One-Dimensional Nanostructures of Polypyrrole for Shielding of Electromagnetic Interference in the Microwave Region
Abstract
1. Introduction
2. Results and Discussion
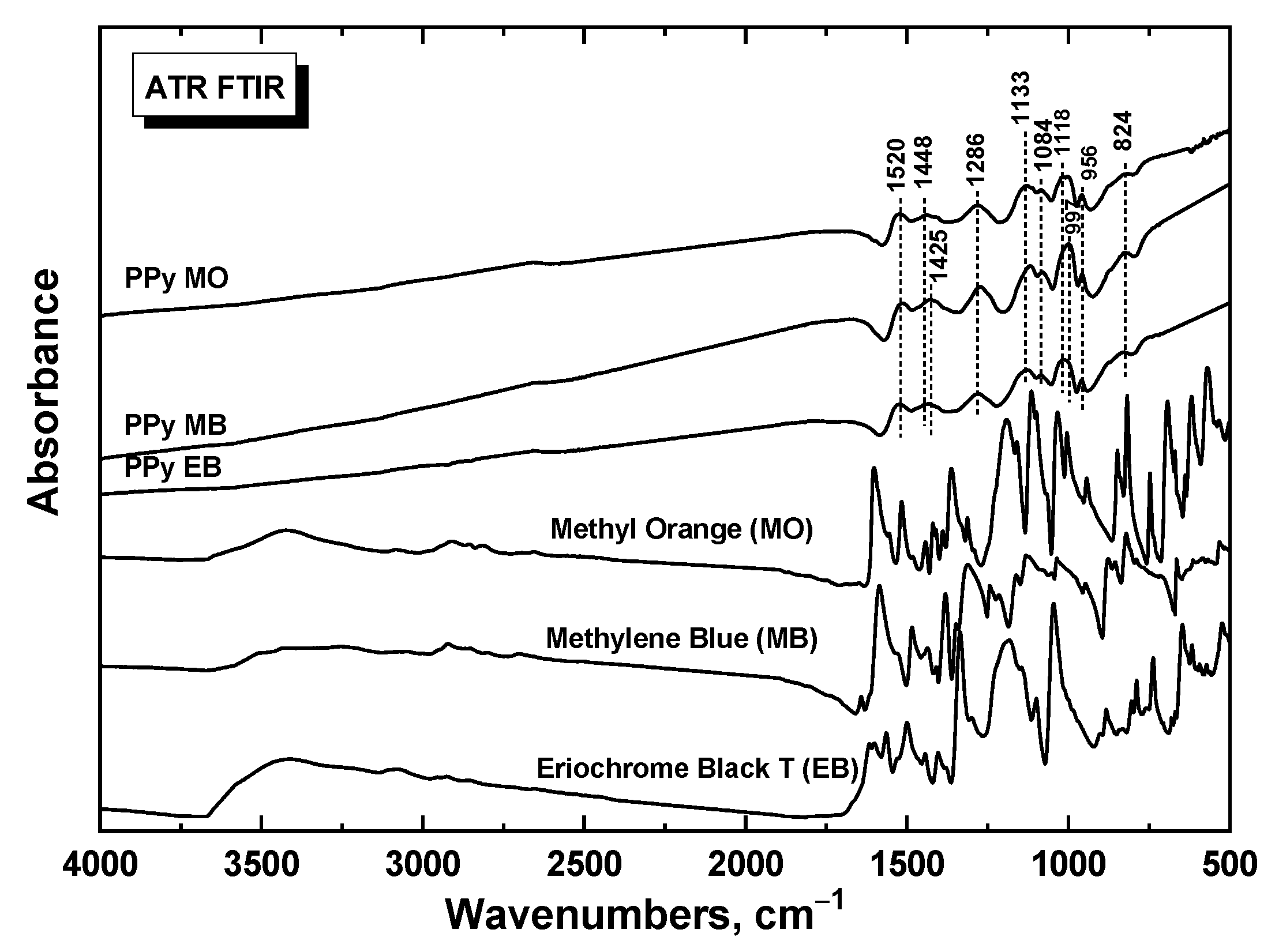
2.1. Morphology, Dynamical and Electrical Properties
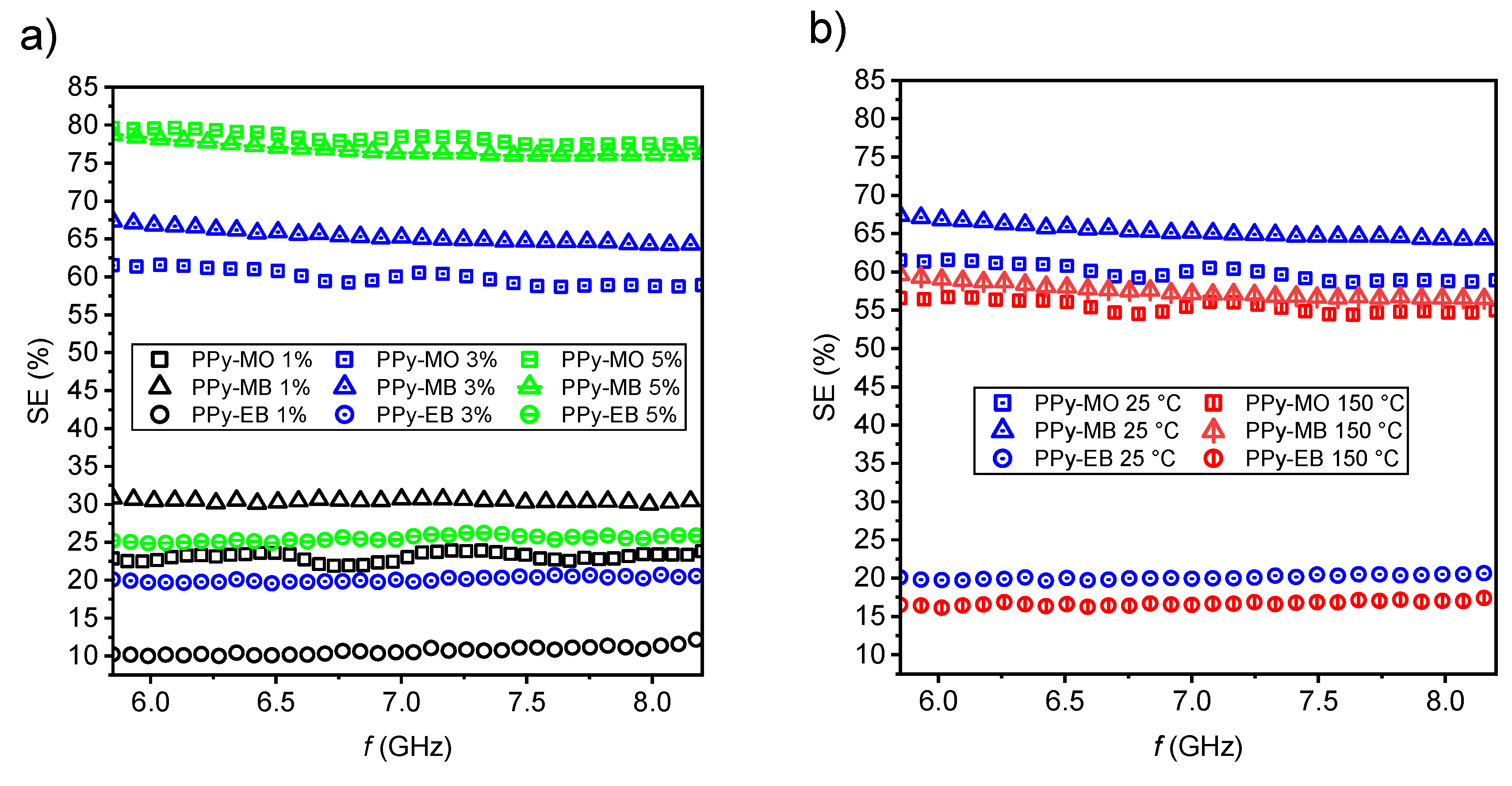
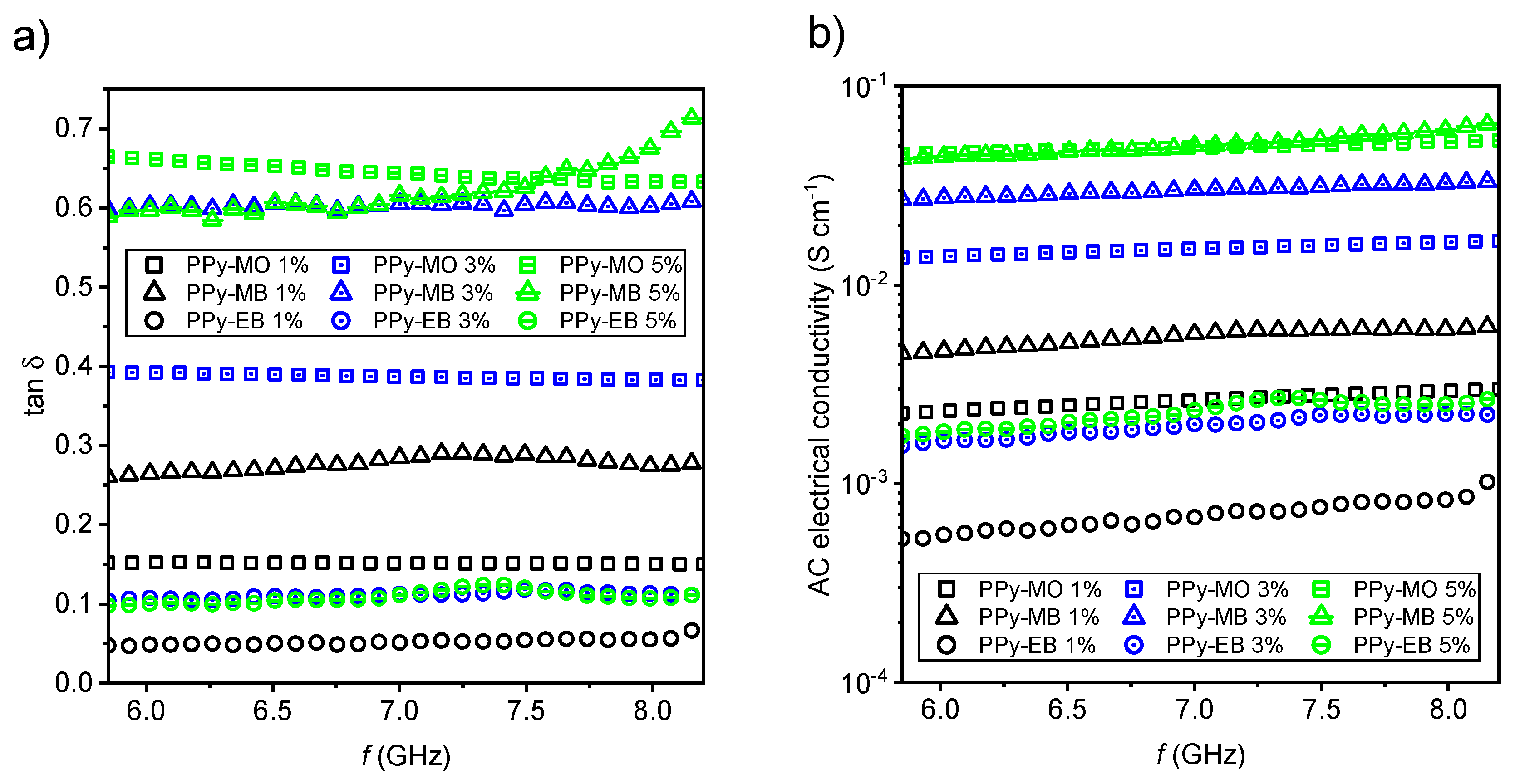
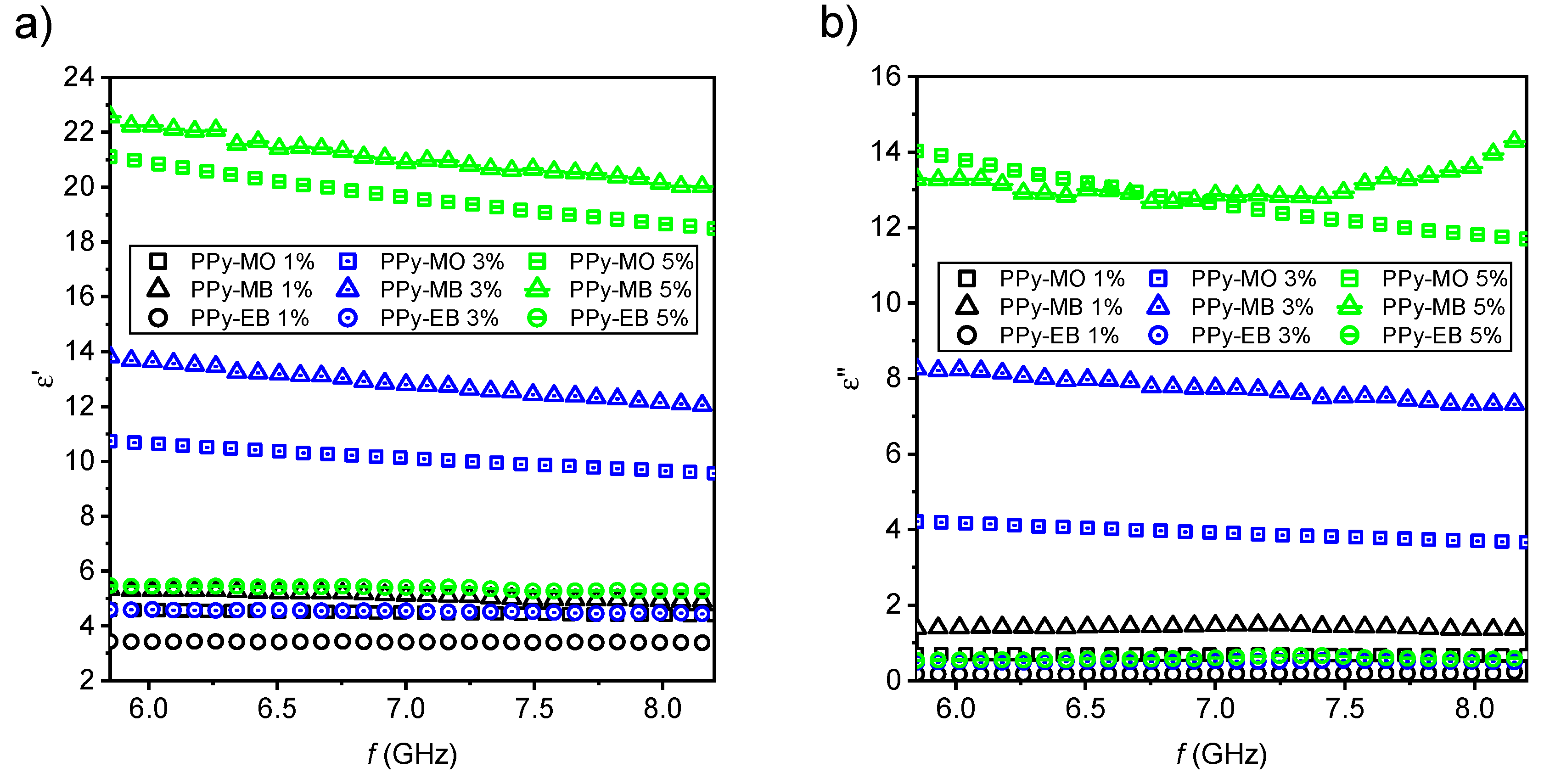
2.2. EMI Shielding Properties
3. Materials and Methods
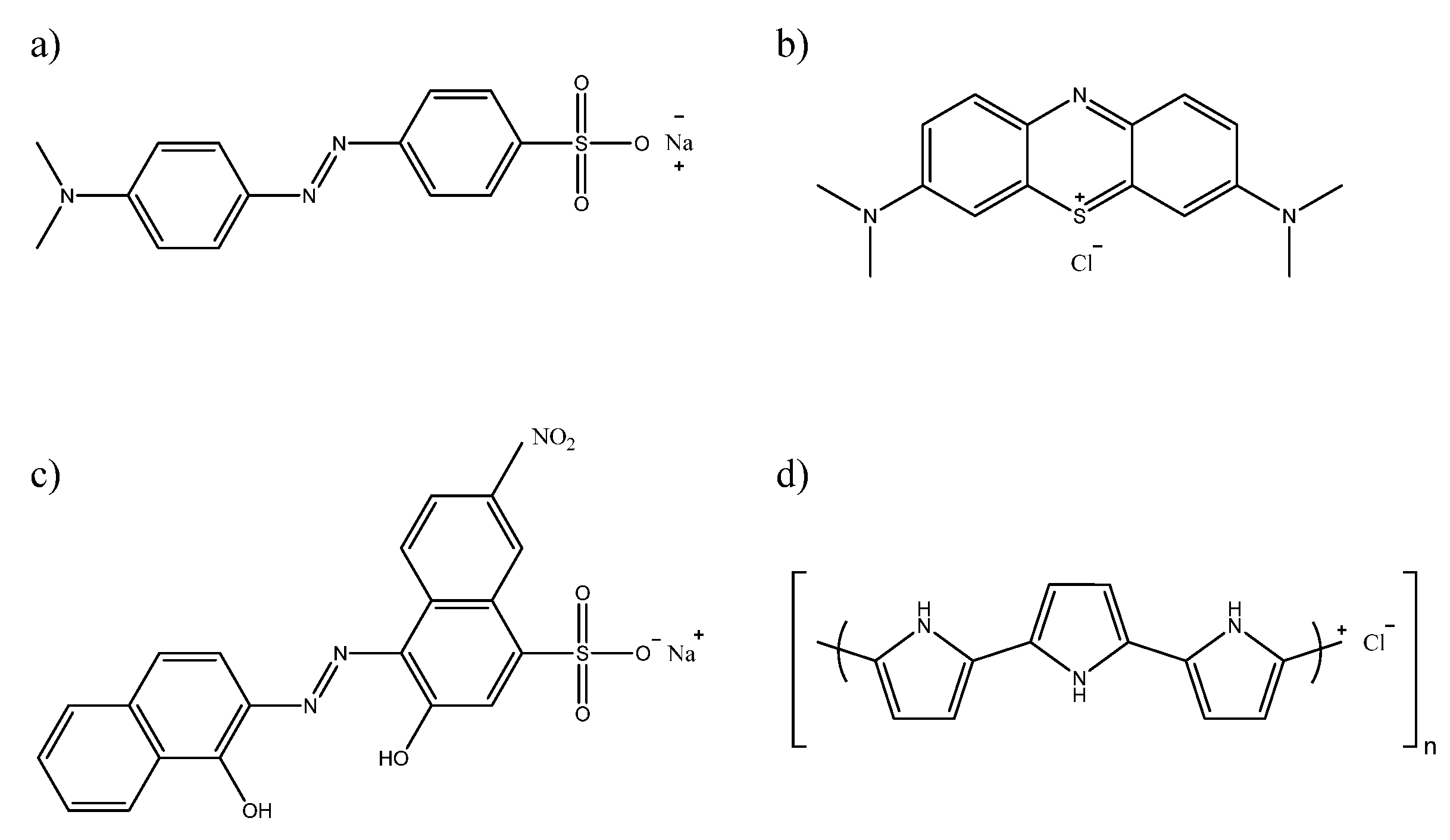
3.1. Chemicals
3.2. Synthesis of 1D Structures of Polypyrrole
3.3. Characterization of 1D Structures by Electron Microanalysis
3.4. Specific Surface Measurement by Brunauer–Emmett–Teller Analysis
3.5. Measurement of DC Electrical Conductivity
3.6. Preparation of Samples for EMI Shielding Measurement
3.7. Study of structure by Infrared Spectroscopy
3.8. Measurement and Evaluation of Shielding Efficiency
3.9. Extraction of Complex Permittivity and AC Electrical Conductivity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Masetti, G.; Graffi, S.; Golzio, D.; KovacsV, Z.M. Failures induced on analog integrated circuits by conveyed electromagnetic interferences: A review. Microelectron. Reliab. 1996, 36, 955–972. [Google Scholar] [CrossRef]
- Turczyn, R.; Krukiewicz, K.; Katunin, A.; Sroka, J.; Sul, P. Fabrication and application of electrically conducting composites for electromagnetic interference shielding of remotely piloted aircraft systems. Compos. Struct. 2020, 232, 111498. [Google Scholar] [CrossRef]
- Carlberg, M.; Hedendahl, L.; Ahonen, M.; Koppel, T.; Hardell, L. Increasing incidence of thyroid cancer in the Nordic countries with main focus on Swedish data. BMC Cancer 2016, 16, 426. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, M.; Koppel, T.; Ahonen, M.; Hardell, L. Case-control study on occupational exposure to extremely low-frequency electromagnetic fields and glioma risk. Am. J. Ind. Med. 2017, 60, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, M.; Koppel, T.; Ahonen, M.; Hardell, L. Case-Control Study on Occupational Exposure to Extremely Low-Frequency Electromagnetic Fields and the Association with Meningioma. BioMed Res. Int. 2018, 2018, 5912394. [Google Scholar] [CrossRef] [PubMed]
- Celozzi, S.; Araneo, R.; Lovat, G. Electromagnetic Shielding; Wiley-Interscience; IEEE Press: Hoboken, NJ, USA, 2008; pp. 358p. ISBN 978-0-470-05536-6. [Google Scholar]
- Chung, D.D.L. Materials for electromagnetic interference shielding. Mater. Chem. Phys. 2020, 255, 123587. [Google Scholar] [CrossRef]
- Singh, A.K.; Shishkin, A.; Koppel, T.; Gupta, N. A review of porous lightweight composite materials for electromagnetic interference shielding. Compos. Part B Eng. 2018, 149, 188–197. [Google Scholar] [CrossRef]
- Kumar, P.; Maiti, U.N.; Sikdar, A.; Das, T.K.; Kumar, A.; Sudarsan, V. Recent Advances in Polymer and Polymer Composites for Electromagnetic Interference Shielding: Review and Future Prospects. Polym. Rev. 2019, 59, 687–738. [Google Scholar] [CrossRef]
- Jiang, D.W.; Murugadoss, V.; Wang, Y.; Lin, J.; Ding, T.; Wang, Z.C.; Shao, Q.; Wang, C.; Liu, H.; Lu, N.; et al. Electromagnetic Interference Shielding Polymers and Nanocomposites-A Review. Polym. Rev. 2019, 59, 280–337. [Google Scholar] [CrossRef]
- Stejskal, J.; Trchová, M. Conducting polypyrrole nanotubes: A review. Chem. Pap. 2018, 72, 1563–1595. [Google Scholar] [CrossRef]
- Stejskal, J.; Prokeš, J. Conductivity and morphology of polyaniline and polypyrrole prepared in the presence of organic dyes. Synth. Met. 2020, 264, 116373. [Google Scholar] [CrossRef]
- Madhusudhan, C.K.; Mahendra, K.; Madhukar, B.S.; Somesh, T.E.; Faisal, M. Incorporation of graphite into iron decorated polypyrrole for dielectric and EMI shielding applications. Synth. Met. 2020, 267, 116450. [Google Scholar] [CrossRef]
- Zhang, K.C.; Chen, X.F.; Gao, X.B.; Chen, L.; Ma, S.Z.; Xie, C.Y.; Zhang, X.G.; Lu, W. Preparation and microwave absorption properties of carbon nanotubes/iron oxide/polypyrrole/carbon composites. Synth. Met. 2020, 260, 116282. [Google Scholar] [CrossRef]
- Xie, A.; Jiang, W.C.; Wu, F.; Dai, X.Q.; Sun, M.X.; Wang, Y.; Wang, M.Y. Interfacial synthesis of polypyrrole microparticles for effective dissipation of electromagnetic waves. J. Appl. Phys. 2015, 118, 204105. [Google Scholar] [CrossRef]
- Green, M.; Tran, A.T.V.; Chen, X.B. Maximizing the microwave absorption performance of polypyrrole by data-driven discovery. Compos. Sci. Technol. 2020, 199, 108332. [Google Scholar] [CrossRef]
- Sapurina, I.; Kazantseva, N.E.; Ryvkina, N.G.; Prokeš, J.; Sáha, P.; Stejskal, J. Electromagnetic radiation shielding by composites of conducting polymers and wood. J. Appl. Polym. Sci. 2005, 95, 807–814. [Google Scholar] [CrossRef]
- Babayan, V.; Kazantseva, N.E.; Moučka, R.; Stejskal, J. Electromagnetic shielding of polypyrrole-sawdust composites: Polypyrrole globules and nanotubes. Cellulose 2017, 24, 3445–3451. [Google Scholar] [CrossRef]
- Yu, L.J.; Yang, Q.X.; Liao, J.L.; Zhu, Y.F.; Li, X.; Yang, W.T.; Fu, Y.Q. A novel 3D silver nanowires@polypyrrole sponge loaded with water giving excellent microwave absorption properties. Chem. Eng. J. 2018, 352, 490–500. [Google Scholar] [CrossRef]
- Jiao, Y.Z.; Li, J.J.; Xie, A.M.; Wu, F.; Zhang, K.; Dong, W.; Zhu, X.F. Confined polymerization strategy to construct polypyrrole/zeolitic imidazolate frameworks (PPy/ZIFs) nanocomposites for tunable electrical conductivity and excellent electromagnetic absorption. Compos. Sci. Technol. 2019, 174, 232–240. [Google Scholar] [CrossRef]
- Hong, Y.K.; Lee, C.Y.; Jeong, C.K.; Sim, J.H.; Kim, K.; Joo, J.; Kim, M.S.; Lee, J.Y.; Jeong, S.H.; Byun, S.W. Electromagnetic interference shielding characteristics of fabric complexes coated with conductive polypyrrole and thermally evaporated Ag. Curr. Appl. Phys. 2001, 1, 439–442. [Google Scholar] [CrossRef]
- Zhao, H.; Hou, L.; Lu, Y.X. Electromagnetic interference shielding of layered linen fabric/polypyrrole/nickel (LF/PPy/Ni) composites. Mater. Des. 2016, 95, 97–106. [Google Scholar] [CrossRef]
- Kim, H.K.; Byun, S.W.; Jeong, S.H.; Hong, Y.K.; Joo, J.S.; Song, K.; Park, Y.H.; Lee, J.Y. Environmental staility of EMI shielding PET fabric/polypyrrole composite. Mol. Cryst. Liq. Cryst. 2002, 377, 369–372. [Google Scholar] [CrossRef]
- Kim, M.S.; Kim, H.K.; Byun, S.W.; Jeong, S.H.; Hong, Y.K.; Joo, J.S.; Song, K.T.; Kim, J.K.; Lee, C.J.; Lee, J.Y. PET fabric/polypyrrole composite with high electrical conductivity for EMI shielding. Synth. Met. 2002, 126, 233–239. [Google Scholar] [CrossRef]
- Kim, H.A.; Kim, M.S.; Chun, S.Y.; Park, Y.H.; Jeon, B.S.; Lee, J.Y.; Hong, Y.K.; Joo, J.; Kim, S.H. Characteristics of electrically conducting polymer-coated textiles. Mol. Cryst. Liq. Cryst. 2003, 405, 161–169. [Google Scholar] [CrossRef]
- Gashti, M.P.; Ghehi, S.T.; Arekhloo, S.V.; Mirsmaeeli, A.; Kiumarsi, A. Electromagnetic Shielding Response of UV-induced Polypyrrole/Silver Coated Wool. Fibers Polym. 2015, 16, 585–592. [Google Scholar] [CrossRef]
- Kathirgamanathan, P. Novel Cable Shielding Materials Based on the Impregnation of Microporous Membranes with Inherently Conducting Polymers. Adv. Mater. 1993, 5, 281–283. [Google Scholar] [CrossRef]
- Xie, A.M.; Wu, F.; Jiang, W.C.; Zhang, K.; Sun, M.X.; Wang, M.Y. Chiral induced synthesis of helical polypyrrole (PPy) nano-structures: A lightweight and high-performance material against electromagnetic pollution. J. Mater. Chem. C 2017, 5, 2175–2181. [Google Scholar] [CrossRef]
- Hu, S.C.; Zhou, Y.; Zhang, L.L.; Liu, S.J.; Cui, K.; Lu, Y.Y.; Li, K.N.; Li, X.D. Effects of indigo carmine concentration on the morphology and microwave absorbing behavior of PPy prepared by template synthesis. J. Mater. Sci. 2018, 53, 3016–3026. [Google Scholar] [CrossRef]
- Moučka, R.; Sedlačík, M.; Prokeš, J.; Kasparyan, H.; Valtera, S.; Kopecký, D. Electromagnetic interference shielding of polypyrrole nanostructures. Synth. Met. 2020, 269. [Google Scholar] [CrossRef]
- Varga, M.; Kopecký, D.; Kopecká, J.; Křivka, I.; Hanuš, J.; Zhigunov, A.; Trchová, M.; Vrňata, M.; Prokes, J. The ageing of polypyrrole nanotubes synthesized with methyl orange. Eur. Polym. J. 2017, 96, 176–189. [Google Scholar] [CrossRef]
- Bober, P.; Li, Y.; Acharya, U.; Panthi, Y.; Pfleger, J.; Humpolíček, P.; Trchová, M.; Stejskal, J. Acid Blue dyes in polypyrrole synthesis: The control of polymer morphology at nanoscale in the promotion of high conductivity and the reduction of cytotoxicity. Synth. Met. 2018, 237, 40–49. [Google Scholar] [CrossRef]
- Valtera, S.; Prokeš, J.; Kopecká, J.; Vrňata, M.; Trchová, M.; Varga, M.; Stejskal, J.; Kopecký, D. Dye-stimulated control of conducting polypyrrole morphology. RSC Adv. 2017, 7, 51495–51505. [Google Scholar] [CrossRef]
- Kopecká, J.; Kopecký, D.; Vrňata, M.; Fitl, P.; Stejskal, J.; Trchová, M.; Bober, P.; Moravková, Z.; Prokeš, J.; Sapurina, I. Polypyrrole nanotubes: Mechanism of formation. RSC Adv. 2014, 4, 1551–1558. [Google Scholar] [CrossRef]
- Kopecký, D.; Varga, M.; Prokeš, J.; Vrňata, M.; Trchová, M.; Kopecká, J.; Václavik, M. Optimization routes for high electrical conductivity of polypyrrole nanotubes prepared in presence of methyl orange. Synth. Met. 2017, 230, 89–96. [Google Scholar] [CrossRef]
- Trchová, M.; Stejskal, J. Resonance Raman Spectroscopy of Conducting Polypyrrole Nanotubes: Disordered Surface versus Ordered Body. J. Phys. Chem. A 2018, 122, 9298–9306. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, H.; Antunes, M.; Velasco, J.I. Recent advances in carbon-based polymer nanocomposites for electromagnetic interference shielding. Prog. Mater. Sci. 2019, 103, 319–373. [Google Scholar] [CrossRef]
- Weir, W.B. Automatic Measurement of Complex Dielectric-Constant and Permeability at Microwave-Frequencies. Proc. IEEE 1974, 62, 33–36. [Google Scholar] [CrossRef]
- Nicolson, A.M.; Ross, G.F. Measurement of Intrinsic Properties of Materials by Time-Domain Techniques. IEEE Trans. Instrum. Meas. 1970, 19, 377–382. [Google Scholar] [CrossRef]


| Sample | Specific Surface (m2 g−1) | Initial Powder Conductivity (S cm−1) | Conductivity after 80 Days (S cm−1) | Approx. Diameter (nm) |
|---|---|---|---|---|
| PPy-MO | 47.81 | 60.9 | 52.0 | 100–400 |
| PPy-MB | 24.41 | 21.5 | 12.4 | 600–4000 |
| PPy-EB | 27.38 | 17.1 | 8.9 | 50–150 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moučka, R.; Sedlačík, M.; Kasparyan, H.; Prokeš, J.; Trchová, M.; Hassouna, F.; Kopecký, D. One-Dimensional Nanostructures of Polypyrrole for Shielding of Electromagnetic Interference in the Microwave Region. Int. J. Mol. Sci. 2020, 21, 8814. https://doi.org/10.3390/ijms21228814
Moučka R, Sedlačík M, Kasparyan H, Prokeš J, Trchová M, Hassouna F, Kopecký D. One-Dimensional Nanostructures of Polypyrrole for Shielding of Electromagnetic Interference in the Microwave Region. International Journal of Molecular Sciences. 2020; 21(22):8814. https://doi.org/10.3390/ijms21228814
Chicago/Turabian StyleMoučka, Robert, Michal Sedlačík, Hayk Kasparyan, Jan Prokeš, Miroslava Trchová, Fatima Hassouna, and Dušan Kopecký. 2020. "One-Dimensional Nanostructures of Polypyrrole for Shielding of Electromagnetic Interference in the Microwave Region" International Journal of Molecular Sciences 21, no. 22: 8814. https://doi.org/10.3390/ijms21228814
APA StyleMoučka, R., Sedlačík, M., Kasparyan, H., Prokeš, J., Trchová, M., Hassouna, F., & Kopecký, D. (2020). One-Dimensional Nanostructures of Polypyrrole for Shielding of Electromagnetic Interference in the Microwave Region. International Journal of Molecular Sciences, 21(22), 8814. https://doi.org/10.3390/ijms21228814







