Stratifying Intraductal Papillary Mucinous Neoplasms by Cyst Fluid Analysis: Present and Future
Abstract
1. Introduction
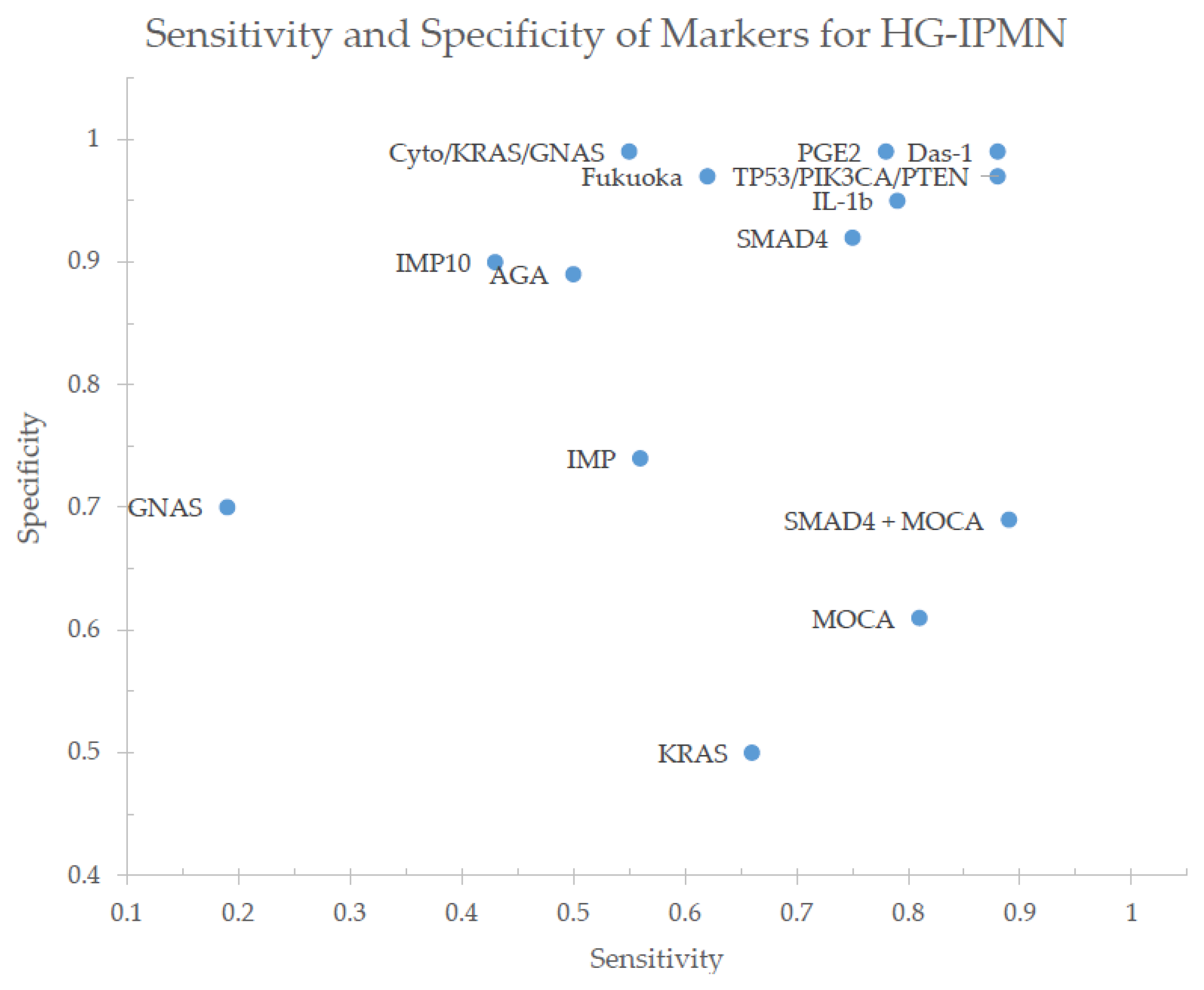
2. Single Marker Analyses
2.1. Mucins
2.2. IL-1b
2.3. PGE2
2.4. Telomere Fusion Status
2.5. miR-216a
2.6. CEP and mAb Das-1
3. Panel Analyses
3.1. Mutational Analysis: KRAS, GNAS, and Cytopathology
3.2. Next-Generation Sequencing Panel: KRAS, GNAS, TP53, PIK3CA, and PTEN
3.3. Whole Exome Sequencing: SMAD4, RNF43, Chromosomal Aneuploidy, and TP53
3.4. Proteomic Array: MMP9, CA72-4, sFASL, and IL-4
3.5. Biosignatures
4. Nomograms and Algorithms: Combining Clinical Features and Biomarker Analyses
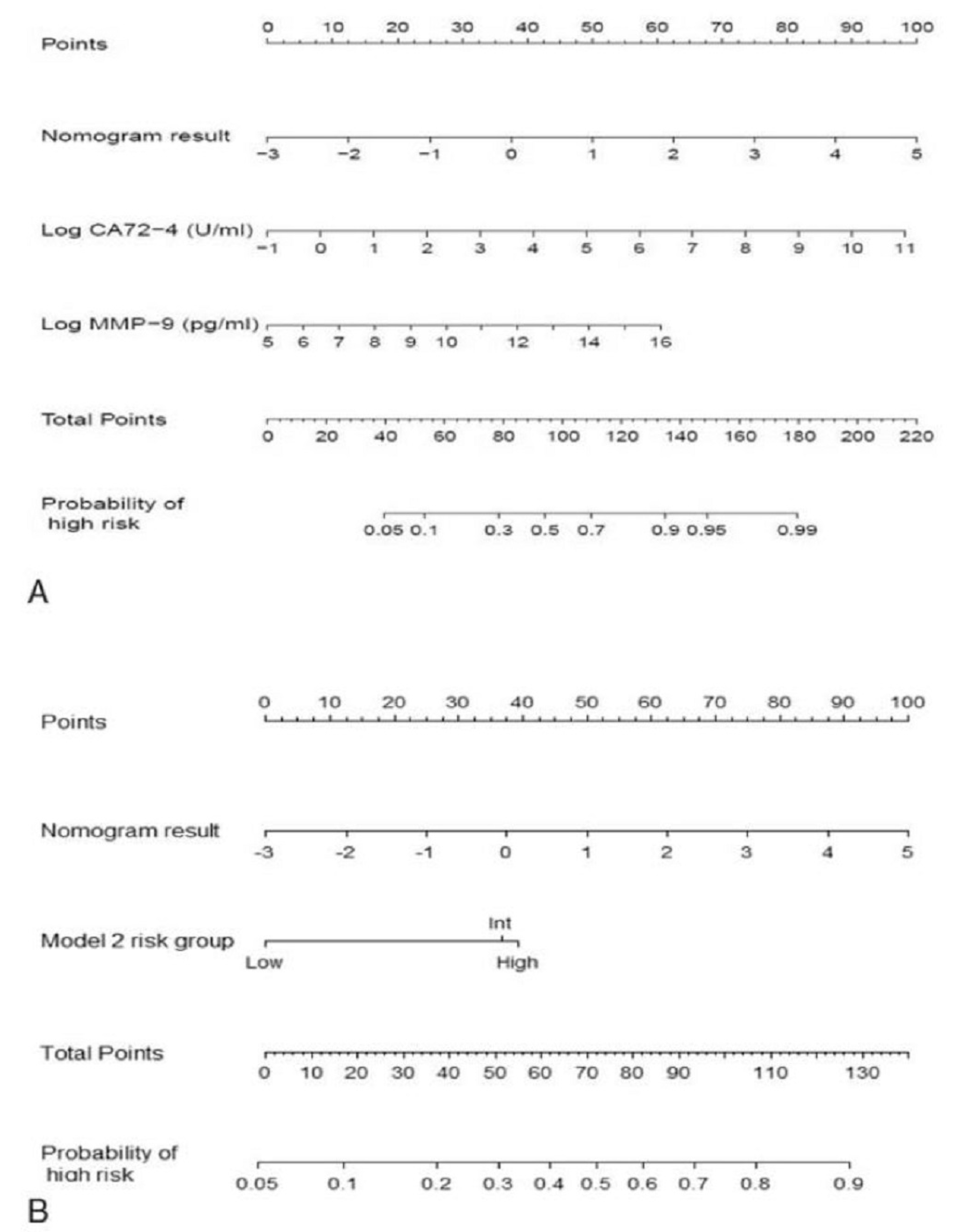
4.1. Nomogram
4.2. MOCA Algorithm
4.3. IMP Testing
5. Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| IPMN | Intraductal papillary mucinous neoplasm |
| CT | Computed tomography |
| MRI | Magnetic resonance imaging |
| MRCP | Magnetic resonance cholangiopancreatography |
| PDAC | Pancreatic ductal adenocarcinoma |
| ICG | International consensus guidelines |
| MD-IPMN | Main duct IPMN |
| BD-IPMN | Branch duct IPMN |
| MPD | Main pancreatic duct |
| AGA | American Gastroenterological Association |
| EUS | Endoscopic ultrasound |
| FNA | Fine needle aspiration |
| HGD | High-grade dysplasia |
| CEA | Carcinoembryonic antigen |
| SCA | Serous cystadenoma |
| SPN | Solid pseudo-papillary neoplasm |
| MCN | Mucinous cystic neoplasm |
| GNAS | Guanine nucleotide binding protein, alpha-stimulating |
| KRAS | Kirsten rat sarcoma viral oncogene homolog |
| NGAL | neutrophil gelatinase-associated lipocalin |
| MUC | Mucin |
| IL | Interleukin |
| PGE2 | Prostaglandin E2 |
| ELISA | Enzyme-linked immunosorbent assay |
| NSAID | Nonsteroidal anti-inflammatory drug |
| LGD | Low-grade dysplasia |
| miRNA | MicroRNA |
| NGS | Next Generation Sequencing |
| mAb | Monoclonal antibody |
| CEP | Colon epithelial protein |
| PCR | Polymerase chain reaction |
| LOH | Loss of heterozygosity |
| MOCA | Multivariate organization of combinatorial alterations |
| IMP | Integrated molecular pathology |
| PANCAN | Pancreatic Cancer Action Network |
| NCI | National Cancer Institute |
References
- Laffan, T.A.; Horton, K.M.; Klein, A.P.; Berlanstein, B.; Siegelman, S.S.; Kawamoto, S.; Johnson, P.T.; Fishman, E.K.; Hruban, R.H. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am. J. Roentgenol. 2008, 191, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Spinelli, K.S.; Fromwiller, T.E.; Daniel, R.A.; Kiely, J.M.; Nakeeb, A.; Komorowski, R.A.; Wilson, S.D.; Pitt, H.A. Cystic pancreatic neoplasms: Observe or operate. Ann. Surg. 2004, 239, 651–657; discussion 657–659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.M.; Mitchell, D.G.; Dohke, M.; Holland, G.A.; Parker, L. Pancreatic cysts: Depiction on single-shot fast spin-echo MR images. Radiology 2002, 223, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Girometti, R.; Intini, S.G.; Cereser, L.; Bazzocchi, M.; Como, G.; Del Pin, M.; Baccarani, U.; Toniutto, P.; Zuiani, C. Incidental pancreatic cysts: A frequent finding in liver-transplanted patients as assessed by 3D T2-weighted turbo spin echo magnetic resonance cholangiopancreatography. JOP 2009, 10, 507–514. [Google Scholar] [PubMed]
- Choi, S.H.; Park, S.H.; Kim, K.W.; Lee, J.Y.; Lee, S.S. Progression of unresected intraductal papillary mucinous neoplasms of the pancreas to cancer: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2017, 15, 1509–1520.e4. [Google Scholar] [CrossRef]
- Chari, S.T.; Yadav, D.; Smyrk, T.C.; DiMagno, E.P.; Miller, L.J.; Raimondo, M.; Clain, J.E.; Norton, I.A.; Pearson, R.K.; Petersen, B.T.; et al. Study of recurrence after surgical resection of intraductal papillary mucinous neoplasm of the pancreas. Gastroenterology 2002, 123, 1500–1507. [Google Scholar] [CrossRef] [PubMed]
- Valsangkar, N.P.; Morales-Oyarvide, V.; Thayer, S.P.; Ferrone, C.R.; Wargo, J.A.; Warshaw, A.L.; Fernández-del Castillo, C. 851 resected cystic tumors of the pancreas: A 33-year experience at the Massachusetts General Hospital. Surgery 2012, 152, S4–S12. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Winter, J.M.; Cameron, J.L.; Lillemoe, K.D.; Campbell, K.A.; Chang, D.; Riall, T.S.; Coleman, J.; Sauter, P.K.; Canto, M.; Hruban, R.H.; et al. Periampullary and pancreatic incidentaloma: A single institution’s experience with an increasingly common diagnosis. Ann. Surg. 2006, 243, 673–680; discussion 680–683. [Google Scholar] [CrossRef]
- Winter, J.M.; Brennan, M.F.; Tang, L.H.; D’Angelica, M.I.; Dematteo, R.P.; Fong, Y.; Klimstra, D.S.; Jarnagin, W.R.; Allen, P.J. Survival after resection of pancreatic adenocarcinoma: Results from a single institution over three decades. Ann. Surg. Oncol. 2012, 19, 169–175. [Google Scholar] [CrossRef]
- Tanaka, M.; Chari, S.; Adsay, V.; Fernandez-del Castillo, C.; Falconi, M.; Shimizu, M.; Yamaguchi, K.; Yamao, K.; Matsuno, S.; International Association of Pancreatology. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006, 6, 17–32. [Google Scholar] [CrossRef]
- Tanaka, M.; Fernández-del Castillo, C.; Adsay, V.; Chari, S.; Falconi, M.; Jang, J.Y.; Kimura, W.; Levy, P.; Pitman, M.B.; Schmidt, C.M.; et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 2012, 12, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Fernández-Del Castillo, C.; Kamisawa, T.; Jang, J.Y.; Levy, P.; Ohtsuka, T.; Salvia, R.; Shimizu, Y.; Tada, M.; Wolfgang, C.L. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology 2017, 17, 738–753. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.M.; Yin, S.; Siddiqui, A.A.; Salem, R.R.; Schrope, B.; Sethi, A.; Poneros, J.M.; Gress, F.G.; Genkinger, J.M.; Do, C.; et al. Comparison of the diagnostic accuracy of three current guidelines for the evaluation of asymptomatic pancreatic cystic neoplasms. Medicine (Baltimore) 2017, 96, e7900. [Google Scholar] [CrossRef] [PubMed]
- Sighinolfi, M.; Quan, S.Y.; Lee, Y.; Ibaseta, A.; Pham, K.; Dua, M.M.; Poultsides, G.A.; Visser, B.C.; Norton, J.A.; Park, W.G. Fukuoka and AGA criteria have superior diagnostic accuracy for advanced cystic neoplasms than Sendai criteria. Dig. Dis. Sci. 2017, 62, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Vege, S.S.; Ziring, B.; Jain, R.; Moayyedi, P.; Clinical Guidelines Committee; American Gastroenterology Association. American gastroenterological association institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology 2015, 148, 819–822, quize 12–13. [Google Scholar] [CrossRef]
- Gaujoux, S.; Brennan, M.F.; Gonen, M.; D’Angelica, M.I.; DeMatteo, R.; Fong, Y.; Schattner, M.; DiMaio, C.; Janakos, M.; Jarnagin, W.R.; et al. Cystic lesions of the pancreas: Changes in the presentation and management of 1424 patients at a single institution over a 15-year time period. J. Am. Coll. Surg. 2011, 212, 590–600; discussion 600-3. [Google Scholar] [CrossRef]
- Ånonsen, K.; Sahakyan, M.A.; Kleive, D.; Waage, A.; Verbeke, C.; Hauge, T.; Buanes, T.; Edwin, B.; Labori, K.J. Trends in management and outcome of cystic pancreatic lesions - analysis of 322 cases undergoing surgical resection. Scand. J. Gastroenterol. 2019, 19, 1–7. [Google Scholar] [CrossRef]
- Vaalavuo, Y.; Antila, A.; Ahola, R.; Siiki, A.; Vornanen, M.; Ukkonen, M.; Sand, J.; Laukkarinen, J. Characteristics and long-term survival of resected pancreatic cystic neoplasms in Finland. The first nationwide retrospective cohort analysis. Pancreatology 2019, 19, 456–461. [Google Scholar] [CrossRef]
- Kang, J.S.; Park, T.; Han, Y.; Lee, S.; Lim, H.; Kim, H.; Kim, S.H.; Kwon, W.; Kim, S.W.; Jang, J.Y. Clinical validation of the 2017 international consensus guidelines on intraductal papillary mucinous neoplasm of the pancreas. Ann. Surg. Treat. Res. 2019, 97, 58–64. [Google Scholar] [CrossRef]
- Pitman, M.B.; Deshpande, V. Endoscopic ultrasound-guided fine needle aspiration cytology of the pancreas: A morphological and multimodal approach to the diagnosis of solid and cystic mass lesions. Cytopathology 2007, 18, 331–347. [Google Scholar] [CrossRef]
- Pitman, M.B.; Centeno, B.A.; Genevay, M.; Fonseca, R.; Mino-Kenudson, M. Grading epithelial atypia in endoscopic ultrasound-guided fine-needle aspiration of intraductal papillary mucinous neoplasms: An international interobserver concordance study. Cancer Cytopathol. 2013, 121, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Yoon, W.J.; Brugge, W.R. The safety of endoscopic ultrasound-guided fine-needle aspiration of pancreatic cystic lesions. Endosc. Ultrasound 2015, 4, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Brugge, W.R.; Dimaio, C.J.; Pitman, M.B. Molecular analysis of pancreatic cyst fluid: A comparative analysis with current practice of diagnosis. Cancer Cytopathol. 2009, 117, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.L.; Arpin, R.N.; Brugge, W.R.; Forcione, D.G.; Basar, O.; Pitman, M.B. Moray micro forceps biopsy improves the diagnosis of specific pancreatic cysts. Cancer Cytopathol. 2018, 126, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.; Zahid, M.; Finkelstein, S.D.; LeBlanc, J.K.; Kaushik, N.; Ahmad, N.; Brugge, W.R.; Edmundowicz, S.A.; Hawes, R.H.; McGrath, K.M. Pancreatic cyst fluid DNA analysis in evaluating pancreatic cysts: A report of the PANDA study. Gastrointest. Endosc. 2009, 69, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Rockacy, M.; Khalid, A. Update on pancreatic cyst fluid analysis. Ann. Gastroenterol. 2013, 26, 122–127. [Google Scholar]
- Brugge, W.R.; Lauwers, G.Y.; Sahani, D.; del Castillo, C.F.; Warshaw, A.L. Cystic neoplasms of the pancreas. N. Engl. J. Med. 2004, 351, 1218–1226. [Google Scholar] [CrossRef]
- Van der Waaij, L.A.; van Dullemen, H.M.; Porte, R.J. Cyst fluid analysis in the differential diagnosis of pancreatic cystic lesions: A pooled analysis. Gastrointest. Endosc. 2005, 62, 383–389. [Google Scholar] [CrossRef]
- Scourtas, A.; Dudley, J.C.; Brugge, W.R.; Kadayifci, A.; Mino-Kenudson, M.; Pitman, M.B. Preoperative characteristics and cytological features of 136 histologically confirmed pancreatic mucinous cystic neoplasms. Cancer 2017, 125, 169–177. [Google Scholar] [CrossRef]
- Brugge, W.R.; Lewandrowski, K.; Lee-Lewandrowski, E.; Centeno, B.A.; Szydio, T.; Regan, S.; del Castillo, C.F.; Warshaw, A.L. Diagnosis of pancreatic cystic neoplasms: A report of the cooperative pancreatic cyst study. Gastroenterology 2004, 126, 1330–1336. [Google Scholar] [CrossRef]
- Park, W.G.; Mascarenhas, R.; Palaez-Luna, M.; Smyrk, T.C.; O’Kane, D.; Clain, J.E.; Levy, M.J.; Pearson, R.K.; Petersen, B.T.; Topazian, M.D.; et al. Diagnostic performance of cyst fluid carcinoembryonic antigen and amylase in histologically confirmed pancreatic cysts. Pancreas 2011, 40, 42–45. [Google Scholar] [CrossRef] [PubMed]
- Thornton, G.D.; McPhail, M.J.; Nayagam, S.; Hewitt, M.J.; Vlavianos, P.; Monahan, K.J. Endoscopic ultrasound guided fine needle aspiration for the diagnosis of pancreatic cystic neoplasms: A meta-analysis. Pancreatology 2013, 13, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.H.; Gonda, T.A. The use of biomarkers in the risk stratification of cystic neoplasms. Gastrointest. Endosc. Clin. N. Am. 2018, 28, 549–568. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Matthaei, H.; Maitra, A.; Dal Molin, M.; Wood, L.D.; Eshleman, J.R.; Goggins, M.; Canto, M.I.; Schulick, R.D.; Edil, B.H.; et al. Recurrent GNAS mutations define an unexpected pathway for pancreatic cyst development. Sci. Transl. Med. 2011, 3, 92ra66. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, M.; Degowska, M.; Rydzewska, G. Cystic fluid neutrophil gelatinase-associated lipocalin (NGAL) concentration in differential diagnosis of pancreatic cystic lesions: A new factor enters the scene? Prz. Gastroenterol. 2018, 13, 132–136. [Google Scholar] [CrossRef]
- Carr, R.A.; Yip-Schneider, M.T.; Simpson, R.E.; Dolejs, S.; Schneider, J.G.; Wu, H.; Ceppa, E.P.; Park, W.; Schmidt, C.M. Pancreatic cyst fluid glucose: Rapid, inexpensive, and accurate diagnosis of mucinous pancreatic cysts. Surgery 2018, 163, 600–605. [Google Scholar] [CrossRef]
- Lopes, C.V. Cyst fluid glucose: An alternative to carcinoembryonic antigen for pancreatic mucinous cysts. World J. Gastroenterol. 2019, 25, 2271–2278. [Google Scholar] [CrossRef]
- Furukawa, T. Subtyping of IPMN. Methods Mol. Biol. 2019, 1882, 1–8. [Google Scholar] [CrossRef]
- Sethi, V.; Giri, B.; Saluja, A.; Dudeja, V. Insights into the Pathogenesis of Pancreatic Cystic Neoplasms. Dig. Dis. Sci. 2017, 62, 1778–1786. [Google Scholar] [CrossRef]
- Maker, A.V.; Katabi, N.; Gonen, M.; DeMatteo, R.P.; D’Angelica, M.I.; Fong, Y.; Jarnagin, W.R.; Brennan, M.F.; Allen, P.J. Pancreatic cyst fluid and serum mucin levels predict dysplasia in intraductal papillary mucinous neoplasms of the pancreas. Ann. Surg. Oncol. 2011, 18, 199–206. [Google Scholar] [CrossRef]
- Sinha, J.; Cao, Z.; Dai, J.; Tang, H.; Partyka, K.; Hostetter, G.; Simeone, D.M.; Feng, Z.; Allen, P.J.; Brand, R.E.; et al. A gastric glycoform of MUC5AC is a biomarker of mucinous cysts of the pancreas. PLoS ONE 2016, 11, e0167070. [Google Scholar] [CrossRef] [PubMed]
- Maker, A.V.; Katabi, N.; Qin, L.X.; Klimstra, D.S.; Schattner, M.; Brennan, M.F.; Jarnagin, W.R.; Allen, P.J. Cyst fluid interleukin-1beta (IL1beta) levels predict the risk of carcinoma in intraductal papillary mucinous neoplasms of the pancreas. Clin. Cancer Res. 2011, 17, 1502–1508. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.M.; Yip-Schneider, M.T.; Ralstin, M.C.; Wentz, S.; DeWitt, J.; Sherman, S.; Howard, T.J.; McHenry, L.; Dutkevitch, S.; Goggins, M.; et al. PGE(2) in pancreatic cyst fluid helps differentiate IPMN from MCN and predict IPMN dysplasia. J. Gastrointest. Surg. 2008, 12, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Yip-Schneider, M.T.; Carr, R.A.; Wu, H.; Schmidt, C.M. Prostaglandin E2: A pancreatic fluid biomarker of intraductal papillary mucinous neoplasm dysplasia. J. Am. Coll Surg. 2017, 225, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, Y.; Murakami, Y.; Uemura, K.; Hayashidani, Y.; Sudo, T.; Ohge, H.; Fukuda, E.; Shimamoto, F.; Sueda, T.; Hiyama, E. Telomere shortening and telomerase expression during multistage carcinogenesis of intraductal papillary mucinous neoplasms of the pancreas. J. Gastrointest. Surg. 2008, 12, 17–28; discussion 28–29. [Google Scholar] [CrossRef] [PubMed]
- Hata, T.; Dal Molin, M.; McGregor-Das, A.; Song, T.J.; Wolfgang, C.; Eshleman, J.R.; Hruban, R.H.; Goggins, M. Simple detection of telomere fusions in pancreatic cancer, intraductal papillary mucinous neoplasm, and pancreatic cyst fluid. J. Mol. Diagn. 2018, 20, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Szafranska, A.E.; Doleshal, M.; Edmunds, H.S.; Gordon, S.; Luttges, J.; Munding, J.B.; Barth, R.J., Jr.; Gutmann, E.J.; Suriawinata, A.A.; Pipas, J.M.; et al. Analysis of microRNAs in pancreatic fine-needle aspirates can classify benign and malignant tissues. Clin. Chem. 2008, 54, 1716–1724. [Google Scholar] [CrossRef]
- Jamieson, N.B.; Morran, D.C.; Morton, J.P.; Ali, A.; Dickson, E.J.; Carter, C.R.; Sansom, O.J.; Evans, T.R.J.; McKay, C.J.; Oien, K.A. MicroRNA molecular profiles associated with diagnosis, clinicopathologic criteria, and overall survival in patients with resectable pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2012, 18, 534–545. [Google Scholar] [CrossRef]
- Wang, J.; Paris, P.L.; Chen, J.; Ngo, V.; Yao, H.; Frazier, M.L.; Killary, A.M.; Liu, C.G.; Liang, H.; Mathy, C.; et al. Next generation sequencing of pancreatic cyst fluid microRNAs from low grade-benign and high grade-invasive lesions. Cancer Lett. 2015, 356, 404–409. [Google Scholar] [CrossRef]
- Das, K.K.; Xiao, H.; Geng, X.; del Castillo, C.F.; Morales-Oyarvide, V.; Daglilar, E.; Forcione, D.G.; Bounds, B.C.; Brugge, W.R.; Pitman, M.B.; et al. mAb Das-1 is specific for high-risk and malignant intraductal papillary mucinous neoplasm IPMN). Gut 2014, 63, 1626–1634. [Google Scholar] [CrossRef]
- Das, K.K.; Geng, X.; Brown, J.W.; Morales-Oyarvide, V.; Huynh, T.; Pergolini, I.; Pitman, M.B.; Ferrone, C.; Al Efishat, M.; Haviland, D.; et al. Cross validation of the monoclonal antibody Das-1 in identification of high-risk mucinous pancreatic cystic lesions. Gastroenterology 2019, 157, 720–730.e2. [Google Scholar] [CrossRef] [PubMed]
- Bournet, B.; Vignolle-Vidoni, A.; Grand, D.; Roques, C.; Breibach, F.; Cros, J.; Muscari, F.; Carrère, N.; Selves, J.; Cordelier, P. Endoscopic ultrasound-guided fine-needle aspiration plus KRAS and GNAS mutation in malignant intraductal papillary mucinous neoplasm of the pancreas. Endosc. Int. Open. 2016, 4, E1228–E1235. [Google Scholar] [CrossRef] [PubMed]
- Faias, S.; Duarte, M.; Albuquerque, C.; da Silva, J.P.; Fonseca, R.; Roque, R.; Dias Pereira, A.; Chaves, P.; Cravo, M. Clinical impact of KRAS and GNAS analysis added to CEA and cytology in pancreatic cystic fluid obtained by EUS-FNA. Dig. Dis. Sci. 2018, 63, 2351–2361. [Google Scholar] [CrossRef] [PubMed]
- Singhi, A.D.; Nikiforova, M.N.; Fasanella, K.E.; McGrath, K.M.; Pai, R.K.; Ohori, N.P.; Bartholow, T.L.; Brand, R.E.; Chennat, J.S.; Lu, X.; et al. Preoperative GNAS and KRAS testing in the diagnosis of pancreatic mucinous cysts. Clin. Cancer Res. 2014, 20, 4381–4389. [Google Scholar] [CrossRef]
- Singhi, A.D.; McGrath, K.; Brand, R.E.; Khalid, A.; Zeh, H.J.; Chennat, J.S.; Fasanella, K.E.; Papachristou, G.I.; Slivka, A.; Bartlett, D.L.; et al. Preoperative next-generation sequencing of pancreatic cyst fluid is highly accurate in cyst classification and detection of advanced neoplasia. Gut 2018, 67, 2131–2141. [Google Scholar] [CrossRef] [PubMed]
- PancreaSeq®—Pancreatic Cyst Fluid NGS Analysis. Available online: https://mgp.upmc.com/Home/Test/PanSeq_details (accessed on 25 August 2019).
- Springer, S.; Wang, Y.; Dal Molin, M.; Masica, D.L.; Jiao, Y.; Kinde, I.; Blackford, A.; Raman, S.P.; Wolfgang, C.L.; Tomita, T.; et al. A combination of molecular markers and clinical features improve the classification of pancreatic cysts. Gastroenterology 2015, 149, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Al Efishat, M.A.; Attiyeh, M.A.; Eaton, A.A.; Gönen, M.; Prosser, D.; Lokshin, A.E.; Castillo, C.F.; Lillemoe, K.D.; Ferrone, C.R.; Pergolini, I.; et al. Multi-institutional validation study of pancreatic cyst fluid protein analysis for prediction of high-risk intraductal papillary mucinous neoplasms of the pancreas. Ann. Surg. 2018, 268, 340–347. [Google Scholar] [CrossRef]
- Maker, A.V.; Hu, V.; Kadkol, S.S.; Hong, L.; Brugge, W.; Winter, J.; Yeo, C.J.; Hackert, T.; Büchler, M.; Lawlor, R.T.; et al. Cyst fluid biosignature to predict intraductal papillary mucinous neoplasms of the pancreas with high malignant potential. J. Am. Coll. Surg. 2019, 228, 721–729. [Google Scholar] [CrossRef]
- Attiyeh, M.A.; Fernández-Del Castillo, C.; Al Efishat, M.; Eaton, A.A.; Gönen, M.; Batts, R.; Pergolini, I.; Rezaee, N.; Lillemoe, K.D.; Ferrone, C.R.; et al. Development and validation of a multi-institutional preoperative nomogram for predicting grade of dysplasia in intraductal papillary mucinous neoplasms (IPMNs) of the pancreas: A report from the pancreatic surgery consortium. Ann. Surg. 2018, 267, 157–163. [Google Scholar] [CrossRef]
- Masica, D.L.; Dal Molin, M.; Wolfgang, C.L.; Tomita, T.; Ostovaneh, M.R.; Blackford, A.; Moran, R.A.; Law, J.K.; Barkley, T.; Goggins, M.; et al. A novel approach for selecting combination clinical markers of pathology applied to a large retrospective cohort of surgically resected pancreatic cysts. J. Am. Med. Inform. Assoc. 2017, 24, 145–152. [Google Scholar] [CrossRef]
- Masica, D.L.; Karchin, R. Correlation among somatic mutation expression identifies genes important in human glioblastoma progression and survival. Cancer Res. 2011, 71, 4550–4561. [Google Scholar] [CrossRef] [PubMed]
- Masica, D.L.; Karchin, R. Collections of simultaneously altered genes as biomarkers of cancer cell drug response. Cancer Res. 2013, 73, 1699–1708. [Google Scholar] [CrossRef] [PubMed]
- Look Below the Surface: Molecular Testing with PancraGEN®. Available online: https://pancragen.com/ (accessed on 25 August 2019).
- Simpson, R.E.; Cockerill, N.J.; Yip-Schneider, M.T.; Ceppa, E.P.; House, M.G.; Zyromski, N.J.; Nakeeb, A.; Al-Haddad, M.A.; Schmidt, C.M. DNA profile components predict malignant outcomes in select cases of intraductal papillary mucinous neoplasm with negative cytology. Surgery 2018, 164, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Simpson, R.E.; Cockerill, N.J.; Yip-Schneider, M.T.; Ceppa, E.P.; House, M.G.; Zyromski, N.J.; Nakeeb, A.; Al-Haddad, M.A.; Schmidt, C.M. Clinical criteria for integrated molecular pathology in intraductal papillary mucinous neoplasm: Less is more. HPB (Oxford) 2019, 21, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Singhi, A.D.; Nikiforova, M.N.; McGrath, K. DNA testing of pancreatic cyst fluid: Is it ready for prime time? Lancet Gastroenterol Hepatol. 2017, 2, 62–73. [Google Scholar] [CrossRef]
- Singhi, A.D.; Zeh, H.J.; Brand, R.E.; Nikiforova, M.N.; Chennat, J.S.; Fasanella, K.E.; Khalid, A.; Papachristou, G.I.; Slivka, A.; Hogg, M.; et al. American Gastroenterological Association guidelines are inaccurate in detecting pancreatic cysts with advanced neoplasia: A clinicopathologic study of 225 patients with supporting molecular data. Gastrointest. Endosc. 2016, 83, 1107–1117. [Google Scholar] [CrossRef]
- Early Detection, Diagnosis, and Prognosis. Available online: https://www.pancan.org/research/grants-program/grants-awarded/by-research-topic/early-detection/ (accessed on 25 August 2019).
- Do, M.; Han, D.; Wang, J.I.; Kim, H.; Kwon, W.; Han, Y.; Jang, J.Y.; Kim, Y. Quantitative proteomic analysis of pancreatic cyst fluid proteins associated with malignancy in intraductal papillary mucinous neoplasms. Clin. Proteomics. 2018, 15, 17. [Google Scholar] [CrossRef]
- Park, J.; Han, D.; Do, M.; Woo, J.; Wang, J.I.; Han, Y.; Kwon, W.; Kim, S.W.; Jang, J.Y.; Kim, Y. Proteome characterization of human pancreatic cyst fluid from intraductal papillary mucinous neoplasm by liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2017, 31, 1761–1772. [Google Scholar] [CrossRef]
- Matthaei, H.; Wylie, D.; Lloyd, M.B.; Dal Molin, M.; Kemppainen, J.; Mayo, S.C.; Wolfgang, C.L.; Schulick, R.D.; Langfield, L.; Andruss, B.F.; et al. miRNA biomarkers in cyst fluid augment the diagnosis and management of pancreatic cysts. Clin. Cancer Res. 2012, 18, 4713–4724. [Google Scholar] [CrossRef]
| Amylase < 250 | Amylase > 250 | |
|---|---|---|
| CEA < 192 | SCA | Pseudocyst |
| CEA > 192 | MCN | IPMN |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, S.; Takahashi, C.; Snyder, R.A.; Parikh, A.A. Stratifying Intraductal Papillary Mucinous Neoplasms by Cyst Fluid Analysis: Present and Future. Int. J. Mol. Sci. 2020, 21, 1147. https://doi.org/10.3390/ijms21031147
Hao S, Takahashi C, Snyder RA, Parikh AA. Stratifying Intraductal Papillary Mucinous Neoplasms by Cyst Fluid Analysis: Present and Future. International Journal of Molecular Sciences. 2020; 21(3):1147. https://doi.org/10.3390/ijms21031147
Chicago/Turabian StyleHao, Scarlett, Caitlin Takahashi, Rebecca A. Snyder, and Alexander A. Parikh. 2020. "Stratifying Intraductal Papillary Mucinous Neoplasms by Cyst Fluid Analysis: Present and Future" International Journal of Molecular Sciences 21, no. 3: 1147. https://doi.org/10.3390/ijms21031147
APA StyleHao, S., Takahashi, C., Snyder, R. A., & Parikh, A. A. (2020). Stratifying Intraductal Papillary Mucinous Neoplasms by Cyst Fluid Analysis: Present and Future. International Journal of Molecular Sciences, 21(3), 1147. https://doi.org/10.3390/ijms21031147





