The Effect of Silver Nanoparticles on Antioxidant/Pro-Oxidant Balance in a Murine Model
Abstract
1. Introduction
2. Results
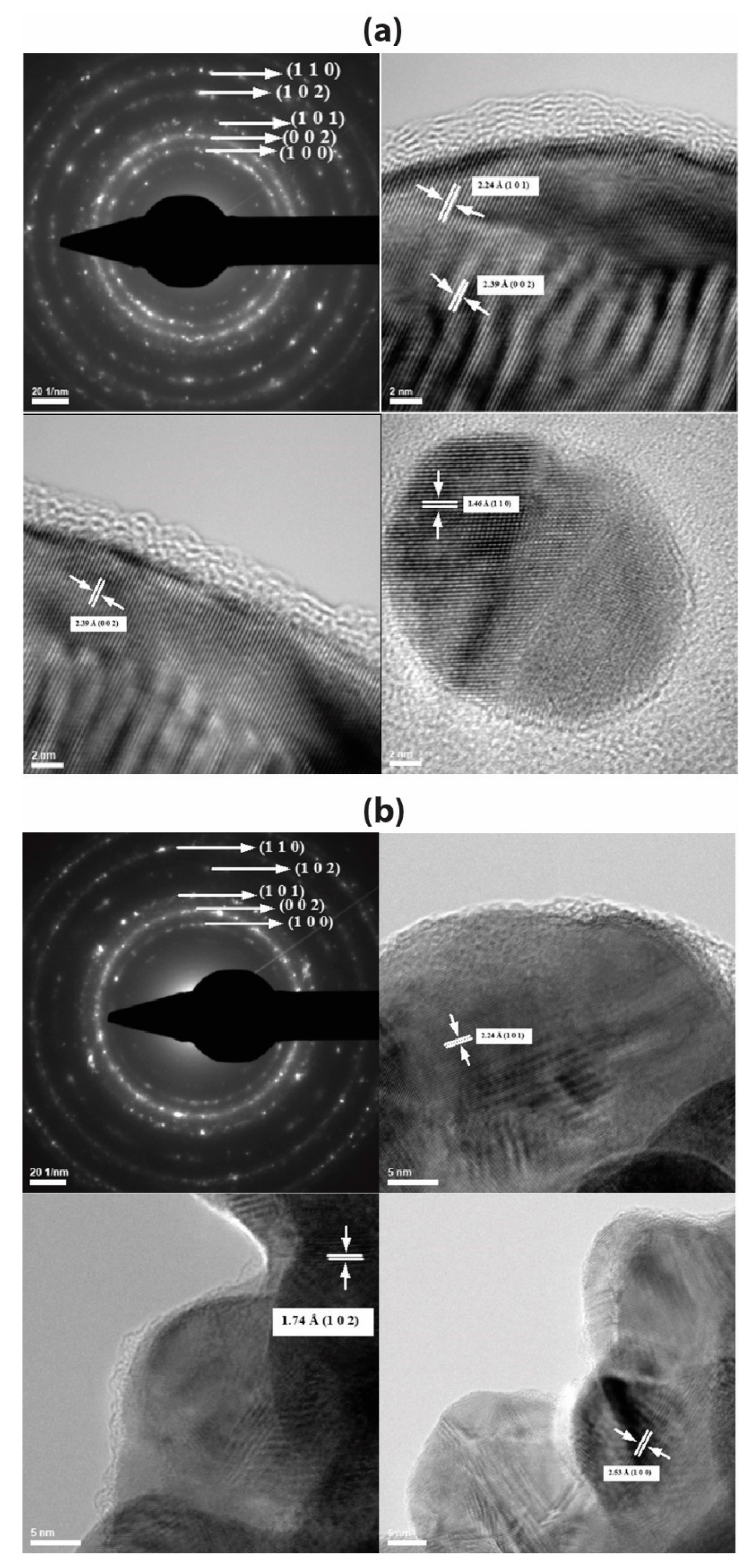
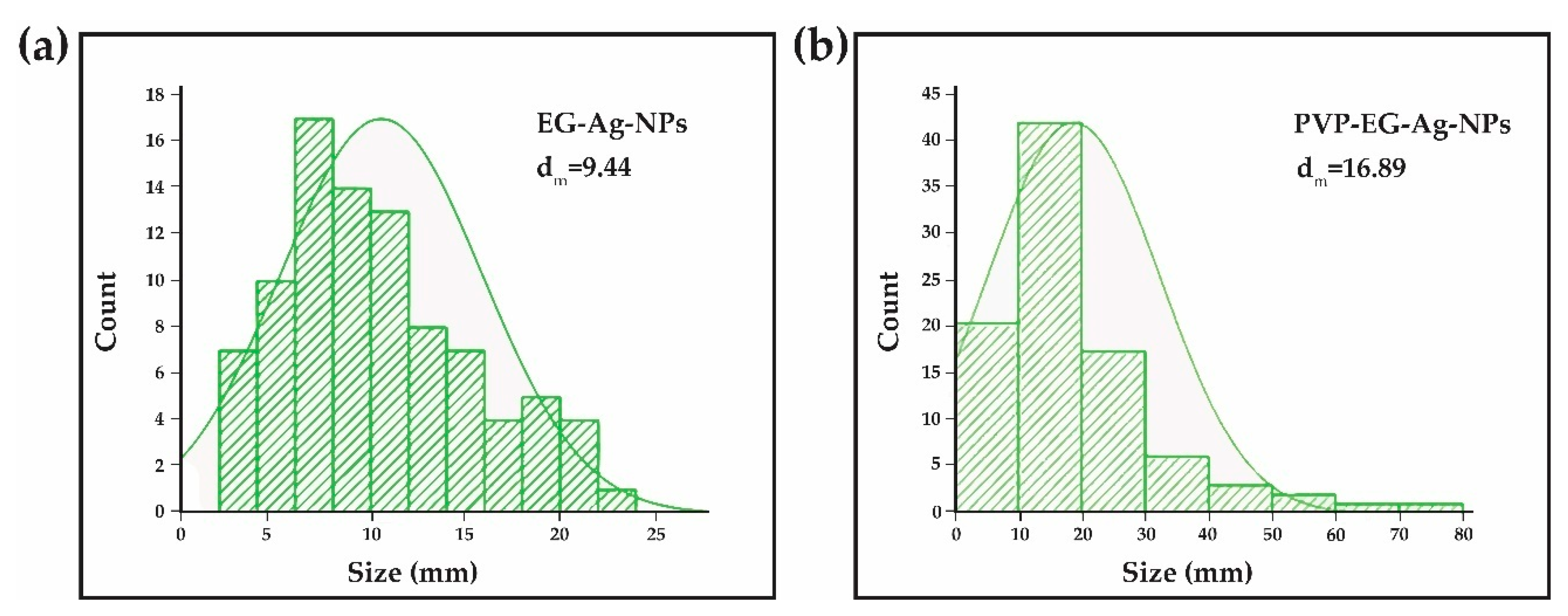
2.1. Transmission Electron Microscopy (TEM) and Selected Area (Electron) Diffraction (SAED)
2.2. The Effect of EG-AgNPs on Anti-Oxidant Markers
2.2.1. TAC Levels
2.2.2. GSH Levels
2.2.3. CAT Activity Levels
2.3. The Effect of PVP-EG-AgNPs on Anti-Oxidant Markers
2.3.1. TAC Levels
2.3.2. GSH Levels
2.3.3. CAT Activity Levels
2.4. The Effect of EG-AgNPs on Pro-Oxidant Markers
2.4.1. TBARS Levels
2.4.2. PROTC Levels
2.5. The Effects of PVP-EG-AgNPs on Pro-Oxidant Markers
2.5.1. TBARS Levels
2.5.2. PROTC Levels
3. Discussion
4. Materials and Methods
4.1. Raw Materials
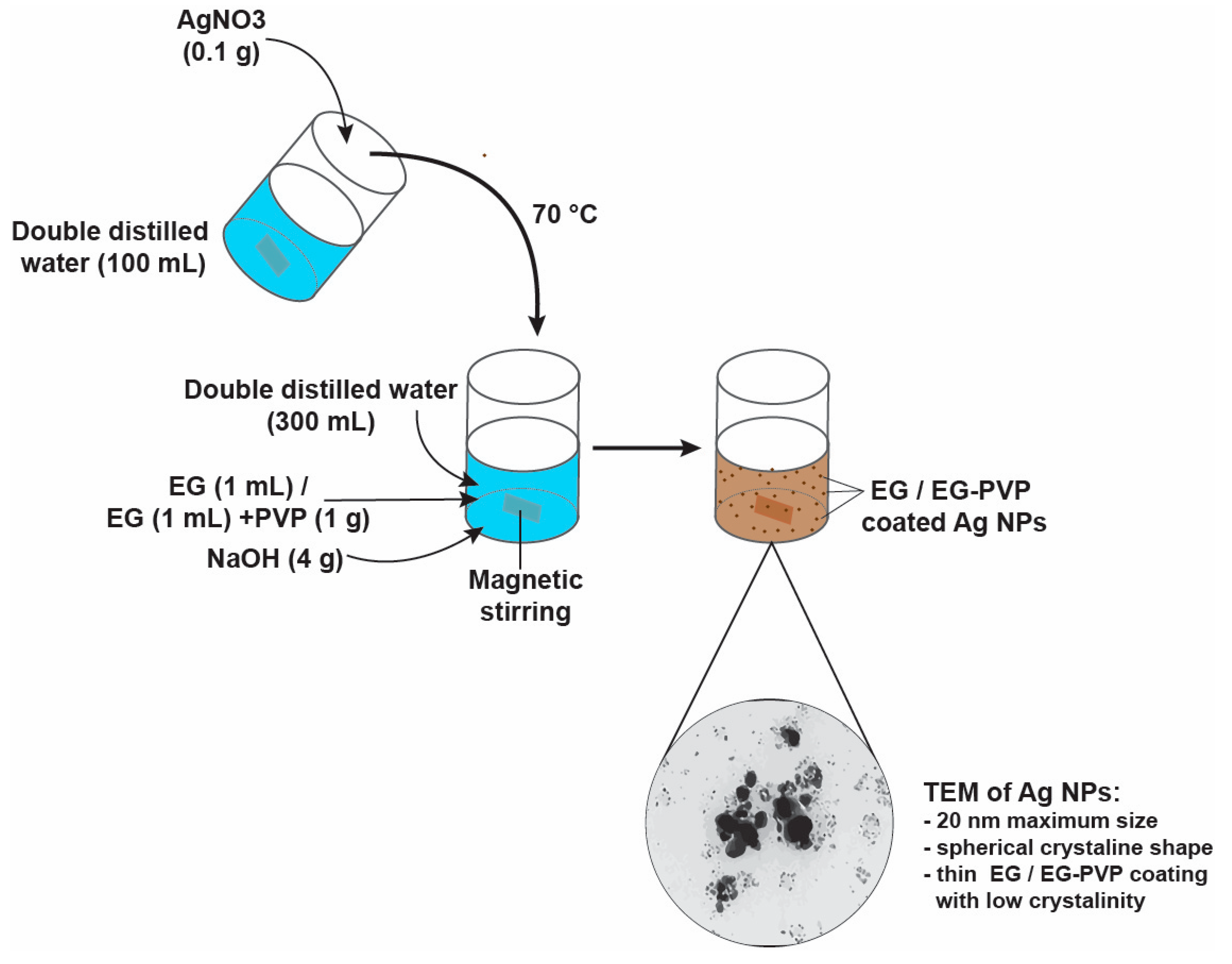
4.2. Synthesis of EG-AgNPs and PVP-EG-AgNPs
4.3. Characterization of EG-AgNPs and PVP-EG-AgNPs
4.3.1. Transmission Electron Microscopy (TEM)
4.3.2. Zeta Potential Measurement
4.4. Animals
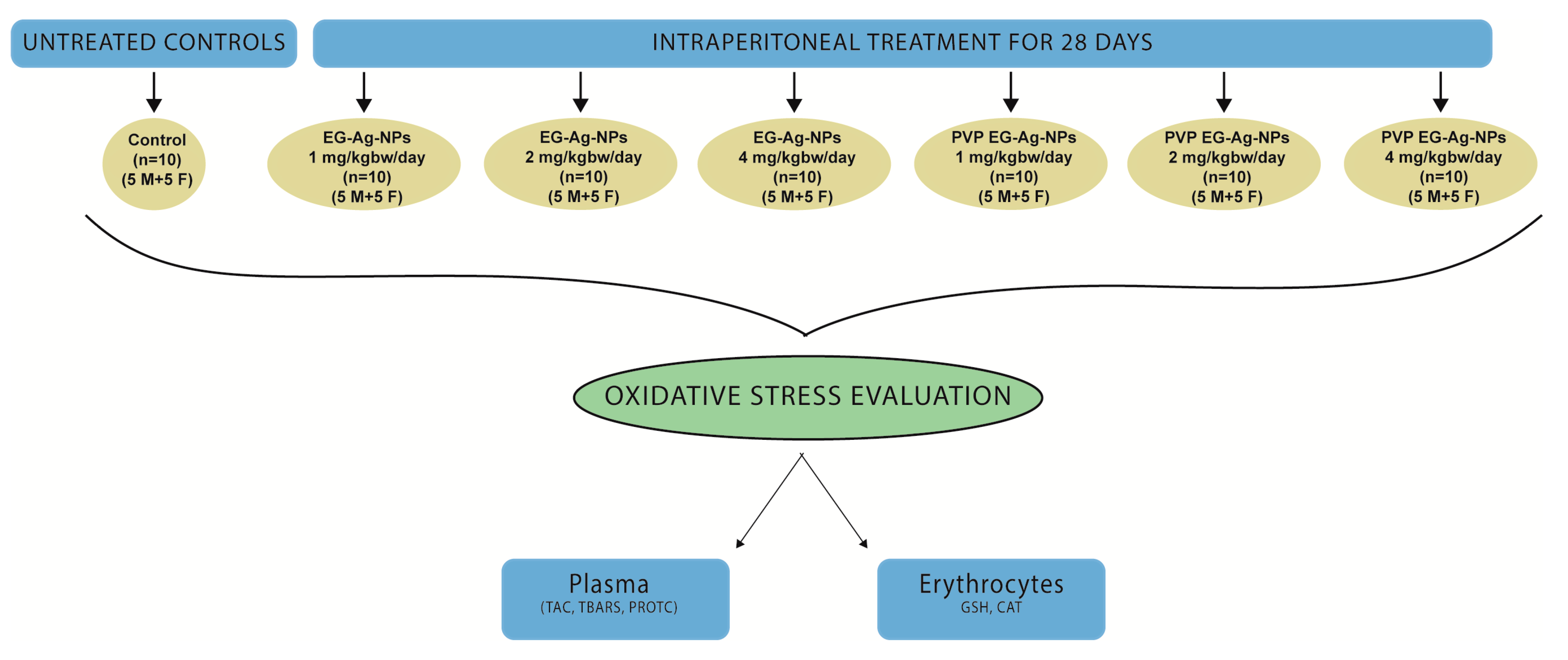
4.5. Experimental Design
4.6. Oxidative Stress Markers Evaluation
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Luss, A.L.; Kulikov, P.P.; Romme, S.B.; Andersen, C.L.; Pennisi, C.P.; Docea, A.O.; Kuskov, A.N.; Velonia, K.; Mezhuev, Y.O.; et al. Nanosized carriers based on amphiphilic poly-N-vinyl-2-pyrrolidone for intranuclear drug delivery. Nanomedicine 2018, 13, 703–715. [Google Scholar] [CrossRef]
- Kuskov, A.N.; Kulikov, P.P.; Goryachaya, A.V.; Tzatzarakis, M.N.; Docea, A.O.; Velonia, K.; Shtilman, M.I.; Tsatsakis, A.M. Amphiphilic poly-N-vinylpyrrolidone nanoparticles as carriers for non-steroidal, anti-inflammatory drugs: In vitro cytotoxicity and in vivo acute toxicity study. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1021–1030. [Google Scholar] [CrossRef]
- Grumezescu, A.M.; Gestal, M.C.; Holban, A.M.; Grumezescu, V.; Vasile, B.S.; Mogoanta, L.; Iordache, F.; Bleotu, C.; Mogosanu, G.D. Biocompatible Fe3O4 increases the efficacy of amoxicillin delivery against Gram-positive and Gram-negative bacteria. Molecules 2014, 19, 5013–5027. [Google Scholar] [CrossRef]
- Radulescu, M.; Andronescu, E.; Cirja, A.; Holban, A.M.; Mogoanta, L.; Balseanu, T.A.; Catalin, B.; Neagu, T.P.; Lascar, I.; Florea, D.A.; et al. Antimicrobial coatings based on zinc oxide and orange oil for improved bioactive wound dressings and other applications. Rom. J. Morphol. Embryol. Rev. Roum. de Morphol. et Embryol. 2016, 57, 107–114. [Google Scholar]
- Ficai, D.; Grumezescu, V.; Fufa, O.M.; Popescu, R.C.; Holban, A.M.; Ficai, A.; Grumezescu, A.M.; Mogoanta, L.; Mogosanu, G.D. Antibiofilm Coatings Based on PLGA and Nanostructured Cefepime-Functionalized Magnetite. Nanomaterials 2018, 8, 633. [Google Scholar] [CrossRef]
- Balaure, P.C.; Holban, A.M.; Grumezescu, A.M.; Mogosanu, G.D.; Balseanu, T.A.; Stan, M.S.; Dinischiotu, A.; Volceanov, A.; Mogoanta, L. In vitro and in vivo studies of novel fabricated bioactive dressings based on collagen and zinc oxide 3D scaffolds. Int. J. Pharm. 2019, 557, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dar, M.Y.; Joshi, B.; Sharma, B.; Shrivastava, S.; Shukla, S. Phytofabrication of Silver nanoparticles: Novel Drug to overcome hepatocellular ailments. Toxicol. Rep. 2018, 5, 333–342. [Google Scholar] [CrossRef]
- Burdusel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoanta, L.; Ficai, A.; Andronescu, E. Biomedical Applications of Silver Nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef] [PubMed]
- Radulescu, M.; Andronescu, E.; Dolete, G.; Popescu, R.C.; Fufa, O.; Chifiriuc, M.C.; Mogoanta, L.; Balseanu, T.A.; Mogosanu, G.D.; Grumezescu, A.M.; et al. Silver Nanocoatings for Reducing the Exogenous Microbial Colonization of Wound Dressings. Materials 2016, 9, 345. [Google Scholar] [CrossRef] [PubMed]
- Kawashita, M.; Tsuneyama, S.; Miyaji, F.; Kokubo, T.; Kozuka, H.; Yamamoto, K. Antibacterial silver-containing silica glass prepared by sol-gel method. Biomaterials 2000, 21, 393–398. [Google Scholar] [CrossRef]
- Buteica, A.S.; Mihaiescu, D.E.; Grumezescu, A.M.; Vasile, B.S.; Popescu, A.; Calina, D.; Mihaiescu, O.M. The Cytotoxicity Of (Non) Magnetic Nanoparticles Tested On Escherichia Coli And Staphylococcus Aureus. Dig. J. Nanomater. Biostruct. 2010, 5, 651–655. [Google Scholar]
- Ali, G.W.; Abd El-Moez, S.H.; Abdel-Fattah, W.A. Synthesis and characterization of nontoxic silver nano-particles with preferential bactericidal activity. Biointerface Res. Appl. Chem. 2019, 9, 4617–4623. [Google Scholar]
- Shen, H.; You, J.; Zhang, G.; Ziemys, A.; Li, Q.; Bai, L.; Deng, X.; Erm, D.R.; Liu, X.; Li, C.; et al. Cooperative, nanoparticle-enabled thermal therapy of breast cancer. Adv. Healthc. Mater. 2012, 1, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Glazer, E.S.; Zhu, C.; Massey, K.L.; Thompson, C.S.; Kaluarachchi, W.D.; Hamir, A.N.; Curley, S.A. Noninvasive radiofrequency field destruction of pancreatic adenocarcinoma xenografts treated with targeted gold nanoparticles. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2010, 16, 5712–5721. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, M.; Thiesen, B.; Jordan, A.; Taymoorian, K.; Gneveckow, U.; Waldofner, N.; Scholz, R.; Koch, M.; Lein, M.; Jung, K.; et al. Magnetic fluid hyperthermia (MFH)reduces prostate cancer growth in the orthotopic Dunning R3327 rat model. Prostate 2005, 64, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Piperigkou, Z.; Karamanou, K.; Engin, A.B.; Gialeli, C.; Docea, A.O.; Vynios, D.H.; Pavao, M.S.; Golokhvast, K.S.; Shtilman, M.I.; Argiris, A.; et al. Emerging aspects of nanotoxicology in health and disease: From agriculture and food sector to cancer therapeutics. Food Chem. Toxicol. 2016, 91, 42–57. [Google Scholar] [CrossRef]
- Engin, A.B.; Nikitovic, D.; Neagu, M.; Henrich-Noack, P.; Docea, A.O.; Shtilman, M.I.; Golokhvast, K.; Tsatsakis, A.M. Mechanistic understanding of nanoparticles’ interactions with extracellular matrix: The cell and immune system. Part. Fibre Toxicol. 2017, 14, 22. [Google Scholar] [CrossRef]
- Neagu, M.; Piperigkou, Z.; Karamanou, K.; Engin, A.B.; Docea, A.O.; Constantin, C.; Negrei, C.; Nikitovic, D.; Tsatsakis, A. Protein bio-corona: Critical issue in immune nanotoxicology. Arch. Toxicol. 2017, 91, 1031–1048. [Google Scholar] [CrossRef]
- Keshvadi, M.; Karimi, F.; Valizadeh, S.; Valizadeh, A. Comparative study of antibacterial inhibitory effect of silver nanoparticles and garlic oil nanoemulsion with their combination. Biointerface Res. Appl. Chem. 2019, 9, 4560–4566. [Google Scholar]
- Sharma, N.; Phutela, K.; Goel, A.; Soni, S.; Batra, N. Exploring the bacterial based silver nanoparticle for their possible application as disinfectants. Biointerface Res. Appl. Chem. 2018, 8, 3100–3104. [Google Scholar]
- Samoilova, N.A.; Krayukhina, M.A.; Popov, D.A.; Anuchina, N.M.; Piskarev, V.E. 3′-sialyllactose-decorated silver nanoparticles: Lectin binding and bactericidal properties. Biointerface Res. Appl. Chem. 2018, 8, 3095–3099. [Google Scholar]
- Jeyaraj, M.; Sathishkumar, G.; Sivanandhan, G.; MubarakAli, D.; Rajesh, M.; Arun, R.; Kapildev, G.; Manickavasagam, M.; Thajuddin, N.; Premkumar, K.; et al. Biogenic silver nanoparticles for cancer treatment: An experimental report. Colloids Surf. B Biointerfaces 2013, 106, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Choi, J.E.; Choi, J.; Chung, K.H.; Park, K.; Yi, J.; Ryu, D.Y. Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol. In Vitro 2009, 23, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Tang, M. Review of the effects of manufactured nanoparticles on mammalian target organs. J. Appl. Toxicol. 2018, 38, 25–40. [Google Scholar] [CrossRef]
- McShan, D.; Ray, P.C.; Yu, H. Molecular toxicity mechanism of nanosilver. J. Food Drug Anal. 2014, 22, 116–127. [Google Scholar] [CrossRef]
- Sabry, N.M.; Tolba, S.; Abdel-Gawad, F.K.; Bassem, S.M.; Nassar, H.F.; El-Taweel, G.E.; Okasha, A.; Ibrahim, M. Interaction between nano silver and bacteria: Modeling approach. Biointerface Res. Appl. Chem. 2018, 8, 3570–3574. [Google Scholar]
- Sharma, N.; Kumar, J.; Thakur, S.; Sharma, S.; Shrivastava, V. Antibacterial study of silver doped zinc oxide nanoparticles against Staphylococcus aureus and Bacillus subtilis. Drug Invent. Today 2013, 5, 50–54. [Google Scholar] [CrossRef]
- Pal, S.; Tak, Y.K.; Song, J.M. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the Gram-negative bacterium Escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720. [Google Scholar] [CrossRef]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramirez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef]
- Smekalova, M.; Aragon, V.; Panacek, A.; Prucek, R.; Zboril, R.; Kvitek, L. Enhanced antibacterial effect of antibiotics in combination with silver nanoparticles against animal pathogens. Vet. J. 2016, 209, 174–179. [Google Scholar] [CrossRef]
- Calderón-Jiménez, B.; Johnson, M.E.; Montoro Bustos, A.R.; Murphy, K.E.; Winchester, M.R.; Vega Baudrit, J.R. Silver Nanoparticles: Technological Advances, Societal Impacts, and Metrological Challenges. Front. Chem. 2017, 5, 6. [Google Scholar] [CrossRef]
- Barcińska, E.; Wierzbicka, J.; Zauszkiewicz-Pawlak, A.; Jacewicz, D.; Dabrowska, A.; Inkielewicz-Stepniak, I. Role of Oxidative and Nitro-Oxidative Damage in Silver Nanoparticles Cytotoxic Effect against Human Pancreatic Ductal Adenocarcinoma Cells. Oxidative Med. Cell. Longev. 2018, 2018, 8251961. [Google Scholar] [CrossRef]
- Bacchetta, C.; Ale, A.; Simoniello, M.F.; Gervasio, S.; Davico, C.; Rossi, A.S.; Desimone, M.F.; Poletta, G.; López, G.; Monserrat, J.M.; et al. Genotoxicity and oxidative stress in fish after a short-term exposure to silver nanoparticles. Ecol. Indic. 2017, 76, 230–239. [Google Scholar] [CrossRef]
- Caballero-Díaz, E.; Pfeiffer, C.; Kastl, L.; Rivera-Gil, P.; Simonet, B.; Valcárcel, M.; Jiménez-Lamana, J.; Laborda, F.; Parak, W.J. The Toxicity of Silver Nanoparticles Depends on Their Uptake by Cells and Thus on Their Surface Chemistry. Part. Part. Syst. Charact. 2013, 30, 1079–1085. [Google Scholar] [CrossRef]
- Pinzaru, I.; Coricovac, D.; Dehelean, C.; Moaca, E.A.; Mioc, M.; Baderca, F.; Sizemore, I.; Brittle, S.; Marti, D.; Calina, C.D.; et al. Stable PEG-coated silver nanoparticles—A comprehensive toxicological profile. Food Chem. Toxicol. 2018, 111, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Travan, A.; Pelillo, C.; Donati, I.; Marsich, E.; Benincasa, M.; Scarpa, T.; Semeraro, S.; Turco, G.; Gennaro, R.; Paoletti, S. Non-cytotoxic silver nanoparticle-polysaccharide nanocomposites with antimicrobial activity. Biomacromolecules 2009, 10, 1429–1435. [Google Scholar] [CrossRef]
- Pallavicini, P.; Taglietti, A.; Dacarro, G.; Diaz-Fernandez, Y.A.; Galli, M.; Grisoli, P.; Patrini, M.; Santucci de Magistris, G.; Zanoni, R. Self-assembled monolayers of silver nanoparticles firmly grafted on glass surfaces: Low Ag+ release for an efficient antibacterial activity. J. Colloid Interface Sci. 2010, 350, 110–116. [Google Scholar] [CrossRef]
- Sharma, V.K.; Siskova, K.M.; Zboril, R.; Gardea-Torresdey, J.L. Organic-coated silver nanoparticles in biological and environmental conditions: Fate, stability and toxicity. Adv. Colloid Interface Sci. 2014, 204, 15–34. [Google Scholar] [CrossRef]
- Bushueva, T.; Minigalieva, I.; Panov, V.; Kuznetsova, A.; Naumova, A.; Shur, V.; Shishkina, E.; Gurvismalles, C.V.; Privalova, L.; Katsnelson, B. More data on in vitro assessment of comparative and combined toxicity of metal oxide nanoparticles. Food Chem. Toxicol. 2019, 133, 110753. [Google Scholar] [CrossRef]
- Nguyen, K.C.; Richards, L.; Massarsky, A.; Moon, T.W.; Tayabali, A.F. Toxicological evaluation of representative silver nanoparticles in macrophages and epithelial cells. Toxicol. In Vitro 2016, 33, 163–173. [Google Scholar] [CrossRef]
- Bergin, I.L.; Wilding, L.A.; Morishita, M.; Walacavage, K.; Ault, A.P.; Axson, J.L.; Stark, D.I.; Hashway, S.A.; Capracotta, S.S.; Leroueil, P.R.; et al. Effects of particle size and coating on toxicologic parameters, fecal elimination kinetics and tissue distribution of acutely ingested silver nanoparticles in a mouse model. Nanotoxicology 2016, 10, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, A.; Sharma, V. Toxicity assessment of nanomaterials: Methods and challenges. Anal. Bioanal. Chem. 2010, 398, 589–605. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.P.; Hernández-Ruiz, J.; Martinez-Subiela, S.; Tvarijonaviciute, A.; Ceron, J.J. Spectrophotometric assays for total antioxidant capacity (TAC) in dog serum: An update. BMC Vet. Res. 2016, 12, 166. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, A.; Klein, K. Chapter 2—Methods for Measuring Oxidative Stress in the Laboratory. In Antioxidants in Food, Vitamins and Supplements; Dasgupta, A., Klein, K., Eds.; Elsevier: San Diego, CA, USA, 2014; pp. 19–40. [Google Scholar]
- Dalle-Donne, I.; Rossi, R.; Giustarini, D.; Milzani, A.; Colombo, R. Protein carbonyl groups as biomarkers of oxidative stress. Clin. Chim. Acta; Int. J. Clin. Chem. 2003, 329, 23–38. [Google Scholar] [CrossRef]
- Lu, S.C. Regulation of glutathione synthesis. Mol. Asp. Med. 2009, 30, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Messner, D.J.; Murray, K.F.; Kowdley, K.V. Chapter 55—Mechanisms of Hepatocyte Detoxification. In Physiology of the Gastrointestinal Tract, 5th ed.; Johnson, L.R., Ghishan, F.K., Kaunitz, J.D., Merchant, J.L., Said, H.M., Wood, J.D., Eds.; Academic Press: Boston, MA, USA, 2012; pp. 1507–1527. [Google Scholar]
- Kander, M.C.; Cui, Y.; Liu, Z. Gender difference in oxidative stress: A new look at the mechanisms for cardiovascular diseases. J. Cell. Mol. Med. 2017, 21, 1024–1032. [Google Scholar] [CrossRef]
- Díaz, A.; López-Grueso, R.; Gambini, J.; Monleón, D.; Mas-Bargues, C.; Abdelaziz, K.M.; Viña, J.; Borrás, C. Sex Differences in Age-Associated Type 2 Diabetes in Rats-Role of Estrogens and Oxidative Stress. Oxidative Med. Cell. Longev. 2019, 2019, 6734836. [Google Scholar] [CrossRef]
- Barp, J.; Araujo, A.S.; Fernandes, T.R.; Rigatto, K.V.; Llesuy, S.; Bello-Klein, A.; Singal, P. Myocardial antioxidant and oxidative stress changes due to sex hormones. Braz. J. Med Biol. Res. 2002, 35, 1075–1081. [Google Scholar] [CrossRef]
- Xue, Y.; Zhang, S.; Huang, Y.; Zhang, T.; Liu, X.; Hu, Y.; Zhang, Z.; Tang, M. Acute toxic effects and gender-related biokinetics of silver nanoparticles following an intravenous injection in mice. J. Appl. Toxicol. 2012, 32, 890–899. [Google Scholar] [CrossRef]
- Chaloupka, K.; Malam, Y.; Seifalian, A.M. Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol. 2010, 28, 580–588. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Kumar, A.; Ansari, A.; Kim, H.; Shin, H.S. Photo-mediated Biosynthesis of Silver Nanoparticles Using the Non-edible Accrescent Fruiting Calyx of Physalis peruviana L. Fruits and Investigation of its Radical Scavenging Potential and Cytotoxicity Activities. J. Photochem. Photobiol. B Biol. 2018, 188, 116–125. [Google Scholar] [CrossRef]
- Das, G.; Patra, J.K.; Debnath, T.; Ansari, A.; Shin, H.-S. Investigation of antioxidant, antibacterial, antidiabetic, and cytotoxicity potential of silver nanoparticles synthesized using the outer peel extract of Ananas comosus (L.). PLoS ONE 2019, 14, e0220950. [Google Scholar] [CrossRef]
- Patil Shriniwas, P. Antioxidant, antibacterial and cytotoxic potential of silver nanoparticles synthesized using terpenes rich extract of Lantana camara L. leaves. Biochem. Biophys. Rep. 2017, 10, 76–81. [Google Scholar]
- Docea, A.O. Experimental designs and protocols for long-term combined exposure studies from methodology to application: Problems and solutions. Toxicol. Lett. 2019, 314, S17-01. [Google Scholar]
- Tsatsakis, A.M.; Docea, A.O.; Tsitsimpikou, C. New challenges in risk assessment of chemicals when simulating real exposure scenarios; simultaneous multi-chemicals’ low dose exposure. Food Chem. Toxicol. 2016, 96, 174–176. [Google Scholar] [CrossRef] [PubMed]
- Docea, A.O.; Gofita, E.; Goumenou, M.; Calina, D.; Rogoveanu, O.; Varut, M.; Olaru, C.; Kerasioti, E.; Fountoucidou, P.; Taitzoglou, I.; et al. Six months exposure to a real life mixture of 13 chemicals’ below individual NOAELs induced non monotonic sex-dependent biochemical and redox status changes in rats. Food Chem. Toxicol. 2018, 115, 470–481. [Google Scholar] [CrossRef]
- Docea, A.O.; Goumenou, M.; Calina, D.; Arsene, A.L.; Dragoi, C.M.; Gofita, E.; Pisoschi, C.G.; Zlatian, O.; Stivaktakis, P.D.; Nikolouzakis, T.K.; et al. Adverse and hormetic effects in rats exposed for 12 months to low dose mixture of 13 chemicals: RLRS part III. Toxicol. Lett. 2019, 310, 70–91. [Google Scholar] [CrossRef]
- Tsatsakis, A.; Docea, A.O.; Constantin, C.; Calina, D.; Zlatian, O.; Nikolouzakis, T.K.; Stivaktakis, P.D.; Kalogeraki, A.; Liesivuori, J.; Tzanakakis, G.; et al. Genotoxic, cytotoxic, and cytopathological effects in rats exposed for 18 months to a mixture of 13 chemicals in doses below NOAEL levels. Toxicol. Lett. 2019, 316, 154–170. [Google Scholar] [CrossRef]
- Patlolla, A.; Hackett, D.; Tchounwou, P. Silver nanoparticle-induced oxidative stress-dependent toxicity in Sprague-Dawley rats. Mol. Cell. Biochem. 2014, 399, 257–268. [Google Scholar] [CrossRef]
- Foldbjerg, R.; Olesen, P.; Hougaard, M.; Dang, D.; Hoffmann, H.; Autrup, H. PVP-coated silver nanoparticles and silver ions induce reactive oxygen species, apoptosis and necrosis in THP-1 monocytes. Toxicol. Lett. 2009, 190, 156–162. [Google Scholar] [CrossRef]
- Carlson, C.; Hussain, S.M.; Schrand, A.M.; Braydich-Stolle, L.K.; Hess, K.L.; Jones, R.L.; Schlager, J.J. Unique cellular interaction of silver nanoparticles: Size-dependent generation of reactive oxygen species. J. Phys. Chem. B 2008, 112, 13608–13619. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Piccapietra, F.; Wagner, B.; Marconi, F.; Kaegi, R.; Odzak, N.; Sigg, L.; Behra, R. Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ. Sci. Technol. 2008, 42, 8959–8964. [Google Scholar] [CrossRef] [PubMed]
- Akter, M.; Sikder, M.T.; Rahman, M.M.; Ullah, A.K.M.A.; Hossain, K.F.B.; Banik, S.; Hosokawa, T.; Saito, T.; Kurasaki, M. A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J. Adv. Res. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sakulkhu, U.; Mahmoudi, M.; Maurizi, L.; Salaklang, J.; Hofmann, H. Protein corona composition of superparamagnetic iron oxide nanoparticles with various physico-chemical properties and coatings. Sci. Rep. 2014, 4, 5020. [Google Scholar] [CrossRef]
- Fountoucidou, P.; Veskoukis, A.S.; Kerasioti, E.; Docea, A.O.; Taitzoglou, I.A.; Liesivuori, J.; Tsatsakis, A.; Kouretas, D. A mixture of routinely encountered xenobiotics induces both redox adaptations and perturbations in blood and tissues of rats after a long-term low-dose exposure regimen: The time and dose issue. Toxicol. Lett. 2019, 317, 24–44. [Google Scholar] [CrossRef]
- Padureanu, R.; Albu, C.V.; Mititelu, R.R.; Bacanoiu, M.V.; Docea, A.O.; Calina, D.; Padureanu, V.; Olaru, G.; Sandu, R.E.; Malin, R.D.; et al. Oxidative Stress and Inflammation Interdependence in Multiple Sclerosis. J. Clin. Med. 2019, 8, 1815. [Google Scholar] [CrossRef]
- Iordache, A.M.; Docea, A.O.; Buga, A.M.; Zlatian, O.; Ciurea, M.E.; Rogoveanu, O.C.; Burada, F.; Sosoi, S.; Mitrut, R.; Mamoulakis, C.; et al. Sildenafil and tadalafil reduce the risk of contrast-induced nephropathy by modulating the oxidant/antioxidant balance in a murine model. Food Chem. Toxicol. 2019, 135, 111038. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Parameter | Males | Females | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | ||
| TAC (mmol DPPH/L) | Average | 0.2422 | 0.4993 * | 0.3758 * | 0.2561 | 0.2316 | 0.5161 * | 0.5450 * | 0.2584 * |
| SD | 0.0138 | 0.492 | 0.0492 | 0.0157 | 0.0159 | 0.0091 | 0.0140 | 0.0060 | |
| % to control | 106.2% | 55.2% | 5.7% | 122.8% | 135.3% | 11.6% | |||
| GSH (µmol/g Hb) | Average | 1.9408 | 1.8092 | 1.9901 | 2.0559 | 2.2039 | 2.4178* | 1.9342* | 2.2270 |
| SD | 0.1628 | 0.0601 | 0.0349 | 0.0349 | 0.0233 | 0.0814 | 0.0221 | 0.1506 | |
| % to control | −6.8% | 2.5% | 5.9% | 9.7% | −12.2% | 1.1% | |||
| CAT (U/g Hb) | Average | 177.38 | 168.5714 | 185.02 | 192.96 | 171.03 | 171.10 | 177.78 | 188.27* |
| SD | 2.38 | 28.9076 | 2.175 | 1.61 | 0.56 | 2.51 | 1.56 | 11.25 | |
| % to control | −5.0% | 4.3% | 8.8% | 0.1% | 3.9% | 10.1% | |||
| Parameter | Males | Females | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | ||
| TAC (mmol DPPH/L) | Average | 0.2422 | 0.3472 * | 0.2545 | 0.2038 * | 0.2316 | 0.4009 * | 0.2171 | 0.2450 |
| SD | 0.0138 | 0.0016 | 0.0138 | 0.0063 | 0.0159 | 0.0315 | 0.0031 | 0.0039 | |
| % to control | 43.4% | −5.1% | −15.9% | 73.1% | −6.3% | 5.8% | |||
| GSH (µmol/g Hb) | Average | 1.9408 | 2.2862 | 2.1875 | 0.8224 * | 2.2039 | 2.1382 | 2.0066 * | 1.5461 * |
| SD | 0.1628 | 0.1977 | 0.1047 | 0.3489 | 0.0233 | 0.1163 | 0.0930 | 0.0698 | |
| % to control | 17.8% | 12.7% | −57.6% | −3.0% | −9.0% | −29.9% | |||
| CAT (U/g Hb) | Average | 177.38 | 169.84 | 216.47* | 181.84 | 171.03 | 199.01* | 188.09 | 179.66 |
| SD | 2.38 | 5.61 | 15.43 | 2.59 | 0.56 | 14.73 | 5.33 | 13.96 | |
| % to control | −4.3% | 22.0% | 2.5% | 16.4% | 10.0% | 5.1% | |||
| Parameter | Males | Females | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | ||
| TBARS (µmol/L) | Average | 0.6290 | 1.1174 * | 0.5323 | 0.4903 | 0.7032 | 0.7118 | 0.4361 * | 0.4619 * |
| SD | 0.0798 | 0.3934 | 0.0205 | 0.0274 | 0.0274 | 0.0249 | 0.0202 | 0.0195 | |
| % to control | 77.7% | −15.4% | −22.1% | 1.2% | −38.0% | −34.3% | |||
| PROTC (nmol/mg protein) | Average | 0.9008 | 1.0297 | 0.9450 | 0.8287 | 1.0792 | 1.7134* | 0.7076 | 0.5934* |
| SD | 0.1029 | 0.0649 | 0.1090 | 0.1711 | 0.0611 | 0.7115 | 0.0257 | 0.1499 | |
| % to control | 14.3% | 4.9% | −8.0% | 58.8% | −34.4% | −45.0% | |||
| Parameter | Males | Females | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | Control | 1 mg/kgbw | 2 mg/kgbw | 4 mg/kgbw | ||
| TBARS (µmol/L) | Average | 0.6290 | 0.6710 | 0.4968* | 0.5194* | 0.7032 | 0.4677* | 0.5065* | 0.8129 |
| SD | 0.0798 | 0.0502 | 0.0137 | 0.0068 | 0.0274 | 0.0205 | 0.0205 | 0.1962 | |
| % to control | −6.7% | −21.0% | −17.4% | −33.5% | −28.0% | 15.6% | |||
| PROTC (nmol/mg protein) | Average | 0.9008 | 0.6718 | 2.1341* | 3.8725* | 1.0792 | 0.7884 | 2.5592* | 2.5274* |
| SD | 0.1029 | 0.0516 | 0.8640 | 0.1741 | 0.0611 | 0.0320 | 0.0994 | 0.4932 | |
| % to control | −25.4% | −136.9% | 330.0% | −27.0% | 137.1% | 134.2% | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Docea, A.O.; Calina, D.; Buga, A.M.; Zlatian, O.; Paoliello, M.M.B.; Mogosanu, G.D.; Streba, C.T.; Popescu, E.L.; Stoica, A.E.; Bîrcă, A.C.; et al. The Effect of Silver Nanoparticles on Antioxidant/Pro-Oxidant Balance in a Murine Model. Int. J. Mol. Sci. 2020, 21, 1233. https://doi.org/10.3390/ijms21041233
Docea AO, Calina D, Buga AM, Zlatian O, Paoliello MMB, Mogosanu GD, Streba CT, Popescu EL, Stoica AE, Bîrcă AC, et al. The Effect of Silver Nanoparticles on Antioxidant/Pro-Oxidant Balance in a Murine Model. International Journal of Molecular Sciences. 2020; 21(4):1233. https://doi.org/10.3390/ijms21041233
Chicago/Turabian StyleDocea, Anca Oana, Daniela Calina, Ana Maria Buga, Ovidiu Zlatian, M.M.B. Paoliello, George Dan Mogosanu, Costin Teodor Streba, Elena Leocadia Popescu, Alexandra Elena Stoica, Alexandra Catalina Bîrcă, and et al. 2020. "The Effect of Silver Nanoparticles on Antioxidant/Pro-Oxidant Balance in a Murine Model" International Journal of Molecular Sciences 21, no. 4: 1233. https://doi.org/10.3390/ijms21041233
APA StyleDocea, A. O., Calina, D., Buga, A. M., Zlatian, O., Paoliello, M. M. B., Mogosanu, G. D., Streba, C. T., Popescu, E. L., Stoica, A. E., Bîrcă, A. C., Vasile, B. Ș., Grumezescu, A. M., & Mogoanta, L. (2020). The Effect of Silver Nanoparticles on Antioxidant/Pro-Oxidant Balance in a Murine Model. International Journal of Molecular Sciences, 21(4), 1233. https://doi.org/10.3390/ijms21041233











