The Microenvironment of Decellularized Extracellular Matrix from Heart Failure Myocardium Alters the Balance between Angiogenic and Fibrotic Signals from Stromal Primitive Cells
Abstract
1. Introduction
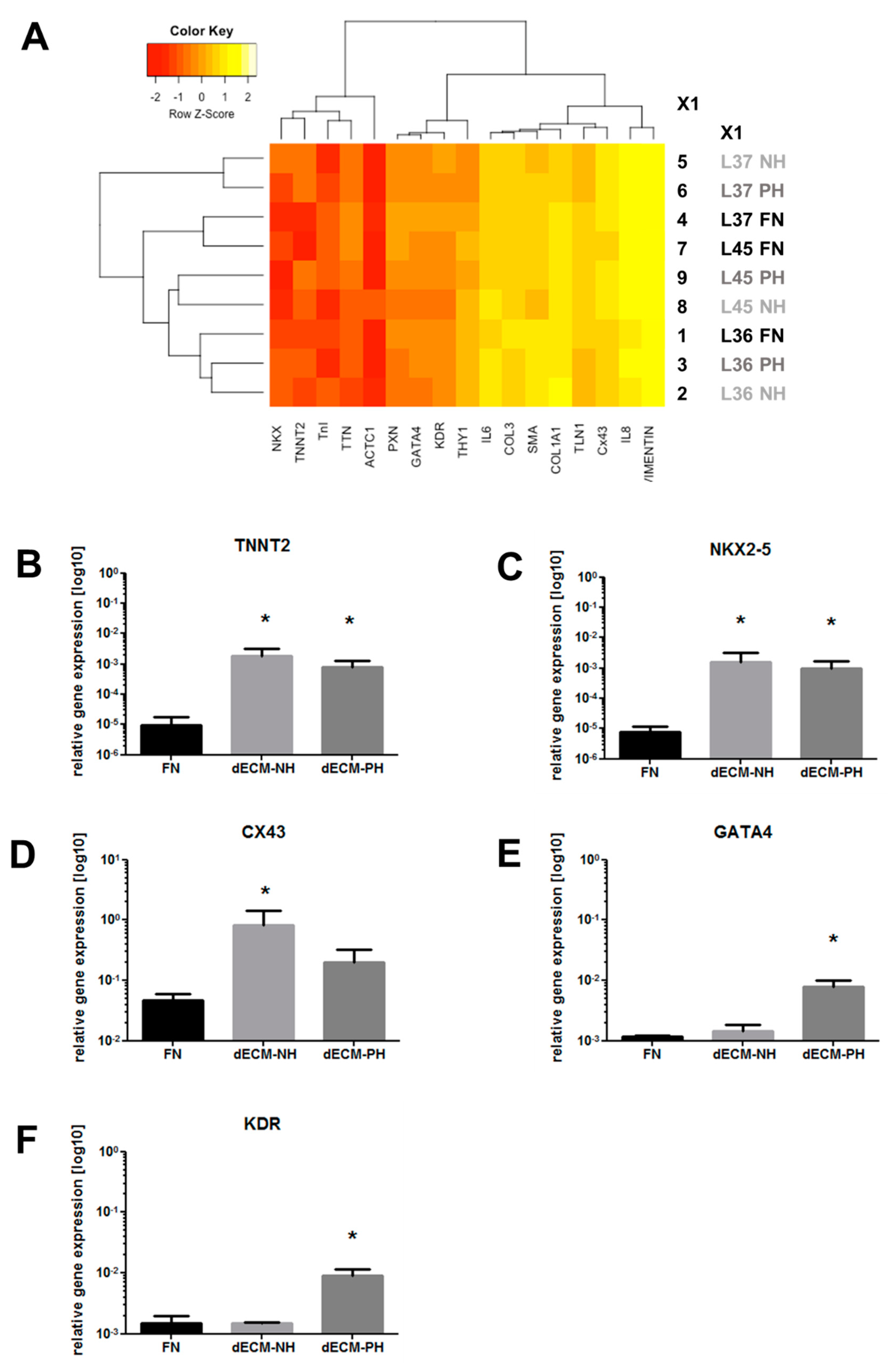
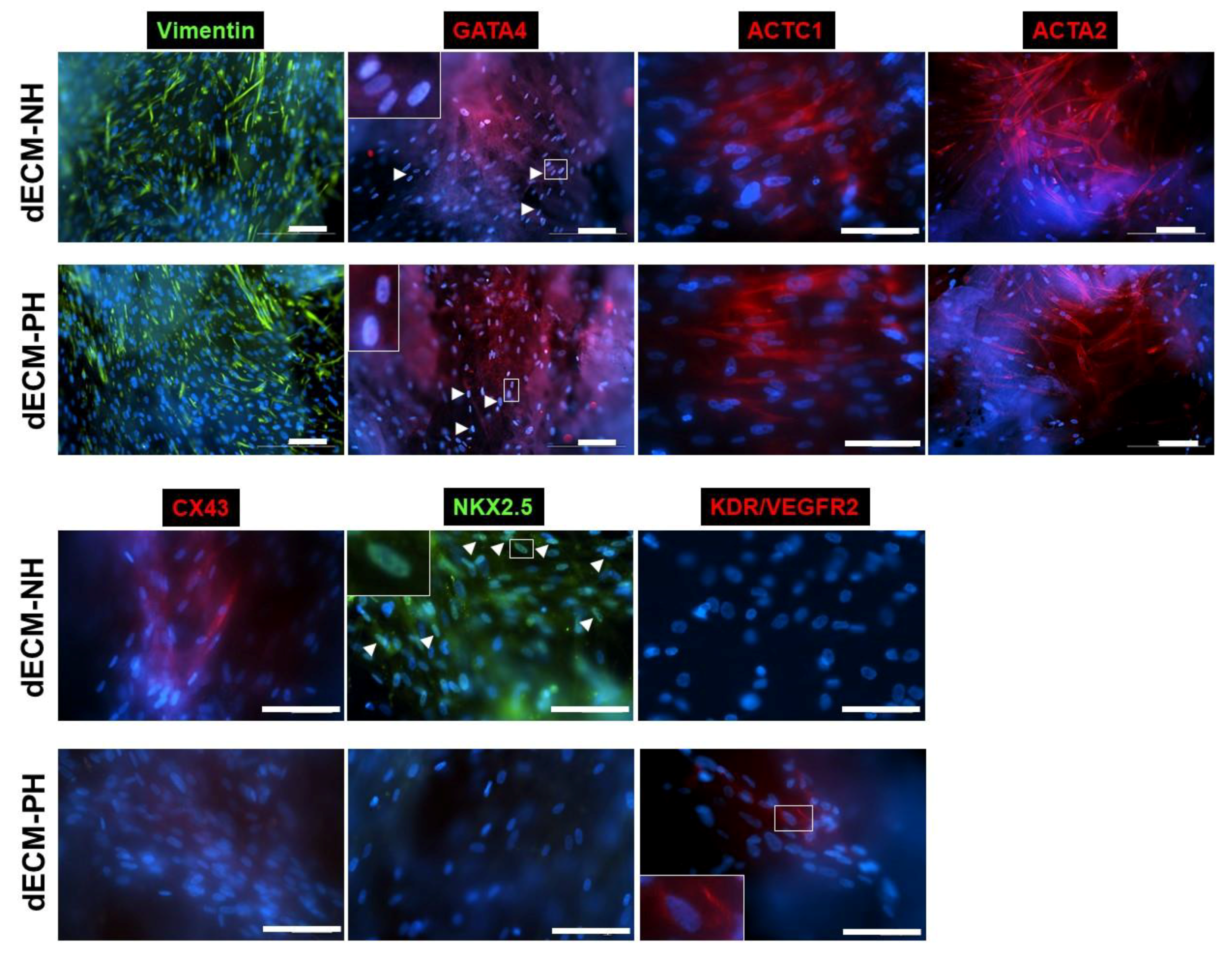
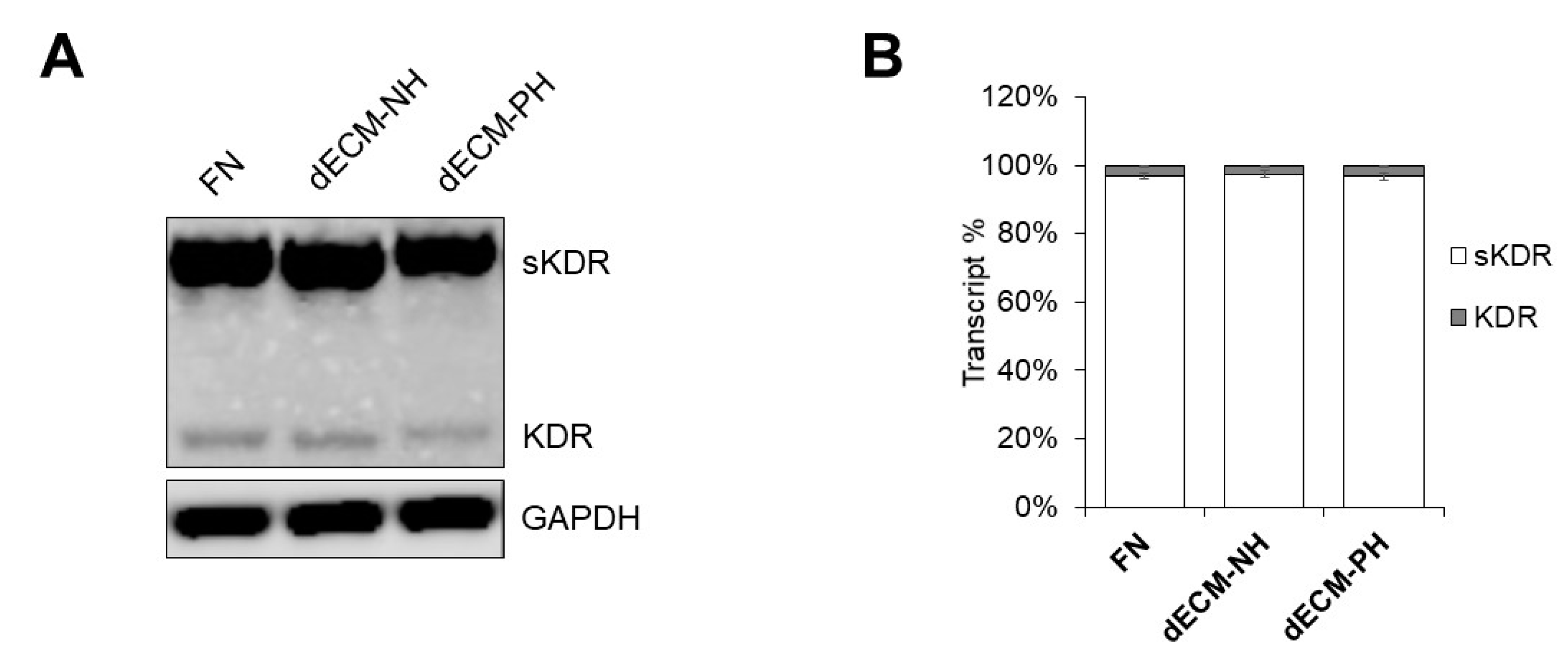
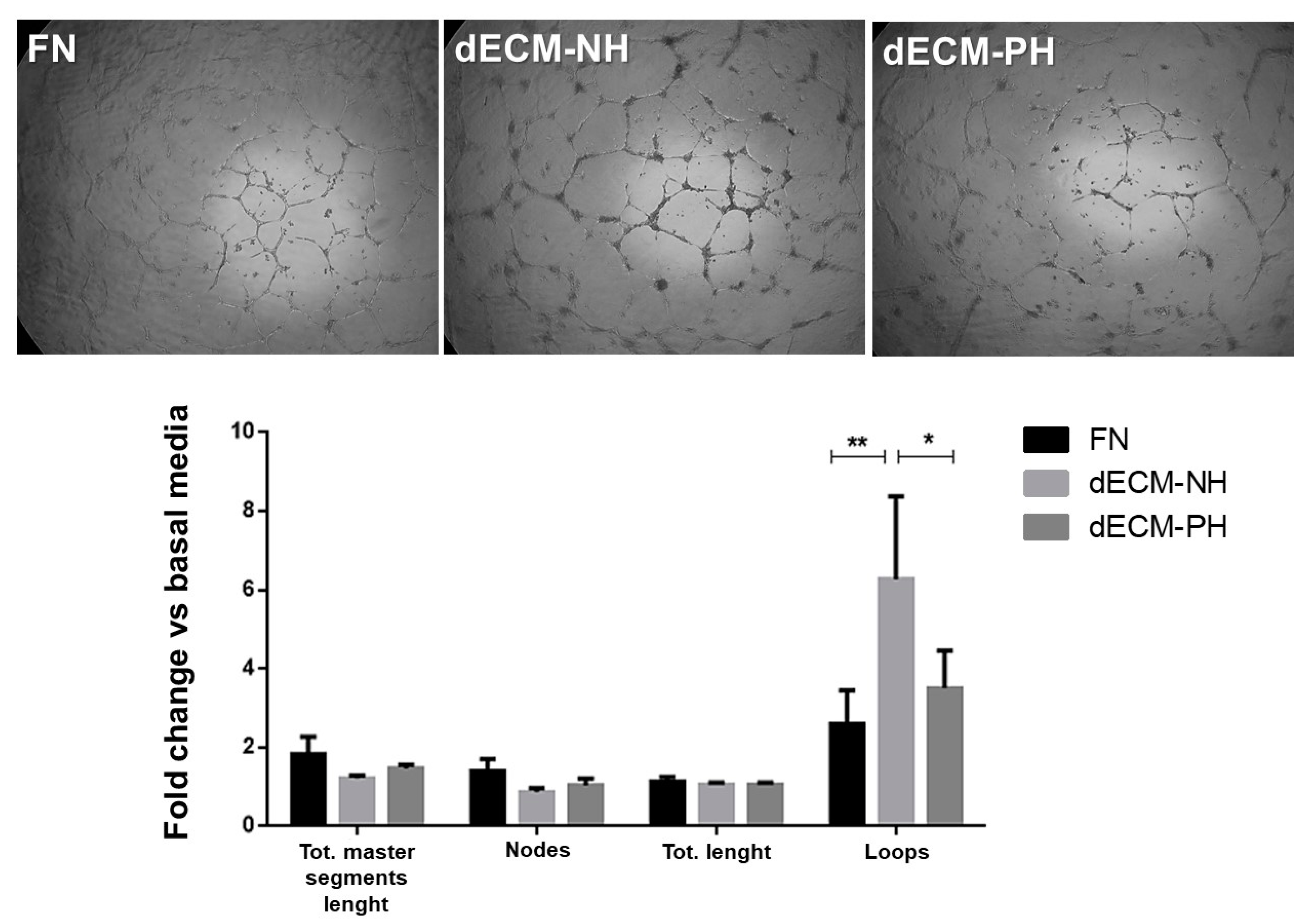
2. Results
3. Discussion
4. Materials and Methods
4.1. Decellularized Extracellular Matrix (dECM) Sections Production
4.2. Cardiosphere-Derived Cell Culture
4.3. Assay of CPC Viability
4.4. Immunostaining and Fluorescence Microscopy Analyses
4.5. RNA Extraction and Real-Time PCR
4.6. Soluble KDR/VEGFR2 mRNA Analysis
4.7. Conditioned Media Collection
4.8. Quantification of H2O2 and NO
4.9. Growth Factor Array
4.10. HUVEC Angiogenic Assay
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Townsend, N.; Wilson, L.; Bhatnagar, P.; Wickramasinghe, K.; Rayner, M.; Nichols, M. Cardiovascular disease in Europe: Epidemiological update 2016. Eur. Heart J. 2016, 37, 3232–3245. [Google Scholar] [CrossRef] [PubMed]
- Schirone, L.; Forte, M.; Palmerio, S.; Yee, D.; Nocella, C.; Angelini, F.; Pagano, F.; Schiavon, S.; Bordin, A.; Carrizzo, A.; et al. A review of the molecular mechanisms underlying the development and progression of cardiac remodeling. Oxid. Med. Cell. Longev. 2017, 2017, 3920195. [Google Scholar] [CrossRef]
- Kajstura, J.; Leri, A.; Castaldo, C.; Nadal-Ginard, B.; Anversa, P. Myocyte growth in the failing heart. Surg. Clin. North Am. 2004, 84, 161–177. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. The extracellular matrix in myocardial injury, repair, and remodeling. J. Clin. Investig. 2017, 127, 1600–1612. [Google Scholar] [CrossRef] [PubMed]
- Marban, E. A mechanistic roadmap for the clinical application of cardiac cell therapies. Nat. Biomed. Eng. 2018, 2, 353–361. [Google Scholar] [CrossRef]
- Peruzzi, M.; De Falco, E.; Abbate, A.; Biondi-Zoccai, G.; Chimenti, I.; Lotrionte, M.; Benedetto, U.; Delewi, R.; Marullo, A.G.; Frati, G. State of the Art on the evidence base in cardiac regenerative therapy: Overview of 41 systematic reviews. Biomed. Res. Int. 2015, 2015, 613782. [Google Scholar] [CrossRef]
- Marotta, P.; Cianflone, E.; Aquila, I.; Vicinanza, C.; Scalise, M.; Marino, F.; Mancuso, T.; Torella, M.; Indolfi, C.; Torella, D. Combining cell and gene therapy to advance cardiac regeneration. Expert Opin. Biol. Ther. 2018, 18, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Emmert, M.Y. Cell-based cardiac regeneration. Eur. Heart J. 2017, 38, 1095–1098. [Google Scholar] [CrossRef][Green Version]
- Mauretti, A.; Spaans, S.; Bax, N.A.M.; Sahlgren, C.; Bouten, C.V.C. Cardiac progenitor cells and the interplay with their microenvironment. Stem Cells Int. 2017, 2017, 7471582. [Google Scholar] [CrossRef]
- Nurzynska, D.; Iruegas, M.E.; Castaldo, C.; Muller-Best, P.; Di Meglio, F. Application of biotechnology in myocardial regeneration-tissue engineering triad: Cells, scaffolds, and signaling molecules. Biomed. Res. Int. 2013, 2013, 236893. [Google Scholar] [CrossRef]
- Castaldo, C.; Chimenti, I. Cardiac progenitor cells: The matrix has you. Stem Cells Transl. Med. 2018, 7, 506–510. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Smith, R.R.; Matsushita, S.; Chakravarty, T.; Czer, L.S.; Burton, K.; Schwarz, E.R.; Davis, D.R.; Wang, Q.; Reinsmoen, N.L.; et al. Intrinsic cardiac origin of human cardiosphere-derived cells. Eur. Heart J. 2013, 34, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Gaetani, R.; Feyen, D.A.; Doevendans, P.A.; Gremmels, H.; Forte, E.; Fledderus, J.O.; Ramjankhan, F.Z.; Messina, E.; Sussman, M.A.; Giacomello, A.; et al. Different types of cultured human adult cardiac progenitor cells have a high degree of transcriptome similarity. J. Cell. Mol. Med. 2014, 18, 2147–2151. [Google Scholar] [CrossRef] [PubMed]
- Santini, M.P.; Forte, E.; Harvey, R.P.; Kovacic, J.C. Developmental origin and lineage plasticity of endogenous cardiac stem cells. Development 2016, 143, 1242–1258. [Google Scholar] [CrossRef] [PubMed]
- Castaldo, C.; Di Meglio, F.; Nurzynska, D.; Romano, G.; Maiello, C.; Bancone, C.; Muller, P.; Bohm, M.; Cotrufo, M.; Montagnani, S. CD117-positive cells in adult human heart are localized in the subepicardium, and their activation is associated with laminin-1 and alpha6 integrin expression. Stem Cells 2008, 26, 1723–1731. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, F.; Castaldo, C.; Nurzynska, D.; Miraglia, R.; Romano, V.; Russolillo, V.; Giuseppina, L.; Vosa, C.; Montagnani, S. Localization and origin of cardiac CD117-positive cells: Identification of a population of epicardially-derived cells in adult human heart. Ital. J. Anat. Embryol. 2010, 115, 71–78. [Google Scholar] [PubMed]
- Fernandez-Aviles, F.; Sanz-Ruiz, R.; Climent, A.M.; Badimon, L.; Bolli, R.; Charron, D.; Fuster, V.; Janssens, S.; Kastrup, J.; Kim, H.S.; et al. Global position paper on cardiovascular regenerative medicine. Eur. Heart J. 2017, 38, 2532–2546. [Google Scholar] [CrossRef]
- Pagano, F.; Picchio, V.; Angelini, F.; Iaccarino, A.; Peruzzi, M.; Cavarretta, E.; Biondi-Zoccai, G.; Sciarretta, S.; De Falco, E.; Chimenti, I.; et al. The biological mechanisms of action of cardiac progenitor cell therapy. Curr. Cardiol. Rep. 2018, 20, 84. [Google Scholar] [CrossRef]
- Chimenti, I.; Forte, E.; Angelini, F.; Giacomello, A.; Messina, E. From ontogenesis to regeneration: Learning how to instruct adult cardiac progenitor cells. Prog. Mol. Biol. Transl. Sci. 2012, 111, 109–137. [Google Scholar]
- Fabrizi, C.; Angelini, F.; Chimenti, I.; Pompili, E.; Somma, F.; Gaetani, R.; Messina, E.; Fumagalli, L.; Giacomello, A.; Frati, G. Thrombin and thrombin-derived peptides promote proliferation of cardiac progenitor cells in the form of cardiospheres without affecting their differentiation potential. J. Biol. Regul. Homeost. Agents 2011, 25 (2 Suppl), S43–S51. [Google Scholar]
- Pagano, F.; Angelini, F.; Siciliano, C.; Tasciotti, J.; Mangino, G.; De Falco, E.; Carnevale, R.; Sciarretta, S.; Frati, G.; Chimenti, I. Beta2-adrenergic signaling affects the phenotype of human cardiac progenitor cells through EMT modulation. Pharmacol. Res. 2018, 127, 41–48. [Google Scholar] [CrossRef]
- Di Meglio, F.; Castaldo, C.; Nurzynska, D.; Romano, V.; Miraglia, R.; Bancone, C.; Langella, G.; Vosa, C.; Montagnani, S. Epithelial-mesenchymal transition of epicardial mesothelium is a source of cardiac CD117-positive stem cells in adult human heart. J. Mol. Cell. Cardiol. 2010, 49, 719–727. [Google Scholar] [CrossRef]
- Aghila Rani, K.G.; Kartha, C.C. Effects of epidermal growth factor on proliferation and migration of cardiosphere-derived cells expanded from adult human heart. Growth Factors 2010, 28, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, F.; Castaldo, C.; Nurzynska, D.; Romano, V.; Miraglia, R.; Montagnani, S. Epicardial cells are missing from the surface of hearts with ischemic cardiomyopathy: A useful clue about the self-renewal potential of the adult human heart? Int. J. Cardiol. 2010, 145, e44–e46. [Google Scholar] [CrossRef] [PubMed]
- Aquila, I.; Cianflone, E.; Scalise, M.; Marino, F.; Mancuso, T.; Filardo, A.; Smith, A.J.; Cappetta, D.; De Angelis, A.; Urbanek, K.; et al. c-kit Haploinsufficiency impairs adult cardiac stem cell growth, myogenicity and myocardial regeneration. Cell Death Dis. 2019, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Akhyari, P.; Kamiya, H.; Haverich, A.; Karck, M.; Lichtenberg, A. Myocardial tissue engineering: The extracellular matrix. Eur. J. Cardiothorac. Surg. 2008, 34, 229–241. [Google Scholar] [CrossRef]
- Forte, E.; Chimenti, I.; Rosa, P.; Angelini, F.; Pagano, F.; Calogero, A.; Giacomello, A.; Messina, E. EMT/MET at the crossroad of stemness, regeneration and oncogenesis: The ying-yang equilibrium recapitulated in cell spheroids. Cancers 2017, 9, 98. [Google Scholar] [CrossRef]
- Pesce, M.; Messina, E.; Chimenti, I.; Beltrami, A.P. Cardiac mechanoperception: A life-long story from early beats to aging and failure. Stem Cells Dev. 2017, 26, 77–90. [Google Scholar] [CrossRef]
- Pampaloni, F.; Reynaud, E.G.; Stelzer, E.H. The third dimension bridges the gap between cell culture and live tissue. Nat. Rev. Mol. Cell Biol. 2007, 8, 839–845. [Google Scholar] [CrossRef]
- Chimenti, I.; Massai, D.; Morbiducci, U.; Beltrami, A.P.; Pesce, M.; Messina, E. Stem cell spheroids and ex vivo niche modeling: Rationalization and scaling-up. J. Cardiovasc. Transl. Res. 2017, 10, 150–166. [Google Scholar] [CrossRef] [PubMed]
- Pagliarosi, O.; Picchio, V.; Chimenti, I.; Messina, E.; Gaetani, R. Building an artificial cardiac microenvironment: A focus on the extracellular matrix. Front. Cell Dev. Biol. 2020, 8, 559032. [Google Scholar] [CrossRef] [PubMed]
- Hinderer, S.; Layland, S.L.; Schenke-Layland, K. ECM and ECM-like materials—biomaterials for applications in regenerative medicine and cancer therapy. Adv. Drug Deliv. Rev. 2016, 97, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Belviso, I.; Romano, V.; Sacco, A.M.; Ricci, G.; Massai, D.; Cammarota, M.; Catizone, A.; Schiraldi, C.; Nurzynska, D.; Terzini, M.; et al. Decellularized human dermal matrix as a biological scaffold for cardiac repair and regeneration. Front. Bioeng. Biotechnol. 2020, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- Castaldo, C.; Di Meglio, F.; Miraglia, R.; Sacco, A.M.; Romano, V.; Bancone, C.; Della Corte, A.; Montagnani, S.; Nurzynska, D. Cardiac fibroblast-derived extracellular matrix (biomatrix) as a model for the studies of cardiac primitive cell biological properties in normal and pathological adult human heart. Biomed. Res. Int. 2013, 2013, 352370. [Google Scholar] [CrossRef]
- Pagano, F.; Angelini, F.; Castaldo, C.; Picchio, V.; Messina, E.; Sciarretta, S.; Maiello, C.; Biondi-Zoccai, G.; Frati, G.; Meglio, F.D.; et al. Normal versus pathological cardiac fibroblast-derived extracellular matrix differentially modulates cardiosphere-derived cell paracrine properties and commitment. Stem Cells Int. 2017, 2017, 7396462. [Google Scholar] [CrossRef]
- Nie, J.; Jiang, M.; Zhang, X.; Tang, H.; Jin, H.; Huang, X.; Yuan, B.; Zhang, C.; Lai, J.C.; Nagamine, Y.; et al. Post-transcriptional regulation of Nkx2-5 by RHAU in heart development. Cell Rep. 2015, 13, 723–732. [Google Scholar] [CrossRef]
- Ebos, J.M.; Bocci, G.; Man, S.; Thorpe, P.E.; Hicklin, D.J.; Zhou, D.; Jia, X.; Kerbel, R.S. A naturally occurring soluble form of vascular endothelial growth factor receptor 2 detected in mouse and human plasma. Mol. Cancer Res. 2004, 2, 315–326. [Google Scholar]
- Mishra, P.K.; Givvimani, S.; Chavali, V.; Tyagi, S.C. Cardiac matrix: a clue for future therapy. Biochim. Biophys. Acta 2013, 1832, 2271–2276. [Google Scholar] [CrossRef]
- Gaetani, R.; Yin, C.; Srikumar, N.; Braden, R.; Doevendans, P.A.; Sluijter, J.P.; Christman, K.L. Cardiac-derived extracellular matrix enhances cardiogenic properties of human cardiac progenitor cells. Cell Transpl. 2016, 25, 1653–1663. [Google Scholar] [CrossRef]
- Hastings, J.F.; Skhinas, J.N.; Fey, D.; Croucher, D.R.; Cox, T.R. The extracellular matrix as a key regulator of intracellular signalling networks. Br. J. Pharmacol. 2019, 176, 82–92. [Google Scholar] [CrossRef]
- Cooke, J.P.; Losordo, D.W. Nitric oxide and angiogenesis. Circulation 2002, 105, 2133–2135. [Google Scholar] [CrossRef] [PubMed]
- Kroll, J.; Waltenberger, J. A novel function of VEGF receptor-2 (KDR): Rapid release of nitric oxide in response to VEGF-A stimulation in endothelial cells. Biochem. Biophys. Res. Commun. 1999, 265, 636–639. [Google Scholar] [CrossRef] [PubMed]
- Daher, Z.; Boulay, P.L.; Desjardins, F.; Gratton, J.P.; Claing, A. Vascular endothelial growth factor receptor-2 activates ADP-ribosylation factor 1 to promote endothelial nitric-oxide synthase activation and nitric oxide release from endothelial cells. J. Biol. Chem. 2010, 285, 24591–24599. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.S.; Wang, H.; Zhang, Q.L.; Yang, Z.J.; Kong, F.X.; Wu, C.T. Hepatocyte growth factor gene therapy for ischemic diseases. Hum. Gene Ther. 2018, 29, 413–423. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, L.; Shu, B.; Tang, J.; Zhang, L.; Xie, J.; Qi, S.; Xu, Y. Granulocyte/macrophage colony-stimulating factor influences angiogenesis by regulating the coordinated expression of VEGF and the Ang/Tie system. PLoS ONE 2014, 9, e92691. [Google Scholar] [CrossRef]
- Oka, T.; Akazawa, H.; Naito, A.T.; Komuro, I. Angiogenesis and cardiac hypertrophy: Maintenance of cardiac function and causative roles in heart failure. Circ. Res. 2014, 114, 565–571. [Google Scholar] [CrossRef]
- Roy, S.; Khanna, S.; Sen, C.K. Redox regulation of the VEGF signaling path and tissue vascularization: Hydrogen peroxide, the common link between physical exercise and cutaneous wound healing. Free Radic. Biol. Med. 2008, 44, 180–192. [Google Scholar] [CrossRef]
- Zhao, T.; Zhao, W.; Chen, Y.; Li, V.S.; Meng, W.; Sun, Y. Platelet-derived growth factor-D promotes fibrogenesis of cardiac fibroblasts. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H1719–H1726. [Google Scholar] [CrossRef]
- Wang, L.; Yue, Y.; Yang, X.; Fan, T.; Mei, B.; Hou, J.; Liang, M.; Chen, G.; Wu, Z. Platelet derived growth factor alpha (PDGFRalpha) induces the activation of cardiac fibroblasts by activating c-kit. Med. Sci. Monit. 2017, 23, 3808–3816. [Google Scholar] [CrossRef]
- Sabbineni, H.; Verma, A.; Somanath, P.R. Isoform-specific effects of transforming growth factor beta on endothelial-to-mesenchymal transition. J. Cell. Physiol. 2018, 233, 8418–8428. [Google Scholar] [CrossRef]
- von Gise, A.; Pu, W.T. Endocardial and epicardial epithelial to mesenchymal transitions in heart development and disease. Circ. Res. 2012, 110, 1628–1645. [Google Scholar] [CrossRef]
- Svystonyuk, D.A.; Ngu, J.M.; Mewhort, H.E.; Lipon, B.D.; Teng, G.; Guzzardi, D.G.; Malik, G.; Belke, D.D.; Fedak, P.W. Fibroblast growth factor-2 regulates human cardiac myofibroblast-mediated extracellular matrix remodeling. J. Transl. Med. 2015, 13, 147. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; Tsuchida, M.; Lampe, P.D.; Murakami, M. Cardiomyocyte FGF signaling is required for Cx43 phosphorylation and cardiac gap junction maintenance. Exp. Cell Res. 2013, 319, 2152–2165. [Google Scholar] [CrossRef] [PubMed]
- Felkin, L.E.; Lara-Pezzi, E.A.; Hall, J.L.; Birks, E.J.; Barton, P.J. Reverse remodelling and recovery from heart failure are associated with complex patterns of gene expression. J. Cardiovasc. Transl. Res. 2011, 4, 321–331. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. Cardiac fibrosis: Cell biological mechanisms, molecular pathways and therapeutic opportunities. Mol. Asp. Med. 2019, 65, 70–99. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, R.; Ross, R.S.; Manso, A.M. Integrins and integrin-related proteins in cardiac fibrosis. J. Mol. Cell. Cardiol. 2016, 93, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Ibrahim, A.; Cheng, K.; Wu, Z.; Liang, W.; Malliaras, K.; Sun, B.; Liu, W.; Shen, D.; Cheol Cho, H.; et al. Importance of cell-cell contact in the therapeutic benefits of cardiosphere-derived cells. Stem Cells 2014, 32, 2397–2406. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, F.; Nurzynska, D.; Romano, V.; Miraglia, R.; Belviso, I.; Sacco, A.M.; Barbato, V.; Di Gennaro, M.; Granato, G.; Maiello, C.; et al. Optimization of human myocardium decellularization method for the construction of implantable patches. Tissue Eng. Part C Methods 2017, 23, 525–539. [Google Scholar] [CrossRef]
- Chimenti, I.; Pagano, F.; Cavarretta, E.; Angelini, F.; Peruzzi, M.; Barretta, A.; Greco, E.; De Falco, E.; Marullo, A.G.; Sciarretta, S.; et al. Beta-blockers treatment of cardiac surgery patients enhances isolation and improves phenotype of cardiosphere-derived cells. Sci. Rep. 2016, 6, 36774. [Google Scholar] [CrossRef]
- Chimenti, I.; Gaetani, R.; Forte, E.; Angelini, F.; De Falco, E.; Zoccai, G.B.; Messina, E.; Frati, G.; Giacomello, A. Serum and supplement optimization for EU GMP-compliance in cardiospheres cell culture. J. Cell. Mol. Med. 2014, 18, 624–634. [Google Scholar] [CrossRef]
- Belviso, I.; Sacco, A.M.; Romano, V.; Schonauer, F.; Nurzynska, D.; Montagnani, S.; Di Meglio, F.; Castaldo, C. Isolation of adult human dermal fibroblasts from abdominal skin and generation of induced pluripotent stem cells using a non-integrating method. J. Vis. Exp. 2020, e60629. [Google Scholar] [CrossRef] [PubMed]
- Nurzynska, D.; Di Meglio, F.; Romano, V.; Miraglia, R.; Sacco, A.M.; Latino, F.; Bancone, C.; Della Corte, A.; Maiello, C.; Amarelli, C.; et al. Cardiac primitive cells become committed to a cardiac fate in adult human heart with chronic ischemic disease but fail to acquire mature phenotype: Genetic and phenotypic study. Basic Res. Cardiol. 2013, 108, 320. [Google Scholar] [CrossRef] [PubMed]
- Stevens, M.; Oltean, S. Modulation of receptor tyrosine kinase activity through alternative splicing of ligands and receptors in the VEGF-A/VEGFR axis. Cells 2019, 8, 288. [Google Scholar] [CrossRef] [PubMed]
- Pagano, F.; Nocella, C.; Sciarretta, S.; Fianchini, L.; Siciliano, C.; Mangino, G.; Ibrahim, M.; De Falco, E.; Carnevale, R.; Chimenti, I.; et al. Cytoprotective and antioxidant effects of steen solution on human lung spheroids and human endothelial cells. Am. J. Transpl. 2017, 17, 1885–1894. [Google Scholar] [CrossRef]
- Sacco, A.M.; Belviso, I.; Romano, V.; Carfora, A.; Schonauer, F.; Nurzynska, D.; Montagnani, S.; Di Meglio, F.; Castaldo, C. Diversity of dermal fibroblasts as major determinant of variability in cell reprogramming. J. Cell. Mol. Med. 2019, 23, 4256–4268. [Google Scholar] [CrossRef]


| Target Gene | Primer Sequence | |
|---|---|---|
| CX43 | FW | AGGAGTTCAATCACTTGGCG |
| RV | GAGTTTGCCTAAGGCGCTC | |
| GATA4 | FW | GTTTTTTCCCCTTTGATTTTTGATC |
| RV | AACGACGGCAACAACGATAAT | |
| KDR | FW | AAAGGGTGGAGGTGACTGAG |
| RV | CGGTAGAAGCACTTGTAGGC | |
| KDR exon 12 | FW | TCAACAAAGTCGGGAGAGGA |
| KDR exon 14 | RV | GAGTCTTCTACAAGGGTCTCA |
| TnI | FW | GGACAAGGTGGATGAAGAGA |
| RV | AGGGTGGGCCGCTTAAACT | |
| SMA | FW | ATGAAGATCCTGACTGAGCG |
| RV | GCAGTGGCCATCTCATTTTC | |
| NKX2-5 | FW | GGTGGAGCTGGAGAAGACAGA |
| RV | CGCCGCTCCAGTTCATAG | |
| TNNT2 | FW | GGAGGAGTCCAAACCAAAGCC |
| RV | TCAAAGTCCACTCTCTCTCCATC | |
| TTN | FW | GCAAGAGTACCAGCACCTGT |
| RV | TCACTGTCGGGGATGGGTAT | |
| COL1A1 | FW | AAGAGGAAGGCCAAGTCGAG |
| RV | CACACGTCTCGGTCATGGTA | |
| COL3A1 | FW | CATGCCCTACTGGTCCTCAG |
| RV | ATAGCCTGCGAGTCCTCCTA | |
| VIM | FW | ACCCACTCAAAAAGGACACTTC |
| RV | GGTCATCGTGATGCTGAGAA | |
| PXN | FW | AAAGTTGCGGGGCATAGAC |
| RV | GTAGACTCCAAGTCCGCGAC | |
| TLN1 | FW | AAGGCACTTTGTGGCTTCAC |
| RV | ACTGTGTGGGCTCCACTAGC | |
| VCL | FW | ACCTTGAACAACTCCGACTAAC |
| RV | AACTCTTCATCCTTTTCCTCTGG | |
| IL-6 | FW | GGTACATCCTCGACGGCATCT |
| RV | GTGCCTCTTTGCTGCTTTCAC | |
| IL-8 | FW | CTTGGCAGCCTTCCTGATTT |
| RV | TTCTTTAGCACTCCTTGGCAAAA | |
| VEGF | FW | CTACCTCCACCATGCCAAGT |
| RV | CCACTTCGTGATGATTCTGC | |
| AREG | FW | AAGGAGAAGCTGAGGAACGAA |
| RV | TGGCAGTGACTCCAATGTGA | |
| HGF | FW | AAGTGAATACTGCAGACCAATGTG |
| RV | AAGGGGAACCAGAGGCATT | |
| IGF-2 | FW | GGACACCCTCCAGTTCGTCT |
| RV | CGGAAACAGCACTCCTCAAC | |
| PDGFA | FW | TTTGGACACCAGCCTGAGAG |
| RV | AGACAGCGGGGACAGCTT | |
| GAPDH | FW | ACAGTCAGCCGCATCTTC |
| RV | GCCCAATACGACCAAATCC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belviso, I.; Angelini, F.; Di Meglio, F.; Picchio, V.; Sacco, A.M.; Nocella, C.; Romano, V.; Nurzynska, D.; Frati, G.; Maiello, C.; et al. The Microenvironment of Decellularized Extracellular Matrix from Heart Failure Myocardium Alters the Balance between Angiogenic and Fibrotic Signals from Stromal Primitive Cells. Int. J. Mol. Sci. 2020, 21, 7903. https://doi.org/10.3390/ijms21217903
Belviso I, Angelini F, Di Meglio F, Picchio V, Sacco AM, Nocella C, Romano V, Nurzynska D, Frati G, Maiello C, et al. The Microenvironment of Decellularized Extracellular Matrix from Heart Failure Myocardium Alters the Balance between Angiogenic and Fibrotic Signals from Stromal Primitive Cells. International Journal of Molecular Sciences. 2020; 21(21):7903. https://doi.org/10.3390/ijms21217903
Chicago/Turabian StyleBelviso, Immacolata, Francesco Angelini, Franca Di Meglio, Vittorio Picchio, Anna Maria Sacco, Cristina Nocella, Veronica Romano, Daria Nurzynska, Giacomo Frati, Ciro Maiello, and et al. 2020. "The Microenvironment of Decellularized Extracellular Matrix from Heart Failure Myocardium Alters the Balance between Angiogenic and Fibrotic Signals from Stromal Primitive Cells" International Journal of Molecular Sciences 21, no. 21: 7903. https://doi.org/10.3390/ijms21217903
APA StyleBelviso, I., Angelini, F., Di Meglio, F., Picchio, V., Sacco, A. M., Nocella, C., Romano, V., Nurzynska, D., Frati, G., Maiello, C., Messina, E., Montagnani, S., Pagano, F., Castaldo, C., & Chimenti, I. (2020). The Microenvironment of Decellularized Extracellular Matrix from Heart Failure Myocardium Alters the Balance between Angiogenic and Fibrotic Signals from Stromal Primitive Cells. International Journal of Molecular Sciences, 21(21), 7903. https://doi.org/10.3390/ijms21217903















