Metals in Imaging of Alzheimer’s Disease
Abstract
1. Introduction
2. Copper Coordination Compounds for PET Imaging of Alzheimer Disease
3. Gd3+ and Ga3+ Coordination Compounds for Aβ Visualization
4. 99mTc3+-Based Coordination Compounds for SPECT Visualization of Aβ
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| [125I]IMPY | ([125I]6-iodo-2-(40-dimethylamino)-phenyl-imidazo [1,2-a]pyridine) |
| ICP-MS | inductively coupled plasma mass spectrometry |
| [18F]FDDNP | (1,1-dicyanopropen-2-yl)-6-(2-[18F]-fluoroethyl)-methylamino-naphthalene |
| SPECT | single-photon emission computed tomography |
| PET | positron emission tomography |
| MRI | magnetic resonance imaging |
| AD | Alzheimer’s disease |
| BBB | Blood-brain barrier |
| CAA | cerebral amyloid angiopathy |
| PgP | P-glycoprotein |
| TEM | transmission electron microscopy |
| Tg-mice | transgenic mice |
| ThT | Thioflavin-T |
| DOTA | 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid |
| DO3A | 1,4,7,10-tetraazacyclododecane-1,4,7-triacetic acid |
| PCTA | 3,6,9,15-tetraaza bicyclo[9.3.1]-pentadeca1(15),11,13-triene-3,6,9-triacetic acid |
| HBED-CC | N,N’-bis[2-hydroxy-5-(carboxyethyl)benzyl]ethylenediamine-N,N’-diacetic acid |
| NODAGA | 1,4,7-triazacyclononane,1-glutaric acid-4,7-acetic acid |
| AAZTA | 1,4-bis(carboxymethyl)-6-[bis(carboxymethyl)]amino-6-methylperhydro-1,4-diazepine |
| BAT- | Bis-amino bis-thiol |
| MAMA | Monoamine-monoamide dithiols |
| IC50 | The half maximal inhibitory concentration |
| MTT | (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| PIB | 2-(4′-[11C]methylaminophenyl)-6-hydroxybenzothiazole |
References
- Penke, B.; Szűcs, M.; Bogár, F. Oligomerization and Conformational Change Turn Monomeric β-Amyloid and Tau Proteins Toxic: Their Role in Alzheimer’s Pathogenesis. Molecules 2020, 25, 1659. [Google Scholar] [CrossRef]
- Chen, G.-F.; Xu, T.-H.; Yan, Y.; Zhou, Y.-R.; Jiang, Y.; Melcher, K.; Xu, H.E. Amyloid beta: Structure, biology and structure-based therapeutic development. Acta Pharmacol. Sin. 2017, 38, 1205–1235. [Google Scholar] [CrossRef]
- Savelieff, M.G.; Lee, S.; Liu, Y.; Lim, M.H. Untangling Amyloid-β, Tau, and Metals in Alzheimer’s Disease. ACS Chem. Biol. 2013, 8, 856–865. [Google Scholar] [CrossRef]
- LaFerla, F.M.; Green, K.N.; Oddo, S. Intracellular amyloid-β in Alzheimer’s disease. Nat. Rev. Neurosci. 2007, 8, 499–509. [Google Scholar] [CrossRef]
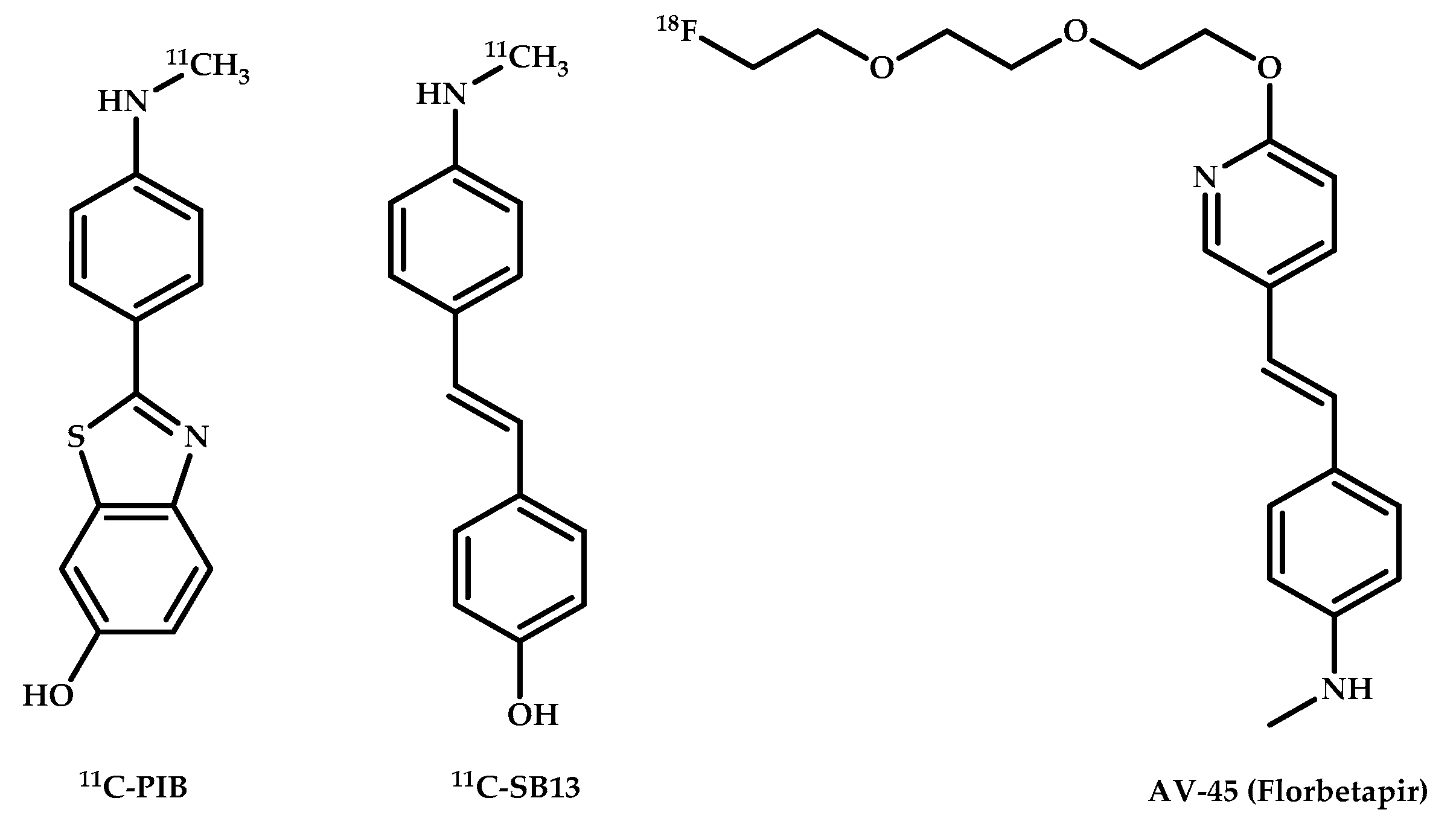
- Verhoeff, N.P.; Wilson, A.A.; Takeshita, S.; Trop, L.; Hussey, D.; Singh, K.; Kung, H.F.; Kung, M.P.; Houle, S. In-vivo imaging of Alzheimer disease beta-amyloid with [11C]SB-13 PET. Am. J. Geriatr. Psychiatry 2004, 12, 584–595. [Google Scholar]
- Rowe, C.C.; Villemagne, V.L. Brain Amyloid Imaging. J. Nucl. Med. Technol. 2013, 41, 11–18. [Google Scholar] [CrossRef]
- Roe, C.M.; Fagan, A.M.; Grant, E.A.; Hassenstab, J.; Moulder, K.L.; Dreyfus, D.M.; Sutphen, C.L.; Benzinger, T.L.S.; Mintun, M.A.; Holtzman, D.M.; et al. Amyloid imaging and CSF biomarkers in predicting cognitive impairment up to 7.5 years later. Neurology 2013, 80, 1784–1791. [Google Scholar] [CrossRef]
- Therriault, J.; Benedet, A.; Pascoal, T.A.; Savard, M.; Ashton, N.; Chamoun, M.; Tissot, C.; Lussier, F.; Kang, M.S.P.; Bezgin, G.; et al. Determining Amyloid-β positivity using [18F]AZD4694 PET imaging. J. Nucl. Med. 2020, 120, 245209. [Google Scholar]
- Chang, Y.; Li, C.; Yang, H.; Wu, Y.; Xu, B.; Zhang, J.; Wang, R. 18F-Florbetaben Amyloid PET Imaging: A Chinese Study in Cognitive Normal Controls, Mild Cognitive Impairment, and Alzheimer’s Disease Patients. Front. Neurosci. 2020, 14, 745. [Google Scholar] [CrossRef]
- Cohen, A.D.; Rabinovici, G.D.; Mathis, C.A.; Jagust, W.J.; Klunk, W.E.; Ikonomovic, M.D. Using Pittsburgh Compound B for In Vivo PET Imaging of Fibrillar Amyloid-Beta. Adv. Pharmacol. 2012, 64, 27–81. [Google Scholar]
- Carotenuto, A.; Giordano, B.; Dervenoulas, G.; Wilson, H.; Veronese, M.; Chappell, Z.; Polychronis, S.; Pagano, G.; Mackewn, J.; Turkheimer, F.E.; et al. [18F] Florbetapir PET/MR imaging to assess demyelination in multiple sclerosis. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Chételat, G.; Arbizu, J.; Barthel, H.; Garibotto, V.; Law, I.; Morbelli, S.; van de Giessen, E.; Agosta, F.; Barkhof, F.; Brooks, D.J.; et al. Amyloid-PET and 18F-FDG-PET in the diagnostic investigation of Alzheimer’s disease and other dementias. Lancet Neurol. 2020, 19, 951–962. [Google Scholar] [CrossRef]
- Moynier, F.; Creech, J.; Dallas, J.; Le Borgne, M. Serum and brain natural copper stable isotopes in a mice model of Alzheimer’s disease. Sci. Rep. 2019, 9, 1–7. [Google Scholar] [CrossRef]
- Lau, J.; Rousseau, E.; Kwon, D.; Lin, K.-S.; Bénard, F.; Chen, X. Insight into the Development of PET Radiopharmaceuticals for Oncology. Cancers 2020, 12, 1312. [Google Scholar] [CrossRef] [PubMed]
- Meisenheimer, M.; Saenko, Y.; Eppard, E. Gallium-68: Radiolabeling of Radiopharmaceuticals for PET Imaging—A Lot to Consider. Med Isot. 2019. [Google Scholar] [CrossRef]
- Romero, E.; Martínez, A.; Oteo, M.; Ibañez, M.; Santos, M.; Morcillo, M.Á. Development and long-term evaluation of a new 68Ge/68Ga generator based on nano-SnO2 for PET imaging. Sci. Rep. 2020, 10, 12756. [Google Scholar] [CrossRef]
- Dash, A.; Chakravarty, R. Radionuclide generators: The prospect of availing PET radiotracers to meet current clinical needs and future research demands. Am. J. Nucl. Med. Mol. Imaging 2019, 9, 30–66. [Google Scholar]
- Boschi, A.; Uccelli, L.; Martini, P. A Picture of Modern Tc-99m Radiopharmaceuticals: Production, Chemistry, and Applications in Molecular Imaging. Appl. Sci. 2019, 9, 2526. [Google Scholar] [CrossRef]
- Wang, C.; Wang, X.; Chan, H.; Chan, H.-N.; Liu, G.; Wang, Z.; Li, H.; Li, H.-W.; Wong, M.S. Amyloid-β Oligomer-Targeted Gadolinium-Based NIR/MR Dual-Modal Theranostic Nanoprobe for Alzheimer’s Disease. Adv. Funct. Mater. 2020, 30, 1909529. [Google Scholar] [CrossRef]
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The Blood-Brain Barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef]
- He, Q.; Liu, J.; Liang, J.; Liu, X.; Li, W.; Liu, Z.; Ding, Z.; Tuo, D. Towards improvements for penetrating the blood-brain barrier—Recent progress from a material and pharmaceutical perspective. Cells 2018, 7, 24. [Google Scholar] [CrossRef]
- Rautio, J.; Laine, K.; Gynther, M.; Savolainen, J. Prodrug approaches for CNS delivery. AAPS J. 2008, 10, 92–102. [Google Scholar] [CrossRef]
- Aryal, M.; Arvanitis, C.D.; Alexander, P.M.; McDannold, N. Ultrasound-mediated blood-brain barrier disruption for targeted drug delivery in the central nervous system. Adv. Drug Deliv. Rev. 2014, 72, 94–109. [Google Scholar] [CrossRef]
- Polat, B.E.; Blankschtein, D.; Langer, R. Low-frequency sonophoresis: Application to the transdermal delivery of macromolecules and hydrophilic drugs. Expert Opin. Drug Deliv. 2010, 7, 1415–1432. [Google Scholar] [CrossRef]
- Wood, I.S.; Trayhurn, P. Glucose transporters (GLUT and SGLT): Expanded families of sugar transport proteins. Br. J. Nutr. 2007, 89, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Oddo, A.; Peng, B.; Tong, Z.; Wei, Y.; Tong, W.Y.; Thissen, H.; Voelcker, N.H. Advances in Microfluidic Blood-Brain Barrier (BBB) Models. Trends Biotechnol. 2019, 12, 1295–1314. [Google Scholar] [CrossRef]
- Di Lullo, E.; Kriegstein, A.R. The use of brain organoids to investigate neural development and disease. Nat. Rev. Neurosci. 2017, 18, 573–584. [Google Scholar] [CrossRef]
- Salman, M.M.; Marsh, G.; Kusters, I.; Delincé, M.; Di Caprio, G.; Upadhyayula, S.; de Nola, G.; Hunt, R.; Ohashi, K.G.; Gray, T.; et al. Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging. Front. Bioeng. Biotechnol. 2020, 8, 1077. [Google Scholar] [CrossRef]
- Mensch, J.; Oyarzabal, J.; Mackie, C.; Augustijns, P. In vivo, in vitro and in silico methods for small molecule transfer across the BBB. J. Pharm. Sci. 2009, 98, 4429–4468. [Google Scholar] [CrossRef]
- Van de Waterbeemd, H.; Camenisch, G.; Folkers, G.; Chretien, J.R.; Raevsky, O.A. Estimation of blood-brain barrier crossing of drugs using molecular size and shape, and H-bonding descriptors. J.Drug Target 1998, 6, 151–165. [Google Scholar] [CrossRef]
- Sedgwick, A.C.; Brewster II, J.T.; Harvey, P.; Iovan, D.A.; Smith, G.; He, X.-P.; Tian, H.; Sessler, J.L.; James, T.D. Metal-based imaging agents: Progress towards interrogating neurodegenerative disease. Chem. Soc. Rev. 2020, 49, 2886–2915. [Google Scholar] [CrossRef]
- Gomes, L.M.F.; Bataglioli, J.C.; Storr, T. Metal complexes that bind to the amyloid-β peptide of relevance to Alzheimer’s disease. Coord. Chem. Rev. 2020, 412, 213255. [Google Scholar] [CrossRef]
- Liu, H.; Qu, Y.; Wang, X. Amyloid β-targeted metal complexes for potential applications in Alzheimer’s disease. Future Med. Chem. 2018, 10, 679–701. [Google Scholar] [CrossRef]
- Daghighian, F.; Sumida, R.; Phelps, M.E. PET Imaging: An Overview and Instrumentation. J. Nucl. Med. Technol. 1990, 18, 5–15. [Google Scholar]
- Sin, I.; Kang, C.S.; Bandara, N.; Sun, X.; Zhong, Y.L.; Rogers, B.E.; Chong, H.S. Novel hexadentate and pentadentate chelators for 64Cu-based targeted PET imaging. Bioorg. Med. Chem. 2014, 22, 2553–2562. [Google Scholar] [CrossRef]
- Kim, D.-K.; Song, J.-W.; Park, J.-D.; Choi, B.-S. Copper induces the accumulation of amyloid-beta in the brain. Mol. Cell. Toxicol. 2013, 9, 57–66. [Google Scholar] [CrossRef]
- Liu, T.; Karlsen, M.; Karlberg, A.M.; Redalen, K.R. Hypoxia imaging and theranostic potential of [64Cu][Cu(ATSM)] and ionic Cu(II) salts: A review of current evidence and discussion of the retention mechanisms. Eur. J. Nucl. Med. Mol. Imaging 2020, 10, 33. [Google Scholar] [CrossRef]
- Sharma, A.K.; Schultz, J.W.; Prior, J.T.; Rath, N.P.; Mirica, L.M. Coordination Chemistry of Bifunctional Chemical Agents Designed for Applications in 64Cu PET Imaging for Alzheimer’s Disease. Inorg. Chem. 2017, 56, 13801–13814. [Google Scholar] [CrossRef]
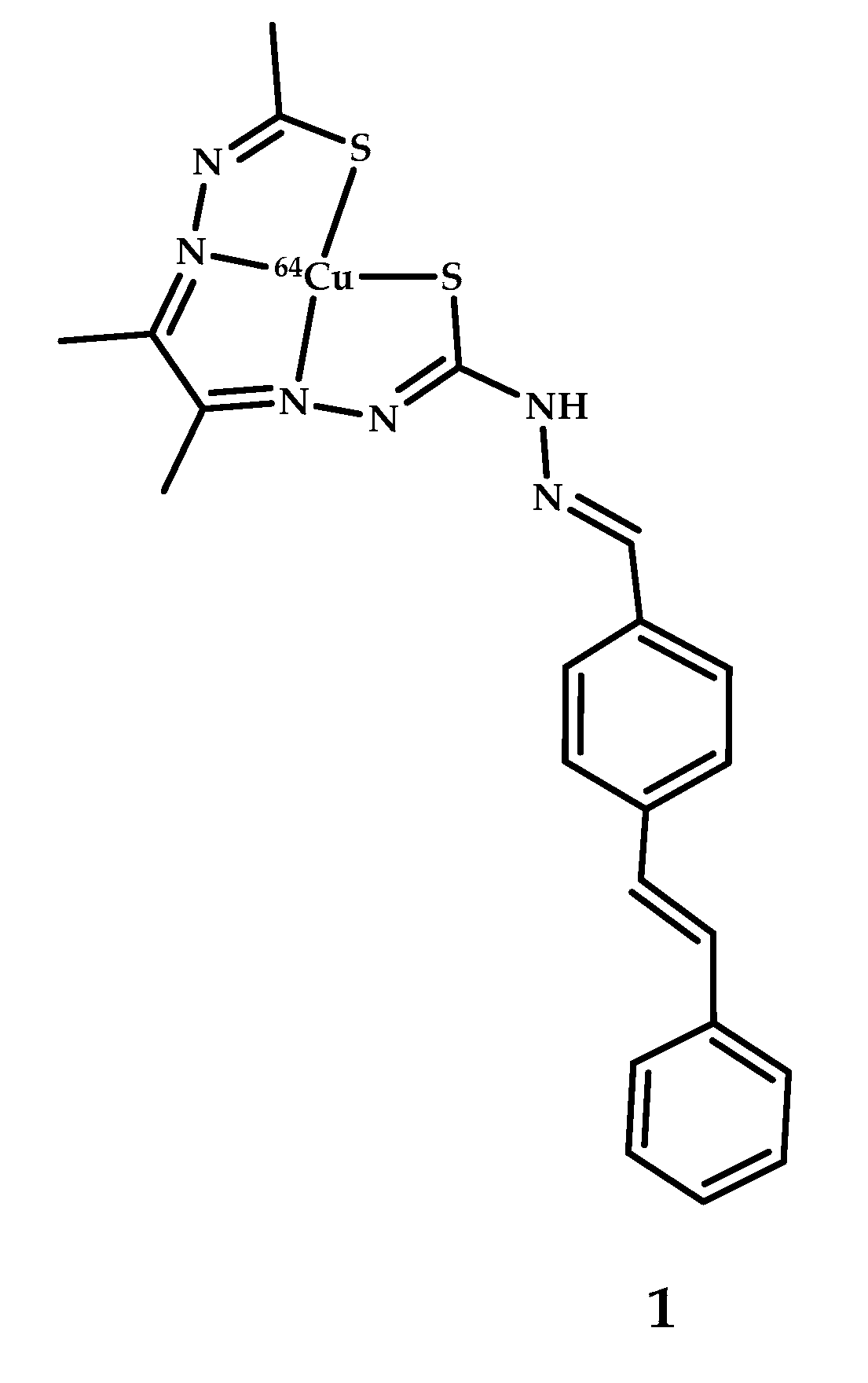
- Lim, S.; Paterson, B.M.; Fodero-Tavoletti, M.T.; O’Keefe, G.J.; Cappai, R.; Barmham, K.J.; Villemagne, V.L.; Donnelly, P.S. A copper radiopharmaceutical for diagnostic imaging of Alzheimer’s disease: A bis(thiosemicarbazonato)copper(II) complex that binds to amyloid-β plaques. Chem. Commun. 2010, 46, 5437–5439. [Google Scholar] [CrossRef]
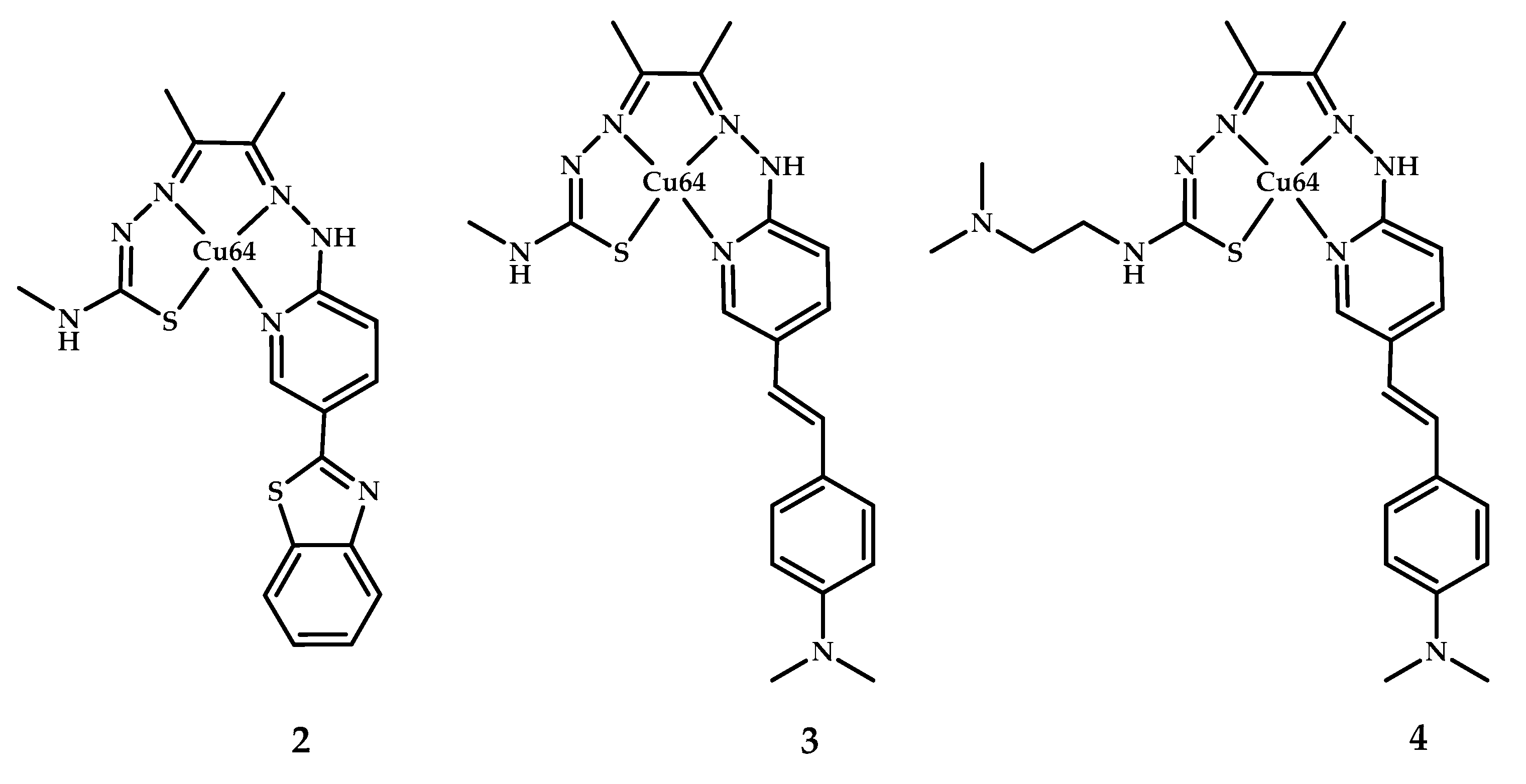
- Hickey, J.L.; Lim, S.; Hayne, D.J.; Paterson, B.M.; White, J.M.; Villemagne, V.L.; Roselt, P.; Binns, D.; Cullinane, C.; Jeffery, C.M.; et al. Diagnostic Imaging Agents for Alzheimer’s Disease: Copper Radiopharmaceuticals that Target Aβ Plaques. J. Am. Chem. Soc. 2013, 135, 16120–16132. [Google Scholar] [CrossRef] [PubMed]
- McInnes, L.E.; Noor, A.; Kysenius, K.; Cullinane, C.; Roselt, P.; McLean, C.A.; Chiu, F.C.K.; Powell, A.K.; Crouch, P.J.; White, J.M.; et al. Potential Diagnostic Imaging of Alzheimer’s Disease with Copper-64 Complexes That Bind to Amyloid-β Plaques. Inorg. Chem. 2019, 58, 3382–3395. [Google Scholar] [CrossRef] [PubMed]
- McInnes, L.E.; Noor, A.; Roselt, P.D.; McLean, C.A.; White, J.M.; Donnelly, P.S. A Copper Complex of a Thiosemicarbazone-Pyridylhydrazone Ligand Containing a Vinylpyridine Functional Group as a Potential Imaging Agent for Amyloid-β Plaques. Aust. J. Chem. 2019, 72, 827. [Google Scholar] [CrossRef]
- Noor, A.; Hayne, D.J.; Lim, S.; Van Zuylekom, J.K.; Cullinane, C.; Roselt, P.D.; McLean, C.A.; White, J.M.; Donnelly, P.S. Copper Bis(thiosemicarbazonato)-stilbenyl Complexes That Bind to Amyloid-β Plaques. Inorg. Chem. 2020, 59, 11658–11669. [Google Scholar] [CrossRef] [PubMed]
- Paterson, B.M.; Cullinane, C.; Crouch, P.J.; White, A.R.; Barnham, K.J.; Roselt, P.D.; Noonan, W.; Binns, D.; Hicks, R.J.; Donnelly, P.S. Modification of Biodistribution and Brain Uptake of Copper Bis(thiosemicarbazonato) Complexes by the Incorporation of Amine and Polyamine Functional Groups. Inorg. Chem. 2019, 58, 4540–4552. [Google Scholar] [CrossRef]
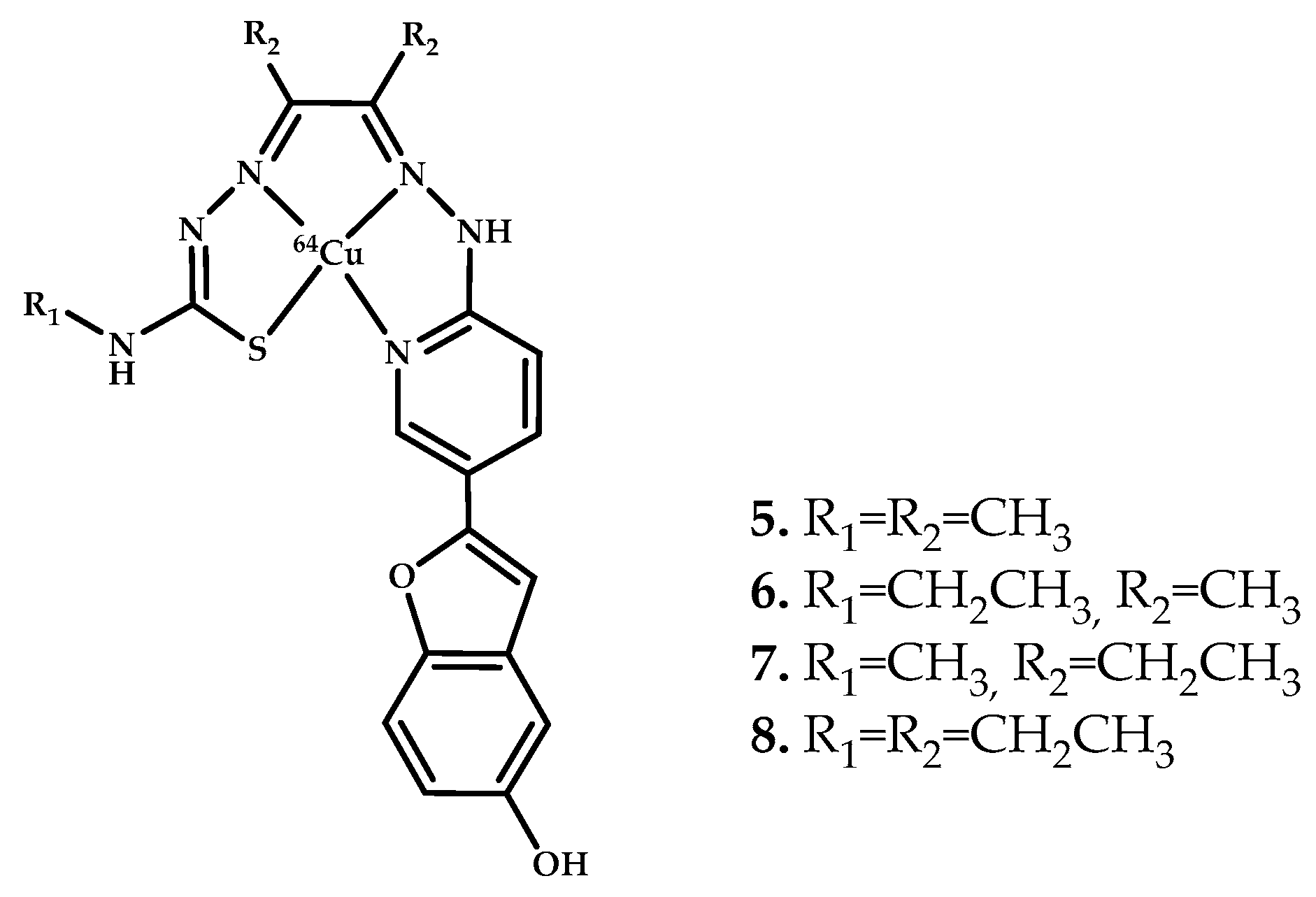
- Watanabe, H.; Kawasaki, A.; Sano, K.; Ono, M.; Saji, H. Synthesis and evaluation of copper-64 labeled benzofuran derivatives targeting β-amyloid aggregates. Bioorg. Med. Chem. 2016, 24, 3618–3623. [Google Scholar] [CrossRef]
- Bandara, N.; Sharma, A.K.; Krieger, S.; Schultz, J.W.; Han, B.H.; Rogers, B.E.; Mirica, L.M. Evaluation of 64Cu-Based Radiopharmaceuticals that Target Aβ Peptide Aggregates as Diagnostic Tools for Alzheimer’s Disease. J. Am. Chem. Soc. 2017, 139, 12550–12558. [Google Scholar] [CrossRef]
- Huang, Y.; Cho, H.-J.; Bandara, N.; Sun, L.; Tran, D.; Rogers, B.E.; Mirica, L.M. Metal-chelating benzothiazole multifunctional compounds for the modulation and 64Cu PET imaging of Aβ aggregation. Chem. Sci. 2020, 11, 7789–7799. [Google Scholar] [CrossRef]
- Sharma, A.K.; Pavlova, S.T.; Kim, J.; Finkelstein, D.; Hawco, N.J.; Rath, N.P.; Kim, J.; Mirica, L.M. Bifunctional Compounds for Controlling Metal-Mediated Aggregation of the Aβ42 Peptide. J. Am. Chem. Soc. 2012, 134, 6625–6636. [Google Scholar] [CrossRef]
- Sharma, A.K.; Kim, J.; Prior, J.T.; Hawco, N.J.; Rath, N.P.; Kim, J.; Mirica, L.M. Small Bifunctional Chelators That Do Not Disaggregate Amyloid β Fibrils Exhibit Reduced Cellular Toxicity. Inorg. Chem. 2014, 53, 11367–11376. [Google Scholar] [CrossRef]
- Banerjee, S.R.; Pomper, M.G. Clinical applications of Gallium-68. Appl. Radiat. Isot. 2013, 76, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Moerlein, S.M.; Welch, M.J. The chemistry of gallium and indium as related to radiopharmaceutical production. Int. J. Nucl. Med. Biol. 1981, 8, 277–287. [Google Scholar] [CrossRef]
- Zhou, Z.; Lu, Z.-R. Gadolinium-based contrast agents for magnetic resonance cancer imaging. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2013, 5, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Caravan, P.; Ellison, J.J.; McMurry, T.J.; Lauffer, R.B. Gadolinium(III) Chelates as MRI Contrast Agents: Structure, Dynamics, and Applications. Chem. Rev. 1999, 99, 2293–2352. [Google Scholar] [CrossRef]
- Martins, A.F.; Morfin, J.-F.; Kubíčková, A.; Kubíček, V.; Buron, F.; Suzenet, F.; Salerno, M.; Lazar, A.N.; Duyckaerts, C.; Arlicot, N.; et al. PiB-Conjugated, Metal-Based Imaging Probes: Multimodal Approaches for the Visualization of β-Amyloid Plaques. ACS Med. Chem. Lett. 2013, 4, 436–440. [Google Scholar] [CrossRef]
- Martins, A.F.; Morfin, J.-F.; Geraldes, C.F.G.C.; Tóth, É. Gd3+ complexes conjugated to Pittsburgh compound B: Potential MRI markers of β-amyloid plaques. J. Biol. Inorg. Chem. 2014, 19, 281–295. [Google Scholar] [CrossRef]
- Bort, G.; Catoen, S.; Borderies, H.; Kebsi, A.; Ballet, S.; Louin, G.; Port, M.; Ferroud, C. Gadolinium-based contrast agents targeted to amyloid aggregates for the early diagnosis of Alzheimer’s disease by MRI. Eur. J. Med. Chem. 2014, 87, 843–861. [Google Scholar] [CrossRef]
- Watanabe, H.; Ono, M.; Iikuni, S.; Yoshimura, M.; Matsumura, K.; Kimura, H.; Saji, H. A 68Ga complex based on benzofuran scaffold for the detection of β-amyloid plaques. Bioorg. Med. Chem. Lett. 2014, 24, 4834–4837. [Google Scholar] [CrossRef]
- Cressier, D.; Dhilly, M.; Cao Pham, T.T.; Fillesoye, F.; Gourand, F.; Maïza, A.; Martins, A.F.; Morfin, J.-F.; Geraldes, C.F.G.C.; Tóth, É.; et al. Gallium-68 Complexes Conjugated to Pittsburgh Compound B: Radiolabeling and Biological Evaluation. Mol. Imaging Biol. 2016, 18, 334–343. [Google Scholar] [CrossRef]
- Zha, Z.; Song, J.; Choi, S.R.; Wu, Z.; Ploessl, K.; Smith, M.; Kung, H. 68Ga-Bivalent Polypegylated Styrylpyridine Conjugates for Imaging Aβ Plaques in Cerebral Amyloid Angiopathy. Bioconjug. Chem. 2016, 27, 1314–1323. [Google Scholar] [CrossRef]
- Chauhan, K.; Datta, A.; Adhikari, A.; Chuttani, K.; Singh, A.K.; Mishra, A.K. 68Ga based probe for Alzheimer’s disease: Synthesis and preclinical evaluation of homodimeric chalcone in β-amyloid imaging. Org. Biomol. Chem. 2014, 12, 7328. [Google Scholar] [CrossRef] [PubMed]
- Asti, M.; Ferrar, E.; Croci, S.; Atti, G.; Rubagotti, S.; Iori, M.; Capponi, P.C.; Zerbini, A.; Saladini, M.; Versari, A. Synthesis and Characterization of 68Ga-Labeled Curcumin and Curcuminoid Complexes as Potential Radiotracers for Imaging of Cancer and Alzheimer’s Disease. Inorg. Chem. 2014, 53, 4922–4933. [Google Scholar] [CrossRef] [PubMed]
- Rubagotti, S.; Croci, S.; Ferrari, E.; Iori, M.; Capponi, P.C.; Lorenzini, L.; Calzà, L.; Versari, A.; Asti, M. Affinity of (nat/68)Ga-Labelled Curcumin and Curcuminoid Complexes for β-Amyloid Plaques: Towards the Development of New Metal-Curcumin Based Radiotracers. Int. J. Mol. Sci. 2016, 17, 1480. [Google Scholar] [CrossRef] [PubMed]
- Lange, J.L.; Hayne, D.J.; Roselt, P.; McLean, C.A.; White, J.M.; Donnelly, P.S. A gallium(III) Schiff base-curcumin complex that binds to amyloid-β plaques. J. Inorg. Biochem. 2016, 162, 274–279. [Google Scholar] [CrossRef]
- Orteca, G.; Sinnes, J.-P.; Rubagotti, S.; Iori, M.; Capponi, P.C.; Piel, M.; Rösch, F.; Ferrari, E.; Asti, M. Gallium-68 and scandium-44 labelled radiotracers based on curcumin structure linked to bifunctional chelators: Synthesis and characterization of potential PET radiotracers. J. Inorg. Biochem. 2020, 204, 110954. [Google Scholar]
- Gniazdowska, E.; Koźmiński, P.; Halik, P.; Bajda, M.; Czarnecka, K.; Mikiciuk-Olasik, E.; Masłowska, K.; Rogulski, Z.; Cheda, Ł.; Kilian, K.; et al. Synthesis, physicochemical and biological evaluation of tacrine derivative labeled with technetium-99m and gallium-68 as a prospective diagnostic tool for early diagnosis of Alzheimer’s disease. Bioorg. Chem. 2019, 91, 103136. [Google Scholar] [CrossRef]
- Singh, S.; Khar, A. Biological Effects of Curcumin and Its Role in Cancer Chemoprevention and Therapy. AntiCancer Agents Med. Chem. 2006, 6, 933–946. [Google Scholar] [CrossRef]
- Bartik, L.; Whitfield, G.K.; Kaczmarska, M.; Lowmiller, C.L.; Moffet, E.W.; Furmick, J.K.; Hernandez, Z.; Haussler, C.A.; Haussler, M.R.; Jurutka, P.W. Curcumin: A novel nutritionally derived ligand of the vitamin D receptor with implications for colon cancer chemoprevention. J. Nutr. Biochem. 2010, 21, 1153–1161. [Google Scholar] [CrossRef]
- Chen, M.; Du, Z.Y.; Zheng, X.; Li, D.L.; Zhou, R.P.; Zhang, K. Use of curcumin in diagnosis, prevention, and treatment of Alzheimer’s disease. Neural Regen. Res. 2018, 13, 742–752. [Google Scholar]
- Sidiqi, A.; Wahl, D.; Lee, S.; Ma, D.; To, E.; Cui, J.; To, E.; Beg, M.F.; Sarunic, M.; Matsubara, J.A. In vivo Retinal Fluorescence Imaging with Curcumin in an Alzheimer Mice Model. Front. Neurosci. 2020, 14, 713. [Google Scholar] [CrossRef]
- McCrate, O.A.; Zhou, X.; Cegelski, L. Curcumin as an amyloid-indicatordye in E. coli. Chem. Commun. 2013, 49, 4193–4195. [Google Scholar] [CrossRef] [PubMed]
- Thapa, A.; Jett, S.D.; Chi, E.Y. Curcumin Attenuates Amyloid-β Aggregate Toxicity and Modulates Amyloid-β Aggregation Pathway. ACS Chem. Neurosci. 2016, 7, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Teoh, C.L.; Su, D.; Sahu, S.; Yun, S.-W.; Drummond, E.; Prelli, F.; Lim, S.; Cho, S.; Ham, S.; Wisniewski, T.; et al. Chemical Fluorescent Probe for Detection of Aβ Oligomers. J. Am. Chem. Soc. 2015, 137, 13503–13509. [Google Scholar] [CrossRef] [PubMed]
- Ono, K.; Hasegawa, K.; Naiki, H.; Yamada, M.J. Curcumin has potent anti-amyloidogenic effects for Alzheimer’s β-amyloid fibrils in vitro. Neurosci. Res. 2004, 75, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Cheng, R.; Fu, H.; Yang, J.; Kumar, M.; Lu, J.; Xu, Y.; Liang, S.H.; Cui, M.; Ran, C. Half-curcumin analogues as PET imaging probes for amyloid beta species. Chem. Comm. 2019, 55, 3630–3633. [Google Scholar] [CrossRef] [PubMed]
- Sherin, S.; Balachandran, S.; Abraham, A. Curcumin incorporated titanium dioxide nanoparticles as MRI contrasting agent for early diagnosis of atherosclerosis- rat model. Vet. Anim. Sci. 2020, 10, 100090. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Haratake, M.; Mori, H.; Nakayama, M. Novel chalcones as probes for in vivo imaging of beta-amyloid plaques in Alzheimer’s brains. Bioorg. Med. Chem. 2007, 15, 6802–6809. [Google Scholar] [CrossRef]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin Inhibits Formation of Amyloid β Oligomers and Fibrils, Binds Plaques, and Reduces Amyloid in Vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar] [CrossRef]
- Jeyarasasingam, G.; Yeluashvili, M.; Quik, M. Tacrine, a reversible acetylcholinesterase inhibitor, induces myopathy. NeuroReport 2000, 11, 1173–1176. [Google Scholar] [CrossRef]
- Meszaros, L.K.; Dose, A.; Biagini, S.C.G.; Blower, P.J. Synthesis and evaluation of analogues of HYNIC as bifunctional chelators for technetium. Dalton Trans. 2011, 40, 6260–6267. [Google Scholar] [CrossRef]
- Fissers, J.; Waldron, A.-M.; De Vijlder, T.; Van Broeck, B.; Pemberton, D.J.; Mercken, M.; Van Der Veken, P.; Joossens, J.; Augustyns, K.; Dedeurwaerdere, S.; et al. Synthesis and Evaluation of a Zr-89-Labeled Monoclonal Antibody for Immuno-PET Imaging of Amyloid-β Deposition in the Brain. Mol. Imaging Biol. 2016, 18, 598–605. [Google Scholar] [PubMed]
- Jia, J.; Cui, M.; Dai, J.; Wang, X.; Ding, Y.-S.; Jia, H.; Liu, B. 99mTc-labeled benzothiazole and stilbene derivatives as imaging agents for Aβ plaques in cerebral amyloid angiopathy. Med. Chem. Comm. 2014, 5, 153–158. [Google Scholar] [CrossRef]
- Li, Z.; Cui, M.; Dai, J.; Wang, X.; Yu, P.; Yang, Y.; Jia, J.; Fu, H.; Ono, M.; Jia, H.; et al. Novel Cyclopentadienyl Tricarbonyl Complexes of 99mTc Mimicking Chalcone as Potential Single-Photon Emission Computed Tomography Imaging Probes for β-Amyloid Plaques in Brain. J. Med. Chem. 2013, 56, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Cui, M.; Jin, B.; Wang, X.; Li, Z.; Yu, P.; Jia, J.; Fu, H.; Jia, H.; Liu, B. 99mTc-labeled dibenzylideneacetone derivatives as potential SPECT probes for in vivo imaging of β-amyloid plaque. Eur. J. Med. Chem. 2013, 64, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Iikuni, S.; Ono, M.; Watanabe, H.; Matsumura, K.; Yoshimura, M.; Harada, N.; Kimura, H.; Nakayama, M.; Saji, H. Enhancement of Binding Affinity for Amyloid Aggregates by Multivalent Interactions of 99mTc-Hydroxamamide Complexes. Mol. Pharm. 2014, 11, 1132–1139. [Google Scholar] [CrossRef]
- Iikuni, S.; Ono, M.; Watanabe, H.; Matsumura, K.; Yoshimura, M.; Kimura, H.; Ishibashi-Ueda, H.; Okamoto, Y.; Ihara, M.; Saji, H. Imaging of Cerebral Amyloid Angiopathy with Bivalent 99mTc-Hydroxamamide Complexes. Sci. Rep. 2016, 6, 25990. [Google Scholar] [CrossRef]
- Hayne, D.J.; North, A.J.; Fodero-Tavoletti, M.; White, J.M.; Hung, L.W.; Rigopoulos, A.; McLean, C.A.; Adlard, P.A.; Ackermann, U.; Tochon-Danguy, H.; et al. Rhenium and technetium complexes that bind to amyloid-β plaques. Dalton Trans. 2015, 44, 4933–4944. [Google Scholar] [CrossRef]
- Wang, X.; Cui, M.; Jia, J.; Liu, B. 99mTc-labeled-2-arylbenzoxazole derivatives as potential Aβ imaging probes for single-photon emission computed tomography. Eur. J. Med. Chem. 2015, 89, 331–339. [Google Scholar] [CrossRef]
- Jia, J.; Cui, M.; Dai, J.; Liu, B. 99mTc(CO)3-Labeled Benzothiazole Derivatives Preferentially Bind Cerebrovascular Amyloid: Potential Use as Imaging Agents for Cerebral Amyloid Angiopathy. Mol. Pharm. 2015, 12, 2937–2946. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, P.; Yang, Y.; Hou, Y.; Peng, C.; Liang, Z.; Lu, J.; Chen, B.; Dai, J.; Liu, B.; et al. 99mTc-Labeled 2-Arylbenzothiazoles: Aβ Imaging Probes with Favorable Brain Pharmacokinetics for Single-Photon Emission Computed Tomography. Bioconj. Chem. 2016, 27, 2493–2504. [Google Scholar] [CrossRef]
- Hayne, D.J.; White, J.M.; McLean, C.A.; Villemagne, V.L.; Barnham, K.J.; Donnelly, P.S. Synthesis of Oxorhenium(V) and Oxotechnetium(V) Complexes That Bind to Amyloid-β Plaques. Inorg. Chem. 2016, 55, 7944–7953. [Google Scholar] [CrossRef] [PubMed]
- Kiritsis, C.; Mavroidi, B.; Shegani, A.; Palamaris, L.; Loudos, G.; Sagnou, M.; Pirmettis, I.; Papadopoulos, M.; Pelecanou, M. 2-(4′-Aminophenyl)benzothiazole Labeled with 99mTc-Cyclopentadienyl for Imaging β-Amyloid Plaques. ACS Med. Chem. Lett. 2017, 8, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Iikuni, S.; Ono, M.; Tanimura, K.; Watanabe, H.; Yoshimuraa, M.; Sajia, H. Synthesis and biological evaluation of novel technetium-99m-labeled phenylquinoxaline derivatives as single photon emission computed tomography imaging probes targeting β-amyloid plaques in Alzheimer’s disease. RSC Adv. 2017, 7, 20582–20590. [Google Scholar] [CrossRef]
- Fletcher, S.P.; Noor, A.; Hickey, J.L.; McLean, C.A.; White, J.M.; Donnelly, P.S. Rhenium and technetium complexes of thioamide derivatives of pyridylhydrazine that bind to amyloid-β plaques. J. Biol. Inorg. Chem. 2018, 23, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Molavipordanjani, S.; Emami, S.; Mardanshahi, A.; Amiri, F.T.; Noaparast, Z.; Hosseinimehr, S.J. Novel 99m Tc-2-arylimidazo[2,1-b]benzothiazole derivatives as SPECT imaging agents for amyloid-β plaques. Eur. J. Med. Chem. 2019, 175, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Sagnou, M.; Mavroidi, B.; Shegani, A.; Paravatou-Petsotas, M.; Raptopoulou, C.; Psycharis, V.; Pirmettis, I.; Papadopoulos, M.S.; Pelecanou, M. Remarkable Brain Penetration of Cyclopentadienyl M(CO)3+ (M = 99mTc, Re) Derivatives of Benzothiazole and Benzimidazole Paves the Way for Their Application as Diagnostic, with Single-Photon-Emission Computed Tomography (SPECT), and Therapeutic Agents for Alzheimer’s Disease. J. Med. Chem. 2019, 62, 2638–2650. [Google Scholar] [PubMed]
- Jokar, S.; Behnammanesh, H.; Erfani, M.; Sharifzadeh, M.; Gholami, M.; Sabzevari, O.; Amini, M.; Geramifae, P.; Hajiramezanali, M.; Beiki, D. Synthesis, biological evaluation and preclinical study of a novel 99mTc-peptide: A targeting probe of amyloid-β plaques as a possible diagnostic agent for Alzheimer’s disease. Bioorg. Chem. 2020, 99, 103857. [Google Scholar] [CrossRef]

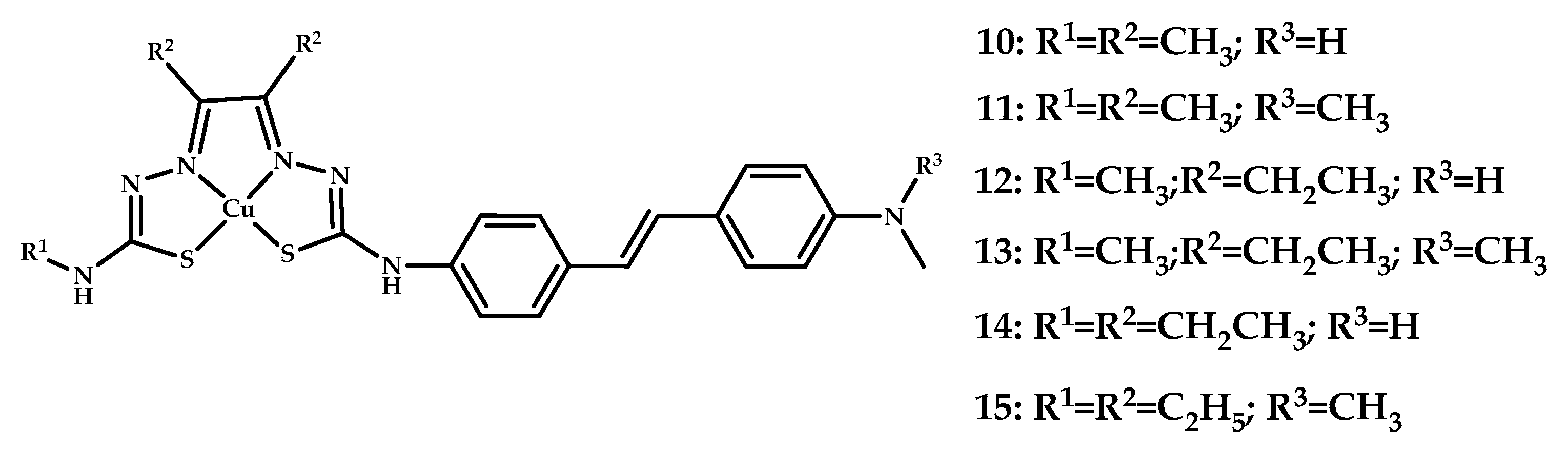
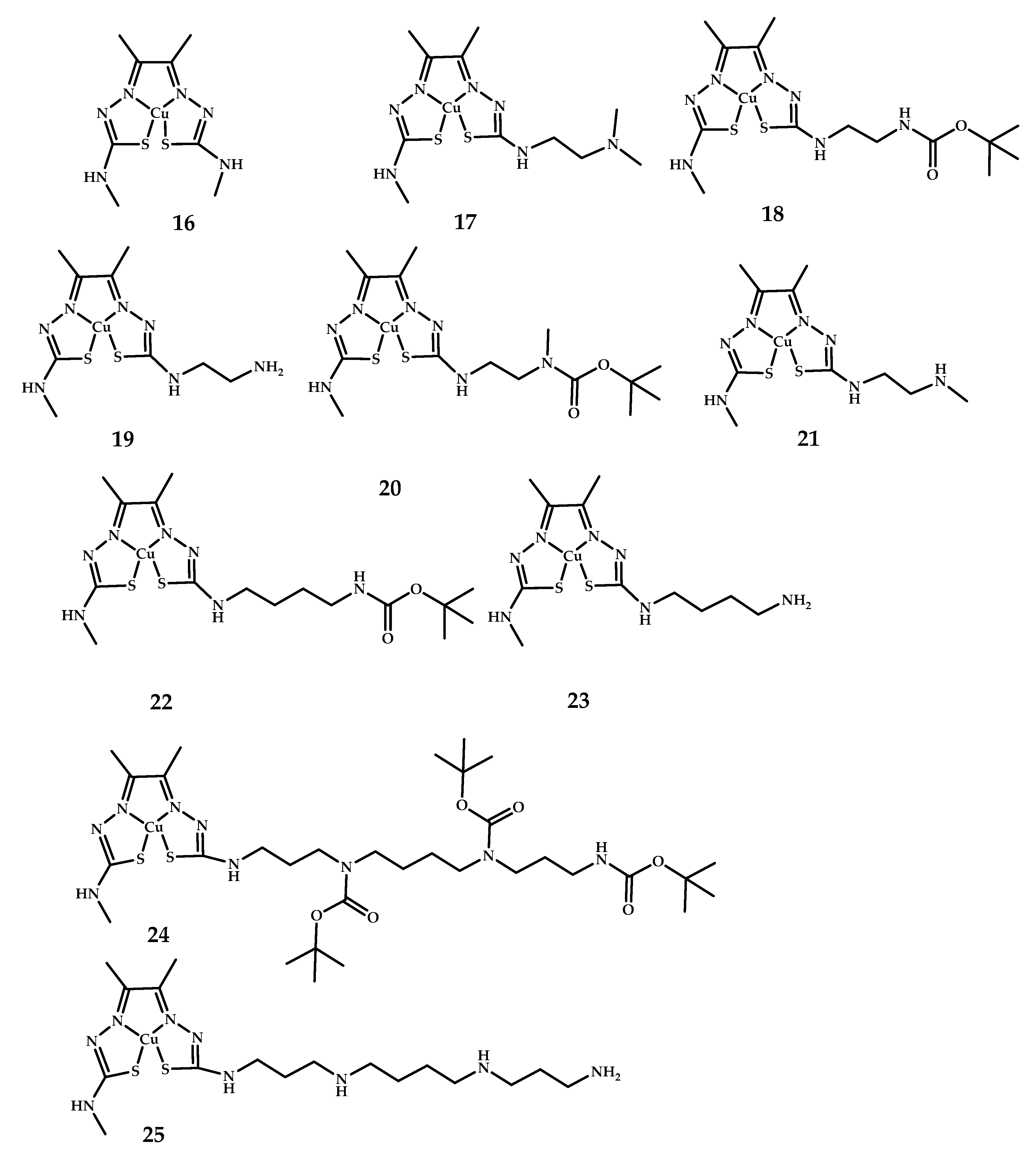
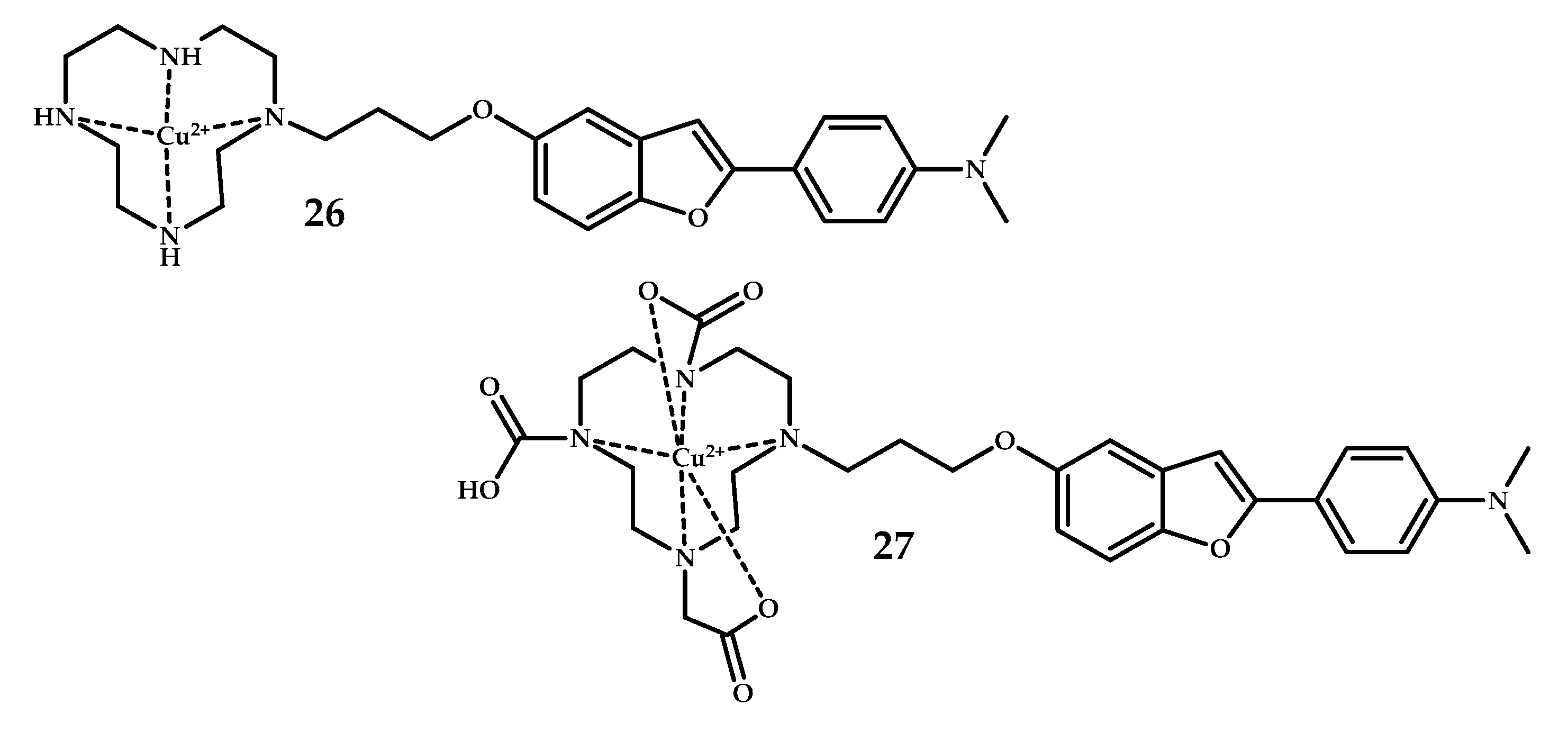

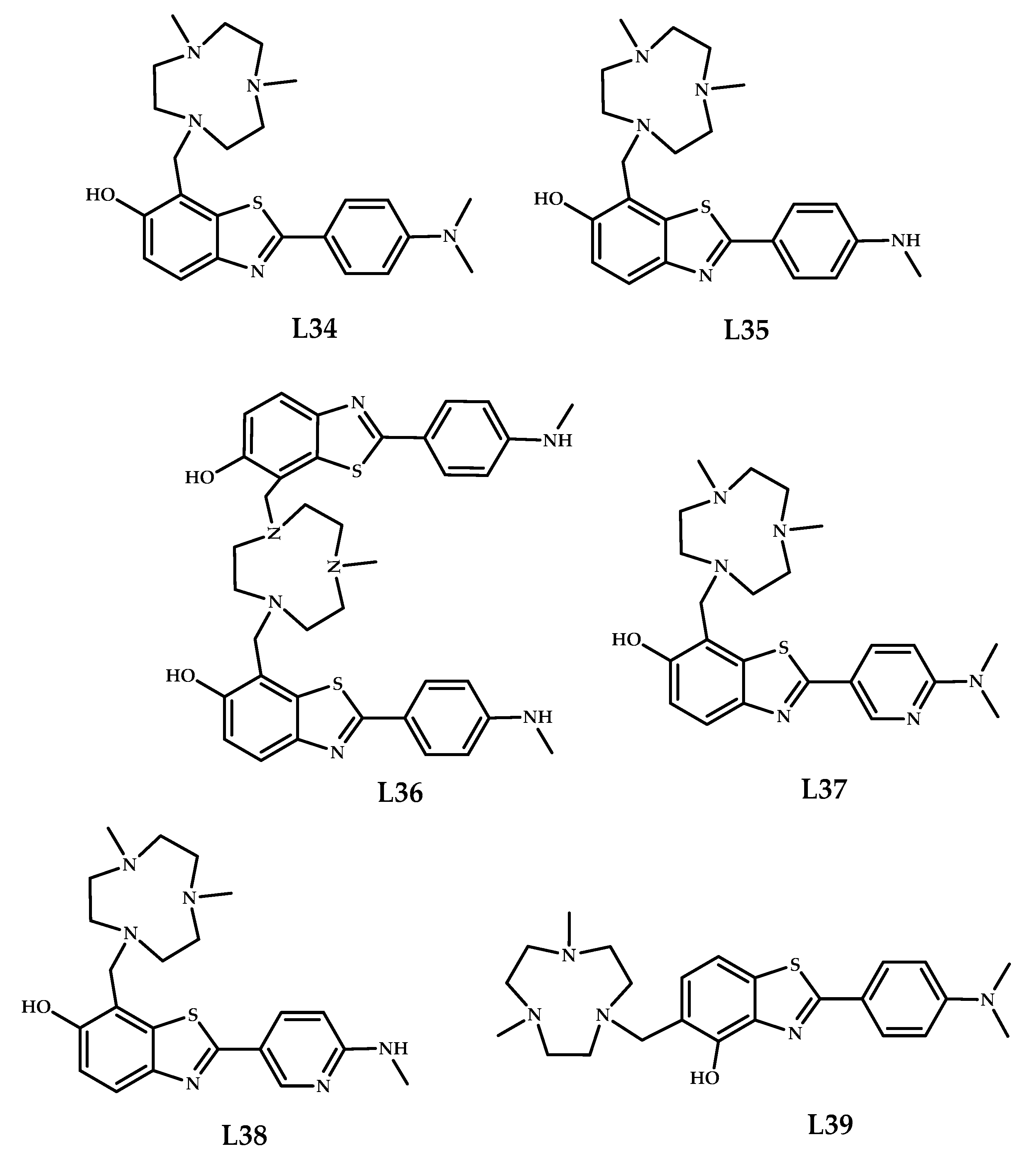

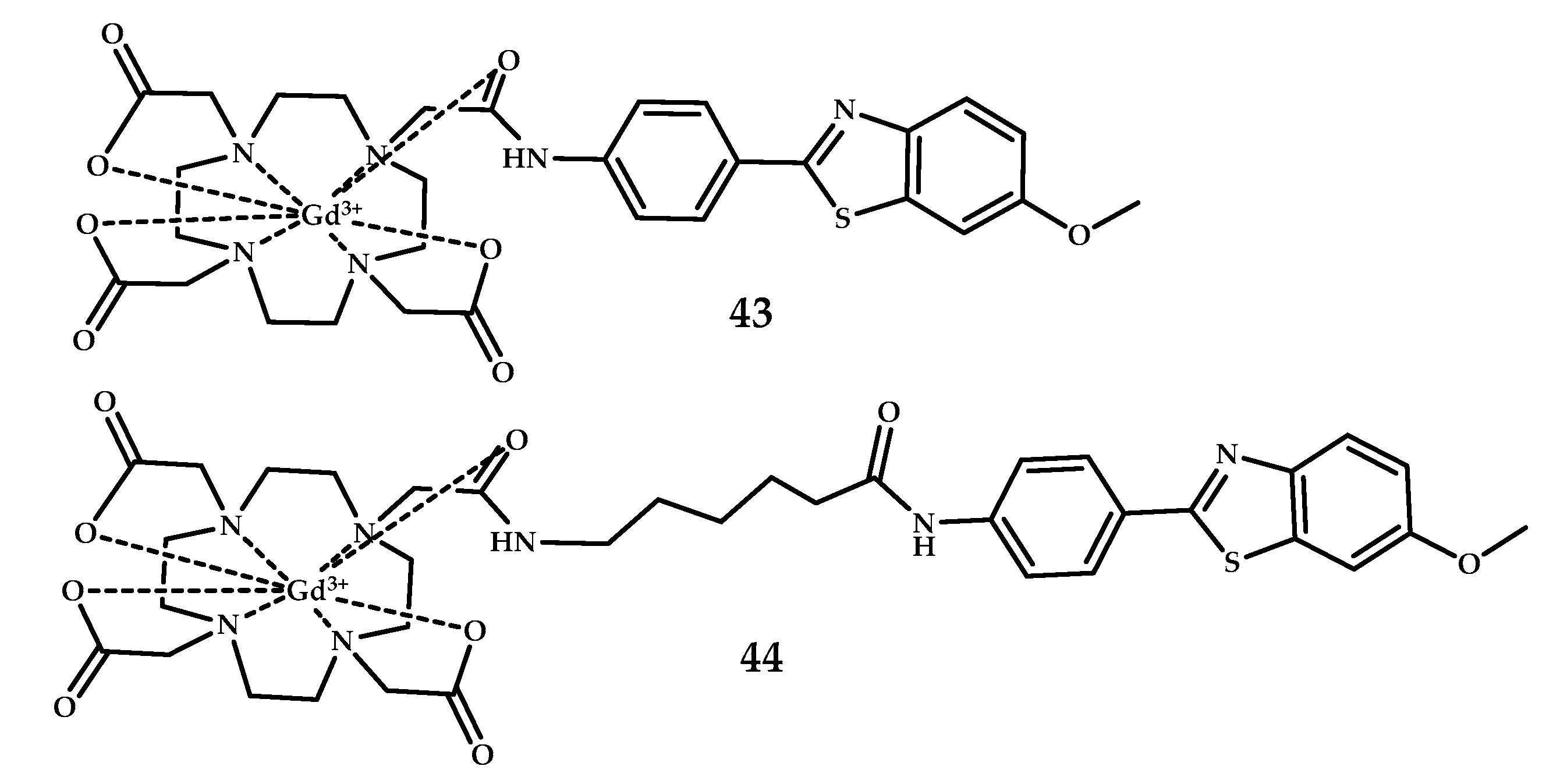
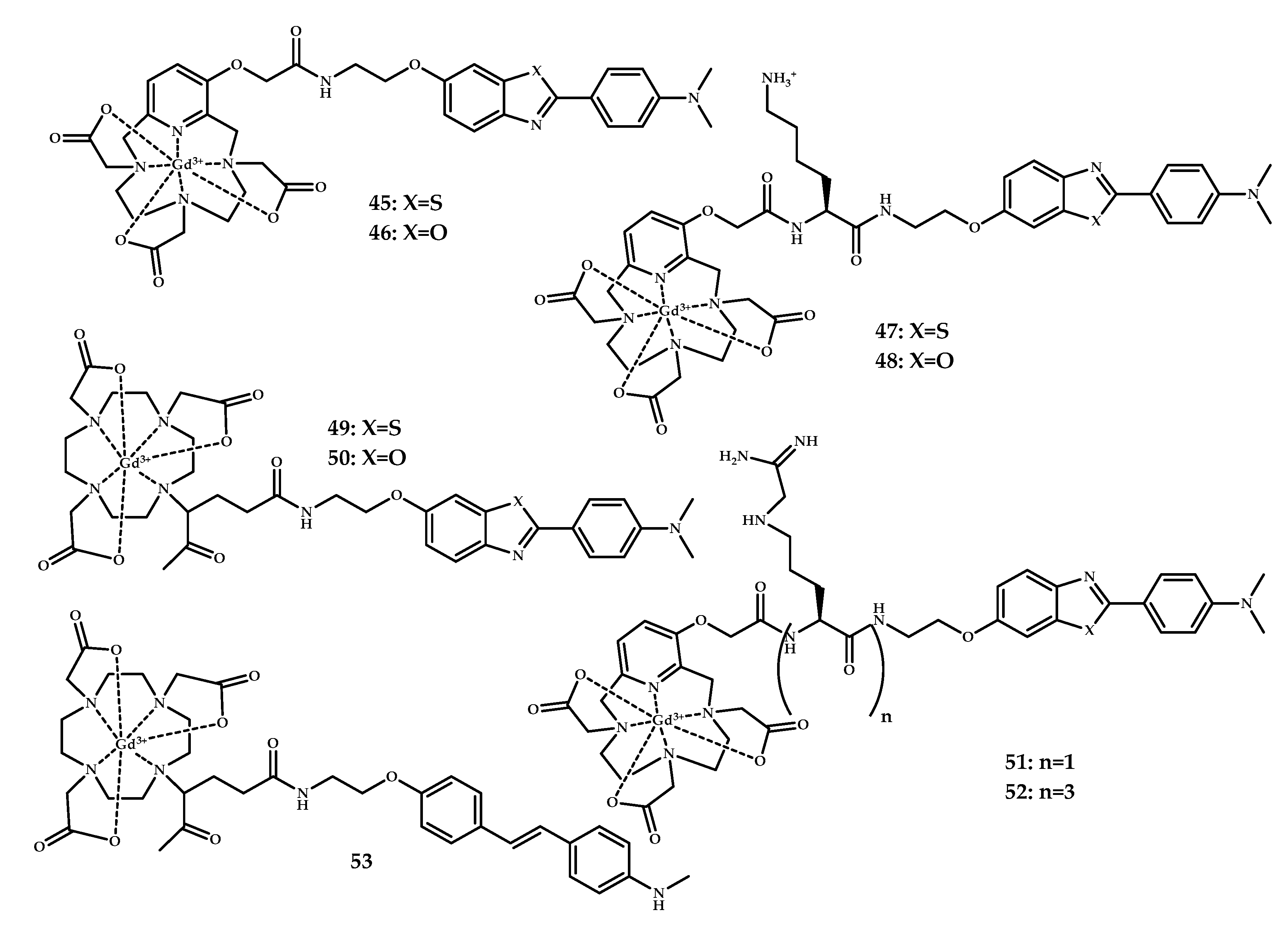
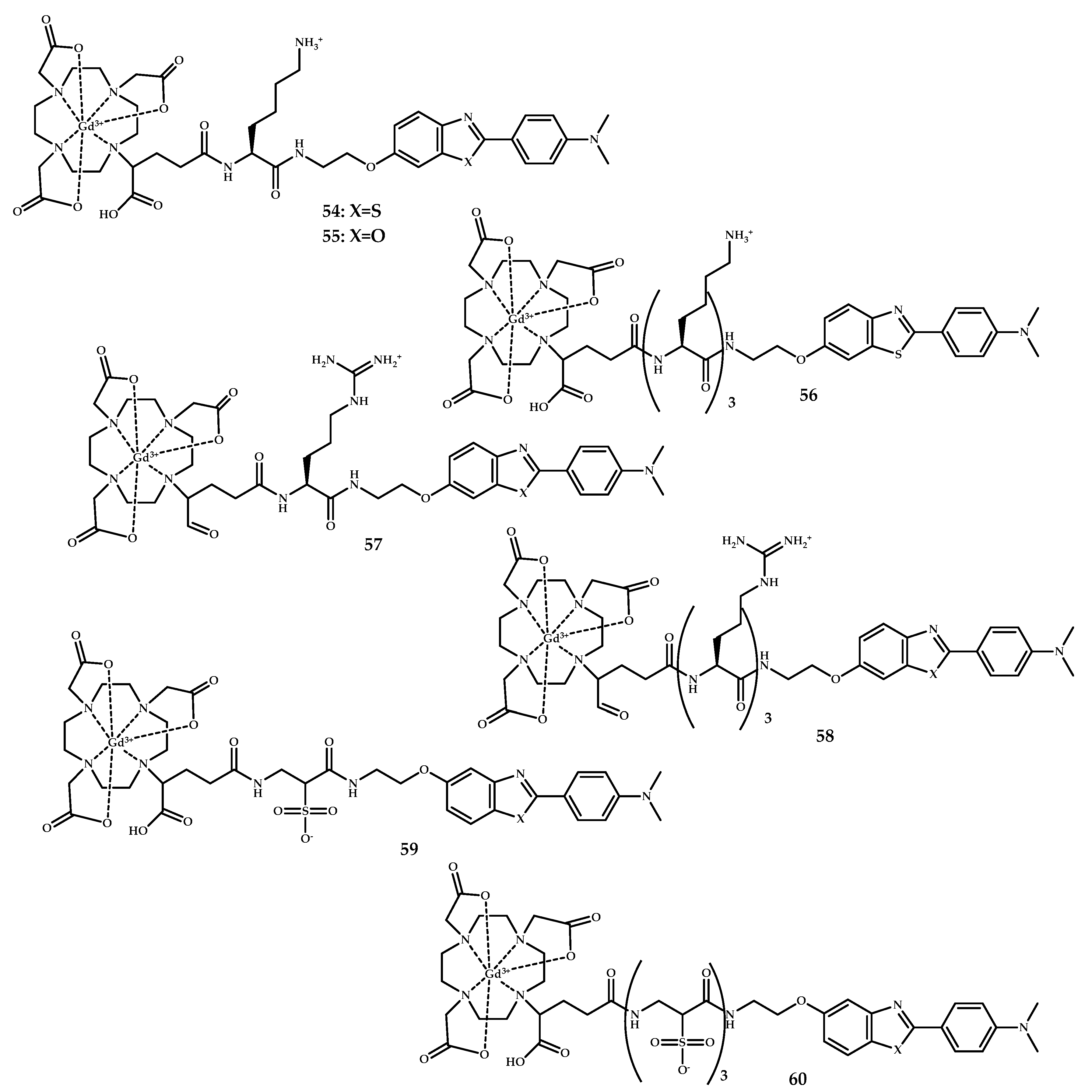
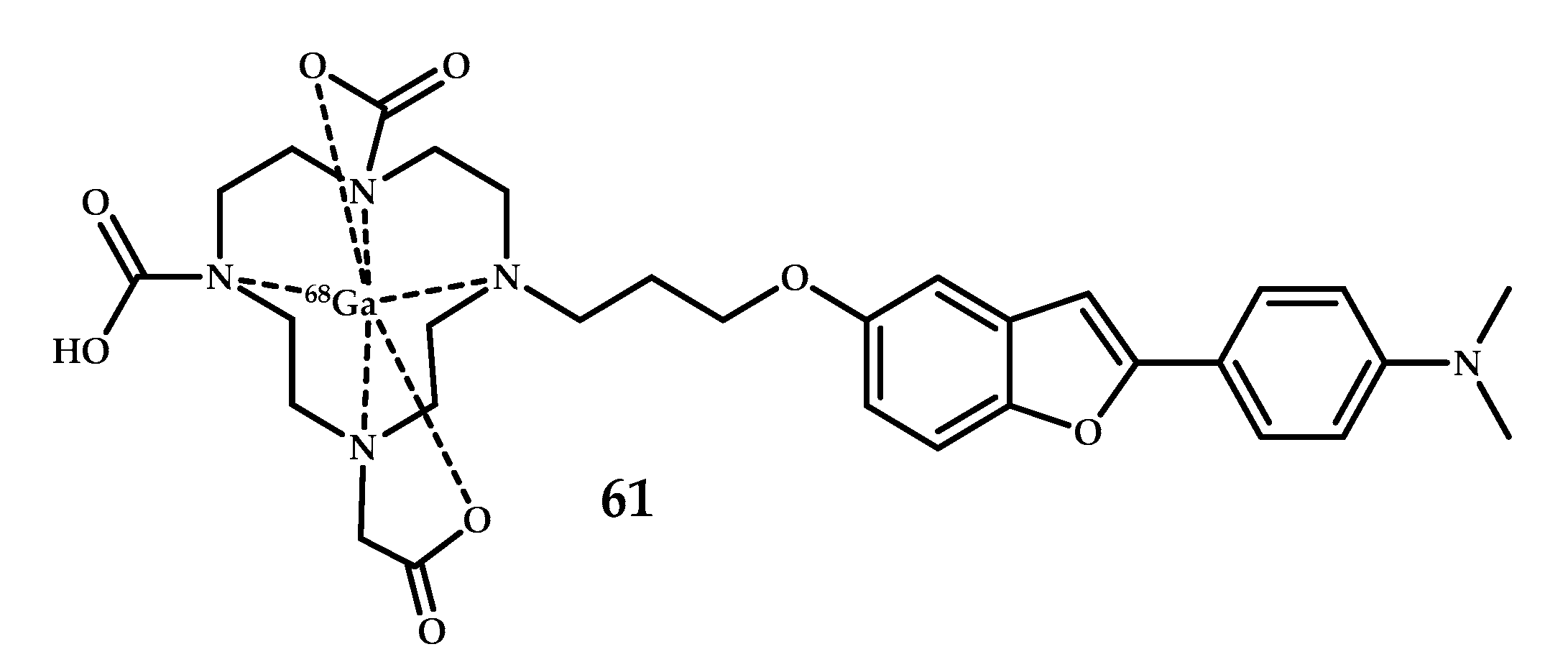

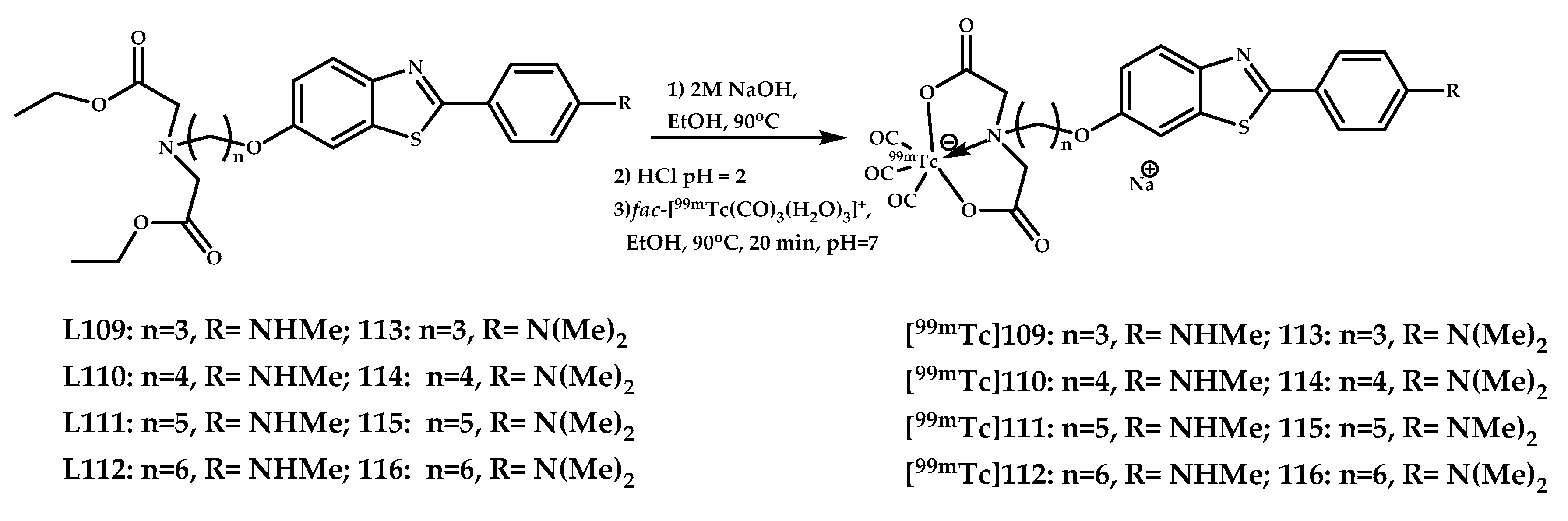
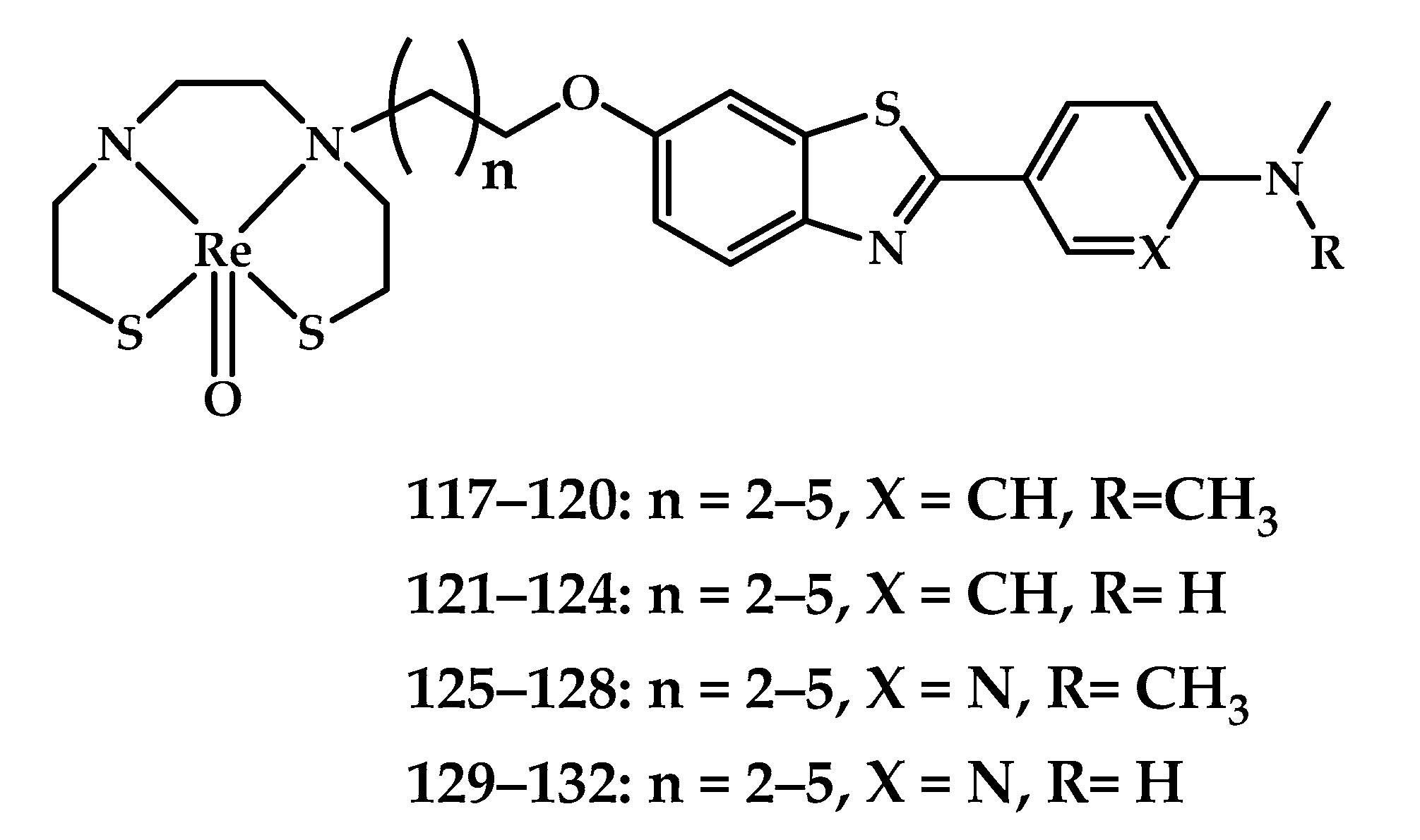
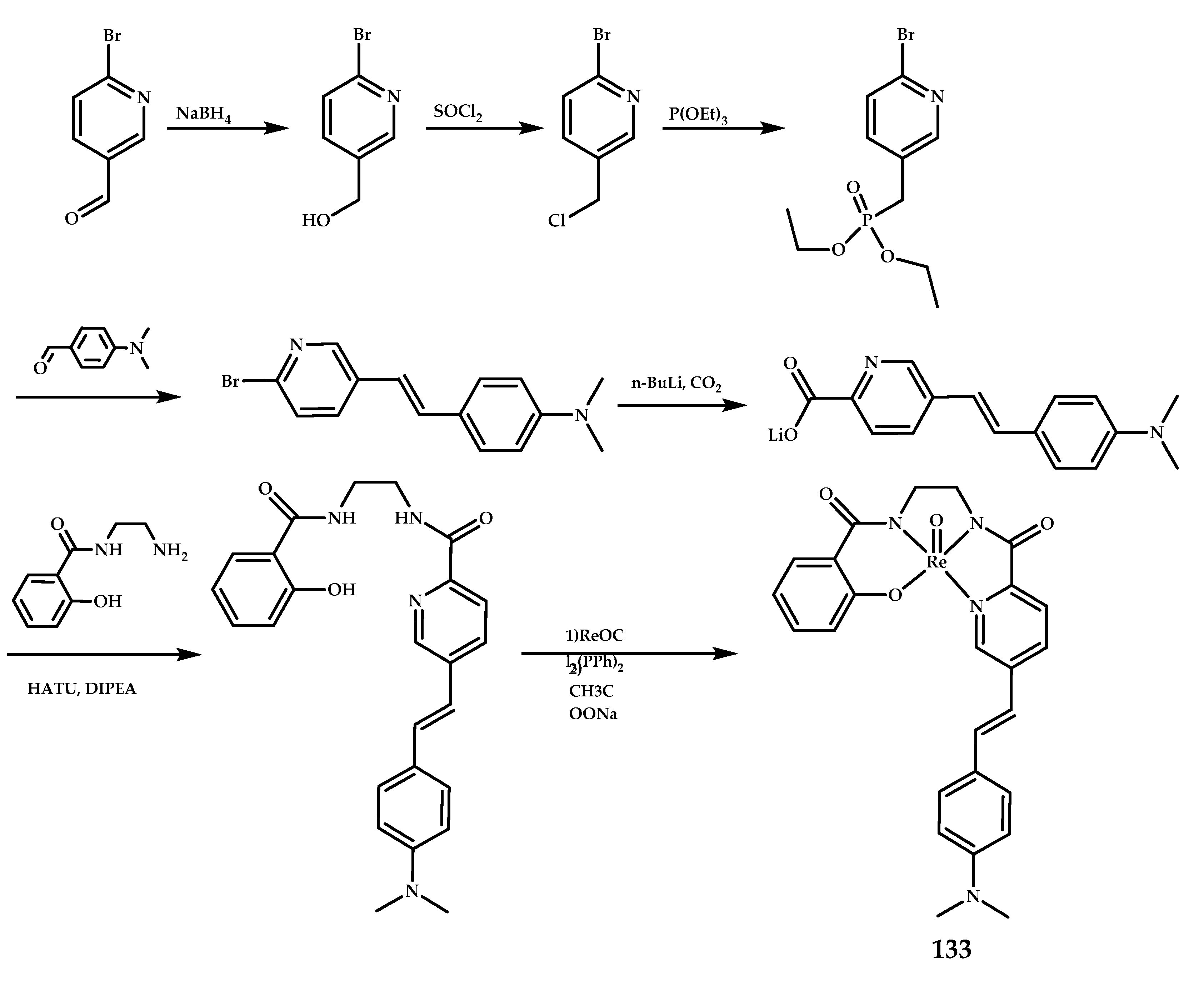
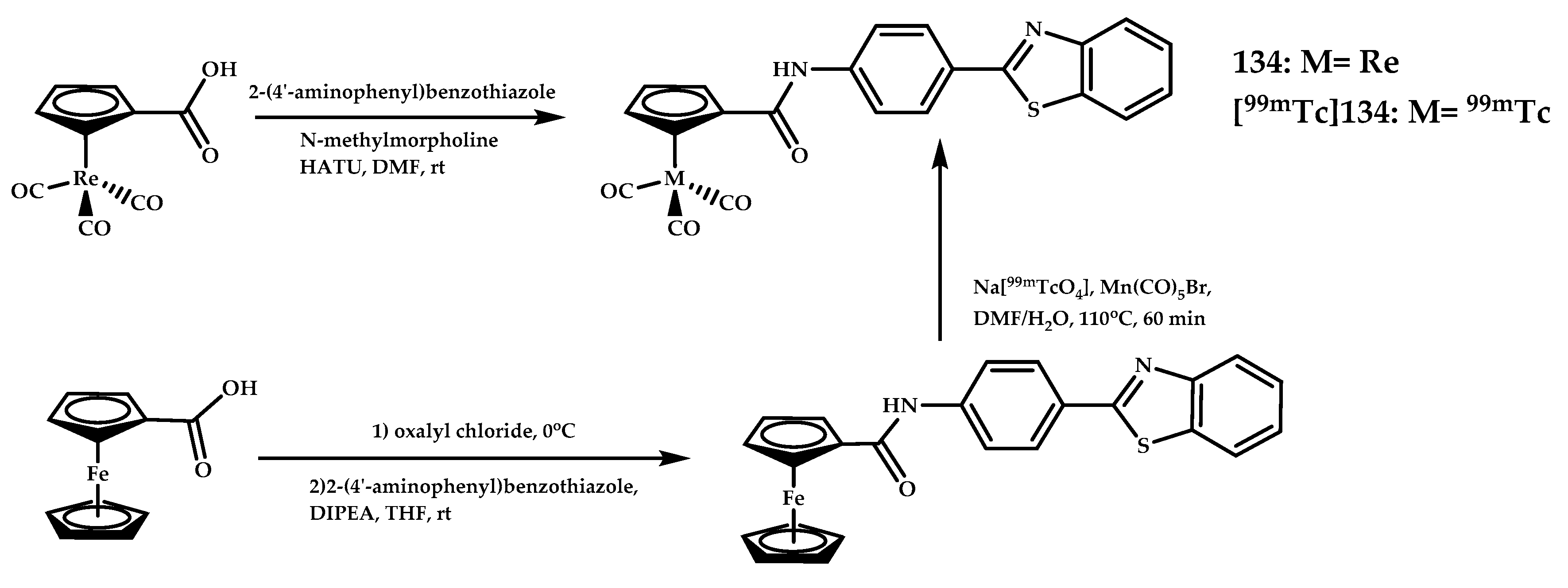
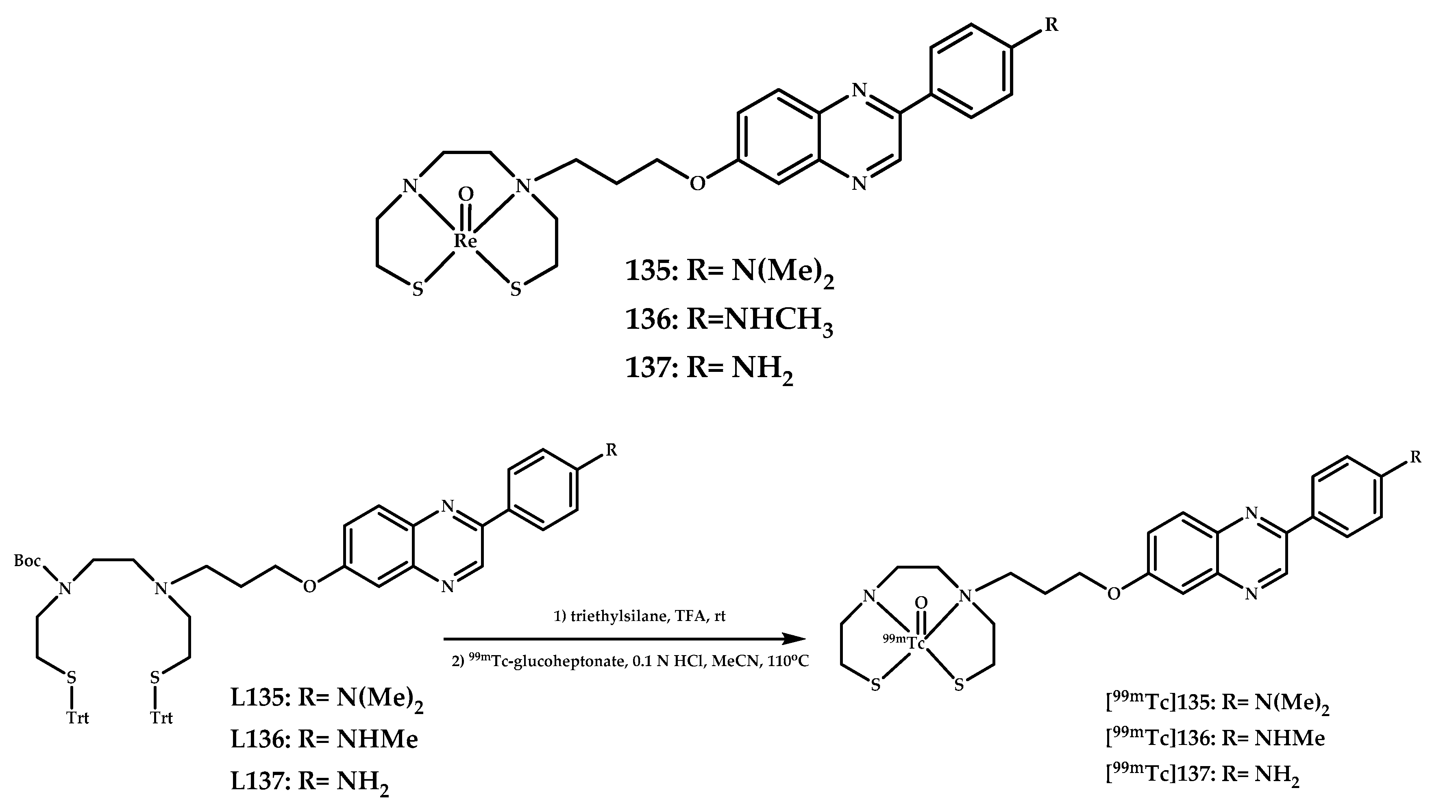
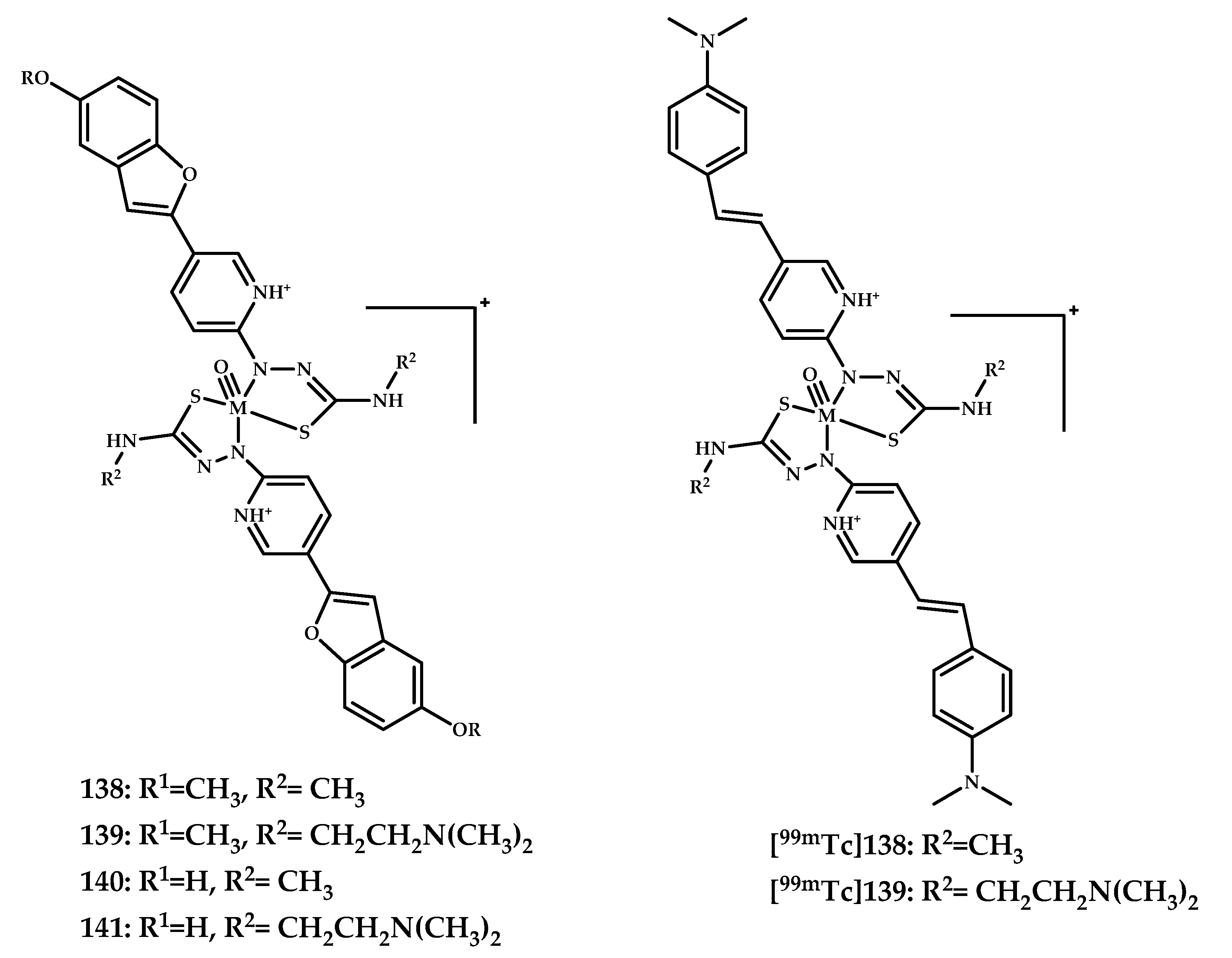
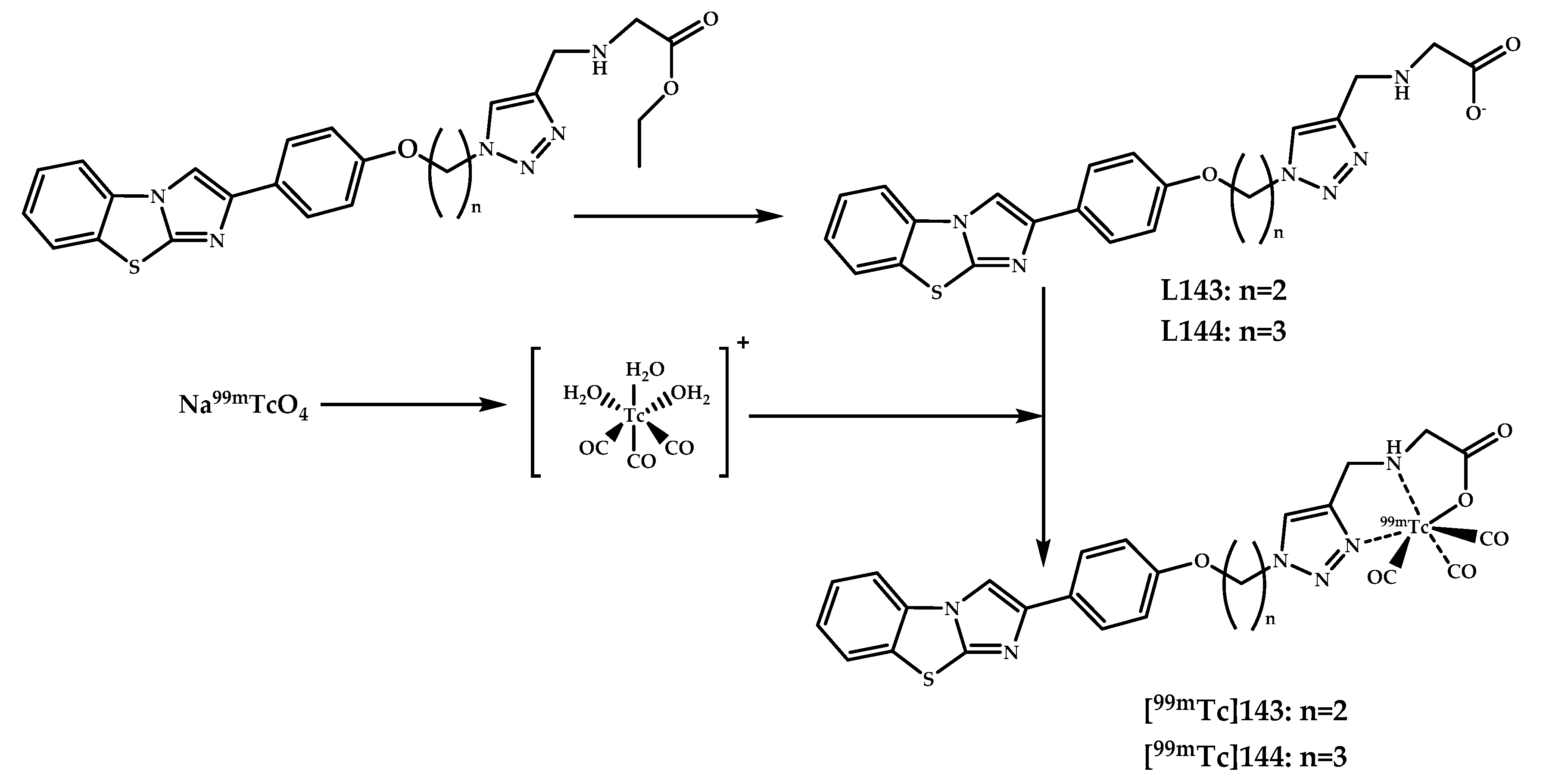
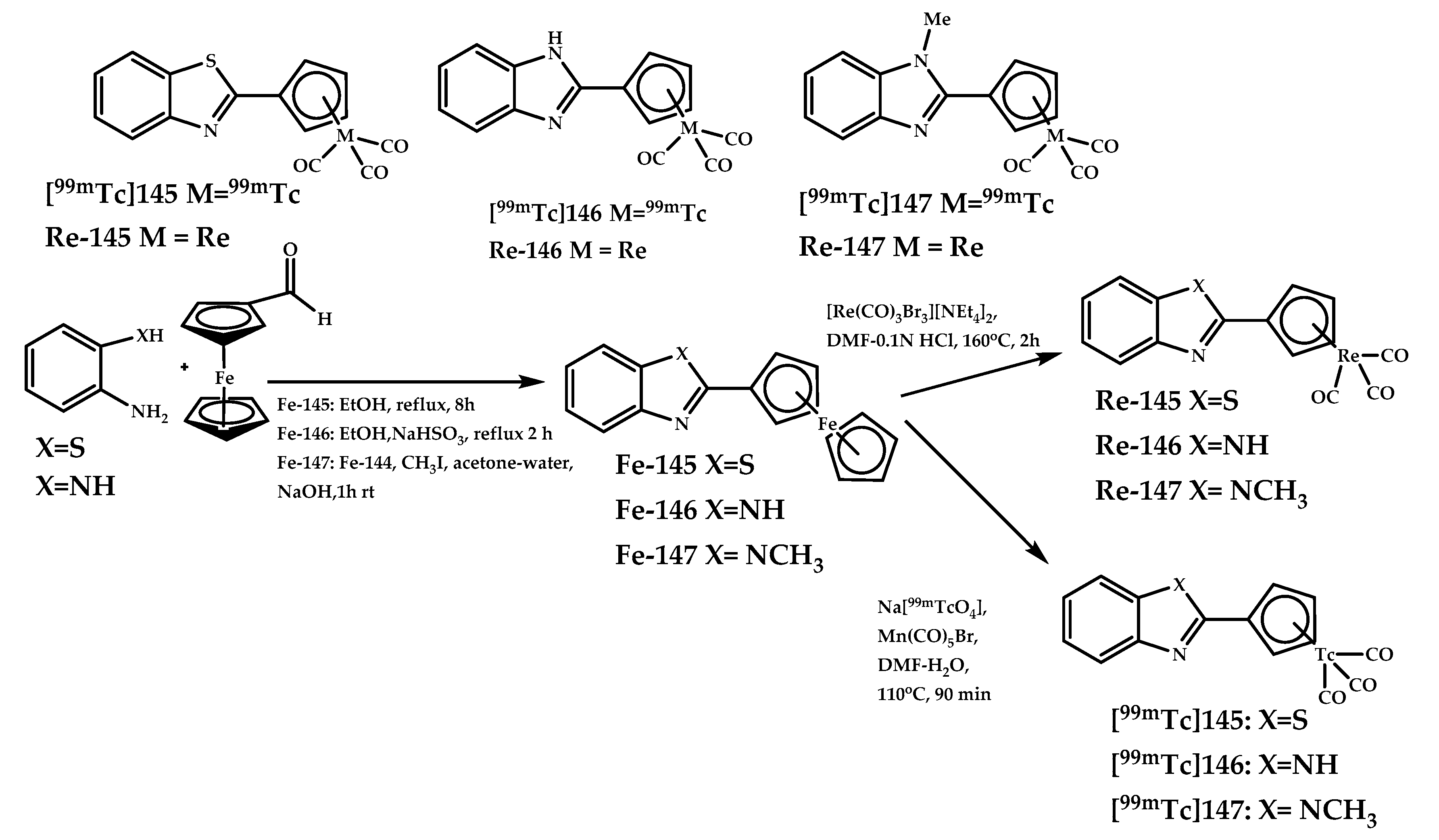
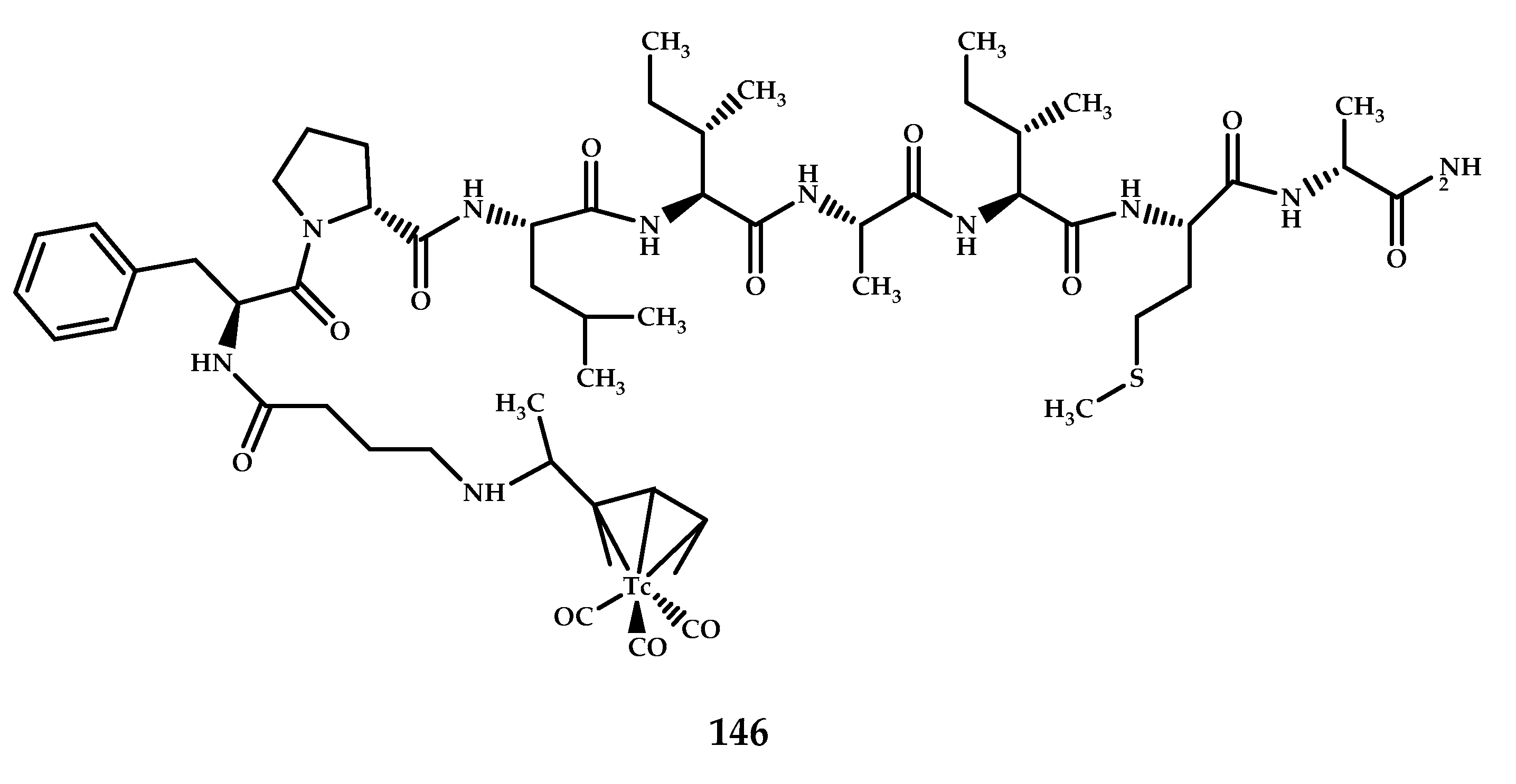
| Coordination Compound Number | Brain Uptake, ID/g, 2 min Post-Injection, % | Brain2min/60min (*Brain2min/30min) Ratio | Brain Tissue Experiments | Aβ Binding Moiety | Reference |
|---|---|---|---|---|---|
| Cu(ATSM)-based coordination compounds | |||||
| 1 | 2.5 ± 0.6 (APP/PS1 transgenic mice) 1.7 ± 0.6 (Wild-type mice) 7 min after injection | - | Epi-fluorescence microscopy of AD human brain sections | Stilbene | [39] |
| 2–4 | 1.11 ± 0.20 | 2.92 * | Epi-fluorescence microscopy of AD human brain sections | 2-benzothiazole,3,4-styrylpyridine | [40] |
| 5–8 | 1.39 ± 0.06 1.06 ± 0.43 0.77 ± 0.19 1.54 ± 0.60 | 1.31 * 2.16 * 1.05 * | Elemental composition of AD human brain tissue using LA-ICP-MS | Benzofuran | [41] |
| 9 | - | - | Epi-fluorescence microscopy of AD human brain tissue (ligand) | Stilbene | [42] |
| 10–15 | 2.2 ± 0.6 1.1 ± 0.2 | 6.47 5 | Epi-fluorescence microscopy of AD human brain sections | Styrylpyridine | [43] |
| 16–25 | 4.41 ± 0.23 (23 h Post-injection similar) | - | PET imagine of BALB/c mice | - | [44] |
| Other metal-chelating moieties | |||||
| 26, 27 | 0.33 ± 0.12 0.36 ± 0.10 | 1.83 2.11 | Fluorescent staining using brain sections from a Tg2576 mice | Benzofuran | [45] |
| 29–33 | 0.37 ± 0.06 0.17 ± 0.02 1.33 ± 0.27 0.49 ± 0.01 0.61 ± 0.14 0.75 ± 0.16 | 2.64 1.30 4.92 2.22 4.69 2.88 | Fluorescent imaging of amyloid plaques in Tg2576 AD mice brain sections | Benzothiazole | [46,47] |
| 34–39 | 0.16 ± 0.02 0.99 ± 0.04 | 1.59 4.95 | Fluorescence imaging of amyloid plaques in 5xFAD mice brain sections | Benzothiazole | [48] |
| № | Brain Uptake, % | Diagnostic Method | Metal | Metal- Chelating Moiety | Aβ-binding Moiety | Reference |
|---|---|---|---|---|---|---|
| 40–42 | Cellebrium 0.50 ± 0.07 Cortex 0.36 ± 0.03 | MRI SPECT | Gd3+, 111In3+ | DO3A | PiB | [55] |
| - | ||||||
| 43, 44 | - | MRI | Gd3+ | DO3A | PiB | [56] |
| 45–60 | MRI | Gd3+ | DOTA PCTA | Benzothiazole Benzoxazole Stilbene | [57] | |
| 61 | - | PET | Ga3+ | DOTA | Benzofuran | [58] |
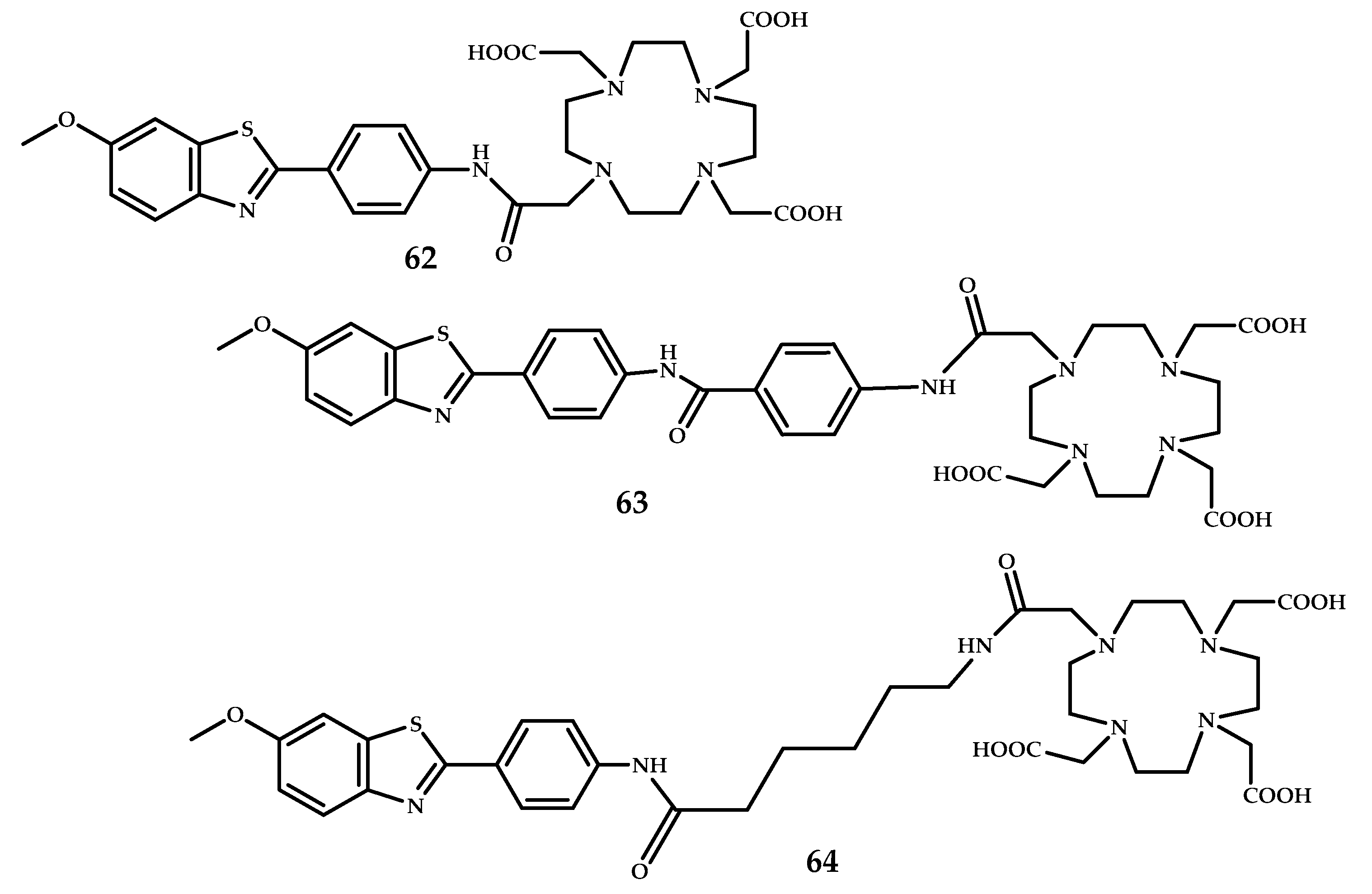
| 62–64 | - | PET | Ga3+ | DOTA | PiB | [59] |
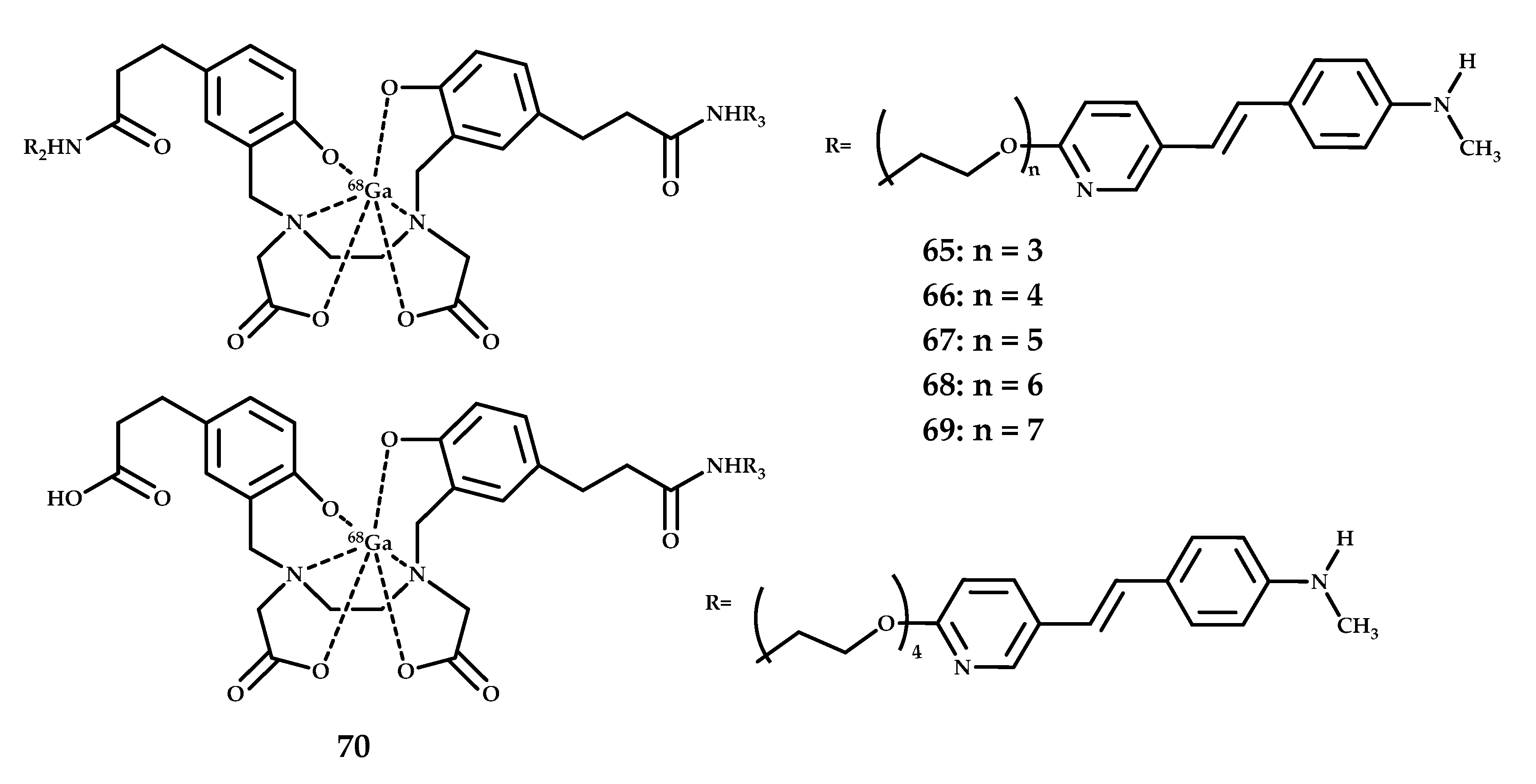
| 65–70 | 0.12 ± 0.05 0.17 ± 0.05 0.31 ± 0.09 0.21 ± 0.05 0.22 ± 0.03 0.11 ± 0.01 | PET | Ga3+ | HBED-CC | Styrylpyridine | [60] |
| 71 | 1.24 ± 0.31 | PET | Ga3+ | Chalkone | [61] | |
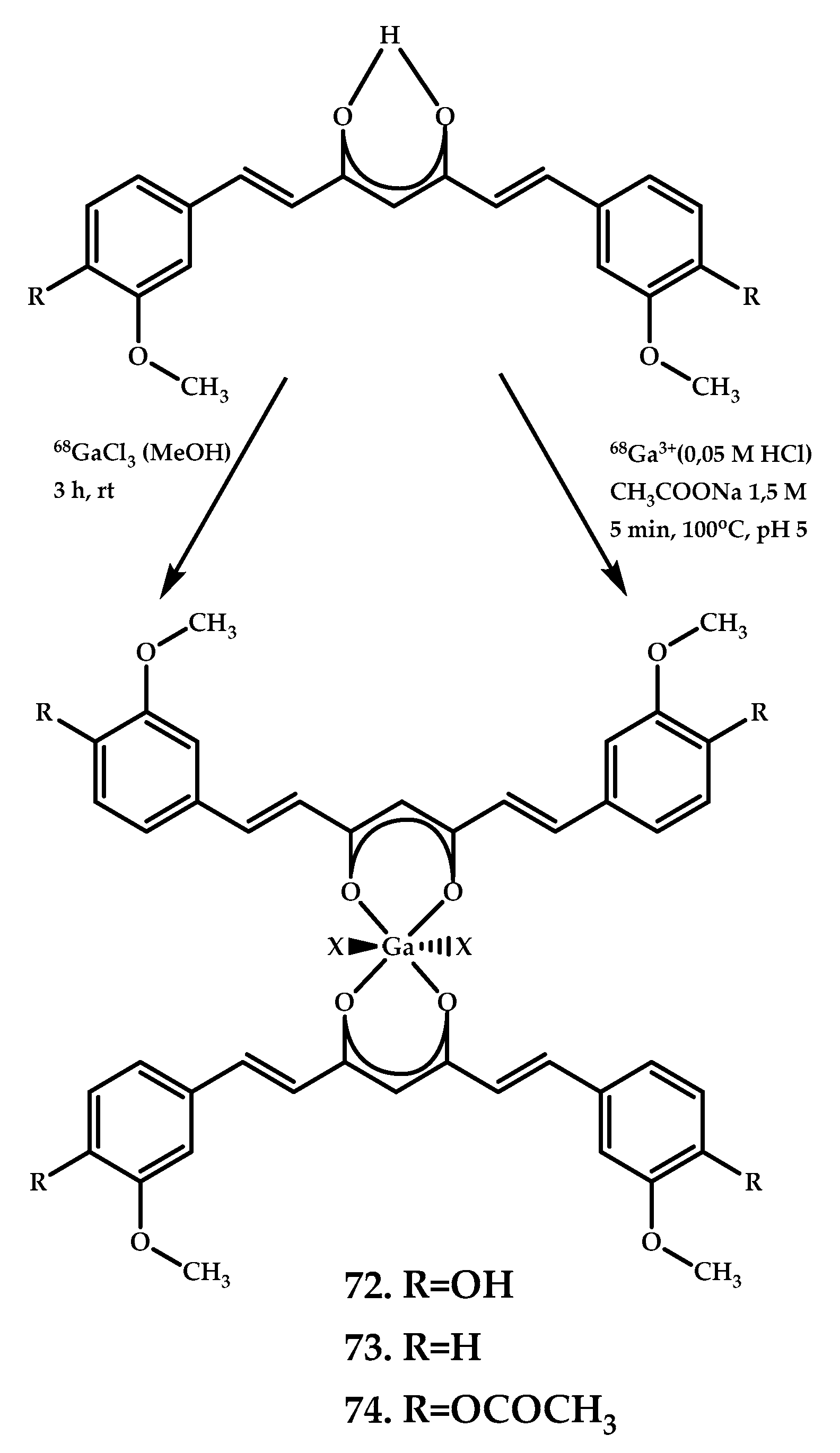
| 72 74 | No brain uptake | - | Ga3+ | Curcumin | [62,63] | |
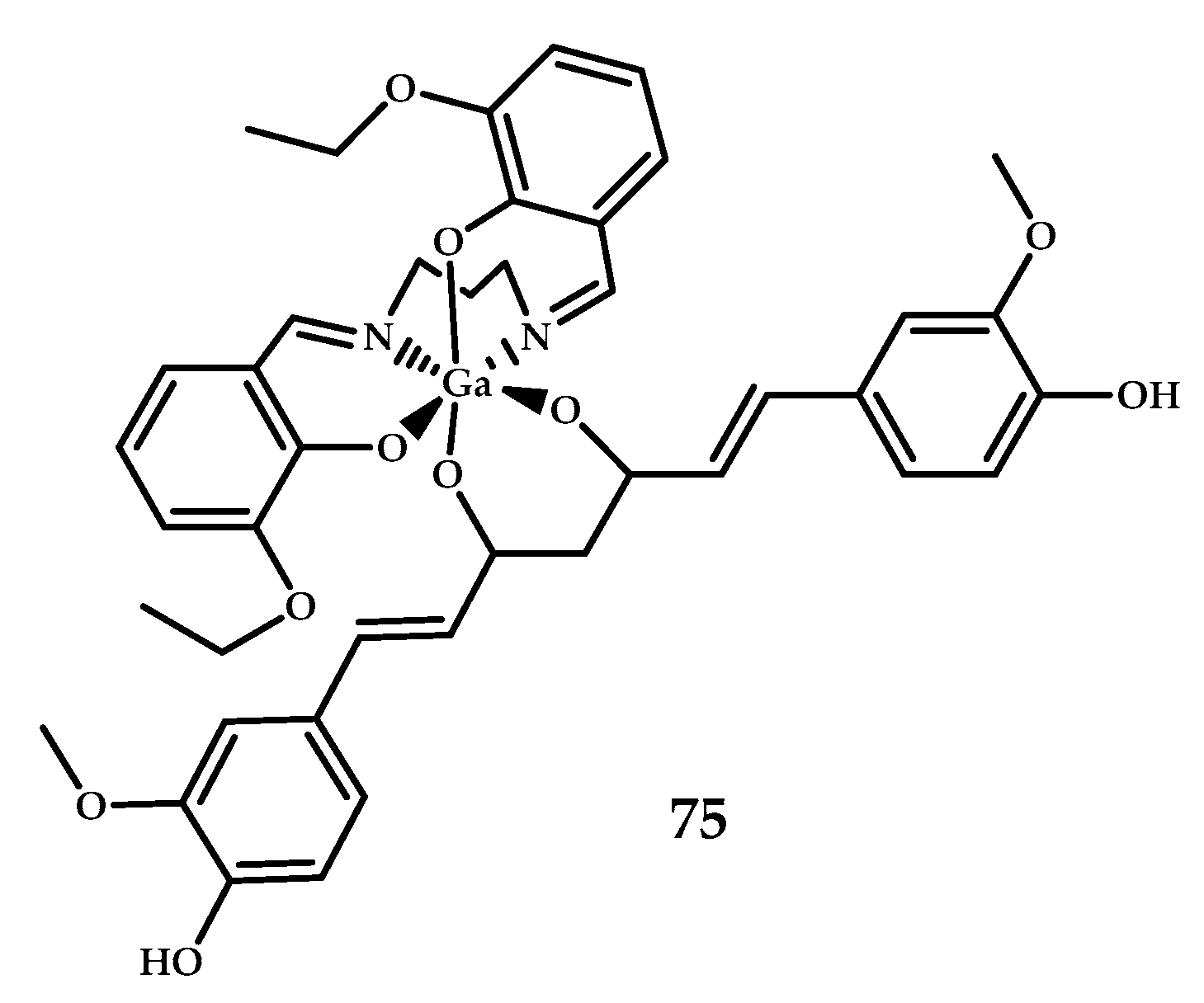
| 75 | No biodistribution experiment | - | Ga3+ | N2O2 Schiff- base ligand | Curcumin | [64] |
| 76, 77 | - | Ga3+ | NODAGA AAZTA | Curcumin | [65] | |
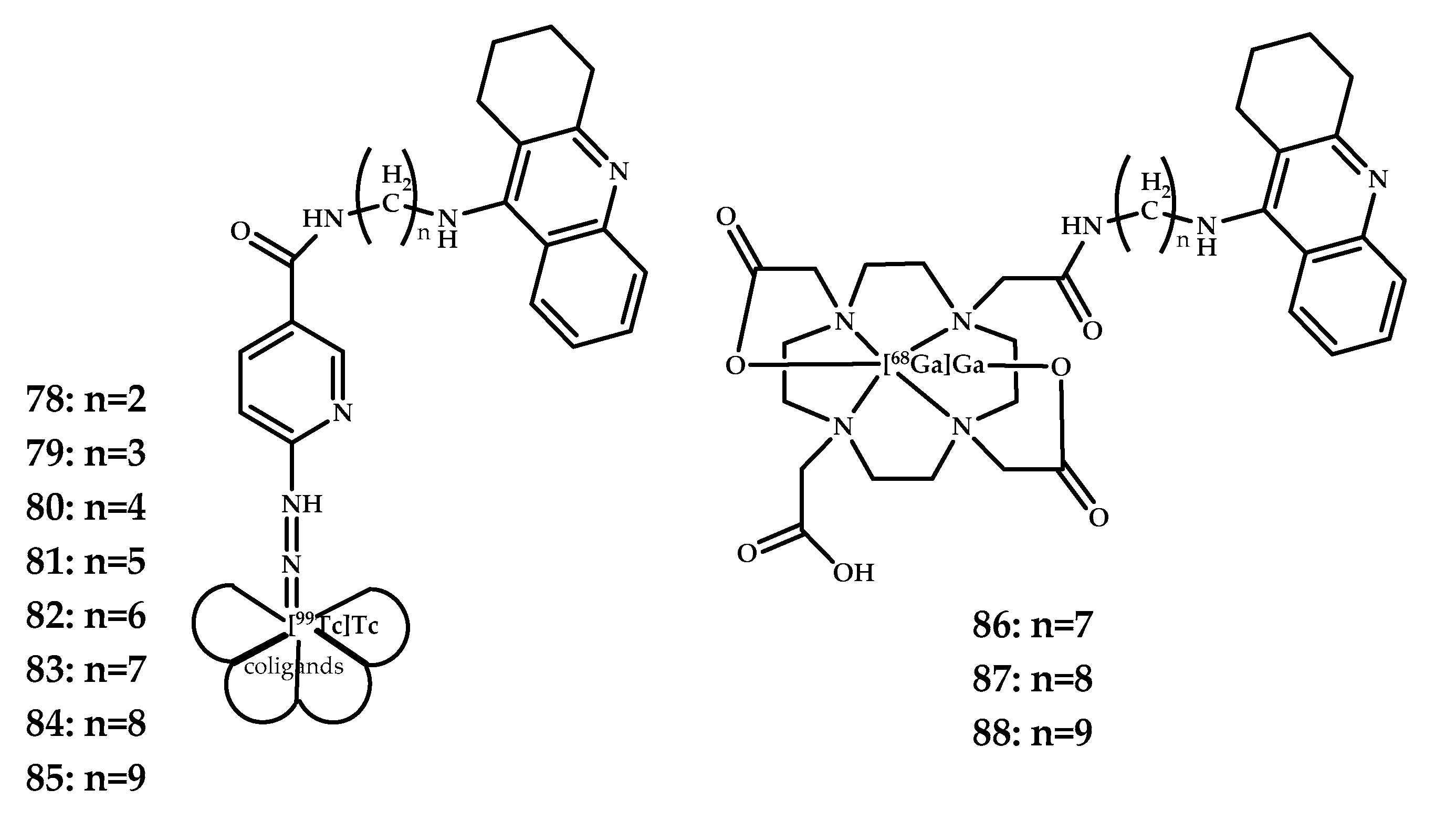
| 78–88 | 0.21 ± 0.07 (5 min p.i.) | PET SPECT | Ga3+ 99mTc3+ | DOTA | Tacrine | [66] |
| (CH2)n | Log D | ||
|---|---|---|---|
| [99mTc]Tc-Hynic-NH(CH2)nTac | [68Ga]Ga-DOTA-NH(CH2)nTac | ||
| 78: n = 2 | −2.95 ± 0.06 | - | |
| 79: n = 3 | −2.80 ± 0.01 | - | |
| 80: n = 4 | −2.53 ± 0.02 | - | |
| 81: n = 5 | −2.41 ± 0.01 | - | |
| 82: n = 6 | −2.08 ± 0.01 | - | |
| 83: n = 7 | −1.86 ± 0.02 | 86: | −2.52 ± 0.01 |
| 84: n = 8 | −1.50 ± 0.01 | 87: | −2.02 ± 0.01 |
| 85: n = 9 | −1.38 ± 0.01 | 88: | −1.52 ± 0.01 |
| Compound | IC50 ± SD ** (nM) | Selectivity for AChE a | Selectivity for BuChE b | |
|---|---|---|---|---|
| AChE | BuChE | |||
| 82 | 0.10 ± 0.01 | 0.12 ± 0.02 | 1.2 | 0.83 |
| 86 | 290 ± 20 | 167 ± 9 | 0.57 | 1.75 |
| Tacrine | 107 ± 9 | 16 ± 1 | 0.15 | 6.67 |
| № | Brain Uptake, ID/g, 2 Min Post-Injection % | Brain2 min/Brain60 min Ratio | Brain Tissue Experiments | Ligand | Reference |
|---|---|---|---|---|---|
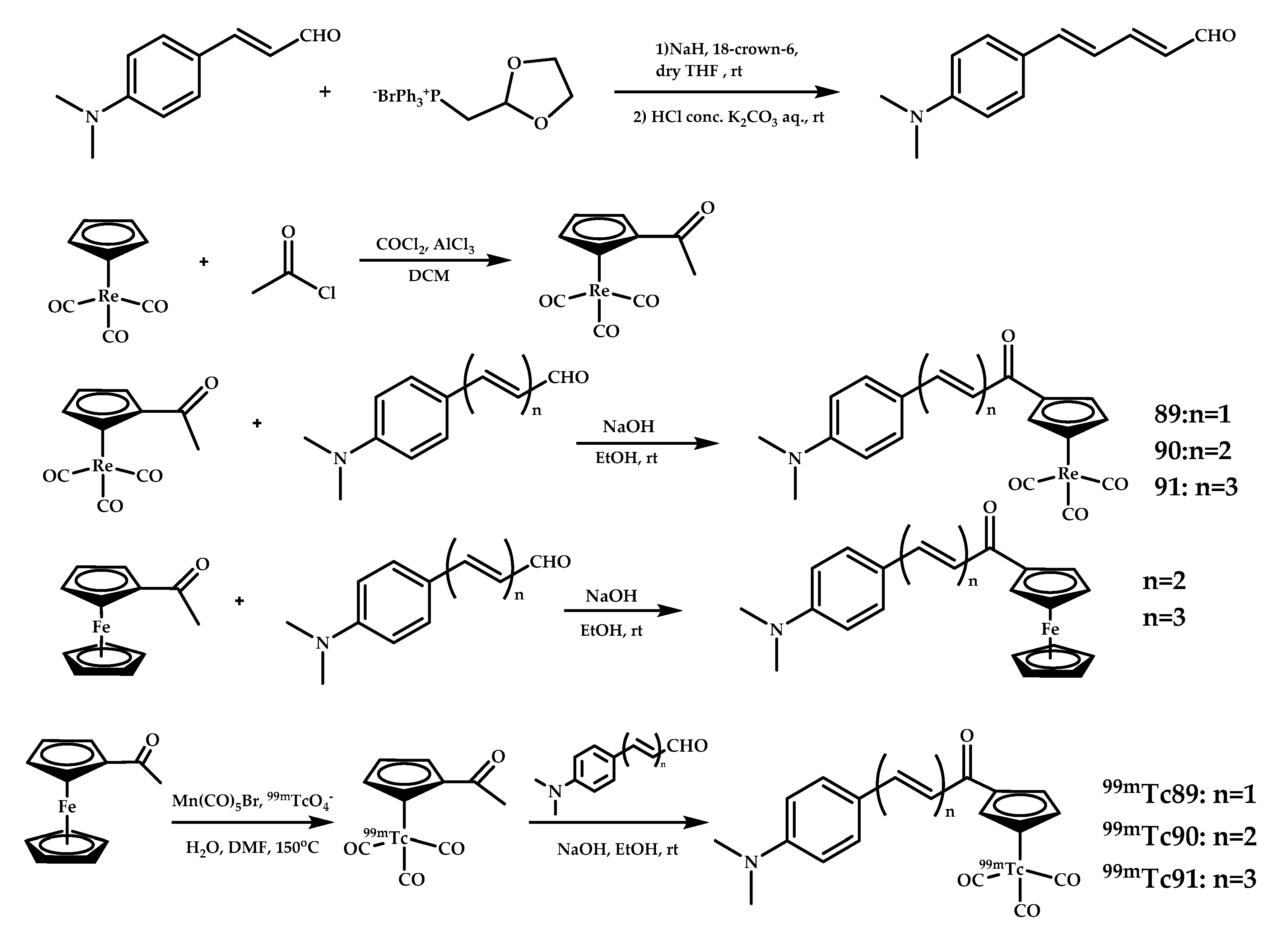
| 89–91 | 4.10 ± 0.38 /6.34 ± 0.81 2.30 ± 0.27 /3.68 ± 0.07 1.11 ± 0.34/ 1.64 ± 0.17 With/without PgP Blocked by Cyclosporin A | 8.20 4.18 1.73 | Fluorescent staining of Re complexes on APPswe/PSEN1 mice and AD patient brain sections Autoradiography on a APPswe/PSEN1 model mice | Chalcone-mimic moiety with [Cp99mTc(CO)3] | [83] |
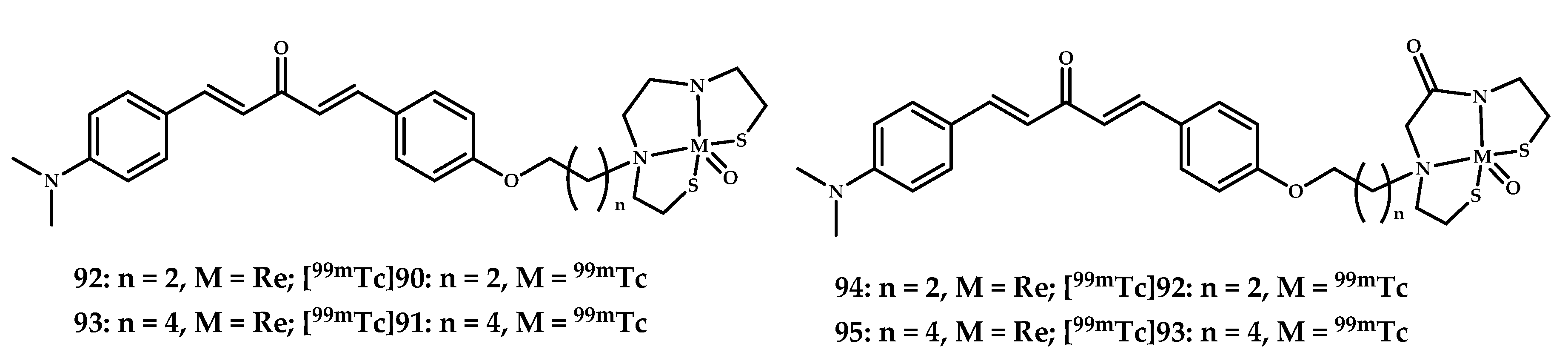
| 92–95 | 0.49 ± 0.08 0.47 ± 0.11 0.48 ± 0.06 0.31 ± 0.06 | 6.13 3.92 5.33 2.06 | In vitro fluorescent staining of Re complexes of brain tissue APPswe/PSEN1 mice | Curcumin-like dibenzylideneacetone conjugated with monoamineemonoamide dithiol (MAMA) and BAT (bis(aminoethanethiol) as chelating moieties | [84] |
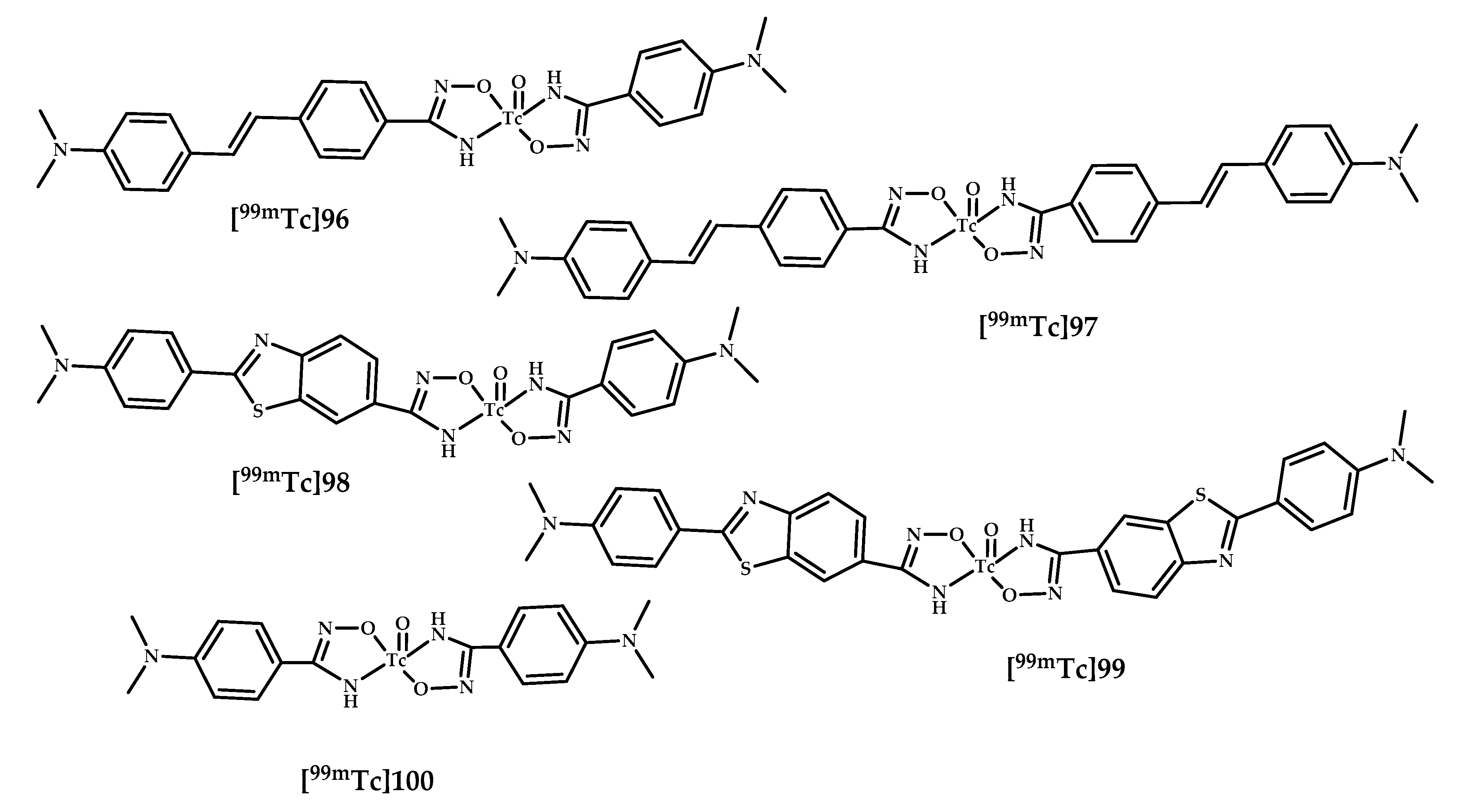
| 96–100 | 0.28 ± 0.03 | 2.54 | Autoradiography Tg2576 and wild-type mice | Benzotiasole/stilbene conjugated with hydroxamamide (Ham) as chelating moiety | [85,86] |
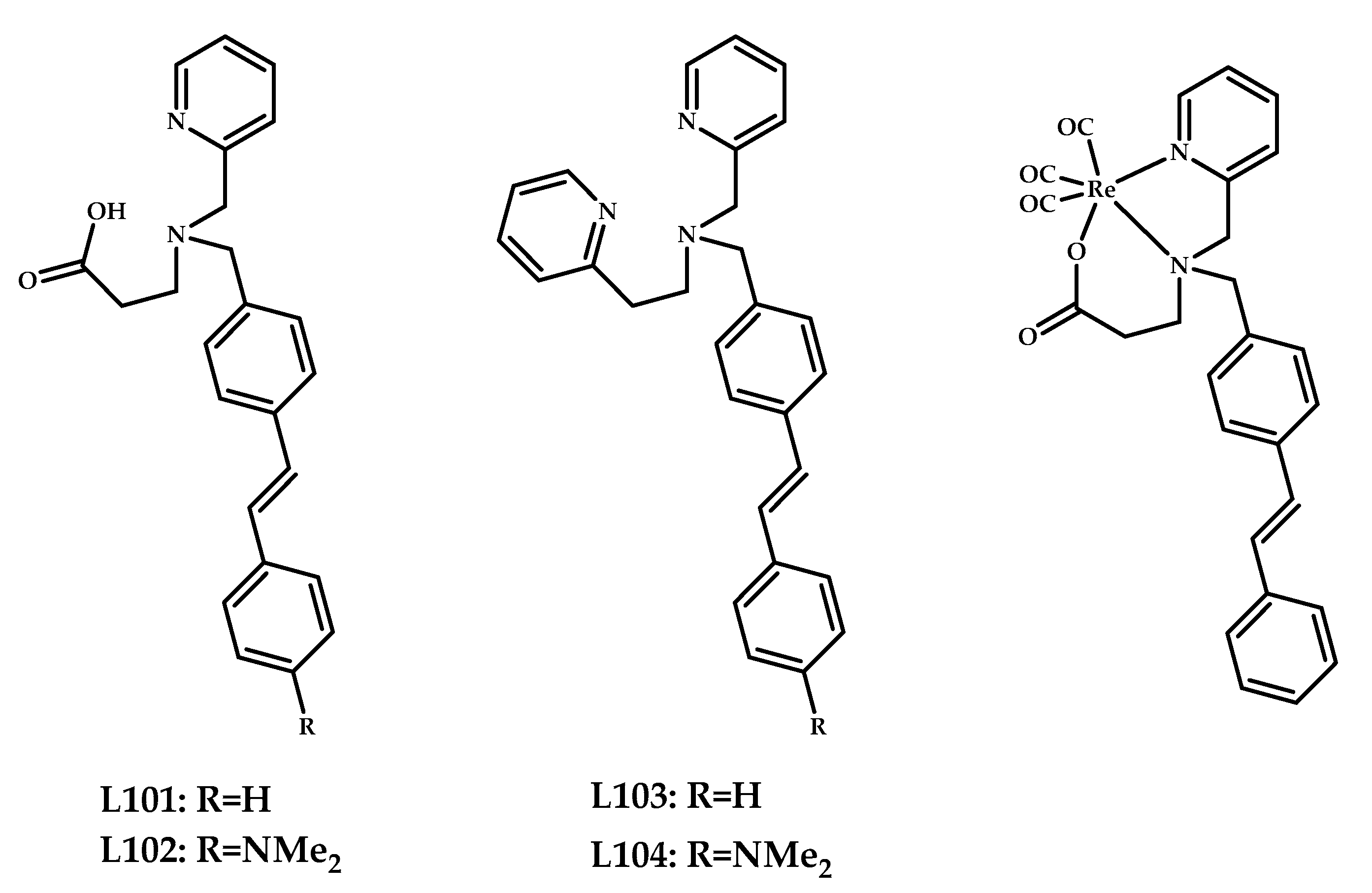
| 101–104 | 0.25 ± 0.05 0.24 ± 0.02 (wild type/APP mice) | 1.26 | SPECT images in APP/ PS1 transgenic mice | Styrilpyridyl conjugated with pyridylamine-carboxylate and dipyridylamine ligands as chelating moiety | [87] |
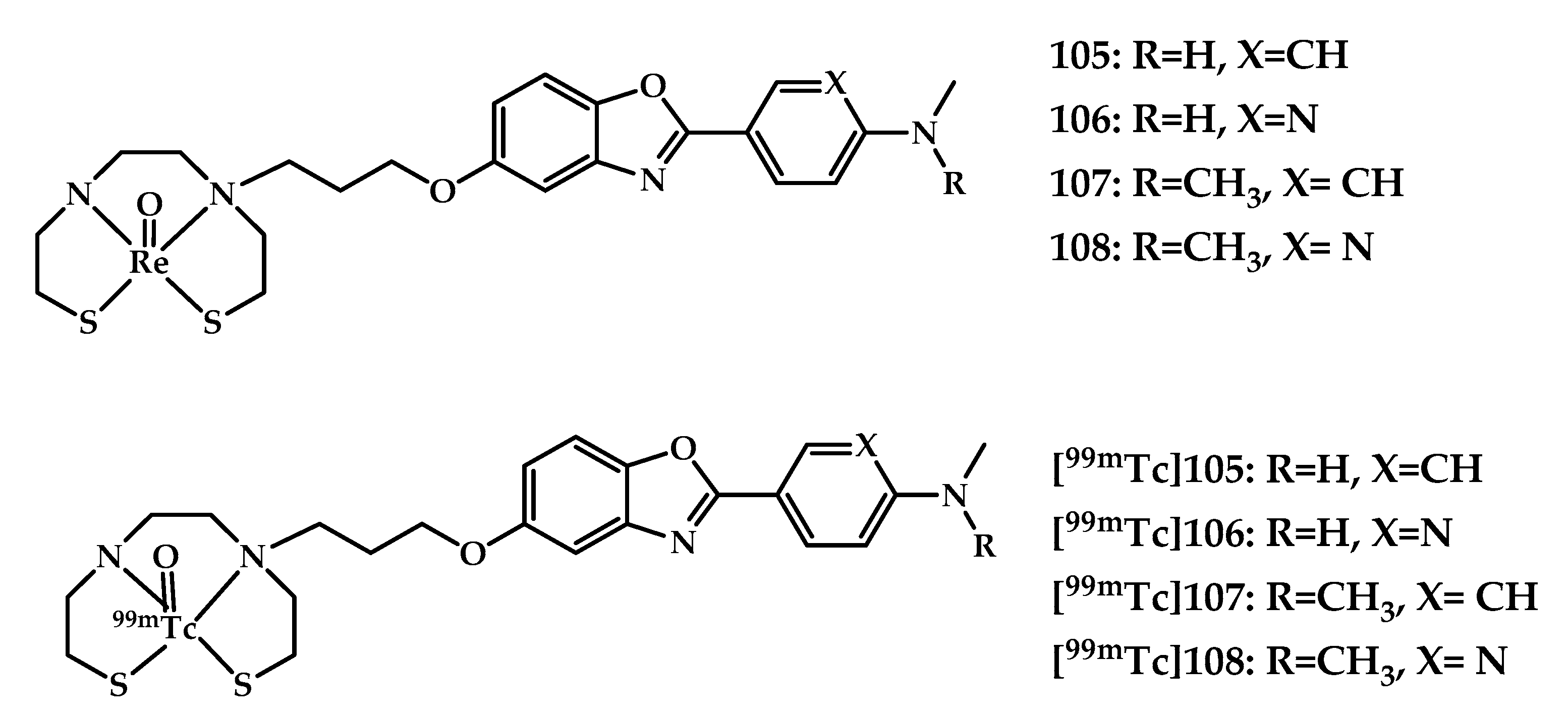
| 105–107 | 1.10 ± 0.08 0.96 ± 0.13 1.55 ± 0.51 1.24 ± 0.17 | 3.54 6.40 3.87 8.64 | In vitro autoradiography Brain tissue from APPswe/PSEN1 mice | Arylbenzoxazole conjugated with bis (aminoethanethiol) (BAT) as chelating moiety | [88] |
| 109–116 | 0.80 ± 0.17 0.61 ± 0.08 0.88 ± 0.14 1.21 ± 0.22 | 26.66 3.38 6.68 20.16 | Fluorescent staining of Re complexes with brain sections of APPswe/PSEN1T mice and AD patients In vitro autoradiography on brain sections of APPswe/PSEN1T mice and AD patients | Benzothiazole conjugated with iminodiacetic acid (IDA) as chelating moiety | [89] |
| 117–132 | 0.69 ± 0.16 0.46 ± 0.09 0.59 ± 0.12 2.11 ± 0.11 0.92 ± 0.09 0.47 ± 0.07 0.60 ± 0.05 | 1.50 1.15 1.37 3.40 1.46 2.47 2.07 | Fluorescent staining of rhenium complexes on brain slices from APPswe/PSEN1 mice and AD patients. Autoradiography on brain slices from APPswe/PSEN1 mice Ex vivo Autoradiography APPswe/PSEN1 mice In vivo SPECT−CT Imaging in Rhesus Monkeys | Arylbenzoxazole conjugated with bis (aminoethanethiol) (BAT) as chelating moiety | [90] |
| 131 | - | - | Fluorescent staining or De complexes of AD human brain tissue | Styrilpyridyl conjugated with 2-aminoethyl-2-hydroxybenzamide as chelating moiety | [91] |
| 132 | 0.53 ± 0.11 0.52 ± 0.08 (healthy/5xFAD mice) | Brain2 min/Brain90 min 2.0 Brain 2 min/Brain 90min 2.1. | Fluorescence staining of Re complexes of AD patient brain and 5x FAD mice | Benzothiazole conjugated with tricarbonyl [M(CO)3]+ | [92] |
| 133–135 | 0.88± 0.08 | 3.52 | Ex vivo autoradiography using Tg2576 mice | Phenylquinoxaline conjugated with bis (aminoethanethiol) (BAT) as chelating moiety | [93] |
| 136–140 | _ | - | Fluorescence staining of Re complexes of AD patient brain | Styrilpyridyl/Benzofuran conjugated with pyridylthiosemicarbazide as chelating moiety | [94] |
| 141, 142 | 0.78 ± 0.07 0.86 ± 0.07 | 8.66 7.16 | Autoradiography of AD rat model (vaccinated with Aβ solution) | Arylimidazo[2,1-b] benzothiazole conjugated with triazole-based N/N/O, N/N/N, N/N/S ligands as chelating moieties | [95] |
| 143–145 | 7.94 ± 1.46 3.99 ± 0.60 5.36 ± 0.65 | 39.7 99.75 59.55 | Fluorescent staining of AD patient brain | Benzothiazole with benzene ring replaced by the cyclopentadienyl tricarbonyl | [96] |
| 146 | 0.38 ± 0.03 0.35 ± 0.01 (AD/normal rats) With/without blocked PgP (Cyclosporine A) (10 min after injection) 0.27 ± 0.01 0.60 ± 0.01 | Brain2 min/brain30 min 2.33 Brain2 min/brain30 min 1.65 | Planar scintigraphy, autoradiography and fluorescent staining with Thioflavin S and Congo Red studies on prepared brain slices of AD rats (vaccinated with Aβ1–42) and brain sections of AD and Schizophrenia patients. | D-(FPLIAIMA)-NH2 peptide | [97] |
| 0–10 Min | 10–20 Min | 20–30 Min | 30–40 Min | Clearance Ratio | |
|---|---|---|---|---|---|
| M04 | 1.23 | 1.13 | 1.01 | 0.88 | 1.40 |
| F27 | 0.78 | 0.70 | 0.67 | 0.64 | 1.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krasnovskaya, O.; Spector, D.; Zlobin, A.; Pavlov, K.; Gorelkin, P.; Erofeev, A.; Beloglazkina, E.; Majouga, A. Metals in Imaging of Alzheimer’s Disease. Int. J. Mol. Sci. 2020, 21, 9190. https://doi.org/10.3390/ijms21239190
Krasnovskaya O, Spector D, Zlobin A, Pavlov K, Gorelkin P, Erofeev A, Beloglazkina E, Majouga A. Metals in Imaging of Alzheimer’s Disease. International Journal of Molecular Sciences. 2020; 21(23):9190. https://doi.org/10.3390/ijms21239190
Chicago/Turabian StyleKrasnovskaya, Olga, Daniil Spector, Alexander Zlobin, Kirill Pavlov, Peter Gorelkin, Alexander Erofeev, Elena Beloglazkina, and Alexander Majouga. 2020. "Metals in Imaging of Alzheimer’s Disease" International Journal of Molecular Sciences 21, no. 23: 9190. https://doi.org/10.3390/ijms21239190
APA StyleKrasnovskaya, O., Spector, D., Zlobin, A., Pavlov, K., Gorelkin, P., Erofeev, A., Beloglazkina, E., & Majouga, A. (2020). Metals in Imaging of Alzheimer’s Disease. International Journal of Molecular Sciences, 21(23), 9190. https://doi.org/10.3390/ijms21239190






