Microbial Degradation of Plastic in Aqueous Solutions Demonstrated by CO2 Evolution and Quantification
Abstract
1. Introduction
2. Results
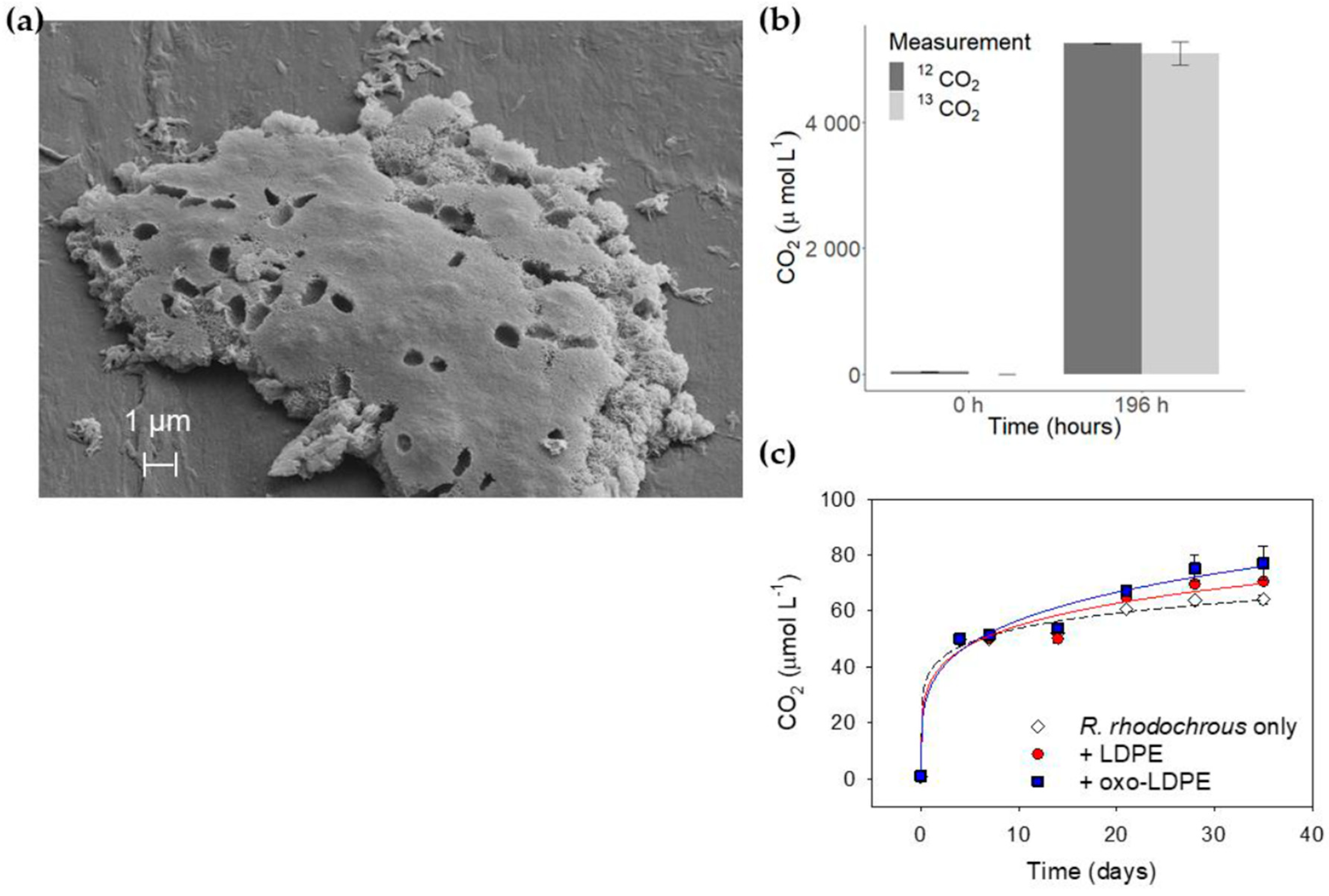
2.1. Assessment of Microbial Growth
2.2. Measurement of Growth by Monitoring CO2 Production
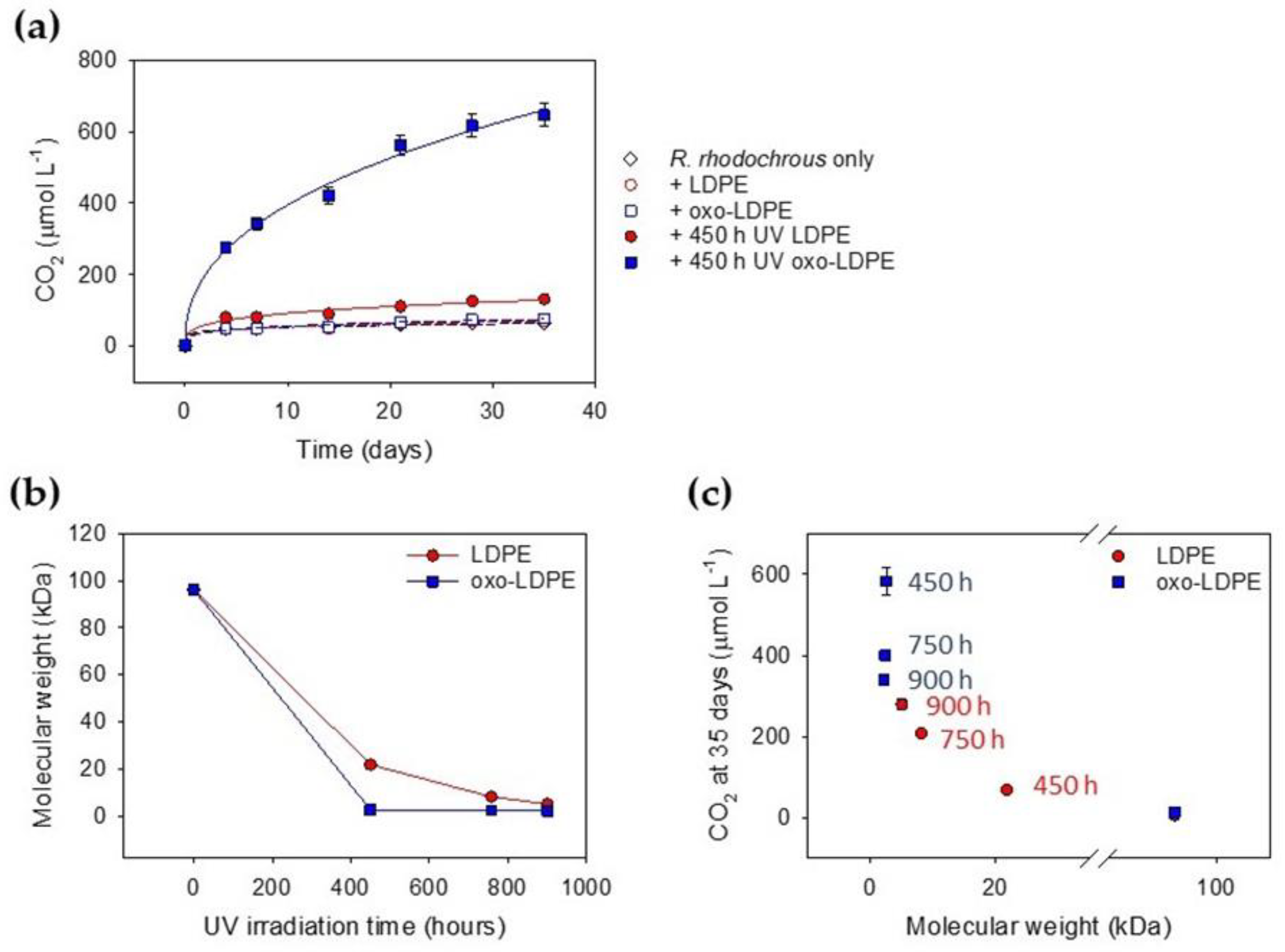
2.3. Effect of UV Aging on Biodegradation
2.4. Influence of Molecular Weight on Biodegradation
2.5. Starch-Based Plastic
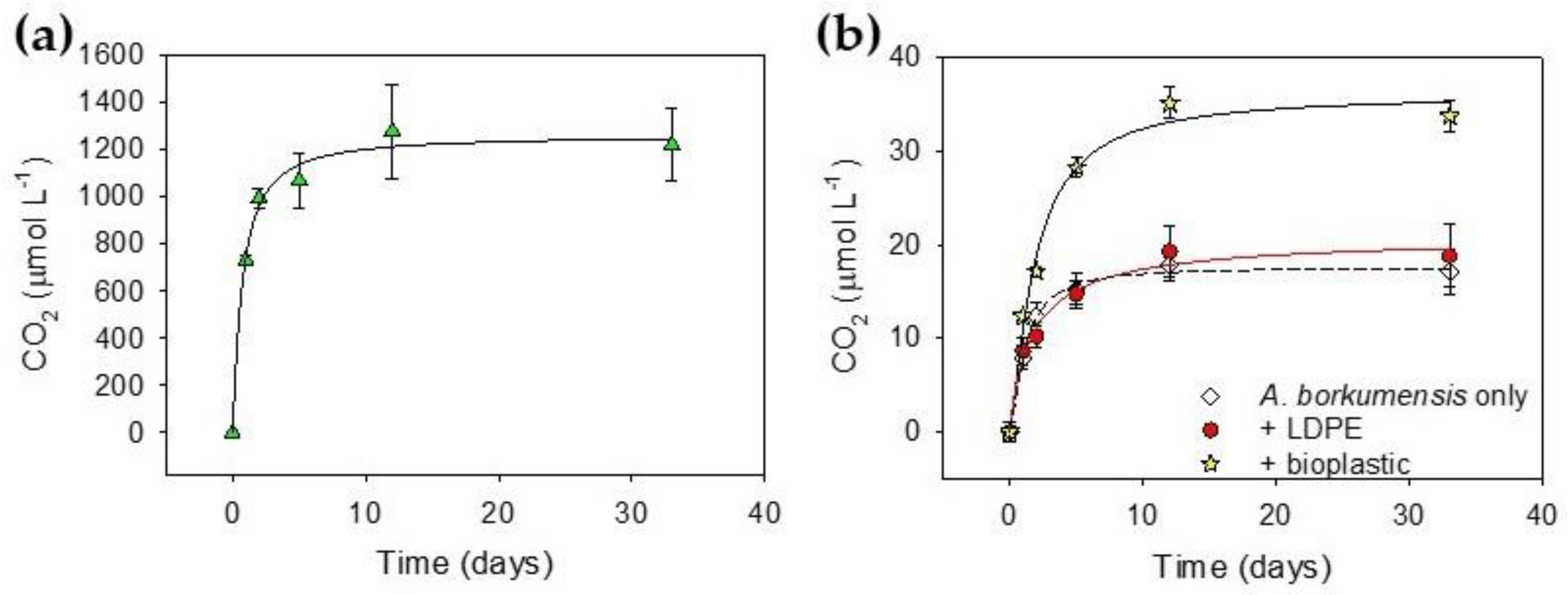
2.6. Biodegradation in Water
2.7. Monitoring of A. Borkumensis Growth on Plastics
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Plastic Preparation
4.3. Preparation of Bacteria on Minimal Media Plates
4.4. Scanning Electron Microscopy
4.5. Preparation of Bacteria for the Plastic CO2 Bioassay
4.6. CO2 Analysis
4.7. 13C Glucose Analysis
4.8. Data Fitting
4.9. Gel Permeation Chromatography
4.10. ATP Assay
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GC | Gas chromatography |
| LDPE | Low-density polyethylene |
| SEM | Scanning electron microscopy |
References
- Isobe, A.; Iwasaki, S.; Uchida, K.; Tokai, T. Abundance of non-conservative microplastics in the upper ocean from 1957 to 2066. Nat. Commun. 2019, 10, 417. [Google Scholar] [CrossRef] [PubMed]
- O’Brine, T.; Thompson, R.C. Degradation of plastic carrier bags in the marine environment. Mar. Pollut. Bull. 2010, 60, 2279–2283. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.; Lebreton, L.C.M.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Galgani, F.; Ryan, P.G.; Reisser, J. Plastic Pollution in the World’s Oceans: More than 5 Trillion Plastic Pieces Weighing over 250,000 Tons Afloat at Sea. PLoS ONE 2014, 9, e111913. [Google Scholar] [CrossRef] [PubMed]
- Waters, C.N.; Zalasiewicz, J.; Summerhayes, C.; Barnosky, A.D.; Poirier, C.; Gałuszka, A.; Cearreta, A.; Edgeworth, M.; Ellis, E.C.; Ellis, M.; et al. The Anthropocene is functionally and stratigraphically distinct from the Holocene. Science 2016, 351, aad2622. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef]
- Haward, M. Plastic pollution of the world’s seas and oceans as a contemporary challenge in ocean governance. Nat. Commun. 2018, 9, 667. [Google Scholar] [CrossRef]
- Romero-Azogil, L.; Benito, E.; Martínez de Ilarduya, A.; García-Martín, M.G.; Galbis, J.A. Hydrolytic degradation of D-mannitol-based polyurethanes. Polym. Degrad. Stab. 2018, 153, 262–271. [Google Scholar] [CrossRef]
- Hermann, B.G.; Debeer, L.; de Wilde, B.; Blok, K.; Patel, M.K. To compost or not to compost: Carbon and energy footprints of biodegradable materials’ waste treatment. Polym. Degrad. Stab. 2011, 96, 1159–1171. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Vivekanandhan, S.; Pin, J.M.; Misra, M. Composites from renewable and sustainable resources: Challenges and innovations. Science 2018, 362, 536–542. [Google Scholar] [CrossRef]
- Muniyasamy, S.; Anstey, A.; Reddy, M.M.; Misra, M.; Mohanty, A. Biodegradability and compostability of lignocellulosic based composite materials. J. Renew. Mater. 2013, 1, 253–272. [Google Scholar] [CrossRef]
- Chen, G.Q.; Patel, M.K. Plastics derived from biological sources: Present and future: A technical and environmental review. Chem. Rev. 2012, 112, 2082–2099. [Google Scholar] [CrossRef] [PubMed]
- Ammala, A.; Bateman, S.; Dean, K.; Petinakis, E.; Sangwan, P.; Wong, S.; Yuan, Q.; Yu, L.; Patrick, C.; Leong, K.H. An overview of degradable and biodegradable polyolefins. Prog. Polym. Sci. 2011, 36, 1015–1049. [Google Scholar] [CrossRef]
- Hann, S.; Ettlinger, S.; Gibbs, A. The Impact of the Use of “Oxo-Degradable” Plastic on the Environment; Publications Office of the European Union: Luxembourg, 2016.
- Harrison, J.P.; Boardman, C.; O’Callaghan, K.; Delort, A.M.; Song, J. Biodegradability standards for carrier bags and plastic films in aquatic environments: A critical review. R. Soc. Open Sci. 2018, 5, 171792. [Google Scholar] [CrossRef] [PubMed]
- Kumar Sen, S.; Raut, S. Microbial degradation of low density polyethylene (LDPE): A review. J. Environ. Chem. Eng. 2015, 3, 462–473. [Google Scholar] [CrossRef]
- Shah, A.A.; Hasan, F.; Hameed, A.; Ahmed, S. Biological degradation of plastics: A comprehensive review. Biotechnol. Adv. 2008, 26, 246–265. [Google Scholar] [CrossRef] [PubMed]
- Castro-Aguirre, E.; Auras, R.; Selke, S.; Rubino, M.; Marsh, T. Insights on the aerobic biodegradation of polymers by analysis of evolved carbon dioxide in simulated composting conditions. Polym. Degrad. Stab. 2017, 137, 251–271. [Google Scholar] [CrossRef]
- Neher, D.A.; Weicht, T.R.; Bates, S.T.; Leff, J.W.; Fierer, N. Changes in bacterial and fungal communities across compost recipes, preparation methods, and composting times. PLoS ONE 2013, 8, e79512. [Google Scholar] [CrossRef]
- Dussud, C.; Meistertzheim, A.L.; Conan, P.; Pujo-Pay, M.; George, M.; Fabre, P.; Coudane, J.; Higgs, P.; Elineau, A.; Pedrotti, M.L.; et al. Evidence of niche partitioning among bacteria living on plastics, organic particles and surrounding seawaters. Environ. Pollut. 2018, 236, 807–816. [Google Scholar] [CrossRef]
- Fontanella, S.; Bonhomme, S.; Koutny, M.; Husarova, L.; Brusson, J.-M.; Courdavault, J.-P.; Pitteri, S.; Samuel, G.; Pichon, G.; Lemaire, J.; et al. Comparison of the biodegradability of various polyethylene films containing pro-oxidant additives. Polym. Degrad. Stab. 2010, 95, 1011–1021. [Google Scholar] [CrossRef]
- Napper, I.E.; Thompson, R.C. Environmental Deterioration of Biodegradable, Oxo-biodegradable, Compostable, and Conventional Plastic Carrier Bags in the Sea, Soil, and Open-Air over a 3-Year Period. Environ. Sci. Technol. 2019, 53, 4775–4783. [Google Scholar] [CrossRef]
- Beckloff, N.; Laube, D.; Castro, T.; Furgang, D.; Park, S.; Perlin, D.; Clements, D.; Tang, H.; Scott, R.W.; Tew, G.N.; et al. Activity of an antimicrobial peptide mimetic against planktonic and biofilm cultures of oral pathogens. Antimicrob. Agents Chemother. 2007, 51, 4125–4132. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yang, Y.; Wu, W.-M.; Zhao, J.; Jiang, L. Evidence of polyethylene biodegradation by bacterial strains from the guts of plastic-eating waxworms. Environ. Sci. Technol. 2014, 48, 13776–13784. [Google Scholar] [CrossRef] [PubMed]
- Sivan, A.; Szanto, M.; Pavlov, V. Biofilm development of the polyethylene-degrading bacterium Rhodococcus ruber. Appl. Microbiol. Biotechnol. 2006, 72, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Gilan, I.; Hadar, Y.; Sivan, A. Colonization, biofilm formation and biodegradation of polyethylene by a strain of Rhodococcus ruber. Appl. Microbiol. Biotechnol. 2004, 65, 97–104. [Google Scholar]
- Bonhomme, S.; Cuer, A.; Delort, A.M.; Lemaire, J.; Sancelme, M.; Scott, G. Environmental biodegradation of polyethylene. Polym. Degrad. Stab. 2003, 81, 441–452. [Google Scholar] [CrossRef]
- Sabirova, J.S.; Becker, A.; Lünsdorf, H.; Nicaud, J.M.; Timmis, K.N.; Golyshin, P.N. Transcriptional profiling of the marine oil-degrading bacterium Alcanivorax borkumensis during growth on n-alkanes. Fems Microbiol. Lett. 2011, 319, 160–168. [Google Scholar] [CrossRef]
- Abbasi, A.; Bothun, G.D.; Bose, A. Attachment of Alcanivorax borkumensis to Hexadecane-In-Artificial Sea Water Emulsion Droplets. Langmuir 2018, 34, 5352–5357. [Google Scholar] [CrossRef]
- Ali, G.S.; Reddy, A.S.N. Inhibition of fungal and bacterial plant pathogens by synthetic peptides: In vitro growth inhibition, interaction between peptides and inhibition of disease progression. Mol. Plant Microbe Interact. 2000, 13, 847–859. [Google Scholar] [CrossRef]
- Jacks, S.; Giguère, S.; Crawford, P.C.; Castleman, W.L. Experimental infection of neonatal foals with Rhodococcus equi triggers adult-like gamma interferon induction. Clin. Vaccine Immunol. 2007, 14, 669–677. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and importance of microplastics in the marine environmentA review of the sources, fate, effects, and potential solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef]
- Lundin, A. Bioluminescence and Chemiluminescence Part C; Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2000; Volume 305, ISBN 9780121822064. [Google Scholar]
- Lundin, A. Optimization of the firefly luciferase reaction for analytical purposes. Adv. Biochem. Eng. Biotechnol. 2014, 145, 31–62. [Google Scholar] [PubMed]
- Pepper, I.L.; Gerba, C.P.; Gentry, T.J. Environmental Microbiology; Elservier: London, UK, 2015. [Google Scholar]
- Zamboni, N.; Fendt, S.-M.; Ruhl, M.; Sauer, U. 13C-based metabolic flux analysis. Nat. Protoc. 2009, 4, 878–892. [Google Scholar] [CrossRef]
- Zumstein, M.T.; Schintlmeister, A.; Nelson, T.F.; Baumgartner, R.; Woebken, D.; Wagner, M.; Kohler, H.P.E.; McNeill, K.; Sander, M. Biodegradation of synthetic polymers in soils: Tracking carbon into CO2 and microbial biomass. Sci. Adv. 2018, 4, 9024–9032. [Google Scholar] [CrossRef]
- Flickinger, M.C.; Panikov, N.S. Kinetics, Microbial Growth. In Encyclopedia of Industrial Biotechnology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010. [Google Scholar]
- Cajal-Medrano, R.; Maske, H. Growth efficiency, growth rate and the remineralization of organic substrate by bacterioplankton—Revisiting the Pirt model. Aquat. Microb. Ecol. 1999, 19, 119–128. [Google Scholar] [CrossRef]
- Pickett, J.E.; Gibson, D.A.; Gardner, M.M. Effects of irradiation conditions on the weathering of engineering thermoplastics. Polym. Degrad. Stab. 2008, 93, 1597–1606. [Google Scholar] [CrossRef]
- Erlandsson, B.; Albertsson, A.C.; Karlsson, S. Molecular weight determination in degraded oxidizable and hydrolyzable polymers giving deviation from accurate using calibration and the Mark-Houwink-Sakaruda (MHS) equation. Polym. Degrad. Stab. 1997, 57, 15–23. [Google Scholar] [CrossRef]
- Eyheraguibel, B.; Traikia, M.; Fontanella, S.; Sancelme, M.; Bonhomme, S.; Fromageot, D.; Lemaire, J.; Lauranson, G.; Lacoste, J.; Delort, A.M. Characterization of oxidized oligomers from polyethylene films by mass spectrometry and NMR spectroscopy before and after biodegradation by a Rhodococcus rhodochrous strain. Chemosphere 2017, 184, 366–374. [Google Scholar] [CrossRef]
- Chiellini, E.; Corti, A.; D’Antone, S. Oxo-biodegradable full carbon backbone polymers—Biodegradation behaviour of thermally oxidized polyethylene in an aqueous medium. Polym. Degrad. Stab. 2007, 92, 1378–1383. [Google Scholar] [CrossRef]
- Kawai, F.; Watanabe, M.; Shibata, M.; Yokoyama, S.; Sudate, Y.; Hayashi, S. Comparative study on biodegradability of polyethylene wax by bacteria and fungi. Polym. Degrad. Stab. 2004, 86, 105–114. [Google Scholar] [CrossRef]
- Wróblewska-Krepsztul, J.; Rydzkowski, T.; Borowski, G.; Szczypiński, M.; Klepka, T.; Thakur, V.K. Recent progress in biodegradable polymers and nanocomposite-based packaging materials for sustainable environment. Int. J. Polym. Anal. Charact. 2018, 23, 383–395. [Google Scholar] [CrossRef]
- Vaverková, M.; Adamcová, D.; Kotovicová, J.; Toman, F. Evaluation of biodegradability of plastics bags in composting conditions. Ecol. Chem. Eng. S 2014, 21, 45–57. [Google Scholar] [CrossRef]
- Lv, Y.; Huang, Y.; Yang, J.; Kong, M.; Yang, H.; Zhao, J.; Li, G. Outdoor and accelerated laboratory weathering of polypropylene: A comparison and correlation study. Polym. Degrad. Stab. 2015, 112, 145–159. [Google Scholar] [CrossRef]
- Santos, R.M.; Botelho, G.L.; Machado, A.V. Artificial and natural weathering of ABS. J. Appl. Polym. Sci. 2010, 116, 2005–2014. [Google Scholar] [CrossRef]
- Patrauchan, M.; Sarkisova, S.; Sauer, K.; Franklin, M.J. Calcium influences cellular and extracellular product formation during biofilm-associated growth of a marine Pseudoalteromonas sp. Microbiology 2005, 151, 2885–2897. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Huang, P.; Huang, C.; Sun, P.; Monroy, G.L.; Wu, W.; Lin, J.; Espinosa-Marzal, R.M.; Boppart, S.A.; Liu, W.T.; et al. Effect of divalent ions and a polyphosphate on composition, structure, and stiffness of simulated drinking water biofilms. Biofilms Microbiomes 2018, 4, 15. [Google Scholar] [CrossRef]
- Jacquin, J.; Cheng, J.; Odobel, C.; Pandin, C.; Conan, P.; Pujo-Pay, M.; Barbe, V.; Meistertzheim, A.L.; Ghiglione, J.F. Microbial ecotoxicology of marine plastic debris: A review on colonization and biodegradation by the “plastisphere”. Front. Microbiol. 2019, 10, 865. [Google Scholar] [CrossRef]
- Wang, W.; Shao, Z. The long-chain alkane metabolism network of Alcanivorax dieselolei. Nat. Commun. 2014, 5, 5755. [Google Scholar] [CrossRef]
- Delacuvellerie, V.; Cyriaque, S.; Gobert, S.; Benali, S.; Wattiez, R. The plastisphere in marine ecosystem hosts potential specific microbial degraders including Alcanivorax borkumensis as a key player for the low-density polyethylene degradation. J. Hazard. Mater. 2019, 380, 120899. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rose, R.-S.; Richardson, K.H.; Latvanen, E.J.; Hanson, C.A.; Resmini, M.; Sanders, I.A. Microbial Degradation of Plastic in Aqueous Solutions Demonstrated by CO2 Evolution and Quantification. Int. J. Mol. Sci. 2020, 21, 1176. https://doi.org/10.3390/ijms21041176
Rose R-S, Richardson KH, Latvanen EJ, Hanson CA, Resmini M, Sanders IA. Microbial Degradation of Plastic in Aqueous Solutions Demonstrated by CO2 Evolution and Quantification. International Journal of Molecular Sciences. 2020; 21(4):1176. https://doi.org/10.3390/ijms21041176
Chicago/Turabian StyleRose, Ruth-Sarah, Katherine H. Richardson, Elmeri Johannes Latvanen, China A. Hanson, Marina Resmini, and Ian A. Sanders. 2020. "Microbial Degradation of Plastic in Aqueous Solutions Demonstrated by CO2 Evolution and Quantification" International Journal of Molecular Sciences 21, no. 4: 1176. https://doi.org/10.3390/ijms21041176
APA StyleRose, R.-S., Richardson, K. H., Latvanen, E. J., Hanson, C. A., Resmini, M., & Sanders, I. A. (2020). Microbial Degradation of Plastic in Aqueous Solutions Demonstrated by CO2 Evolution and Quantification. International Journal of Molecular Sciences, 21(4), 1176. https://doi.org/10.3390/ijms21041176





