Breast Cancer and Microcalcifications: An Osteoimmunological Disorder?
Abstract
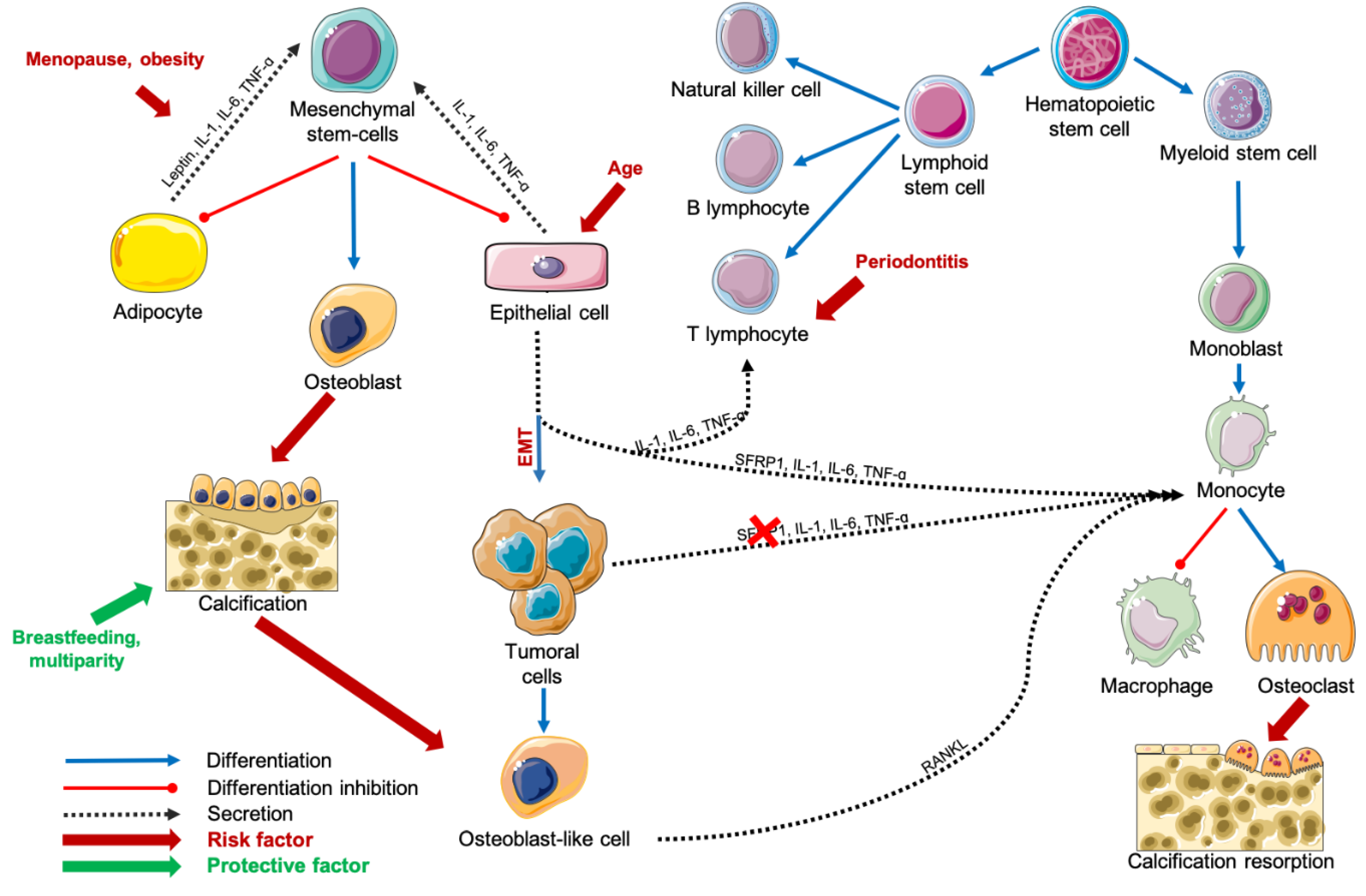
1. Introduction
2. Overview of the Most Frequent Osteoimmunological Disorders
2.1. Osteoporosis (OP)
2.2. Osteoarthritis
2.3. Rheumatoid Arthritis
2.4. Periodontitis
2.5. Other Osteoimmunological Disorders
3. Osteoimmunology of the Breast Tissue during a Woman’s Lifetime
3.1. Pregnancy and Lactation
3.2. Postlactation Lobular Involution
3.3. Age-Related Lobular Involution
3.4. Inflammation in Breast Carcinogenesis
4. Evidence of the Presence of Osteoblastic and Osteoclastic Lineages in the Breast Tissue
4.1. Osteoblast-Like Breast Cells
4.2. Osteoclast-Like Giant Breast Cells
5. Osteoblastic and Osteoclastic Breast Cancer Metastases in Bone Microenvironment
5.1. Osteolytic Lesions
5.2. Osteoblastic and Mixed Lesions
6. Drugs Commonly Prescribed for Osteoporosis or Rheumatoid Arthritis Treatment and Their Impact on Breast Cancer Prevalence
6.1. Hormone Replacement Therapies
6.2. Antiresorptive Drugs
6.2.1. Bisphosphonates
6.2.2. Selective Estrogen Receptor Modulators (SERMs)
6.2.3. Denosumab
6.3. Bone Anabolic Drugs
6.3.1. Parathyroid Hormone Analog
6.3.2. Parathyroid Hormone Related Protein Analog
6.3.3. Romosozumab, an Anti-Sclerostin Monoclonal Antibody
6.4. Biological Agents Targeting Pro-Inflammatory Cytokines in Rheumatoid Arthritis
6.4.1. IL-1
6.4.2. IL-6
6.4.3. TNF-α
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| OP | osteoporosis |
| IL-1 | interleukin 1 |
| TNF-α | tumor necrosis factor-alpha |
| RANKL | Receptor Activator of Nuclear factor Kappa-B Ligand |
| Ob-R | leptin receptor |
| MSCs | mesenchymal stem cells |
| SFRP1 | secreted Frizzled-Related Protein 1 |
| OA | osteoarthritis |
| IL-6 | interleukin 6 |
| Th17 | T helper 17 |
| IL-17 | interleukin 17 |
| ADRB2 | beta-2 adrenergic receptor |
| RA | rheumatoid arthritis |
| Th1 | T helper 1 |
| Treg | T regulator |
| Dkk-1 | dikkopf-1 |
| TGF-ß | transforming growth factor-beta |
| PD | periodontitis |
| CD4 | surface glycoprotein CD4 |
| Th2 | T helper 2 |
| IL-8 | interleukin 8 |
| SLE | systemic lupus erythematosus |
| SSc | systemic sclerosis |
| PsA | psoriatic arthritis |
| BMI | body mass index |
| PMCA2 | plasma Membrane Ca2+-ATPase |
| JAK | tyrosine-protein kinase JAK |
| STAT | signal transducer and activator of transcription |
| MAPK | mitogen-activated protein kinase |
| ERK | extracellular signal-regulated kinase |
| ER-α | estrogen receptor-alpha |
| NF-κB | Nuclear Factor-Kappa B |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| OPN | osteopontin |
| SPP1 | secreted phosphoprotein 1 |
| ER | estrogen receptor |
| PR | progesterone receptor |
| HER2 | receptor tyrosine-protein kinase erbB-2 |
| CRP | C-reactive protein |
| COX-2 | cyclooxygenase 2 |
| SAA1 | serum amyloid 1 |
| IL-10 | interleukin 10 |
| IDC | invasive ductal carcinoma |
| DCIS | ductal carcinoma in situ |
| RANTES | regulated upon activation, normal T-cell expressed and secreted |
| EMT | epithelial to mesenchymal transition |
| RUNX2 | runt-related transcription factor 2 |
| OBL | osteoblast-like cell |
| VDR | vitamin D3 receptor |
| BMP-2 | bone morphogenetic protein 2 |
| BMP-4 | bone morphogenetic protein 4 |
| PTX3 | pentraxin-related protein PTX3 |
| OCL | osteoclast-like giant cell |
| ICAM-1 | intracellular adhesion molecule 1 |
| PTHrP | parathyroid hormone-related protein |
| OPG | osteoprotegerin |
| ET-1 | endothelin-1 |
| HRT | hormone replacement therapies |
| SERM | selective estrogen receptor modulator |
| MORE | Multiple Outcomes Raloxifene Evaluation |
| CORE | Continuing Outcomes Relevant to Evista |
| NSABP | National Surgical Adjuvant Breast and Bowel Project |
| BCPT | Breast Cancer Prevention Trial |
| WT | wild type |
| PTHPTHrP | parathyroid hormoneparathyroid hormone related protein |
| FDA | Food and Drug Administration |
| US | United-States |
| SOST | sclerostin |
| LEC | lymphatic endothelial cells |
References
- Arron, J.R.; Choi, Y. Bone versus immune system. Nature 2000, 408, 535–536. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, J. Cytokines and Bone: Osteoimmunology. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Okamoto, K.; Takayanagi, H. Osteoimmunology. Cold Spring Harb. Perspect. Med. 2019, 9, a031245. [Google Scholar] [CrossRef] [PubMed]
- Ralston, S.H.; Schett, G. Osteoimmunology. Calcif. Tissue Int. 2018, 102, 501–502. [Google Scholar] [CrossRef] [PubMed]
- Guder, C.; Gravius, S.; Burger, C.; Wirtz, D.C.; Schildberg, F.A. Osteoimmunology: A Current Update of the Interplay Between Bone and the Immune System. Front. Immunol. 2020, 11, 58. [Google Scholar] [CrossRef]
- Tsukasaki, M.; Takayanagi, H. Osteoimmunology: Evolving concepts in bone–immune interactions in health and disease. Nat. Rev. Immunol. 2019, 19, 626–642. [Google Scholar] [CrossRef]
- Subramani, R.; Lakshmanaswamy, R. Pregnancy and Breast Cancer. In Progress in Molecular Biology and Translational Science; Elsevier: Amsterdam, The Netherlands, 2017; Volume 151, pp. 81–111. ISBN 978-0-12-812772-8. [Google Scholar]
- Bernier, M.O. Breastfeeding and risk of breast cancer: A meta-analysis of published studies. Hum. Reprod. Update 2000, 6, 374–386. [Google Scholar] [CrossRef]
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and breastfeeding: Collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50 302 women with breast cancer and 96 973 women without the disease. Lancet 2002, 360, 187–195. [Google Scholar] [CrossRef]
- Faupel-Badger, J.M.; Arcaro, K.F.; Balkam, J.J.; Eliassen, A.H.; Hassiotou, F.; Lebrilla, C.B.; Michels, K.B.; Palmer, J.R.; Schedin, P.; Stuebe, A.M.; et al. Postpartum Remodeling, Lactation, and Breast Cancer Risk: Summary of a National Cancer Institute–Sponsored Workshop. JNCI J. Natl. Cancer Inst. 2013, 105, 166–174. [Google Scholar] [CrossRef]
- Walker, R.; Martin, C. The aged breast. J. Pathol. 2007, 211, 232–240. [Google Scholar] [CrossRef]
- Gierach, G.L.; Patel, D.A.; Pfeiffer, R.M.; Figueroa, J.D.; Linville, L.; Papathomas, D.; Johnson, J.M.; Chicoine, R.E.; Herschorn, S.D.; Shepherd, J.A.; et al. Relationship of Terminal Duct Lobular Unit Involution of the Breast with Area and Volume Mammographic Densities. Cancer Prev. Res. 2016, 9, 149–158. [Google Scholar] [CrossRef]
- Ferretti, G.; Felici, A.; Cognetti, F. Re: Age-Related Lobular Involution and Risk of Breast Cancer. JNCI J. Natl. Cancer Inst. 2007, 99, 571–572. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hanna, M.; Dumas, I.; Jacob, S.; Têtu, B.; Diorio, C. Physical activity, mammographic density, and age-related lobular involution among premenopausal and postmenopausal women. Menopause 2015, 22, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Henson, D.E. On the possible role of involution in the natural history of breast cancer. Cancer 1993, 71, 2154–2156. [Google Scholar] [CrossRef]
- Radisky, D.C.; Visscher, D.W.; Frank, R.D.; Vierkant, R.A.; Winham, S.; Stallings-Mann, M.; Hoskin, T.L.; Nassar, A.; Vachon, C.M.; Denison, L.A.; et al. Natural history of age-related lobular involution and impact on breast cancer risk. Breast Cancer Res. Treat. 2016, 155, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, O.M.; Martin, L.J.; Boyd, N.F. Mammographic density, lobular involution, and risk of breast cancer. Br. J. Cancer 2008, 99, 1369–1374. [Google Scholar] [CrossRef]
- Radisky, D.C.; Hartmann, L.C. Mammary Involution and Breast Cancer Risk: Transgenic Models and Clinical Studies. J. Mammary Gland Biol. Neoplasia 2009, 14, 181–191. [Google Scholar] [CrossRef]
- Henson, D.E.; Tarone, R.E.; Nsouli, H. Lobular Involution: The Physiological Prevention of Breast Cancer. JNCI J. Natl. Cancer Inst. 2006, 98, 1589–1590. [Google Scholar] [CrossRef][Green Version]
- Henson, D.E.; Tarone, R.E. Involution and the etiology of breast cancer. Cancer 1994, 74, 424–429. [Google Scholar] [CrossRef]
- Ghosh, K.; Vachon, C.M.; Pankratz, V.S.; Vierkant, R.A.; Anderson, S.S.; Brandt, K.R.; Visscher, D.W.; Reynolds, C.; Frost, M.H.; Hartmann, L.C. Independent Association of Lobular Involution and Mammographic Breast Density With Breast Cancer Risk. JNCI J. Natl. Cancer Inst. 2010, 102, 1716–1723. [Google Scholar] [CrossRef]
- Bodelon, C.; Oh, H.; Chatterjee, N.; Garcia-Closas, M.; Palakal, M.; Sherman, M.E.; Pfeiffer, R.M.; Geller, B.M.; Vacek, P.M.; Weaver, D.L.; et al. Association between breast cancer genetic susceptibility variants and terminal duct lobular unit involution of the breast: SNPs and TDLU involution of the breast. Int. J. Cancer 2017, 140, 825–832. [Google Scholar] [CrossRef]
- Yang, X.R.; Figueroa, J.D.; Falk, R.T.; Zhang, H.; Pfeiffer, R.M.; Hewitt, S.M.; Lissowska, J.; Peplonska, B.; Brinton, L.; Garcia-Closas, M.; et al. Analysis of terminal duct lobular unit involution in luminal A and basal breast cancers. Breast Cancer Res. 2012, 14. [Google Scholar] [CrossRef] [PubMed]
- Milanese, T.R.; Hartmann, L.C.; Sellers, T.A.; Frost, M.H.; Vierkant, R.A.; Maloney, S.D.; Pankratz, V.S.; Degnim, A.C.; Vachon, C.M.; Reynolds, C.A.; et al. Age-Related Lobular Involution and Risk of Breast Cancer. JNCI J. Natl. Cancer Inst. 2006, 98, 1600–1607. [Google Scholar] [CrossRef] [PubMed]
- Rominger, M.; Wisgickl, C.; Timmesfeld, N. Breast Microcalcifications as Type Descriptors to Stratify risk of Malignancy: A Systematic Review and Meta-Analysis of 10665 Cases with Special Focus on Round/Punctate Microcalcifications. Fortschr Röntgenstr 2012, 184, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Kim, H.Y.; Kim, E.-K.; Kim, M.J.; Moon, H.J.; Yoon, J.H. Evaluation of Malignancy Risk Stratification of Microcalcifications Detected on Mammography: A Study Based on the 5th Edition of BI-RADS. Ann. Surg. Oncol. 2015, 22, 2895–2901. [Google Scholar] [CrossRef] [PubMed]
- Sharma, T.; Radosevich, J.A.; Pachori, G.; Mandal, C.C. A Molecular View of Pathological Microcalcification in Breast Cancer. J. Mammary Gland Biol. Neoplasia 2016, 21, 25–40. [Google Scholar] [CrossRef]
- Bonfiglio, R.; Scimeca, M.; Toschi, N.; Pistolese, C.A.; Giannini, E.; Antonacci, C.; Ciuffa, S.; Tancredi, V.; Tarantino, U.; Albonici, L.; et al. Radiological, Histological and Chemical Analysis of Breast Microcalcifications: Diagnostic Value and Biological Significance. J. Mammary Gland Biol. Neoplasia 2018, 23, 89–99. [Google Scholar] [CrossRef]
- Ward, M.M.; Alehashemi, S. Risks of solid cancers in elderly persons with osteoarthritis or ankylosing spondylitis. Rheumatology 2020, keaa166. [Google Scholar] [CrossRef] [PubMed]
- Matzelle, M.M.; Gallant, M.A.; Condon, K.W.; Walsh, N.C.; Manning, C.A.; Stein, G.S.; Lian, J.B.; Burr, D.B.; Gravallese, E.M. Resolution of inflammation induces osteoblast function and regulates the Wnt signaling pathway. Arthritis Rheum. 2012, 64, 1540–1550. [Google Scholar] [CrossRef]
- Chen, H.-H.; Lin, C.-H.; Chen, D.-Y.; Chao, W.-C.; Chen, Y.-H.; Hung, W.-T.; Chou, Y.-Y.; Wu, Y.-D.; Chen, C.-C. Risk of major autoimmune diseases in female breast cancer patients: A nationwide, population-based cohort study. PLoS ONE 2019, 14, e0222860. [Google Scholar] [CrossRef]
- Bhandari, B.; Basyal, B.; Sarao, M.S.; Nookala, V.; Thein, Y. Prevalence of Cancer in Rheumatoid Arthritis: Epidemiological Study Based on the National Health and Nutrition Examination Survey (NHANES). Cureus 2020. [Google Scholar] [CrossRef]
- Tian, G.; Liang, J.-N.; Wang, Z.-Y.; Zhou, D. Breast Cancer Risk in Rheumatoid Arthritis: An Update Meta-Analysis. Biomed Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Wadström, H.; Pettersson, A.; Smedby, K.E.; Askling, J. Risk of breast cancer before and after rheumatoid arthritis, and the impact of hormonal factors. Ann. Rheum. Dis. 2020, 79, 581–586. [Google Scholar] [CrossRef]
- Shi, T.; Min, M.; Sun, C.; Zhang, Y.; Liang, M.; Sun, Y. Periodontal disease and susceptibility to breast cancer: A meta-analysis of observational studies. J. Clin Periodontol. 2018, 45, 1025–1033. [Google Scholar] [CrossRef]
- Sfreddo, C.S.; Maier, J.; De David, S.C.; Susin, C.; Moreira, C.H.C. Periodontitis and breast cancer: A case-control study. Community Dent. Oral Epidemiol. 2017, 45, 545–551. [Google Scholar] [CrossRef]
- Shao, J.; Wu, L.; Leng, W.-D.; Fang, C.; Zhu, Y.-J.; Jin, Y.-H.; Zeng, X.-T. Periodontal Disease and Breast Cancer: A Meta-Analysis of 1,73,162 Participants. Front. Oncol. 2018, 8, 601. [Google Scholar] [CrossRef]
- Cummings, S.R.; Eckert, S.; Krueger, K.A.; Grady, D.; Powles, T.J.; Cauley, J.A.; Norton, L.; Nickelsen, T.; Bjarnason, N.H.; Morrow, M.; et al. The Effect of Raloxifene on Risk of Breast Cancer in Postmenopausal Women: Results From the MORE Randomized Trial. Obstet. Gynecol. Surv. 2000, 55, 100. [Google Scholar] [CrossRef]
- Cauley, J.A.; Norton, L.; Lippman, M.E.; Eckert, S.; Krueger, K.A.; Purdie, D.W.; Farrerons, J.; Karasik, A.; Mellstrom, D.; Ng, K.W.; et al. Continued Breast Cancer Risk Reduction in Postmenopausal Women Treated with Raloxifene: 4-Year Results from the MORE Trial. Breast Cancer Res. Treat. 2001, 65, 125–134. [Google Scholar] [CrossRef]
- Gnant, M.; Pfeiler, G.; Steger, G.G.; Egle, D.; Greil, R.; Fitzal, F.; Wette, V.; Balic, M.; Haslbauer, F.; Melbinger-Zeinitzer, E.; et al. Adjuvant denosumab in postmenopausal patients with hormone receptor-positive breast cancer (ABCSG-18): Disease-free survival results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 339–351. [Google Scholar] [CrossRef]
- Vetter, M.; Landin, J.; Szczerba, B.M.; Castro-Giner, F.; Gkountela, S.; Donato, C.; Krol, I.; Scherrer, R.; Balmelli, C.; Malinovska, A.; et al. Denosumab treatment is associated with the absence of circulating tumor cells in patients with breast cancer. Breast Cancer Res. 2018, 20, 141. [Google Scholar] [CrossRef]
- Holen, I.; Lefley, D.V.; Francis, S.E.; Rennicks, S.; Bradbury, S.; Coleman, R.E.; Ottewell, P. IL-1 drives breast cancer growth and bone metastasis in vivo. Oncotarget 2016, 7, 75571–75584. [Google Scholar] [CrossRef]
- Wu, T.-C.; Xu, K.; Martinek, J.; Young, R.R.; Banchereau, R.; George, J.; Turner, J.; Kim, K.I.; Zurawski, S.; Wang, X.; et al. IL1 Receptor Antagonist Controls Transcriptional Signature of Inflammation in Patients with Metastatic Breast Cancer. Cancer Res. 2018, 78, 5243–5258. [Google Scholar] [CrossRef] [PubMed]
- Tulotta, C.; Lefley, D.V.; Freeman, K.; Gregory, W.M.; Hanby, A.M.; Heath, P.R.; Nutter, F.; Wilkinson, J.M.; Spicer-Hadlington, A.R.; Liu, X.; et al. Endogenous Production of IL1B by Breast Cancer Cells Drives Metastasis and Colonization of the Bone Microenvironment. Clin Cancer Res. 2019, 25, 2769–2782. [Google Scholar] [CrossRef] [PubMed]
- Korkaya, H.; Kim, G.; Davis, A.; Malik, F.; Henry, N.L.; Ithimakin, S.; Quraishi, A.A.; Tawakkol, N.; D’Angelo, R.; Paulson, A.K.; et al. Activation of an IL6 Inflammatory Loop Mediates Trastuzumab Resistance in HER2+ Breast Cancer by Expanding the Cancer Stem Cell Population. Mol. Cell 2012, 47, 570–584. [Google Scholar] [CrossRef]
- Rodriguez-Barrueco, R.; Yu, J.; Saucedo-Cuevas, L.P.; Olivan, M.; Llobet-Navas, D.; Putcha, P.; Castro, V.; Murga-Penas, E.M.; Collazo-Lorduy, A.; Castillo-Martin, M.; et al. Inhibition of the autocrine IL-6–JAK2–STAT3–calprotectin axis as targeted therapy for HR−/HER2+ breast cancers. Genes Dev. 2015, 29, 1631–1648. [Google Scholar] [CrossRef]
- Alraouji, N.N.; Al-Mohanna, F.H.; Ghebeh, H.; Arafah, M.; Almeer, R.; Al-Tweigeri, T.; Aboussekhra, A. Tocilizumab potentiates cisplatin cytotoxicity and targets cancer stem cells in triple-negative breast cancer. Mol. Carcinog. 2020, 59, 1041–1051. [Google Scholar] [CrossRef]
- Jin, K.; Pandey, N.B.; Popel, A.S. Simultaneous blockade of IL-6 and CCL5 signaling for synergistic inhibition of triple-negative breast cancer growth and metastasis. Breast Cancer Res. 2018, 20, 54. [Google Scholar] [CrossRef]
- Ettinger, B.; Pressman, A.; Sklarin, P.; Bauer, D.C.; Cauley, J.A.; Cummings, S.R. Associations between Low Levels of Serum Estradiol, Bone Density, and Fractures among Elderly Women: The Study of Osteoporotic Fractures. J. Clin. Endocrinol. Metab. 1998, 83, 2239–2243. [Google Scholar] [CrossRef]
- Cummings, S.R.; Ensrud, K. Endogenous Hormones and the Risk of Hip and Vertebral Fractures among Older Women. N. Engl. J. Med. 1998, 339, 733–738. [Google Scholar] [CrossRef]
- Garnero, P.; Sornay-Rendu, E.; Claustrat, B.; Delmas, P.D. Biochemical Markers of Bone Turnover, Endogenous Hormones and the Risk of Fractures in Postmenopausal Women: The OFELY Study. J. Bone Miner. Res. 2000, 15, 1526–1536. [Google Scholar] [CrossRef]
- Pacifici, R.; Brown, C.; Puscheck, E.; Friedrich, E.; Slatopolsky, E.; Maggio, D.; Mccracken, R.; Avioli, L.V. Effect of surgical menopause and estrogen replacement on cytokine release from human blood mononuclear cells. Proc. Natl. Acad. Sci. USA 1991, 88, 5134–5138. [Google Scholar] [CrossRef]
- Breuil, V.; Ticchioni, M.; Testa, J.; Roux, C.H.; Ferrari, P.; Breittmayer, J.P.; Albert-Sabonnadière, C.; Durant, J.; De Perreti, F.; Bernard, A.; et al. Immune changes in post-menopausal osteoporosis: The Immunos study. Osteoporos. Int. 2010, 21, 805–814. [Google Scholar] [CrossRef]
- D’Amelio, P.; Grimaldi, A.; Di Bella, S.; Brianza, S.Z.M.; Cristofaro, M.A.; Tamone, C.; Giribaldi, G.; Ulliers, D.; Pescarmona, G.P.; Isaia, G. Estrogen deficiency increases osteoclastogenesis up-regulating T cells activity: A key mechanism in osteoporosis. Bone 2008, 43, 92–100. [Google Scholar] [CrossRef]
- Adeel, S.; Singh, K.; Vydareny, K.H.; Kumari, M.; Shah, E.; Weitzmann, M.N.; Tangpricha, V. Bone Loss in Surgically Ovariectomized Premenopausal Women Is Associated With T Lymphocyte Activation and Thymic Hypertrophy. J. Investig. Med. 2013, 61, 1178–1183. [Google Scholar] [CrossRef]
- Dumond, H.; Presle, N.; Terlain, B.; Mainard, D.; Loeuille, D.; Netter, P.; Pottie, P. Evidence for a key role of leptin in osteoarthritis. Arthritis Rheum. 2003, 48, 3118–3129. [Google Scholar] [CrossRef]
- Wang, T.; He, C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018, 44, 38–50. [Google Scholar] [CrossRef]
- Astudillo, P.; Ríos, S.; Pastenes, L.; Pino, A.M.; Rodríguez, J.P. Increased adipogenesis of osteoporotic human-mesenchymal stem cells (MSCs) characterizes by impaired leptin action. J. Cell. Biochem. 2008, 103, 1054–1065. [Google Scholar] [CrossRef]
- Rehn, M.; Pihlajaniemi, T. Identification of three N-terminal ends of type XVIII collagen chains and tissue-specific differences in the expression of the corresponding transcripts. J. Biol. Chem. 1995, 270, 4705–4711. [Google Scholar] [CrossRef]
- Bhanot, P.; Brink, M.; Samos, C.H.; Hsieh, J.C.; Wang, Y.; Macke, J.P.; Andrew, D.; Nathans, J.; Nusse, R. A new member of the frizzled family from Drosophila functions as a Wingless receptor. Nature 1996, 382, 225–230. [Google Scholar] [CrossRef]
- Finch, P.W.; He, X.; Kelley, M.J.; Uren, A.; Schaudies, R.P.; Popescu, N.C.; Rudikoff, S.; Aaronson, S.A.; Varmus, H.E.; Rubin, J.S. Purification and molecular cloning of a secreted, Frizzled-related antagonist of Wnt action. Proc. Natl. Acad. Sci. USA 1997, 94, 6770–6775. [Google Scholar] [CrossRef]
- Bafico, A.; Gazit, A.; Pramila, T.; Finch, P.W.; Yaniv, A.; Aaronson, S.A. Interaction of Frizzled Related Protein (FRP) with Wnt Ligands and the Frizzled Receptor Suggests Alternative Mechanisms for FRP Inhibition of Wnt Signaling. J. Biol. Chem. 1999, 274, 16180–16187. [Google Scholar] [CrossRef]
- Üren, A.; Reichsman, F.; Anest, V.; Taylor, W.G.; Muraiso, K.; Bottaro, D.P.; Cumberledge, S.; Rubin, J.S. Secreted frizzled-related protein-1 binds directly to wingless and is a biphasic modulator of wnt signaling. J. Biol. Chem. 2000, 275, 4374–4382. [Google Scholar] [CrossRef]
- Bodine, P.V.N.; Zhao, W.; Kharode, Y.P.; Bex, F.J.; Lambert, A.-J.; Goad, M.B.; Gaur, T.; Stein, G.S.; Lian, J.B.; Komm, B.S. The Wnt Antagonist Secreted Frizzled-Related Protein-1 Is a Negative Regulator of Trabecular Bone Formation in Adult Mice. Mol. Endocrinol. 2004, 18, 1222–1237. [Google Scholar] [CrossRef]
- Bodine, P.V.N.; Billiard, J.; Moran, R.A.; Ponce-de-Leon, H.; McLarney, S.; Mangine, A.; Scrimo, M.J.; Bhat, R.A.; Stauffer, B.; Green, J.; et al. The Wnt antagonist secreted frizzled-related protein-1 controls osteoblast and osteocyte apoptosis. J. Cell. Biochem. 2005, 96, 1212–1230. [Google Scholar] [CrossRef]
- Boudin, E.; Fijalkowski, I.; Piters, E.; Van Hul, W. The role of extracellular modulators of canonical Wnt signaling in bone metabolism and diseases. Semin. Arthritis Rheum. 2013, 43, 220–240. [Google Scholar] [CrossRef]
- Monroe, D.G.; McGee-Lawrence, M.E.; Oursler, M.J.; Westendorf, J.J. Update on Wnt signaling in bone cell biology and bone disease. Gene 2012, 492, 1–18. [Google Scholar] [CrossRef]
- Taipaleenmäki, H.; Abdallah, B.M.; AlDahmash, A.; Säämänen, A.-M.; Kassem, M. Wnt signalling mediates the cross-talk between bone marrow derived pre-adipocytic and pre-osteoblastic cell populations. Exp. Cell Res. 2011, 317, 745–756. [Google Scholar] [CrossRef]
- Häusler, K.D.; Horwood, N.J.; Chuman, Y.; Fisher, J.L.; Ellis, J.; Martin, T.J.; Rubin, J.S.; Gillespie, M.T. Secreted Frizzled-Related Protein-1 Inhibits RANKL-Dependent Osteoclast Formation. J. Bone Miner. Res. 2004, 19, 1873–1881. [Google Scholar] [CrossRef]
- Tang, L.; Lu, W.; Huang, J.; Tang, X.; Zhang, H.; Liu, S. miR-144 promotes the proliferation and differentiation of bone mesenchymal stem cells by downregulating the expression of SFRP1. Mol. Med. Rep. 2019. [Google Scholar] [CrossRef]
- Gu, H.; Wu, L.; Chen, H.; Huang, Z.; Xu, J.; Zhou, K.; Zhang, Y.; Chen, J.; Xia, J.; Yin, X. Identification of differentially expressed microRNAs in the bone marrow of osteoporosis patients. Am. J. Transl. Res. 2019, 11, 2940–2954. [Google Scholar]
- Kothari, C.; Diorio, C.; Durocher, F. The Importance of Breast Adipose Tissue in Breast Cancer. Int. J. Mol. Sci. 2020, 21, 5760. [Google Scholar] [CrossRef]
- Fontanges, E.; Fontana, A.; Delmas, P. Osteoporosis and breast cancer. Jt. Bone Spine 2004, 71, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Woodell-May, J.E.; Sommerfeld, S.D. Role of Inflammation and the Immune System in the Progression of Osteoarthritis. J. Orthop. Res. 2020, 38, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Michou, L.; Numan, M.; Amiable, N.; Brown, J.P. Paget’s disease of bone: An osteoimmunological disorder? DDDT 2015, 4695. [Google Scholar] [CrossRef]
- Wojdasiewicz, P.; Poniatowski, Ł.A.; Szukiewicz, D. The Role of Inflammatory and Anti-Inflammatory Cytokines in the Pathogenesis of Osteoarthritis. Mediat. Inflamm. 2014, 2014, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Chow, Y.Y.; Chin, K.-Y. The Role of Inflammation in the Pathogenesis of Osteoarthritis. Mediat. Inflamm. 2020, 2020, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Elefteriou, F.; Ahn, J.D.; Takeda, S.; Starbuck, M.; Yang, X.; Liu, X.; Kondo, H.; Richards, W.G.; Bannon, T.W.; Noda, M.; et al. Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature 2005, 434, 514–520. [Google Scholar] [CrossRef]
- Pasold, J.; Osterberg, A.; Peters, K.; Taipaleenmäki, H.; Säämänen, A.-M.; Vollmar, B.; Müller-Hilke, B. Reduced expression of Sfrp1 during chondrogenesis and in articular chondrocytes correlates with osteoarthritis in STR/ort mice. Exp. Cell Res. 2013, 319, 649–659. [Google Scholar] [CrossRef]
- Di Sante, G.; Tolusso, B.; Fedele, A.L.; Gremese, E.; Alivernini, S.; Nicolò, C.; Ria, F.; Ferraccioli, G. Collagen Specific T-Cell Repertoire and HLA-DR Alleles: Biomarkers of Active Refractory Rheumatoid Arthritis. EBioMedicine 2015, 2, 2037–2045. [Google Scholar] [CrossRef]
- McInnes, I.B.; Schett, G. The Pathogenesis of Rheumatoid Arthritis. N. Engl. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef]
- Pandolfi, F.; Franza, L.; Carusi, V.; Altamura, S.; Andriollo, G.; Nucera, E. Interleukin-6 in Rheumatoid Arthritis. Int. J. Mol. Sci. 2020, 21, 5238. [Google Scholar] [CrossRef]
- McInnes, I.B.; Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat. Rev. Immunol. 2007, 7, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Taghadosi, M.; Samimi, Z.; Assar, S.; Salahshoor, M.R.; Jalili, C. Plasma Leptin Does Not Reflect the Effect of High Body Mass Index on Disease Activity in Rheumatoid Arthritis. Immunol. Investig. 2020, 49, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.; Lafaurie, G.I.; Bautista-Molano, W.; Chila-Moreno, L.; Bello-Gualtero, J.M.; Romero-Sánchez, C. Adipokines and periodontal markers as risk indicators of early rheumatoid arthritis: A cross-sectional study. Clin. Oral Investig. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.A.; English, A.; Henshaw, K.; Kinsey, S.E.; Markham, A.F.; Emery, P.; McGonagle, D. Enumeration and phenotypic characterization of synovial fluid multipotential mesenchymal progenitor cells in inflammatory and degenerative arthritis. Arthritis Rheum. 2004, 50, 817–827. [Google Scholar] [CrossRef]
- Djouad, F.; Bony, C.; Häupl, T.; Uzé, G.; Lahlou, N.; Louis-Plence, P.; Apparailly, F.; Canovas, F.; Rème, T.; Sany, J.; et al. Transcriptional profiles discriminate bone marrow-derived and synovium-derived mesenchymal stem cells. Arthritis Res. Ther. 2005, 7, R1304. [Google Scholar] [CrossRef]
- Diarra, D.; Stolina, M.; Polzer, K.; Zwerina, J.; Ominsky, M.S.; Dwyer, D.; Korb, A.; Smolen, J.; Hoffmann, M.; Scheinecker, C.; et al. Dickkopf-1 is a master regulator of joint remodeling. Nat. Med. 2007, 13, 156–163. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Lee, K.-A.; Yoon, H.-B.; Yoo, S.-A.; Park, Y.W.; Chung, Y.; Kim, W.-U.; Kang, C.-Y. The Wnt inhibitor secreted Frizzled-Related Protein 1 (sFRP1) promotes human Th17 differentiation: Cellular immune response. Eur. J. Immunol. 2012, 42, 2564–2573. [Google Scholar] [CrossRef]
- Clemenceau, A.; Hanna, M.; Ennour-Idrissi, K.; Burguin, A.; Diorio, C.; Durocher, F. Secreted Frizzled-Related Protein 1 as a Biomarker against Incomplete Age-Related Lobular Involution and Microcalcifications’ Development. Cancers 2020, 12, 2693. [Google Scholar] [CrossRef]
- Ponzetti, M.; Rucci, N. Updates on Osteoimmunology: What’s New on the Cross-Talk Between Bone and Immune System. Front. Endocrinol. 2019, 10, 236. [Google Scholar] [CrossRef]
- Brunetti, G.; Colucci, S.; Pignataro, P.; Coricciati, M.; Mori, G.; Cirulli, N.; Zallone, A.; Grassi, F.R.; Grano, M. T Cells Support Osteoclastogenesis in an In Vitro Model Derived From Human Periodontitis Patients. J. Periodontol. 2005, 76, 1675–1680. [Google Scholar] [CrossRef]
- Kawai, T.; Matsuyama, T.; Hosokawa, Y.; Makihira, S.; Seki, M.; Karimbux, N.Y.; Goncalves, R.B.; Valverde, P.; Dibart, S.; Li, Y.-P.; et al. B and T Lymphocytes Are the Primary Sources of RANKL in the Bone Resorptive Lesion of Periodontal Disease. Am. J. Pathol. 2006, 169, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Campbell, L.; Millhouse, E.; Malcolm, J.; Culshaw, S. T cells, teeth and tissue destruction—What do T cells do in periodontal disease? Mol. Oral Microbiol. 2016, 31, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Cekici, A.; Kantarci, A.; Hasturk, H.; Van Dyke, T.E. Inflammatory and immune pathways in the pathogenesis of periodontal disease: Inflammatory and immune pathways in periodontal disease. Periodontology 2000 2014, 64, 57–80. [Google Scholar] [CrossRef]
- Hoare, A.; Soto, C.; Rojas-Celis, V.; Bravo, D. Chronic Inflammation as a Link between Periodontitis and Carcinogenesis. Mediat. Inflamm. 2019, 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Purwar, P.; Khan, M.A.; Mahdi, A.A.; Pandey, S.; Singh, B.; Dixit, J.; Sareen, S. Salivary and Serum Leptin Concentrations in Patients With Chronic Periodontitis. J. Periodontol. 2015, 86, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Huang, B.; Liu, K.; Hou, J.; Meng, H. Upregulated Leptin in Periodontitis Promotes Inflammatory Cytokine Expression in Periodontal Ligament Cells. J. Periodontol. 2015, 86, 917–926. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Guo, B.; Gan, X.; Zhang, L.; He, Y.; Liu, B.; Chen, X.; Zhang, S.; Yu, H. Association of circulating leptin and adiponectin with periodontitis: A systematic review and meta-analysis. BMC Oral Health 2017, 17, 104. [Google Scholar] [CrossRef]
- Li, C.H.; Amar, S. Inhibition of SFRP1 Reduces Severity of Periodontitis. J. Dent. Res. 2007, 86, 873–877. [Google Scholar] [CrossRef]
- Chan, K.; Clarke, A.E.; Ramsey-Goldman, R.; Foulkes, W.; Tessier Cloutier, B.; Urowitz, M.B.; Gladman, D.; Nived, O.; Romero-Diaz, J.; Petri, M.; et al. Breast cancer in systemic lupus erythematosus (SLE): Receptor status and treatment. Lupus 2018, 27, 120–123. [Google Scholar] [CrossRef]
- Bernatsky, S.; Ramsey-Goldman, R.; Petri, M.; Urowitz, M.B.; Gladman, D.D.; Fortin, P.F.; Ginzler, E.; Romero-Diaz, J.; Peschken, C.; Jacobsen, S.; et al. Breast cancer in systemic lupus. Lupus 2017, 26, 311–315. [Google Scholar] [CrossRef]
- Bernatsky, S.; Kale, M.; Ramsey-Goldman, R.; Gordon, C.; Clarke, A.E. Systemic lupus and malignancies. Curr. Opin. Rheumatol. 2012, 24, 177–181. [Google Scholar] [CrossRef]
- Bernatsky, S.; Ramsey-Goldman, R.; Foulkes, W.D.; Gordon, C.; Clarke, A.E. Breast, ovarian, and endometrial malignancies in systemic lupus erythematosus: A meta-analysis. Br. J. Cancer 2011, 104, 1478–1481. [Google Scholar] [CrossRef] [PubMed]
- Bernatsky, S.; Ramsey-Goldman, R.; Labrecque, J.; Joseph, L.; Boivin, J.-F.; Petri, M.; Zoma, A.; Manzi, S.; Urowitz, M.B.; Gladman, D.; et al. Cancer risk in systemic lupus: An updated international multi-centre cohort study. J. Autoimmun. 2013, 42, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Parikh-Patel, A.; White, R.H.; Allen, M.; Cress, R. Cancer risk in a cohort of patients with systemic lupus erythematosus (SLE) in California. Cancer Causes Control 2008, 19, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Colaci, M.; Giuggioli, D.; Vacchi, C.; Lumetti, F.; Iachetta, F.; Marcheselli, L.; Federico, M.; Ferri, C. Breast cancer in systemic sclerosis: Results of a cross-linkage of an Italian Rheumatologic Center and a population-based Cancer Registry and review of the literature. Autoimmun. Rev. 2014, 13, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Wilton, K.M.; Crowson, C.S.; Matteson, E.L. Malignancy incidence in patients with psoriatic arthritis: A comparison cohort-based incidence study. Clin. Rheumatol. 2016, 35, 2603–2607. [Google Scholar] [CrossRef]
- Rohekar, S.; Tom, B.D.M.; Hassa, A.; Schentag, C.T.; Farewell, V.T.; Gladman, D.D. Prevalence of malignancy in psoriatic arthritis. Arthritis Rheum. 2008, 58, 82–87. [Google Scholar] [CrossRef]
- Bostoen, J.; Van Praet, L.; Brochez, L.; Mielants, H.; Lambert, J. A cross-sectional study on the prevalence of metabolic syndrome in psoriasis compared to psoriatic arthritis: Metabolic syndrome in psoriatic disease. J. Eur. Acad. Derm. Venereol. 2014, 28, 507–511. [Google Scholar] [CrossRef]
- Haroon, M.; Gallagher, P.; Heffernan, E.; FitzGerald, O. High Prevalence of Metabolic Syndrome and of Insulin Resistance in Psoriatic Arthritis is Associated with the Severity of Underlying Disease. J. Rheumatol. 2014, 41, 1357–1365. [Google Scholar] [CrossRef]
- Eder, L.; Harvey, P.; Chandran, V.; Rosen, C.F.; Dutz, J.; Elder, J.T.; Rahman, P.; Ritchlin, C.T.; Rohekar, S.; Hayday, R.; et al. Gaps in Diagnosis and Treatment of Cardiovascular Risk Factors in Patients with Psoriatic Disease: An International Multicenter Study. J. Rheumatol. 2018, 45, 378–384. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Press, M.F.; Haile, R.W.; Lynch, C.F.; Glaser, S.L.; Schildkraut, J.; Gammon, M.D.; Douglas Thompson, W.; Bernstein, J.L. Risk factors by molecular subtypes of breast cancer across a population-based study of women 56 years or younger. Breast Cancer Res. Treat. 2011, 130, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Turkoz, F.P.; Solak, M.; Petekkaya, I.; Keskin, O.; Kertmen, N.; Sarici, F.; Arik, Z.; Babacan, T.; Ozisik, Y.; Altundag, K. Association between common risk factors and molecular subtypes in breast cancer patients. Breast 2013, 22, 344–350. [Google Scholar] [CrossRef]
- Protani, M.; Coory, M.; Martin, J.H. Effect of obesity on survival of women with breast cancer: Systematic review and meta-analysis. Breast Cancer Res. Treat. 2010, 123, 627–635. [Google Scholar] [CrossRef]
- Cross, B.M.; Breitwieser, G.E.; Reinhardt, T.A.; Rao, R. Cellular calcium dynamics in lactation and breast cancer: From physiology to pathology. Am. J. Physiol. -Cell Physiol. 2014, 306, C515–C526. [Google Scholar] [CrossRef] [PubMed]
- VanHouten, J.; Sullivan, C.; Bazinet, C.; Ryoo, T.; Camp, R.; Rimm, D.L.; Chung, G.; Wysolmerski, J. PMCA2 regulates apoptosis during mammary gland involution and predicts outcome in breast cancer. Proc. Natl. Acad. Sci. USA 2010, 107, 11405–11410. [Google Scholar] [CrossRef] [PubMed]
- Koktener, A.; Cakir, B.; Akin, K.; Kosehan, D.; Bayrak, R.; Yenidunya, S. Pregnancy-like (pseudolactational) hyperplasia: Uncommon cause of microcalcifications and mass in two cases. J. Belg. Soc. Radiol. 2013, 96, 25. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gauger, K.J.; Shimono, A.; Crisi, G.M.; Schneider, S. Loss of sfrp1 promotes ductal branching in the murine mammary gland. BMC Dev. Biol. 2012, 12, 25. [Google Scholar] [CrossRef]
- Zheng, X.; Ning, C.; Dong, Y.; Zhao, P.; Li, J.; Fan, Z.; Li, J.; Yu, Y.; Mrode, R.; Liu, J. Quantitative proteome analysis of bovine mammary gland reveals protein dynamic changes involved in peak and late lactation stages. Biochem. Biophys. Res. Commun. 2017, 494, 292–297. [Google Scholar] [CrossRef]
- Aoki, N. Lactation-dependent down regulation of leptin production in mouse mammary gland. Biochim. Biophys. Acta (Bba)—Gen. Subj. 1999, 1427, 298–306. [Google Scholar] [CrossRef]
- Basree, M.M.; Shinde, N.; Koivisto, C.; Cuitino, M.; Kladney, R.; Zhang, J.; Stephens, J.; Palettas, M.; Zhang, A.; Kim, H.K.; et al. Abrupt involution induces inflammation, estrogenic signaling, and hyperplasia linking lack of breastfeeding with increased risk of breast cancer. Breast Cancer Res. 2019, 21, 80. [Google Scholar] [CrossRef]
- Brann, W.D.; Wade, M.F.; Dhandapani, K.M.; Mahesh, V.B.; Buchanan, C.D. Leptin and reproduction. Steroids 2001, 67, 95–104. [Google Scholar] [CrossRef]
- Diano, S.; Kalra, S.P.; Sakamoto, H.; Horvath, T.L. Leptin receptors in estrogen receptor-containing neurons of the female rat hypothalamus. Brain Res. 1998, 812, 256–259. [Google Scholar] [CrossRef]
- Pensa, S.; Watson, C.J.; Poli, V. Stat3 and the Inflammation/Acute Phase Response in Involution and Breast Cancer. J. Mammary Gland Biol. Neoplasia 2009, 14, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Poli, V. The Role of C/EBP Isoforms in the Control of Inflammatory and Native Immunity Functions. J. Biol. Chem. 1998, 273, 29279–29282. [Google Scholar] [CrossRef]
- Chapman, R.S.; Lourenco, P.C.; Tonner, E.; Flint, D.J.; Selbert, S.; Takeda, K.; Akira, S.; Clarke, A.R.; Watson, C.J. Suppression of epithelial apoptosis and delayed mammary gland involution in mice with a conditional knockout of Stat3. Genes Dev. 1999, 13, 2604–2616. [Google Scholar] [CrossRef]
- Humphreys, R.C.; Bierie, B.; Zhao, L.; Raz, R.; Levy, D.; Hennighausen, L. Deletion of Stat3 Blocks Mammary Gland Involution and Extends Functional Competence of the Secretory Epithelium in the Absence of Lactogenic Stimuli. Endocrinology 2002, 143, 3641–3650. [Google Scholar] [CrossRef]
- Martinson, H.A.; Jindal, S.; Durand-Rougely, C.; Borges, V.F.; Schedin, P. Wound healing-like immune program facilitates postpartum mammary gland involution and tumor progression: Immune cells in postpartum involution and breast cancer. Int. J. Cancer 2015, 136, 1803–1813. [Google Scholar] [CrossRef]
- Prokesch, A.; Smorlesi, A.; Perugini, J.; Manieri, M.; Ciarmela, P.; Mondini, E.; Trajanoski, Z.; Kristiansen, K.; Giordano, A.; Bogner-Strauss, J.G.; et al. Molecular Aspects of Adipoepithelial Transdifferentiation in Mouse Mammary Gland: Adipoepithelial Transdifferentiation. Stem Cells 2014, 32, 2756–2766. [Google Scholar] [CrossRef]
- Rittling, R.; Novick, E. Osteopontin Expression in Mammary Gland Development and Tumorigenesis. Cell Growth Differ. 1997, 10, 1061–1069. [Google Scholar]
- Huan, J.-L.; Xing, L.; Qin, X.-J.; Gao, Z.-G.; Pan, X.-F.; Zhao, Z.-D. Expression and Clinical Significance of Osteopontin in Calcified Breast Tissue. Asian Pac. J. Cancer Prev. 2012, 13, 5219–5223. [Google Scholar] [CrossRef][Green Version]
- Bellahc, A. Increased Expression of Osteonectin and Osteopontin, Two Bone Matrix Proteins, in Human Breast Cancer. Am. J. Pathol. 1995, 146, 95. [Google Scholar]
- Kothari, C.; Ouellette, G.; Labrie, Y.; Jacob, S.; Diorio, C.; Durocher, F. Identification of a gene signature for different stages of breast cancer development that could be used for early diagnosis and specific therapy. Oncotarget 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, A.; Paidi, S.K.; Zheng, C.; Cheng, M.; Barman, I.; Glunde, K. Mapping the genetic basis of breast microcalcifications and their role in metastasis. Sci. Rep. 2018, 8, 11067. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, S.M.; Rumer, K.K.; Biroc, S.L.; Metz, R.P.; Singh, M.; Porter, W.; Schedin, P. Remodeling of the Mammary Microenvironment after Lactation Promotes Breast Tumor Cell Metastasis. Am. J. Pathol. 2006, 168, 608–620. [Google Scholar] [CrossRef]
- Hanna, M.; Dumas, I.; Orain, M.; Jacob, S.; Têtu, B.; Sanschagrin, F.; Bureau, A.; Poirier, B.; Diorio, C. Association between local inflammation and breast tissue age-related lobular involution among premenopausal and postmenopausal breast cancer patients. PLoS ONE 2017, 12, e0183579. [Google Scholar] [CrossRef]
- Veeck, J.; Niederacher, D.; An, H.; Klopocki, E.; Wiesmann, F.; Betz, B.; Galm, O.; Camara, O.; Dürst, M.; Kristiansen, G.; et al. Aberrant methylation of the Wnt antagonist SFRP1 in breast cancer is associated with unfavourable prognosis. Oncogene 2006, 25, 3479–3488. [Google Scholar] [CrossRef]
- Huth, L.; Rose, M.; Kloubert, V.; Winkens, W.; Schlensog, M.; Hartmann, A.; Knüchel, R.; Dahl, E. BDNF Is Associated with SFRP1 Expression in Luminal and Basal-Like Breast Cancer Cell Lines and Primary Breast Cancer Tissues: A Novel Role in Tumor Suppression? PLoS ONE 2014, 9, e102558. [Google Scholar] [CrossRef]
- Yang, Y.; Xing, Y.; Liang, C.; Hu, L.; Xu, F.; Chen, Y. Crucial microRNAs and genes of human primary breast cancer explored by microRNA-mRNA integrated analysis. Tumor Biol. 2015, 36, 5571–5579. [Google Scholar] [CrossRef]
- Wang, Z.; Li, R.; He, Y.; Huang, S. Effects of secreted frizzled-related protein 1 on proliferation, migration, invasion, and apoptosis of colorectal cancer cells. Cancer Cell Int. 2018, 18. [Google Scholar] [CrossRef]
- Gregory, K.J.; Schneider, S.S. Estrogen-mediated signaling is differentially affected by the expression levels of Sfrp1 in mammary epithelial cells: Estrogen signaling and Sfrp1 expression. Cell Biol. Int. 2015, 39, 873–879. [Google Scholar] [CrossRef]
- Dahl, E.; Wiesmann, F.; Woenckhaus, M.; Stoehr, R.; Wild, P.J.; Veeck, J.; Knüchel, R.; Klopocki, E.; Sauter, G.; Simon, R.; et al. Frequent loss of SFRP1 expression in multiple human solid tumours: Association with aberrant promoter methylation in renal cell carcinoma. Oncogene 2007, 26, 5680–5691. [Google Scholar] [CrossRef] [PubMed]
- Gregory, K.J.; Roberts, A.L.; Conlon, E.M.; Mayfield, J.A.; Hagen, M.J.; Crisi, G.M.; Bentley, B.A.; Kane, J.J.; Makari-Judson, G.; Mason, H.S.; et al. Gene expression signature of atypical breast hyperplasia and regulation by SFRP1. Breast Cancer Res. 2019, 21. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.-C.; Wang, L.-J.; Hsiao, T.-H.; Chuang, E.Y.; Chen, Y. Genome-wide identification of key modulators of gene-gene interaction networks in breast cancer. BMC Genom. 2017, 18. [Google Scholar] [CrossRef]
- Bernemann, C.; Hülsewig, C.; Ruckert, C.; Schäfer, S.; Blümel, L.; Hempel, G.; Götte, M.; Greve, B.; Barth, P.J.; Kiesel, L.; et al. Influence of secreted frizzled receptor protein 1 (SFRP1) on neoadjuvant chemotherapy in triple negative breast cancer does not rely on WNT signaling. Mol. Cancer 2014, 13, 174. [Google Scholar] [CrossRef] [PubMed]
- Meli, R.; Pacilio, M.; Raso, G.M.; Esposito, E.; Coppola, A.; Nasti, A.; Di Carlo, C.; Nappi, C.; Di Carlo, R. Estrogen and Raloxifene Modulate Leptin and Its Receptor in Hypothalamus and Adipose Tissue from Ovariectomized Rats. Endocrinology 2004, 145, 3115–3121. [Google Scholar] [CrossRef] [PubMed]
- Naseem, M.; Murray, J.; Hilton, J.F.; Karamchandani, J.; Muradali, D.; Faragalla, H.; Polenz, C.; Han, D.; Bell, D.C.; Brezden-Masley, C. Mammographic microcalcifications and breast cancer tumorigenesis: A radiologic-pathologic analysis. BMC Cancer 2015, 15, 307. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Quigley, D.A.; Tahiri, A.; Lüders, T.; Riis, M.H.; Balmain, A.; Børresen-Dale, A.-L.; Bukholm, I.; Kristensen, V. Age, estrogen, and immune response in breast adenocarcinoma and adjacent normal tissue. OncoImmunology 2017, 6, e1356142. [Google Scholar] [CrossRef]
- Hanna, M.; Dumas, I.; Orain, M.; Jacob, S.; Têtu, B.; Sanschagrin, F.; Bureau, A.; Poirier, B.; Diorio, C. Association between expression of inflammatory markers in normal breast tissue and mammographic density among premenopausal and postmenopausal women. Menopause 2017, 24, 524–535. [Google Scholar] [CrossRef]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef]
- Desmedt, C.; Salgado, R.; Fornili, M.; Pruneri, G.; Van den Eynden, G.; Zoppoli, G.; Rothé, F.; Buisseret, L.; Garaud, S.; Willard-Gallo, K.; et al. Immune Infiltration in Invasive Lobular Breast Cancer. JNCI J. Natl. Cancer Inst. 2018, 110, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Autenshlyus, A.I.; Kunts, T.A.; Karpukhina, K.V.; Mikhaylova, E.S.; Varaksin, N.A.; Marinkin, I.O.; Lyakhovich, V.V. Cytokine pattern of the breast tumor supernatant. Dokl. Biol. Sci 2016, 470, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Clemenceau, A.; Diorio, C.; Durocher, F. Role of Secreted Frizzled-Related Protein 1 in Early Mammary Gland Tumorigenesis and Its Regulation in Breast Microenvironment. Cells 2020, 9, 208. [Google Scholar] [CrossRef] [PubMed]
- Ercan, C.; van Diest, P.J.; Vooijs, M. Mammary Development and Breast Cancer: The Role of Stem Cells. CMM 2011, 11, 270–285. [Google Scholar] [CrossRef]
- Macias, H.; Hinck, L. Mammary gland development: Mammary gland development. Wires Dev. Biol. 2012, 1, 533–557. [Google Scholar] [CrossRef]
- Reya, T.; Clevers, H. Wnt signalling in stem cells and cancer. Nature 2005, 434, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.-C.; Li, G.-X.; Tan, L.-D.; Du, X.; Li, X.-Q.; He, R.; Wang, Q.-S.; Feng, Y.-M. Breast cancer cells obtain an osteomimetic feature via epithelial-mesenchymal transition that have undergone BMP2/RUNX2 signaling pathway induction. Oncotarget 2016, 7, 79688–79705. [Google Scholar] [CrossRef]
- Scimeca, M.; Urbano, N.; Bonfiglio, R.; Schillaci, O.; Bonanno, E. Breast osteoblast-like cells: A new biomarker for the management of breast cancer. Br. J. Cancer 2018, 119, 1129–1132. [Google Scholar] [CrossRef]
- Scimeca, M.; Bonfiglio, R.; Menichini, E.; Albonici, L.; Urbano, N.; De Caro, M.T.; Mauriello, A.; Schillaci, O.; Gambacurta, A.; Bonanno, E. Microcalcifications Drive Breast Cancer Occurrence and Development by Macrophage-Mediated Epithelial to Mesenchymal Transition. Int. J. Mol. Sci. 2019, 20, 5633. [Google Scholar] [CrossRef]
- Valentin-Opran, A.; Eilon, G.; Saez, S.; Mundy, G.R. Estrogens and antiestrogens stimulate release of bone resorbing activity by cultured human breast cancer cells. J. Clin. Investig. 1985, 75, 726–731. [Google Scholar] [CrossRef]
- Vicard, E.; Valentin-Opran, A.; Chenu, C.; Delmas, P.D.; Meunier, P.J.; Saez, S. Androgens increase osteoblast-stimulating activity of human breast cancer cells in vitro. J. Steroid Biochem. 1986, 24, 401–403. [Google Scholar] [CrossRef]
- Cox, R.F.; Hernandez-Santana, A.; Ramdass, S.; McMahon, G.; Harmey, J.H.; Morgan, M.P. Microcalcifications in breast cancer: Novel insights into the molecular mechanism and functional consequence of mammary mineralisation. Br. J. Cancer 2012, 106, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.F.; Morgan, M.P. Microcalcifications in breast cancer: Lessons from physiological mineralization. Bone 2013, 53, 437–450. [Google Scholar] [CrossRef] [PubMed]
- Agnantis, N.T.; Rosen, P.P. Mammary Carcinoma with Osteoclast-like Giant Cells: A Study of Eight Cases with Follow-up Data. Am. J. Clin. Pathol. 1979, 72, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Athanasou, N.; Wells, C.; Quinn, J.; Ferguson, D.; Heryet, A.; McGee, J. The origin and nature of stromal osteoclast-like multinucleated giant cells in breast carcinoma: Implications for tumour osteolysis and macrophage biology. Br. J. Cancer 1989, 59, 491–498. [Google Scholar] [CrossRef]
- Zagelbaum, N.K.; Ward, M.F.; Okby, N.; Karpoff, H. Invasive ductal carcinoma of the breast with osteoclast-like giant cells and clear cell features: A case report of a novel finding and review of the literature. World J. Surg. Oncol. 2016, 14, 227. [Google Scholar] [CrossRef]
- Xu, Z.; Gu, J.; Zhang, S.; Zhang, Z.; Fang, W. Leiomyosarcoma with osteoclast-like (LMS-OGC) giant cells the breast: A report of a rare case. Thorac. Cancer 2019, 10, 2054–2056. [Google Scholar] [CrossRef]
- Cai, N.; Koizumi, J.; Vazquez, M. Mammary carcinoma with osteoclast-like giant cells: A study of four cases and a review of literature. Diagn. Cytopathol. 2005, 33, 246–251. [Google Scholar] [CrossRef]
- Richter, G.; Uleer, C.; Noesselt, T. Multifocal invasive ductal breast cancer with osteoclast-like giant cells: A case report. J. Med. Case Rep. 2011, 5, 85. [Google Scholar] [CrossRef]
- Cozzolino, I.; Ciancia, G.; Limite, G.; Di Micco, R.; Varone, V.; Cortese, A.; Vatrella, A.; Di Crescenzo, V.; Zeppa, P. Neuroendocrine differentiation in breast carcinoma with osteoclast-like giant cells. Report of a case and review of the literature. Int. J. Surg. 2014, 12, S8–S11. [Google Scholar] [CrossRef][Green Version]
- Peña-Jaimes, L.; González-García, I.; Reguero-Callejas, M.E.; Pinilla-Pagnon, I.; Pérez-Mies, B.; Albarrán-Artahona, V.; Martínez-Jañez, N.; Rosa-Rosa, J.M.; Palacios, J. Pleomorphic lobular carcinoma of the breast with osteoclast-like giant cells: A case report and review of the literature. Diagn. Pathol. 2018, 13, 62. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, C.; Longacre, T.A. Ductal carcinoma in situ of the breast with osteoclast-like giant cells. Hum. Pathol. 2006, 37, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Romics, L.; Mallon, E.A.; Reid, R.; Cordiner, C.M.; Doughty, J.C. Osteoclast-like giant cell tumor arising in the soft tissue of the breast: Report of a case. Surg. Today 2009, 39, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Mammary carcinoma with osteoclast-like giant cells: Additional observations on six cases. Cancer 1984, 53, 1963–1973. [CrossRef]
- Ohashi, R.; Hayama, A.; Matsubara, M.; Watarai, Y.; Sakatani, T.; Naito, Z.; Shimizu, A. Breast carcinoma with osteoclast-like giant cells: A cytological-pathological correlation with a literature review. Ann. Diagn. Pathol. 2018, 33, 1–5. [Google Scholar] [CrossRef]
- Ohashi, R.; Yanagihara, K.; Namimatsu, S.; Sakatani, T.; Takei, H.; Naito, Z.; Shimizu, A. Osteoclast-like giant cells in invasive breast cancer predominantly possess M2-macrophage phenotype. Pathol. Res. Pract. 2018, 214, 253–258. [Google Scholar] [CrossRef]
- Tsai, C.-F.; Chen, J.-H.; Wu, C.-T.; Chang, P.-C.; Wang, S.-L.; Yeh, W.-L. Induction of osteoclast-like cell formation by leptin-induced soluble intercellular adhesion molecule secreted from cancer cells. Adv. Med. Oncol. 2019, 11, 175883591984680. [Google Scholar] [CrossRef]
- Guise, T.A.; Yin, J.J.; Taylor, S.D.; Kumagai, Y.; Dallas, M.; Boyce, B.F.; Yoneda, T.; Mundy, G.R. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J. Clin. Investig. 1996, 98, 1544–1549. [Google Scholar] [CrossRef]
- Powell, G.J.; Southby, J.; Danks, J.A.; Stillwell, R.G.; Hayman, J.A.; Henderson, M.A.; Bennett, R.C.; Martin, T.J. Localization of Parathyroid Hormone-related Protein in Breast Cancer Metastases: Increased Incidence in Bone Compared with Other Sites. Cancer Res. 1991, 51, 3059–3061. [Google Scholar]
- Taipaleenmäki, H.; Farina, N.H.; van Wijnen, A.J.; Stein, J.L.; Hesse, E.; Stein, G.S.; Lian, J.B. Antagonizing miR-218-5p attenuates Wnt signaling and reduces metastatic bone disease of triple negative breast cancer cells. Oncotarget 2016, 7, 79032–79046. [Google Scholar] [CrossRef]
- Bendre, M.S.; Montague, D.C.; Peery, T.; Akel, N.S.; Gaddy, D.; Suva, L.J. Interleukin-8 stimulation of osteoclastogenesis and bone resorption is a mechanism for the increased osteolysis of metastatic bone disease. Bone 2003, 33, 28–37. [Google Scholar] [CrossRef]
- Kovacheva, M.; Zepp, M.; Schraad, M.; Berger, S.; Berger, M.R. Conditional Knockdown of Osteopontin Inhibits Breast Cancer Skeletal Metastasis. Int. J. Mol. Sci. 2019, 20, 4918. [Google Scholar] [CrossRef] [PubMed]
- Mariz, K.; Ingolf, J.-B.; Daniel, H.; Teresa, N.J.; Erich-Franz, S. The Wnt inhibitor dickkopf-1: A link between breast cancer and bone metastases. Clin. Exp. Metastasis 2015, 32, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Voorzanger-Rousselot, N.; Goehrig, D.; Journe, F.; Doriath, V.; Body, J.J.; Clézardin, P.; Garnero, P. Increased Dickkopf-1 expression in breast cancer bone metastases. Br. J. Cancer 2007, 97, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Guise, T.A.; Kozlow, W.M.; Heras-Herzig, A.; Padalecki, S.S.; Yin, J.J.; Chirgwin, J.M. Molecular Mechanisms of Breast Cancer Metastases to Bone. Clin. Breast Cancer 2005, 5, S46–S53. [Google Scholar] [CrossRef]
- Fang, J.; Xu, Q. Differences of osteoblastic bone metastases and osteolytic bone metastases in clinical features and molecular characteristics. Clin. Transl. Oncol. 2015, 17, 173–179. [Google Scholar] [CrossRef]
- Nelson, J.B.; Hedican, S.P.; George, D.J.; Reddi, A.H.; Piantadosi, S.; Eisenberger, M.A.; Simons, J.W. Identification of endothelin–1 in the pathophysiology of metastatic adenocarcinoma of the prostate. Nat. Med. 1995, 1, 944–949. [Google Scholar] [CrossRef]
- Yin, J.J.; Mohammad, K.S.; Kakonen, S.M.; Harris, S.; Wu-Wong, J.R.; Wessale, J.L.; Padley, R.J.; Garrett, I.R.; Chirgwin, J.M.; Guise, T.A. A causal role for endothelin-1 in the pathogenesis of osteoblastic bone metastases. Proc. Natl. Acad. Sci. USA 2003, 100, 10954–10959. [Google Scholar] [CrossRef]
- Clines, G.A.; Mohammad, K.S.; Bao, Y.; Stephens, O.W.; Suva, L.J.; Shaughnessy, J.D.; Fox, J.W.; Chirgwin, J.M.; Guise, T.A. Dickkopf Homolog 1 Mediates Endothelin-1-Stimulated New Bone Formation. Mol. Endocrinol. 2007, 21, 486–498. [Google Scholar] [CrossRef]
- Beral, V. Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet 2003, 362, 419–427. [Google Scholar] [CrossRef]
- Newcomb, P.A.; Titus-Ernstoff, L.; Egan, K.M.; Trentham-Dietz, A.; Baron, J.A.; Storer, B.E.; Willett, W.C.; Stampfer, M.J. Postmenopausal Estrogen and Progestin Use in Relation to Breast Cancer Risk. Cancer Epidemiol. Biomark. Prev. 2002, 11, 593–600. [Google Scholar]
- Stahlberg, C.; Pedersen, A.T.; Lynge, E.; Andersen, Z.J.; Keiding, N.; Hundrup, Y.A.; Obel, E.B.; Ottesen, B. Increased risk of breast cancer following different regimens of hormone replacement therapy frequently used in Europe. Int. J. Cancer 2004, 109, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Fournier, A.; Berrino, F.; Clavel-Chapelon, F. Unequal risks for breast cancer associated with different hormone replacement therapies: Results from the E3N cohort study. Breast Cancer Res. Treat. 2007, 107, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Ewertz, M.; Mellemkjaer, L.; Poulsen, A.H.; Friis, S.; Sørensen, H.T.; Pedersen, L.; McLaughlin, J.K.; Olsen, J.H. Hormone use for menopausal symptoms and risk of breast cancer. A Danish cohort study. Br. J. Cancer 2005, 92, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kolonel, L.; Wilkens, L.; Wan, P.; Henderson, B.; Pike, M. Postmenopausal hormone therapy and breast cancer risk: The multiethnic cohort. Int. J. Cancer 2006, 118, 1285–1291. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, C.; Baron, J.A.; Correia, N.; Bergström, R.; Adami, H.O.; Persson, I. Breast-cancer risk following long-term oestrogen- and oestrogen-progestin-replacement therapy. Int. J. Cancer 1999, 81, 339–344. [Google Scholar] [CrossRef]
- Chen, W.Y.; Manson, J.E.; Hankinson, S.E.; Rosner, B.; Holmes, M.D.; Willett, W.C.; Colditz, G.A. Unopposed Estrogen Therapy and the Risk of Invasive Breast Cancer. Arch. Intern. Med. 2006, 166, 1027–1032. [Google Scholar] [CrossRef]
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormone replacement therapy: Collaborative reanalysis of data from 51 epidemiological studies of 52 705 women with breast cancer and 108 411 women without breast cancer. Lancet 1997, 350, 1047–1059. [Google Scholar] [CrossRef]
- Ishtiaq, S.; Fogelman, I.; Hampson, G. Treatment of post-menopausal osteoporosis: Beyond bisphosphonates. J. Endocrinol. Investig. 2015, 38, 13–29. [Google Scholar] [CrossRef]
- Ewelina, B.; Fengfeng, C.; Marcus, V. Bone targeted therapies in advanced breast cancer. Swiss Med. Wkly. 2017. [Google Scholar] [CrossRef]
- Elomaa, I.; Blomqvist, C.; Gröhn, P.; Porkaa, L.; Kairento, A.L.; Selander, K.; Lamberg-Allardt, C.; Holmström, T. Long-term controlled trial with diphosphonate in patients with osteolytic bone metastases. Lancet 1983, 8317, 146–149. [Google Scholar] [CrossRef]
- Coleman, R. Bisphosphonates and breast cancer—From cautious palliation to saving lives. Bone 2020, 140, 115570. [Google Scholar] [CrossRef] [PubMed]
- Dhesy-Thind, S.; Fletcher, G.G.; Blanchette, P.S.; Clemons, M.J.; Dillmon, M.S.; Frank, E.S.; Gandhi, S.; Gupta, R.; Mates, M.; Moy, B.; et al. Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: A Cancer Care Ontario and American Society of Clinical Oncology Clinical Practice Guideline. JCO 2017, 35, 2062–2081. [Google Scholar] [CrossRef] [PubMed]
- Gnant, M. The evolving role of zoledronic acid in early breast cancer. OTT 2009, 95. [Google Scholar] [CrossRef] [PubMed]
- Gnant, M. Role of bisphosphonates in postmenopausal women with breast cancer. Cancer Treat. Rev. 2014, 40, 476–484. [Google Scholar] [CrossRef]
- Gnant, M. Zoledronic Acid in the Treatment of Early-Stage Breast Cancer: Is There a Final Verdict? Curr. Oncol. Rep. 2012, 14, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Gnant, M. Zoledronic acid in breast cancer: Latest findings and interpretations. Ther. Adv. Med. Oncol. 2011, 3, 293–301. [Google Scholar] [CrossRef]
- Gnant, M.; Mlineritsch, B.; Stoeger, H.; Luschin-Ebengreuth, G.; Heck, D.; Menzel, C.; Jakesz, R.; Seifert, M.; Hubalek, M.; Pristauz, G.; et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-month follow-up from the ABCSG-12 randomised trial. Lancet Oncol. 2011, 12, 631–641. [Google Scholar] [CrossRef]
- Gnant, M.; Mlineritsch, B.; Stoeger, H.; Luschin-Ebengreuth, G.; Knauer, M.; Moik, M.; Jakesz, R.; Seifert, M.; Taucher, S.; Bjelic-Radisic, V.; et al. Zoledronic acid combined with adjuvant endocrine therapy of tamoxifen versus anastrozol plus ovarian function suppression in premenopausal early breast cancer: Final analysis of the Austrian Breast and Colorectal Cancer Study Group Trial 12. Ann. Oncol. 2015, 26, 313–320. [Google Scholar] [CrossRef]
- Gnant, M.; Mlineritsch, B.; Luschin-Ebengreuth, G.; Kainberger, F.; Kässmann, H.; Piswanger-Sölkner, J.C.; Seifert, M.; Ploner, F.; Menzel, C.; Dubsky, P.; et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 5-year follow-up of the ABCSG-12 bone-mineral density substudy. Lancet Oncol. 2008, 9, 840–849. [Google Scholar] [CrossRef]
- Coleman, R.E.; Marshall, H.; Cameron, D.; Dodwell, D.; Burkinshaw, R.; Keane, M.; Gil, M.; Houston, S.J.; Grieve, R.J.; Barrett-Lee, P.J.; et al. Breast-Cancer Adjuvant Therapy with Zoledronic Acid. N. Engl. J. Med. 2011, 365, 1396–1405. [Google Scholar] [CrossRef]
- Coleman, R.; Cameron, D.; Dodwell, D.; Bell, R.; Wilson, C.; Rathbone, E.; Keane, M.; Gil, M.; Burkinshaw, R.; Grieve, R.; et al. Adjuvant zoledronic acid in patients with early breast cancer: Final efficacy analysis of the AZURE (BIG 01/04) randomised open-label phase 3 trial. Lancet Oncol. 2014, 15, 997–1006. [Google Scholar] [CrossRef]
- Coleman, R.E.; Collinson, M.; Gregory, W.; Marshall, H.; Bell, R.; Dodwell, D.; Keane, M.; Gil, M.; Barrett-Lee, P.; Ritchie, D.; et al. Benefits and risks of adjuvant treatment with zoledronic acid in stage II/III breast cancer. 10 years follow-up of the AZURE randomized clinical trial (BIG 01/04). J. Bone Oncol. 2018, 13, 123–135. [Google Scholar] [CrossRef]
- Suarez-Almazor, M.E.; Herrera, R.; Lei, X.; Chavez-MacGregor, M.; Zhao, H.; Giordano, S.H. Survival in older women with early stage breast cancer receiving low-dose bisphosphonates or denosumab. Cancer 2020, 126, 3929–3938. [Google Scholar] [CrossRef]
- van Hellemond, I.E.G.; Smorenburg, C.H.; Peer, P.G.M.; Swinkels, A.C.P.; Seynaeve, C.M.; van der Sangen, M.J.C.; Kroep, J.R.; de Graaf, H.; Honkoop, A.H.; Erdkamp, F.L.; et al. Breast cancer outcome in relation to bone mineral density and bisphosphonate use: A sub-study of the DATA trial. Breast Cancer Res. Treat. 2020, 180, 675–685. [Google Scholar] [CrossRef]
- Perrone, F.; De Laurentiis, M.; De Placido, S.; Orditura, M.; Cinieri, S.; Riccardi, F.; Ribecco, A.S.; Putzu, C.; Del Mastro, L.; Rossi, E.; et al. Adjuvant zoledronic acid and letrozole plus ovarian function suppression in premenopausal breast cancer: HOBOE phase 3 randomised trial. Eur. J. Cancer 2019, 118, 178–186. [Google Scholar] [CrossRef]
- Park, Y.-E.; Bava, U.; Lin, J.; Cornish, J.; Naot, D.; Reid, I.R. Bone-Bound Bisphosphonates Inhibit Proliferation of Breast Cancer Cells. Calcif. Tissue Int. 2019, 105, 497–505. [Google Scholar] [CrossRef]
- Buranrat, B.; Bootha, S. Antiproliferative and antimigratory activities of bisphosphonates in human breast cancer cell line MCF-7. Oncol. Lett. 2019. [Google Scholar] [CrossRef]
- Simpson, E.L.; Martyn-St James, M.; Hamilton, J.; Wong, R.; Gittoes, N.; Selby, P.; Davis, S. Clinical effectiveness of denosumab, raloxifene, romosozumab, and teriparatide for the prevention of osteoporotic fragility fractures: A systematic review and network meta-analysis. Bone 2020, 130, 115081. [Google Scholar] [CrossRef]
- Christodoulakos, G.E.; Lambrinoudaki, I.V.; Economou, E.V.; Papadias, C.; Vitoratos, N.; Panoulis, C.P.; Kouskouni, E.E.; Vlachou, S.A.; Creatsas, G.C. Circulating chemoattractants RANTES, negatively related to endogenous androgens, and MCP-1 are differentially suppressed by hormone therapy and raloxifene. Atherosclerosis 2007, 193, 142–150. [Google Scholar] [CrossRef]
- Sebastián-Ochoa, A.; Fernández-García, D.; Reyes-García, R.; Mezquita-Raya, P.; Rozas-Moreno, P.; Alonso-Garcia, G.; Muñoz-Torres, M. Adiponectin and leptin serum levels in osteoporotic postmenopausal women treated with raloxifene or alendronate. Menopause J. N. Am. Menopause Soc. 2012, 19, 172–177. [Google Scholar] [CrossRef]
- Lambrinoudaki, I.V.; Christodoulakos, G.E.; Economou, E.V.; Vlachou, S.A.; Panoulis, C.P.; Alexandrou, A.P.; Kouskouni, E.E.; Creatsas, G.C. Circulating leptin and ghrelin are differentially influenced by estrogen/progestin therapy and raloxifene. Maturitas 2008, 59, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Tommaselli, G.A.; Di Carlo, C.; Di Spiezio Sardo, A.; Bifulco, G.; Cirillo, D.; Guida, M.; Capasso, R.; Nappi, C. Serum leptin levels and body composition in postmenopausal women treated with tibolone and raloxifene. Menopause 2006, 13, 660–668. [Google Scholar] [CrossRef]
- Corson, S.L. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. J. Minim. Invasive Gynecol. 2006, 13, 492–493. [Google Scholar] [CrossRef]
- Garofalo, C. Increased Expression of Leptin and the Leptin Receptor as a Marker of Breast Cancer Progression: Possible Role of Obesity-Related Stimuli. Clin. Cancer Res. 2006, 12, 1447–1453. [Google Scholar] [CrossRef]
- Niu, J.; Jiang, L.; Guo, W.; Shao, L.; Liu, Y.; Wang, L. The Association between Leptin Level and Breast Cancer: A Meta-Analysis. PLoS ONE 2013, 8, e67349. [Google Scholar] [CrossRef]
- Haque, I.; Ghosh, A.; Acup, S.; Banerjee, S.; Dhar, K.; Ray, A.; Sarkar, S.; Kambhampati, S.; Banerjee, S.K. Leptin-induced ER-α-positive breast cancer cell viability and migration is mediated by suppressing CCN5-signaling via activating JAK/AKT/STAT-pathway. BMC Cancer 2018, 18, 99. [Google Scholar] [CrossRef]
- Wardell, S.E.; Nelson, E.R.; Chao, C.A.; McDonnell, D.P. Bazedoxifene Exhibits Antiestrogenic Activity in Animal Models of Tamoxifen-Resistant Breast Cancer: Implications for Treatment of Advanced Disease. Clin. Cancer Res. 2013, 19, 2420–2431. [Google Scholar] [CrossRef]
- Tian, J.; Chen, X.; Fu, S.; Zhang, R.; Pan, L.; Cao, Y.; Wu, X.; Xiao, H.; Lin, H.-J.; Lo, H.-W.; et al. Bazedoxifene is a novel IL-6/GP130 inhibitor for treating triple-negative breast cancer. Breast Cancer Res. Treat. 2019, 175, 553–566. [Google Scholar] [CrossRef]
- Fu, S.; Lin, J. Blocking Interleukin-6 and Interleukin-8 Signaling Inhibits Cell Viability, Colony-forming Activity, and Cell Migration in Human Triple-negative Breast Cancer and Pancreatic Cancer Cells. Anticancer Res. 2018, 38, 6271–6279. [Google Scholar] [CrossRef] [PubMed]
- Fanning, S.W.; Jeselsohn, R.; Dharmarajan, V.; Mayne, C.G.; Karimi, M.; Buchwalter, G.; Houtman, R.; Toy, W.; Fowler, C.E.; Han, R.; et al. The SERM/SERD bazedoxifene disrupts ESR1 helix 12 to overcome acquired hormone resistance in breast cancer cells. eLife 2018, 7, e37161. [Google Scholar] [CrossRef]
- Santen, R.J.; Song, Y.; Wang, J.; Yue, W. Preclinical breast effects of a tissue selective estrogen complex (TSEC) including conjugated estrogen with bazedoxifene. J. Steroid Biochem. Mol. Biol. 2017, 170, 61–64. [Google Scholar] [CrossRef]
- Fu, S.; Chen, X.; Lo, H.-W.; Lin, J. Combined bazedoxifene and paclitaxel treatments inhibit cell viability, cell migration, colony formation, and tumor growth and induce apoptosis in breast cancer. Cancer Lett. 2019, 448, 11–19. [Google Scholar] [CrossRef]
- Fabian, C.J.; Nye, L.; Powers, K.R.; Nydegger, J.L.; Kreutzjans, A.L.; Phillips, T.A.; Metheny, T.; Winblad, O.; Zalles, C.M.; Hagan, C.R.; et al. Effect of Bazedoxifene and Conjugated Estrogen (Duavee) on Breast Cancer Risk Biomarkers in High-Risk Women: A Pilot Study. Cancer Prev. Res. 2019, 12, 711–720. [Google Scholar] [CrossRef]
- Giannakeas, V.; Cadarette, S.M.; Ban, J.K.; Lipscombe, L.; Narod, S.A.; Kotsopoulos, J. Denosumab and breast cancer risk in postmenopausal women: A population-based cohort study. Br. J. Cancer 2018, 119, 1421–1427. [Google Scholar] [CrossRef]
- Coleman, R.; Finkelstein, D.M.; Barrios, C.; Martin, M.; Iwata, H.; Hegg, R.; Glaspy, J.; Periañez, A.M.; Tonkin, K.; Deleu, I.; et al. Adjuvant denosumab in early breast cancer (D-CARE): An international, multicentre, randomised, controlled, phase 3 trial. Lancet Oncol. 2020, 21, 60–72. [Google Scholar] [CrossRef]
- Crane, J.L.; Cao, X. Bone marrow mesenchymal stem cells and TGF-β signaling in bone remodeling. J. Clin. Investig. 2014, 124, 466–472. [Google Scholar] [CrossRef]
- Capriani, C.; Irani, D.; Bilezikian, J.P. Safety of osteoanabolic therapy: A decade of experience. J. Bone Miner. Res. 2012, 27, 2419–2428. [Google Scholar] [CrossRef]
- Gilsenan, A.; Harding, A.; Kellier-Steele, N.; Harris, D.; Midkiff, K.; Andrews, E. The Forteo Patient Registry linkage to multiple state cancer registries: Study design and results from the first 8 years. Osteoporos. Int. 2018, 29, 2335–2343. [Google Scholar] [CrossRef]
- Andrews, E.B.; Gilsenan, A.W.; Midkiff, K.; Sherrill, B.; Wu, Y.; Mann, B.H.; Masica, D. The US postmarketing surveillance study of adult osteosarcoma and teriparatide: Study design and findings from the first 7 years. J. Bone Miner. Res. 2012, 27, 2429–2437. [Google Scholar] [CrossRef]
- Swami, S.; Johnson, J.; Bettinson, L.A.; Kimura, T.; Zhu, H.; Albertelli, M.A.; Johnson, R.W.; Wu, J.Y. Prevention of breast cancer skeletal metastases with parathyroid hormone. JCI Insight 2017, 2, e90874. [Google Scholar] [CrossRef] [PubMed]
- Grill, V.; Hillary, J.; Ho, P.M.W.; Law, F.M.K.; Macisaac, R.J.; Maclsaac, I.A.; Moseley, J.M.; Martin, T.J. Parathyroid hormone-related protein: A possible endocrine function in lactation. Clin. Endocrinol. 1992, 37, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Rakopoulos, M.; Vargas, S.J.; Gillespie, M.T.; Ho, P.W.; Diefenbach-Jagger, H.; Leaver, D.D.; Grill, V.; Moseley, J.M.; Danks, J.A.; Martin, T.J. Production of parathyroid hormone-related protein by the rat mammary gland in pregnancy and lactation. Am. J. Physiol. 1993, 263, E1077–E1085. [Google Scholar]
- Martin, T.J.; Johnson, R.W. Multiple actions of parathyroid hormone-related protein in breast cancer bone metastasis. Br. J. Pharm. 2019, bph.14709. [Google Scholar] [CrossRef]
- Kovacs, C.S.; Lanske, B.; Hunzelman, J.L.; Guo, J.; Karaplis, A.C.; Kronenberg, H.M. Parathyroid hormone-related peptide (PTHrP) regulates fetal-placental calcium transport through a receptor distinct from the PTH/PTHrP receptor. Proc. Natl. Acad. Sci. USA 1996, 93, 15233–15238. [Google Scholar] [CrossRef]
- Thomas, R.J.; Guise, T.A.; Yin, J.J.; Elliott, J.; Horwood, N.J.; Martin, T.J.; Gillespie, M.T. Breast Cancer Cells Interact with Osteoblasts to Support Osteoclast Formation. Endocrinology 1999, 140, 4451–4458. [Google Scholar] [CrossRef]
- Sleeman, A.; Clements, J.N. Abaloparatide: A new pharmacological option for osteoporosis. Am. J. Health Syst. Pharm. 2019, 76, 130–135. [Google Scholar] [CrossRef]
- Boyce, E.G.; Mai, Y.; Pham, C. Abaloparatide: Review of a next-generation parathyroid hormone agonist. Ann. Pharm. 2018, 52, 462–472. [Google Scholar] [CrossRef]
- Tella, S.H.; Kommalapati, A.; Correa, R. Profile of abaloparatide and its potential in the treatment of postmenopausal osteoporosis. Cureus 2017, 9, e1300. [Google Scholar] [CrossRef]
- Gkotzamanidou, M.; Dimopoulos, M.A.; Kastritis, E.; Christoulas, D.; Moulopoulos, L.A.; Terpos, E. Sclerostin: A possible target for the management of cancer-induced bone disease. Expert Opin. Ther. Targets 2012, 16, 761–769. [Google Scholar] [CrossRef]
- Hesse, E.; Schröder, S.; Brandt, D.; Pamperin, J.; Saito, H.; Taipaleenmäki, H. Sclerostin inhibition alleviates breast cancer–induced bone metastases and muscle weakness. JCI Insight 2019, 4, e125543. [Google Scholar] [CrossRef] [PubMed]
- Lewiecki, E.M.; Blicharski, T.; Goemaere, S.; Lippuner, K.; Meisner, P.D.; Miller, P.D.; Miyauchi, A.; Maddox, J.; Chen, L.; Horlait, S. A Phase III Randomized Placebo-Controlled Trial to Evaluate Efficacy and Safety of Romosozumab in Men With Osteoporosis. J. Clin. Endocrinol. Metab. 2018, 103, 3183–3193. [Google Scholar] [CrossRef] [PubMed]
- Langdahl, B.L.; Libanati, C.; Crittenden, D.B.; Bolognese, M.A.; Brown, J.P.; Daizadeh, N.S.; Dokoupilova, E.; Engelke, K.; Finkelstein, J.S.; Genant, H.K.; et al. Romosozumab (sclerostin monoclonal antibody) versus teriparatide in postmenopausal women with osteoporosis transitioning from oral bisphosphonate therapy: A randomised, open-label, phase 3 trial. Lancet 2017, 390, 1585–1594. [Google Scholar] [CrossRef]
- Cosman, F.; Crittenden, D.B.; Adachi, J.D.; Binkley, N.; Czerwinski, E.; Ferrari, S.; Hofbauer, L.C.; Lau, E.; Lewiecki, E.M.; Miyauchi, A.; et al. Romosozumab Treatment in Postmenopausal Women with Osteoporosis. N. Engl. J. Med. 2016, 375, 1532–1543. [Google Scholar] [CrossRef]
- Morales-Santana, S.; Garcia-Fontana, B.; Garcia-Martin, A.; Rozas-Moreno, P.; Garcia-Salcedo, J.A.; Reyes-Garcia, R.; Munoz-Torres, M. Atherosclerotic Disease in Type 2 Diabetes Is Associated With an Increase in Sclerostin Levels. Diabetes Care 2013, 36, 1667–1674. [Google Scholar] [CrossRef]
- Hampson, G.; Edwards, S.; Conroy, S.; Blake, G.M.; Fogelman, I.; Frost, M.L. The relationship between inhibitors of the Wnt signalling pathway (Dickkopf-1(DKK1) and sclerostin), bone mineral density, vascular calcification and arterial stiffness in post-menopausal women. Bone 2013, 56, 42–47. [Google Scholar] [CrossRef]
- Claes, K.J.; Viaene, L.; Heye, S.; Meijers, B.; d’Haese, P.; Evenepoel, P. Sclerostin: Another Vascular Calcification Inhibitor? J. Clin. Endocrinol. Metab. 2013, 98, 3221–3228. [Google Scholar] [CrossRef]
- Cooke, M.M.; McCarthy, G.M.; Sallis, J.D.; Morgan, M.P. Phosphocitrate Inhibits Calcium Hydroxyapatite Induced Mitogenesis and Upregulation of Matrix Metalloproteinase-1, Interleukin-1β and Cyclooxygenase-2 mRNA in Human Breast Cancer Cell Lines. Breast Cancer Res. Treat. 2003, 79, 253–263. [Google Scholar] [CrossRef]
- Lin, C.; Liao, W.; Jian, Y.; Peng, Y.; Zhang, X.; Ye, L.; Cui, Y.; Wang, B.; Wu, X.; Xiong, Z.; et al. CGI-99 promotes breast cancer metastasis via autocrine interleukin-6 signaling. Oncogene 2017, 36, 3695–3705. [Google Scholar] [CrossRef]
- Burmester, G.R.; Lin, Y.; Patel, R.; van Adelsberg, J.; Mangan, E.K.; Graham, N.M.H.; van Hoogstraten, H.; Bauer, D.; Ignacio Vargas, J.; Lee, E.B. Efficacy and safety of sarilumab monotherapy versus adalimumab monotherapy for the treatment of patients with active rheumatoid arthritis (MONARCH): A randomised, double-blind, parallel-group phase III trial. Ann. Rheum. Dis. 2017, 76, 840–847. [Google Scholar] [CrossRef]
- Huizinga, T.W.J.; Fleischmann, R.M.; Jasson, M.; Radin, A.R.; van Adelsberg, J.; Fiore, S.; Huang, X.; Yancopoulos, G.D.; Stahl, N.; Genovese, M.C. Sarilumab, a fully human monoclonal antibody against IL-6Rα in patients with rheumatoid arthritis and an inadequate response to methotrexate: Efficacy and safety results from the randomised SARIL-RA-MOBILITY Part A trial. Ann. Rheum. Dis. 2014, 73, 1626–1634. [Google Scholar] [CrossRef] [PubMed]
- Emery, P.; Rondon, J.; Parrino, J.; Lin, Y.; Pena-Rossi, C.; van Hoogstraten, H.; Graham, N.M.H.; Liu, N.; Paccaly, A.; Wu, R.; et al. Safety and tolerability of subcutaneous sarilumab and intravenous tocilizumab in patients with rheumatoid arthritis. Rheumatology 2019, 58, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Mitoma, H.; Horiuchi, T.; Tsukamoto, H.; Ueda, N. Molecular mechanisms of action of anti-TNF-α agents—Comparison among therapeutic TNF-α antagonists. Cytokine 2018, 101, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Madhusudan, S. A Phase II Study of Etanercept (Enbrel), a Tumor Necrosis Factor Inhibitor in Patients with Metastatic Breast Cancer. Clin. Cancer Res. 2004, 10, 6528–6534. [Google Scholar] [CrossRef]
- Raaschou, P.; Frisell, T.; Askling, J. TNF inhibitor therapy and risk of breast cancer recurrence in patients with rheumatoid arthritis: A nationwide cohort study. Ann. Rheum. Dis. 2015, 74, 2137–2143. [Google Scholar] [CrossRef]
- Mamtani, R.; Clark, A.S.; Scott, F.I.; Brensinger, C.M.; Boursi, B.; Chen, L.; Xie, F.; Yun, H.; Osterman, M.T.; Curtis, J.R.; et al. Association Between Breast Cancer Recurrence and Immunosuppression in Rheumatoid Arthritis and Inflammatory Bowel Disease: A Cohort Study: BREAST CANCER RECURRENCE WITH IMMUNOSUPPRESSION IN RA AND IBD. Arthritis Rheumatol. 2016, 68, 2403–2411. [Google Scholar] [CrossRef]
- Chiesa Fuxench, Z.C.; Shin, D.B.; Ogdie Beatty, A.; Gelfand, J.M. The Risk of Cancer in Patients With Psoriasis: A Population-Based Cohort Study in the Health Improvement Network. JAMA Derm. 2016, 152, 282. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, J.; Yang, Y.; Huang, Y.; Liu, G. Malignancy risk of anti-tumor necrosis factor alpha blockers: An overview of systematic reviews and meta-analyses. Clin. Rheumatol. 2016, 35, 1–18. [Google Scholar] [CrossRef]
- Chen, Y.; Friedman, M.; Liu, G.; Deodhar, A.; Chu, C.-Q. Do tumor necrosis factor inhibitors increase cancer risk in patients with chronic immune-mediated inflammatory disorders? Cytokine 2018, 101, 78–88. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, W.; Chen, H.; Yu, M.-X. Risk of Breast Cancer and Total Malignancies in Rheumatoid Arthritis Patients Undergoing TNF-α Antagonist Therapy: A Meta-analysis of Randomized Control Trials. Asian Pac. J. Cancer Prev. 2014, 15, 3403–3410. [Google Scholar] [CrossRef][Green Version]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007, 449, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Liu, J.; Bai, L.; Tian, J.; Zhu, J. Interleukin-6 induces malignant transformation of rat mesenchymal stem cells in association with enhanced signaling of signal transducer and activator of transcription 3. Cancer Sci. 2014, 105, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Mandal, G.; Roy Chowdhury, S.; Purohit, S.; Payne, K.K.; Anadon, C.; Gupta, A.; Swanson, P.; Yu, X.; Conejo-Garcia, J.R.; et al. Exosomes Produced by Mesenchymal Stem Cells Drive Differentiation of Myeloid Cells into Immunosuppressive M2-Polarized Macrophages in Breast Cancer. J. Immunol. 2019, 203, 3447–3460. [Google Scholar] [CrossRef] [PubMed]
- Ijiri, K.; Nagayoshi, R.; Matsushita, N.; Tsuruga, H.; Taniguchi, N.; Gushi, A.; Sakakima, H.; Komiya, S.; Matsuyama, T. Differential Expression Patterns of Secreted Frizzled Related Protein Genes in Synovial Cells from Patients with Arthritis. J. Rheumatol. 2002, 29, 2266–2270. [Google Scholar]
- Imai, K.; Morikawa, M.; D’Armiento, J.; Matsumoto, H.; Komiya, K.; Okada, Y. Differential expression of WNTs and FRPs in the synovium of rheumatoid arthritis and osteoarthritis. Biochem. Biophys. Res. Commun. 2006, 345, 1615–1620. [Google Scholar] [CrossRef]

| PI | ARLI | Carcinogenesis | OP | RA | OA | PD | |
|---|---|---|---|---|---|---|---|
| References | [72,126,129,130,131] | [90,151] | [44,151,159,160,161,162,164,165,166,167,273,274,275] | [52,53,54,55,56,57,58,70] | [30,81,82,83,84,85,86,87,88] | [56,57,58,75,76,77,276,277] | [91,92,93,94,95,96,97,99,100] |
| Cytokines | |||||||
| IL-1 | + | + | + | + | + | ||
| IL-6 | + | + | + | + | + | + | + |
| IL-8 | + | + | |||||
| IL-17 | + | + | |||||
| TNF-α | + | + | + | + | + | + | |
| Leptin | + | + | + | + | + | + | + |
| RANKL | + | + | + | + | |||
| IL-10 | - | - | |||||
| TGF-ß | - | ||||||
| Others secreted protein | |||||||
| SFRP1 | + | + | + | + | + | ||
| Myeloid lineage | |||||||
| T-cells | + | + | + | + | + | + | + |
| Treg | - | - | |||||
| B-cells | + | ||||||
| Macrophages | + | + | + | + | + | + | |
| Osteoclasts/OCL | + | + | + | + | + | ||
| Bone resorption makers | |||||||
| SPP1 | + | + | + | + | |||
| OPN | + | + | + | ||||
| Mesenchymal lineage | |||||||
| MSCs | + | + | + | ||||
| Osteoblasts/OBL | + | + | + | + | |||
| Bone formation markers | |||||||
| ALP | + | ||||||
| OC | + | - | - | + | + or = | ||
| Osteoblastogenesis markers | |||||||
| RUNX2 | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clemenceau, A.; Michou, L.; Diorio, C.; Durocher, F. Breast Cancer and Microcalcifications: An Osteoimmunological Disorder? Int. J. Mol. Sci. 2020, 21, 8613. https://doi.org/10.3390/ijms21228613
Clemenceau A, Michou L, Diorio C, Durocher F. Breast Cancer and Microcalcifications: An Osteoimmunological Disorder? International Journal of Molecular Sciences. 2020; 21(22):8613. https://doi.org/10.3390/ijms21228613
Chicago/Turabian StyleClemenceau, Alisson, Laetitia Michou, Caroline Diorio, and Francine Durocher. 2020. "Breast Cancer and Microcalcifications: An Osteoimmunological Disorder?" International Journal of Molecular Sciences 21, no. 22: 8613. https://doi.org/10.3390/ijms21228613
APA StyleClemenceau, A., Michou, L., Diorio, C., & Durocher, F. (2020). Breast Cancer and Microcalcifications: An Osteoimmunological Disorder? International Journal of Molecular Sciences, 21(22), 8613. https://doi.org/10.3390/ijms21228613





