Correct Identification of Cell of Origin May Explain Many Aspects of Cancer: The Role of Neuroendocrine Cells as Exemplified from the Stomach
Abstract
1. Highlights
2. Introduction
3. Causes of Mutations
4. Genes of Importance in Neoplasia
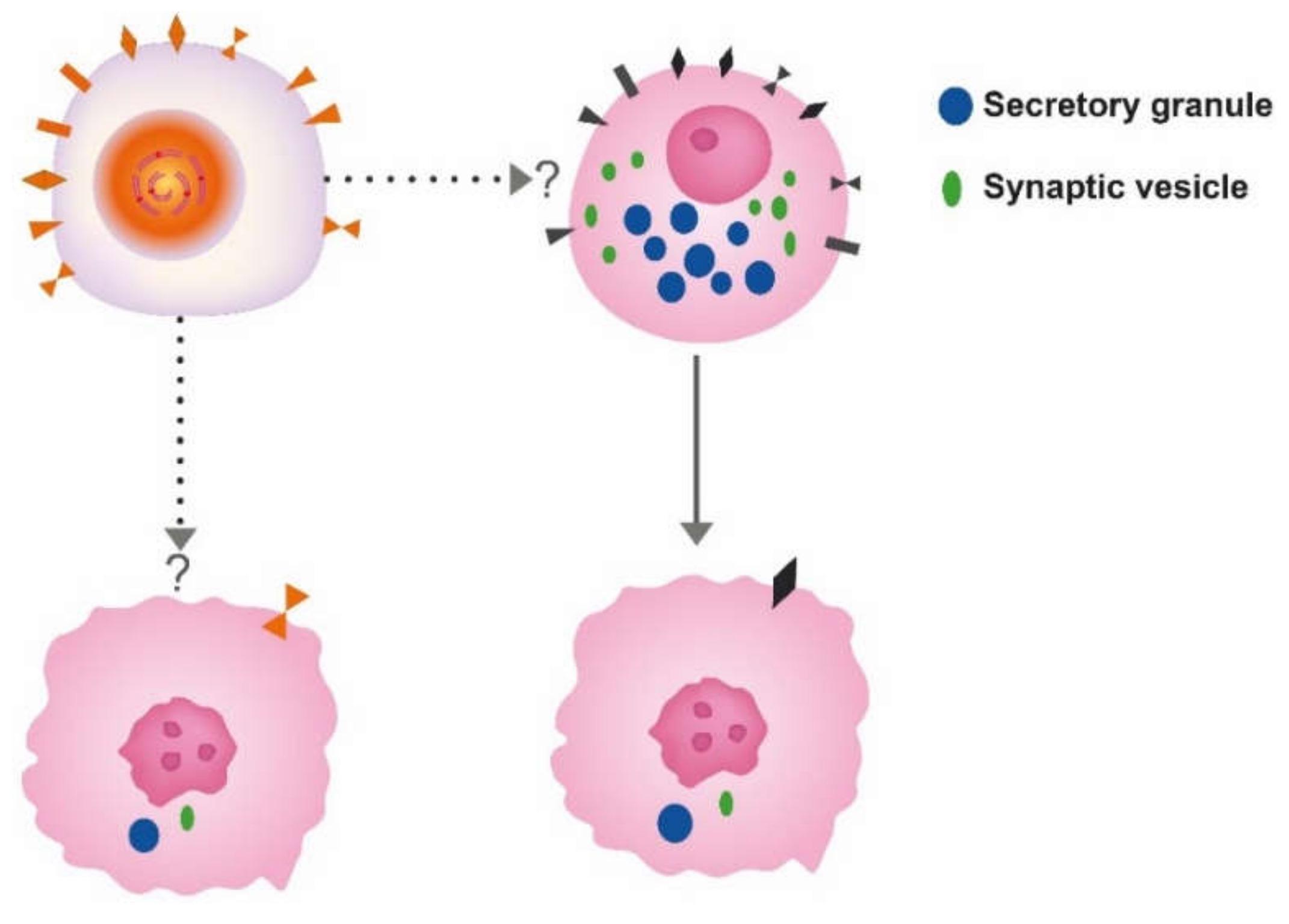
5. Cell of Origin
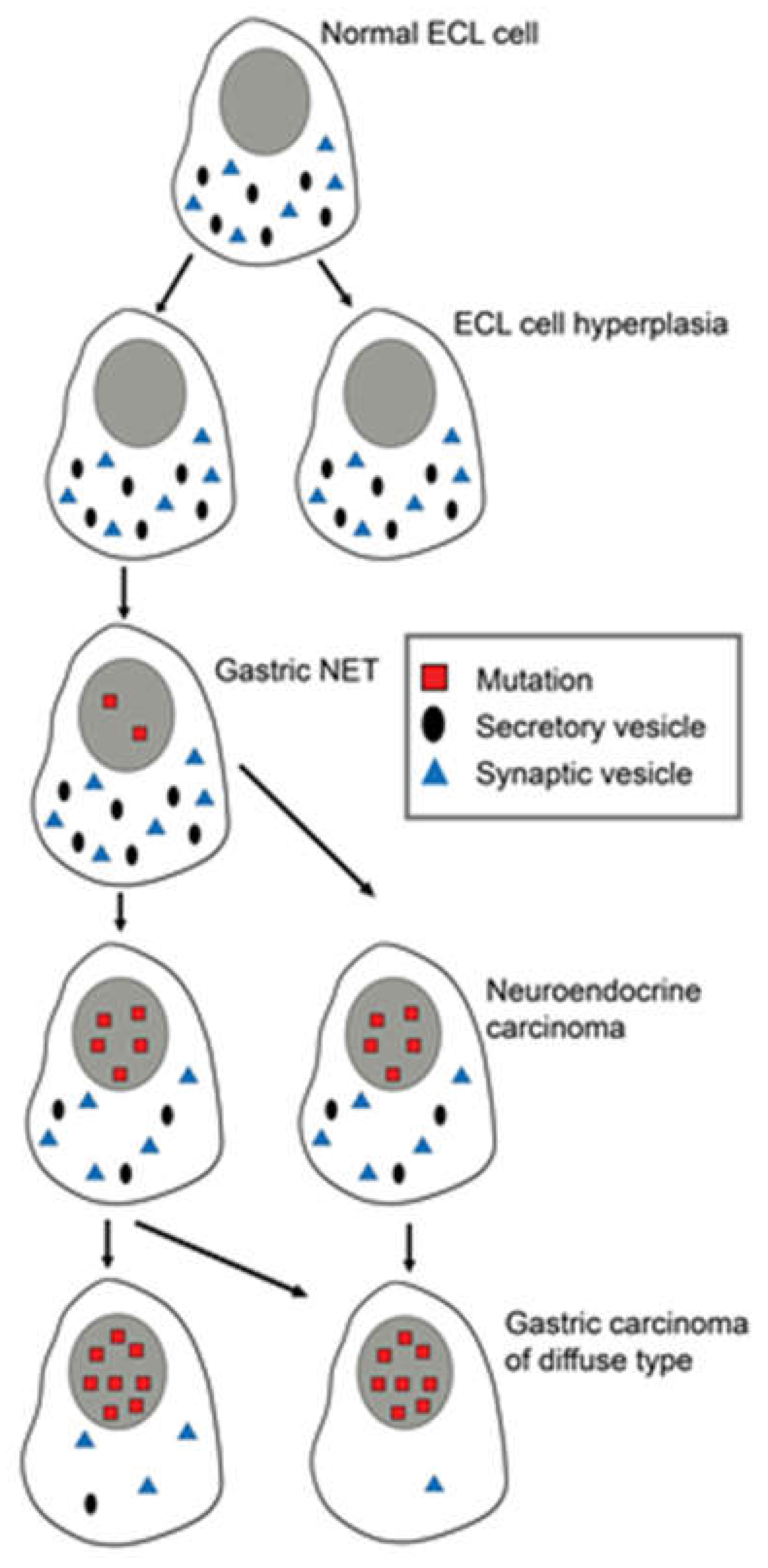
6. Gastric Cancer
7. Hormonal Carcinogenesis in General
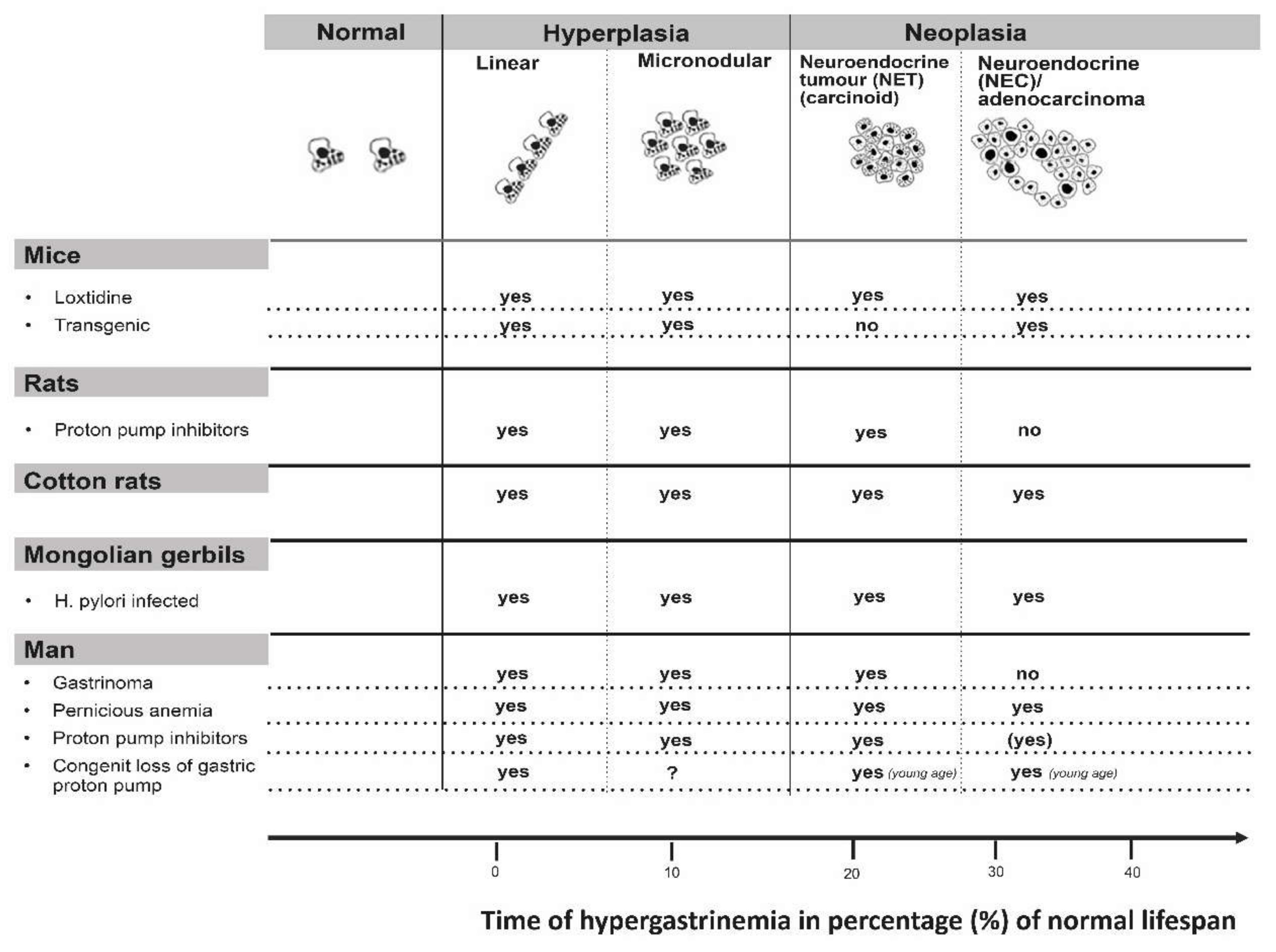
8. The Role of Neuroendocrine (NE) Cells in Carcinogenesis
9. Tumor Dormancy
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Antonelli, A.; Furlan, M.; Sodano, M.; Cindolo, L.; Belotti, S.; Tardanico, R.; Cozzoli, A.; Zanotelli, T.; Simeone, C. Features, risk factors and clinical outcome of “very late” recurrences after surgery for localized renal carcinoma: A retrospective evaluation of a cohort with a minimum of 10 years of follow up. Int. J. Urol. 2016, 23, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Ashina, K.; Tsubosaka, Y.; Nakamura, T.; Omori, K.; Kobayashi, K.; Hori, M.; Ozaki, H.; Murata, T. Histamine Induces Vascular Hyperpermeability by Increasing Blood Flow and Endothelial Barrier Disruption In Vivo. PLoS ONE 2015, 10, e0132367. [Google Scholar] [CrossRef] [PubMed]
- Azzopardi, J.G.; Pollock, D.J. Argentaffin and Argyrophil Cells in Gastric Carcinoma. J. Pathol. Bacteriol. 1963, 86, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Bakke, I.; Qvigstad, G.; Brenna, E.; Sandvik, A.K.; Waldum, H.L. Gastrin has a specific proliferative effect on the rat enterochromaffin-like cell, but not on the parietal cell: A study by elutriation centrifugation. Acta Physiol. Scand. 2000, 169, 29–37. [Google Scholar] [CrossRef]
- Bakke, I.; Qvigstad, G.; Sandvik, A.K.; Waldum, H.L. The CCK-2 receptor is located on the ECL cell, but not on the parietal cell. Scand. J. Gastroenterol. 2001, 36, 1128–1133. [Google Scholar] [CrossRef]
- Bakkelund, K.; Fossmark, R.; Nordrum, I.; Waldum, H. Signet ring cells in gastric carcinomas are derived from neuroendocrine cells. J. Histochem. Cytochem. 2006, 54, 615–621. [Google Scholar] [CrossRef]
- Barrett, P.; Hobbs, R.C.; Coates, P.J.; Risdon, R.A.; Wright, N.A.; Hall, P.A. Endocrine cells of the human gastrointestinal tract have no proliferative capacity. Histochem. J. 1995, 27, 482–486. [Google Scholar] [CrossRef]
- Bergquist, J.R.; Leiting, J.L.; Habermann, E.B.; Cleary, S.P.; Kendrick, M.L.; Smoot, R.L.; Nagorney, D.M.; Truty, M.J.; Grotz, T.E. Early-onset gastric cancer is a distinct disease with worrisome trends and oncogenic features. Surgery 2019, 166, 547–555. [Google Scholar] [CrossRef]
- Blair, A.J., 3rd; Richardson, C.T.; Walsh, J.H.; Feldman, M. Variable contribution of gastrin to gastric acid secretion after a meal in humans. Gastroenterology 1987, 92, 944–949. [Google Scholar] [CrossRef]
- Bliss, P.W.; Healey, Z.V.; Arebi, N.; Calam, J. Nalpha-methyl histamine and histamine stimulate gastrin release from rabbit G-cells via histamine H2-receptors. Aliment. Pharmacol. Ther. 1999, 13, 1669–1674. [Google Scholar] [CrossRef]
- Blokzijl, F.; de Ligt, J.; Jager, M.; Sasselli, V.; Roerink, S.; Sasaki, N.; Huch, M.; Boymans, S.; Kuijk, E.; Prins, P.; et al. Tissue-specific mutation accumulation in human adult stem cells during life. Nature 2016, 538, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Borch, K.; Renvall, H.; Liedberg, G. Gastric endocrine cell hyperplasia and carcinoid tumors in pernicious anemia. Gastroenterology 1985, 88, 638–648. [Google Scholar] [CrossRef]
- Boyce, M.; Moore, A.R.; Sagatun, L.; Parsons, B.N.; Varro, A.; Campbell, F.; Fossmark, R.; Waldum, H.L.; Pritchard, D.M. Netazepide, a gastrin/cholecystokinin-2 receptor antagonist, can eradicate gastric neuroendocrine tumours in patients with autoimmune chronic atrophic gastritis. Br. J. Clin. Pharmacol 2017, 83, 466–475. [Google Scholar] [CrossRef]
- Brenna, E.; Swarts, H.G.; Klaassen, C.H.; de Pont, J.J.; Waldum, H.L. Evaluation of the trophic effect of longterm treatment with the histamine H2 receptor antagonist loxtidine on rat oxyntic mucosa by differential counting of dispersed cells. Gut 1994, 35, 1547–1550. [Google Scholar] [CrossRef] [PubMed]
- Brenna, E.; Waldum, H.L.; Sandvik, A.K.; Sognen, B.S.; Kristensen, A. Effects on the rat oxyntic mucosa of the histamine2-antagonist loxtidine and the H+, K(+)-ATPase inhibitor omeprazole. Aliment. Pharmacol. Ther. 1992, 6, 335–349. [Google Scholar] [CrossRef]
- Calvete, O.; Reyes, J.; Zuniga, S.; Paumard-Hernandez, B.; Fernandez, V.; Bujanda, L.; Rodriguez-Pinilla, M.S.; Palacios, J.; Heine-Suner, D.; Banka, S.; et al. Exome sequencing identifies ATP4A gene as responsible of an atypical familial type I gastric neuroendocrine tumour. Hum. Mol. Genet. 2015, 24, 2914–2922. [Google Scholar] [CrossRef]
- Capp, J.P. Cancer Stem Cells: From Historical Roots to a New Perspective. J. Oncol. 2019, 5189232. [Google Scholar] [CrossRef]
- Cheung, K.S.; Chan, E.W.; Wong, A.Y.S.; Chen, L.; Wong, I.C.K.; Leung, W.K. Long-term proton pump inhibitors and risk of gastric cancer development after treatment for Helicobacter pylori: A population-based study. Gut 2018, 67, 28–35. [Google Scholar] [CrossRef]
- Conforti, F.; Pala, L.; Pagan, E.; Viale, G.; Bagnardi, V.; Peruzzotti, G.; de Pas, T.; Bianco, N.; Graffeo, R.; Rocco, E.G.; et al. Endocrine-responsive lobular carcinoma of the breast: Features associated with risk of late distant recurrence. Breast Cancer Res. 2019, 21, 153. [Google Scholar] [CrossRef]
- Cunningham, M.L. Role of increased DNA replication in the carcinogenic risk of nonmutagenic chemical carcinogens. Mutat. Res. 1996, 365, 59–69. [Google Scholar] [CrossRef]
- Davis, J.E., Jr.; Kirk, J.; Ji, Y.; Tang, D.G. Tumor Dormancy and Slow-Cycling Cancer Cells. Adv. Exp. Med. Biol. 2019, 1164, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Endo, H.; Inoue, M. Dormancy in cancer. Cancer Sci. 2019, 110, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Fiebiger, W.; Penz, M.; Längle, F.; Kurtaran, A.; Oberhuber, G.; Raderer, M. Malignancy in slow motion: Diagnosis of biochemically apparent, but otherwise occult persistent disease 21 years after resection of a carcinoid tumour of the terminal ileum. Scand. J. Gastroenterol. 2001, 36, 1116–1118. [Google Scholar] [CrossRef] [PubMed]
- Fossmark, R.; Sordal, O.; Jianu, C.S.; Qvigstad, G.; Nordrum, I.S.; Boyce, M.; Waldum, H.L. Treatment of gastric carcinoids type 1 with the gastrin receptor antagonist netazepide (YF476) results in regression of tumours and normalisation of serum chromogranin A. Aliment. Pharmacol. Ther. 2012, 36, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Friberg, S.; Nyström, A. Cancer Metastases: Early Dissemination and Late Recurrences. Cancer Growth Metastasis 2015, 8, 43–49. [Google Scholar] [CrossRef]
- Gao, F.; Zafar, M.I.; Jüttner, S.; Höcker, M.; Wiedenmann, B. Expression and Molecular Regulation of the Cox2 Gene in Gastroenteropancreatic Neuroendocrine Tumors and Antiproliferation of Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Med. Sci. Monit. 2018, 24, 8125–8140. [Google Scholar] [CrossRef]
- Guilford, P.J.; Hopkins, J.B.; Grady, W.M.; Markowitz, S.D.; Willis, J.; Lynch, H.; Rajput, A.; Wiesner, G.L.; Lindor, N.M.; Burgart, L.J.; et al. E-cadherin germline mutations define an inherited cancer syndrome dominated by diffuse gastric cancer. Hum. Mutat. 1999, 14, 249–255. [Google Scholar] [CrossRef]
- Hakanson, R.; Oscarson, J.; Sundler, F. Gastrin and the trophic control of gastric mucosa. Scand. J. Gastroenterol. Suppl. 1986, 118, 18–30. [Google Scholar] [CrossRef]
- Havu, N. Enterochromaffin-like cell carcinoids of gastric mucosa in rats after life-long inhibition of gastric secretion. Digestion 1986, 35 (Suppl. 1), 42–55. [Google Scholar] [CrossRef]
- Herrera-Martínez, A.D.; Gahete, M.D.; Sánchez-Sánchez, R.; Alors-Perez, E.; Pedraza-Arevalo, S.; Serrano-Blanch, R.; Martínez-Fuentes, A.J.; Gálvez-Moreno, M.A.; Castaño, J.P.; Luque, R.M. Ghrelin-O-Acyltransferase (GOAT) Enzyme as a Novel Potential Biomarker in Gastroenteropancreatic Neuroendocrine Tumors. Clin. Transl. Gastroenterol. 2018, 9, 196. [Google Scholar] [CrossRef]
- Huo, D.; Anderson, D.; Palmer, J.R.; Herbst, A.L. Incidence rates and risks of diethylstilbestrol-related clear-cell adenocarcinoma of the vagina and cervix: Update after 40-year follow-up. Gynecol. Oncol. 2017, 146, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Ishikura, N.; Usui, Y.; Ito, H.; Kasugai, Y.; Oze, I.; Kato, S.; Yatabe, Y.; Nakamura, S.; Matsuo, K. Helicobacter pylori (HP) infection alone, but not HP-induced atrophic gastritis, increases the risk of gastric lymphoma: A case-control study in Japan. Ann. Hematol. 2019, 98, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, Y.; Ishihara, S.; Kadowaki, Y.; Fukui, H.; Chiba, T. Reg protein is a unique growth factor of gastric mucosal cells. J. Gastroenterol. 2004, 39, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Kreipe, H.H. Neuroendocrine differentiation in breast cancer. Pathologe 2019, 40 (Suppl. 3), 325–330. [Google Scholar] [CrossRef]
- Krump, N.A.; You, J. Molecular mechanisms of viral oncogenesis in humans. Nat. Rev. Microbiol. 2018, 16, 684–698. [Google Scholar] [CrossRef]
- Li, C.C.; Xu, B.; Hirokawa, M.; Qian, Z.; Yoshimoto, K.; Horiguchi, H.; Tashiro, T.; Sano, T. Alterations of E-cadherin, alpha-catenin and beta-catenin expression in neuroendocrine tumors of the gastrointestinal tract. Virchows Arch. 2002, 440, 145–154. [Google Scholar] [CrossRef]
- Marshall, B.J.; Barrett, L.J.; Prakash, C.; McCallum, R.W.; Guerrant, R.L. Urea protects Helicobacter (Campylobacter) pylori from the bactericidal effect of acid. Gastroenterology 1990, 99, 697–702. [Google Scholar] [CrossRef]
- Martinsen, T.C.; Skogaker, N.E.; Fossmark, R.; Nordrum, I.S.; Sandvik, A.K.; Bendheim, M.O.; Bakkelund, K.E.; Waldum, H.L. Neuroendocrine cells in diffuse gastric carcinomas: An ultrastructural study with immunogold labeling of chromogranin A. Appl. Immunohistochem. Mol. Morphol. 2010, 18, 62–68. [Google Scholar] [CrossRef]
- Mjelle, R.; Hegre, S.A.; Aas, P.A.; Slupphaug, G.; Drablos, F.; Saetrom, P.; Krokan, H.E. Cell cycle regulation of human DNA repair and chromatin remodeling genes. DNA Repair 2015, 30, 53–67. [Google Scholar] [CrossRef]
- Mjones, P.G.; Nordrum, I.S.; Qvigstad, G.; Sordal, O.; Rian, L.L.; Waldum, H.L. Expression of erythropoietin and neuroendocrine markers in clear cell renal cell carcinoma. APMIS 2017, 125, 213–222. [Google Scholar] [CrossRef]
- Mjones, P.; Nordrum, I.S.; Sordal, O.; Sagatun, L.; Fossmark, R.; Sandvik, A.; Waldum, H.L. Expression of the Cholecystokinin-B Receptor in Neoplastic Gastric Cells. Horm. Cancer 2018, 9, 40–54. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Abnet, C.C.; Choo-Wosoba, H.; Vogtmann, E.; Weinstein, S.J.; Taylor, P.R.; Mannisto, S.; Albanes, D.; Dawsey, S.M.; Rehfeld, J.F.; et al. Serum gastrin and cholecystokinin are associated with subsequent development of gastric cancer in a prospective cohort of Finnish smokers. Int. J. Epidemiol. 2017, 46, 914–923. [Google Scholar] [CrossRef] [PubMed]
- Panaccione, A.; Guo, Y.; Yarbrough, W.G.; Ivanov, S.V. Expression Profiling of Clinical Specimens Supports the Existence of Neural Progenitor-Like Stem Cells in Basal Breast Cancers. Clin. Breast Cancer 2017, 17, 298–306.e7. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, G.; Scarpa, A.; Puppa, G.; Veronesi, G.; Spaggiari, L.; Pasini, F.; Maisonneuve, P.; Iannucci, A.; Arrigoni, G.; Viale, G. Alteration of the E-cadherin/beta-catenin cell adhesion system is common in pulmonary neuroendocrine tumors and is an independent predictor of lymph node metastasis in atypical carcinoids. Cancer 2005, 103, 1154–1164. [Google Scholar] [CrossRef]
- Pezzuto, A.; Carico, E. Role of HIF-1 in Cancer Progression: Novel Insights. A Review. Curr. Mol. Med. 2018, 18, 343–351. [Google Scholar] [CrossRef]
- Poynter, D.; Selway, S.A.; Papworth, S.A.; Riches, S.R. Changes in the gastric mucosa of the mouse associated with long lasting unsurmountable histamine H2 blockade. Gut 1986, 27, 1338–1346. [Google Scholar] [CrossRef]
- Qvigstad, G.; Falkmer, S.; Westre, B.; Waldum, H.L. Clinical and histopathological tumour progression in ECL cell carcinoids ("ECLomas"). APMIS 1999, 107, 1085–1092. [Google Scholar] [CrossRef]
- Qvigstad, G.; Qvigstad, T.; Westre, B.; Sandvik, A.K.; Brenna, E.; Waldum, H.L. Neuroendocrine differentiation in gastric adenocarcinomas associated with severe hypergastrinemia and/or pernicious anemia. APMIS 2002, 110, 132–139. [Google Scholar] [CrossRef]
- Qvigstad, G.; Sandvik, A.K.; Brenna, E.; Aase, S.; Waldum, H.L. Detection of chromogranin A in human gastric adenocarcinomas using a sensitive immunohistochemical technique. Histochem. J. 2000, 32, 551–556. [Google Scholar] [CrossRef]
- Schwitalla, S.; Fingerle, A.A.; Cammareri, P.; Nebelsiek, T.; Goktuna, S.I.; Ziegler, P.K.; Canli, O.; Heijmans, J.; Huels, D.J.; Moreaux, G.; et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 2013, 152, 25–38. [Google Scholar] [CrossRef]
- Sheng, W.; Malagola, E.; Nienhüser, H.; Zhang, Z.; Kim, W.; Zamechek, L.; Sepulveda, A.; Hata, M.; Hayakawa, Y.; Zhao, C.M.; et al. Hypergastrinemia Expands Gastric ECL Cells Through CCK2R(+) Progenitor Cells via ERK Activation. Cell. Mol. Gastroenterol. Hepatol. 2020, 10, 434–449.e1. [Google Scholar] [CrossRef] [PubMed]
- Sjoblom, S.M.; Sipponen, P.; Karonen, S.L.; Jarvinen, H.J. Mucosal argyrophil endocrine cells in pernicious anaemia and upper gastrointestinal carcinoid tumours. J. Clin. Pathol. 1989, 42, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Solcia, E.; Fiocca, R.; Villani, L.; Luinetti, O.; Capella, C. Hyperplastic, dysplastic, and neoplastic enterochromaffin-like-cell proliferations of the gastric mucosa. Classification and histogenesis. Am. J. Surg. Pathol. 1995, 19 (Suppl. 1), S1–S7. [Google Scholar] [PubMed]
- Sordal, O.; Qvigstad, G.; Nordrum, I.S.; Sandvik, A.K.; Gustafsson, B.I.; Waldum, H. The PAS positive material in gastric cancer cells of signet ring type is not mucin. Exp. Mol. Pathol. 2014, 96, 274–278. [Google Scholar] [CrossRef]
- Steinbichler, T.B.; Dudás, J.; Skvortsov, S.; Ganswindt, U.; Riechelmann, H.; Skvortsova, I.I. Therapy resistance mediated by cancer stem cells. Semin. Cancer Biol. 2018, 53, 156–167. [Google Scholar] [CrossRef]
- Stroff, T.; Plate, S.; Respondek, M.; Müller, K.M.; Peskar, B.M. Protection by gastrin in the rat stomach involves afferent neurons, calcitonin gene-related peptide, and nitric oxide. Gastroenterology 1995, 109, 89–97. [Google Scholar] [CrossRef]
- Take, S.; Mizuno, M.; Ishiki, K.; Kusumoto, C.; Imada, T.; Hamada, F.; Yoshida, T.; Yokota, K.; Mitsuhashi, T.; Okada, H. Risk of gastric cancer in the second decade of follow-up after Helicobacter pylori eradication. J. Gastroenterol. 2020, 55, 281–288. [Google Scholar] [CrossRef]
- Tielemans, Y.; Willems, G.; Sundler, F.; Hakanson, R. Self-replication of enterochromaffin-like cells in the mouse stomach. Digestion 1990, 45, 138–146. [Google Scholar] [CrossRef]
- Tsolakis, A.V.; Stridsberg, M.; Grimelius, L.; Portela-Gomes, G.M.; Falkmer, S.E.; Waldum, H.L.; Janson, E.T. Ghrelin immunoreactive cells in gastric endocrine tumors and their relation to plasma ghrelin concentration. J. Clin. Gastroenterol. 2008, 42, 381–388. [Google Scholar] [CrossRef]
- Uemura, N.; Okamoto, S.; Yamamoto, S.; Matsumura, N.; Yamaguchi, S.; Yamakido, M.; Taniyama, K.; Sasaki, N.; Schlemper, R.J. Helicobacter pylori infection and the development of gastric cancer. N. Engl. J. Med. 2001, 345, 784–789. [Google Scholar] [CrossRef]
- Waldum, H.L. The increase in early-onset gastric carcinomas from 1995 is probably due to the introduction of proton pump inhibitors. Surgery 2020. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H.L.; Aase, S.; Kvetnoi, I.; Brenna, E.; Sandvik, A.K.; Syversen, U.; Johnsen, G.; Vatten, L.; Polak, J.M. Neuroendocrine differentiation in human gastric carcinoma. Cancer 1998, 83, 435–444. [Google Scholar] [CrossRef]
- Waldum, H.L.; Brenna, E.; Sandvik, A.K. Relationship of ECL cells and gastric neoplasia. Yale J. Biol. Med. 1998, 71, 325–335. [Google Scholar] [PubMed]
- Waldum, H.L.; Brenna, E.; Sandvik, A.K. Long-term safety of proton pump inhibitors: Risks of gastric neoplasia and infections. Expert Opin. Drug Saf. 2002, 1, 29–38. [Google Scholar] [PubMed]
- Waldum, H.L.; Haugen, O.A.; Isaksen, C.; Mecsei, R.; Sandvik, A.K. Enterochromaffin-like tumour cells in the diffuse but not the intestinal type of gastric carcinomas. Eur. J. Gastroenterol. Hepatol. 1991, 3, 245–249. [Google Scholar] [CrossRef]
- Waldum, H.L.; Hauso, O.; Sordal, O.F.; Fossmark, R. Gastrin May Mediate the Carcinogenic Effect of Helicobacter pylori Infection of the Stomach. Dig. Dis. Sci. 2015, 60, 1522–1527. [Google Scholar] [CrossRef]
- Waldum, H.L.; Oberg, K.; Sordal, O.F.; Sandvik, A.K.; Gustafsson, B.I.; Mjones, P.; Fossmark, R. Not only stem cells, but also mature cells, particularly neuroendocrine cells, may develop into tumours: Time for a paradigm shift. Ther. Adv. Gastroenterol. 2018, 11, 1756284818775054. [Google Scholar] [CrossRef]
- Waldum, H.L.; Rehfeld, J.F. Gastric cancer and gastrin: On the interaction of Helicobacter pylori gastritis and acid inhibitory induced hypergastrinemia. Scand. J. Gastroenterol. 2019, 54, 1118–1123. [Google Scholar] [CrossRef]
- Waldum, H.L.; Ringnes, E.; Nordbo, H.; Sordal, O.; Nordrum, I.S.; Hauso, O. The normal neuroendocrine cells of the upper gastrointestinal tract lack E-cadherin. Scand. J. Gastroenterol. 2014, 49, 974–978. [Google Scholar] [CrossRef]
- Waldum, H.L.; Sagatun, L.; Mjones, P. Gastrin and Gastric Cancer. Front. Endocrinol. 2017, 8, 1. [Google Scholar] [CrossRef]
- Waldum, H.L.; Sandvik, A.K.; Brenna, E.; Petersen, H. Gastrin-histamine sequence in the regulation of gastric acid secretion. Gut 1991, 32, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H.L.; Sordal, O.F. Classification of Epithelial Malignant Tumors-the Differentiation Between Adenocarcinomas and Neuroendocrine Carcinomas: Why Rely on Nonspecific Histochemistry and Dismiss Specific Methods Like Immunohistochemistry and In Situ Hybridization? Appl. Immunohistochem. Mol. Morphol. 2016, 24, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H. Gastrin drives gastric cancer due to oxyntic atrophy also after Helicobacter pylori eradication. Ther. Adv. Gastroenterol. 2020, 13, 1756284820931717. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.C.; Schaeffer, D.F.; Kirsch, R.; Pollett, A.; Manzoni, M.; Riddell, R.H.; Albarello, L. Ileal “carcinoid” tumors-small size belies deadly intent: High rate of nodal metastasis in tumors ≤1 cm in size. Hum. Pathol 2016, 56, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Wilander, E. Achylia, pernicious anaemia, ECL cells and gastric carcinoids. Virchows Arch. Pathol. Anat Histol. 1980, 387, 371–373. [Google Scholar] [CrossRef]
- Wilander, E.; El-Salhy, M.; Pitkanen, P. Histopathology of gastric carcinoids: A survey of 42 cases. Histopathology 1984, 8, 183–193. [Google Scholar] [CrossRef]
- Zabala Torrres, B.; Lucero, Y.; Lagomarcino, A.J.; Orellana-Manzano, A.; George, S.; Torres, J.P.; O’Ryan, M. Review: Prevalence and dynamics of Helicobacter pylori infection during childhood. Helicobacter 2017, 22. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waldum, H.; Mjønes, P.G. Correct Identification of Cell of Origin May Explain Many Aspects of Cancer: The Role of Neuroendocrine Cells as Exemplified from the Stomach. Int. J. Mol. Sci. 2020, 21, 5751. https://doi.org/10.3390/ijms21165751
Waldum H, Mjønes PG. Correct Identification of Cell of Origin May Explain Many Aspects of Cancer: The Role of Neuroendocrine Cells as Exemplified from the Stomach. International Journal of Molecular Sciences. 2020; 21(16):5751. https://doi.org/10.3390/ijms21165751
Chicago/Turabian StyleWaldum, Helge, and Patricia G. Mjønes. 2020. "Correct Identification of Cell of Origin May Explain Many Aspects of Cancer: The Role of Neuroendocrine Cells as Exemplified from the Stomach" International Journal of Molecular Sciences 21, no. 16: 5751. https://doi.org/10.3390/ijms21165751
APA StyleWaldum, H., & Mjønes, P. G. (2020). Correct Identification of Cell of Origin May Explain Many Aspects of Cancer: The Role of Neuroendocrine Cells as Exemplified from the Stomach. International Journal of Molecular Sciences, 21(16), 5751. https://doi.org/10.3390/ijms21165751





