cells. J. Physiol. Sci. 2009, 59, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Sung, K.K.; Young, K.C.; Woo, J.; Myoung, S.K.; Jong, C.P.; Lee, J.; Soria, J.C.; Se, J.J.; Sidransky, D.; Moon, C. Role of human aquaporin 5 in colorectal carcinogenesis. Am. J. Pathol. 2008, 173, 518–525. [Google Scholar]
- Nakamachi, T.; Ohtaki, H.; Seki, T.; Yofu, S.; Kagami, N.; Hashimoto, H.; Shintani, N.; Baba, A.; Mark, L.; Lanekoff, I.; et al. PACAP suppresses dry eye signs by stimulating tear secretion. Nat. Commun. 2016, 7, 12034. [Google Scholar] [CrossRef]
- Johansson, I.; Karlsson, M.; Shukla, V.K.; Chrispeels, M.J.; Larsson, C.; Kjellbom, P. Water transport activity of plasma membrane aquaporin PM28A is regulated by phosphorylation. Plant Cell 1998, 10, 451–459. [Google Scholar] [CrossRef]
- Rodrigues, C.; Mósca, A.F.; Martins, A.P.; Nobre, T.; Prista, C.; Antunes, F.; Gasparovic, A.C.; Soveral, G. Rat aquaporin-5 is pH-gated induced by phosphorylation and is implicated in oxidative stress. Int. J. Mol. Sci. 2016, 17, 2090. [Google Scholar] [CrossRef]
- Satoh, K.; Narita, T.; Matsuki-Fukushima, M.; Okabayashi, K.; Ito, T.; Senpuku, H.; Sugiya, H. E2f1-deficient NOD/SCID mice have dry mouth due to a change of acinar/duct structure and the down-regulation of AQP5 in the salivary gland. Pflügers Arch. Eur. J. Physiol. 2013, 465, 271–281. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hosoi, K.; Yao, C.; Hasegawa, T.; Yoshimura, H.; Akamatsu, T. Dynamics of Salivary Gland AQP5 under Normal and Pathologic Conditions. Int. J. Mol. Sci. 2020, 21, 1182. https://doi.org/10.3390/ijms21041182
Hosoi K, Yao C, Hasegawa T, Yoshimura H, Akamatsu T. Dynamics of Salivary Gland AQP5 under Normal and Pathologic Conditions. International Journal of Molecular Sciences. 2020; 21(4):1182. https://doi.org/10.3390/ijms21041182
Chicago/Turabian StyleHosoi, Kazuo, Chenjuan Yao, Takahiro Hasegawa, Hiroshi Yoshimura, and Tetsuya Akamatsu. 2020. "Dynamics of Salivary Gland AQP5 under Normal and Pathologic Conditions" International Journal of Molecular Sciences 21, no. 4: 1182. https://doi.org/10.3390/ijms21041182
APA StyleHosoi, K., Yao, C., Hasegawa, T., Yoshimura, H., & Akamatsu, T. (2020). Dynamics of Salivary Gland AQP5 under Normal and Pathologic Conditions. International Journal of Molecular Sciences, 21(4), 1182. https://doi.org/10.3390/ijms21041182



