Beyond Microcystins: Cyanobacterial Extracts Induce Cytoskeletal Alterations in Rice Root Cells
Abstract
1. Introduction
2. Results
2.1. Enzyme Inhibition
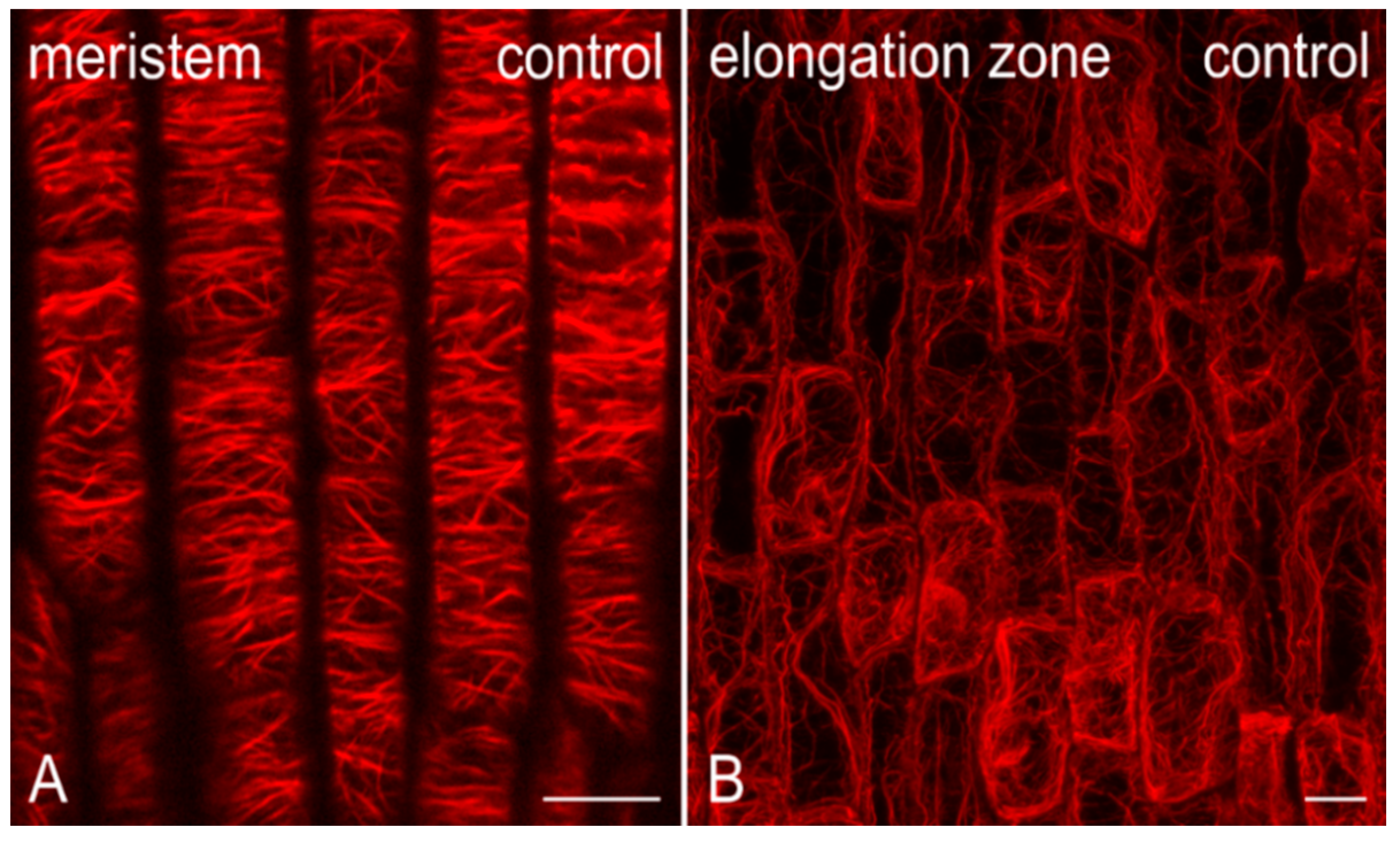
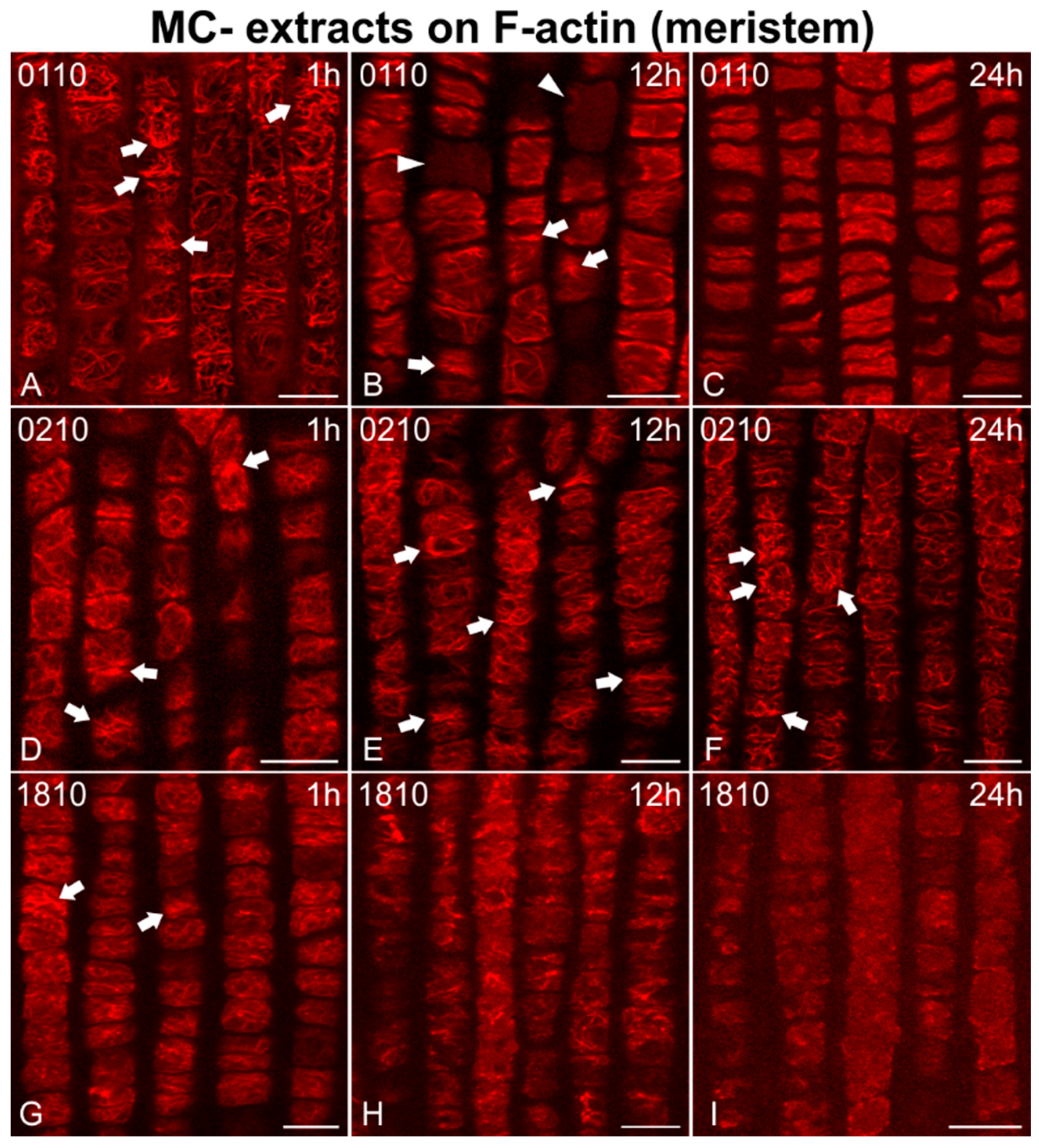
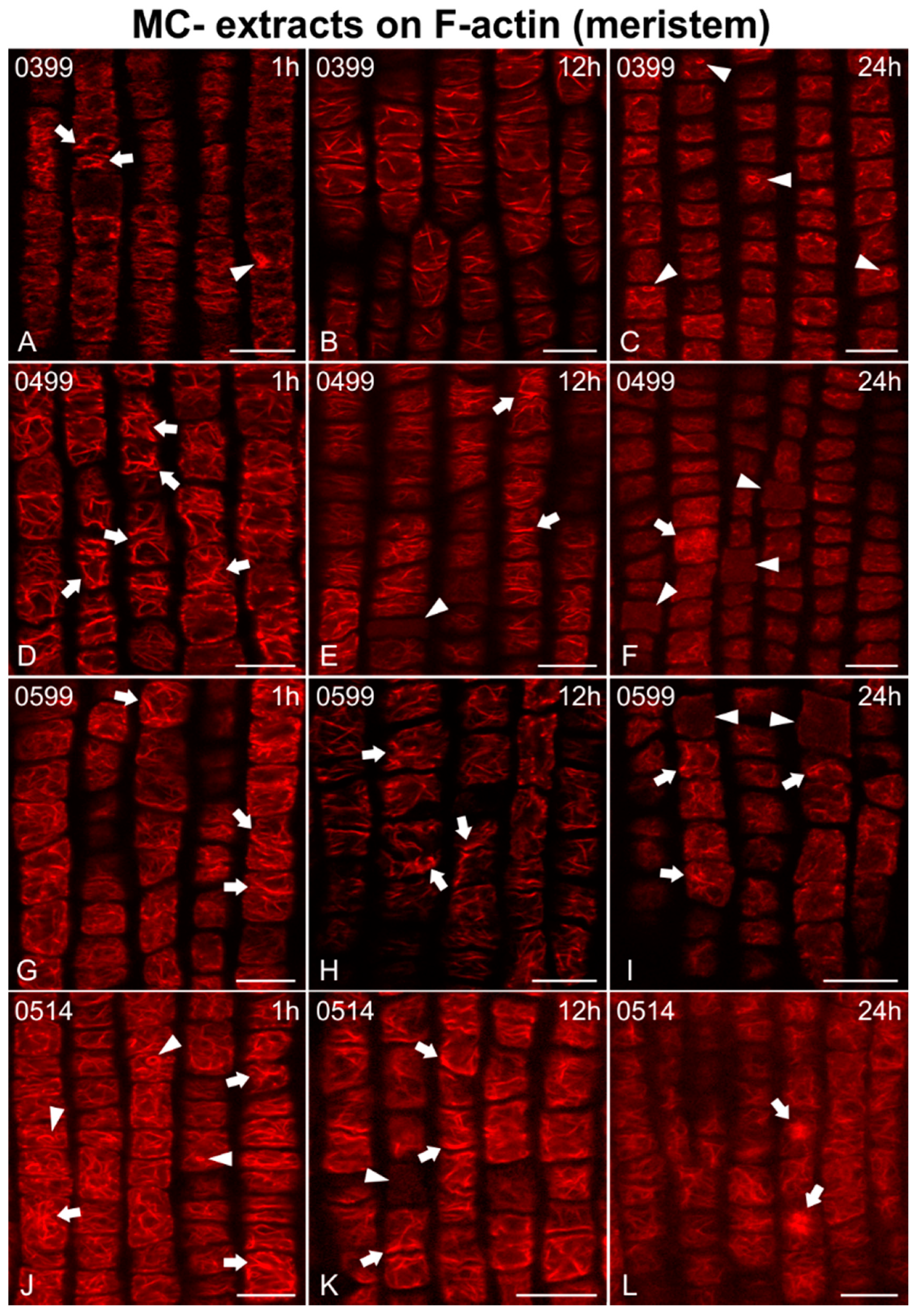
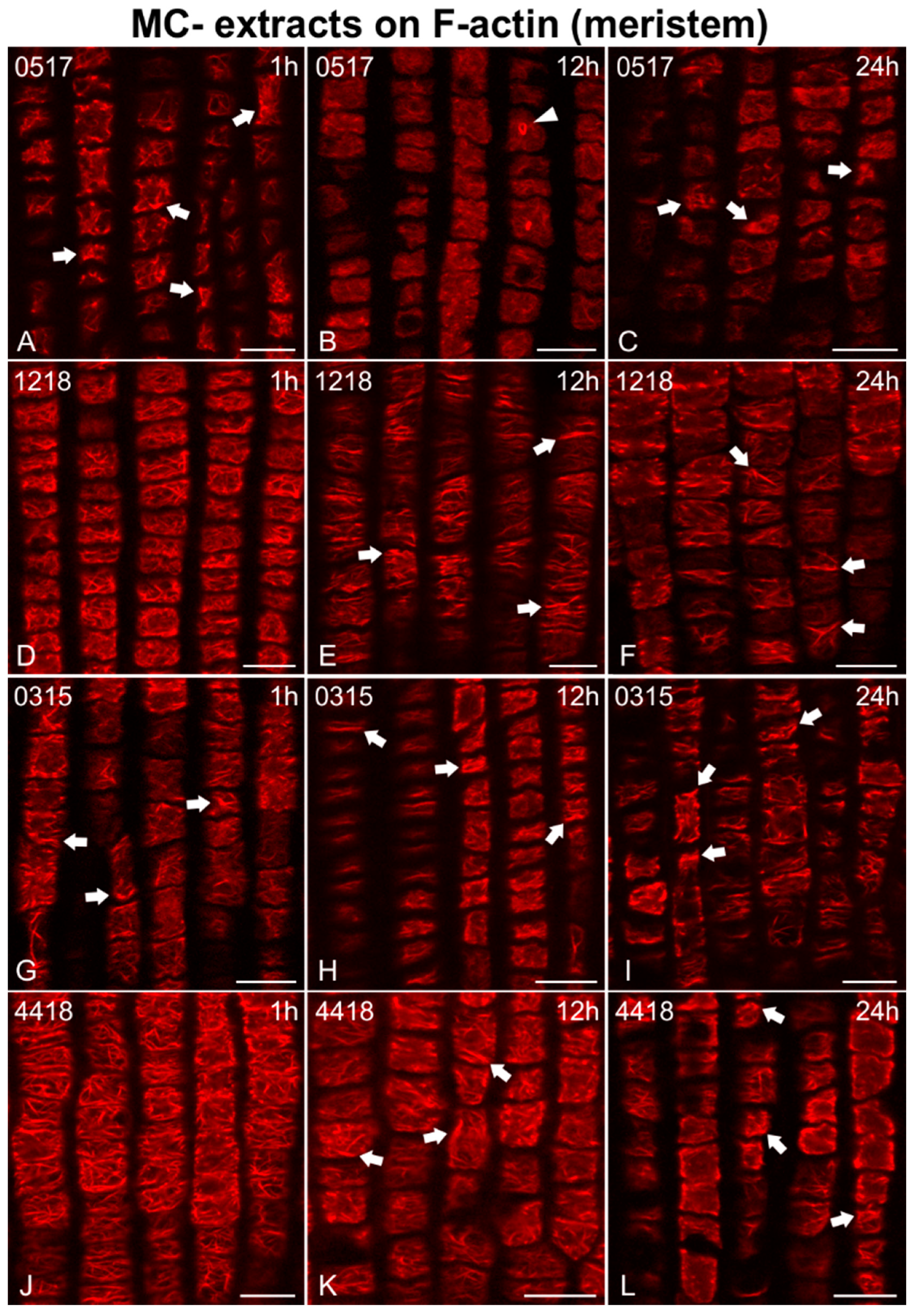
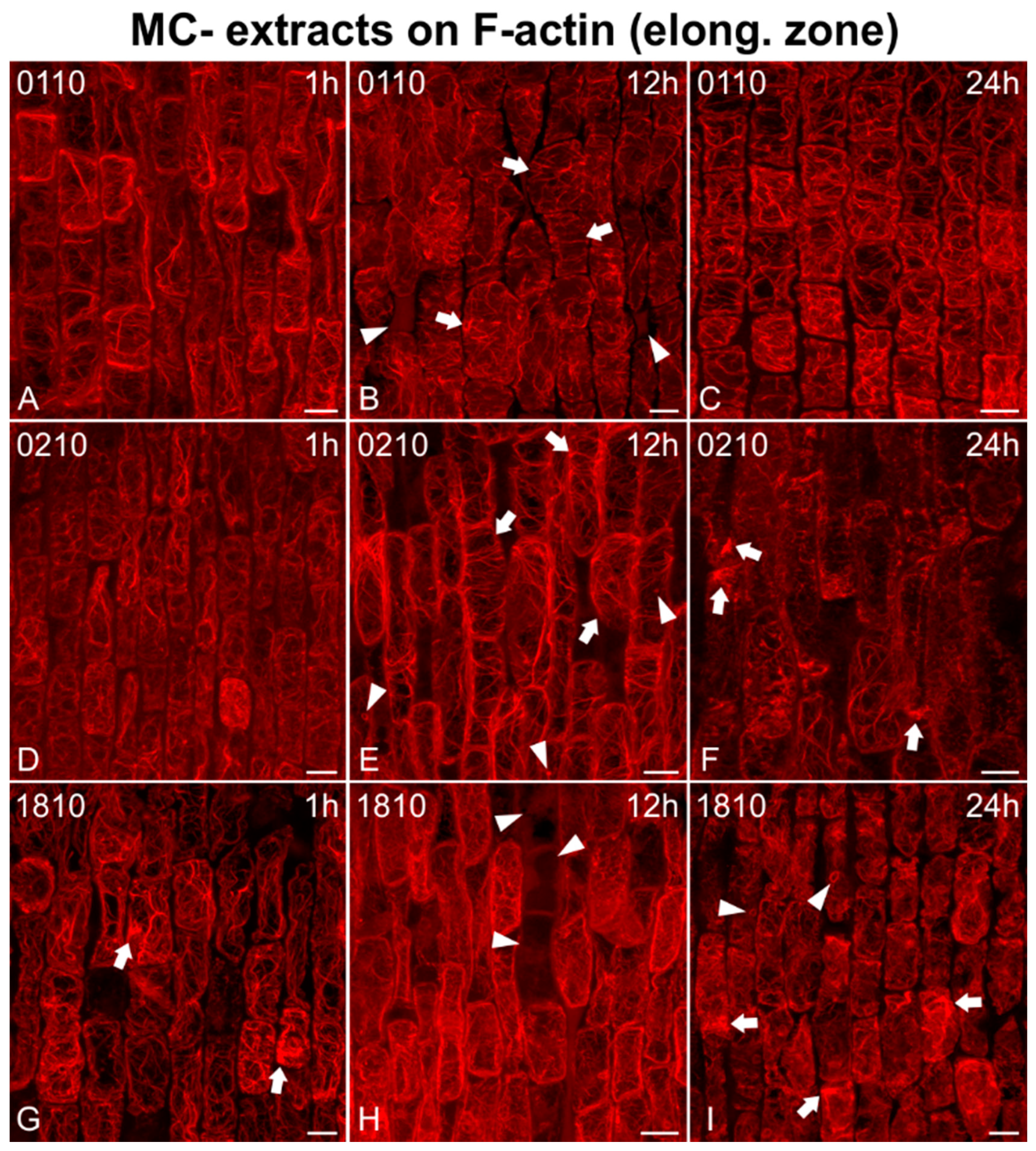
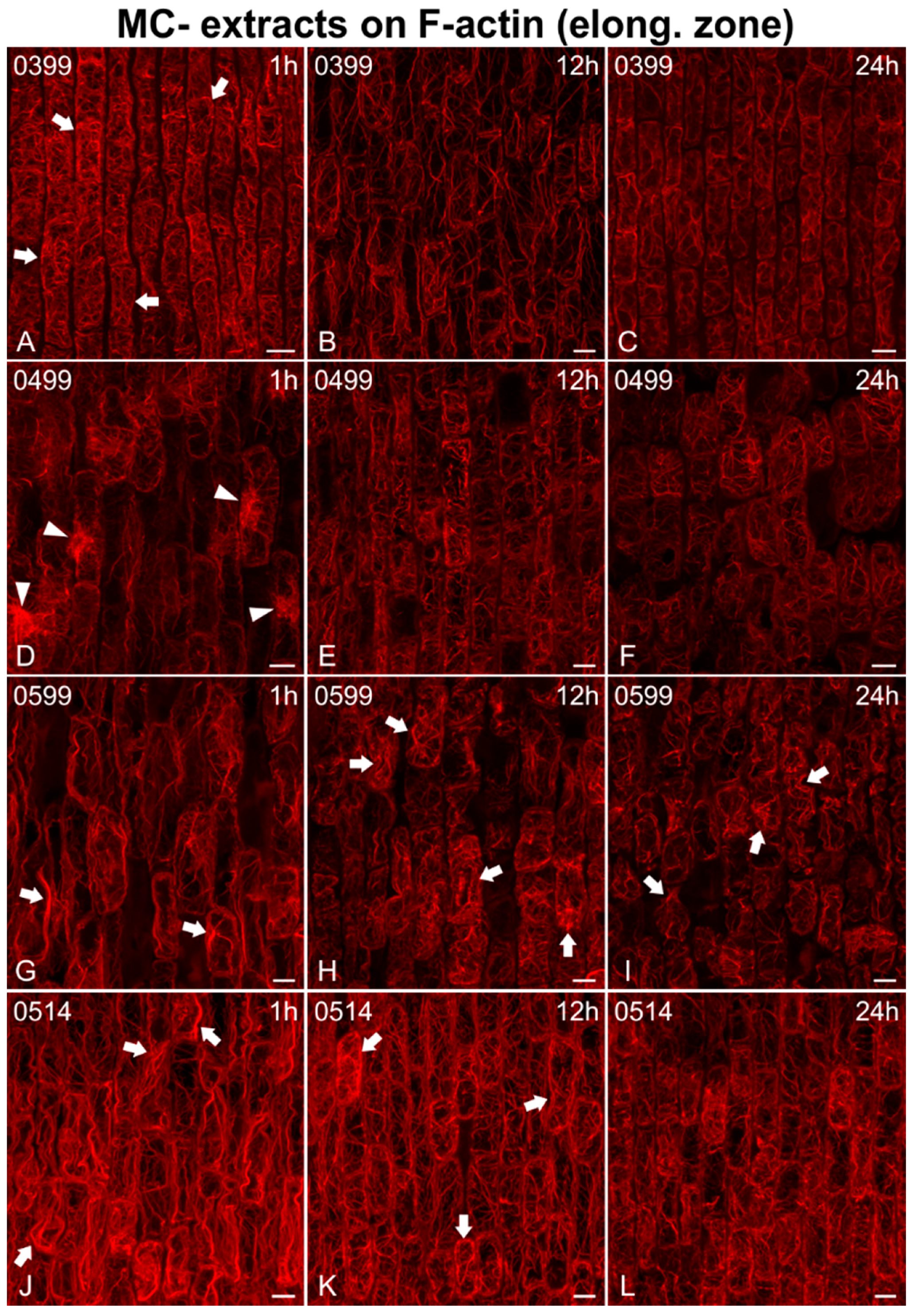
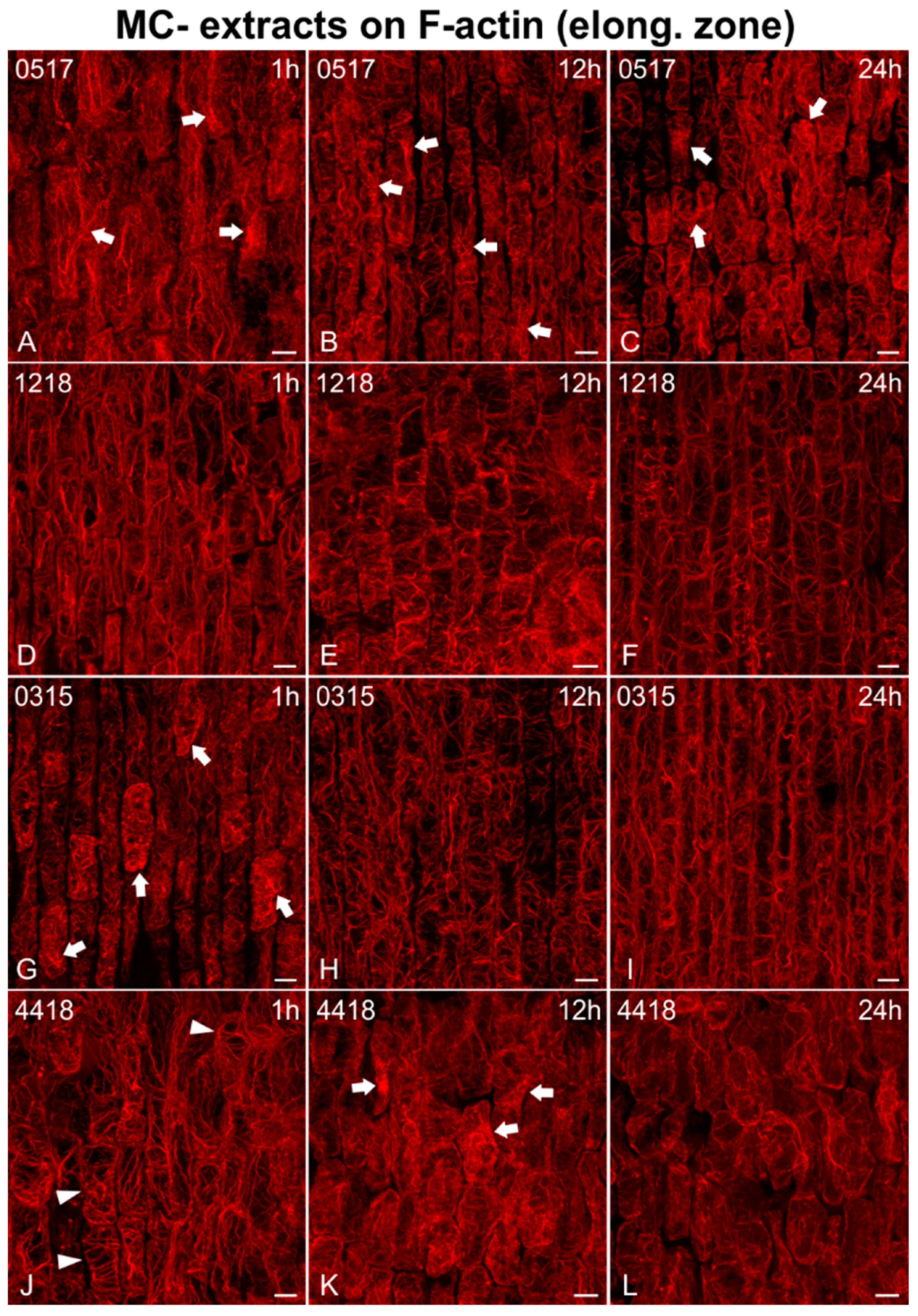
2.2. Effects on F-actin
2.2.1. Morphological Alterations
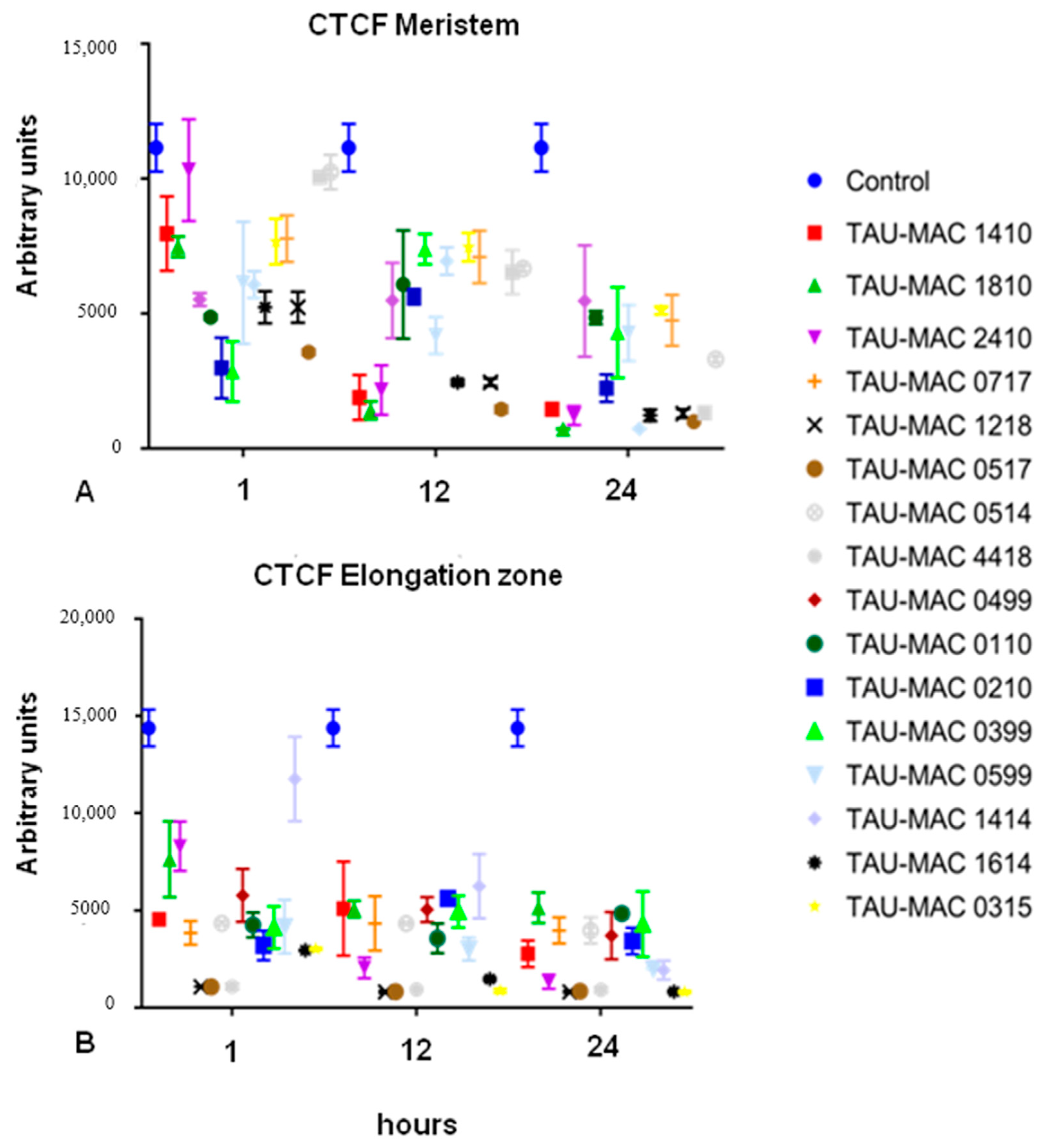
2.2.2. F-actin Fluorescence Intensity Measurements
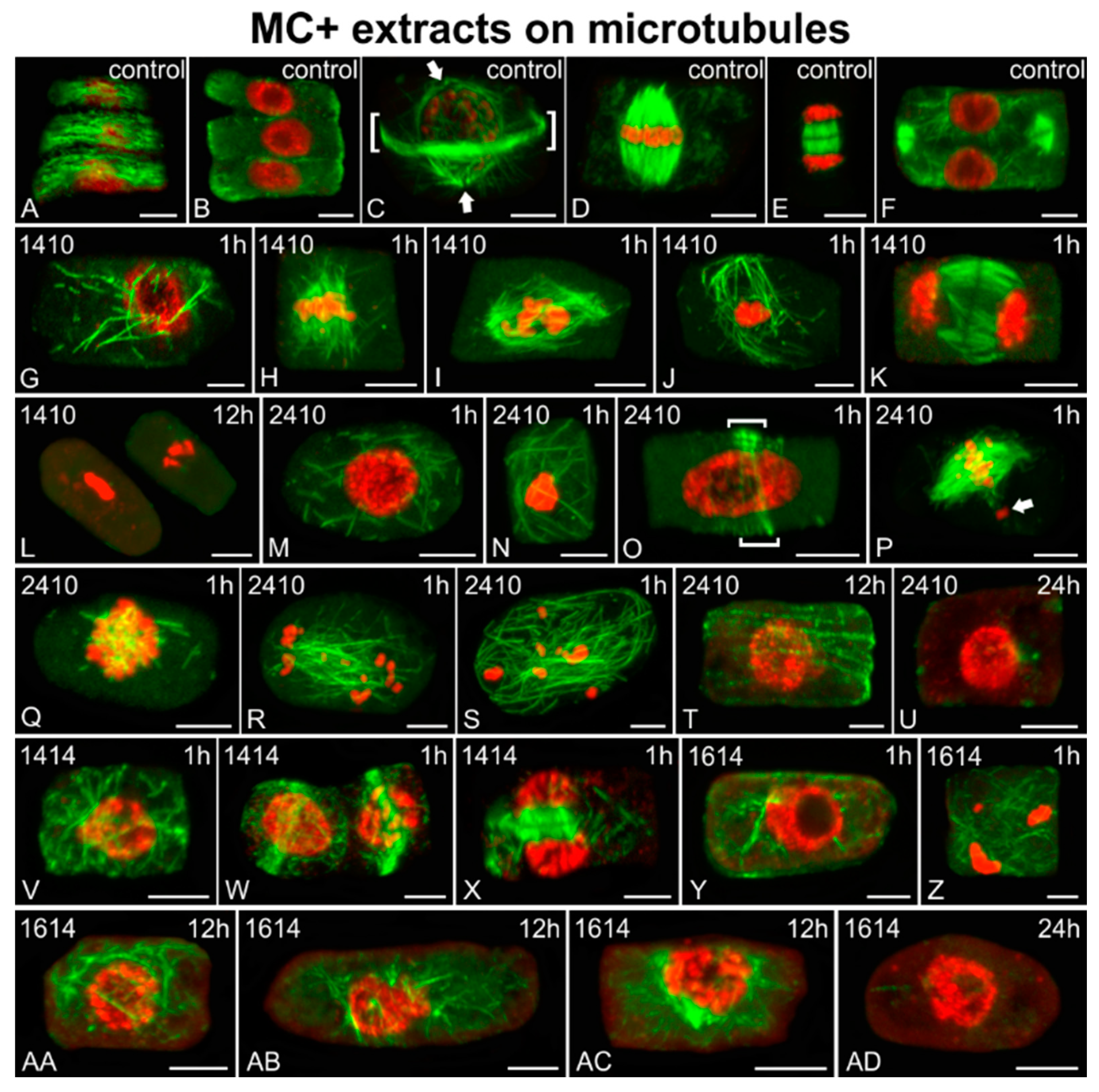
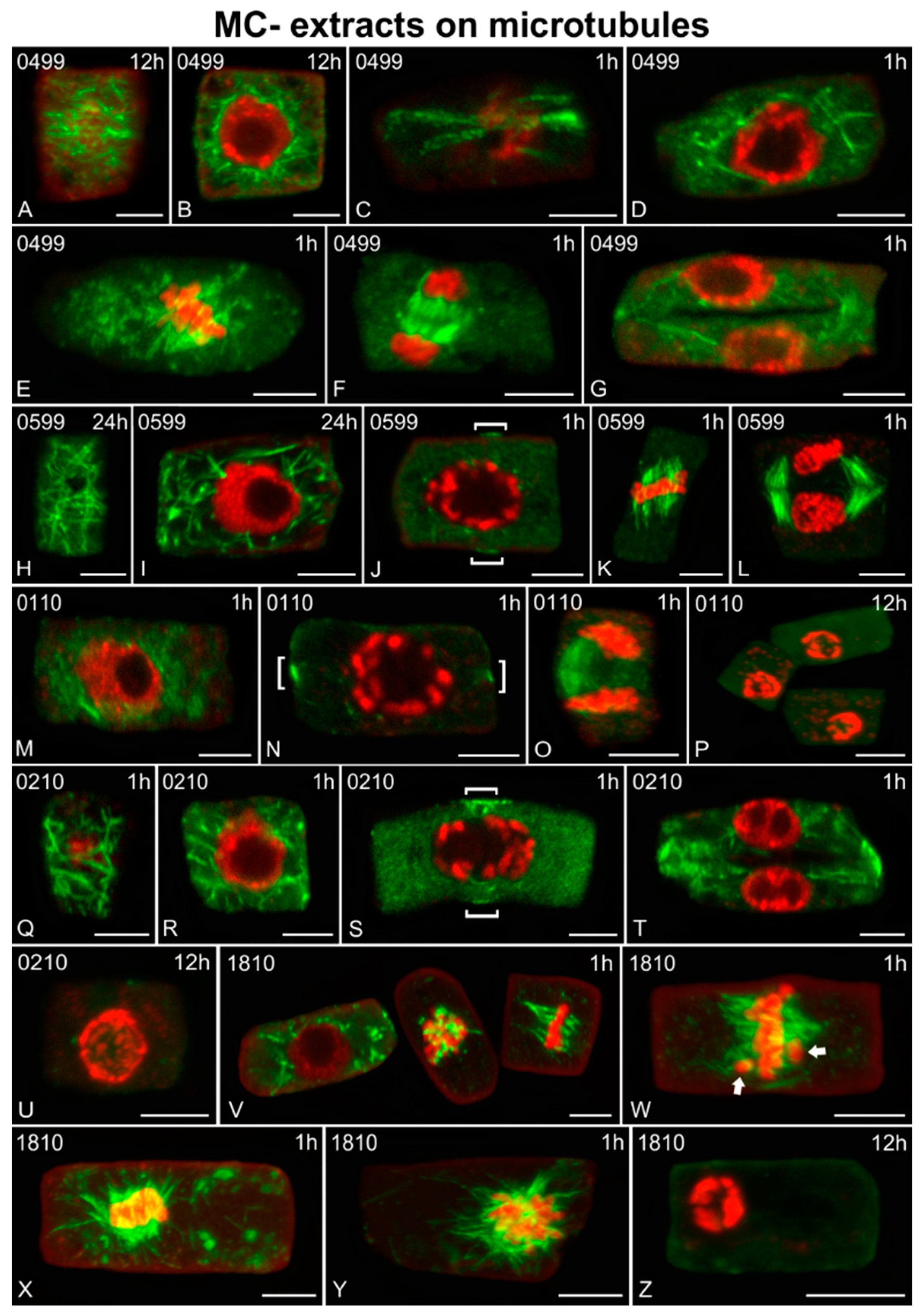
2.3. Effects on Microtubules and Chromatin
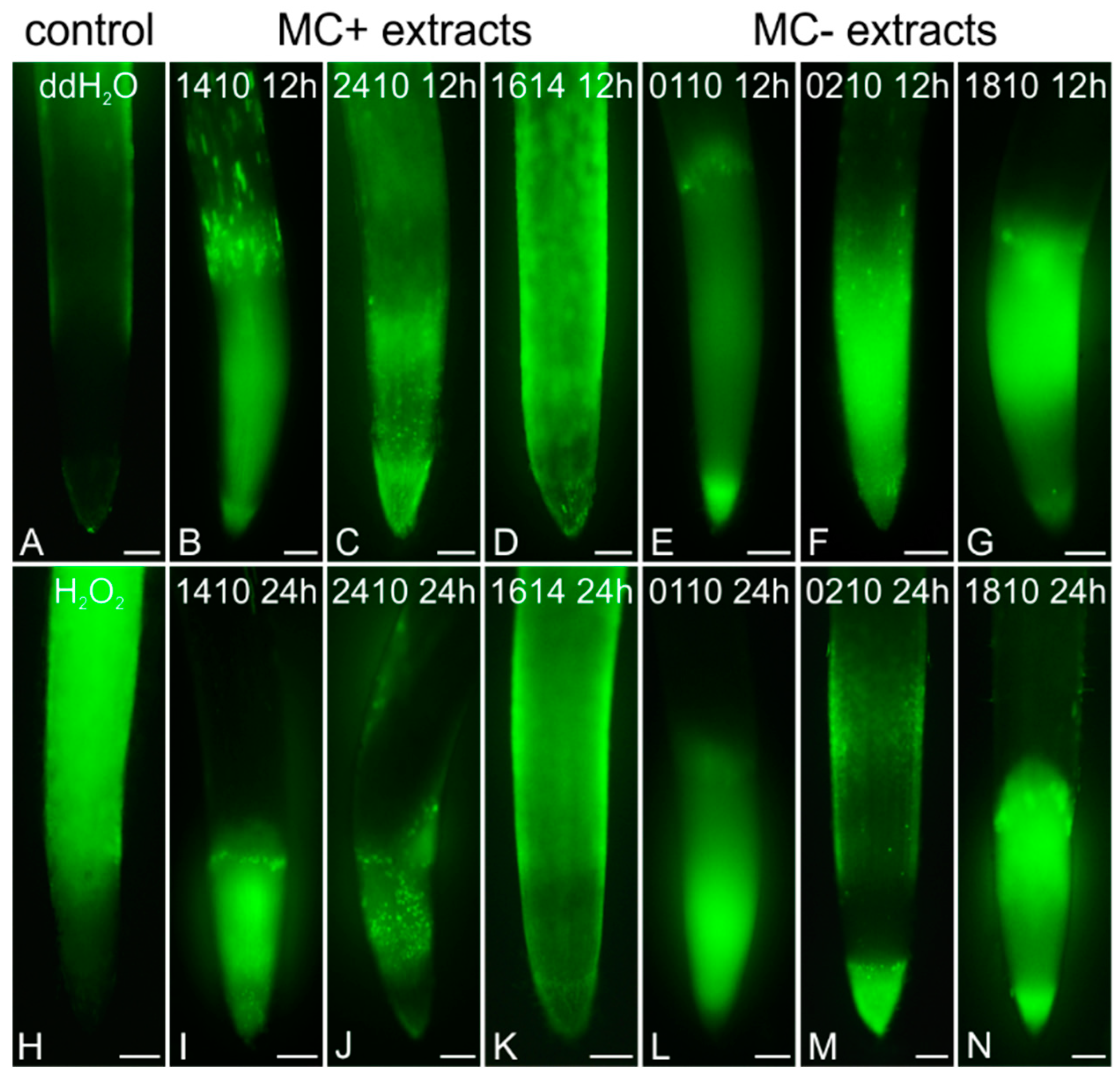
2.4. Induction of Oxidative Stress
3. Discussion
4. Materials and Methods
4.1. Culture of Cyanobacteria, Biomass Collection, and Preparation of Extracts
| Taxonomic Order | Strain | Habitat | Lifestyle | Detection of MCs | References |
|---|---|---|---|---|---|
| Chroococcales | Microcystis flos-aquae TAU-MAC 1410 | Freshwater | Planktic | + | [29,59] |
| Microcystis viridis TAU-MAC 1810 | Freshwater | Planktic | − | [29,59] | |
| Microcystis sp. TAU-MAC 2410 | Freshwater | Planktic | + | [29,59] | |
| Synechococcales | Synechococcus sp. TAU-MAC 0499 | Freshwater | Planktic | − | [29,59] |
| Jaaginema sp. TAU-MAC 0110 | Freshwater | Planktic | − | [29,59] | |
| Jaaginema sp. TAU-MAC 0210 | Freshwater | Planktic | − | [29,59] | |
| Nostocales | Calothrix epiphytica TAU-MAC 0399 | Freshwater | Epiphytic | − | [29,59] |
| Chlorogloeopsis fritschii TAU-MAC 0599 | Freshwater | Epiphytic | − | [29,59] | |
| Raphidiopsis raciborskii TAU-MAC 1414 | Freshwater | Planktic | + 1 | [59,60] | |
| Trichormus variabilis TAU-MAC 1614 | Freshwater | Planktic | + | Unpublished data | |
| Nostoc oryzae TAU-MAC 0315 | Freshwater | Epiphytic | − | Unpublished data | |
| Nodularia sp. TAU-MAC 0717 | Brackish | Planktic | − | Unpublished data | |
| Scytonema sp. TAU-MAC 1218 | Terrestrial cave | Epilithic | − | Unpublished data | |
| Oscillatoriales | Planktothrix agardhii TAU-MAC 0514 | Freshwater | Planktic | − | [29,59] |
| Phormidium sp. TAU-MAC 0517 | Terrestrial cave | Epilithic | − | Unpublished data | |
| Lyngbya sp. TAU-MAC 4418 | Intertidal zone | Epilithic | − | Unpublished data |
4.2. Enzyme Inhibition Assays
4.2.1. Protein Phosphatase Inhibition Assay
4.2.2. Trypsin Inhibition Assay
4.2.3. Elastase Inhibition Assay
4.3. Plant Material and Treatments
4.4. Cytoskeletal Studies
4.4.1. Tubulin Immunolabeling
4.4.2. F-actin Labeling
4.4.3. Confocal Fluorescence Microscopy
4.4.4. Fluorescence Intensity Measurements
4.5. Detection of Hydrogen Peroxide Production
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CLSM | Confocal laser scanning microscope |
| CTCF | Corrected total cell fluorescence |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DCF-DA | 2,7-dichlorofluorescein diacetate |
| DMSO | Dimethyl sulfoxide |
| LC–MS/MS | Liquid chromatography with tandem mass spectrometry |
| MC | Microcystin |
| MC+ | Microcystin-rich |
| MC− | Microcystin-devoid |
| PBS | Phosphate buffer saline |
| PEM | PIPES EGTA MgSO4 |
| PFA | Paraformaldehyde |
| PPB | Preprophase band |
| PP1 | Protein phosphatase 1 |
| PP2A | Protein phosphatase 2A |
| ROS | Reactive oxygen species |
Appendix A
| Taxonomic Order | Strain | Culture Medium | References |
|---|---|---|---|
| Chroococcales | Microcystis flos-aquae TAU-MAC 1410 | BG-11 | [61,69] |
| Microcystis viridis TAU-MAC 1810 | BG-11 | ||
| Microcystis sp. TAU-MAC 2410 | BG-11 | ||
| Synechococcales | Synechococcus sp. TAU-MAC 0499 | BG-11 | |
| Jaaginema sp. TAU-MAC 0110 | BG-11 | ||
| Jaaginema sp. TAU-MAC 0210 | BG-11 | ||
| Nostocales | Calothrix epiphytica TAU-MAC 0399 | BG-110 | |
| Chlorogloeopsis fritschii TAU-MAC 0599 | BG-110 | ||
| Raphidiopsis raciborskii TAU-MAC 1414 | BG-110 | ||
| Trichormus variabilis TAU-MAC 1614 | BG-110 | ||
| Nostoc oryzae TAU-MAC 0315 | BG-110 | ||
| Nodularia sp. TAU-MAC 0717 | MN0 | ||
| Scytonema sp. TAU-MAC 1218 | BG-110 | ||
| Oscillatoriales | Planktothrix agardhii TAU-MAC 0514 | BG-11 | |
| Phormidium sp. TAU-MAC 0517 | BG-11 | ||
| Lyngbya sp. TAU-MAC 4418 | MN |
References
- Codd, G.A.; Meriluoto, J.; Metcalf, J.S. Introduction: Cyanobacteria, Cyanotoxins, Their Human Impact, and Risk Management. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; John Wiley & Sons: Chichester, West Sussex, UK, 2017; pp. 3–8. [Google Scholar]
- Huang, I.S.; Zimba, P.V. Cyanobacterial bioactive metabolites—A review of their chemistry and biology. Harmful Algae 2019, 83, 42–94. [Google Scholar] [CrossRef]
- Demay, J.; Bernard, C.; Reinhardt, A.; Marie, B. Natural products from cyanobacteria: Focus on Beneficial Activities. Mar. Drugs 2019, 17, 320. [Google Scholar] [CrossRef]
- Campos, A.; Vasconcelos, V. Molecular mechanisms of microcystin toxicity in animal cells. Int. J. Mol. Sci. 2010, 11, 268–287. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Tu, W.; Xu, J. Mechanisms of microcystin-LR-induced cytoskeletal disruption in animal cells. Toxicon 2015, 101, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Zilliges, Y.; Kehr, J.-C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef] [PubMed]
- MacKintosh, C.; Beattie, K.A.; Klumpp, S.; Cohen, P.; Codd, G.A. Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett. 1990, 264, 187–192. [Google Scholar] [CrossRef]
- Honkanen, R.E.; Zwiller, J.; Mooren, R.E.; Daily, S.L.; Khatra, B.S.; Dukelow, M.; Boynton, A.L. Characterization of microcystin- LR, a potent inhibitor of type 1 and type 2A protein phosphatases. J. Biol. Chem. 1990, 265, 19401–19404. [Google Scholar]
- Moura, M.; Conde, C. Phosphatases in mitosis: Roles and regulation. Biomolecules 2019, 9, 55. [Google Scholar] [CrossRef]
- Hoffman, A.; Taleski, G.; Sontag, E. The protein serine/threonine phosphatases PP2A, PP1 and calcineurin: A triple threat in the regulation of the neuronal cytoskeleton. Mol. Cell. Neurosci. 2017, 84, 119–131. [Google Scholar] [CrossRef]
- Machado, J.; Campos, A.; Vasconcelos, V.; Freitas, M. Effects of microcystin-LR and cylindrospermopsin on plant-soil systems: A review of their relevance for agricultural plant quality and public health. Environ. Res. 2017, 153, 191–204. [Google Scholar] [CrossRef]
- Máthé, C.; M-Hamvas, M.; Vasas, G. Microcystin-LR and cylindrospermopsin induced alterations in chromatin organization of plant cells. Mar. Drugs 2013, 11, 3689–3717. [Google Scholar] [CrossRef]
- Pappas, D.; Gkelis, S.; Panteris, E. The effects of microcystin-LR in Oryza sativa root cells: F-actin as a new target of cyanobacterial toxicity. Plant Biol. 2020, 22, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, C.; Azimzadeh, J.; Pastuglia, M.; Bellini, C.; Grandjean, O.; Bouchez, D. The Arabidopsis TONNEAU 2 gene encodes a putative novel protein phosphatase 2A regulatory subunit essential for the control of the cortical cytoskeleton. Plant Cell 2002, 14, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Wen, F.; Wang, J.; Xing, D. A protein phosphatase 2A catalytic subunit modulates blue light-induced chloroplast avoidance movements through regulating actin cytoskeleton in Arabidopsis. Plant Cell Physiol. 2012, 53, 1366–1379. [Google Scholar] [CrossRef]
- Samofalova, D.O.; Karpov, P.A.; Raevsky, A.V.; Blume, Y.B. Protein phosphatases potentially associated with regulation of microtubules, their spatial structure reconstruction and analysis. Cell Biol. Int. 2019, 43, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Pflugmacher, S.; Jung, K.; Lundvall, L.; Neumann, S.; Peuthert, A. Effects of cyanobacterial toxins and cyanobacterial cell-free crude extract on germination of alfalfa (Medicago sativa) and induction of oxidative stress. Environ. Toxicol. Chem. 2006, 25, 2381–2387. [Google Scholar] [CrossRef] [PubMed]
- Saqrane, S.; El Ghazali, I.; Ouahid, Y.; El Hassni, M.; El Hadrami, I.; Bouarab, L.; del Campo, F.F.; Oudra, B.; Vasconcelos, V. Phytotoxic effects of cyanobacteria extract on the aquatic plant Lemna gibba: Microcystin accumulation, detoxication and oxidative stress induction. Aquat. Toxicol. 2007, 83, 284–294. [Google Scholar] [CrossRef]
- Prieto, A.; Campos, A.; Cameán, A.; Vasconcelos, V. Effects on growth and oxidative stress status of rice plants (Oryza sativa) exposed to two extracts of toxin-producing cyanobacteria (Aphanizomenon ovalisporum and Microcystis aeruginosa). Ecotoxicol. Environ. Saf. 2011, 74, 1973–1980. [Google Scholar] [CrossRef]
- Quan, L.J.; Zhang, B.; Shi, W.W.; Li, H.Y. Hydrogen peroxide in plants: A versatile molecule of the reactive oxygen species network. J. Integr. Plant Biol. 2008, 50, 2–18. [Google Scholar] [CrossRef]
- Pflugmacher, S. Promotion of oxidative stress in the aquatic macrophyte Ceratophyllum demersum during biotransformation of the cyanobacterial toxin microcystin-LR. Aquat. Toxicol. 2004, 70, 169–178. [Google Scholar] [CrossRef]
- Chen, J.; Han, F.X.; Wang, F.; Zhang, H.; Shi, Z. Accumulation and phytotoxicity of microcystin-LR in rice (Oryza sativa). Ecotoxicol. Environ. Saf. 2012, 76, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Huang, J.; Huang, W.; Li, D.; Wang, G.; Liu, Y. Microcystin-RR-induced accumulation of reactive oxygen species and alteration of antioxidant systems in tobacco BY-2 cells. Toxicon 2005, 46, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Gu, X.; Song, R.; Wang, X.; Yang, L. Microcystin-LR induced oxidative stress and ultrastructural alterations in mesophyll cells of submerged macrophyte Vallisneria natans (Lour.) Hara. J. Hazard. Mater. 2011, 190, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Xing, W.; Li, D.; Liu, Y. Microcystin-RR induced apoptosis in tobacco BY-2 suspension cells is mediated by reactive oxygen species and mitochondrial permeability transition pore status. Toxicol. Vitr. 2008, 22, 328–337. [Google Scholar] [CrossRef]
- Janssen, E.M.L. Cyanobacterial peptides beyond microcystins—A review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Zervou, S.K.; Gkelis, S.; Kaloudis, T.; Hiskia, A.; Mazur-Marzec, H. New microginins from cyanobacteria of Greek freshwaters. Chemosphere 2020, 248, 125961. [Google Scholar] [CrossRef]
- Gkelis, S.; Lanaras, T.; Sivonen, K. The presence of microcystins and other cyanobacterial bioactive peptides in aquatic fauna collected from Greek freshwaters. Aquat. Toxicol. 2006, 78, 32–41. [Google Scholar] [CrossRef]
- Gkelis, S.; Panou, M.; Konstantinou, D.; Apostolidis, P.; Kasampali, A.; Papadimitriou, S.; Kati, D.; Di Lorenzo, G.M.; Ioakeim, S.; Zervou, S.-K.; et al. Diversity, cyanotoxin production, and bioactivities of cyanobacteria isolated from freshwaters of Greece. Toxins 2019, 11, 436. [Google Scholar] [CrossRef]
- Chen, J.; Song, L.; Dai, J.; Gan, N.; Liu, Z. Effects of microcystins on the growth and the activity of superoxide dismutase and peroxidase of rape (Brassica napus L.) and rice (Oryza sativa L.). Toxicon 2004, 43, 393–400. [Google Scholar] [CrossRef]
- Pflugmacher, S.; Aulhorn, M.; Grimm, B. Influence of a cyanobacterial crude extract containing microcystin-LR on the physiology and antioxidative defence systems of different spinach variants. New Phytol. 2007, 175, 482–489. [Google Scholar] [CrossRef]
- Liang, C.; Liu, H. Response of hormone in rice seedlings to irrigation contaminated with cyanobacterial extract containing microcystins. Chemosphere 2020, 256, 127157. [Google Scholar] [CrossRef]
- Fairhurst, T.H.; Dobermann, A. Rice in the Global Food Supply. Better Crops Int. 2002, 16, 3–6. [Google Scholar]
- Peuthert, A.; Chakrabarti, S.; Pflugmacher, S. Uptake of microcystins-LR and -LF (cyanobacterial toxins) in seedlings of several important agricultural plant species and the correlation with cellular damage (lipid peroxidation). Environ. Toxicol. 2007, 22, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Kittler, K.; Schreiner, M.; Krumbein, A.; Manzei, S.; Koch, M.; Rohn, S.; Maul, R. Uptake of the cyanobacterial toxin cylindrospermopsin in Brassica vegetables. Food Chem. 2012, 133, 875–879. [Google Scholar] [CrossRef]
- Mazur-Marzec, H.; Błaszczyk, A.; Felczykowska, A.; Hohlfeld, N.; Kobos, J.; Toruńska-Sitarz, A.; Devi, P.; Montalvão, S.; D’souza, L.; Tammela, P.; et al. Baltic cyanobacteria—A source of biologically active compounds. Eur. J. Phycol. 2015, 50, 343–360. [Google Scholar] [CrossRef]
- Lifshits, M.; Zafrir-Ilan, E.; Raveh, A.; Carmeli, S. Protease inhibitors from three fishpond water blooms of Microcystis spp. Tetrahedron 2011, 67, 4017–4024. [Google Scholar] [CrossRef]
- Namikoshi, M.; Rinehart, K.L. Bioactive compounds produced by cyanobacteria. J. Ind. Microbiol. Biotechnol. 1996, 17, 373–384. [Google Scholar] [CrossRef]
- Jaspars, M.; Lawton, L.A. Cyanobacteria—A novel source of pharmaceuticals. Curr. Opin. Drug Discov. Dev. 1998, 1, 77–84. [Google Scholar]
- Kodani, S.; Ishida, K.; Murakami, M. Dehydroradiosumin, a trypsin inhibitor from the cyanobacterium Anabaena cylindrica. J. Nat. Prod. 1998, 61, 854–856. [Google Scholar] [CrossRef] [PubMed]
- Ploutno, A.; Shoshan, M.; Carmeli, S. Three novel protease inhibitors from a natural bloom of the cyanobacterium Microcystis aeruginosa. J. Nat. Prod. 2002, 65, 973–978. [Google Scholar] [CrossRef]
- Pluotno, A.; Carmeli, S. Banyasin A and banyasides A and B, three novel modified peptides from a water bloom of the cyanobacterium Nostoc sp. Tetrahedron 2005, 61, 575–583. [Google Scholar] [CrossRef]
- Grach-Pogrebinsky, O.; Sedmak, B.; Carmeli, S. Protease inhibitors from a Slovenian Lake Bled toxic waterbloom of the cyanobacterium Planktothrix rubescens. Tetrahedron 2003, 59, 8329–8336. [Google Scholar] [CrossRef]
- Bubik, A.; Sedmak, B.; Novinec, M.; Lenarčič, B.; Lah, T.T. Cytotoxic and peptidase inhibitory activities of selected non-hepatotoxic cyclic peptides from cyanobacteria. Biol. Chem. 2008, 389, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, M.; Maruthanayagam, V.; Sundararaman, M. A review of pharmacological and toxicological potentials of marine cyanobacterial metabolites. J. Appl. Toxicol. 2012, 32, 153–185. [Google Scholar] [CrossRef]
- Reeck, G.R.; Kramer, K.J.; Baker, J.E.; Kanost, M.R.; Fabrick, J.A.; Behnke, C.A. Proteinase inhibitors and resistance of transgenic plants to insects. In Advances in Insect Control: The Role of Transgenic Plants; Carozzi, N., Koziel, M., Eds.; Taylor & Francis: London, UK, 1997; pp. 157–183. [Google Scholar]
- Lawrence, P.; Koundal, K. Plant protease inhibitors in control of phytophagous insects. Electron. J. Biotechnol. 2002, 5, 5–6. [Google Scholar] [CrossRef]
- McElhiney, J.; Lawton, L.A.; Leifert, C. Investigations into the inhibitory effects of microcystins on plant growth, and the toxicity of plant tissues following exposure. Toxicon 2001, 39, 1411–1420. [Google Scholar] [CrossRef]
- Llana-Ruiz-Cabello, M.; Jos, A.; Cameán, A.; Oliveira, F.; Barreiro, A.; MacHado, J.; Azevedo, J.; Pinto, E.; Almeida, A.; Campos, A.; et al. Analysis of the use of cylindrospermopsin and/or microcystin-contaminated water in the growth, mineral content, and contamination of Spinacia oleracea and Lactuca sativa. Toxins 2019, 11, 624. [Google Scholar] [CrossRef]
- Beyer, D.; Surányi, G.; Vasas, G.; Roszik, J.; Erdodi, F.; M-Hamvas, M.; Bácsi, I.; Bátori, R.; Serfozo, Z.; Szigeti, Z.M.; et al. Cylindrospermopsin induces alterations of root histology and microtubule organization in common reed (Phragmites australis) plantlets cultured in vitro. Toxicon 2009, 54, 440–449. [Google Scholar] [CrossRef]
- Beyer, D.; Tándor, I.; Kónya, Z.; Bátori, R.; Roszik, J.; Vereb, G.; Erddi, F.; Vasas, G.; M-Hamvas, M.; Jambrovics, K.; et al. Microcystin-LR, a protein phosphatase inhibitor, induces alterations in mitotic chromatin and microtubule organization leading to the formation of micronuclei in Vicia faba. Ann. Bot. 2012, 110, 797–808. [Google Scholar] [CrossRef]
- Livanos, P.; Galatis, B.; Apostolakos, P. The interplay between ROS and tubulin cytoskeleton in plants. Plant Signal. Behav. 2014, 9, e28069. [Google Scholar] [CrossRef]
- Li, J.; Cao, L.; Staiger, C.J. Capping protein modulates actin remodeling in response to reactive oxygen species during plant innate immunity. Plant Physiol. 2017, 173, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Calteau, A.; Fewer, D.P.; Latifi, A.; Coursin, T.; Laurent, T.; Jokela, J.; Kerfeld, C.A.; Sivonen, K.; Piel, J.; Gugger, M. Phylum-wide comparative genomics unravel the diversity of secondary metabolism in Cyanobacteria. BMC Genom. 2014, 15, 977. [Google Scholar] [CrossRef] [PubMed]
- Mazard, S.; Penesyan, A.; Ostrowski, M.; Paulsen, I.; Egan, S. Tiny microbes with a big impact: The role of cyanobacteria and their Metabolites in shaping our future. Mar. Drugs 2016, 14, 97. [Google Scholar] [CrossRef] [PubMed]
- Codd, G.A.; Lindsay, J.; Young, F.M.; Morrison, L.F.; Metcalf, J.S. Harmful Cyanobacteria. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P.M., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 1–23. [Google Scholar]
- Gkelis, S.; Panou, M.; Chronis, I.; Zervou, S.K.; Christophoridis, C.; Manolidi, K.; Ntislidou, C.; Triantis, T.M.; Kaloudis, T.; Hiskia, A.; et al. Monitoring a newly re-born patient: Water quality and cyanotoxin occurrence in a reconstructed shallow mediterranean lake. Adv. Oceanogr. Limnol. 2017, 8. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Gkelis, S.; Panou, M. Capturing biodiversity: Linking a cyanobacteria culture collection to the “scratchpads” virtual research environment enhances biodiversity knowledge. Biodivers. Data J. 2016, 4, e7965-1. [Google Scholar] [CrossRef]
- Panou, M.; Zervou, S.K.; Kaloudis, T.; Hiskia, A.; Gkelis, S. A Greek Cylindrospermopsis raciborskii strain: Missing link in tropic invader’s phylogeography tale. Harmful Algae 2018, 80, 96–106. [Google Scholar] [CrossRef]
- Rippka, R. Isolation and purification of cyanobacteria. Methods Enzymol. 1988, 167, 3–27. [Google Scholar]
- An, J.S.; Carmichael, W.W. Use of a colorimetric protein phosphatase inhibition assay and enzyme linked immunosorbent assay for the study of microcystins and nodularins. Toxicon 1994, 32, 1495–1507. [Google Scholar] [CrossRef]
- Kwan, J.C.; Taori, K.; Paul, V.J.; Luesch, H. Lyngbyastatins 8–10, elastase inhibitors with cyclic depsipeptide scaffolds isolated from the marine cyanobacterium Lyngbya semiplena. Mar. Drugs 2009, 7, 528–538. [Google Scholar] [CrossRef]
- Panteris, E.; Achlati, T.; Daras, G.; Rigas, S. Stomatal complex development and F-actin organization in maize leaf epidermis depend on cellulose synthesis. Molecules 2018, 23, 1365. [Google Scholar] [CrossRef] [PubMed]
- Adamakis, I.-D.S.; Panteris, E.; Eleftheriou, E.P. The nitrate reductase inhibitor, tungsten, disrupts actin microfilaments in Zea mays L. Protoplasma 2014, 251, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Gavet, O.; Pines, J. Activation of cyclin B1-Cdk1 synchronizes events in the nucleus and the cytoplasm at mitosis. J. Cell Biol. 2010, 189, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Livanos, P.; Galatis, B.; Quader, H.; Apostolakos, P. Disturbance of reactive oxygen species homeostasis induces atypical tubulin polymer formation and affects mitosis in root-tip cells of Triticum turgidum and Arabidopsis thaliana. Cytoskeleton 2012, 69, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriou, E.P.; Adamakis, I.-D.S.; Panteris, E.; Fatsiou, M. Chromium-induced ultrastructural changes and oxidative stress in roots of Arabidopsis thaliana. Int. J. Mol. Sci. 2015, 16, 15852–15871. [Google Scholar] [CrossRef] [PubMed]
- Waterbury, J.B.; Stanier, R.Y. Patterns of growth and development in pleurocapsalean cyanobacteria. Microbiol. Rev. 1978, 42, 2–44. [Google Scholar] [CrossRef] [PubMed]


| MCs Production Status | TAU-MAC Strain | PP1 1 | Trypsin 1 | Elastase 2 |
|---|---|---|---|---|
| MC-producing | Microcystis flos-aquae 1410 | + | − | + |
| Microcystis sp. 2410 | + | − | + | |
| Trichormus variabilis 1614 | + | + | + | |
| Raphidiopsis raciborskii 1414 3 | − | − | + | |
| Non-MC-producing | Jaaginema sp. 0110 | − | − | + |
| Jaaginema sp. 0210 | − | − | + | |
| Microcystis viridis 1810 | − | − | + | |
| Synechococcus sp. 0499 | − | − | + | |
| Chlorogloeopsis fritschii 0599 | − | − | + | |
| Calothrix epiphytica 0399 | − | − | + | |
| Planktothrix agardhii 0514 | − | − | + | |
| Phormidium sp. 0517 | − | − | + | |
| Scytonema sp. 1218 | − | − | + | |
| Nostoc oryzae 0315 | − | − | + | |
| Lyngbya sp. 4418 | − | − | + | |
| Nodularia sp. 0717 | − | − | − |
| MCs Production Status | TAU-MAC Strain | F-actin | Microtubules | H2O2 Levels |
|---|---|---|---|---|
| MC-producing | Microcystis flos-aquae 1410 | ✓ | ✓ | ✓ |
| Microcystis sp. 2410 | ✓ | ✓ | ✓ | |
| Trichormus variabilis 1614 | ✓ | ✓ | ✓ | |
| Raphidiopsis raciborskii 1414 1 | ✓ | ✓ 3 | ✘ | |
| Non-MC-producing | Jaaginema sp. 0110 | ✓ | ✓ | ✓ |
| Jaaginema sp. 0210 | ✓ | ✓ | ✓ | |
| Microcystis viridis 1810 | ✓ | ✓ | ✓ | |
| Synechococcus sp. 0499 | ✓ | ✓ 3 | ✘ | |
| Chlorogloeopsis fritschii 0599 | ✓ | ✓ 3 | ✘ | |
| Planktothrix agardhii 0514 | ✓ | ✓ 3,4 | ✘ | |
| Calothrix epiphytica 0399 | ✓ | ✘ | ✘ | |
| Phormidium sp. 0517 | ✓ | ✘ | ✘ | |
| Scytonema sp. 1218 | ✓ | ✘ | ✘ | |
| Nostoc oryzae 0315 | ✓ 2 | ✘ | ✘ | |
| Lyngbya sp. 4418 | ✓ 2 | ✘ | ✘ | |
| Nodularia sp. 0717 | ✘ | ✘ | ✘ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pappas, D.; Panou, M.; Adamakis, I.-D.S.; Gkelis, S.; Panteris, E. Beyond Microcystins: Cyanobacterial Extracts Induce Cytoskeletal Alterations in Rice Root Cells. Int. J. Mol. Sci. 2020, 21, 9649. https://doi.org/10.3390/ijms21249649
Pappas D, Panou M, Adamakis I-DS, Gkelis S, Panteris E. Beyond Microcystins: Cyanobacterial Extracts Induce Cytoskeletal Alterations in Rice Root Cells. International Journal of Molecular Sciences. 2020; 21(24):9649. https://doi.org/10.3390/ijms21249649
Chicago/Turabian StylePappas, Dimitris, Manthos Panou, Ioannis-Dimosthenis S. Adamakis, Spyros Gkelis, and Emmanuel Panteris. 2020. "Beyond Microcystins: Cyanobacterial Extracts Induce Cytoskeletal Alterations in Rice Root Cells" International Journal of Molecular Sciences 21, no. 24: 9649. https://doi.org/10.3390/ijms21249649
APA StylePappas, D., Panou, M., Adamakis, I.-D. S., Gkelis, S., & Panteris, E. (2020). Beyond Microcystins: Cyanobacterial Extracts Induce Cytoskeletal Alterations in Rice Root Cells. International Journal of Molecular Sciences, 21(24), 9649. https://doi.org/10.3390/ijms21249649







