Evaluation of Sterilization/Disinfection Methods of Fibrous Polyurethane Scaffolds Designed for Tissue Engineering Applications
Abstract
:1. Introduction
2. Results
2.1. Evaluation of Sterilization Efficiency
2.1.1. Visual Inspection
2.1.2. Contact with Medium
2.1.3. Agar Diffusion Method
2.2. Material Characterization
2.2.1. Surface Morphology
2.2.2. Chemical Analysis
2.2.3. Mechanical Tests Results
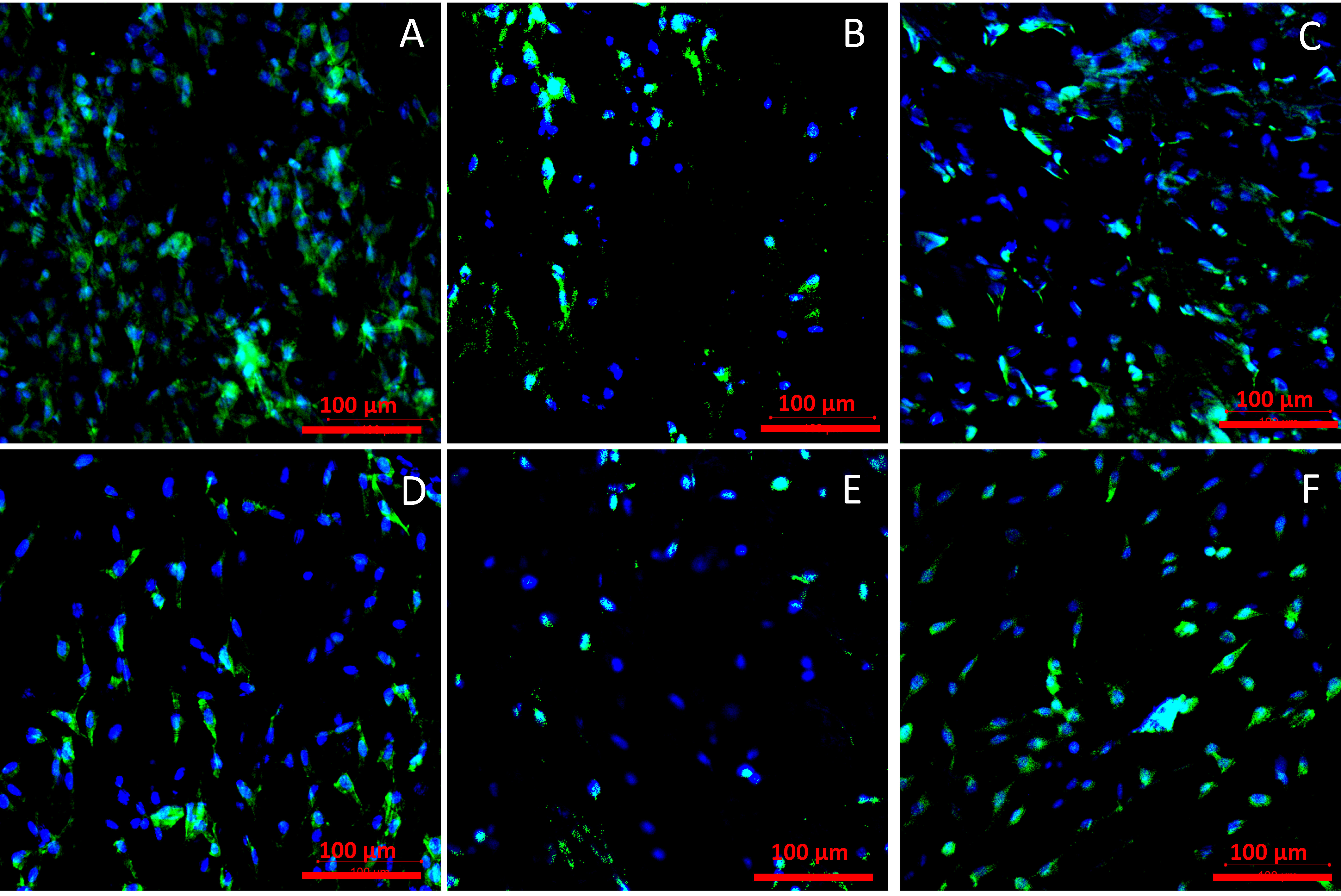
2.3. Cell Viability
2.3.1. Cell Viability
2.3.2. Cell Adhesion
3. Discussion
4. Materials and Methods
4.1. Materials Fabrication
4.2. Sterilization Techniques
4.2.1. Antimicrobial Solution (AMS)
4.2.2. Ethanol Solution
4.2.3. UV Irradiation
4.2.4. Gamma Irradiation
4.2.5. E-Beam Irradiation
4.3. Evaluation of Sterilization Efficiency
4.3.1. Contact with Cell Culture Medium
4.3.2. Agar Diffusion
4.4. Materials Characterization
4.4.1. Surface Morphology
4.4.2. Chemical Analysis
4.4.3. Mechanical Properties
- Fmax—maximum force acting on the sample [N]
- A—cross-section area [m2]
- —sample porosity [-]
- L—sample deformation [m]
4.4.4. Materials’ Porosity
- ρPU—ChronoFlex®C75D density [g/cm3]
- ρsample—sample density [g/cm3] [51]
- m—sample weight [g]
- A—sample area [g/cm3]
- δ—sample thickness [cm]
4.5. Cell Viability
4.5.1. Cell Culture Preparation
4.5.2. Cell Viability
- V—cells viability [%]
- FIsample—sample’s fluorescent intensity [-]
- FIcontrol24h—control’s fluorescent intensity after 24 h of culture [-]
4.5.3. Cell Adhesion
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ma, P.X. Scaffolds for tissue fabrication. Mater. Today 2004, 7, 30–40. [Google Scholar] [CrossRef]
- Rediguieri, C.F.; Sassonia, R.C.; Dua, K.; Kikuchi, I.S.; de Jesus Andreoli Pinto, T. Impact of sterilization methods on electrospun scaffolds for tissue engineering. Eur. Polym. J. 2016, 82, 181–195. [Google Scholar] [CrossRef]
- Dai, Z.; Ronholm, J.; Tian, Y.; Sethi, B.; Cao, X. Sterilization techniques for biodegradable scaffolds in tissue engineering applications. J. Tissue Eng. 2016, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silindir, M.; Özer, A.Y. Sterilization methods and the comparison of E-beam sterilization with gamma radiation sterilization. Fabad J. Pharm. Sci. 2009, 34, 43–53. [Google Scholar]
- Hsiao, C.Y.; Liu, S.J.; Wen-Neng Ueng, S.; Chan, E.C. The influence of γ irradiation and ethylene oxide treatment on the release characteristics of biodegradable poly(lactide-co-glycolide) composites. Polym. Degrad. Stab. 2012, 97, 715–720. [Google Scholar] [CrossRef]
- Rutala, W.A.; Gergen, M.F.; Weber, D.J. Comparative evaluation of the sporicidal activity of new low-temperature sterilization technologies: Ethylene oxide, 2 plasma sterilization systems, and liquid peracetic acid. Am. J. Infect. Control 1998, 26, 393–398. [Google Scholar] [CrossRef]
- Mendes, G.C.C.; Brandão, T.R.S.; Silva, C.L.M. Ethylene oxide sterilization of medical devices: A review. Am. J. Infect. Control 2007, 35, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomaterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- Marreco, P.R.; Da Luz Moreira, P.; Genari, S.C.; Moraes, Â.M. Effects of different sterilization methods on the morphology, mechanical properties, and cytotoxicity of chitosan membranes used as wound dressings. J. Biomed. Mater. Res. Part B Appl. Biomater. 2004, 71, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Gualandi, C.; Govoni, M.; Foroni, L.; Valente, S.; Bianchi, M.; Giordano, E.; Pasquinelli, G.; Biscarini, F.; Focarete, M.L. Ethanol disinfection affects physical properties and cell response of electrospun poly(l-lactic acid) scaffolds. Eur. Polym. J. 2012, 48, 2008–2018. [Google Scholar] [CrossRef]
- Selim, M.; Bullock, A.J.; Blackwood, K.A.; Chapple, C.R.; MacNeil, S. Developing biodegradable scaffolds for tissue engineering of the urethra. BJU Int. 2011, 107, 296–302. [Google Scholar] [CrossRef]
- Braghirolli, D.I.; Steffens, D.; Quintiliano, K.; Acasigua, G.A.X.; Gamba, D.; Fleck, R.A.; Petzhold, C.L.; Pranke, P. The effect of sterilization methods on electronspun poly(lactide-co-glycolide) and subsequent adhesion efficiency of mesenchymal stem cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 102, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Yunoki, S.; Ikoma, T.; Monkawa, A.; Ohta, K.; Tanaka, J.; Sotome, S.; Shinomiya, K. Influence of γ irradiation on the mechanical strength and in vitro biodegradation of porous hydroxyapatite/collagen composite. J. Am. Ceram. Soc. 2006, 89, 2977–2979. [Google Scholar] [CrossRef]
- Cottam, E.; Hukins, D.W.L.; Lee, K.; Hewitt, C.; Jenkins, M.J. Effect of sterilisation by gamma irradiation on the ability of polycaprolactone (PCL) to act as a scaffold material. Med. Eng. Phys. 2009, 31, 221–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischbach, C.; Tessmar, J.; Lucke, A.; Schnell, E.; Schmeer, G.; Blunk, T.; Göpferich, A. Does UV irradiation affect polymer properties relevant to tissue engineering? Surf. Sci. 2001, 491, 333–345. [Google Scholar] [CrossRef]
- Leeming, J.P.; Lovering, A.M.; Hunt, C.J. Residual antibiotics in allograft heart valve tissue samples following antibiotic disinfection. J. Hosp. Infect. 2005, 60, 231–234. [Google Scholar] [CrossRef]
- Jashari, R.; Tabaku, M.; Van Hoeck, B.; Cochéz, C.; Callant, M.; Vanderkelen, A. Decontamination of heart valve and arterial allografts in the European Homograft Bank (EHB): Comparison of two different antibiotic cocktails in low temperature conditions. Cell Tissue Bank. 2007, 8, 247–255. [Google Scholar] [CrossRef]
- Daristotle, J.L.; Behrens, A.M.; Sandler, A.D.; Kofinas, P. A Review of the Fundamental Principles and Applications of Solution Blow Spinning. ACS Appl. Mater. Interfaces 2016, 8, 34951–34963. [Google Scholar] [CrossRef] [Green Version]
- Medeiros, E.S.; Glenn, G.M.; Klamczynski, A.P.; Orts, W.J.; Mattoso, L.H.C. Solution blow spinning: A new method to produce micro- and nanofibers from polymer solutions. J. Appl. Polym. Sci. 2009, 113, 2322–2330. [Google Scholar] [CrossRef]
- Zhang, Y.; Chwee, T.L.; Ramakrishna, S.; Huang, Z.M. Recent development of polymer nanofibers for biomedical and biotechnological applications. J. Mater. Sci. Mater. Med. 2005, 16, 933–946. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Bowlin, G.L.; Mansfield, K.; Layman, J.; Simpson, D.G.; Sanders, E.H.; Wnek, G.E. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Control. Release 2002, 81, 57–64. [Google Scholar] [CrossRef]
- Pashneh-Tala, S.; MacNeil, S.; Claeyssens, F. The tissue-engineered vascular graft-Past, present, and future. Tissue Eng. Part B Rev. 2016, 22, 68–100. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, H.; Shin, Y.M.; Terai, H.; Vacanti, J.P. A biodegradable nanofiber scaffold by electrospinning and its potential for bone tissue engineering. Biomaterials 2003, 24, 2077–2082. [Google Scholar] [CrossRef]
- Yang, F.; Murugan, R.; Ramakrishna, S.; Wang, X.; Ma, Y.-X.; Wang, S. Fabrication of nano-structured porous PLLA scaffold intended for nerve tissue engineering. Biomaterials 2004, 25, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Danielson, K.G.; Alexander, P.G.; Tuan, R.S. Biological response of chondrocytes cultured in three-dimensional nanofibrous poly(ϵ-caprolactone) scaffolds. J. Biomed. Mater. Res. Part A 2003, 67A, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Reis, T.C.; Castleberry, S.; Rego, A.M.B.; Aguiar-Ricardo, A.; Hammond, P.T. Three-dimensional multilayered fibrous constructs for wound healing applications. Biomater. Sci. 2016, 4, 319. [Google Scholar] [CrossRef] [PubMed]
- Asefnejad, A.; Khorasani, M.T.; Behnamghader, A.; Farsadzadeh, B.; Bonakdar, S. Manufacturing of biodegradable polyurethane scaffolds based on polycaprolactone using a phase separation method: Physical properties and in vitro assay. Int. J. Nanomed. 2011, 6, 2375–2384. [Google Scholar] [CrossRef] [Green Version]
- Tcharkhtchi, A.; Farzaneh, S.; Abdallah-Elhirtsi, S.; Esmaeillou, B.; Nony, F.; Baron, A. Thermal Aging Effect on Mechanical Properties of Polyurethane. Int. J. Polym. Anal. Charact. 2017, 19. [Google Scholar] [CrossRef] [Green Version]
- Kurimoto, Y.; Takeda, M.; Koizumi, A.; Yamauchi, S.; Doi, S.; Tamura, Y. Mechanical properties of polyurethane films prepared from liquefied wood with polymeric MDI. Bioresour. Technol. 2000, 74, 151–157. [Google Scholar] [CrossRef]
- Doss, J.R.; Shanahan, M.H.; Wohl, C.J.; Connell, J.W. Synthesis, characterization and evaluation of urethane co-oligomers containing pendant fluoroalkyl ether groups. Prog. Org. Coat. 2016, 95, 72–78. [Google Scholar] [CrossRef]
- Luzzatto, L.; Apirion, D.; Schlessinger, D. Mechanism of action of streptomycin in E. coli: Interruption of the ribosome cycle at the initiation of protein synthesis. Proc. Natl. Acad. Sci. USA 1968, 60, 873–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yocum, R.R.; Waxman, D.J.; Rasmussen, J.R.; Strominger, J.L. Mechanism of penicillin action: Penicillin and substrate bind covalently to the same active site serine in two bacterial D-alanine carboxypeptidases. Proc. Natl. Acad. Sci. USA 1979, 76, 2730–2734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noor, A.; Preuss, C.V. Antifungal Membrane Function Inhibitors (Amphotericin B); StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Düzyer, S.; Koral Koç, S.; Hockenberger, A.; Evke, E.; Kahveci, Z.; Uǧuz, A. Effects of different sterilization methods on polyester surfaces. Tekstil ve Konfeksiyon 2013, 23, 319–324. [Google Scholar]
- Dai, T.; Vrahas, M.S.; Murray, C.K.; Hamblin, M.R. Ultraviolet C irradiation: An alternative antimicrobial approach to localized infections? Expert Rev. Anti. Infect. Ther. 2012, 10, 185–195. [Google Scholar] [CrossRef] [Green Version]
- Holy, C.E.; Cheng, C.; Davies, J.E.; Shoichet, M.S. Optimizing the sterilization of PLGA scaffolds for use in tissue engineering. Biomaterials 2000, 22, 25–31. [Google Scholar] [CrossRef]
- Mcdonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef] [Green Version]
- Navone, S.E.; Pascucci, L.; Dossena, M.; Ferri, A.; Invernici, G.; Acerbi, F.; Cristini, S.; Bedini, G.; Tosetti, V.; Ceserani, V.; et al. Decellularized silk fibroin scaffold primed with adipose mesenchymal stromal cells improves wound healing in diabetic mice. Stem Cell Res. Ther. 2014, 5, 7. [Google Scholar] [CrossRef] [Green Version]
- Beringer, L.T.; Xu, X.; Shih, W.; Shih, W.H.; Habas, R.; Schauer, C.L. An electrospun PVDF-TrFe fiber sensor platform for biological applications. Sens. Actuators A Phys. 2015, 222, 293–300. [Google Scholar] [CrossRef]
- He, S.; Li, X.; Yang, Y.; Jia, G.; Zou, J. Correlations of In Vitro in In vivo Degradation Tests on Electrospun Poly-DL-Lactide-Poly(ethylene glycol) Fibers. J. Appl. Polym. Sci. 2012, 116, 2207–2215. [Google Scholar] [CrossRef]
- Bye, F.J.; Wang, L.; Bullock, A.J.; Blackwood, K.A.; Ryan, A.J.; Macneil, S. Postproduction processing of electrospun fibres for tissue engineering. J. Vis. Exp. 2012. [Google Scholar] [CrossRef] [Green Version]
- Gorna, K.; Gogolewski, S. The effect of gamma radiation on molecular stability and mechanical properties of biodegradable polyurethanes for medical applications. Polym. Degrad. Stab. 2003, 79, 465–474. [Google Scholar] [CrossRef]
- Bosworth, L.A.; Gibb, A.; Downes, S. Gamma Irradiation of Electrospun Poly (e-caprolactone) Fibers Affects Material Properties but Not Cell Response. J. Polym. Sci. Part B Polym. Phys. 2012, 50, 870–876. [Google Scholar] [CrossRef]
- Sefat, F.; Mckean, R.; Deshpande, P.; Ramachandran, C.; Hill, C.J.; Sangwan, V.S.; Ryan, A.J.; Macneil, S. Production, sterilisation and storage of biodegradable electrospun PLGA membranes for delivery of limbal stem cells to the cornea. Procedia Eng. 2013, 59, 101–116. [Google Scholar] [CrossRef]
- Augustine, R.; Saha, A.; Jayachandran, V.P.; Thomas, S. Dose-dependent effects of gamma irradiation on the materials properties and cell proliferation of electrospun polycaprolactone tissue engineering scaffolds. Int. J. Polym. Mater. Polym. Biomater. 2015, 64, 526–533. [Google Scholar] [CrossRef] [Green Version]
- Da Silva Aquino, K.A. Sterilization by Gamma Irradiation. In Gamma Radiation; Adrovic, F., Ed.; InTech: London, UK, 2012. [Google Scholar]
- De Cassan, D.; Lena, A.; Glasmacher, B.; Menzel, H. Impact of sterilization by electron beam, gamma radiation and X-rays on electrospun poly-(ε-caprolactone) fi ber mats. J. Mater. Sci. Mater. Med. 2019, 30, 42. [Google Scholar] [CrossRef]
- Zhang, X.; Kotaki, M.; Okubayashi, S.; Sukigara, S. Acta Biomaterialia Effect of electron beam irradiation on the structure and properties of electrospun PLLA and PLLA/PDLA blend nanofibers. Acta Biomater. 2010, 6, 123–129. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2019, 9. [Google Scholar] [CrossRef] [Green Version]
- ASTM International. ASTM D638-10, Standard Test Method for Tensile Properties of Plastics; ASTM International: West Conshohocken, PA, USA, 2010; Volume 8, pp. 1–16. [Google Scholar] [CrossRef]
- MatWeb: Online Materials Information Resource. Available online: http://www.matweb.com/search/datasheet.aspx?matguid=7c9858f90b2843ff8894d7a18ca40415&n=1&ckck=1 (accessed on 26 October 2020).







| Unsterile | AMS 1 h | AMS 24 h | UV Irradiation | EtOH | Gamma Irradiation | E-Beam Irradiation | |
|---|---|---|---|---|---|---|---|
| Material morphology (visual inspection) | − | − | − | − | − | + * | − |
| Contact with cell culture medium | − | − | − | − | − | − | − |
| Contact with Agar | + | − | − | + | + | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łopianiak, I.; Butruk-Raszeja, B.A. Evaluation of Sterilization/Disinfection Methods of Fibrous Polyurethane Scaffolds Designed for Tissue Engineering Applications. Int. J. Mol. Sci. 2020, 21, 8092. https://doi.org/10.3390/ijms21218092
Łopianiak I, Butruk-Raszeja BA. Evaluation of Sterilization/Disinfection Methods of Fibrous Polyurethane Scaffolds Designed for Tissue Engineering Applications. International Journal of Molecular Sciences. 2020; 21(21):8092. https://doi.org/10.3390/ijms21218092
Chicago/Turabian StyleŁopianiak, Iwona, and Beata A. Butruk-Raszeja. 2020. "Evaluation of Sterilization/Disinfection Methods of Fibrous Polyurethane Scaffolds Designed for Tissue Engineering Applications" International Journal of Molecular Sciences 21, no. 21: 8092. https://doi.org/10.3390/ijms21218092







