Homo-Tris-Nitrones Derived from α-Phenyl-N-tert-butylnitrone: Synthesis, Neuroprotection and Antioxidant Properties
Abstract
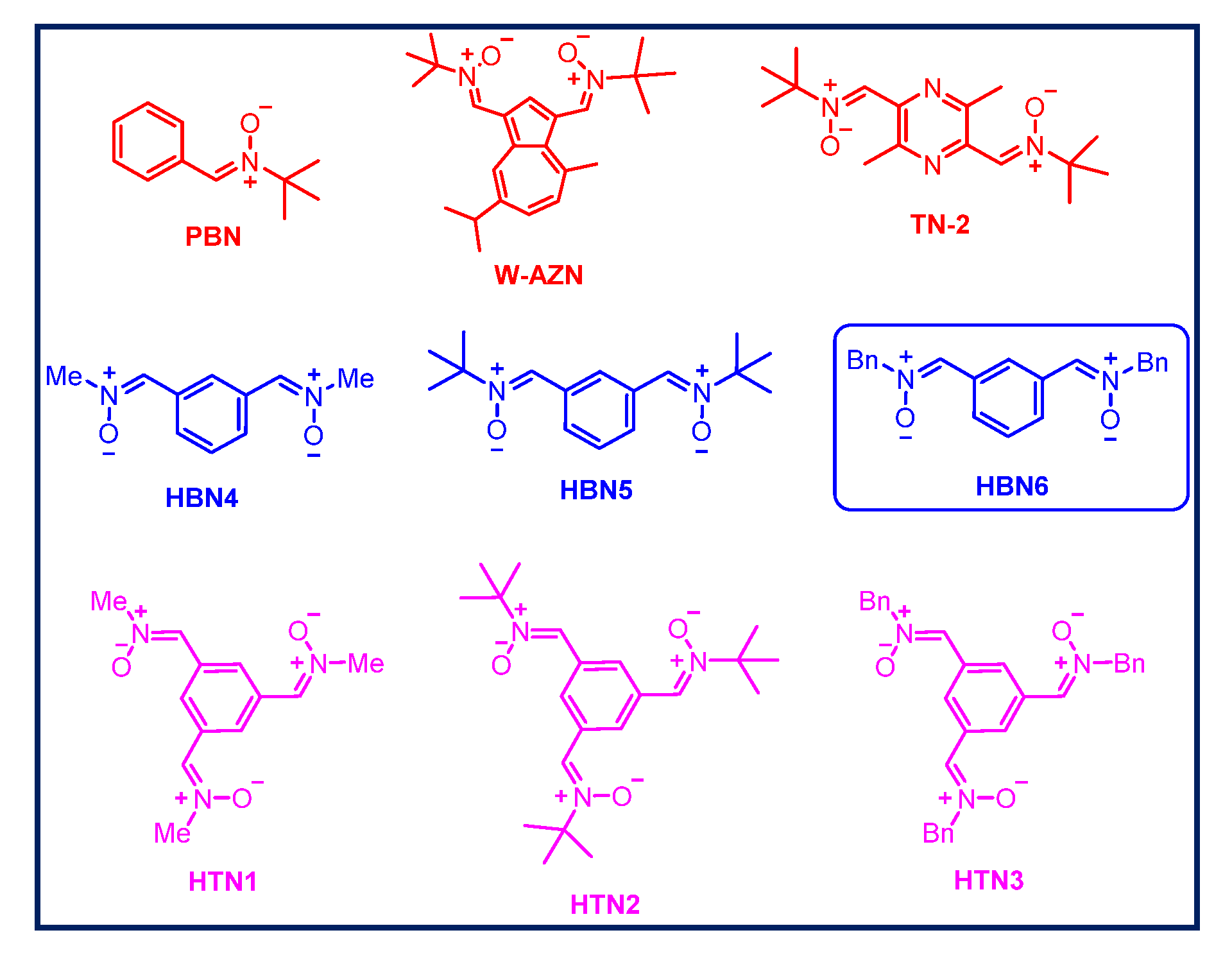
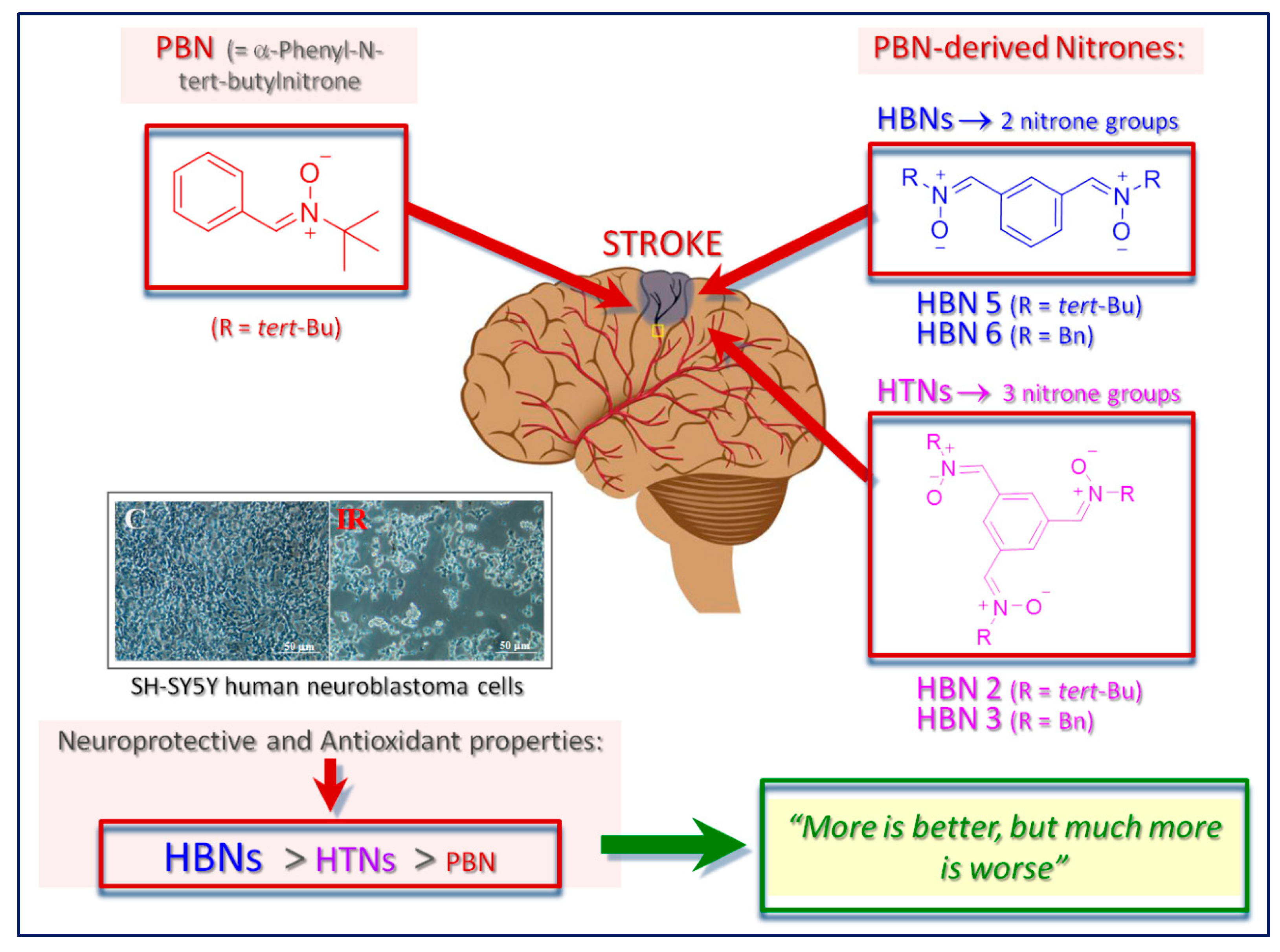
1. Introduction
2. Results and Discussion
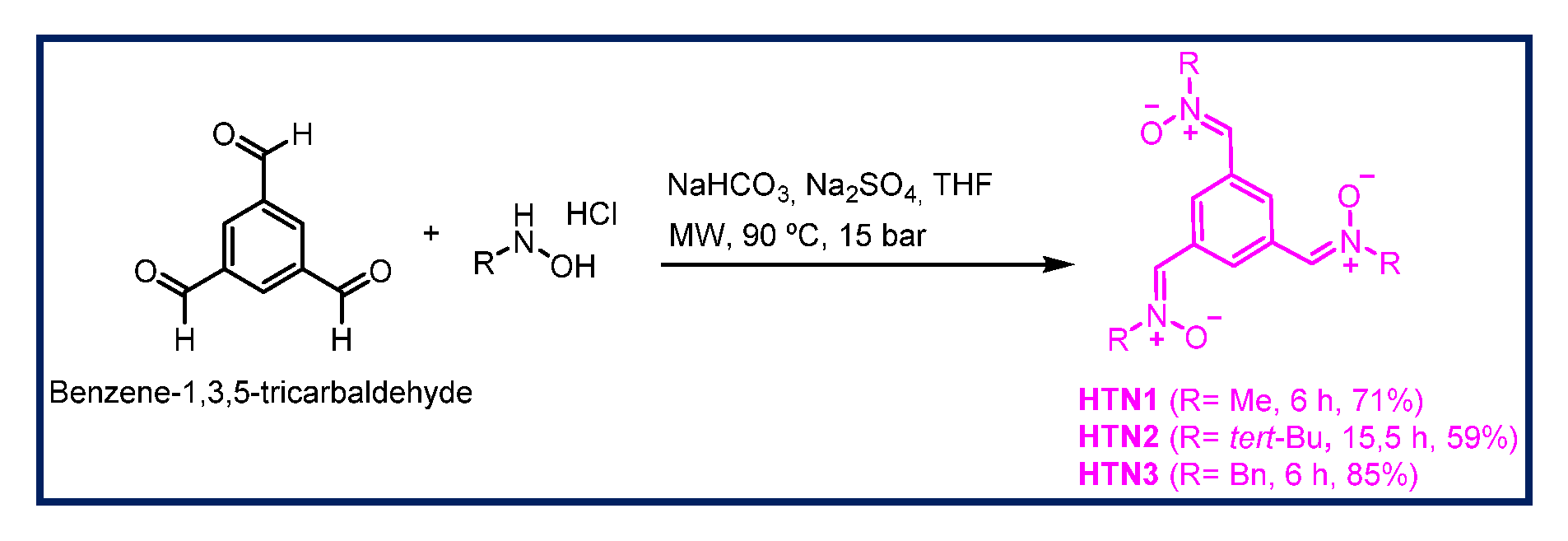
2.1. Chemistry
2.2. Neuroprotection Studies of HTNs 1-3
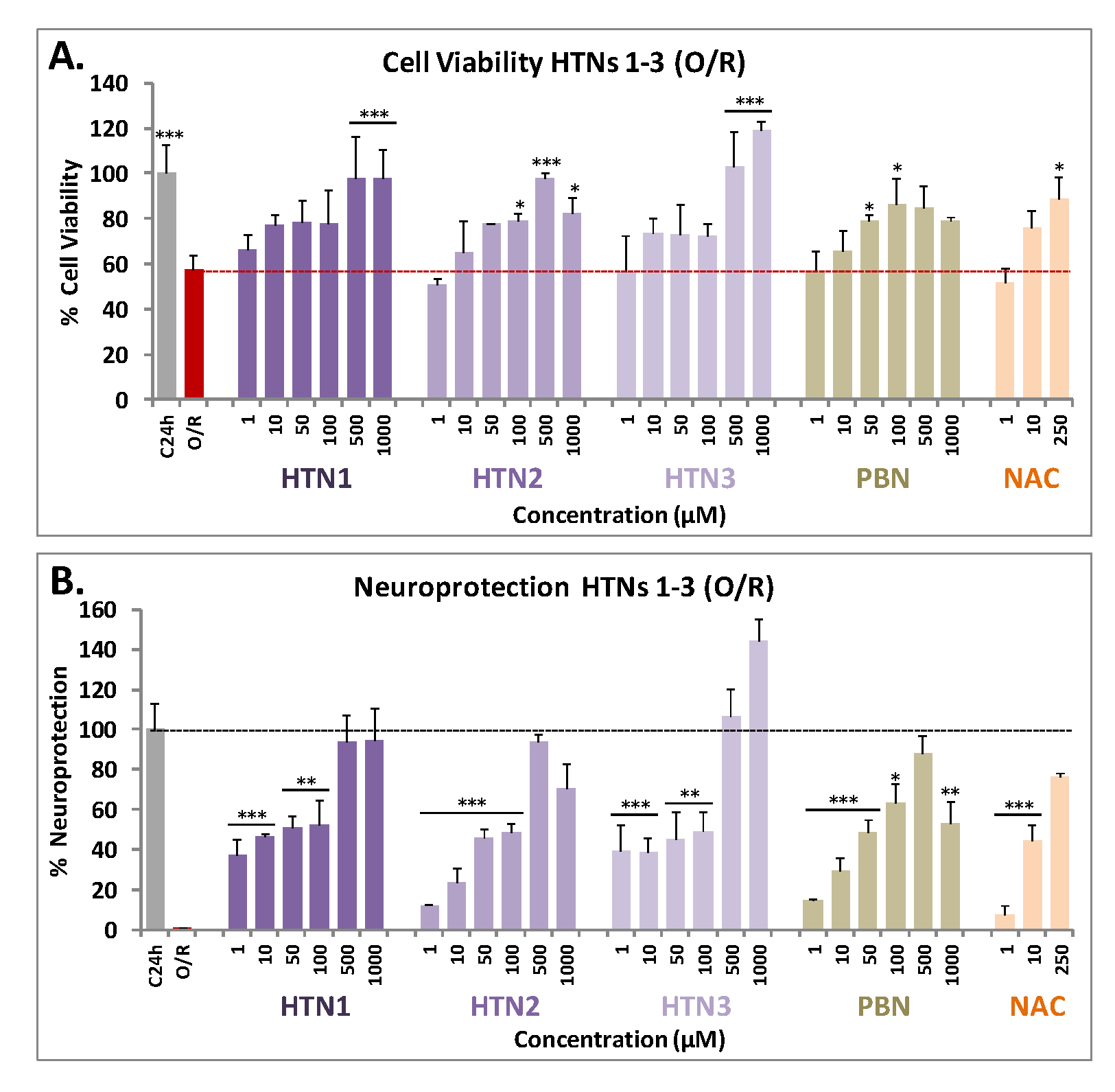
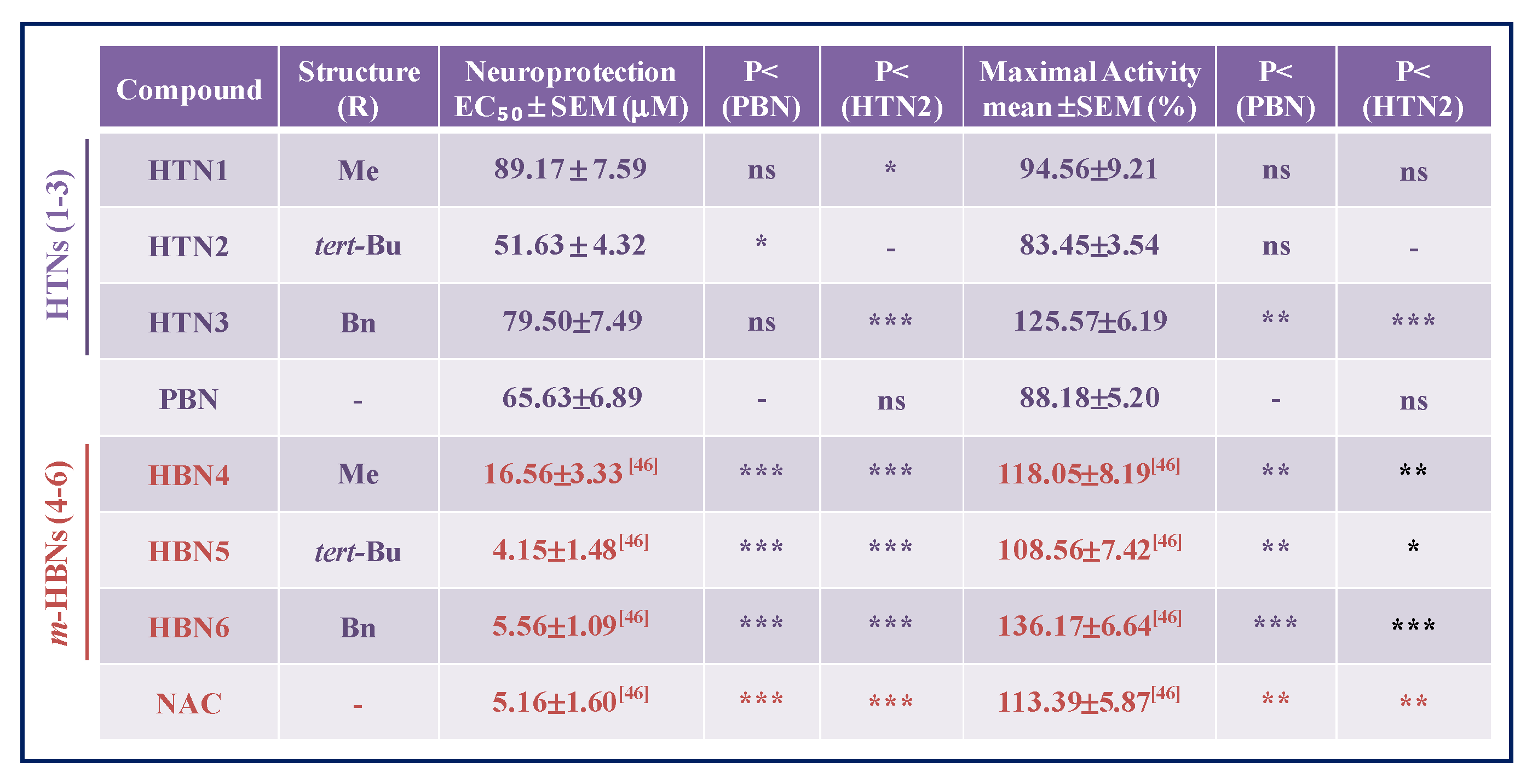
2.2.1. Neuroprotection Analysis in an Oligomycin A/Rotenone Model
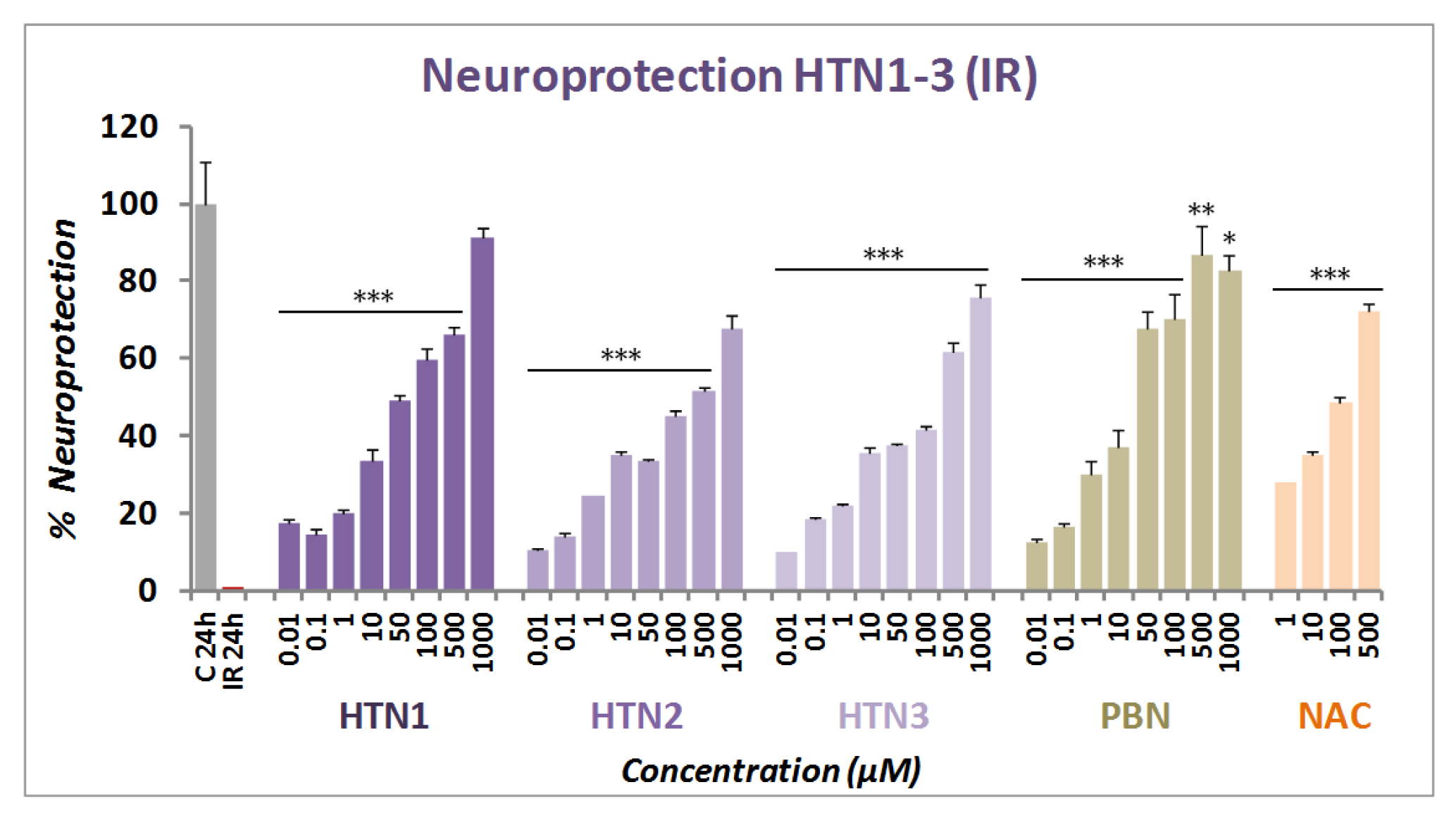
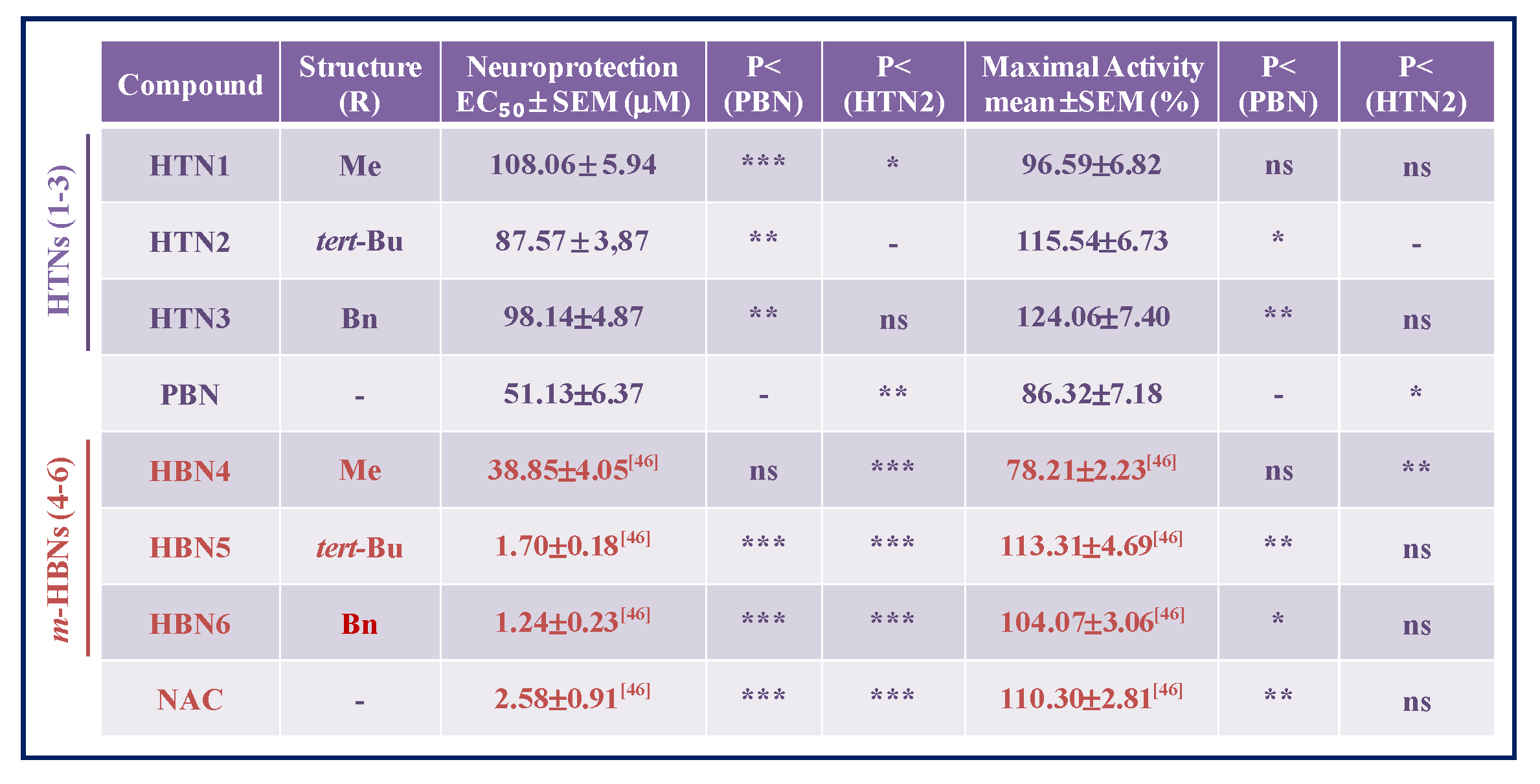
2.2.2. Neuroprotection Analysis in an Oxygen Glucose Deprivation Model
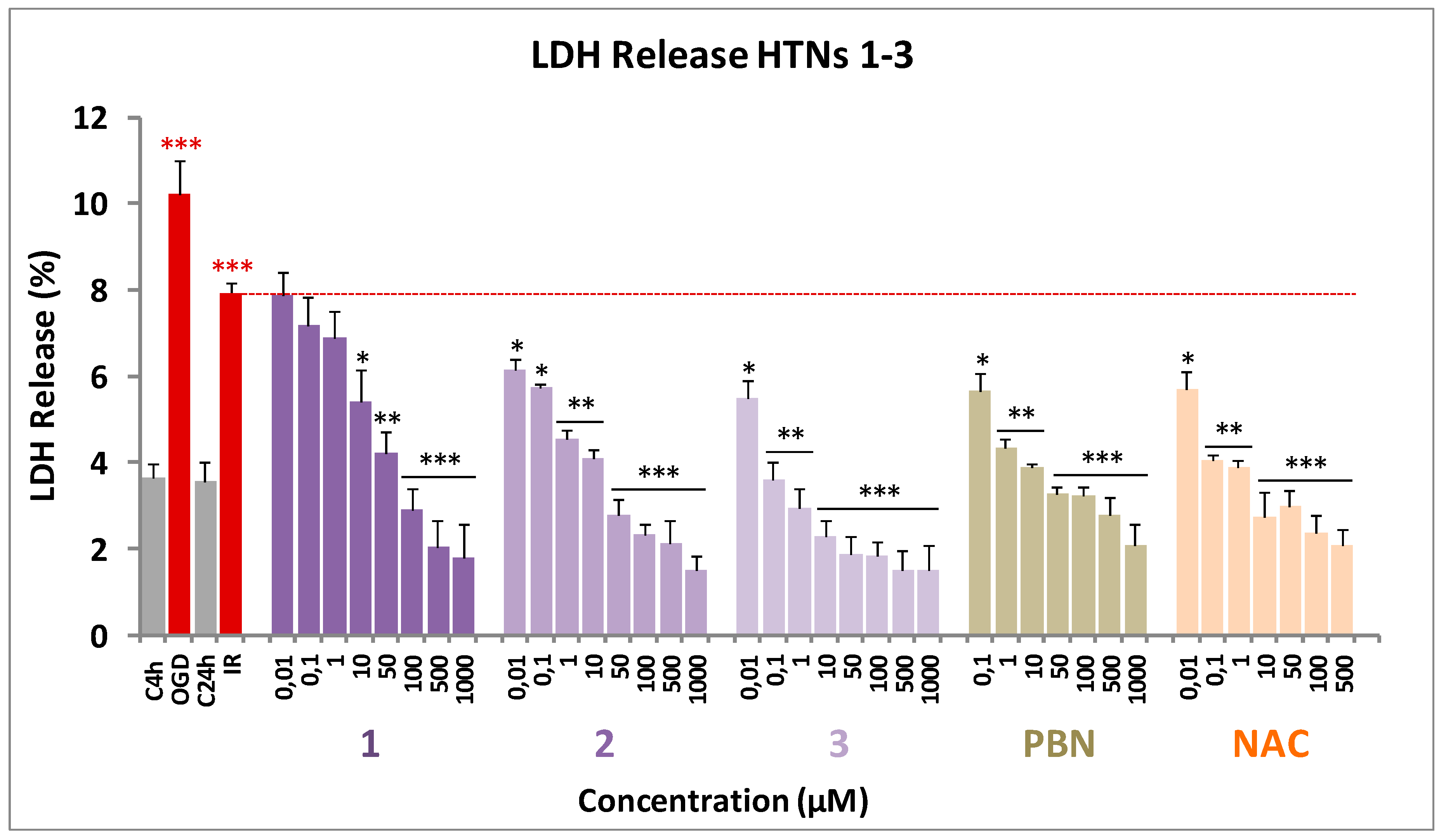
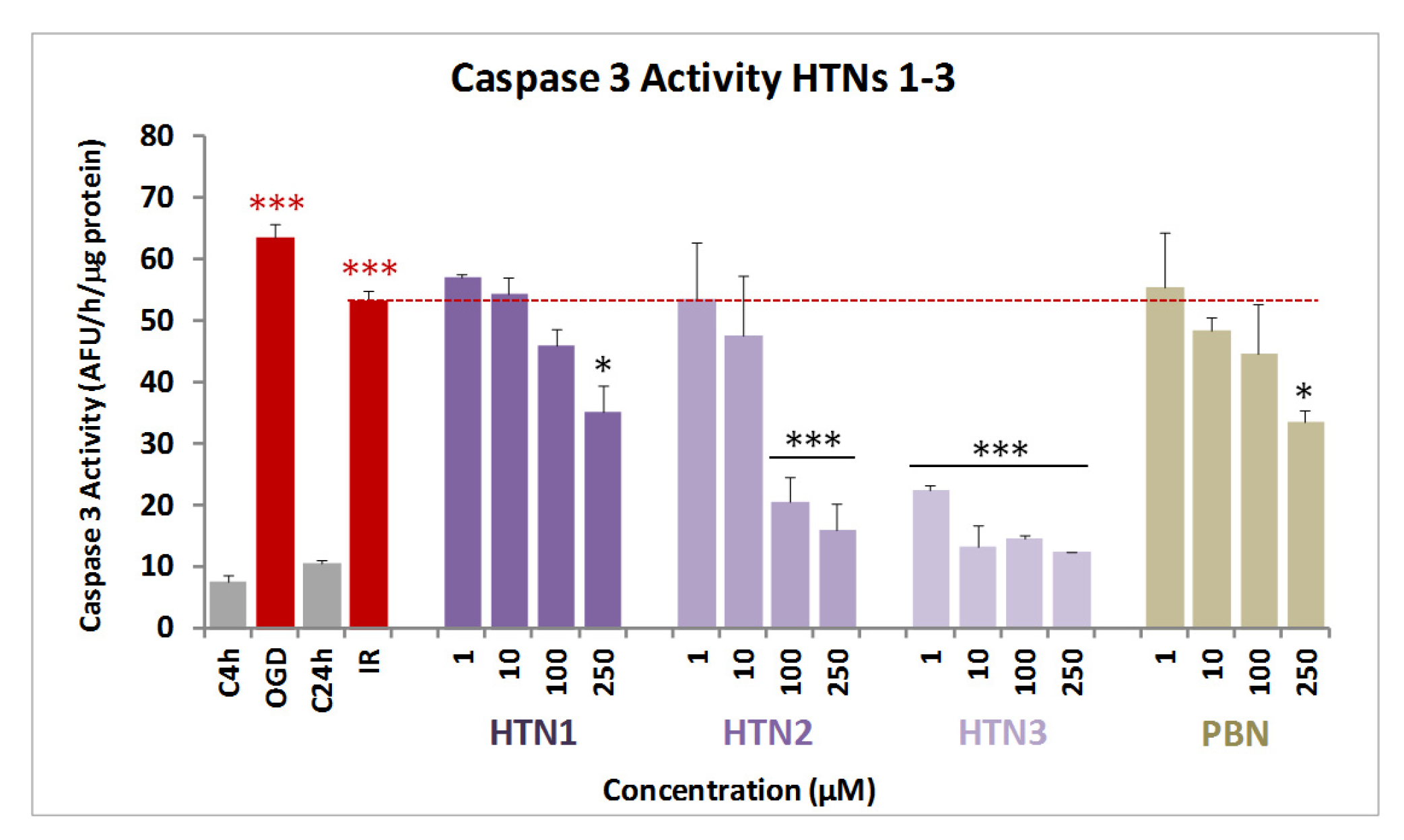
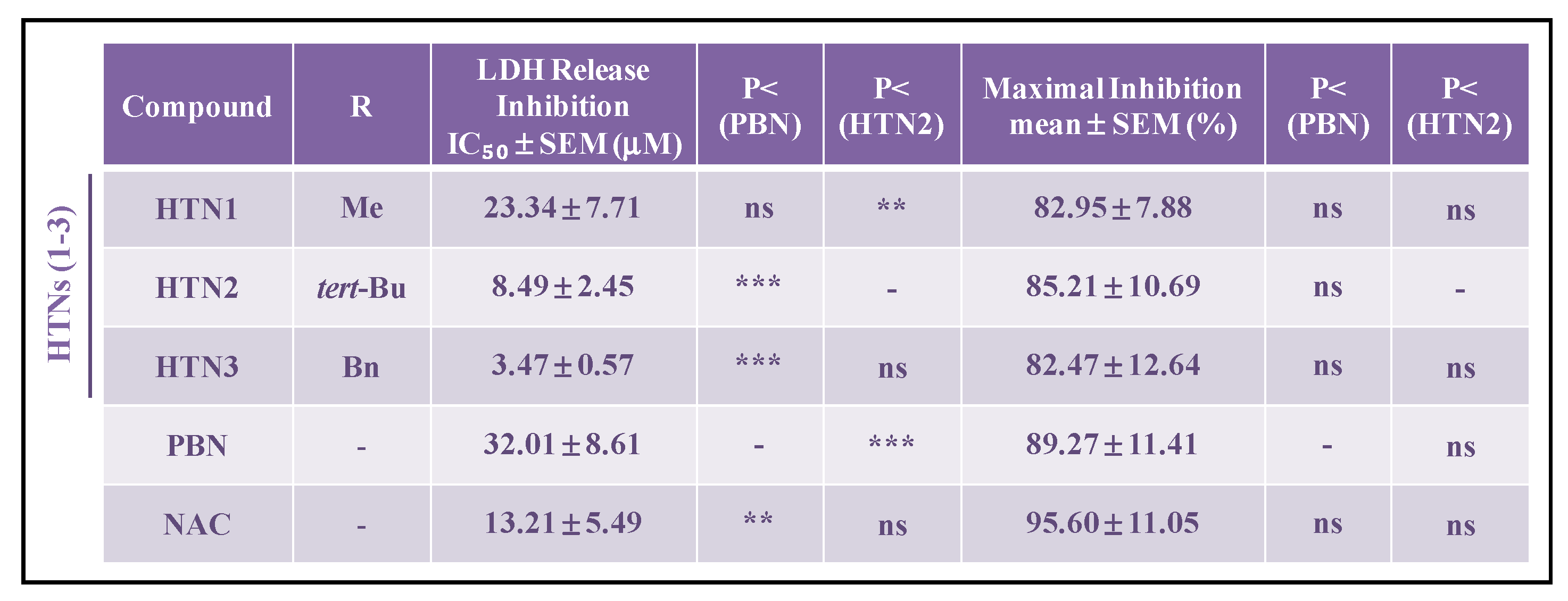
2.2.3. Effect of HTNs on Necrotic and Apoptotic Cell Death Induced by Induced by Oxygen Glucose Deprivation
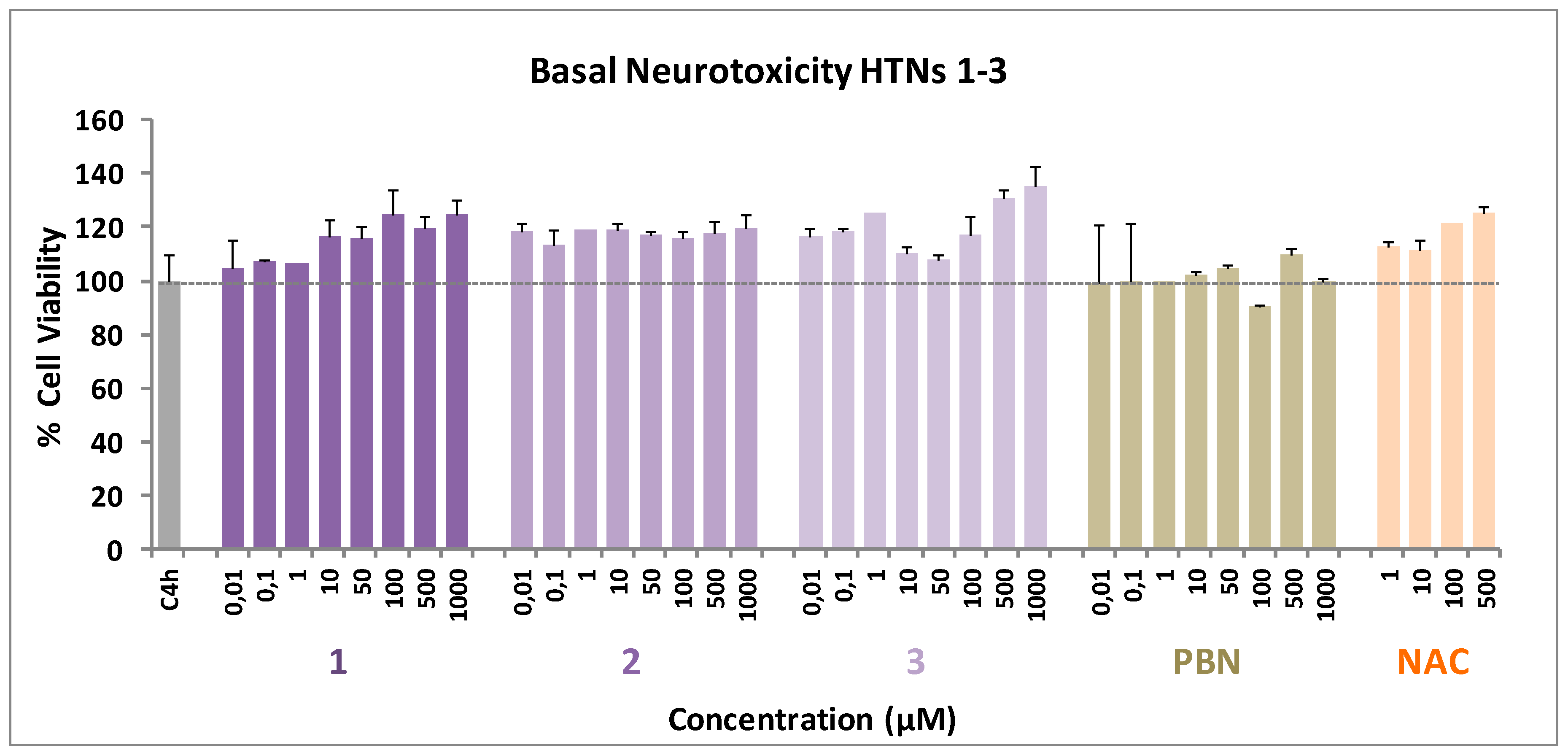
2.2.4. Basal Neurotoxicity of HTNs 1-3
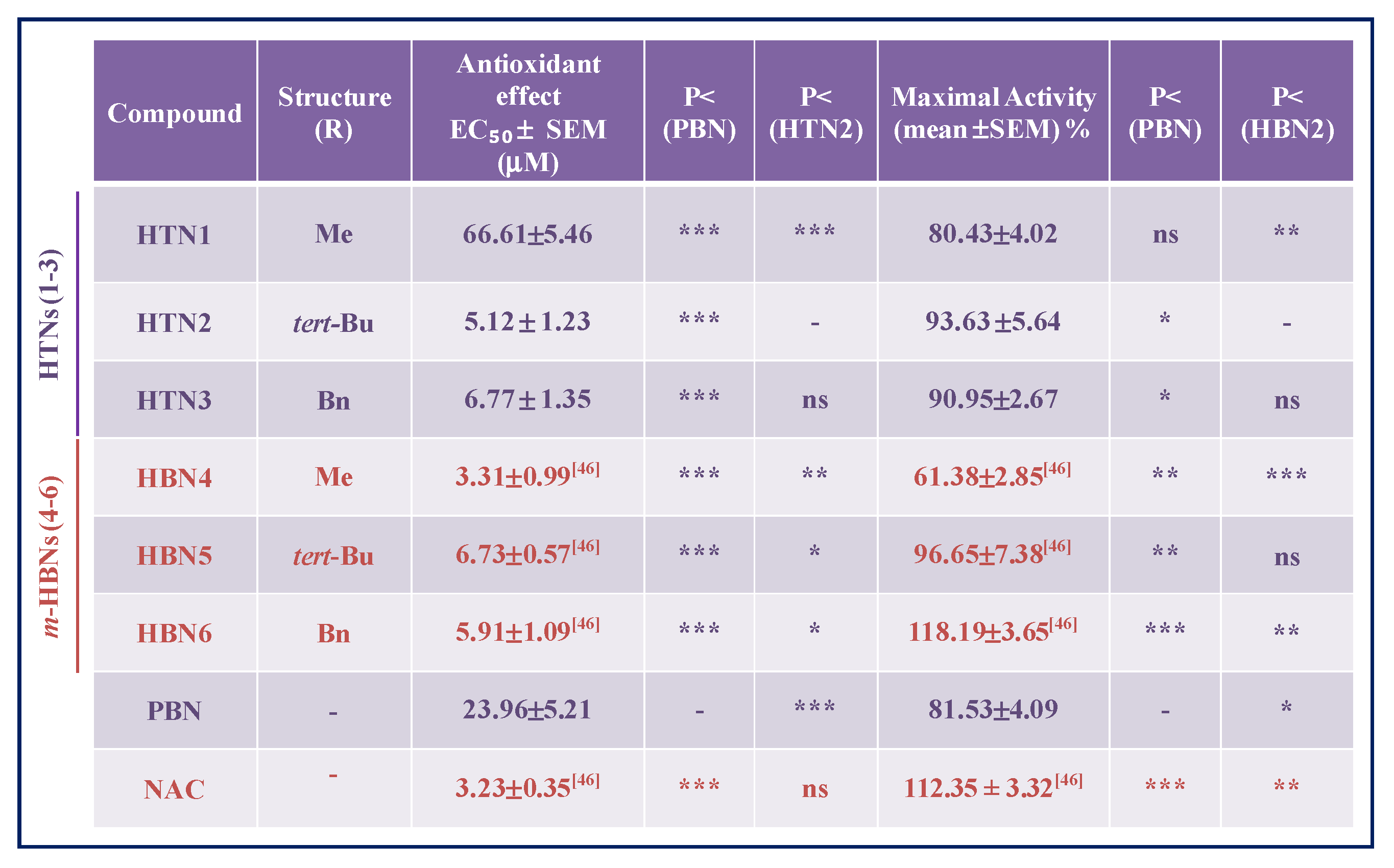
2.2.5. Antioxidant Capacity of HTNs 1-3. Production and Scavenging of Radical Superoxide Radical in Human Neuroblastoma SH-SY5Y Cells
2.3. Antioxidant Tests
3. Materials and Methods
3.1. Chemistry
3.1.1. General Methods
3.1.2. General Method for the Synthesis of HTNs
3.1.3. (1. Z,1′Z,1″Z)-1,1′,1″-(Benzene-1,3,5-triyl)tris(N-methylmethanimine oxide) (HTN1)
3.1.4. (1. Z,1′Z,1″Z)-1,1′,1″-(Benzene-1,3,5-triyl)tris(N-tert-butylmethanimine oxide) (HTN2)
3.1.5. (1. Z,1′Z,1″Z)-1,1′,1″-(Benzene-1,3,5-triyl)tris(N-benzylmethanimine oxide) (HTN3)
3.2. Neuroprotection Assessment Assays
3.2.1. Neuroblastoma Cell Cultures
3.2.2. Neuroblastoma Cell Cultures Exposure to Oxygen Glucose Deprivation
3.2.3. Assessment of Cell Viability
3.2.4. Measurement of LDH Activity
3.2.5. Analysis of Caspase-3 Activity
3.2.6. Measurement of Reactive Oxygen Species Formation
3.2.7. Statistical Analysis
3.3. Antioxidant Activity Tests of HTNs 1-3, PBN, Trolox, and NDGA in Vitro
3.3.1. Estimation of Lipophilicity as Clog P
3.3.2. General Methods
3.3.3. Inhibition of Linoleic Acid Peroxidation
3.3.4. Soybean Lipoxygenase Inhibition Study
3.3.5. ABTS+•—Decolorization Assay for Antioxidant Activity
3.3.6. Hydroxyl Radical Scavenging Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAPH | 2,2′-Azobis(2-amidinopropane) dihydrochloride |
| NAC | N-Acetyl-L-cysteine |
| BBB | Blood−brain barrier |
| HBNs | Homo-bis-nitrones |
| HTNs | Homo-tris-nitrones |
| IR | Ischemic reperfusion |
| LDH | Lactate dehydrogenase |
| LOX | Lipoxygenase |
| LP | Lipid peroxidation |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NDGA | Nordihydroguaiaretic acid |
| O/R | Oligomycin A/Rotenone |
| OGD | Oxygen Glucose Deprivation |
| PBN | α-Phenyl-N-tert-butylnitrone |
| ROS | Reactive oxygen species |
References
- Tanaka, M.; Bohár, Z.; Vécsei, L. Are kynurenines accomplices or principal villains in dementia? Maintenance of kynurenine metabolism. Molecules 2020, 25, 564. [Google Scholar] [CrossRef]
- Tanaka, M.; Vécsei, L. Monitoring the redox status in multiple sclerosis. Biomedicines 2020, 8, 406. [Google Scholar] [CrossRef]
- Catanesi, M.; d’Angelo, M.; Tupone, M.G.; Benedetti, E.; Giordano, A.; Castelli, V.; Cimini, A. 438 MicroRNAs dysregulation and mitochondrial dysfunction in neurodegenerative diseases. Int. J. Mol. Sci. 2020, 21, 5986. [Google Scholar] [CrossRef]
- Azzi, A. Antioxidants: Wonder drugs or quackery? BioFactors 2017, 43, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Nicole, O.; Docagne, F.; Ali, C.; Margaill, I.; Carmeliet, P.; MacKenzie, E.T.; Vivien, D.; Buisson, A. The proteolytic activity of tissue-plasminogen activator enhances NMDA receptor-mediated signaling. Nat. Med. 2001, 7, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.H. Cellular Antioxidant Defense Mechanisms; Chow, C.K., Ed.; CRC Press: Boca Ratón, FL, USA, 1988; Volume 3, pp. 89–109. [Google Scholar]
- Rosselin, M.; Poeggeler, B.; Durand, G. Nitrone derivatives as therapeutics: From chemical modification to specific-targeting. Curr. Top. Med. Chem. 2017, 17, 2006–2022. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Benfeito, S.; Fernandes, C.; Cagide, F.; Silva, T.; Borges, F. NO and HNO donors, nitrones, and nitroxides: Past, present, and future. Med. Res. Rev. 2018, 38, 1159–1187. [Google Scholar] [CrossRef] [PubMed]
- Floyd, R.A.; Kopke, R.D.; Choi, C.-H.; Foster, S.B.; Doblas, S.; Towner, R.A. Nitrones as therapeutics. Free Radic. Biol. Med. 2008, 45, 1361–1374. [Google Scholar] [CrossRef] [PubMed]
- Croitoru, M.D.; Ibolya, F.; Pop, M.C.; Dergez, T.; Mitroi, B.; Dogaru, M.T.; Tőkes, B. Nitrones are able to release nitric oxide in aqueous environment under hydroxyl free radical attack. Nitric Oxide 2011, 25, 309–315. [Google Scholar] [CrossRef]
- Durand, G.; Choteau, F.; Pucci, B.; Villamena, F.A. Reactivity of superoxide radical anion and hydroperoxyl radical with alpha-phenyl-N-tert-butylnitrone (PBN) derivatives. J. Phys. Chem. A 2008, 112, 12498–12509. [Google Scholar] [CrossRef]
- Cao, X.; Phillis, J.W. Phenyl-tert-butyl-nitrone reduces cortical infarct and edema in rats subjected to focal ischemia. Brain Res. 1994, 644, 267–272. [Google Scholar] [CrossRef]
- Escobar-Peso, A.; Chioua, M.; Frezza, V.; Martínez-Alonso, E.; Marco-Contelles, J.A.; Alcázar, A. Nitrones, Old fellows for new therapies in ischemic stroke. In Neuroprotective Therapy for Stroke and Ischemic Disease; Lapchak, P.A., Zhang, J.H., Eds.; Springer: Cham, Switzerland, 2017; Chapter 9; pp. 251–283. [Google Scholar]
- Maples, K.R.; Green, A.R.; Floyd, R.A. Nitrone-related therapeutics: Potential of NXY-059 for the treatment of acute ischaemic stroke. CNS Drugs 2004, 18, 1071–1084. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-Q. Bridging free radical chemistry with drug discovery: A promising way for finding novel drugs efficiently. Eur. J. Med. Chem. 2020, 189, 112020. [Google Scholar] [CrossRef] [PubMed]
- Floyd, R.A.; Neto, H.C.C.F.; Zimmerman, G.A.; Hensley, K.; Towner, R.A. Nitrone-based therapeutics for neurodegenerative diseases: Their use alone or in combination with lanthionines. Free Rad. Biol. Med. 2013, 62, 145–156. [Google Scholar] [CrossRef]
- Floyd, R.A.; Towner, R.A.; He, T.; Hensley, K.; Maples, K.R. Translational research involving oxidative stress and diseases of aging. Free Radic. Biol. Med. 2011, 51, 931–941. [Google Scholar] [CrossRef]
- Mandal, M.N.; Moiseyev, G.P.; Elliott, M.H.; Kasus-Jacobi, A.; Li, X.; Chen, H.; Zheng, L.; Nikolaeva, O.; Floyd, R.A.; Ma, J.-X.; et al. Alpha-phenyl-N-tert-butylnitrone (PBN) prevents light-induced degeneration of the retina by inhibiting RPE65 protein isomerohydrolase activity. J. Biol. Chem. 2011, 286, 32491–32501. [Google Scholar] [CrossRef]
- Ewert, D.; Hu, N.; Du, X.; Li, W.; West, M.B.; Choi, C.-H.; Floyd, R.; Kopke, R.D. HPN-07, a free radical spin trapping agent, protects against functional, cellular and electrophysiological changes in the cochlea induced by acute acoustic trauma. PLoS ONE 2017, 12, e0183089. [Google Scholar] [CrossRef]
- Novelli, G.P.; Angiolini, P.; Tani, R.; Consales, G.; Bordi, L. Phenyl-t-butyl-nitrone is active against traumatic shock in rats. Free Radic. Res. Commun. 1986, 1, 321–327. [Google Scholar] [CrossRef]
- Dias, A.G.; Santos, C.E.V.; Cyrino, F.Z.G.A.; Bouskela, E.; Costa, P.R.R. N-tert-Butyl and N-methyl nitrones derived from aromatic aldehydes inhibit macromolecular permeability increase induced by ischemia/reperfusion in hamsters. Bioorg. Med. Chem. 2009, 17, 3995–3998. [Google Scholar] [CrossRef]
- Sun, Y.; Jiang, J.; Zhang, Z.; Yu, P.; Wang, L.; Xu, C.; Liu, W.; Wang, Y. Antioxidative and Thrombolytic TMP Nitrone for Treatment of Ischemic Stroke. Bioorg. Med. Chem. 2008, 16, 8868–8874. [Google Scholar] [CrossRef]
- Sun, Y.; Yu, P.; Zhang, G.; Wang, L.; Zhong, H.; Zhai, Z.; Wang, L.; Wang, Y. Therapeutic effects of tetramethylpyrazine nitrone in rat ischemic stroke models. J. Neurosci. Res. 2012, 90, 1662–1669. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, G.; Sun, Y.; Szeto, S.S.W.; Law, H.C.H.; Quan, Q.; Li, G.; Yu, P.; Sho, E.; Siu, M.K.W.; et al. Tetramethylpyrazine nitrone, a multifunctional neuroprotective agent for ischemic stroke therapy. Sci. Rep. 2016, 6, 37148. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, G.; Zhang, Z.; Yu, P.; Zhong, H.; Du, J.; Wang, Y. Novel multi-functional nitrones for treatment of ischemic stroke. Bioorg. Med. Chem. 2012, 20, 3939–3945. [Google Scholar] [CrossRef] [PubMed]
- Socrier, L.; Rosselin, M.; Choteau, F.; Durand, G.; Morandat, S. Cholesterol-nitrone conjugates as protective agents against lipid oxidation: A model membrane study. BBA Biomembranes 2017, 1859, 2495–2504. [Google Scholar] [CrossRef] [PubMed]
- Choteau, F.; Durand, G.; Ranchon-Cole, I.; Cercy, C.; Pucci, B. Cholesterol-based-phenyl-N-tert-butyl nitrone derivatives as antioxidants against light-induced retinal degeneration. Bioorg. Med. Chem. Lett. 2010, 20, 7405–7409. [Google Scholar] [CrossRef] [PubMed]
- Rosselin, M.; Tuccio, B.; Perio, P.; Villamena, F.A.; Fabre, P.-L.; Durand, G. Electrochemical and spin-trapping properties of para-substituted α-phenyl-N-tert-butyl nitrones. Electrochim. Acta 2016, 193, 231–239. [Google Scholar] [CrossRef]
- Zeghdaoui, A.; Tuccio, B.; Finet, J.-P.; Cerri, V.; Tordo, P. β-Phosphorylated α-phenyl-N-tert-butylnitrone (PBN) analogues: A new series of spin traps for oxyl radicals. J. Chem. Soc. Perkin Trans. 1995, 2, 2087–2089. [Google Scholar] [CrossRef]
- Roubaud, V.; Lauricella, R.; Bouteiller, J.-C.; Tuccio, B. N-2-(2-Ethoxycarbonyl-propyl) α-phenylnitrone: An efficacious lipophilic spin trap for superoxide detection. Arch. Biochem. Biophys. 2002, 397, 51–56. [Google Scholar] [CrossRef]
- Rosselin, M.; Choteau, F.; Zéamari, K.; Nash, K.M.; Das, A.; Lauricella, R.; Lojou, E.; Tuccio, B.; Villamena, F.A.; Durand, G. Reactivities of substituted α-phenyl-N-tert -butyl nitrones. J. Org. Chem. 2014, 79, 6615–6626. [Google Scholar] [CrossRef]
- Deletraz, A.; Zéamari, K.; Hua, K.; Combes, M.; Villamena, F.A.; Tuccio, B.; Callizot, N.; Durand, G. Substituted α-phenyl and α-naphthlyl-N-tert-butyl nitrones: Synthesis, spin-trapping, and neuroprotection evaluation. J. Org. Chem. 2020, 85, 6073–6085. [Google Scholar] [CrossRef]
- Balogh, G.T.; Vukics, K.; Könczöl, A.; Kis-Varga, A.; Gere, A.; Fischer, J. Nitrone derivatives of trolox as neuroprotective agents. Bioorg. Med. Chem. Lett. 2005, 15, 3012–3015. [Google Scholar] [CrossRef] [PubMed]
- Chioua, M.; Sucunza, D.; Soriano, E.; Hadjipavlou-Litina, D.; Alcázar, A.; Ayuso, I.; Oset-Gasque, M.J.; González, M.P.; Monjas, L.; Rodríguez-Franco, M.I.; et al. Aryl-N-alkyl nitrones, as potential agents for stroke treatment: Synthesis, theoretical calculations, antioxidant, anti-inflammatory, neuroprotective and brain-blood barrier permeability properties. J. Med. Chem. 2012, 55, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, M.I.; Chioua, M.; Martínez-Alonso, E.; Soriano, E.; Montaner, J.; Masjuán, J.; Hadjipavlou-Litina, D.; Marco-Contelles, J.; Alcázar, A. Cholesteronitrones for stroke. J. Med. Chem. 2015, 58, 6704–6709. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, M.I.; Martínez-Alonso, E.; Chioua, M.; Escobar-Peso, A.; Gonzalo-Gobernado, R.; Montaner, J.; Marco-Contelles, J.; Alcázar, A. Quinolinylnitrone RP19 induces neuroprotection after transient brain ischemia. ACS Chem. Neurosci. 2017, 8, 2202–2213. [Google Scholar] [CrossRef]
- Chioua, M.; Salgado-Ramos, M.; Diez-Iriepa, D.; Escobar-Peso, A.; Isabel Iriepa, I.; Hadjipavlou-Litina, D.; Martínez-Alonso, E.; Alcázar, A.; Marco-Contelles, J. Novel quinolylnitrones combining neuroprotective and antioxidant properties. ACS Chem. Neurosci. 2019, 10, 2703–2706. [Google Scholar] [CrossRef]
- Chioua, M.; Martinez-Alonso, E.; Gonzalo-Gobernado, R.; Ayuso, M.I.; Escobar-Peso, A.; Infantes, L.; Hadjipavlou-Litina, D.; Montoya, J.J.; Montaner, J.; Alcazar, A.; et al. New quinolylnitrones for stroke therapy: Antioxidant and neuroprotective (Z)-N-tert-butyl-1-(2-chloro-6-methoxyquinolin-3-yl)methanimine oxide as a new lead-compound for ischemic stroke treatment. J. Med. Chem. 2019, 62, 2184–2201. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.A.; Ley, J.J.; Echegoyen, L.; Alvarado, R. Stilbazulenyl nitrone (STAZN): A nitronyl-substituted hydrocarbon with the potency of classical phenolic chain-breaking antioxidants. J. Am. Chem. Soc. 2002, 124, 4678–4684. [Google Scholar] [CrossRef]
- Ginsberg, M.D.; Becker, D.A.; Busto, R.; Belayev, A.; Zhang, Y.; Khoutorova, L.; Ley, J.J.; Zhao, W.; Belayev, L. Stilbazulenyl nitrone, a novel antioxidant, is highly neuroprotective in focal ischemia. Ann. Neurol. 2003, 54, 330–342. [Google Scholar] [CrossRef]
- Lapchak, P.A.; Schubert, D.R.; Maher, P.A. De-risking of stilbazulenyl nitrone (stazn), a lipophilic nitrone to treat stroke using a unique panel of in vitro assays. Trans. Stroke Res. 2011, 2, 209–217. [Google Scholar] [CrossRef][Green Version]
- Althaus, J.S.; Fleck, T.J.; Becker, D.A.; Hall, E.D.; Von Voigtlander, P.F. Azulenyl nitrones: Colorimetric detection of oxyradical end products and neuroprotection in the gerbil transient forebrain ischemia/reperfusion model. Free Radical Biol. Med. 1998, 24, 738–744. [Google Scholar] [CrossRef]
- Xu, D.; Duan, H.; Zhang, Z.; Cui, W.; Wang, L.; Sun, Y.; Lang, M.; Hoy, P.M.; Han, Y.; Wang, Y.; et al. The novel tetramethypyrazine bis-nitrone (TN-2) protects against MPTP/MPP+-induced neurotoxicity via inhibition of mitochondrial-dependent apoptosis. J. Neuroimmune Pharmacol. 2014, 9, 245–258. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.-P.; Zhang, K.; Zhang, Z.-J.; Sun, Y.-W.; Guo, B.-J.; Wang, Y.-Q.; Hoi, P.-M.; Han, Y.-F.; Lee, S.M.-Y. A Novel Tetramethylpyrazine bis-nitrone (TN-2) protects against 6-hydroxyldopamine-induced neurotoxicity via modulation of the NF-κB and the PKCα/PI3-K/Akt pathways. Neurochem. Int. 2014, 78, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liao, K.; Li, S.; Zhang, Z.; Yu, P.; Wang, Y. Pharmacokinetic analysis of tetramethylpyrazine bis-nitrone TN-2 in rats and its protein binding in vitro. Lett. Drug Des. Discov. 2014, 11, 770–777. [Google Scholar] [CrossRef]
- Chamorro, B.; Diez-Iriepa, D.; Merás-Sáiz, B.; Chioua, M.; García-Vieira, D.; Iriepa, I.; Hadjipavlou-Litina, D.; López-Muñoz, F.; Martínez-Murillo, R.; González-Nieto, D.; et al. Synthesis, antioxidant properties and neuroprotection of α-phenyl-tert-butylnitrone derived HomoBisNitrones in in vitro and in vivo ischemia models. Sci. Rep. 2020, 10, 14150. [Google Scholar] [CrossRef] [PubMed]
- Tien, M.-J.; Wang, Y.; Ho, T.-I.; Chiang, L.Y. Synthesis and magnetic properties of stable nitroxyl tri-radicals. Synth. Metals 1997, 85, 1675–1676. [Google Scholar] [CrossRef]
- Carney, J.M.; Floyd, R.A. Spin trapping agents for the treatment of diseases associated with oxidation of lipids and proteins. PCT Int. Appl. 1992, 32, S22–S27, WO9222290. [Google Scholar]
- Jiménez-Almarza, A.; Diez-Iriepa, D.; Chioua, M.; Chamorro, B.; Iriepa, I.; Martínez-Murillo, R.; Hadjipavlou-Litina, D.; Oset-Gasque, M.J.; Marco-Contelles, J. Synthesis, neuroprotective and antioxidant capacity of PBN-related indanonitrones. Bioorg. Chem. 2019, 86, 445–451. [Google Scholar] [CrossRef]
- García-Font, N.; Hayour, H.; Belfaitah, A.; Pedraz, J.; Moraleda, I.; Iriepa, I.; Bouraiou, A.; Chioua, M.; Marco-Contelles, J.; Oset-Gasque, M.J. Potent anticholinesterasic and neuroprotective pyranotacrines as inhibitors of beta-amyloid aggregation, oxidative stress and tau-phosphorylation for Alzheimer’s disease. Eur. J. Med. Chem. 2016, 118, 178–192. [Google Scholar] [CrossRef]
- Vicente, S.; Pérez-Rodríguez, R.; Oliván, A.M.; Martínez-Palacián, A.; González, M.P.; Oset-Gasque, M.J. Nitric oxide andperoxynitrite induce cellular death in bovine chromaffin cells: Evidence for a mixed necrotic and apoptotic mechanism with caspases activation. J. Neurosci. Res. 2006, 84, 78–96. [Google Scholar] [CrossRef]
- Piotrowska, D.G.; Mediavilla, L.; Cuarental, L.; Głowacka, I.E.; Marco-Contelles, J.; Hadjipavlou-Litina, D.; López-Muñoz, F.; Oset-Gasque, M.J. Synthesis and neuroprotective properties of N-substituted C-dialkoxyphosphorylated nitrones. ACS Omega 2019, 16, 8581–8587. [Google Scholar] [CrossRef]
- Pontiki, E.; Hadjipavlou-Litina, D.; Litinas, K.; Geromichalos, G. Novel cinnamic acid derivatives as antioxidant and anticancer agents: Design, synthesis and modeling studies. Molecules 2014, 19, 9655–9674. [Google Scholar] [CrossRef] [PubMed]
- Chioua, M.; González-Camuñas, A.; Catarozzo, M.T.; Alcaro, S.; Ortuso, F.; Yáñez, M.; Marco-Contelles, J. Synthesis, monoamine oxidase inhibition and computational analysis of diversely substituted N-propargylated-1,3,5-triazines. ChemistrySelect 2019, 4, 8334–8337. [Google Scholar] [CrossRef]

 |
 |
 |
 |
| Nitrones/ Standards | ClogP * | ILPO (%) | LOX Inhibition (% or IC50 [μM]) (±SEM) | Scavenger Activity for •OH (%) | ABTS+. (%) |
|---|---|---|---|---|---|
| HTN1 | −2.94 | 46 | 100 ± 1.1 μΜ | 59 | No |
| HTN2 | 0.77 | 55 | 26 ± 0.2% | 81 | No |
| HTN3 | 2.73 | 92 | 70 ± 2.6 μΜ | 83 | 10 |
| PBN | 3.02 | 11 | 23 ± 0.1% | No | 5 |
| HBN5 [46] | 4.51 | 55 | 6 ± 0.1% | 67 | No |
| HBN6 [46] | 4.96 | 37 | 29 ± 0.3% | 81 | No |
| NDGA | 0.45 μM | ||||
| Trolox | 88 | 83 | 91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diez-Iriepa, D.; Chamorro, B.; Talaván, M.; Chioua, M.; Iriepa, I.; Hadjipavlou-Litina, D.; López-Muñoz, F.; Marco-Contelles, J.; Oset-Gasque, M.J. Homo-Tris-Nitrones Derived from α-Phenyl-N-tert-butylnitrone: Synthesis, Neuroprotection and Antioxidant Properties. Int. J. Mol. Sci. 2020, 21, 7949. https://doi.org/10.3390/ijms21217949
Diez-Iriepa D, Chamorro B, Talaván M, Chioua M, Iriepa I, Hadjipavlou-Litina D, López-Muñoz F, Marco-Contelles J, Oset-Gasque MJ. Homo-Tris-Nitrones Derived from α-Phenyl-N-tert-butylnitrone: Synthesis, Neuroprotection and Antioxidant Properties. International Journal of Molecular Sciences. 2020; 21(21):7949. https://doi.org/10.3390/ijms21217949
Chicago/Turabian StyleDiez-Iriepa, Daniel, Beatriz Chamorro, Marta Talaván, Mourad Chioua, Isabel Iriepa, Dimitra Hadjipavlou-Litina, Francisco López-Muñoz, José Marco-Contelles, and María Jesús Oset-Gasque. 2020. "Homo-Tris-Nitrones Derived from α-Phenyl-N-tert-butylnitrone: Synthesis, Neuroprotection and Antioxidant Properties" International Journal of Molecular Sciences 21, no. 21: 7949. https://doi.org/10.3390/ijms21217949
APA StyleDiez-Iriepa, D., Chamorro, B., Talaván, M., Chioua, M., Iriepa, I., Hadjipavlou-Litina, D., López-Muñoz, F., Marco-Contelles, J., & Oset-Gasque, M. J. (2020). Homo-Tris-Nitrones Derived from α-Phenyl-N-tert-butylnitrone: Synthesis, Neuroprotection and Antioxidant Properties. International Journal of Molecular Sciences, 21(21), 7949. https://doi.org/10.3390/ijms21217949










