Prostaglandin E2 Pathway Is Dysregulated in Gastric Adenocarcinoma in a Caucasian Population
Abstract
1. Introduction
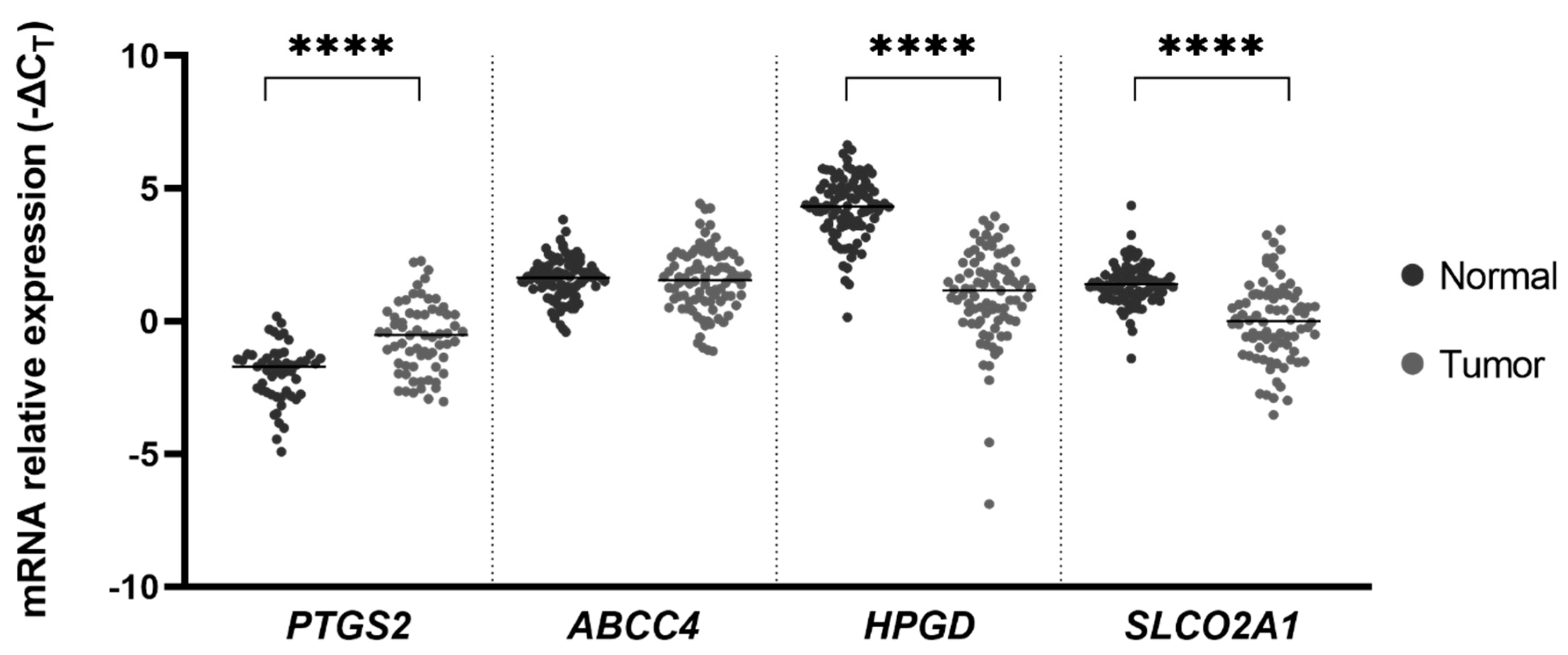
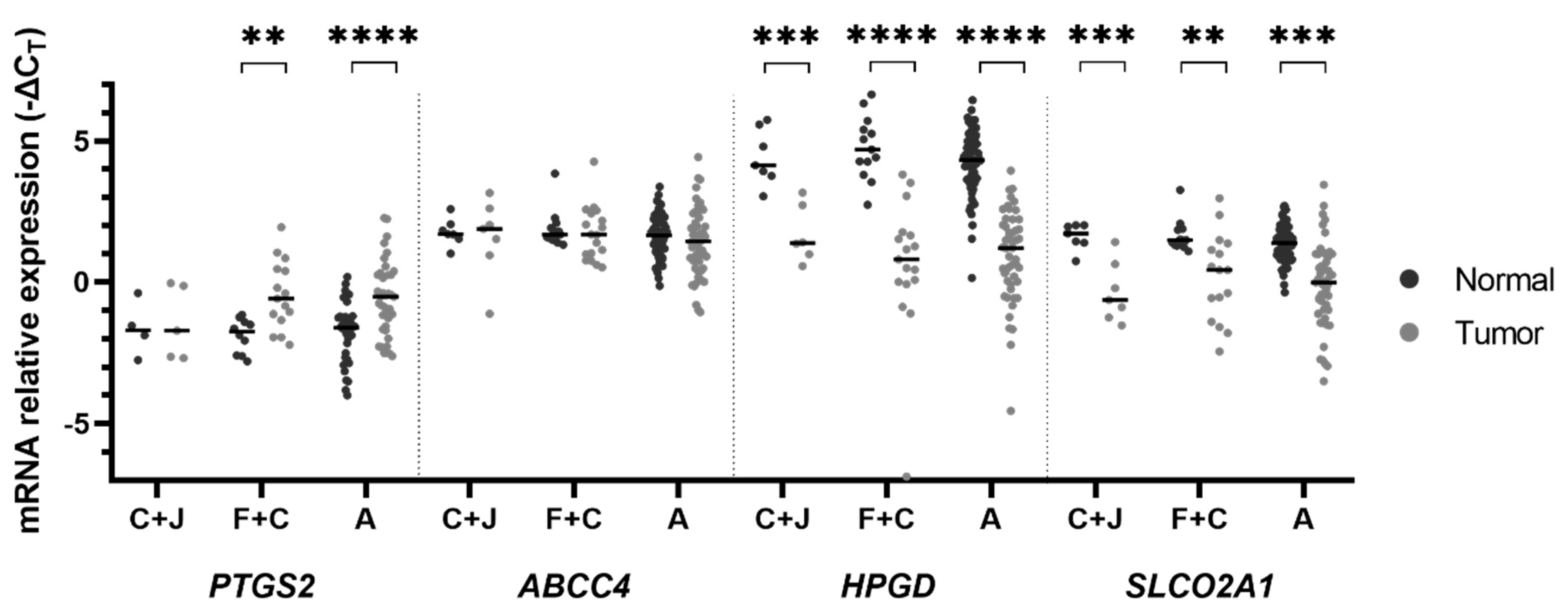
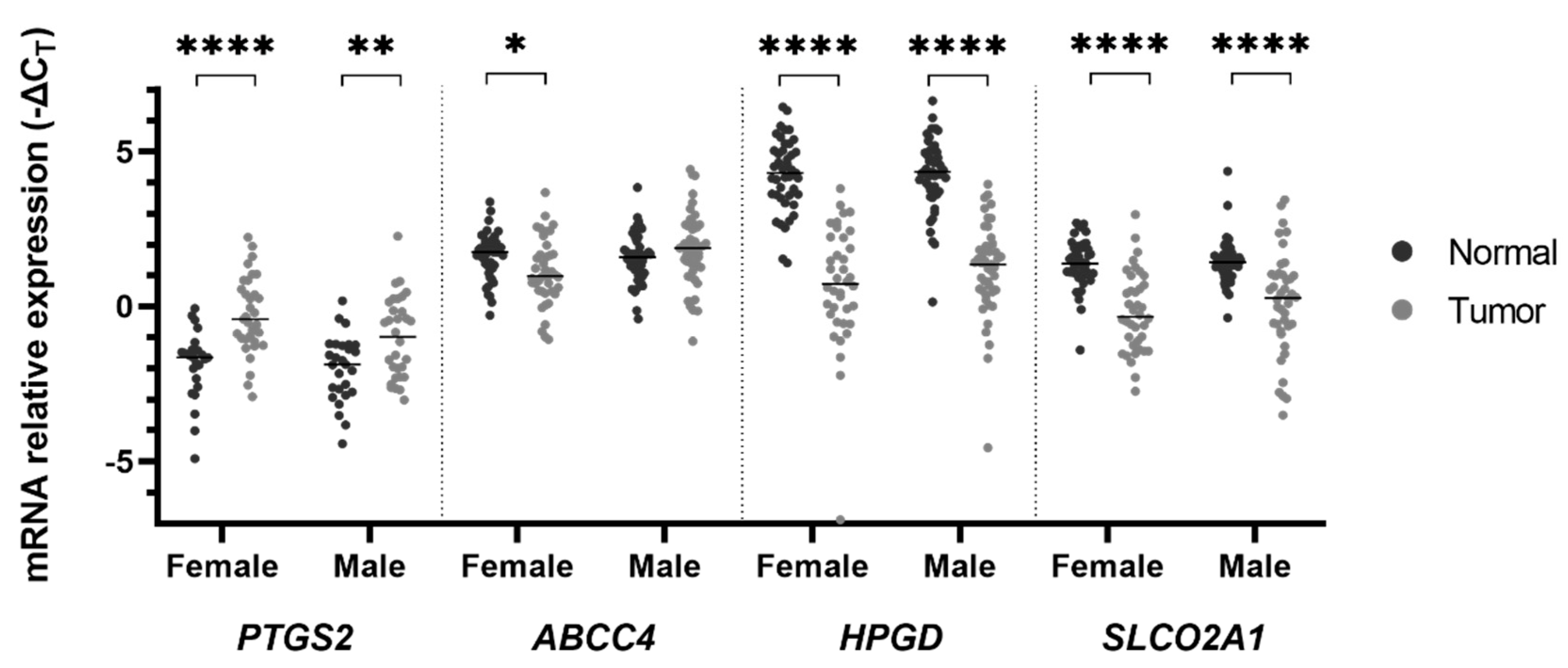
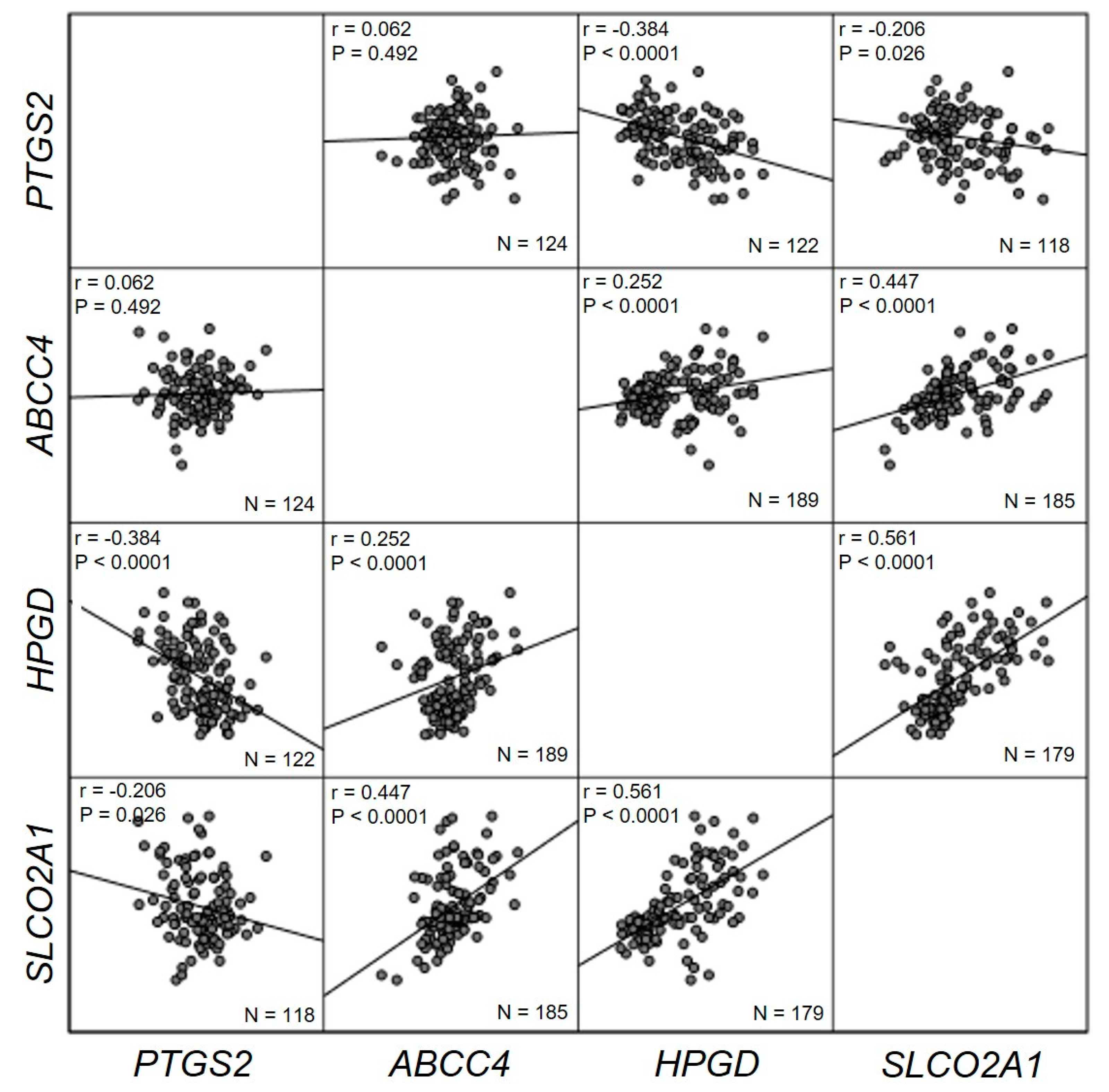
2. Results
3. Discussion
4. Materials and Methods
4.1. Patient Samples
4.2. Nucleic Acid Isolation and Quantification
4.3. Reverse Transcription Reaction
4.4. Real-Time PCR
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 15-PGDH | 15-hydroxyprostaglandin dehydrogenase |
| ABCC4 | ATP-binding cassette subfamily C member 4 |
| AJCC | American Joint Committee on Cancer |
| B2M | Beta-2-microglobulin |
| cDNA | Complementary deoxyribonucleic acid |
| COX-2 | Cyclooxygenase-2 |
| CT | Cycle threshold |
| EP | Prostaglandin E2 receptor |
| FFPE | Formalin-fixed paraffin-embedded |
| GC | Gastric cancer |
| GUSB | Glucuronidase beta |
| H&E | Hematoxylin & eosin |
| H. pylori | Helicobacter pylori |
| HPGD | Hydroxyprostaglandin dehydrogenase 15-(NAD) |
| HPRT1 | Hypoxanthine phosphoribosyltransferase 1 |
| IPO8 | Importin 8 |
| mRNA | Messenger ribonucleic acid |
| MRP4 | Multidrug resistance protein 4 |
| NSAIDs | Non-steroidal anti-inflammatory drugs |
| OD | Optical density |
| PCR | Polymerase chain reaction |
| PGE2 | Prostaglandin E2 |
| PGT | Prostaglandin transporter |
| PPIA | Peptidylprolyl isomerase A |
| PTGS2 | Prostaglandin-endoperoxide synthase 2 |
| RPL29 | Ribosomal protein L29 |
| RT | Reverse transcription |
| SD | Standard deviation |
| siRNA | Small interfering ribonucleic acid |
| SLCO2A1 | Solute carrier organic anion transporter family member 2A1 |
| SPSS | Statistical package for the social sciences |
| VEGF | Vascular endothelial growth factor |
| WHO | World Health Organization |
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Rugge, M.; Fassan, M.; Graham, D.Y. Epidemiology of Gastric Cancer. In Gastric Cancer: Principles and Practice; Strong, V.E., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 23–34. [Google Scholar] [CrossRef]
- Israel, D.A.; Peek, R.M. Chapter 63—Mechanisms of Helicobacter Pylori-Induced Gastric Inflammation. In Physiology of the Gastrointestinal Tract, 6th ed.; Said, H.M., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 1517–1545. [Google Scholar] [CrossRef]
- Nagaraju, G.P.; El-Rayes, B.F. Cyclooxygenase-2 in gastrointestinal malignancies. Cancer 2019, 125, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Tong, D.; Liu, Q.; Wang, L.-A.; Xie, Q.; Pang, J.; Huang, Y.; Wang, L.; Liu, G.; Zhang, D.; Lan, W.; et al. The roles of the COX2/PGE2/EP axis in therapeutic resistance. Cancer Metastasis Rev. 2018, 37, 355–368. [Google Scholar] [CrossRef]
- Song, J.; Su, H.; Zhou, Y.-Y.; Guo, L.-L. Cyclooxygenase-2 Expression Is Associated with Poor Overall Survival of Patients with Gastric Cancer: A Meta-Analysis. Dig. Dis. Sci. 2014, 59, 436–445. [Google Scholar] [CrossRef]
- Kasper, H.U.; Konze, E.; Dienes, H.P.; Stippel, D.L.; Schirmacher, P.; Kern, M. COX-2 expression and effects of COX-2 inhibition in colorectal carcinomas and their liver metastases. Anticancer Res. 2010, 30, 2017–2023. [Google Scholar] [PubMed]
- Song, H.J.; Myung, S.J.; Kim, I.W.; Jeong, J.Y.; Park, Y.S.; Lee, S.M.; Nam, W.H.; Ryu, Y.M.; Fink, S.P.; Yang, D.H.; et al. 15-hydroxyprostaglandin dehydrogenase is downregulated and exhibits tumor suppressor activity in gastric cancer. Cancer Investig. 2011, 29, 257–265. [Google Scholar] [CrossRef]
- Wolf, I.; O’Kelly, J.; Rubinek, T.; Tong, M.; Nguyen, A.; Lin, B.T.; Tai, H.-H.; Karlan, B.Y.; Koeffler, H.P. 15-Hydroxyprostaglandin Dehydrogenase Is a Tumor Suppressor of Human Breast Cancer. Cancer Res. 2006, 66, 7818–7823. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.L.; Kench, J.G.; Handelsman, D.J.; Scheffer, G.L.; Stricker, P.D.; Grygiel, J.G.; Sutherland, R.L.; Henshall, S.M.; Allen, J.D.; Horvath, L.G. Androgen regulation of multidrug resistance-associated protein 4 (MRP4/ABCC4) in prostate cancer. Prostate 2008, 68, 1421–1429. [Google Scholar] [CrossRef]
- Wlcek, K.; Svoboda, M.; Sellner, F.; Krupitza, G.; Jaeger, W.; Thalhammer, T. Altered expression of organic anion transporter polypeptide (OATP) genes in human breast carcinoma. Cancer Biol. Ther. 2008, 7, 1450–1455. [Google Scholar] [CrossRef] [PubMed]
- Hays, A.; Apte, U.; Hagenbuch, B. Organic Anion Transporting Polypeptides Expressed in Pancreatic Cancer May Serve As Potential Diagnostic Markers and Therapeutic Targets for Early Stage Adenocarcinomas. Pharm. Res. 2013, 30, 2260–2269. [Google Scholar] [CrossRef]
- Kochel, T.J.; Goloubeva, O.G.; Fulton, A.M. Upregulation of Cyclooxygenase-2/Prostaglandin E2 (COX-2/PGE2) Pathway Member Multiple Drug Resistance-Associated Protein 4 (MRP4) and Downregulation of Prostaglandin Transporter (PGT) and 15-Prostaglandin Dehydrogenase (15-PGDH) in Triple-Negative Breast Cancer. Breast Cancer Basic Clin. Res. 2016, 10, 61–70. [Google Scholar] [CrossRef]
- Park, S.R. Management of gastric cancer: East vs West. Curr. Probl. Cancer 2015, 39, 315–341. [Google Scholar] [CrossRef]
- Zmigrodzka, M.; Rzepecka, A.; Krzyzowska, M.; Witkowska-Pilaszewicz, O.; Cywinska, A.; Winnicka, A. The cyclooxygenase-2/prostaglandin E2 pathway and its role in the pathogenesis of human and dog hematological malignancies. J. Physiol. Pharmacol. 2018, 69, 653–661. [Google Scholar] [CrossRef]
- Goradel, N.H.; Najafi, M.; Salehi, E.; Farhood, B.; Mortezaee, K. Cyclooxygenase-2 in cancer: A review. J. Cell. Phisiol. 2019, 234, 5683–5699. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Omori, K.; Murata, T. Role of prostaglandins in tumor microenvironment. Cancer Metastasis Rev. 2018, 37, 347–354. [Google Scholar] [CrossRef]
- Greenhough, A.; Smartt, H.J.M.; Moore, A.E.; Roberts, H.R.; Williams, A.C.; Paraskeva, C.; Kaidi, A. The COX-2/PGE2 pathway: Key roles in the hallmarks of cancer and adaptation to the tumour microenvironment. Carcinogenesis 2009, 30, 377–386. [Google Scholar] [CrossRef]
- Karpisheh, V.; Nikkhoo, A.; Hojjat-Farsangi, M.; Namdar, A.; Azizi, G.; Ghalamfarsa, G.; Sabz, G.; Yousefi, M.; Yousefi, B.; Jadidi-Niaragh, F. Prostaglandin E2 as a potent therapeutic target for treatment of colon cancer. Prostaglandins Other Lipid Mediat. 2019, 144, 106338. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, M.; Rosenberg, D.W. Multifaceted roles of PGE2 in inflammation and cancer. Semin. Immunopathol. 2013, 35, 123–137. [Google Scholar] [CrossRef]
- Thorat, M.A.; Cuzick, J. Role of Aspirin in Cancer Prevention. Curr. Oncol. Rep. 2013, 15, 533–540. [Google Scholar] [CrossRef]
- Lin, S.J.; Gagnon-Bartsch, J.A.; Tan, I.B.; Earle, S.; Ruff, L.; Pettinger, K.; Ylstra, B.; van Grieken, N.; Rha, S.Y.; Chung, H.C.; et al. Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas. Gut 2015, 64, 1721–1731. [Google Scholar] [CrossRef]
- Zhao, X.; Guo, Y.; Yue, W.; Zhang, L.; Gu, M.; Wang, Y. ABCC4 is required for cell proliferation and tumorigenesis in non-small cell lung cancer. OncoTargets Ther. 2014, 7, 343–351. [Google Scholar] [CrossRef]
- Borel, F.; Han, R.; Visser, A.; Petry, H.; van Deventer, S.J.H.; Jansen, P.L.M.; Konstantinova, P.; Collaboration of the Réseau Centre de Ressources Biologiques Foie. Adenosine triphosphate-binding cassette transporter genes up-regulation in untreated hepatocellular carcinoma is mediated by cellular microRNAs. Hepatology 2012, 55, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.M.; Slitt, A.L.; Cherrington, N.J.; Cheng, X.; Klaassen, C.D. Tissue Distribution and Hepatic and Renal Ontogeny of the Multidrug Resistance-Associated Protein (MRP) Family in Mice. Drug Metab. Dispos. 2005, 33, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.M.; Cheng, X.; Tanaka, Y.; Scheffer, G.L.; Klaassen, C.D. Hormonal regulation of renal multidrug resistance-associated proteins 3 and 4 (Mrp3 and Mrp4) in mice. Biochem. Pharmacol. 2006, 71, 1470–1478. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, T.; Ohta, T.; Oyama, K.; Miyashita, T.; Miwa, K. Cyclooxygenase-2 (COX-2) in carcinogenesis and selective COX-2 inhibitors for chemoprevention in gastrointestinal cancers. J. Gastrointest. Cancer 2007, 38, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Da, M.-X.; Wu, X.-T.; Wang, J.; Guo, T.-K.; Zhao, Z.-G.; Luo, T.; Zhang, M.-M.; Qian, K. Expression of cyclooxygenase-2 and vascular endothelial growth factor-C correlates with lymphangiogenesis and lymphatic invasion in human gastric cancer. Arch. Med Res. 2008, 39, 92–99. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, K.-F.; Zhang, L.; Ma, J.-L.; Zhou, T.; Li, J.-Y.; Shen, L.; You, W.-C. Helicobacter pylori, cyclooxygenase-2 and evolution of gastric lesions: Results from an intervention trial in China. Carcinogenesis 2015, 36, 1572–1579. [Google Scholar] [CrossRef]
- Gou, H.-F.; Chen, X.-C.; Zhu, J.; Jiang, M.; Yang, Y.; Cao, D.; Hou, M. Expressions of COX-2 and VEGF-C in gastric cancer: Correlations with lymphangiogenesis and prognostic implications. J. Exp. Clin. Cancer Res. 2011, 30, 14. [Google Scholar] [CrossRef]
- Zhao, J.; Wen, S.; Wang, X.; Zhang, Z. Helicobacter pylori modulates cyclooxygenase-2 and 15-hydroxy prostaglandin dehydrogenase in gastric cancer. Oncol. Lett. 2017, 14, 5519–5525. [Google Scholar] [CrossRef][Green Version]
- Song, L.; Guo, Y.; Xu, B. Expressions of ras homolog gene family, member A (RhoA) and cyclooxygenase-2 (COX-2) proteins in early gastric cancer and their role in the development of gastric cancer. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 2979–2984. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; Wang, M. Prognostic significance of expression of cysteine-rich 61 and cyclooxygenase-2 in gastric cancer. BMC Gastroenterol. 2016, 16, 74. [Google Scholar] [CrossRef] [PubMed]
- Jang, T.J. Expression of proteins related to prostaglandin E2 biosynthesis is increased in human gastric cancer and during gastric carcinogenesis. Virchows Arch. 2004, 445, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.Y.; Joo, H.J.; Choi, J.H.; Yi, J.W.; Yang, M.S.; Cho, D.Y.; Kim, H.S.; Nam, D.K.; Lee, K.B.; Kim, H.C. Increased expression of cyclooxygenase-2 protein in human gastric carcinoma. Clin. Cancer Res. 2000, 6, 519–525. [Google Scholar] [PubMed]
- Sung, J.J.Y.; Leung, W.K.; Go, M.Y.Y.; To, K.F.; Cheng, A.S.L.; Ng, E.K.W.; Chan, F.K.L. Cyclooxygenase-2 expression in Helicobacter pylori-associated premalignant and malignant gastric lesions. Am. J. Pathol. 2000, 157, 729–735. [Google Scholar] [CrossRef]
- McCarthy, C.J.; Crofford, L.J.; Greenson, J.; Scheiman, J.M. Cyclooxygenase-2 expression in gastric antral mucosa before and after eradication of Helicobacter pylori infection. Am. J. Gastroenterol. 1999, 94, 1218–1223. [Google Scholar] [CrossRef] [PubMed]
- Ben Ayed-Guerfali, D.; Charfi, S.; Khabir, A.; Sellami-Boudawara, T.; Gargouri, A.; Mokdad-Gargouri, R. Clinical and prognosis relevance of COX-2 expression in Tunisian patients with primary gastric adenocarcinoma. Cancer Biomark. 2016, 17, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, P.; Bateman, A.C.; Mehta, R.L.; Patel, P. Mucosal expression of cyclooxygenase isoforms 1 and 2 is increased with worsening damage to the gastric mucosa. Histopathology 2005, 46, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Mrena, J.; Wiksten, J.-P.; Kokkola, A.; Nordling, S.; Ristimäki, A.; Haglund, C. COX-2 is associated with proliferation and apoptosis markers and serves as an independent prognostic factor in gastric cancer. Tumor Biol. 2010, 31, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Saukkonen, K.; Nieminen, O.; van Rees, B.; Vilkki, S.; Härkönen, M.; Juhola, M.; Mecklin, J.-P.; Sipponen, P.; Ristimäki, A. Expression of Cyclooxygenase-2 in Dysplasia of the Stomach and in Intestinal-type Gastric Adenocarcinoma. Clin. Cancer Res. 2001, 7, 1923–1931. [Google Scholar]
- Nardone, G.; Rocco, A.; Vaira, D.; Staibano, S.; Budillon, A.; Tatangelo, F.; Sciulli, M.G.; Perna, F.; Salvatore, G.; Di Benedetto, M.; et al. Expression of COX-2, mPGE-synthase1, MDR-1 (P-gp), and Bcl-xL: A molecular pathway of H. pylori-related gastric carcinogenesis. J. Pathol. 2004, 202, 305–312. [Google Scholar] [CrossRef]
- Hu, M.; Li, K.; Maskey, N.; Xu, Z.; Peng, C.; Tian, S.; Li, Y.; Yang, G. 15-PGDH expression as a predictive factor response to neoadjuvant chemotherapy in advanced gastric cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 6910–6918. [Google Scholar]
- Liu, Z.; Wang, X.; Lu, Y.; Du, R.; Luo, G.; Wang, J.; Zhai, H.; Zhang, F.; Wen, Q.; Wu, K.; et al. 15-Hydroxyprostaglandin dehydrogenase is a tumor suppressor of human gastric cancer. Cancer Biol. Ther. 2010, 10, 780–787. [Google Scholar] [CrossRef]
- Jang, T.J.; Ji, Y.S.; Jung, K.H. Decreased expression of 15-hydroxyprostaglandin dehydrogenase in gastric carcinomas. Yonsei Med. J. 2008, 49, 917–922. [Google Scholar] [CrossRef]
- Lou, L.-H.; Jing, D.-D.; Lai, Y.-X.; Lu, Y.-Y.; Li, J.-K.; Wu, K. 15-PGDH is reduced and induces apoptosis and cell cycle arrest in gastric carcinoma. World J. Gastroenterol. 2012, 18, 1028–1037. [Google Scholar] [CrossRef]
- Seo, S.H.; Kang, M.S.; Kim, K.H.; An, M.S.; Ha, T.K.; Bae, K.B.; Oh, M.K.; Choi, C.S.; Oh, S.H.; Choi, Y.K. Correlation of 15-prostagladin dehydrogenase expression with clinicopathological factors and survival rate in gastric adenocarcinoma. Int. J. Surg. 2015, 13, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Thiel, A.; Ganesan, A.; Mrena, J.; Junnila, S.; Nykänen, A.; Hemmes, A.; Tai, H.-H.; Monni, O.; Kokkola, A.; Haglund, C.; et al. 15-Hydroxyprostaglandin dehydrogenase is down-regulated in gastric cancer. Clin. Cancer Res. 2009, 15, 4572–4580. [Google Scholar] [CrossRef]
- Tatsuwaki, H.; Tanigawa, T.; Watanabe, T.; Machida, H.; Okazaki, H.; Yamagami, H.; Shiba, M.; Watanabe, K.; Tominaga, K.; Fujiwara, Y.; et al. Reduction of 15-hydroxyprostaglandin dehydrogenase expression is an independent predictor of poor survival associated with enhanced cell proliferation in gastric adenocarcinoma. Cancer Sci. 2010, 101, 550–558. [Google Scholar] [CrossRef]
- Ryu, Y.-M.; Myung, S.-J.; Park, Y.S.; Yang, D.-H.; Song, H.J.; Jeong, J.-Y.; Lee, S.M.; Song, M.; Kim, D.H.; Lee, H.-J.; et al. Inhibition of 15-hydroxyprostaglandin dehydrogenase by Helicobacter pylori in human gastric carcinogenesis. Cancer Prev. Res. 2013. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Tamai, I. Roles of Organic Anion Transporting Polypeptide 2A1 (OATP2A1/SLCO2A1) in Regulating the Pathophysiological Actions of Prostaglandins. AAPS J. 2018, 20, 13. [Google Scholar] [CrossRef] [PubMed]
- Bujok, K.; Glaeser, H.; Schuh, W.; Rau, T.T.; Schmidt, I.; Fromm, M.F.; Mandery, K. Interplay between the prostaglandin transporter OATP2A1 and prostaglandin E2-mediated cellular effects. Cell. Signal. 2015, 27, 663–672. [Google Scholar] [CrossRef]
- Takeda, S.; Tanigawa, T.; Watanabe, T.; Tatsuwaki, H.; Nadatani, Y.; Otani, K.; Nagami, Y.; Tanaka, F.; Kamata, N.; Yamagami, H.; et al. Reduction of prostaglandin transporter predicts poor prognosis associated with angiogenesis in gastric adenocarcinoma. J. Gastroenterol. Hepatol. 2016, 31, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Ohno, Y.; Aotani, R.; Maruyama, S.; Shimada, H.; Kamo, S.; Oshima, H.; Oshima, M.; Schuetz, J.D.; Tamai, I. A novel role for OATP2A1/SLCO2A1 in a murine model of colon cancer. Sci. Rep. 2017, 7, 16567. [Google Scholar] [CrossRef]
- Buxhofer-Ausch, V.; Secky, L.; Wlcek, K.; Svoboda, M.; Kounnis, V.; Briasoulis, E.; Tzakos, A.G.; Jaeger, W.; Thalhammer, T. Tumor-specific expression of organic anion-transporting polypeptides: Transporters as novel targets for cancer therapy. J. Drug Deliv. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, N.; Lockhart, A.C.; Lee, W. Role of organic anion-transporting polypeptides (oatps) in cancer therapy. AAPS J. 2015, 17, 535–545. [Google Scholar] [CrossRef]
- Aran, D.; Camarda, R.; Odegaard, J.; Paik, H.; Oskotsky, B.; Krings, G.; Goga, A.; Sirota, M.; Butte, A.J. Comprehensive analysis of normal adjacent to tumor transcriptomes. Nat. Commun. 2017, 8, 1077. [Google Scholar] [CrossRef]
- Russi, S.; Calice, G.; Ruggieri, V.; Laurino, S.; La Rocca, F.; Amendola, E.; Lapadula, C.; Compare, D.; Nardone, G.; Musto, P.; et al. Gastric normal adjacent mucosa versus healthy and cancer tissues: Distinctive transcriptomic profiles and biological features. Cancers 2019, 11, 1248. [Google Scholar] [CrossRef] [PubMed]
- Areia, M.; Spaander, M.C.; Kuipers, E.J.; Dinis-Ribeiro, M. Endoscopic screening for gastric cancer: A cost-utility analysis for countries with an intermediate gastric cancer risk. United Eur. Gastroenterol. J. 2018, 6, 192–202. [Google Scholar] [CrossRef]
- Amin, M.B.; Edge, S.B.; Greene, F.L.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. AJCC Cancer Staging Manual; Springer: Cham, Switzerland, 2018; p. 1032. [Google Scholar] [CrossRef]
- Livak, K.; Schmittgen, T. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta (T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
| Cases | |
|---|---|
| Demographics (n = 121) | |
| Age (years) | |
| Mean ± sd | 69.49 ± 0.91 |
| Median (min-max) | 70 (50–89) |
| Sex, n (%) | |
| Male | 60 (50) |
| Female | 61 (50) |
| Tumor characteristics (n = 89) | |
| Tumor location, n (%) | |
| Cardia and GEJ | 7 (8) |
| Fundus and corpus | 18 (20) |
| Antrum and corpus-antrum transition | 53 (60) |
| Angularis incisura | 3 (3) |
| Others * | 8 (9) |
| Grade, n (%) | |
| Well-differentiated | 11 (13) |
| Moderately differentiated | 56 (63) |
| Poorly differentiated | 19 (21) |
| Cannot be assessed | 3 (3) |
| Stage, n (%) | |
| I-II | 44 (49) |
| III-IV | 45 (51) |
| Synchronous tumors, n (%) | |
| Yes | 7 (8) |
| No | 82 (92) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, C.; Pereira, C.; Farinha, M.; Medeiros, R.; Dinis-Ribeiro, M. Prostaglandin E2 Pathway Is Dysregulated in Gastric Adenocarcinoma in a Caucasian Population. Int. J. Mol. Sci. 2020, 21, 7680. https://doi.org/10.3390/ijms21207680
Lopes C, Pereira C, Farinha M, Medeiros R, Dinis-Ribeiro M. Prostaglandin E2 Pathway Is Dysregulated in Gastric Adenocarcinoma in a Caucasian Population. International Journal of Molecular Sciences. 2020; 21(20):7680. https://doi.org/10.3390/ijms21207680
Chicago/Turabian StyleLopes, Catarina, Carina Pereira, Mónica Farinha, Rui Medeiros, and Mário Dinis-Ribeiro. 2020. "Prostaglandin E2 Pathway Is Dysregulated in Gastric Adenocarcinoma in a Caucasian Population" International Journal of Molecular Sciences 21, no. 20: 7680. https://doi.org/10.3390/ijms21207680
APA StyleLopes, C., Pereira, C., Farinha, M., Medeiros, R., & Dinis-Ribeiro, M. (2020). Prostaglandin E2 Pathway Is Dysregulated in Gastric Adenocarcinoma in a Caucasian Population. International Journal of Molecular Sciences, 21(20), 7680. https://doi.org/10.3390/ijms21207680






