Transplantation of Neural Precursors Derived from Induced Pluripotent Cells Preserve Perineuronal Nets and Stimulate Neural Plasticity in ALS Rats
Abstract
1. Introduction
2. Results
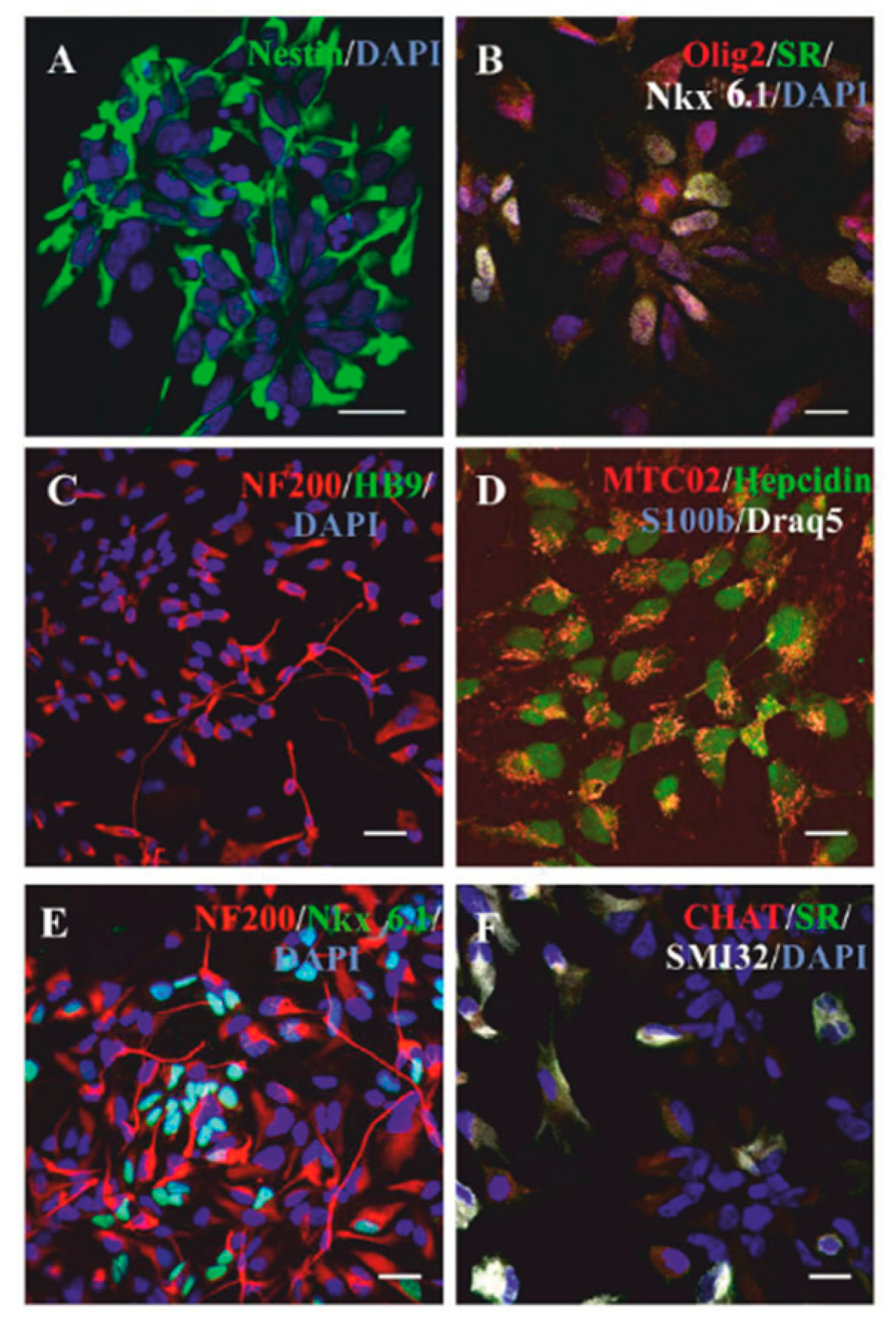
2.1. Differentiation of NP-iPS in Culture
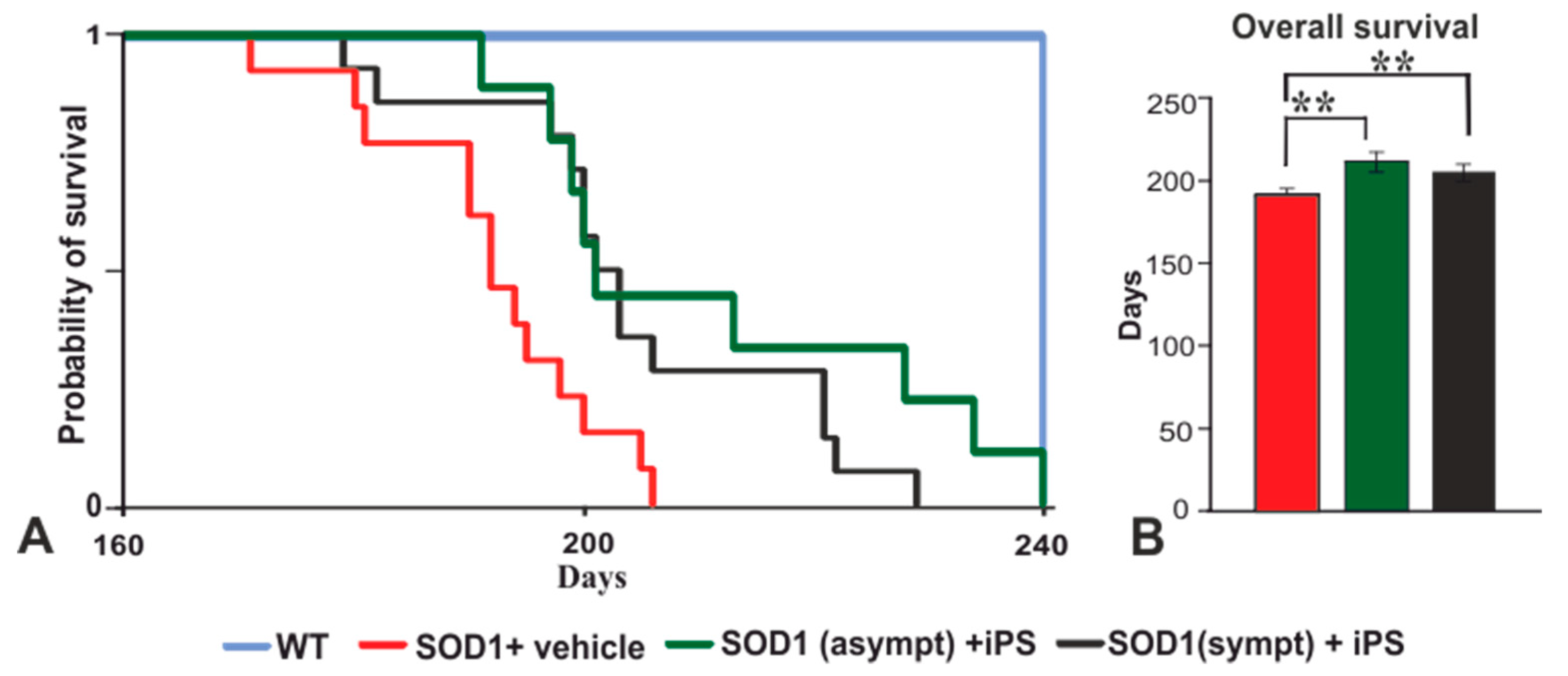
2.2. NP-iPS Transplantation Improves Motor Activity and Extends the Survival of SOD1 Rats
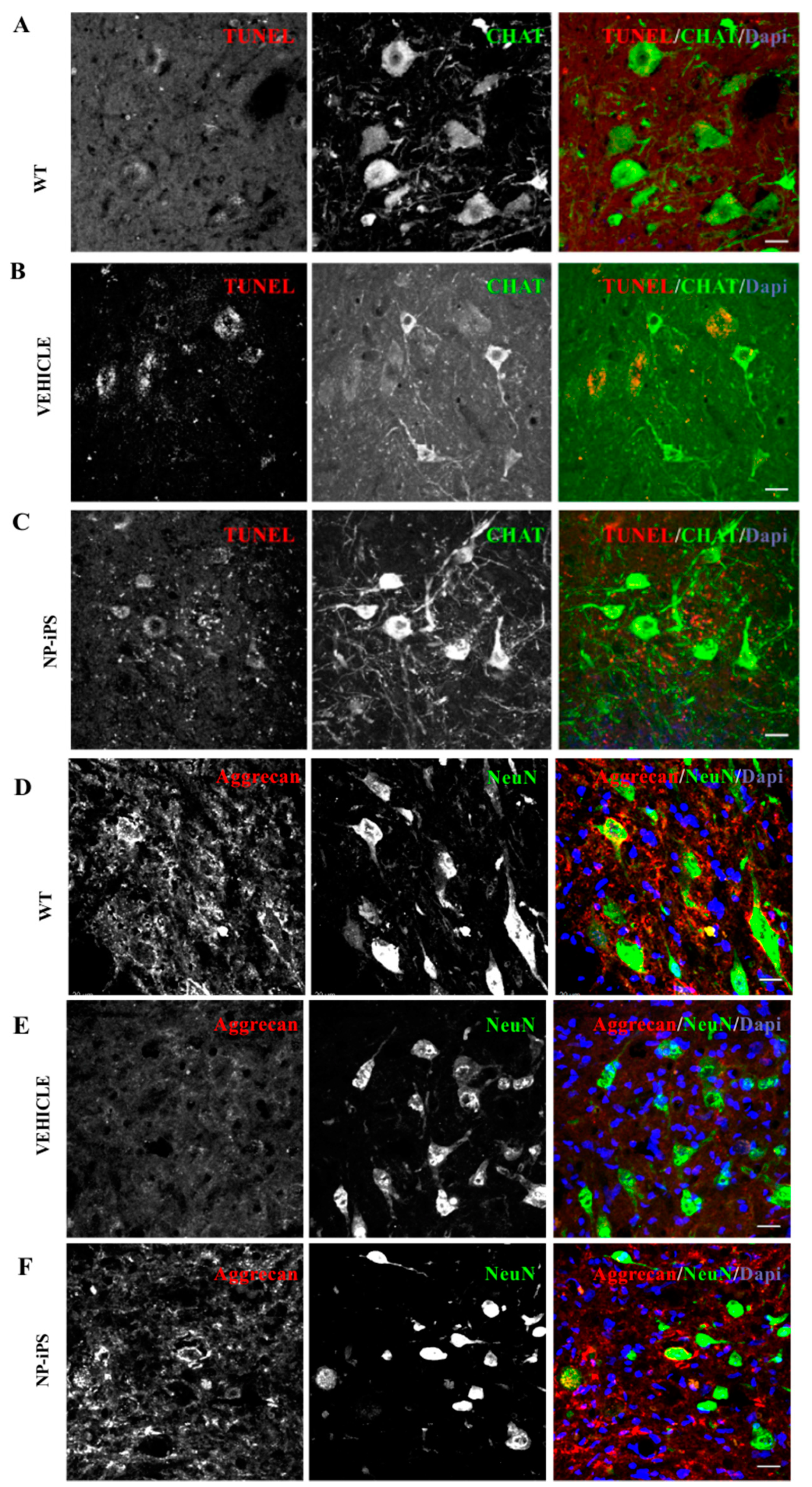
2.3. Spinally Grafted NP-iPS Are Neuroprotective and Do Not Cause Tumors
2.4. NP-iPS Graft Modifies Spinal PNN-, Neurotrophic-, and Apoptosis-Related Gene Expression
2.5. Intraspinally Grafted NP-iPS Normalizes Expression of PNN-Related Chondroitin Sulfate Proteoglycans in Terminal SOD1 Rats
3. Discussion
4. Materials and Methods
4.1. Culture of Human Induced Pluripotent Stem Cell-Derived Neural Precursors
4.2. Animals
4.3. Behavioral Testing
4.3.1. Grip Strength Test (GrST)
4.3.2. BBB Test
4.3.3. Rotarod
4.3.4. Thirty Second Test
- (I)
- NP-iPS transplantation into asymptomatic SOD1 rats (n = 9)
- (II)
- NP-iPS transplantation into symptomatic SOD1 rats (n = 14)
- (III)
- vehicle injection into SOD1symptomatic rats (n = 13)
- (IV)
- vehicle injection into wild type (WT) littermates (n = 7)
4.4. Intraspinal Transplantation of NP-iPS
4.5. Postoperative Care
4.6. Tissue Processing and Immunohistochemistry
4.7. Quantification of Motoneuron Staining in the Spinal Ventral Horns
4.8. Immunocytochemistry
4.9. Electrophoresis and Western Blotting (WB)
4.10. Gene Expression
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| acan | Aggrecan |
| ALS | Amyotrophic lateral sclerosis |
| BDNF | Brain derived neurotrophic factor |
| Bcan | Brevican |
| Casp-3 | Caspase-3 |
| Casp-7 | Caspase-7 |
| CSPGs | Chondroitin sulfate proteoglycans |
| EAAT2 | Excitatory amino acid transporter2 |
| EGF | Epidermal growth factor |
| ECM | Extracellular matrix |
| FGF | Fibroblast growth factor |
| GFAP | Glial fibrillary acidic protein |
| GRNP | Glial-rich neural precursor |
| GrST | Grip strength test |
| Has 1 | Hyaluronan synthase-1 |
| ICC | Immunocytochemistry |
| IHC | Immunohistochemistry |
| iPSC | Induced pluripotent stem cells |
| IGF-1 | Insulin growth factor-1 |
| hapln-1 | Hyaluronan and proteoglycan link protein-1 |
| MMP | Matrix metalloproteinase |
| ADAMST-4 | Metalloproteinase with thrombospondin motifs four |
| MNs | Motoneurons |
| NF | Neurofilaments |
| NGF | Nerve growth factor |
| NP-iPS | Neural precursors derived from induced pluripotent stem cells |
| PNN | Perineuronal nets |
| SR | Serotonin |
| SC | Stem cells |
| SOD1 | Superoxide dismutase-1 |
| TnR | Tenascin-R |
| Tg | Transgenic animals |
| VEGF | Vascular endothelial growth factor |
| vcan | Versican |
| WB | Western Blot |
| WT | Wild type animal |
Appendix A
| Weeks | BBB | Rotarod | GRST | Weight |
|---|---|---|---|---|
| 23 | p ≤ 0.0221 | p = 0.054 | p = 0.08 | p = 0.2 |
| 24 | p ≤ 0.003 | p ≤ 0.0018 | p ≤ 0.024 | p = 0.055 |
| 25 | p ≤ 0.003 | p ≤ 0.0000004 | p ≤ 0.000061 | p = 0.10 |
| 26 | p ≤ 0.003 | p ≤ 0.0000004 | p ≤ 0.000061 | p = 0.20 |
| 27 | p ≤ 0.004 | p ≤ 0.0035 | p ≤ 0.000013 | p = 0.09 |
| 28 | p ≤ 0.024 | p ≤ 0.04 | p ≤ 0.022 | p = 0.1 |
| 29 | p ≤ 0.05 | p ≤ 0.014 | p ≤ 0.04 | p = 0.2 |
| Weeks | BBB | Rotarod | GRST | Weight |
|---|---|---|---|---|
| 23 | p = 0.2 | p = 0.53 | p = 0.37 | p = 0.6 |
| 24 | p ≤ 0.037 | p ≤ 0.21 | p ≤ 0.053 | p = 0.2 |
| 25 | p ≤ 0.021 | p ≤ 0.002 | p ≤ 0.011 | p = 0.5 |
| 26 | p ≤ 0.02 | p ≤ 0.032 | p ≤ 0.04 | p = 0.2 |
| 27 | p ≤ 0.0019 | p = 0.053 | p ≤ 0.0028 | p = 0.1 |
| 28 | p ≤ 0.012 | p ≤ 0.011 | p ≤ 0.012 | p = 0.5 |
| 29 | p ≤ 0.01 | p ≤ 0.026 | p ≤ 0.03 | p = 0.2 |
Appendix B
Appendix C
| AB Name | Protein of Interest/Application | Species & Antibody Type | Concentrations | Manufacturer |
|---|---|---|---|---|
| AB1031 | Aggrecan/WB, IHC | Rabbit Polyclonal | 1:1000 | Millipore, Burlington, MA, USA |
| 12C5 | Versican/WB, IHC | Mouse monoclonal | 1:100 | DSHB, Iowa city, IA, USA |
| Anti-Tenascin-R | Tenascin-R/WB, IHC | Goat polyclonal | 1:1000 | R&D Systems, Minneapolis, MN, USA |
| 1F6 | Neurocan/WB, IHC | Mouse monoclonal | 1:100 | DHSB |
| Anti-Brevican | Brevican/WB, IHC | Mouse polyclonal | 1:100 | Abcam, Cambridge, MA, USA |
| 3F8 | Phosphacan/WB, IHC | Mouse monoclonal | 1:1000 | DHSB |
| Anti-Crtl1 | Link protein-1/WB, IHC | Goat monoclonal | 1:1000 | R&D Systems |
| Anti-NF200 | Neural filaments 200 kDa/IHC | Mouse monoclonal | 1:200 | Sigma-Aldrich, St. Louis, MO, USA |
| SMI-32 | Nonphosphorylated neurofilament H/IHC | Mouse monoclonal | 1:1000 | Covance, Princeton, NJ, USA |
| Anti-NeuN | Neural DNA/IHC | Rabbit monoclonal | 1:200 | Millipore |
| Anti-MTC02 | Non-glycosylated protein component of human mitochondria/IHC | Mouse monoclonal | 1:200 | Abcam |
| Anti-β-III tubulin | β-tubulin isotype III/IHC | Mouse monoclonal | Sigma-Aldrich | |
| Anti-S100β | β chain/IHC/ICC | Rabbit polyclonal | 1:400 | DAKO Denmark, Glostrup, Denmark |
| Anti-l-type Ca2+ CP α1C | L-type Ca2+ CP α1C/ICC | Rabbit polyclonal | 1:200 | Alomone Labs, Jerusalem, Israel |
| Anti-N type Ca2+ CP α1B | N type Ca2+ CP α1B/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-P/Q-type Ca2+ CP α1A | P/Q-type Ca2+ CP α1A/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-Ryanodine Receptor 1 | Ryanodine Receptor 1/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-Ryanodine Receptor 2 | Ryanodine Receptor 2/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-Ryanodine Receptor 3 | Ryanodine Receptor 3/ICC | Rabbit polyclonal | 1:100 | Alomone Labs |
| Anti-ORAI Receptor 1 | ORAI Receptor 1/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-ORAI Receptor 2 | ORAI Receptor 2/ICC | Rabbit polyclonal | 1:200 | Alomone Labs |
| Anti-ORAI Receptor 3 | ORAI Receptor 3/ICC | Rabbit polyclonal | 1:1000 | Sigma-Aldrich |
| Anti-Hepcidin | Hepcidin | Goat monoclonal | 1:200 | Dr. Raha-Chowdhury |
| Anti-HB9 | HB9 homeobox transcription factor | Goat polyclonal | 1:200 | Santa Cruz Inc., Dallas, TX, USA |
| Anti-SR | Serotonin | Goat polyclonal | 1:600 | Abcam |
| Anti-ParAlb | PV 25 Rabbit anti Parvalbumin | Rabbit | 1.2000 | Swant, Marly, Switzerland |
| NKx6.1 | NK homeodomain protein, Nkx6.1 | Mouse monoclonal | 1:20 | DSHB |
| Anti-Islet1 | Islet 1- Neural Stem Cell Marker | Rabbit polyclonal | 1:500 | Abcam |
| Anti-Islet2 | GST fusion from E. coli linker region of protein | Mouse | 1:20 | DSHB |
| Anti-CHAT | Choline Acetyltransferase | Chicken polyclonal | 1:50 | Abcam |
| Anti-Nestin | Anti-NESTIN | Mouse monoclonal | 1:2500 | Millipore, |
| Anti-GFAP | Anti-glial fibrillary acidic protein | Mouse monoclonal | 1:1000 | Cell Signaling, Danvers, MA, USA |
| Assay | Gene Name |
|---|---|
| Rn00573424_m1 | aggrecan |
| Rn00581331_m1 | neurocan |
| Rn00563814_m1 | brevican |
| Rn00564869_m1 | tenascin-R |
| Rn01493755_m1 | versican |
| Rn00569884_m1 | hapln1 |
| Rn99999125_m1 | bcl2 |
| Rn00563902_m1 | casp3 |
| Rn00573917_m1 | casp7 |
| Rn01480161_g1 | bax |
| Rn01533872_m1 | ngf |
| Rn02531967_s1 | bdnf |
| Rn 00710306_m1 | igf-1 |
| Rn 00580526_m1 | ncam |
References
- Renton, A.E.; Chio, A.; Traynor, B.J. State of play in amyotrophic lateral sclerosis genetics. Nat. Neurosci. 2014, 17, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Riva, N.; Agosta, F.; Lunetta, C.; Filippi, M.; Quattrini, A. Recent advances in amyotrophic lateral sclerosis. J. Neurol. 2016, 263, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
- Bunton-Stasyshyn, R.K.; Saccon, R.A.; Fratta, P.; Fisher, E.M. Sod1 function and its implications for amyotrophic lateral sclerosis pathology: New and renascent themes. Neuroscientist 2015, 21, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, M.F.; Martinez, F.J.; Wright, S.; Ramos, C.; Volfson, D.; Mason, M.; Garnes, J.; Dang, V.; Lievers, J.; Shoukat-Mumtaz, U.; et al. A cellular model for sporadic als using patient-derived induced pluripotent stem cells. Mol. Cell. Neurosci. 2013, 56, 355–364. [Google Scholar] [CrossRef]
- Oksanen, M.; Lehtonen, S.; Jaronen, M.; Goldsteins, G.; Hamalainen, R.H.; Koistinaho, J. Astrocyte alterations in neurodegenerative pathologies and their modeling in human induced pluripotent stem cell platforms. Cell Mol. Life Sci. 2019, 76, 2739–2760. [Google Scholar] [CrossRef]
- Tyzack, G.E.; Hall, C.E.; Sibley, C.R.; Cymes, T.; Forostyak, S.; Carlino, G.; Meyer, I.F.; Schiavo, G.; Zhang, S.C.; Gibbons, G.M.; et al. A neuroprotective astrocyte state is induced by neuronal signal ephb1 but fails in als models. Nat. Commun. 2017, 8, 1164. [Google Scholar] [CrossRef]
- Almad, A.A.; Doreswamy, A.; Gross, S.K.; Richard, J.P.; Huo, Y.; Haughey, N.; Maragakis, N.J. Connexin 43 in astrocytes contributes to motor neuron toxicity in amyotrophic lateral sclerosis. Glia 2016, 64, 1154–1169. [Google Scholar] [CrossRef]
- Yamanaka, K.; Komine, O. The multi-dimensional roles of astrocytes in als. Neurosci. Res. 2018, 126, 31–38. [Google Scholar] [CrossRef]
- Kondo, T.; Funayama, M.; Tsukita, K.; Hotta, A.; Yasuda, A.; Nori, S.; Kaneko, S.; Nakamura, M.; Takahashi, R.; Okano, H.; et al. Focal transplantation of human ipsc-derived glial-rich neural progenitors improves lifespan of als mice. Stem Cell Rep. 2014, 3, 242–249. [Google Scholar] [CrossRef]
- Sareen, D.; Gowing, G.; Sahabian, A.; Staggenborg, K.; Paradis, R.; Avalos, P.; Latter, J.; Ornelas, L.; Garcia, L.; Svendsen, C.N. Human induced pluripotent stem cells are a novel source of neural progenitor cells (inpcs) that migrate and integrate in the rodent spinal cord. J. Comp. Neurol. 2014, 522, 2707–2728. [Google Scholar] [CrossRef]
- Richard, J.P.; Maragakis, N.J. Induced pluripotent stem cells from als patients for disease modeling. Brain Res. 2015, 1607, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pak, C.; Han, Y.; Ahlenius, H.; Zhang, Z.; Chanda, S.; Marro, S.; Patzke, C.; Acuna, C.; Covy, J.; et al. Rapid single-step induction of functional neurons from human pluripotent stem cells. Neuron 2013, 78, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.Y.; Zhang, S.C. Differentiation of spinal motor neurons from pluripotent human stem cells. Nat. Protoc. 2009, 4, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.; Ferraiuolo, L.; Miranda, C.J.; Likhite, S.; McElroy, S.; Renusch, S.; Ditsworth, D.; Lagier-Tourenne, C.; Smith, R.A.; Ravits, J.; et al. Direct conversion of patient fibroblasts demonstrates non-cell autonomous toxicity of astrocytes to motor neurons in familial and sporadic als. Proc. Natl. Acad. Sci. USA 2014, 111, 829–832. [Google Scholar] [CrossRef]
- Amemori, T.; Ruzicka, J.; Romanyuk, N.; Jhanwar-Uniyal, M.; Sykova, E.; Jendelova, P. Comparison of intraspinal and intrathecal implantation of induced pluripotent stem cell-derived neural precursors for the treatment of spinal cord injury in rats. Stem Cell Res. Ther. 2015, 6, 257. [Google Scholar] [CrossRef] [PubMed]
- Forostyak, O.; Romanyuk, N.; Verkhratsky, A.; Sykova, E.; Dayanithi, G. Plasticity of calcium signaling cascades in human embryonic stem cell-derived neural precursors. Stem Cells Dev. 2013, 22, 1506–1521. [Google Scholar] [CrossRef]
- Romanyuk, N.; Amemori, T.; Turnovcova, K.; Prochazka, P.; Onteniente, B.; Sykova, E.; Jendelova, P. Beneficial effect of human induced pluripotent stem cell-derived neural precursors in spinal cord injury repair. Cell Transpl. 2015, 24, 1781–1797. [Google Scholar] [CrossRef]
- Sekiya, T.; Holley, M.C.; Hashido, K.; Ono, K.; Shimomura, K.; Horie, R.T.; Hamaguchi, K.; Yoshida, A.; Sakamoto, T.; Ito, J. Cells transplanted onto the surface of the glial scar reveal hidden potential for functional neural regeneration. Proc. Natl. Acad. Sci. USA 2015, 112, E3431–E3440. [Google Scholar] [CrossRef]
- Yu, D.X.; Marchetto, M.C.; Gage, F.H. Therapeutic translation of ipscs for treating neurological disease. Cell Stem Cell 2013, 12, 678–688. [Google Scholar] [CrossRef]
- Feldman, E.L.; Boulis, N.M.; Hur, J.; Johe, K.; Rutkove, S.B.; Federici, T.; Polak, M.; Bordeau, J.; Sakowski, S.A.; Glass, J.D. Intraspinal neural stem cell transplantation in amyotrophic lateral sclerosis: Phase 1 trial outcomes. Ann. Neurol. 2014, 75, 363–373. [Google Scholar] [CrossRef]
- Forostyak, S.; Homola, A.; Turnovcova, K.; Svitil, P.; Jendelova, P.; Sykova, E. Intrathecal delivery of mesenchymal stromal cells protects the structure of altered perineuronal nets in sod1 rats and amends the course of als. Stem Cells 2014, 32, 3163–3172. [Google Scholar] [CrossRef] [PubMed]
- Forostyak, S.; Jendelova, P.; Kapcalova, M.; Arboleda, D.; Sykova, E. Mesenchymal stromal cells prolong the lifespan in a rat model of amyotrophic lateral sclerosis. Cytotherapy 2011, 13, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Popescu, I.R.; Nicaise, C.; Liu, S.; Bisch, G.; Knippenberg, S.; Daubie, V.; Bohl, D.; Pochet, R. Neural progenitors derived from human induced pluripotent stem cells survive and differentiate upon transplantation into a rat model of amyotrophic lateral sclerosis. Stem Cells Transl. Med. 2013, 2, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ryugo, D.K.; Pongstaporn, T.; Johe, K.; Koliatsos, V.E. Human neural stem cell grafts in the spinal cord of sod1 transgenic rats: Differentiation and structural integration into the segmental motor circuitry. J. Comp. Neurol. 2009, 514, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Polentes, J.; Jendelova, P.; Cailleret, M.; Braun, H.; Romanyuk, N.; Tropel, P.; Brenot, M.; Itier, V.; Seminatore, C.; Baldauf, K.; et al. Human induced pluripotent stem cells improve stroke outcome and reduce secondary degeneration in the recipient brain. Cell Transpl. 2012, 21, 2587–2602. [Google Scholar] [CrossRef]
- Cabungcal, J.H.; Steullet, P.; Morishita, H.; Kraftsik, R.; Cuenod, M.; Hensch, T.K.; Do, K.Q. Perineuronal nets protect fast-spiking interneurons against oxidative stress. Proc. Natl. Acad. Sci. USA 2013, 110, 9130–9135. [Google Scholar] [CrossRef]
- Suttkus, A.; Morawski, M.; Arendt, T. Protective properties of neural extracellular matrix. Mol. Neurobiol. 2014, 53, 73–82. [Google Scholar] [CrossRef]
- Kwok, J.C.; Dick, G.; Wang, D.; Fawcett, J.W. Extracellular matrix and perineuronal nets in cns repair. Dev. Neurobiol. 2011, 71, 1073–1089. [Google Scholar] [CrossRef]
- Ganz, T. Hepcidin in iron metabolism. Curr. Opin. Hematol. 2004, 11, 251–254. [Google Scholar] [CrossRef]
- Lu, H.; Lian, L.; Shi, D.; Zhao, H.; Dai, Y. Hepcidin promotes osteogenic differentiation through the bone morphogenetic protein 2/small mothers against decapentaplegic and mitogen-activated protein kinase/p38 signaling pathways in mesenchymal stem cells. Mol. Med. Rep. 2015, 11, 143–150. [Google Scholar] [CrossRef]
- Raha-Chowdhury, R.; Raha, A.A.; Zhao, J.W.; Stott, S.; Bomford, A. Iron regulatory protein hepcidin present in embryonic brain and increased in the glial scar after mechanical injury. In F1000 Posters 2014; Demyelination and Remyelination: From Mechanism to Therapy; The New York Academy of Science: New York, NY, USA, 2014; Volume 5. [Google Scholar]
- Hutchinson, S.A.; Cheesman, S.E.; Hale, L.A.; Boone, J.Q.; Eisen, J.S. Nkx6 proteins specify one zebrafish primary motoneuron subtype by regulating late islet1 expression. Development 2007, 134, 1671–1677. [Google Scholar] [CrossRef] [PubMed]
- Raha-Chowdhury, R.; Raha, A.A.; Forostyak, S.; Zhao, J.W.; Stott, S.R.; Bomford, A. Expression and cellular localization of hepcidin mrna and protein in normal rat brain. BMC Neurosci. 2015, 16, 24. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, T.J.; Rossi, S.L.; Siegenthaler, M.M.; Frame, J.; Robles, R.; Nistor, G.; Keirstead, H.S. Human motor neuron progenitor transplantation leads to endogenous neuronal sparing in 3 models of motor neuron loss. Stem Cells Int. 2011, 2011, 207230. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gonzalez, R.; Kunckles, P.; Velasco, I. Transient recovery in a rat model of familial amyotrophic lateral sclerosis after transplantation of motor neurons derived from mouse embryonic stem cells. Cell Transpl. 2009, 18, 1171–1181. [Google Scholar] [CrossRef] [PubMed]
- Lepore, A.C.; Rauck, B.; Dejea, C.; Pardo, A.C.; Rao, M.S.; Rothstein, J.D.; Maragakis, N.J. Focal transplantation-based astrocyte replacement is neuroprotective in a model of motor neuron disease. Nat. Neurosci. 2008, 11, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yan, J.; Chen, D.; Welsh, A.M.; Hazel, T.; Johe, K.; Hatfield, G.; Koliatsos, V.E. Human neural stem cell grafts ameliorate motor neuron disease in sod-1 transgenic rats. Transplantation 2006, 82, 865–875. [Google Scholar] [CrossRef]
- Forostyak, S.; Jendelova, P.; Sykova, E. The role of mesenchymal stromal cells in spinal cord injury, regenerative medicine and possible clinical applications. Biochimie 2013, 95, 2257–2270. [Google Scholar] [CrossRef]
- Ziv, Y.; Avidan, H.; Pluchino, S.; Martino, G.; Schwartz, M. Synergy between immune cells and adult neural stem/progenitor cells promotes functional recovery from spinal cord injury. Proc. Natl. Acad. Sci. USA 2006, 103, 13174–13179. [Google Scholar] [CrossRef]
- Butovsky, O.; Ziv, Y.; Schwartz, A.; Landa, G.; Talpalar, A.E.; Pluchino, S.; Martino, G.; Schwartz, M. Microglia activated by il-4 or ifn-gamma differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol. Cell. Neurosci. 2006, 31, 149–160. [Google Scholar] [CrossRef]
- Devlin, A.C.; Burr, K.; Borooah, S.; Foster, J.D.; Cleary, E.M.; Geti, I.; Vallier, L.; Shaw, C.E.; Chandran, S.; Miles, G.B. Human ipsc-derived motoneurons harbouring tardbp or c9orf72 als mutations are dysfunctional despite maintaining viability. Nat. Commun. 2015, 6, 5999. [Google Scholar] [CrossRef]
- Kwok, J.C.; Afshari, F.; Garcia-Alias, G.; Fawcett, J.W. Proteoglycans in the central nervous system: Plasticity, regeneration and their stimulation with chondroitinase abc. Restor. Neurol. Neurosci. 2008, 26, 131–145. [Google Scholar] [PubMed]
- Rehorova, M.; Vargova, I.; Forostyak, S.; Vackova, I.; Turnovcova, K.; Kupcova Skalnikova, H.; Vodicka, P.; Kubinova, S.; Sykova, E.; Jendelova, P. A combination of intrathecal and intramuscular application of human mesenchymal stem cells partly reduces the activation of necroptosis in the spinal cord of sod1(g93a) rats. Stem Cells Transl. Med. 2019, 8, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Jirak, D.; Ziolkowska, N.; Turnovcova, K.; Karova, K.; Sykova, E.; Jendelova, P.; Romanyuk, N. Metabolic changes in focal brain ischemia in rats treated with human induced pluripotent stem cell-derived neural precursors confirm the beneficial effect of transplanted cells. Front. Neurol. 2019, 10, 1074. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; McHugh, J.; Tork, C.; Shelley, B.; Klein, S.M.; Aebischer, P.; Svendsen, C.N. Gdnf secreting human neural progenitor cells protect dying motor neurons, but not their projection to muscle, in a rat model of familial als. PLoS ONE 2007, 2, e689. [Google Scholar] [CrossRef]
- Lunn, J.S.; Sakowski, S.A.; McGinley, L.M.; Pacut, C.; Hazel, T.G.; Johe, K.; Feldman, E.L. Autocrine production of igf-i increases stem cell-mediated neuroprotection. Stem Cells 2015, 33, 1480–1489. [Google Scholar] [CrossRef]
- Ciucci, F.; Putignano, E.; Baroncelli, L.; Landi, S.; Berardi, N.; Maffei, L. Insulin-like growth factor 1 (igf-1) mediates the effects of enriched environment (ee) on visual cortical development. PLoS ONE 2007, 2, e475. [Google Scholar] [CrossRef]
- Kostic, V.; Jackson-Lewis, V.; de Bilbao, F.; Dubois-Dauphin, M.; Przedborski, S. Bcl-2: Prolonging life in a transgenic mouse model of familial amyotrophic lateral sclerosis. Science 1997, 277, 559–562. [Google Scholar] [CrossRef]
- Van Loo, G.; Saelens, X.; van Gurp, M.; MacFarlane, M.; Martin, S.J.; Vandenabeele, P. The role of mitochondrial factors in apoptosis: A russian roulette with more than one bullet. Cell Death Differ. 2002, 9, 1031–1042. [Google Scholar] [CrossRef]
- Boston-Howes, W.; Gibb, S.L.; Williams, E.O.; Pasinelli, P.; Brown, R.H., Jr.; Trotti, D. Caspase-3 cleaves and inactivates the glutamate transporter eaat2. J. Biol. Chem. 2006, 281, 14076–14084. [Google Scholar] [CrossRef]
- Fawcett, J.W. The extracellular matrix in plasticity and regeneration after cns injury and neurodegenerative disease. Prog. Brain Res 2015, 218, 213–226. [Google Scholar]
- Rowlands, D.; Sugahara, K.; Kwok, J.C. Glycosaminoglycans and glycomimetics in the central nervous system. Molecules 2015, 20, 3527–3548. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, H.; Warita, H.; Aoki, M.; Itoyama, Y. Accumulation of chondroitin sulfate proteoglycans in the microenvironment of spinal motor neurons in amyotrophic lateral sclerosis transgenic rats. J. Neurosci. Res. 2008, 86, 2512–2523. [Google Scholar] [CrossRef] [PubMed]
- Lemarchant, S.; Pomeshchik, Y.; Kidin, I.; Karkkainen, V.; Valonen, P.; Lehtonen, S.; Goldsteins, G.; Malm, T.; Kanninen, K.; Koistinaho, J. Adamts-4 promotes neurodegeneration in a mouse model of amyotrophic lateral sclerosis. Mol. Neurodegener. 2016, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, E.J.; Moon, L.D.; Popat, R.J.; King, V.R.; Bennett, G.S.; Patel, P.N.; Fawcett, J.W.; McMahon, S.B. Chondroitinase abc promotes functional recovery after spinal cord injury. Nature 2002, 416, 636–640. [Google Scholar] [CrossRef]
- Pizzorusso, T.; Medini, P.; Berardi, N.; Chierzi, S.; Fawcett, J.W.; Maffei, L. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 2002, 298, 1248–1251. [Google Scholar] [CrossRef]
- Carulli, D.; Pizzorusso, T.; Kwok, J.C.; Putignano, E.; Poli, A.; Forostyak, S.; Andrews, M.R.; Deepa, S.S.; Glant, T.T.; Fawcett, J.W. Animals lacking link protein have attenuated perineuronal nets and persistent plasticity. Brain A J. Neurol. 2010, 133, 2331–2347. [Google Scholar] [CrossRef]
- Van Velthoven, C.T.; Kavelaars, A.; van Bel, F.; Heijnen, C.J. Mesenchymal stem cell transplantation changes the gene expression profile of the neonatal ischemic brain. Brain Behav. Immun. 2011, 25, 1342–1348. [Google Scholar] [CrossRef]
- Barkho, B.Z.; Munoz, A.E.; Li, X.; Li, L.; Cunningham, L.A.; Zhao, X. Endogenous matrix metalloproteinase (mmp)-3 and mmp-9 promote the differentiation and migration of adult neural progenitor cells in response to chemokines. Stem Cells 2008, 26, 3139–3149. [Google Scholar] [CrossRef]
- Beurdeley, M.; Spatazza, J.; Lee, H.H.; Sugiyama, S.; Bernard, C.; Di Nardo, A.A.; Hensch, T.K.; Prochiantz, A. Otx2 binding to perineuronal nets persistently regulates plasticity in the mature visual cortex. J. Neurosci. Off. J. Soc. Neurosci. 2012, 32, 9429–9437. [Google Scholar] [CrossRef]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 2007, 318, 1917–1920. [Google Scholar] [CrossRef]
- Basso, D.M.; Beattie, M.S.; Bresnahan, J.C. A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 1995, 12, 1–21. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The miqe guidelines: Minimum information for publication of quantitative real-time pcr experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.; Spiller, K.J.; Towne, C.; Kanning, K.C.; Choe, G.T.; Geber, A.; Akay, T.; Aebischer, P.; Henderson, C.E. Neuronal matrix metalloproteinase-9 is a determinant of selective neurodegeneration. Neuron 2014, 81, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Israelson, A.; Ditsworth, D.; Sun, S.; Song, S.; Liang, J.; Hruska-Plochan, M.; McAlonis-Downes, M.; Abu-Hamad, S.; Zoltsman, G.; Shani, T.; et al. Macrophage migration inhibitory factor as a chaperone inhibiting accumulation of misfolded sod1. Neuron 2015, 86, 218–232. [Google Scholar] [CrossRef] [PubMed]
- Bai, F.; Asojo, O.A.; Cirillo, P.; Ciustea, M.; Ledizet, M.; Aristoff, P.A.; Leng, L.; Koski, R.A.; Powell, T.J.; Bucala, R.; et al. A novel allosteric inhibitor of macrophage migration inhibitory factor (mif). J. Biol. Chem. 2012, 287, 30653–30663. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forostyak, S.; Forostyak, O.; Kwok, J.C.F.; Romanyuk, N.; Rehorova, M.; Kriska, J.; Dayanithi, G.; Raha-Chowdhury, R.; Jendelova, P.; Anderova, M.; et al. Transplantation of Neural Precursors Derived from Induced Pluripotent Cells Preserve Perineuronal Nets and Stimulate Neural Plasticity in ALS Rats. Int. J. Mol. Sci. 2020, 21, 9593. https://doi.org/10.3390/ijms21249593
Forostyak S, Forostyak O, Kwok JCF, Romanyuk N, Rehorova M, Kriska J, Dayanithi G, Raha-Chowdhury R, Jendelova P, Anderova M, et al. Transplantation of Neural Precursors Derived from Induced Pluripotent Cells Preserve Perineuronal Nets and Stimulate Neural Plasticity in ALS Rats. International Journal of Molecular Sciences. 2020; 21(24):9593. https://doi.org/10.3390/ijms21249593
Chicago/Turabian StyleForostyak, Serhiy, Oksana Forostyak, Jessica C. F. Kwok, Nataliya Romanyuk, Monika Rehorova, Jan Kriska, Govindan Dayanithi, Ruma Raha-Chowdhury, Pavla Jendelova, Miroslava Anderova, and et al. 2020. "Transplantation of Neural Precursors Derived from Induced Pluripotent Cells Preserve Perineuronal Nets and Stimulate Neural Plasticity in ALS Rats" International Journal of Molecular Sciences 21, no. 24: 9593. https://doi.org/10.3390/ijms21249593
APA StyleForostyak, S., Forostyak, O., Kwok, J. C. F., Romanyuk, N., Rehorova, M., Kriska, J., Dayanithi, G., Raha-Chowdhury, R., Jendelova, P., Anderova, M., Fawcett, J. W., & Sykova, E. (2020). Transplantation of Neural Precursors Derived from Induced Pluripotent Cells Preserve Perineuronal Nets and Stimulate Neural Plasticity in ALS Rats. International Journal of Molecular Sciences, 21(24), 9593. https://doi.org/10.3390/ijms21249593








