The Epigenetics of the Endocannabinoid System
Abstract
1. Introduction
2. ES in Summary
3. Epigenetic Mechanisms: A Brief Overview
4. The Epigenetics of ES
4.1. Effects on Peripheral Tissues, Brain Functions, and Disease State
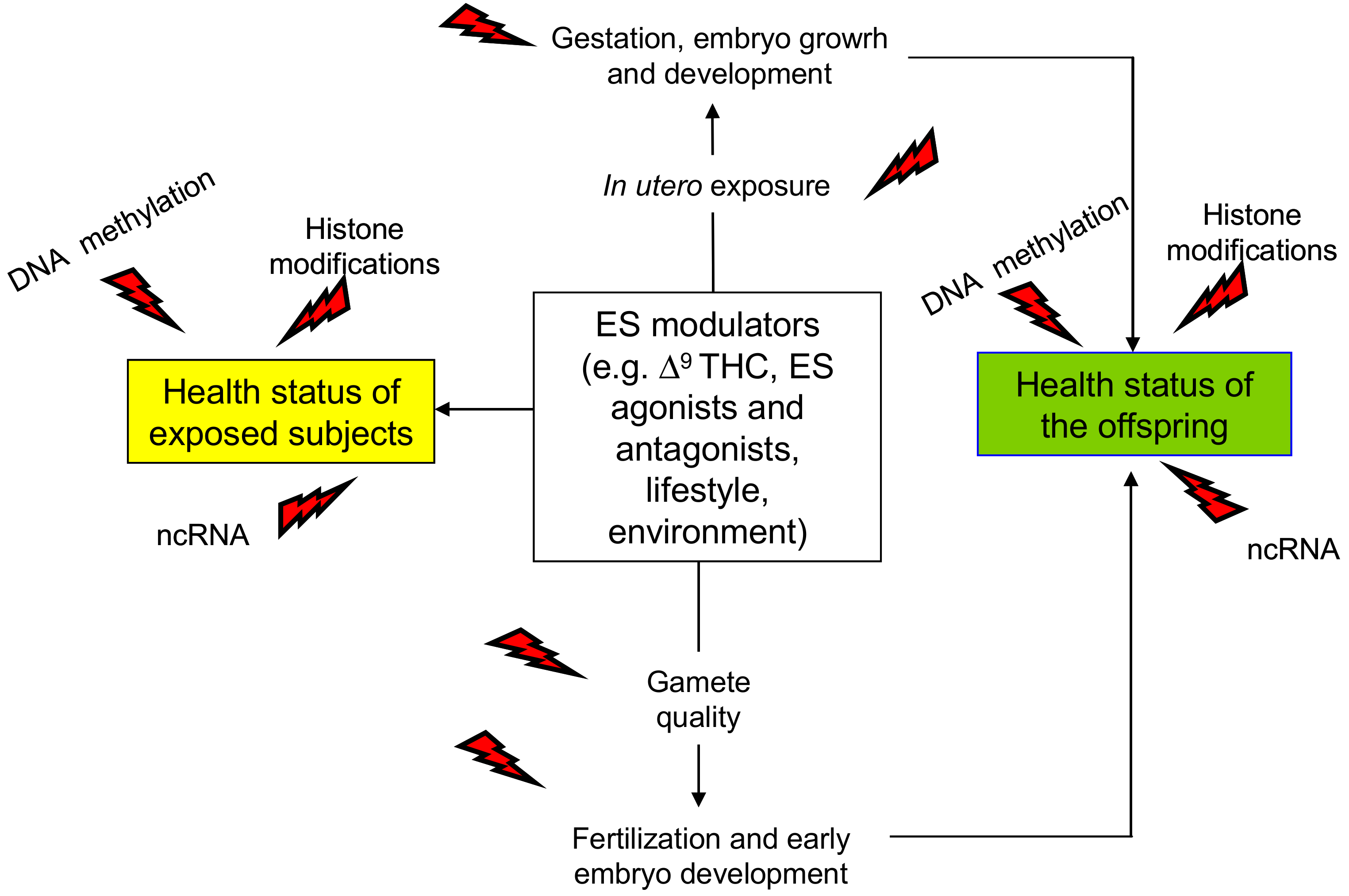
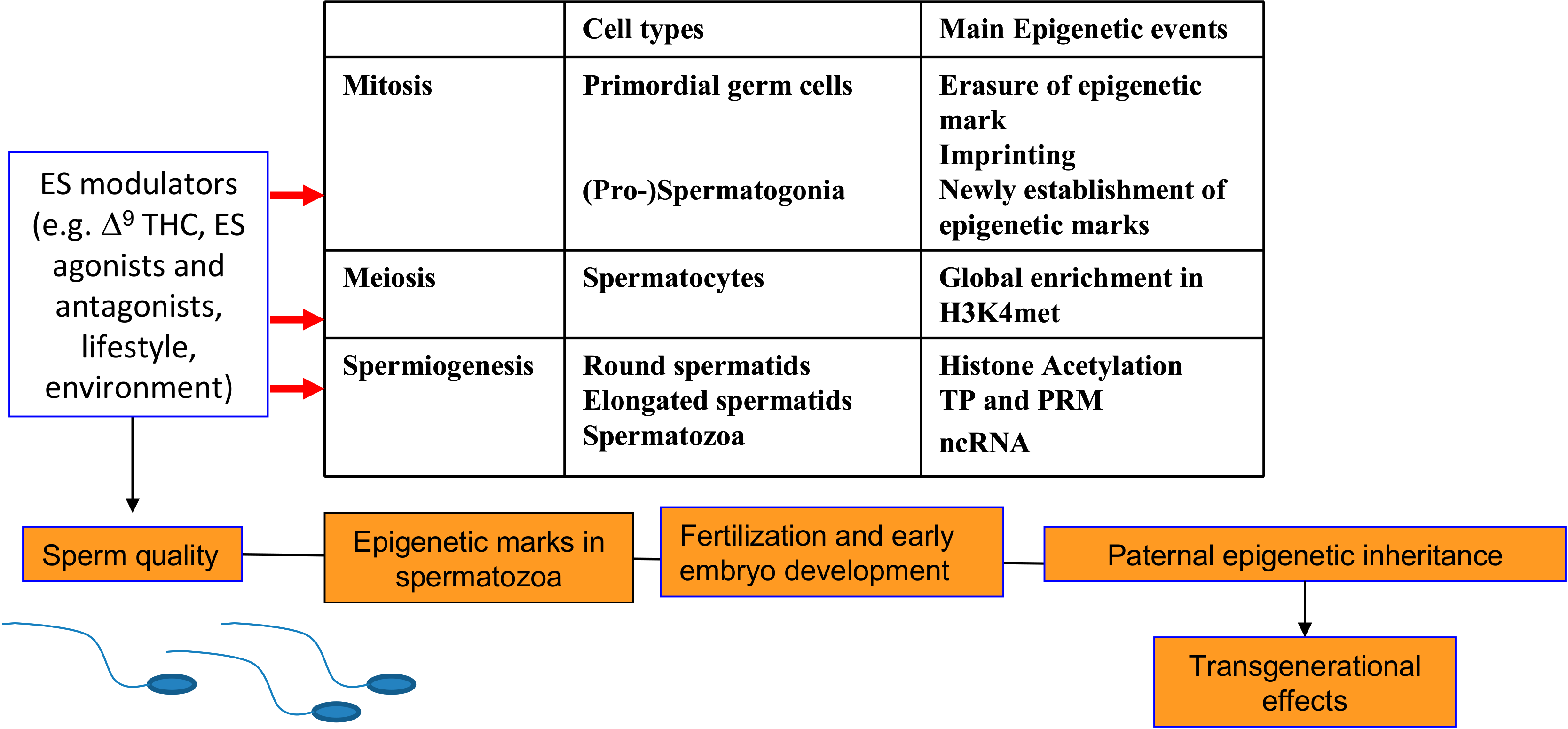
4.2. Effects on Male Reproduction and Embryo Development
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACEA | arachidonyl-2′-chloroethylamine |
| ACPA | arachidonylcyclopropylamide |
| AEA | anandamide |
| 2AG | 2-arachydonoylglicerol |
| APN | adiponectin |
| ARA | arachidonic acid |
| CB1 | type 1 cannabinoid receptor |
| CB2 | type 2 cannabinoid receptor |
| CHF | chronic heart failure |
| circRNA | circular RNA |
| CNR1 | gene encoding for CB1 |
| CpG | cytosine–phosphate-guanine |
| CRC | Colon Rectal Cancer |
| DAGL | sn-1-diacylglycerol lipase |
| Dex | dexmedetomidine |
| DIO | diet-induced obesity |
| DLGAP2 | discs large associated protein 2 |
| DNMT | DNA methyltransferases |
| Drd2 | gene encoding for dopamine receptor D2 |
| Δ9THC | Δ9-tetrahydrocannabinol |
| ER | oestrogen receptor |
| ERE | oestrogen responsive element |
| ES | endocannabinoid system |
| EVOO | extra-virgin olive oil |
| FAAH1 | fatty acid amide hydrolase 1 |
| FAAH2 | fatty acid amide hydrolase 2 |
| FABP5 | fatty acid binding protein 5 |
| FLAT-1 | FAAH-like AEA transporter |
| FSH | follicle stimulating hormone |
| GABA | γ-aminobutyric acid |
| GlcNAcylation | O-linked β-d-N-acetylglucosaminylation |
| GnRH | gonadotropin releasing hormone |
| HAT | histone acetyltransferases |
| HDAC | histone deacetylases |
| HFD | high-fat diet |
| 5hmC | 5-hydroxymethylcytosine |
| HMT | histone methyltransferases |
| HPG | hypothalamus-pituitary-gonad |
| HT | hydroxytyrosol |
| KDM1 | amino oxidase homolog lysine demethylase 1 |
| LH | luteinizing hormone |
| lncRNA | long non-coding RNA |
| LOAD | late-onset Alzheimer’s disease |
| MAGL | monoacylglycerol lipase |
| 5mC | 5-methylcytosine |
| miRNA | microRNA |
| NAPE-PLD | N-acyl-phosphatidylethanolamine-specific phospholipase D |
| ncRNA | non coding RNA |
| OPE | olive oil phenolic extract |
| PBMC | peripheral blood mononuclear cells |
| Penk | gene encoding for Proenkephalin |
| PGC | primordial germ cell |
| P-LGG | paediatric low-grade gliomas |
| piRNA | PIWI-interacting RNA |
| piwi | P-element induced wimpy testis |
| PPARγ | peroxisome proliferator-activated receptor γ |
| PRM | protamine |
| PUFA | poly unsaturated fatty acids |
| SAM | S-adenosyl-L-methionine |
| siRNA | short interfering RNA |
| TET | Ten-eleven translocation methylcytosine dioxygenases |
| tiRNA and tRF | tRNA fragments |
| TP | transition protein |
| TRPV1 | cationic channel type 1 vanilloid receptor |
| TSS | transcription start site |
References
- Lu, H.-C.; Mackie, K. An Introduction to the Endogenous Cannabinoid System. Biol. Psychiatr. 2016, 79, 516–525. [Google Scholar] [CrossRef]
- Meccariello, R.; Chianese, R. (Eds.) Cannabinoids in Health and Disease; IntechOpen: Rijeka, Croatia, 2016; pp. 1–248. ISBN 978-953-51-2429-0. [Google Scholar] [CrossRef]
- Laezza, C.; Pagano, C.; Navarra, G.; Pastorino, O.; Proto, M.C.; Fiore, D.; Piscopo, C.; Gazzerro, P.; Bifulco, M. The Endocannabinoid System: A Target for Cancer Treatment. Int. J. Mol. Sci. 2020, 21, 747. [Google Scholar] [CrossRef]
- Serrano, A.; Parsons, L.H. Endocannabinoid influence in drug reinforcement, dependence and addiction-related behaviors. Pharmacol. Ther. 2011, 132, 215–241. [Google Scholar] [CrossRef] [PubMed]
- Basavarajappa, B.S.; Joshi, V.; Shivakumar, M.; Subbanna, S. Distinct functions of endogenous cannabinoid system in alcohol abuse disorders. Br. J. Pharmacol. 2019, 176, 3085–3109. [Google Scholar] [CrossRef] [PubMed]
- Hayase, T. Epigenetic mechanisms associated with addiction-related behavioural effects of nicotine and/or cocaine: Implication of the endocannabinoid system. Behav. Pharmacol. 2017, 28, 493–511. [Google Scholar] [CrossRef]
- Jaenisch, R.; Bird, A. Epigenetic regulation of gene expression: How the genome integrates intrinsic and environmental signals. Nat. Genet. 2003, 33, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Samaranayake, M.; Pradhan, S. Epigenetic mechanisms in mammals. Cell. Mol. Life Sci. 2009, 66, 596–612. [Google Scholar] [CrossRef] [PubMed]
- Seisenberger, S.; Peat, J.R.; Hore, T.A.; Santos, F.; Dean, W.; Reik, W. Reprogramming DNA methylation in the mammalian life cycle: Building and breaking epigenetic barriers. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2013, 368, 20110330. [Google Scholar] [CrossRef]
- Hogg, K.; Western, P.S. Refurbishing the germline epigenome: Out with the old, in with the new. Semin. Cell. Dev. Biol. 2015, 45, 104–113. [Google Scholar] [CrossRef]
- Bakhshandeh, B.; Kamaleddin, M.A.; Aalishah, K.A. Comprehensive review on exosomes and microvesicles as epigenetic factors. Curr. Stem Cell Res. Ther. 2017, 12, 31–36. [Google Scholar] [CrossRef]
- Qian, Z.; Shen, Q.; Yang, X.; Qiu, Y.; Zhang, W. The role of extracellular vesicles: An epigenetic view of the cancer microenvironment. BioMed Res. Int. 2015, 2015, 649161. [Google Scholar] [CrossRef] [PubMed]
- Motti, M.L.; D’Angelo, S.; Meccariello, R. MicroRNAs, cancer and diet: Facts and new exciting perspectives. Curr. Mol. Pharmacol. 2018, 11, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Daxinger, L.; Whitelaw, E. Understanding transgenerational epigenetic inheritance via the gametes in mammals. Nat. Rev. Genet. 2012, 13, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Derghal, A.; Djelloul, M.; Trouslard, J.; Mounien, L. An Emerging Role of micro-RNA in the Effect of the Endocrine Disruptors. Front. Neurosci. 2016, 10, 318. [Google Scholar] [CrossRef] [PubMed]
- Norouzitallab, P.; Baruah, K.; Vanrompay, D.; Bossier, P. Can epigenetics translate environmental cues into phenotypes? Sci. Total Environ. 2019, 647, 1281–1293. [Google Scholar] [CrossRef] [PubMed]
- Gaoni, Y.; Mechoulam, R. Isolation, structure and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647. [Google Scholar] [CrossRef]
- Cacciola, G.; Chianese, R.; Chioccarelli, T.; Ciaramella, V.; Fasano, S.; Pierantoni, R.; Meccariello, R.; Cobellis, G. Cannabinoids and Reproduction: A Lasting and Intriguing History. Pharmaceuticals 2010, 3, 3275–3323. [Google Scholar] [CrossRef]
- Di Marzo, V. Endocannabinoids: Synthesis and degradation. Rev. Physiol. Biochem. Pharmacol. 2008, 160, 1–24. [Google Scholar] [CrossRef]
- Maccarrone, M. Metabolism of the Endocannabinoid Anandamide: Open Questions after 25 Years. Front. Mol. Neurosci. 2017, 10, 166. [Google Scholar] [CrossRef]
- Kaczocha, M.; Vivieca, S.; Sun, J.; Glaser, S.T.; Deutsch, D.G. Fatty acid-binding proteins transport N-acylethanolamines to nuclear receptors and are targets of endocannabinoid transport inhibitors. J. Biol. Chem. 2012, 287, 3415–3424. [Google Scholar] [CrossRef]
- Liedhegner, E.S.; Vogt, C.D.; Sem, D.S.; Cunningham, C.W.; Hillard, C.J. Sterol carrier protein-2: Binding protein for endocannabinoids. Mol. Neurobiol. 2014, 50, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Bottegoni, G.; Sasso, O.; Bertorelli, R.; Rocchia, W.; Masetti, M.; Guijarro, A.; Lodola, A.; Armirotti, A.; Garau, G.; et al. A catalytically silent FAAH-1 variant drives anandamide transport in neurons. Nat. Neurosci. 2011, 15, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.D.; Le, T.; Fan, G. DNA Methylation and Its Basic Function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Smith, A.C. Genomic imprinting: The emergence of an epigenetic paradigm. Nat. Rev. Genet. 2011, 12, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Li, Y.; Robertson, K.D. DNA Methylation Superior or Subordinate in the Epigenetic Hierarchy? Genes Cancer 2011, 2, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhang, Y. Reversing DNA methylation: Mechanisms, genomics, and biological functions. Cell 2014, 156, 45–68. [Google Scholar] [CrossRef]
- Javaid, N.; Choi, S. Acetylation- and Methylation-Related Epigenetic Proteins in the Context of Their Targets. Genes 2017, 8, 196. [Google Scholar] [CrossRef]
- Wu, D.; Cai, Y.; Jin, J. Potential coordination role between O-GlcNAcylation and epigenetics. Protein Cell 2017, 8, 713–723. [Google Scholar] [CrossRef]
- Xu, Y.M.; Du, J.Y.; Lau, A.T. Posttranslational modifications of human histone H3: An update. Proteomics 2014, 14, 2047–2060. [Google Scholar] [CrossRef]
- Taft, R.J.; Pang, K.C.; Mercer, T.R.; Dinger, M.; Mattick, J.S. Non-coding RNAs: Regulators of disease. J. Pathol. 2010, 220, 126–139. [Google Scholar] [CrossRef]
- Chianese, R.; Troisi, J.; Richards, S.; Scafuro, M.; Fasano, S.; Guida, M.; Pierantoni, R.; Meccariello, R. Bisphenol A in Reproduction: Epigenetic Effects. Curr. Med. Chem. 2018, 25, 748–770. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.A.; Webb, W.M.; Lubin, F.D. Regulatory RNAs and control of epigenetic mechanisms: Expectations for cognition and cognitive dysfunction. Epigenomics 2016, 8, 135–151. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar]
- Saikia, M.; Hatzoglou, M. The Many Virtues of tRNA-derived Stress-induced RNAs (tiRNAs): Discovering Novel Mechanisms of Stress Response and Effect on Human Health. J. Biol. Chem. 2015, 290, 29761–29768. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kuscu, C.; Dutta, A. Biogenesis and Function of Transfer RNA-Related Fragments (tRFs). Trends Biochem. Sci. 2016, 41, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Ransohoff, J.D.; Wei, Y.; Khavari, P.A. The functions and unique features of long intergenic non-coding RNA. Nat. Rev. Mol. Cell Biol. 2018, 19, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, M.; Barbagallo, D.; Chioccarelli, T.; Manfrevola, F.; Cobellis, G.; Di Pietro, C.; Brex, D.; Battaglia, R.; Fasano, S.; Ferraro, B.; et al. CircNAPEPLD is expressed in human and murine spermatozoa and physically interacts with oocyte miRNAs. RNA Biol. 2019, 16, 1237–1248. [Google Scholar] [CrossRef]
- Rong, D.; Sun, H.; Li, Z.; Liu, S.; Dong, C.; Fu, K.; Tang, W.; Cao, H. An emerging function of circRNA-miRNAs-mRNA axis in human diseases. Oncotarget 2017, 8, 73271–73281. [Google Scholar] [CrossRef] [PubMed]
- Kanherkar, R.R.; Bhatia-Dey, N.; Csoka, A.B. Epigenetics across the human lifespan. Front. Cell. Dev. Biol. 2014, 2, 49. [Google Scholar] [CrossRef]
- Horvath, S.; Raj, K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat. Rev. Genet. 2018, 19, 371–384. [Google Scholar] [CrossRef]
- Feinberg, A.P. Phenotypic plasticity and the epigenetics of human disease. Nature 2007, 447, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.D.; Issa, J.J. The promise of epigenetic therapy: Reprogramming the cancer epigenome. Curr. Opin. Genet. Dev. 2017, 42, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hutchison, K.E.; Bryan, A.D.; Filbey, F.M.; Calhoun, V.D.; Claus, E.D.; Lin, D.; Sui, J.; Du, Y.; Liu, J. Opposite Epigenetic Associations With Alcohol Use and Exercise Intervention. Front. Psychiatry 2018, 9, 594. [Google Scholar] [CrossRef]
- Pucci, M.; Micioni Di Bonaventura, M.V.; Zaplatic, E.; Bellia, F.; Maccarrone, M.; Cifani, C.; D’Addario, C. Transcriptional regulation of the endocannabinoid system in a rat model of binge-eating behavior reveals a selective modulation of the hypothalamic fatty acid amide hydrolase gene. Int. J. Eat Disord. 2019, 52, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Rotter, A.; Bayerlein, K.; Hansbauer, M.; Weiland, J.; Sperling, W.; Kornhuber, J.; Biermann, T. CB1 and CB2 receptor expression and promoter methylation in patients with cannabis dependence. Eur. Addict. Res. 2013, 19, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Lomazzo, E.; König, F.; Abassi, L.; Jelinek, R.; Lutz, B. Chronic stress leads to epigenetic dysregulation in the neuropeptide-Y and cannabinoid CB1 receptor genes in the mouse cingulate cortex. Neuropharmacology 2017, 113 Pt A, 301–313. [Google Scholar] [CrossRef]
- Subbanna, S.; Nagre, N.N.; Umapathy, N.S.; Pace, B.S.; Basavarajappa, B.S. Ethanol exposure induces neonatal neurodegeneration by enhancing CB1R Exon1 histone H4K8 acetylation and up-regulating CB1R function causing neurobehavioral abnormalities in adult mice. Int. J. Neuropsychopharmacol. 2014, 18, pyu028. [Google Scholar] [CrossRef] [PubMed]
- Nagre, N.N.; Subbanna, S.; Shivakumar, M.; Psychoyos, D.; Basavarajappa, B.S. CB1-receptor knockout neonatal mice are protected against ethanol-induced impairments of DNMT1, DNMT3A, and DNA methylation. J. Neurochem. 2015, 132, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Correa, F.; De Laurentiis, A.; Franchi, A.M. Ethanol downregulates N-acyl phosphatidylethanolamine-phospholipase D expression in BV2 microglial cells via epigenetic mechanisms. Eur. J. Pharmacol. 2016, 786, 224–233. [Google Scholar] [CrossRef]
- Di Francesco, A.; Falconi, A.; Di Germanio, C.; Micioni Di Bonaventura, M.V.; Costa, A.; Caramuta, S.; Del Carlo, M.; Compagnone, D.; Dainese, E.; Cifani, C.; et al. Extravirgin olive oil up-regulates CB1 tumor suppressor gene in human colon cancer cells and in rat colon via epigenetic mechanisms. J. Nutr. Biochem. 2015, 26, 250–258. [Google Scholar] [CrossRef]
- Almeida, M.M.; Dias-Rocha, C.P.; Reis-Gomes, C.F.; Wang, H.; Atella, G.C.; Cordeiro, A.; Pazos-Moura, C.C.; Joss-Moore, L.; Trevenzoli, I.H. Maternal high-fat diet impairs leptin signaling and up-regulates type-1 cannabinoid receptor with sex-specific epigenetic changes in the hypothalamus of newborn rats. Psychoneuroendocrinology 2019, 103, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yang, W.; Wang, W.; Wang, Z.; Pu, Y.; Chen, H.; Wang, F.; Qian, J. Involvement of miR-665 in protection effect of dexmedetomidine against Oxidative Stress Injury in myocardial cells via CB2 and CK1. Biomed. Pharmacother. 2019, 115, 108894. [Google Scholar] [CrossRef] [PubMed]
- Börner, C.; Martella, E.; Höllt, V.; Kraus, J. Regulation of opioid and cannabinoid receptor genes in human neuroblastoma and T cells by theepigenetic modifiers trichostatin A and 5-aza-2′-deoxycytidine. Neuroimmunomodulation 2012, 19, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Proto, M.C.; Gazzerro, P.; Di Croce, L.; Santoro, A.; Malfitano, A.M.; Pisanti, S.; Laezza, C.; Bifulco, M. Interaction of endocannabinoid system and steroid hormones in the control of colon cancer cell growth. J. Cell. Physiol. 2012, 227, 250–258. [Google Scholar] [CrossRef]
- D’Addario, C.; Di Francesco, A.; Arosio, B.; Gussago, C.; Dell’Osso, B.; Bari, M.; Galimberti, D.; Scarpini, E.; Altamura, A.C.; Mari, D.; et al. Epigenetic regulation of fatty acid amide hydrolase in Alzheimer disease. PLoS ONE 2012, 7, e39186. [Google Scholar] [CrossRef] [PubMed]
- Etcheverry, A.; Aubry, M.; de Tayrac, M.; Vauleon, E.; Boniface, R.; Guenot, F.; Saikali, S.; Hamlat, A.; Riffaud, L.; Menei, P.; et al. DNA methylation in glioblastoma: Impact on gene expression and clinical outcome. BMC Genom. 2010, 11, 701. [Google Scholar] [CrossRef] [PubMed]
- Hasenoehrl, C.; Feuersinger, D.; Sturm, E.M.; Bärnthaler, T.; Heitzer, E.; Graf, R.; Grill, M.; Pichler, M.; Beck, S.; Butcher, L.; et al. G protein-coupled receptor GPR55 promotes colorectal cancer and has opposing effects to cannabinoid receptor 1. Int. J. Cancer 2018, 142, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Qian, X.; Zhu, M.; Li, A.; Fang, M.; Zhu, Y.; Zhang, J. miR-1273g-3p promotes proliferation, migration and invasion of LoVo cells via cannabinoid receptor 1 through activation of ERBB4/PIK3R3/mTOR/S6K2 signaling pathway. Mol. Med. Rep. 2018, 17, 4619–4626. [Google Scholar] [CrossRef]
- Tung, C.W.; Ho, C.; Hsu, Y.C.; Huang, S.C.; Shih, Y.H.; Lin, C.L. MicroRNA-29a Attenuates Diabetic Glomerular Injury through Modulating Cannabinoid Receptor 1 Signaling. Molecules 2019, 24, 264. [Google Scholar] [CrossRef]
- Möhnle, P.; Schütz, S.V.; Schmidt, M.; Hinske, C.; Hübner, M.; Heyn, J.; Beiras-Fernandez, A.; Kreth, S. MicroRNA-665 is involved in the regulation of the expression of the cardioprotective cannabinoid receptor CB2 in patients with severe heart failure. Biochem. Biophys. Res. Commun. 2014, 451, 516–521. [Google Scholar] [CrossRef]
- Sredni, S.T.; Huang, C.C.; Suzuki, M.; Pundy, T.; Chou, P.; Tomita, T. Spontaneous involution of pediatric low-grade gliomas: High expression of cannabinoid receptor 1 (CNR1) at the time of diagnosis may indicate involvement of the endocannabinoid system. Childs Nerv. Syst. 2016, 32, 2061–2067. [Google Scholar] [CrossRef] [PubMed]
- Chiarlone, A.; Börner, C.; Martín-Gómez, L.; Jiménez-González, A.; García-Concejo, A.; García-Bermejo, M.L.; Lorente, M.; Blázquez, C.; García-Taboada, E.; de Haro, A.; et al. MicroRNA let-7d is a target of cannabinoid CB1 receptor and controls cannabinoid signaling. Neuropharmacology 2016, 108, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Most, D.; Salem, N.A.; Tiwari, G.R.; Blednov, Y.A.; Mayfield, R.D.; Harris, R.A. Silencing synaptic MicroRNA-411 reduces voluntary alcohol consumption in mice. Addict. Biol. 2019, 24, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, J.; Ehrlich, S.; Walton, E.; Whitem, T.; Perrone-Bizzozero, N.; Bustillo, J.; Turner, J.A.; Calhoun, V.D. Methylation patterns in whole blood correlate with symptoms in schizophrenia patients. Schizophr. Bull. 2014, 40, 769–776. [Google Scholar] [CrossRef]
- Pucci, M.; Rapino, C.; Di Francesco, A.; Dainese, E.; D’Addario, C.; Maccarrone, M. Epigenetic control of skin differentiation genes by phytocannabinoids. Br. J. Pharmacol. 2013, 170, 581–591. [Google Scholar] [CrossRef]
- Molina, P.E.; Amedee, A.; LeCapitaine, N.J.; Zabaleta, J.; Mohan, M.; Winsauer, P.; Vande Stouwe, C. Cannabinoid neuroimmune modulation of SIV disease. J. Neuroimmune Pharmacol. 2011, 6, 516–527. [Google Scholar] [CrossRef]
- Khare, M.; Taylor, A.H.; Konje, J.C.; Bell, S.C. Delta9-tetrahydrocannabinol inhibits cytotrophoblast cell proliferation and modulates gene transcription. Mol. Hum. Reprod. 2006, 12, 321–333. [Google Scholar] [CrossRef]
- Tomasiewicz, H.C.; Jacobs, M.M.; Wilkinson, M.B.; Wilson, S.P.; Nestler, E.J.; Hurd, Y.L. Proenkephalin mediates the enduring effects of adolescent cannabis exposure associated with adult opiate vulnerability. Biol. Psychiatry 2012, 72, 803–810. [Google Scholar] [CrossRef]
- DiNieri, J.A.; Wang, X.; Szutorisz, H.; Spano, S.M.; Kaur, J.; Casaccia, P.; Dow-Edwards, D.; Hurd, Y.L. Maternal cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol. Psychiatry 2011, 70, 763–769. [Google Scholar] [CrossRef]
- Prini, P.; Penna, F.; Sciuccati, E.; Alberio, T.; Rubino, T. Chronic Δ⁸-THC Exposure Differently Affects Histone Modifications in the Adolescent and Adult Rat Brain. Int. J. Mol. Sci. 2017, 18, 2094. [Google Scholar] [CrossRef]
- Prini, P.; Rusconi, F.; Zamberletti, E.; Gabaglio, M.; Penna, F.; Fasano, M.; Battaglioli, E.; Parolaro, D.; Rubino, T. Adolescent THC exposure in female rats leads to cognitive deficits through a mechanism involving chromatin modifications in the prefrontal cortex. J. Psychiatry Neurosci. 2017, 42, 170082. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Hegde, V.L.; Rao, R.; Zhang, J.; Nagarkatti, P.S.; Nagarkatti, M. Histone modifications are associated with Δ9-tetrahydrocannabinol-mediated alterations in antigen-specific T cell responses. J. Biol. Chem. 2014, 289, 18707–18718. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Bam, M.; Nagarkatti, P.S.; Nagarkatti, M. RNA-seq Analysis of δ9-Tetrahydrocannabinol-treated T Cells Reveals Altered Gene Expression Profiles That Regulate Immune Response and Cell Proliferation. J. Biol. Chem. 2016, 291, 15460–15472. [Google Scholar] [CrossRef] [PubMed]
- Szutorisz, H.; DiNieri, J.A.; Sweet, E.; Egervari, G.; Michaelides, M.; Carter, J.M.; Ren, Y.; Miller, M.L.; Blitzer, R.D.; Hurd, Y.L. Parental THC Exposure Leads to Compulsive Heroin-Seeking and Altered Striatal Synaptic Plasticity in the Subsequent Generation. Neuropsychopharmacology 2014, 39, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- Hegde, V.L.; Tomar, S.; Jackson, A.; Rao, R.; Yang, X.; Singh, U.P.; Singh, N.P.; Nagarkatti, P.S.; Nagarkatti, M. Distinct microRNA expression profile and targeted biological pathways in functional myeloid-derived suppressor cells induced by Delta9-tetrahydrocannabinol in vivo: Regulation of CCAAT/enhancer-binding protein alpha by microRNA-690. J. Biol. Chem. 2013, 288, 36810–36826. [Google Scholar] [CrossRef]
- Chandra, L.C.; Kumar, V.; Torben, W.; Vande Stouwe, C.; Winsauer, P.; Amedee, A.; Molina, P.E.; Mohan, M. Chronic administration of Delta9-tetrahydrocannabinol induces intestinal anti-inflammatory microRNA expression during acute simian immunodeficiency virus infection of rhesus macaques. J. Virol. 2015, 89, 1168–1181. [Google Scholar] [CrossRef]
- Levin, E.D.; Hawkey, A.B.; Hall, B.J.; Cauley, M.; Slade, S.; Yazdani, E.; Kenou, B.; White, H.; Wells, C.; Rezvani, A.H.; et al. Paternal THC exposure in rats causes long-lasting neurobehavioral effects in the offspring. Neurotoxicol. Teratol. 2019, 74, 106806. [Google Scholar] [CrossRef]
- Watson, C.T.; Szutorisz, H.; Garg, P.; Martin, Q.; Landry, J.A.; Sharp, A.J.; Hurd, Y.L. Genome-Wide DNA Methylation Profiling Reveals Epigenetic Changes in the Rat Nucleus Accumbens Associated With Cross-Generational Effects of Adolescent THC Exposure. Neuropsychopharmacology 2015, 40, 2993–3005. [Google Scholar] [CrossRef]
- Murphy, S.K.; Itchon-Ramos, N.; Visco, Z.; Huang, Z.; Grenier, C.; Schrott, R.; Acharya, K.; Boudreau, M.H.; Price, T.M.; Raburn, D.J.; et al. Cannabinoid exposure and altered DNA methylation in rat and human sperm. Epigenetics 2018, 13, 1208–1221. [Google Scholar] [CrossRef]
- Schrott, R.; Acharya, K.; Itchon-Ramos, N.; Hawkey, A.B.; Pippen, E.; Mitchell, J.T.; Kollins, S.H.; Levin, E.D.; Murphy, S.K. Cannabis use is associated with potentially heritable widespread changes in autism candidate gene DLGAP2 DNA methylation in sperm. Epigenetics 2020, 15, 161–173. [Google Scholar] [CrossRef]
- Ernst, J.; Grabiec, U.; Greither, T.; Fischer, B.; Dehghani, F. The endocannabinoid system in the human granulosa cell line KGN. Mol. Cell. Endocrinol. 2016, 423, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wu, Y.; Valverde, P.; Murray, D.; Tang, J.; Yao, Q.; Han, Q.; Zhang, J.; Zhang, L.; Sui, L.; et al. Central adiponectin induces trabecular bone mass partly through epigenetic downregulation of cannabinoid receptor CB1. J. Cell Physiol. 2019, 234, 7062–7069. [Google Scholar] [CrossRef] [PubMed]
- Hayase, T. Putative Epigenetic Involvement of the Endocannabinoid System in Anxiety- and Depression-Related Behaviors Caused by Nicotine as a Stressor. PLoS ONE 2016, 11, e0158950. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, A.; Pasquariello, N.; Barcaroli, D.; Maccarrone, M. Anandamide regulates keratinocyte differentiation by inducing DNA methylation in a CB1 receptor-dependent manner. J. Biol. Chem. 2008, 283, 6005–6012. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.R.; Nagarkatti, P.; Nagarkatti, M. Anandamide attenuates Th-17 cell-mediated delayed-type hypersensitivity response by triggering IL-10 production and consequent microRNA induction. PLoS ONE 2014, 9, e93954. [Google Scholar] [CrossRef]
- Večeřa, J.; Bártová, E.; Krejčí, J.; Legartová, S.; Komůrková, D.; Rudá-Kučerová, J.; Štark, T.; Dražanová, E.; Kašpárek, T.; Šulcová, A.; et al. HDAC1 and HDAC3 underlie dynamic H3K9 acetylation during embryonic neurogenesis and in schizophrenia-like animals. J. Cell Physiol. 2018, 233, 530–548. [Google Scholar] [CrossRef]
- Miranda, K.; Mehrpouya-Bahrami, P.; Nagarkatti, P.S.; Nagarkatti, M. Cannabinoid Receptor 1 Blockade Attenuates Obesity and Adipose Tissue Type 1 Inflammation Through miR-30e-5p Regulation of Delta-Like-4 in Macrophages and Consequently Downregulation of Th1 Cells. Front. Immunol. 2019, 10, 1049. [Google Scholar] [CrossRef]
- Shamran, H.; Singh, N.P.; Zumbrun, E.E.; Murphy, A.; Taub, D.D.; Mishra, M.K.; Price, R.L.; Chatterjee, S.; Nagarkatti, M.; Nagarkatti, P.S.; et al. Fatty acid amide hydrolase (FAAH) blockade ameliorates experimental colitis by altering microRNA expression and suppressing inflammation. Brain Behav. Immun. 2017, 59, 10–20. [Google Scholar] [CrossRef]
- Hollins, S.L.; Zavitsanou, K.; Walker, F.R.; Cairns, M.J. Alteration of imprinted Dlk1-Dio3 miRNA cluster expression in the entorhinal cortex induced by maternal immune activation and adolescent cannabinoid exposure. Transl. Psychiatry 2014, 4, e452. [Google Scholar] [CrossRef]
- Aguado, T.; Carracedo, A.; Julien, B.; Velasco, G.; Milman, G.; Mechoulam, R.; Alvarez, L.; Guzmán, M.; Galve-Roperh, I. Cannabinoids induce glioma stem-like cell differentiation and inhibit gliomagenesis. J. Biol. Chem. 2007, 282, 6854–6862. [Google Scholar] [CrossRef]
- Di Giacomo, D.; De Domenico, E.; Sette, C.; Geremia, R.; Grimaldi, P. Type 2 cannabinoid receptor contributes to the physiological regulation of spermatogenesis. FASEB J. 2016, 30, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Innocenzi, E.; De Domenico, E.; Ciccarone, F.; Zampieri, M.; Rossi, G.; Cicconi, R.; Bernardini, R.; Mattei, M.; Grimaldi, P. Paternal activation of CB2 cannabinoid receptor impairs placental and embryonic growth via an epigenetic mechanism. Sci. Rep. 2019, 9, 17034. [Google Scholar] [CrossRef] [PubMed]
- Mehrpouya-Bahrami, P.; Miranda, K.; Singh, N.P.; Zumbrun, E.E.; Nagarkatti, M.; Nagarkatti, P.S. Role of microRNA in CB1 antagonist-mediated regulation of adipose tissue macrophage polarization and chemotaxis during diet-induced obesity. J. Biol. Chem. 2019, 294, 7669–7681. [Google Scholar] [CrossRef] [PubMed]
- Ibn Lahmar Andaloussi, Z.; Taghzouti, K.; Abboussi, O. Behavioural and epigenetic effects of paternal exposure to cannabinoids during adolescence on offspring vulnerability to stress. Int. J. Dev. Neurosci. 2019, 72, 48–54. [Google Scholar] [CrossRef]
- Tomas-Roig, J.; Benito, E.; Agis-Balboa, R.C.; Piscitelli, F.; Hoyer-Fender, S.; Di Marzo, V.; Havemann-Reinecke, U. Chronic exposure to cannabinoids during adolescence causes long-lasting behavioral deficits in adult mice. Addict Biol. 2017, 22, 1778–1789. [Google Scholar] [CrossRef]
- Chianese, R.; Coccurello, R.; Viggiano, A.; Scafuro, M.; Fiore, M.; Coppola, G.; Operto, F.F.; Fasano, S.; Layé, S.; Pierantoni, R.; et al. Impact of dietary fat on brain functions. Curr. Neuropharmacol. 2018, 16, 1059–1085. [Google Scholar] [CrossRef]
- Sam, A.H.; Salem, V.; Ghatei, M.A. Rimonabant: From RIO to Ban. J. Obes. 2011, 2011, 432607. [Google Scholar] [CrossRef]
- Di Marzo, V.; Goparaju, S.K.; Wang, L.; Liu, J.; Bátkai, S.; Járai, Z.; Fezza, F.; Miura, G.I.; Palmiter, R.D.; Sugiura, T.; et al. Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature 2001, 410, 822–825. [Google Scholar] [CrossRef]
- Swerdloff, R.S.; Batt, R.A.; Bray, G.A. Reproductive hormonal function in the genetically obese (ob/ob) mouse. Endocrinology 1976, 98, 1359–1364. [Google Scholar] [CrossRef]
- Hay, E.A.; McEwan, A.; Wilson, D.; Barrett, P.; D’Agostino, G.; Pertwee, R.G.; MacKenzie, A. Disruption of an enhancer associated with addictive behaviour within the cannabinoid receptor-1 gene suggests a possible role in alcohol intake, cannabinoid response and anxiety-related behaviour. Psychoneuroendocrinology 2019, 109, 104407. [Google Scholar] [CrossRef]
- Wang, H.; Dey, S.K.; Maccarrone, M. Jekyll and Hyde: Two faces of cannabinoid signaling in male and female fertility. Endocr. Rev. 2006, 27, 427–448. [Google Scholar] [CrossRef] [PubMed]
- Fasano, S.; Meccariello, R.; Cobellis, G.; Chianese, R.; Cacciola, G.; Chioccarelli, T.; Pierantoni, R. The endocannabinoid system: An ancient signaling involved in the control of male fertility. Annals Ann. N. Y. Acad. Sci. 2009, 1163, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Pierantoni, R.; Cobellis, G.; Meccariello, R.; Cacciola, G.; Chianese, R.; Chioccarelli, T.; Fasano, S. Testicular gonadotropin-releasing hormone activity, progression of spermatogenesis and sperm transport in vertebrates. Ann. N. Y. Acad. Sci. 2009, 1163, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Battista, N.; Meccariello, R.; Cobellis, G.; Fasano, S.; Di Tommaso, M.; Pirazzi, V.; Konje, J.C.; Pierantoni, R.; Maccarrone, M. The role of endocannabinoids in gonadal function and fertility along the evolutionary axis. Mol. Cell. Endocrinol. 2012, 355, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Meccariello, R.; Battista, N.; Bradshaw, H.B.; Wang, H. Updates in reproduction coming from the endocannabinoid system. Int. J. Endocrinol. 2014, 2014, 412354. [Google Scholar] [CrossRef]
- Bovolin, P.; Cottone, E.; Pomatto, V.; Fasano, S.; Pierantoni, R.; Cobellis, G.; Meccariello, R. Endocannabinoids are involved in male vertebrate reproduction: Regulatory mechanisms at central and gonadal level. Front. Endocrinol. 2014, 5, 54. [Google Scholar] [CrossRef]
- Cobellis, G.; Meccariello, R.; Chianese, R.; Chioccarelli, T.; Fasano, S.; Pierantoni, R. Effects of neuroendocrine CB1 activity on adult Leydig cells. Front. Endocrinol. 2016, 7, 47. [Google Scholar] [CrossRef]
- Cecconi, S.; Rapino, C.; Di Nisio, V.; Rossi, G.; Maccarrone, M. The (endo)cannabinoid signaling in female reproduction: What are the latest advances? Prog. Lipid Res. 2019, 77, 101019. [Google Scholar] [CrossRef]
- Meccariello, R.; Franzoni, M.F.; Chianese, R.; Cottone, E.; Scarpa, D.; Donna, D.; Cobellis, G.; Guastalla, A.; Pierantoni, R.; Fasano, S. Interplay between the endocannabinoid system and GnRH-I in the forebrain of the anuran amphibian Rana esculenta. Endocrinology 2008, 149, 2149–2158. [Google Scholar] [CrossRef]
- Cacciola, G.; Chioccarelli, T.; Mackie, K.; Meccariello, R.; Ledent, C.; Fasano, S.; Pierantoni, R.; Cobellis, G. Expression of type-1 cannabinoid receptor during rat postnatal testicular development: Possible involvement in adult Leydig cell differentiation. Biol. Reprod. 2008, 79, 758–765. [Google Scholar] [CrossRef]
- Grimaldi, P.; Orlando, P.; Di Siena, S.; Lolicato, F.; Petrosino, S.; Bisogno, T.; Geremia, R.; De Petrocellis, L.; Di Marzo, V. The endocannabinoid system and pivotal role of the CB2 receptor in mouse spermatogenesis. Proc. Natl. Acad. Sci. USA 2009, 106, 11131–11136. [Google Scholar] [CrossRef]
- Trabucco, E.; Acone, G.; Marenna, A.; Pierantoni, R.; Cacciola, G.; Chioccarelli, T.; Mackie, K.; Fasano, S.; Colacurci, N.; Meccariello, R.; et al. Endocannabinoid System in First Trimester Placenta: Low FAAH and High CB1 Expression Characterize Spontaneous Miscarriage. Placenta 2009, 30, 516–522. [Google Scholar] [CrossRef]
- Acone, G.; Trabucco, E.; Colacurci, N.; Cobellis, L.; Mackie, K.; Meccariello, R.; Cacciola, G.; Chioccarelli, T.; Fasano, S.; Pierantoni, R.; et al. Low type I cannabinoid receptor levels characterize placental villous in labouring delivery. Placenta 2009, 30, 203–205. [Google Scholar] [CrossRef]
- Aquila, S.; Guido, C.; Laezza, C.; Santoro, A.; Pezzi, V.; Panza, S.; Andò, S.; Bifulco, M. A new role of anandamide in human sperm: Focus on metabolism. J. Cell Physiol. 2009, 221, 147–153. [Google Scholar] [CrossRef]
- Aquila, S.; Guido, C.; Santoro, A.; Perrotta, I.; Laezza, C.; Bifulco, M.; Sebastiano, A. Human sperm anatomy: Ultrastructural localization of the cannabinoid1 receptor and a potential role of anandamide in sperm survival and acrosome reaction. Anat. Rec. 2010, 293, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Chioccarelli, T.; Cacciola, G.; Altucci, L.; Lewis, S.E.; Simon, L.; Ricci, G.; Ledent, C.; Meccariello, R.; Fasano, S.; Pierantoni, R.; et al. Cannabinoid Receptor 1 Influences Chromatin Remodeling in Mouse Spermatids by Affecting Content of Transition Protein 2 mRNA and Histone Displacement. Endocrinology 2010, 151, 5017–5029. [Google Scholar] [CrossRef] [PubMed]
- Catanzaro, G.; Battista, N.; Rossi, G.; Di Tommaso, M.; Pucci, M.; Pirazzi, V.; Cecconi, S.; Maccarrone, M. Effect of capacitation on the endocannabinoid system of mouse sperm. Mol. Cell. Endocrinol. 2011, 343, 88–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chianese, R.; Ciaramella, V.; Scarpa, D.; Fasano, S.; Pierantoni, R.; Meccariello, R. Anandamide regulates the expression of GnRH1, GnRH2 and GnRHRs in frog testis. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E475–E487. [Google Scholar] [CrossRef]
- Cacciola, G.; Chioccarelli, T.; Altucci, L.; Ledent, C.; Mason, J.I.; Fasano, S.; Pierantoni, R.; Cobellis, G. Low 17beta-estradiol levels in CNR1 knock-out mice affect spermatid chromatin remodeling by interfering with chromatin reorganization. Biol. Reprod. 2013, 88, 152. [Google Scholar] [CrossRef]
- Ciaramella, V.; Meccariello, R.; Chioccarelli, T.; Sirleto, M.; Fasano, S.; Pierantoni, R.; Chianese, R. Anandamide acts via kisspeptin in the regulation of testicular activity of the frog, Pelophylax esculentus. Mol. Cell. Endocrinol. 2016, 420, 75–84. [Google Scholar] [CrossRef]
- Migliaccio, M.; Ricci, G.; Suglia, A.; Manfrevola, F.; Mackie, K.; Fasano, S.; Pierantoni, R.; Chioccarelli, T.; Cobellis, G. Analysis of Endocannabinoid System in Rat Testis During the First Spermatogenetic Wave. Front. Endocrinol. 2018, 9, 269. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.E.; Rolland, A.D.; Rajpert-De Meyts, E.; Janfelt, C.; Jørgensen, A.; Winge, S.B.; Kristensen, D.M.; Juul, A.; Chalmel, F.; Jégou, B.; et al. Characterisation and localisation of the endocannabinoid system components in the adult human testis. Sci. Rep. 2019, 9, 12866. [Google Scholar] [CrossRef] [PubMed]
- Barchi, M.; Innocenzi, E.; Giannattasio, T.; Dolci, S.; Rossi, P.; Grimaldi, P. Cannabinoid receptor signalling in the development, epigenetics and tumors of male germ cells. Int. J. Mol. Sci. 2019, 21, 25. [Google Scholar] [CrossRef] [PubMed]
- Cobellis, G.; Ricci, G.; Cacciola, G.; Orlando, P.; Petrosino, S.; Cascio, M.G.; Bisogno, T.; De Petrocellis, L.; Chioccarelli, T.; Altucci, L.; et al. A Gradient of 2-Arachidonoylglycerol Regulates Mouse Epididymal Sperm Cell Start-Up. Biol. Reprod. 2010, 82, 451–458. [Google Scholar] [CrossRef]
- Santoro, A.; Chianese, R.; Troisi, J.; Richards, S.; Nori, S.L.; Fasano, S.; Guida, M.; Plunk, E.; Viggiano, A.; Pierantoni, R.; et al. Neuro-toxic and Reproductive Effects of BPA. Curr. Neuropharmacol. 2019, 17, 1109–1132. [Google Scholar] [CrossRef]
- Motti, M.L.; Meccariello, R. Minireview: The epigenetic modulation of KISS1 in cancer and reproduction. Int. J. Environ. Res. Public Health 2019, 16, 2607. [Google Scholar] [CrossRef]
- Chamani, I.J.; Keefe, D.L. Epigenetics and Female Reproductive Aging. Front. Endocrinol. 2019, 10, 473. [Google Scholar] [CrossRef]
- Fernandez-Fernandez, R.; Martini, A.C.; Navarro, V.M.; Castellano, J.M.; Dieguez, C.; Aguilar, E.; Pinilla, L.; Tena-Sempere, M. Novel signals for the integration of energy balance and reproduction. Mol. Cell. Endocrinol. 2006, 254–255, 127–132. [Google Scholar] [CrossRef]
- Rossi, G.; Gasperi, V.; Paro, R.; Barsacchi, D.; Cecconi, S.; Maccarrone, M. Follicle-stimulating hormone activates fatty acid amide hydrolase by protein kinase A and aromatase-dependent pathways in mouse primary Sertoli cells. Endocrinology 2007, 148, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Waleh, N.S.; Cravatt, B.F.; Apte-Deshpande, A.; Terao, A.; Kilduff, T.S. Transcriptional regulation of the mouse fatty acid amide hydrolase gene. Gene 2002, 291, 203–210. [Google Scholar] [CrossRef]
- Grimaldi, P.; Pucci, M.; Di Siena, S.; Di Giacomo, D.; Pirazzi, V.; Geremia, R.; Maccarrone, M. The faah gene is the first direct target of estrogen in the testis: Role of histone demethylase LSD1. Cell. Mol. Life Sci. 2012, 69, 4177–4190. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R. Epididymosomes: Role of extracellular microvesicles in sperm maturation. Front. Biosci. (Schol. Ed.) 2016, 8, 106–114. [Google Scholar] [CrossRef]
- Sharma, U.; Conine, C.C.; Shea, J.M.; Boskovic, A.; Derr, A.G.; Bing, X.Y.; Belleannee, C.; Kucukural, A.; Serra, R.W.; Sun, F.; et al. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 2016, 351, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.W.; Li, H.M.; Qing, X.R.; Huang, D.H.; Li, H.G. Identification and characterization of human testis derived circular RNAs and their existence in seminal plasma. Sci. Rep. 2016, 6, 39080. [Google Scholar] [CrossRef] [PubMed]
- Fereshteh, Z.; Schmidt, S.A.; Al-Dossary, A.A.; Accerbi, M.; Arighi, C.; Cowart, J.; Song, J.L.; Green, P.J.; Choi, K.; Yoo, S.; et al. Murine Oviductosomes (OVS) microRNA profiling during the estrous cycle: Delivery of OVS-borne microRNAs to sperm where miR-34c-5p localizes at the centrosome. Sci. Rep. 2018, 8, 16094. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Shi, J.; Zhang, Y.; Zhang, H.; Liao, S.; Li, W.; Lei, L.; Han, C.; Ning, L.; Cao, Y.; et al. A novel class of tRNA-derived small RNAs extremely enriched in mature mouse sperm. Cell Res. 2012, 22, 1609–1612. [Google Scholar] [CrossRef] [PubMed]
- Kotaja, N. MicroRNAs and spermatogenesis. Fertil. Steril. 2014, 101, 1552–1562. [Google Scholar] [CrossRef]
- Rathke, C.; Baarends, W.M.; Awe, S.; Renkawitz-Pohl, R. Chromatin dynamics during spermiogenesis. Biochim. Biophys. Acta 2014, 1839, 155–168. [Google Scholar] [CrossRef]
- Cacciola, G.; Chioccarelli, T.; Altucci, L.; Viggiano, A.; Fasano, S.; Pierantoni, R.; Cobellis, G. Nuclear size as estrogen-responsive chromatin quality parameter of mouse spermatozoa. Gen. Comp. Endocrinol. 2013, 193, 201–209. [Google Scholar] [CrossRef]
| Epigenetic Cues | Epigenetic Modification/Target | Experimental Model and Subjects (n) | Results | References | |
|---|---|---|---|---|---|
| Environ-mental factors/lifestyle | Alcohol and exercise | ↑ DNA methylation | Human saliva (Exercise cohort, n = 53; case-control cohort, n = 81 drinkers and n = 81 controls; drinking cohort, n = 281) | CpG sites in the FAAH gene show opposite DNA methylation patterns in the drinkers vs. exercise cohort | [44] |
| Binge-eating episodes | ↓ H3K4ac at the FAAH gene promoter | Rat brain | Selective down-regulation of FAAH gene expression in the hypothalamus | [45] | |
| Δ9THC smokers, cigarette smokers and non-smokers | ↑ methylation rate of the CNR1 promoter | Human peripheral blood cells (n = 77, 36 with Δ9THC dependence, 21 cigarette smokers, and 20 non-smokers) | Mean promoter methylation negatively correlated with CB1 expression levels | [46] | |
| Chronic unpredictable stress | ↓ H3K9ac and ↑ HDAC2 activity | Mice | Reduced expression of NPY and CB1 in the cingulated cortex | [47] | |
| Ethanol | ↑ H4K8ac at CNR1 exon 1 | Postnatal day 7 mice | Increased CB1 expression in the hippocampus and neocortex, causing neurobehavioral abnormalities in adult mice | [48] | |
| Ethanol | ↓ DNMT1 and DNMT3A levels impairing DNA methylation | Mouse model of foetal alcohol spectrum disorder. PD7 wild type and CB1-/- mice | The lack of CB1 rescues the loss of DNMT1, DNMT3A, and DNA methylation | [49] | |
| Ethanol | ↑ histone acetyltransferase activity and ↑ histone H3 acetylation | Murine BV2 microglial cells | Down regulation of Nape-pld | [50] | |
| Extra-virgin olive oil (EVOO) | ↓ DNA methylation of CNR1 promoter ↓ miR23A ↓miR-301a | Short- and long-term dietary EVOO rats and human colon cancer (CaCo-2) cells | Increased expression of the CB1 and reduced proliferation of colorectal cancer cells | [51] | |
| Maternal high-fat diet (HFD) | ↑ histone acetylation rate | Rat hypothalamus | Chromatin remodelling and increased binding of androgen receptor at CNR1 promoter leading to over expression of CB1 | [52] | |
| Drugs | Dex | miR-665 | Rat heart Langendorff preparation | Heart protective effect against ischemia/reperfusion injury via regulation of AK1 and CNR2 | [53] |
| Epigenetic modifiers (trichostatin A and 5-aza-2′-deoxycytidine) | Differential de novo expression of CB1, CB2 and μ-opioid receptors | Human SH SY5Y neuroblastoma cells and human Jurkat T lymphocytes | Selective de novo induction of CB1, CB2, and μ-opioid receptors depending on cell type | [54] | |
| 17β estradiol | ↓ H3K27 ↑ H3 and H4 acetylation | SW620 and DLD1 human colon cancer cells | Increased expression of CB1 by enhancing the binding of oestrogen receptor (ER) α and ERβ to CNR1 depending on cell type | [55] | |
| Disease | Alzheimer’s disease | ↓ DNA methylation at FAAH gene | Peripheral blood mononuclear cells (PBMCs) from subjects with late-onset Alzheimer disease (LOAD) (n = 33) and healthy controls (n = 33) | FAAH protein, and activity increased in PBMCs of LOAD subjects | [56] |
| Glioblastoma | Differential DNA methylation | Tumour samples (n = 55) and non-neoplastic brain tissues (n = 5) for methylation analyses; tumor samples (n = 40) and control (n = 3) for gene expression | Under expression of FAAH with hypermethylated promoter | [57] | |
| CRC | ↑ DNA methylation of CNR1 at CpGs located from –755 to +268 ↑ GPR55 demethylation | Human CRC tissues (n = 566) | GPR55 is highly expressed in CRC patients while CB1 levels are reduced | [58] | |
| ncRNA | miR-1273g-3p | ↓ CB1 | Human colorectal cancer LoVo cell lines | Promotion of proliferation, migration, and invasion | [59] |
| miR-29a | ↓CB1 ↑PPAR-γ | Gain-of-function transgenic mice | Block of the expressions of proinflammatory and profibrogenic mediators; attenuation of renal hypertrophy | [60] | |
| miR-494 | CB1 | Myocardial biopsy specimens (n = 12 chronic heart failure (CHF) and n = 4 healthy controls) | In CHF miR-494 is slightly increased leading to a compensatory response of the diseased myocardium. | [61] | |
| miR-665 | CB2 | Myocardial biopsy specimens (n = 12 CHF and n = 4 healthy controls) | In CHF miR-665 expression is significantly decreased leading to a compensatory response of the diseased myocardium. | [61] | |
| hsa-miR-29b-3p | CB1 | Paediatric low-grade gliomas (P-LGG) (n = 33) and control brains (n = 6) | Spontaneous involution of P-LGG may be induced by endocannabinoids | [62] | |
| CB1 hsa-let-7d | ↑ hsa-let-7d and other miRNA ↓CB1 | Various in vitro and in vivo systems | CB1 receptor up-regulates let-7d, which, in turn, impairs CB1 receptor signalling and cannabinoid-opioid cross-signalling. | [63] | |
| AntagoMir-411 | ↓ miR-411 ↑FAAH ↑Pparδ ↑glutamate receptor AMPA-2 | Prefrontal cortex of female C57BL/6J mice | Reversion of alcohol-related neuro-adaptations and reduction of chronic alcohol consumption | [64] | |
| Substances | Epigenetic Modification | Experimental Model and Subjects (n) | Results | References |
|---|---|---|---|---|
| Phytocannabinoids | ||||
| Cannabis | Changes in DNA methylation | Blood from schizophrenia patients (n = 98) and healthy controls (n = 108) | Modulation of the immune response and protection against the neurobiological substrate of reality distortion symptoms in schizophrenia patients | [65] |
| Cannabidiol Cannabigerol | ↑ DNA methylation of keratin 10 gene | Human keratinocytes (HaCaT cells) | ↓keratin 10 mRNA through a CB1-dependent mechanism, whereas cannabigerol did not affect either CB1 or CB2 | [66] |
| Cannabidiol | ↑Global DNA methylation levels ↑DNMT1 expression No effect on DNMT 3a, 3b, or 3L | HaCaT cells | Modulation of gene repression | [66] |
| Δ9THC | Changes in DNA methylation profile | Non-human primates, brain (lateral cerebellum) during simian immunodeficiency virus infection | Altered gene expression | [67] |
| Δ9THC | Dose-dependent increase of HDAC3 expression | Human BeWo trophoblast cell line | Inhibition of proliferation | [68] |
| Δ9THC | ↓H3K9me3 ↓H3K4me3 in the nucleus accumbens shell | Adolescent rats | Proenkephalin (Penk) upregulation in the adult and opiate vulnerability | [69] |
| Δ9THC | ↑H3K9m2 ↓H3K4m3 | Rats, prenatal exposure | Decreased dopamine receptor D2 (Drd2) RNA expression in the ventral striatum (nucleus accumbens) in adult animals | [70] |
| Δ9THC | Histone modifications (H3K9me2, H3K9me3, H3K27me3, H3K9ac and H3K14ac) | Adolescent and adult brain areas (hippocampus, amygdala and nucleus accumbens) of female rats | Region- and age-specific histone modifications leading to transcriptional repression in the adolescence and transcriptional activation in the adults | [71] |
| Δ9THC | Histone modifications, mainly H3K9me3 | Adolescent female rats, prefrontal cortex | Increased expression of the histone-lysine N-methyltransferase SUV39H1 Cognitive deficit | [72] |
| Δ9THC | Histone modifications (H3K4me3, H3K9me3, H3K27me3, H3K36me3 and H3K9ac) | Differentiating lymph node cells of mice immunized with a superantigen, staphylococcal enterotoxin B | Alterations in antigen-specific T cell response due to altered gene expression | [73] |
| Δ9THC | ↓ miR-17/92 cluster ↓ miR-374b/421 cluster ↑ miR-146 ↑ LncRNAs expressed from the opposite strand of CD27 and Appbp2 | Mouse super antigen-activated lymph node cells and CD4+ T cells | Altered transcripts mainly related to immune response and cell proliferation | [74] |
| Δ9THC | Not Assayed | Long-Evans rats with parental Δ9THC exposure | Deregulated mRNA levels (i.e., CNR1, glutamate and dopamine-related genes) in the striatum of adolescent and adult F1 offspring; behavioural and neurobiological abnormalities in the F1 offspring | [75] |
| Δ9THC | Up-down regulation of several miRNAs | Non-human primates, CD4+ T cells, during simian immunodeficiency virus infection | Immunomodulatory role for cannabinoids | [67] |
| Δ9THC | Modulation of miRNAs, including ↑miRNA-690 and its target Transcription factor CCAAT/enhancer-binding protein α | Mouse myeloid-derived suppressor cells | Altered myeloid expansion and differentiation | [76] |
| Δ9THC | Up/down regulation of several miRNAs like ↑miR-10a, ↑miR-24, ↑miR-99b, ↑miR-145, ↑miR-149, ↑miR-187 | Intestine of simian immunodeficiency virus infected macaques | Altered miRNA profile and changes in anti-inflammatory response | [77] |
| Δ9THC | DNA methylation in sperm | Paternal exposure of rats | Long-lasting neurobehavioral effects in the offspring | [78] |
| Δ9THC | 1027 differentially methylated regions in F1 adults | Paternal exposure of rats | Cross-generational epigenomic alterations in the rat nucleus accumbens, including differentially methylated regions localized to genes with important roles in neural function, complex psychiatric diseases, and addiction-related traits | [79] |
| Δ9THC | Global DNA methylation | Human (n= 24 including 12 cannabis smokers and 12 cannabis non-smokers) and rat sperm | Changes in DNA sperm methylome, with altered CpG sites associated with genes involved in Hippo signalling and cancer pathways | [80] |
| Δ9THC/Cannabis | DNA methylation | Humans (n= 24 including 12 cannabis smokers and 12 cannabis non-smokers) and paternal exposure of rats | Changes in the methylation of the autism candidate gene DLGAP2 in human and rat sperm and in the nucleus accumbens of the offspring of the Δ9THC exposed rats | [81] |
| Endocannabinoids and ES synthetic agonists and antagonists | ||||
| ACEA JWH-133 AM-281 AM-630 | ↓miR-23a ↓miR-24, ↓miR-181a ↓miR-320a | Human granulosa cell line KGN | Modulating role of the intrinsic ovarian ES in the regulation of oestradiol synthesis and alteration in miRNA profile following CB1 manipulation only | [82] |
| ACEA SR141716A | Enhanced expression levels of HDACs- especially HDAC5- which binds CNR1promoter | Embryonic mouse hypothalamus N1 cell line and mouse hypothalamus | Attenuated or enhanced central adiponectin (APN) induction of bone formation | [83] |
| ACPA SR141716A | Modulation of HDAC activity | Mouse | Combined involvement of histone acetylation and ES system in anxiety- and depression-related behaviours | [84] |
| AEA | Increased DNA methylation and DNMT activity | HaCaT cells | Inhibition of differentiation | [85] |
| AEA | 609 miRNA differentially regulated | Methylated bovine serum albumin-induced delayed type hypersensitivity response in C57BL/6 mice, mediated by Th17 cells | Altered interleukin production and inflammatory response | [86] |
| AM-251 | Restoration of H3K9ac at control levels | Hippocampus of schizophrenia like animals | ES-dependent epigenetic mechanisms involved in both embryonic brain development and neuro-differentiation as well as in the pathophysiology of a schizophrenia like phenotype | [87] |
| AM-251 | ↑miR-30e-5p ↓DLL4 in adipose tissue macrophage | F4/80+ cells from stromal vascular fractions of epididymal fat collected from DIO mice fed HFD | Suppression of DLL4-Notch signalling-induced polarization of inflammatory Th1 cells and adipocyte energy storage with anti-inflammatory state and attenuation of DIO phenotype | [88] |
| FAAH-II | Up-down regulation of several miRNAs, including imprinted Dlk1-Dio3 miRNA cluster | Mouse mesenteric lymph nodes and Peyer’s patches | Suppression of colitis through regulation of pro-inflammatory miRNA expression | [89] |
| HU-210 | Differential miRNA expression | Postnatal day 35 rats | Significant differences in the expression of miRNA in the left hemisphere of the entorhinal cortex, in a manner that is relevant to schizophrenia | [90] |
| HU-210 JWH-133 | ↑H3K9me3 | Glioma cell lines U87MG and U373MG expressing CBs | Induction of differentiation, inhibition of gliomagenesis | [91] |
| JWH-133 | ↑H3K4m3 ↓H3K9m2 | Mouse spermatogonia in vitro | Increased expression of the meiotic genes c-Kit and Stra8 with accelerated meiosis entry | [92] |
| JWH-133 | Altered DNA methylation and hydroxymethylation at specific imprinted genes in sperm and placenta | Paternal exposure in mouse | Reduced sperm count in exposed animals and defects in placental and embryonic development | [93] |
| SR141716A | ↓ miR-466 family ↓ miR-762 and other | DIO mice fed HFD | Attenuation of DIO-associated inflammation | [94] |
| WIN55,212-2 | Increased expression of DNMTs and DNA methylation in prefrontal cortex | Paternal exposure in rats during adolescence | Increased vulnerability to stress in the offspring | [95] |
| WIN55,212-2 | DNA hypermethylation at the intragenic region of the intracellular signalling modulator Rgs7 | Adolescent mice | Reduced expression of Rgs7 in the hippocampus and memory impairment in adult mice | [96] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meccariello, R.; Santoro, A.; D'Angelo, S.; Morrone, R.; Fasano, S.; Viggiano, A.; Pierantoni, R. The Epigenetics of the Endocannabinoid System. Int. J. Mol. Sci. 2020, 21, 1113. https://doi.org/10.3390/ijms21031113
Meccariello R, Santoro A, D'Angelo S, Morrone R, Fasano S, Viggiano A, Pierantoni R. The Epigenetics of the Endocannabinoid System. International Journal of Molecular Sciences. 2020; 21(3):1113. https://doi.org/10.3390/ijms21031113
Chicago/Turabian StyleMeccariello, Rosaria, Antonietta Santoro, Stefania D'Angelo, Rossella Morrone, Silvia Fasano, Andrea Viggiano, and Riccardo Pierantoni. 2020. "The Epigenetics of the Endocannabinoid System" International Journal of Molecular Sciences 21, no. 3: 1113. https://doi.org/10.3390/ijms21031113
APA StyleMeccariello, R., Santoro, A., D'Angelo, S., Morrone, R., Fasano, S., Viggiano, A., & Pierantoni, R. (2020). The Epigenetics of the Endocannabinoid System. International Journal of Molecular Sciences, 21(3), 1113. https://doi.org/10.3390/ijms21031113







