Calcium Ionophore-Induced Extracellular Vesicles Mediate Cytoprotection against Simulated Ischemia/Reperfusion Injury in Cardiomyocyte-Derived Cell Lines by Inducing Heme Oxygenase 1
Abstract
1. Introduction
2. Results
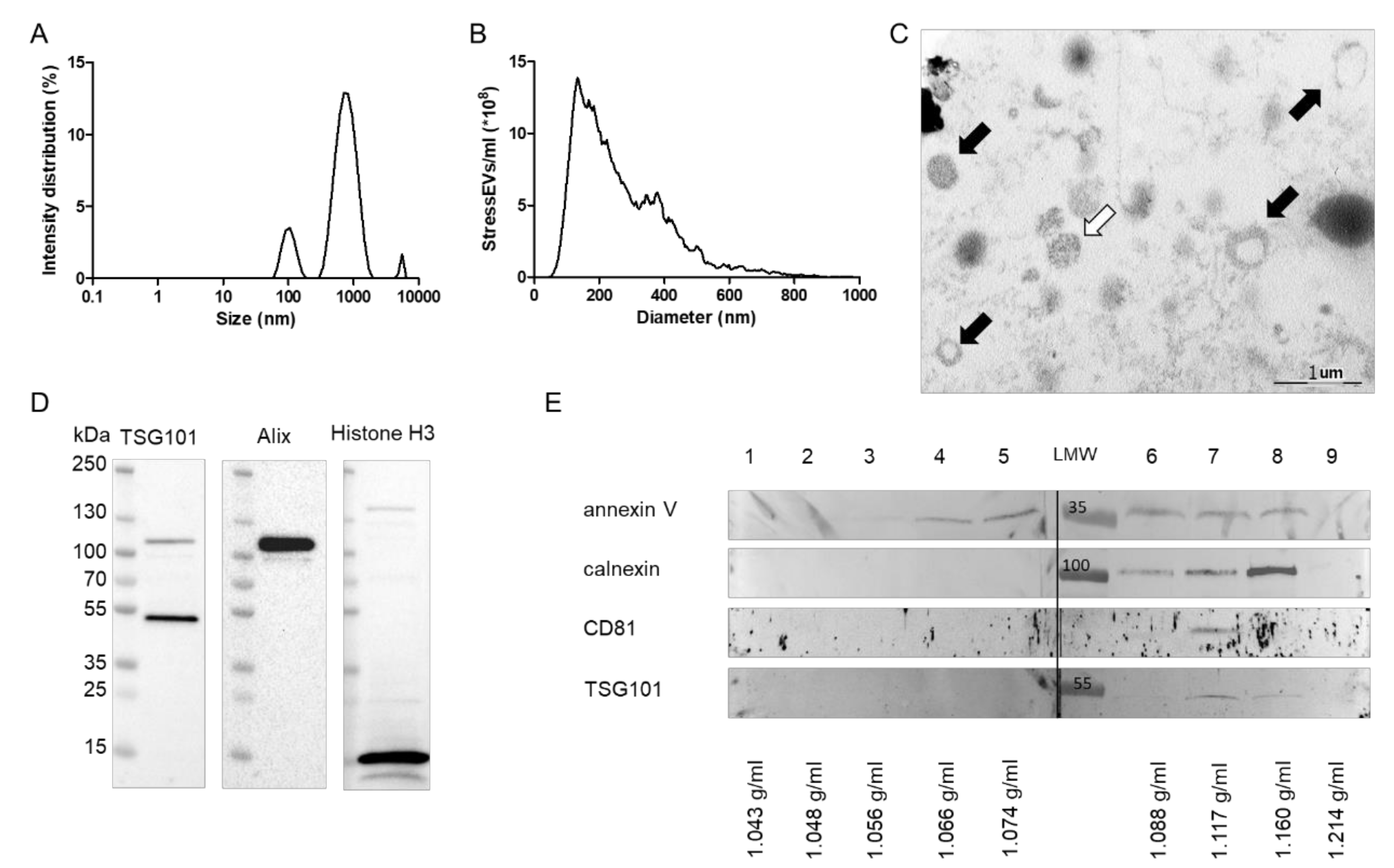
2.1. Calcium-Ionophore Treatment Induces the Release of StressEVs from HEK293 Cells
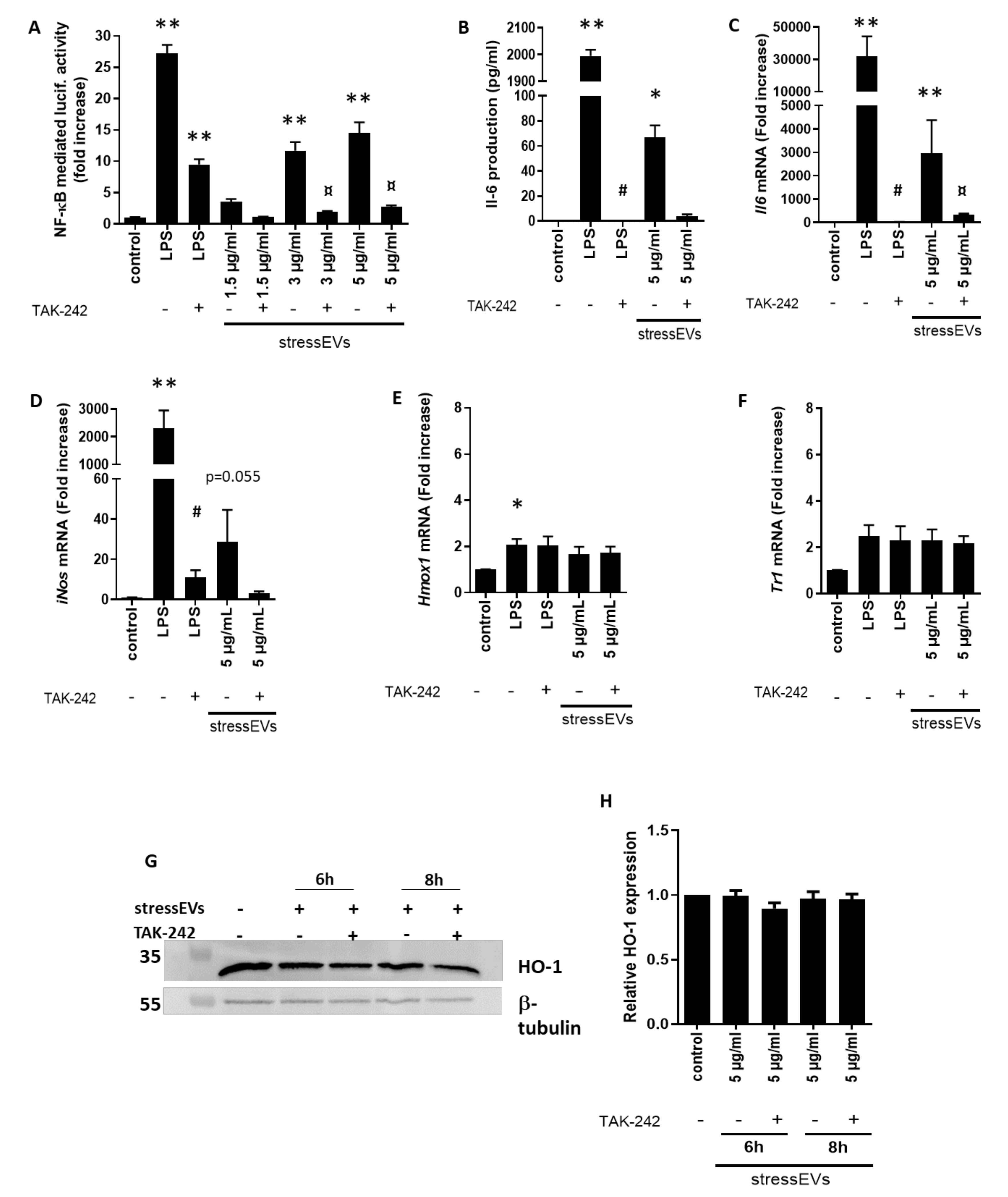
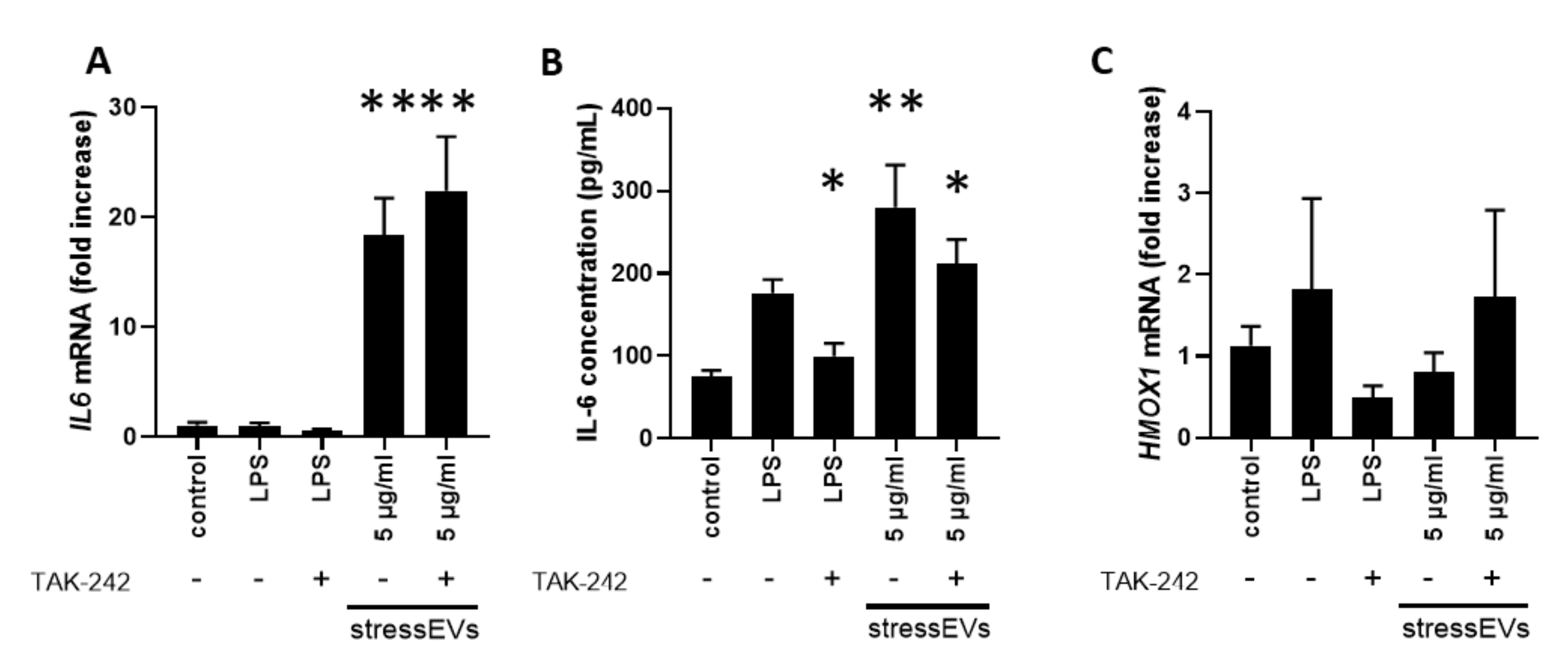
2.2. StressEVs Activate the TLR4 Receptor, but Not the Adaptive Response in Macrophages
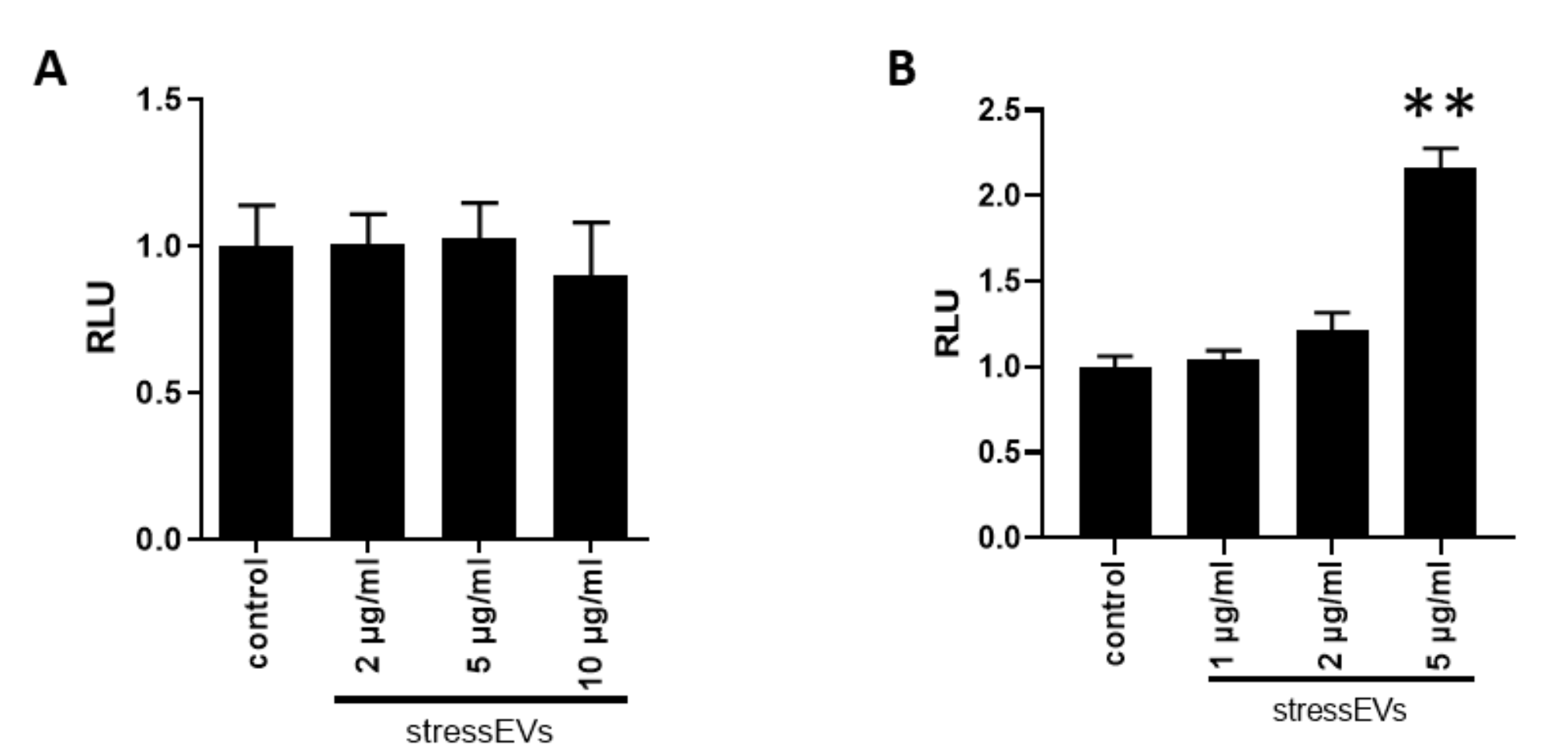
2.3. StressEVs Activate Cardioprotective Signaling in H9c2 and AC16 Cell Lines
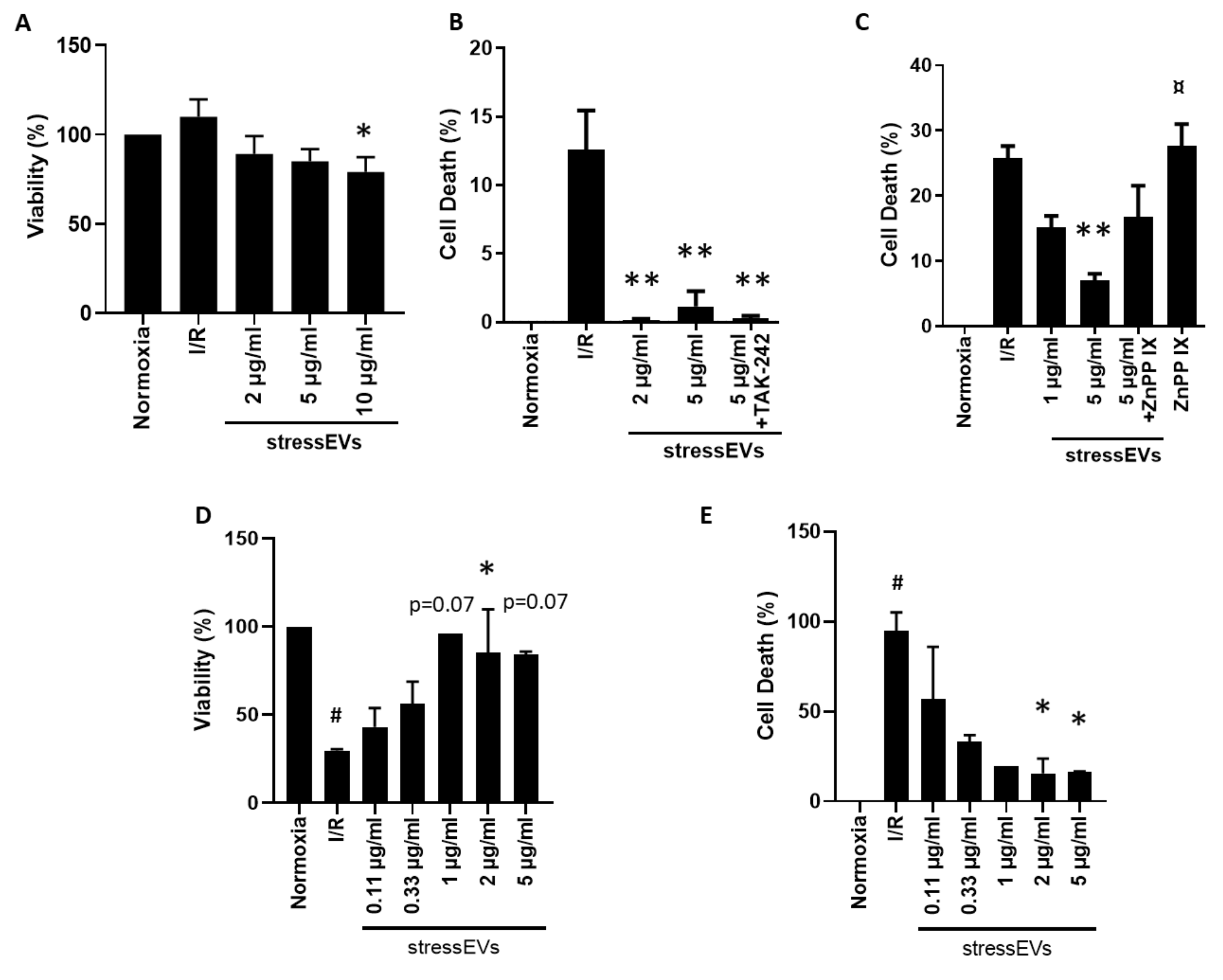
2.4. StressEVs Protect against Simulated I/R-Induced Cytotoxicity via the Induction of HO-1, Independently of TLR4 Receptor Signaling
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culturing
4.3. Isolation and Characterization of StressEVs
4.4. Assessment of TLR4 Induction and the Activation of Cardioprotective Signaling
4.5. Assessment of Toxicity and Cardioprotection by EVs
4.6. TNF-α and IL-6 ELISA
4.7. Apoptosis Assay
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Alix | ALG-2-interacting protein X |
| CD81 | Cluster of differentiation 81 |
| DAMP | Damage-associated molecular pattern |
| DLS | Dynamic light scattering |
| DMEM | Dulbecco’s modified Eagle’s medium |
| DOAJ | Directory of open access journals |
| DPBS | Dulbecco’s phosphate-buffered saline |
| ECACC | The European Collection of Authenticated Cell Cultures |
| ESCRT-III | Endosomal Sorting Complexes Required for Transport-III |
| EV | Extracellular vesicle |
| FBS | Fetal bovine serum |
| HBSS | Hank’s Balanced Salt Solution |
| HEK293 | Human embryonal kidney 293 |
| HO-1 | Heme oxygenase 1 |
| HSP | Heat shock protein |
| IL-6 | Interleukin 6 |
| iNOS | Nitric oxide synthase, inducible |
| I/R | Ischemia/reperfusion |
| LD | Linear dichroism |
| LDH | Lactate dehydrogenase |
| LPS | Lipopolysaccharide |
| MD-2 | Lymphocyte antigen 96 |
| MDPI | Multidisciplinary Digital Publishing Institute |
| MLKL | Mixed lineage kinase domain-like protein |
| MyD88 | Myeloid differentiation primary response 88 |
| NF-κB | Nuclear factor kappa-B |
| NO | Nitric oxide |
| Nrf2/Keap-1 | NF-E2-related factor 2/Kelch-like ECH-associated protein 1 |
| NTA | Nanoparticle tracking analysis |
| oxPL | Oxidized phospholipid |
| PAMP | Pathogen-associated molecular patterns |
| PBS | Phosphate buffered saline |
| PI3K/Akt | Phosphatidylinositol 3-kinase/Akt |
| PKC | Protein kinase C |
| PRR | Pattern recognition receptor |
| RIC | Remote ischemic conditioning |
| RPMI 1640 | Roswell Park Memorial Institute 1640 Medium |
| TLA | Three letter acronym |
| TLR | Toll-like receptor |
| TNFα | Tumor necrosis factor alpha |
| Tr1 | Thioredoxin reductase 1 |
| TSG101 | Tumor susceptibility gene 101 |
| ZnPP IX | Zinc protoporphyrine IX |
References
- Gottlieb, R.A. Cell death pathways in acute ischemia/reperfusion injury. J. Cardiovasc. Pharmacol. Ther. 2011, 16, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Heusch, G. Coronary microvascular obstruction: The new frontier in cardioprotection. Basic Res. Cardiol. 2019, 114, 45. [Google Scholar] [CrossRef] [PubMed]
- Heusch, G. Critical Issues for the Translation of Cardioprotection. Circ. Res. 2017, 120, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.P.; Streed, S.A. Surveillance and use of computers in hospital infection control. J. Hosp. Infect. 1989, 13, 217–229. [Google Scholar] [CrossRef]
- Przyklenk, K.; Whittaker, P. Remote ischemic preconditioning: Current knowledge, unresolved questions, and future priorities. J. Cardiovasc. Pharmacol. Ther. 2011, 16, 255–259. [Google Scholar] [CrossRef]
- Davidson, S.M.; Riquelme, J.A.; Takov, K.; Vicencio, J.M.; Boi-Doku, C.; Khoo, V.; Doreth, C.; Radenkovic, D.; Lavandero, S.; Yellon, D.M. Cardioprotection mediated by exosomes is impaired in the setting of type II diabetes but can be rescued by the use of non-diabetic exosomes in vitro. J. Cell Mol. Med. 2018, 22, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Schulz, R.; Rose, J.; Post, H.; Heusch, G. Involvement of endogenous adenosine in ischaemic preconditioning in swine. Pflugers Arch. 1995, 430, 273–282. [Google Scholar] [CrossRef]
- Lecour, S.; Suleman, N.; Deuchar, G.A.; Somers, S.; Lacerda, L.; Huisamen, B.; Opie, L.H. Pharmacological preconditioning with tumor necrosis factor-alpha activates signal transducer and activator of transcription-3 at reperfusion without involving classic prosurvival kinases (Akt and extracellular signal-regulated kinase). Circulation 2005, 112, 3911–3918. [Google Scholar] [CrossRef]
- Caricati-Neto, A.; Errante, P.R.; Menezes-Rodrigues, F.S. Recent Advances in Pharmacological and Non-Pharmacological Strategies of Cardioprotection. Int. J. Mol. Sci. 2019, 20, 4002. [Google Scholar] [CrossRef]
- Yanez-Mo, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borras, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J. Biological properties of extracellular vesicles and their physiological functions. J. Extracell Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef]
- van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Garcia-Dorado, D.; Botker, H.E.; Davidson, S.M.; Downey, J.; Engel, F.B.; Jennings, R.; Lecour, S.; Leor, J.; Madonna, R. Novel targets and future strategies for acute cardioprotection: Position Paper of the European Society of Cardiology Working Group on Cellular Biology of the Heart. Cardiovasc. Res. 2017, 113, 564–585. [Google Scholar] [CrossRef] [PubMed]
- Davidson, S.M.; Andreadou, I.; Barile, L.; Birnbaum, Y.; Cabrera-Fuentes, H.A.; Cohen, M.V.; Downey, J.M.; Girao, H.; Pagliaro, P.; Penna, C. Circulating blood cells and extracellular vesicles in acute cardioprotection. Cardiovasc. Res. 2019, 115, 1156–1166. [Google Scholar] [CrossRef] [PubMed]
- Sluijter, J.P.G.; Davidson, S.M.; Boulanger, C.M.; Buzas, E.I.; de Kleijn, D.P.V.; Engel, F.B.; Giricz, Z.; Hausenloy, D.J.; Kishore, R.; Lecour, S. Extracellular vesicles in diagnostics and therapy of the ischaemic heart: Position Paper from the Working Group on Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc. Res. 2018, 114, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Moccetti, T.; Marban, E.; Vassalli, G. Roles of exosomes in cardioprotection. Eur. Heart J. 2017, 38, 1372–1379. [Google Scholar] [CrossRef] [PubMed]
- Giricz, Z.; Varga, Z.V.; Baranyai, T.; Sipos, P.; Paloczi, K.; Kittel, A.; Buzas, E.I.; Ferdinandy, P. Cardioprotection by remote ischemic preconditioning of the rat heart is mediated by extracellular vesicles. J. Mol. Cell Cardiol. 2014, 68, 75–78. [Google Scholar] [CrossRef]
- Bartekova, M.; Jelemensky, M.; Dhalla, N.S. Emerging role of non-coding RNAs and extracellular vesicles in cardioprotection by remote ischemic conditioning of the heart. Rev. Cardiovasc. Med. 2019, 20, 59–71. [Google Scholar] [CrossRef]
- Yeang, C.; Hasanally, D.; Que, X.; Hung, M.Y.; Stamenkovic, A.; Chan, D.; Chaudhary, R.; Margulets, V.; Edel, A.L.; Hoshijima, M. Reduction of myocardial ischaemia-reperfusion injury by inactivating oxidized phospholipids. Cardiovasc. Res. 2019, 115, 179–189. [Google Scholar] [CrossRef]
- Mancek-Keber, M.; Frank-Bertoncelj, M.; Hafner-Bratkovic, I.; Smole, A.; Zorko, M.; Pirher, N.; Hayer, S.; Kralj-Iglic, V.; Rozman, B.; Ilc, N. Toll-like receptor 4 senses oxidative stress mediated by the oxidation of phospholipids in extracellular vesicles. Sci. Signal 2015, 8, ra60. [Google Scholar] [CrossRef]
- Ha, V.T.; Lainscek, D.; Gesslbauer, B.; Jarc-Jovicic, E.; Hyotylainen, T.; Ilc, N.; Lakota, K.; Tomsic, M.; van de Loo, F.A.J.; Bochkov, V. Synergy between 15-lipoxygenase and secreted PLA2 promotes inflammation by formation of TLR4 agonists from extracellular vesicles. Proc. Natl. Acad. Sci. USA 2020, 117, 25679–25689. [Google Scholar] [CrossRef]
- Chen, Z.H.; Yoshida, Y.; Saito, Y.; Noguchi, N.; Niki, E. Adaptive response induced by lipid peroxidation products in cell cultures. FEBS Lett. 2006, 580, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, N. Role of oxidative stress in adaptive responses in special reference to atherogenesis. J. Clin. Biochem. Nutr. 2008, 43, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kadl, A.; Meher, A.K.; Sharma, P.R.; Lee, M.Y.; Doran, A.C.; Johnstone, S.R.; Elliott, M.R.; Gruber, F.; Han, J.; Chen, W. Identification of a novel macrophage phenotype that develops in response to atherogenic phospholipids via Nrf2. Circ. Res. 2010, 107, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Guo, X.; Liu, X.M.; Liu, L.; Weng, Q.F.; Dong, S.J.; Knowlton, A.A.; Yuan, W.J.; Lin, L. Extracellular HSP60 induces inflammation through activating and up-regulating TLRs in cardiomyocytes. Cardiovasc. Res. 2013, 98, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Dolasia, K.; Bisht, M.K.; Pradhan, G.; Udgata, A.; Mukhopadhyay, S. TLRs/NLRs: Shaping the landscape of host immunity. Int. Rev. Immunol. 2018, 37, 3–19. [Google Scholar] [CrossRef]
- Lee, S.M.; Hutchinson, M.; Saint, D.A. The role of Toll-like receptor 4 (TLR4) in cardiac ischaemic-reperfusion injury, cardioprotection and preconditioning. Clin. Exp. Pharmacol. Physiol. 2016, 43, 864–871. [Google Scholar] [CrossRef]
- Gaspar, R.; Pipicz, M.; Hawchar, F.; Kovacs, D.; Djirackor, L.; Gorbe, A.; Varga, Z.V.; Kiricsi, M.; Petrovski, G.; Gacser, A. The cytoprotective effect of biglycan core protein involves Toll-like receptor 4 signaling in cardiomyocytes. J. Mol. Cell Cardiol. 2016, 99, 138–150. [Google Scholar] [CrossRef]
- Ha, T.; Liu, L.; Kelley, J.; Kao, R.; Williams, D.; Li, C. Toll-like receptors: New players in myocardial ischemia/reperfusion injury. Antioxid. Redox Signal 2011, 15, 1875–1893. [Google Scholar] [CrossRef]
- Li, C.; Ha, T.; Kelley, J.; Gao, X.; Qiu, Y.; Kao, R.L.; Browder, W.; Williams, D.L. Modulating Toll-like receptor mediated signaling by (1-->3)-beta-D-glucan rapidly induces cardioprotection. Cardiovasc. Res. 2004, 61, 538–547. [Google Scholar] [CrossRef]
- Gedik, N.; Maciel, L.; Schulte, C.; Skyschally, A.; Heusch, G.; Kleinbongard, P. Cardiomyocyte mitochondria as targets of humoral factors released by remote ischemic preconditioning. Arch. Med. Sci. 2017, 13, 448–458. [Google Scholar] [CrossRef]
- Thuy Tran Quang, R.H.; Rousseau, G.; Gosselin, A.A.; Schampaert, E.; Charron, T. Dual effect of pre-ischemic administration of TNF-alpha on myocardial infarct size. World J. Cardiovasc. Dis. 2013, 3, 7. [Google Scholar] [CrossRef][Green Version]
- Vicencio, J.M.; Yellon, D.M.; Sivaraman, V.; Das, D.; Boi-Doku, C.; Arjun, S.; Zheng, Y.; Riquelme, J.A.; Kearney, J.; Sharma, V. Plasma exosomes protect the myocardium from ischemia-reperfusion injury. J. Am. Coll. Cardiol. 2015, 65, 1525–1536. [Google Scholar] [CrossRef]
- Pan, J.; Alimujiang, M.; Chen, Q.; Shi, H.; Luo, X. Exosomes derived from miR-146a-modified adipose-derived stem cells attenuate acute myocardial infarction-induced myocardial damage via downregulation of early growth response factor 1. J. Cell Biochem. 2019, 120, 4433–4443. [Google Scholar] [CrossRef] [PubMed]
- Onodi, Z.; Pelyhe, C.; Terezia Nagy, C.; Brenner, G.B.; Almasi, L.; Kittel, A.; Mancek-Keber, M.; Ferdinandy, P.; Buzas, E.I.; Giricz, Z. Isolation of High-Purity Extracellular Vesicles by the Combination of Iodixanol Density Gradient Ultracentrifugation and Bind-Elute Chromatography From Blood Plasma. Front Physiol. 2018, 9, 1479. [Google Scholar] [CrossRef]
- Sodar, B.W.; Kittel, A.; Paloczi, K.; Vukman, K.V.; Osteikoetxea, X.; Szabo-Taylor, K.; Nemeth, A.; Sperlagh, B.; Baranyai, T.; Giricz, Z. Low-density lipoprotein mimics blood plasma-derived exosomes and microvesicles during isolation and detection. Sci. Rep. 2016, 6, 24316. [Google Scholar] [CrossRef]
- Baranyai, T.; Herczeg, K.; Onodi, Z.; Voszka, I.; Modos, K.; Marton, N.; Nagy, G.; Mager, I.; Wood, M.J.; El Andaloussi, S. Isolation of Exosomes from Blood Plasma: Qualitative and Quantitative Comparison of Ultracentrifugation and Size Exclusion Chromatography Methods. PLoS ONE 2015, 10, e0145686. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, J.; Wang, Q.; Zhang, Y.; Zhang, N.; Lu, L.; Wu, Y.; Zhang, Q.; Wang, W.; Wang, Y. Qishen granules inhibit myocardial inflammation injury through regulating arachidonic acid metabolism. Sci. Rep. 2016, 6, 36949. [Google Scholar] [CrossRef]
- Pan, L.L.; Liu, X.H.; Gong, Q.H.; Zhu, Y.Z. S-Propargyl-cysteine (SPRC) attenuated lipopolysaccharide-induced inflammatory response in H9c2 cells involved in a hydrogen sulfide-dependent mechanism. Amino Acids 2011, 41, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Collino, M.; Pini, A.; Mugelli, N.; Mastroianni, R.; Bani, D.; Fantozzi, R.; Papucci, L.; Fazi, M.; Masini, E. Beneficial effect of prolonged heme oxygenase 1 activation in a rat model of chronic heart failure. Dis. Model Mech. 2013, 6, 1012–1020. [Google Scholar] [CrossRef]
- Gong, Y.N.; Guy, C.; Olauson, H.; Becker, J.U.; Yang, M.; Fitzgerald, P.; Linkermann, A.; Green, D.R. ESCRT-III Acts Downstream of MLKL to Regulate Necroptotic Cell Death and Its Consequences. Cell 2017, 169, 286–300.e16. [Google Scholar] [CrossRef]
- Xie, Q.; Li, X.X.; Zhang, P.; Li, J.C.; Cheng, Y.; Feng, Y.L.; Huang, B.S.; Zhuo, Y.F.; Xu, G.H. Hydrogen gas protects against serum and glucose deprivationinduced myocardial injury in H9c2 cells through activation of the NFE2related factor 2/heme oxygenase 1 signaling pathway. Mol. Med. Rep. 2014, 10, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Zhou, Y.; Xia, Q.; Yao, L.; Chang, X. Astragaloside IV Regulates the PI3K/Akt/HO-1 Signaling Pathway and Inhibits H9c2 Cardiomyocyte Injury Induced by Hypoxia-Reoxygenation. Biol. Pharm. Bull 2019, 42, 721–727. [Google Scholar] [CrossRef]
- Yu, H.; Chen, B.; Ren, Q. Baicalin relieves hypoxia-aroused H9c2 cell apoptosis by activating Nrf2/HO-1-mediated HIF1alpha/BNIP3 pathway. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3657–3663. [Google Scholar] [CrossRef] [PubMed]
- Issan, Y.; Kornowski, R.; Aravot, D.; Shainberg, A.; Laniado-Schwartzman, M.; Sodhi, K.; Abraham, N.G.; Hochhauser, E. Heme oxygenase-1 induction improves cardiac function following myocardial ischemia by reducing oxidative stress. PLoS ONE 2014, 9, e92246. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Wilkinson, F.L.; McCarthy, E.M.; Moreno-Martinez, D.; Langford-Smith, A.; Romero, M.; Duarte, J.; Alexander, M.Y. Endothelial microparticles prevent lipid-induced endothelial damage via Akt/eNOS signaling and reduced oxidative stress. FASEB J. 2017, 31, 4636–4648. [Google Scholar] [CrossRef]
- Subramani, J.; Kundumani-Sridharan, V.; Hilgers, R.H.; Owens, C.; Das, K.C. Thioredoxin Uses a GSH-independent Route to Deglutathionylate Endothelial Nitric-oxide Synthase and Protect against Myocardial Infarction. J. Biol. Chem. 2016, 291, 23374–23389. [Google Scholar] [CrossRef]
- Zuurbier, C.J.; Abbate, A.; Cabrera-Fuentes, H.A.; Cohen, M.V.; Collino, M.; de Kleijn, D.P.V.; Downey, J.M.; Pagliaro, P.; Preissner, K.T.; Takahashi, M. Innate immunity as a target for acute cardioprotection. Cardiovasc. Res. 2019, 115, 1131–1142. [Google Scholar] [CrossRef]
- Li, X.; Heinzel, F.R.; Boengler, K.; Schulz, R.; Heusch, G. Role of connexin 43 in ischemic preconditioning does not involve intercellular communication through gap junctions. J. Mol. Cell. Cardiol. 2004, 36, 161–163. [Google Scholar] [CrossRef]
- Bencsik, P.; Paloczi, J.; Kocsis, G.F.; Pipis, J.; Belecz, I.; Varga, Z.V.; Csonka, C.; Gorbe, A.; Csont, T.; Ferdinandy, P. Moderate inhibition of myocardial matrix metalloproteinase-2 by ilomastat is cardioprotective. Pharmacol. Res. 2014, 80, 36–42. [Google Scholar] [CrossRef]

| Mouse | Forward 5′→3′ | Reverse 5′→3′ |
|---|---|---|
| Gapdh | TTCACCACCATGGAGAAGGC | GGCATGGACTGTGGTCATGA |
| Il1beta | AAGGAGAACCAAGCAACGACAAAA | TGGGGAACTCTGCAGACTCAAACT |
| Hmox1 | CAGGATTTGTCTGAGGCCTT | CATAGACTGGGTTCTGCTTGT |
| Txnrd1 | GCTGGTCTTGGATTTTGTCAC | CTTCACTGTGTCTTCGACTTTC |
| Il6 | CGGAGGCTTAATTACACATGTTC | CTGGCTTTGTCTTTCTTGTTATC |
| iNos | GCCATTGAGTTCATCAACCAGTA | CTGGTAGGTTCCTGTTGTTTCTA |
| Rat | Forward 5′→3′ | Reverse 5′→3′ |
| Gapdh | GTATTGGGCGCCTGGTCACC | CGCTCCTGGAAGATGGTGATGG |
| Tnfalpha | ACTGAACTTCGGGGTGATTG | GCTTGGTGGTTTGCTACGAC |
| Hmox1 | GATTTGTCCGAGGCCTTGAA | GTTCTGCTTGTTTCGCTCTATC |
| Il6 | TGATGGATGCTTCCAAACTG | GAGCATTGGAAGTTGGGGTA |
| Txnrd1 | GCCAAATTTGACAAGAAGGTGA | CTTTCAGAGCTTGTCCTAACAGA |
| Human | Forward 5′→3′ | Reverse 5′→3′ |
| HPRT | AGATGGTCAAGGTCGCAAG | TTCATTATAGTCAAGGGCATATCC |
| TNFA | CCTGTGAGGAGGACGAAC | CGAAGTGGTGGTCTTGTTG |
| HMOX1 | GCCCCAGGATTTGTCAGAG | CATAGATGTGGTACAGGGAG |
| IL6 | ACAGCCACTCACCTCTTC | AAGTCTCCTCATTGAATCCAG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pečan, P.; Hambalkó, S.; Ha, V.T.; Nagy, C.T.; Pelyhe, C.; Lainšček, D.; Kenyeres, B.; Brenner, G.B.; Görbe, A.; Kittel, Á.; et al. Calcium Ionophore-Induced Extracellular Vesicles Mediate Cytoprotection against Simulated Ischemia/Reperfusion Injury in Cardiomyocyte-Derived Cell Lines by Inducing Heme Oxygenase 1. Int. J. Mol. Sci. 2020, 21, 7687. https://doi.org/10.3390/ijms21207687
Pečan P, Hambalkó S, Ha VT, Nagy CT, Pelyhe C, Lainšček D, Kenyeres B, Brenner GB, Görbe A, Kittel Á, et al. Calcium Ionophore-Induced Extracellular Vesicles Mediate Cytoprotection against Simulated Ischemia/Reperfusion Injury in Cardiomyocyte-Derived Cell Lines by Inducing Heme Oxygenase 1. International Journal of Molecular Sciences. 2020; 21(20):7687. https://doi.org/10.3390/ijms21207687
Chicago/Turabian StylePečan, Peter, Szabolcs Hambalkó, Van Thai Ha, Csilla T. Nagy, Csilla Pelyhe, Duško Lainšček, Bence Kenyeres, Gábor B. Brenner, Anikó Görbe, Ágnes Kittel, and et al. 2020. "Calcium Ionophore-Induced Extracellular Vesicles Mediate Cytoprotection against Simulated Ischemia/Reperfusion Injury in Cardiomyocyte-Derived Cell Lines by Inducing Heme Oxygenase 1" International Journal of Molecular Sciences 21, no. 20: 7687. https://doi.org/10.3390/ijms21207687
APA StylePečan, P., Hambalkó, S., Ha, V. T., Nagy, C. T., Pelyhe, C., Lainšček, D., Kenyeres, B., Brenner, G. B., Görbe, A., Kittel, Á., Barteková, M., Ferdinandy, P., Manček-Keber, M., & Giricz, Z. (2020). Calcium Ionophore-Induced Extracellular Vesicles Mediate Cytoprotection against Simulated Ischemia/Reperfusion Injury in Cardiomyocyte-Derived Cell Lines by Inducing Heme Oxygenase 1. International Journal of Molecular Sciences, 21(20), 7687. https://doi.org/10.3390/ijms21207687





