The Diverse Roles of TAO Kinases in Health and Diseases
Abstract
1. Introduction
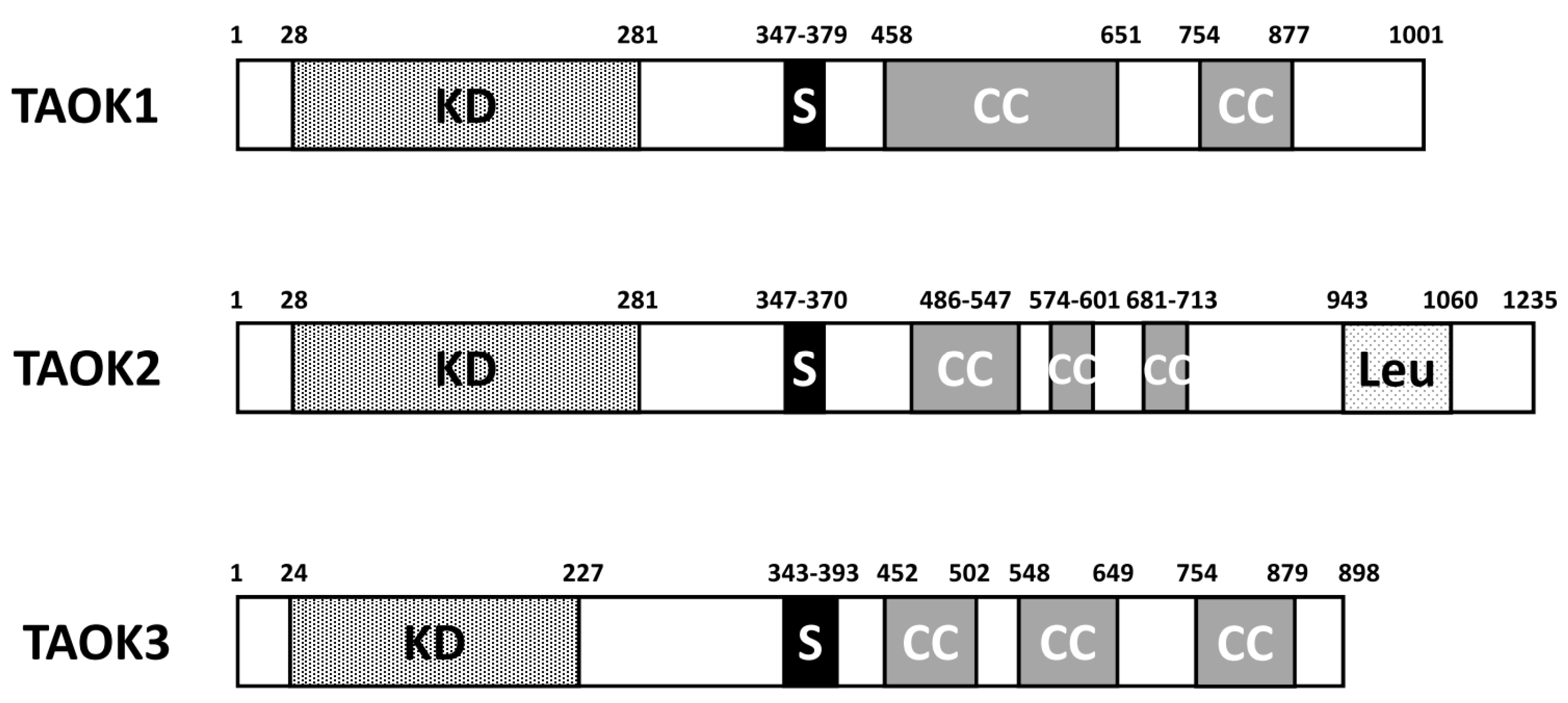
2. Structure and Function of TAO Kinases
3. Signaling Pathways and Cellular Physiologies Regulate by TAO Kinases
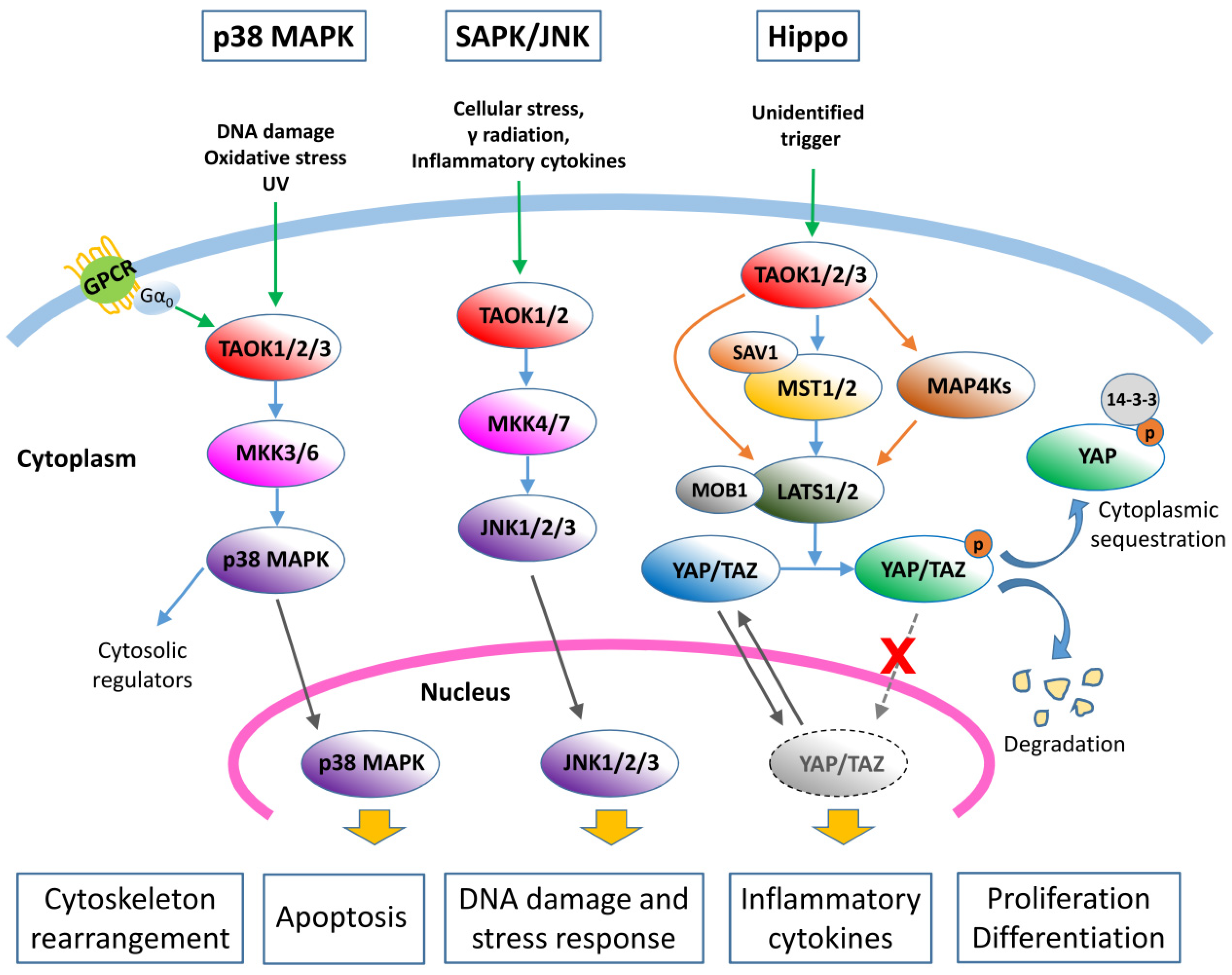
3.1. TAO Kinases Regulate the p38/MAPK Pathway
3.2. TAO Kinases Regulate the SAPK/JNK Pathway
3.3. TAO Kinases Regulate the Hippo Signaling Pathway
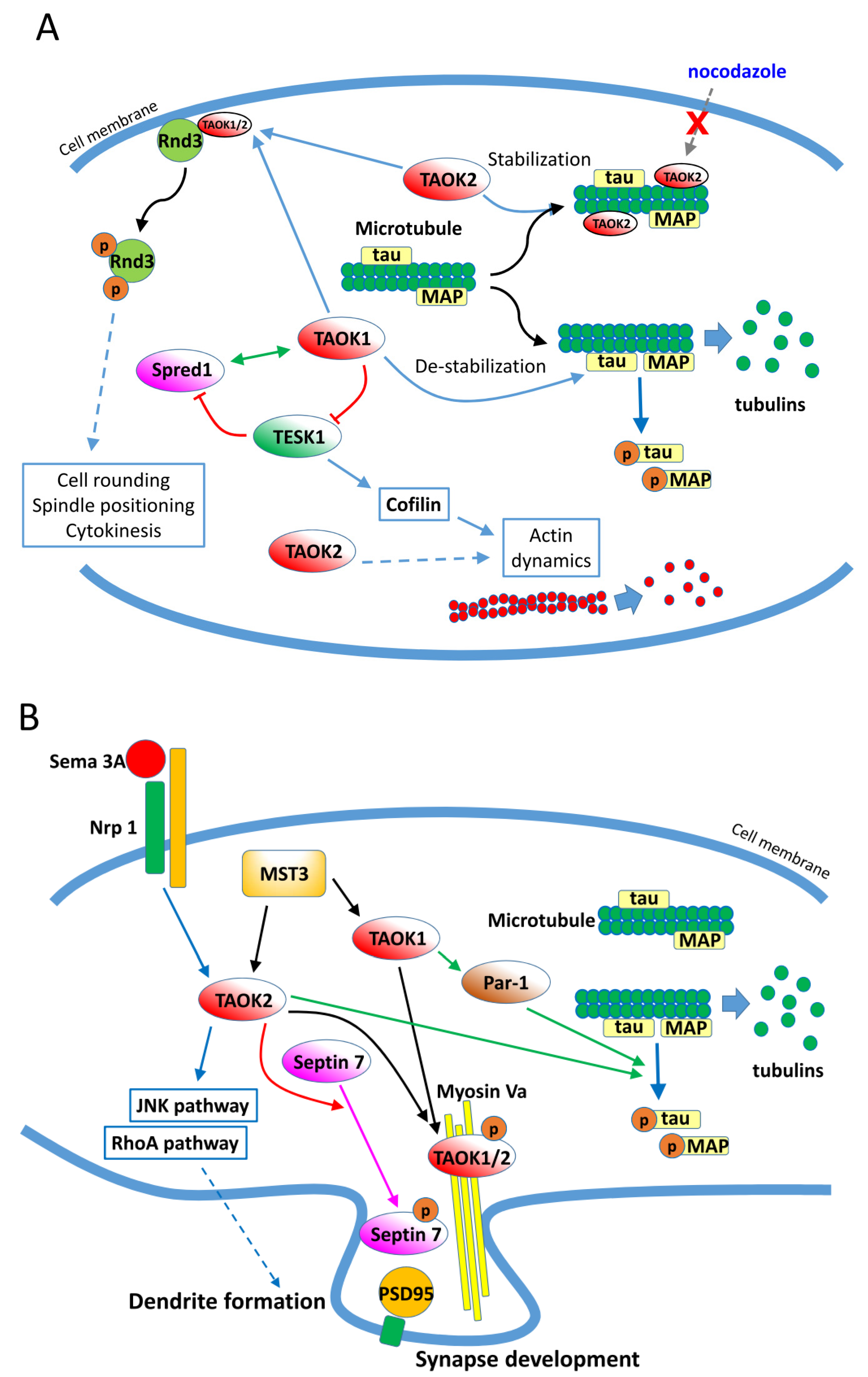
3.4. Cytoskeleton Regulations by TAO Kinases
3.5. TAO Kinases Regulate Neuron Development, Neuritogenesis, and Homeostasis
3.6. Regulation of Inflammation, Immunity, Apoptosis, and other Cellular Pathways by TAOKs
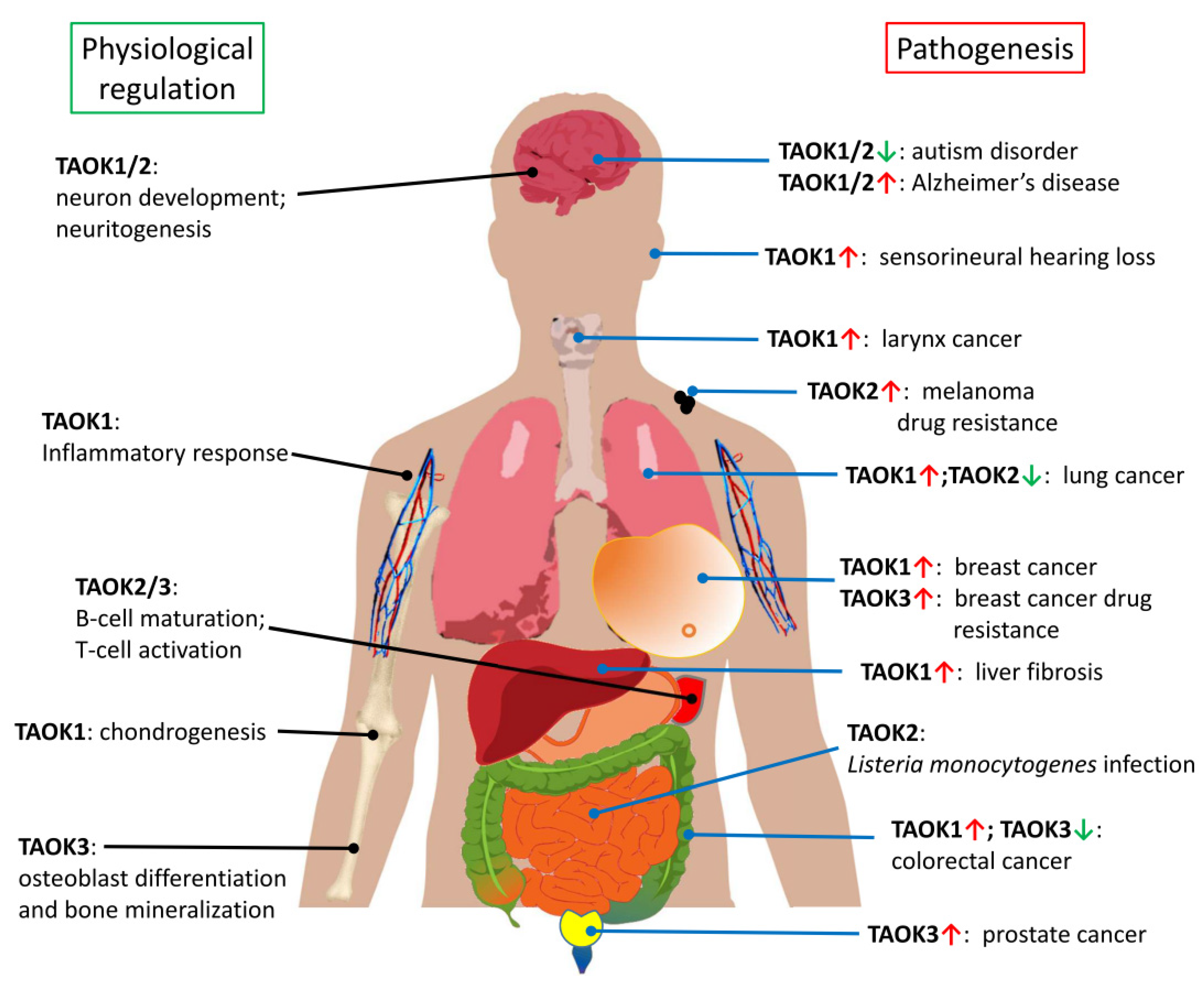
4. Role of TAO Kinases in Cancers
4.1. TAO Kinases in Breast Cancer
4.2. TAO Kinases in Colorectal Cancer
4.3. TAO Kinases in Lung Cancer
4.4. TAO Kinases in Pancreatic Cancer
4.5. TAO Kinases in Prostate Cancer
4.6. TAO Kinases in Melanoma
4.7. TAO Kinases in Larynx Cancer and Leukemia
5. TAO Kinases in Cognitive Disorders and Neurodegenerative Diseases
5.1. TAO Kinases in Autism Spectrum Disorder
5.2. TAO Kinases in Alzheimer’s Disease
5.3. TAO Kinases in Parkinson’s Disease
5.4. TAO Kinases in Cerebral Ischemic Stroke
6. TAO Kinases in Other Physiological Processes and Diseases
7. Current Development of TAOK Inhibitors
8. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pearson, G.; Robinson, F.; Gibson, T.B.; Xu, B.E.; Karandikar, M.; Berman, K.; Cobb, M.H. Mitogen-activated protein (MAP) kinase pathways: Regulation and physiological functions. Endocr. Rev. 2001, 22, 153–183. [Google Scholar] [CrossRef] [PubMed]
- Leberer, E.; Dignard, D.; Harcus, D.; Thomas, D.Y.; Whiteway, M. The Protein-Kinase Homolog Ste20p Is Required to Link the Yeast Pheromone Response G-Protein Beta-Gamma Subunits to Downstream Signaling Components. Embo J. 1992, 11, 4815–4824. [Google Scholar] [CrossRef] [PubMed]
- Ramer, S.W.; Davis, R.W. A Dominant Truncation Allele Identifies a Gene, Ste20, That Encodes a Putative Protein-Kinase Necessary for Mating in Saccharomyces-Cerevisiae. Proc. Natl. Acad. Sci. USA 1993, 90, 452–456. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, M.; Berman, K.S.; Cobb, M.H. Isolation of TAO1, a protein kinase that activates MEKs in stress-activated protein kinase cascades. J. Biol. Chem. 1998, 273, 28625–28632. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Hutchison, M.; Cobb, M.H. Isolation of the protein kinase TAO2 and identification of its mitogen-activated protein kinase/extracellular signal-regulated kinase kinase binding domain. J. Biol. Chem. 1999, 274, 28803–28807. [Google Scholar] [CrossRef]
- Tassi, E.; Biesova, Z.; Di Fiore, P.P.; Gutkind, J.S.; Wong, W.T. Human JIK, a novel member of the STE20 kinase family that inhibits JNK and is negatively regulated by epidermal growth factor. J. Biol. Chem. 1999, 274, 33287–33295. [Google Scholar] [CrossRef]
- Moore, T.M.; Garg, R.; Johnson, C.; Coptcoat, M.J.; Ridley, A.J.; Morris, J.D.H. PSK, a novel STE20-like kinase derived from prostatic carcinoma that activates the c-Jun N-terminal kinase mitogen-activated protein kinase pathway and regulates actin cytoskeletal organization. J. Biol. Chem. 2000, 275, 4311–4322. [Google Scholar] [CrossRef]
- Yustein, J.T.; Xia, L.; Kahlenburg, J.M.; Robinson, D.; Templeton, D.; Kung, H.J. Comparative studies of a new subfamily of human Ste20-like kinases: Homodimerization, subcellular localization, and selective activation of MKK3 and p38. Oncogene 2003, 22, 6129–6141. [Google Scholar] [CrossRef]
- Morrison, D.K. MAP Kinase Pathways. Csh Perspect Biol. 2012, 4, a011254. [Google Scholar] [CrossRef]
- Cuevas, B.D.; Abell, A.N.; Johnson, G.L. Role of mitogen-activated protein kinase kinase kinases in signal integration. Oncogene 2007, 26, 3159–3171. [Google Scholar] [CrossRef]
- Yu, F.X.; Zhao, B.; Guan, K.L. Hippo Pathway in Organ Size Control, Tissue Homeostasis, and Cancer. Cell 2015, 163, 811–828. [Google Scholar] [CrossRef] [PubMed]
- Saucedo, L.J.; Edgar, B.A. Filling out the Hippo pathway. Nat. Rev. Mol. Cell Biol. 2007, 8, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Raman, M.; Earnest, S.; Zhang, K.; Zhao, Y.M.; Cobb, M.H. TAO kinases mediate activation of p38 in response to DNA damage. Embo J. 2007, 26, 2005–2014. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Raman, M.; Chen, L.; Lee, S.F.; Gilman, A.G.; Cobb, M.H. TAO (thousand-and-one amino acid) protein kinases mediate signaling from carbachol to p38 mitogen-activated protein kinase and ternary complex factors. J. Biol. Chem. 2003, 278, 22278–22283. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Cobb, M.H. Regulation of stress-responsive mitogen-activated protein (MAP) kinase pathways by TAO2. J. Biol. Chem. 2001, 276, 16070–16075. [Google Scholar] [CrossRef]
- Weston, C.R.; Davis, R.J. The JNK signal transduction pathway. Curr. Opin. Cell Biol. 2007, 19, 142–149. [Google Scholar] [CrossRef]
- Bogoyevitch, M.A.; Ngoei, K.R.W.; Zhao, T.T.; Yeap, Y.Y.C.; Ng, D.C.H. c-Jun N-terminal kinase (JNK) signaling: Recent advances and challenges. Bba-Proteins. Proteom. 2010, 1804, 463–475. [Google Scholar] [CrossRef]
- Zihni, C.; Mitsopoulos, C.; Tavares, I.A.; Ridley, A.J.; Morris, J.D.H. Prostate-derived sterile 20-like kinase 2 (PSK2) regulates apoptotic morphology via C-jun N-terminal kinase and Rho kinase-1. J. Biol. Chem. 2006, 281, 7317–7323. [Google Scholar] [CrossRef]
- Zihni, C.; Mitsopoulos, C.; Tavares, I.A.; Baum, B.; Ridley, A.J.; Morris, J.D.H. Prostate-derived sterile 20-like kinase 1-alpha induces apoptosis—JNK- and caspase-dependent nuclear localization is a requirement for membrane blebbing. J. Biol. Chem. 2007, 282, 6484–6493. [Google Scholar] [CrossRef]
- Zhang, W.P.; Chen, T.Y.; Wan, T.; He, L.; Li, N.; Yuan, Z.L.; Cao, X.T. Cloning of DPK, a novel dendritic cell-derived protein kinase activating the ERK1/ERK2 and JNK/SAPK pathways. Biochem. Bioph. Res. Commun. 2000, 274, 872–879. [Google Scholar] [CrossRef]
- MacKeigan, J.P.; Murphy, L.O.; Blenis, J. Sensitized RNAi screen of human kinases and phosphatases identifies new regulators of apoptosis and chemoresistance. Nat. Cell Biol. 2005, 7, 591–600. [Google Scholar] [CrossRef]
- Kapfhamer, D.; King, I.; Zou, M.E.; Lim, J.P.; Heberlein, U.; Wolf, F.W. JNK Pathway Activation Is Controlled by Tao/TAOK3 to Modulate Ethanol Sensitivity. PLoS ONE 2012, 7, e50594. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, S.; Dupont, S.; Cordenonsi, M. The Biology of Yap/Taz: Hippo Signaling and Beyond. Physiol. Rev. 2014, 94, 1287–1312. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.X.; Guan, K.L. The Hippo pathway: Regulators and regulations. Gene Dev. 2013, 27, 355–371. [Google Scholar] [CrossRef]
- Boggiano, J.C.; Vanderzalm, P.J.; Fehon, R.G. Tao-1 Phosphorylates Hippo/MST Kinases to Regulate the Hippo-Salvador-Warts Tumor Suppressor Pathway. Dev. Cell 2011, 21, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Poon, C.L.C.; Lin, J.I.; Zhang, X.M.; Harvey, K.F. The Sterile 20-like Kinase Tao-1 Controls Tissue Growth by Regulating the Salvador-Warts-Hippo Pathway. Dev. Cell 2011, 21, 896–906. [Google Scholar] [CrossRef]
- Plouffe, S.W.; Meng, Z.P.; Lin, K.C.; Lin, B.A.; Hong, A.W.; Chun, J.V.; Guan, K.L. Characterization of Hippo Pathway Components by Gene Inactivation. Mol. Cell 2016, 64, 993–1008. [Google Scholar] [CrossRef]
- Meng, Z.; Moroishi, T.; Mottier-Pavie, V.; Plouffe, S.W.; Hansen, C.G.; Hong, A.W.; Park, H.W.; Mo, J.S.; Lu, W.; Lu, S.; et al. MAP4K family kinases act in parallel to MST1/2 to activate LATS1/2 in the Hippo pathway. Nat. Commun. 2015, 6, 8357. [Google Scholar] [CrossRef]
- Mitsopoulos, C.; Zihni, C.; Garg, R.; Ridley, A.J.; Morris, J.D. The prostate-derived sterile 20-like kinase (PSK) regulates microtubule organization and stability. J. Biol. Chem. 2003, 278, 18085–18091. [Google Scholar] [CrossRef]
- Timm, T.; Li, X.Y.; Biernat, J.; Jiao, J.; Mandelkow, E.; Vandekerckhove, J.; Mandelkow, E.M. MARKK, a Ste20-like kinase, activates the polarity-inducing kinase MARK/PAR-1. Embo J. 2003, 22, 5090–5101. [Google Scholar] [CrossRef]
- Liu, T.; Rohn, J.L.; Picone, R.; Kunda, P.; Baum, B. Tao-1 is a negative regulator of microtubule plus-end growth. J. Cell Sci. 2010, 123, 2708–2716. [Google Scholar] [CrossRef] [PubMed]
- Giacomini, C.; Koo, C.Y.; Yankova, N.; Tavares, I.A.; Wray, S.; Noble, W.; Hanger, D.P.; Morris, J.D.H. A new TAO kinase inhibitor reduces tau phosphorylation at sites associated with neurodegeneration in human tauopathies. Acta Neuropathol. Commun. 2018, 6, 37. [Google Scholar] [CrossRef] [PubMed]
- Koo, C.Y.; Giacomini, C.; Reyes-Corral, M.; Olmos, Y.; Tavares, I.A.; Marson, C.M.; Linardopoulos, S.; Tutt, A.N.; Morris, J.D.H. Targeting TAO Kinases Using a New Inhibitor Compound Delays Mitosis and Induces Mitotic Cell Death in Centrosome Amplified Breast Cancer Cells. Mol. Cancer Ther. 2017, 16, 2410–2421. [Google Scholar] [CrossRef] [PubMed]
- de Anda, F.C.; Rosario, A.L.; Durak, O.; Tran, T.; Graff, J.; Meletis, K.; Rei, D.; Soda, T.; Madabhushi, R.; Ginty, D.D.; et al. Autism spectrum disorder susceptibility gene TAOK2 affects basal dendrite formation in the neocortex. Nat. Neurosci. 2012, 15, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Wojtala, R.L.; Tavares, I.A.; Morton, P.E.; Valderrama, F.; Thomas, N.S.B.; Morris, J.D.H. Prostate-derived Sterile 20-like Kinases (PSKs/TAOKs) Are Activated in Mitosis and Contribute to Mitotic Cell Rounding and Spindle Positioning. J. Biol. Chem. 2011, 286, 30161–30170. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, R.L.; Tamura, N.; Fries, A.; Levin, N.; Clark, J.; Draviam, V.M. TAO1 kinase maintains chromosomal stability by facilitating proper congression of chromosomes. Open Biol. 2014, 4, 130108. [Google Scholar] [CrossRef]
- Tavares, I.A.; Touma, D.; Lynham, S.; Troakes, C.; Schober, M.; Causevic, M.; Garg, R.; Noble, W.; Killick, R.; Bodi, I.; et al. Prostate-derived Sterile 20-like Kinases (PSKs/TAOKs) Phosphorylate Tau Protein and Are Activated in Tangle-bearing Neurons in Alzheimer Disease. J. Biol. Chem. 2013, 288, 15418–15429. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.; Koo, C.Y.; Infante, E.; Giacomini, C.; Ridley, A.J.; Morris, J.D.H. Rnd3 interacts with TAO kinases and contributes to mitotic cell rounding and spindle positioning. J. Cell Sci. 2020, 133. [Google Scholar] [CrossRef]
- Johne, C.; Matenia, D.; Li, X.Y.; Timm, T.; Balusamy, K.; Mandelkow, E.M. Spred1 and TESK1--two new interaction partners of the kinase MARKK/TAO1 that link the microtubule and actin cytoskeleton. Mol. Biol. Cell 2008, 19, 1391–1403. [Google Scholar] [CrossRef]
- Ultanir, S.K.; Yadav, S.; Hertz, N.T.; Oses-Prieto, J.A.; Claxton, S.; Burlingame, A.L.; Shokat, K.M.; Jan, L.Y.; Jan, Y.N. MST3 kinase phosphorylates TAO1/2 to enable Myosin Va function in promoting spine synapse development. Neuron 2014, 84, 968–982. [Google Scholar] [CrossRef]
- Yadav, S.; Oses-Prieto, J.A.; Peters, C.J.; Zhou, J.; Pleasure, S.J.; Burlingame, A.L.; Jan, L.Y.; Jan, Y.N. TAOK2 Kinase Mediates PSD95 Stability and Dendritic Spine Maturation through Septin7 Phosphorylation. Neuron 2017, 93, 379–393. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Murtaza, N.; Scharrenberg, R.; White, S.H.; Johanns, O.; Walker, S.; Yuen, R.K.C.; Schwanke, B.; Bedurftig, B.; Henis, M.; et al. Altered TAOK2 activity causes autism-related neurodevelopmental and cognitive abnormalities through RhoA signaling. Mol. Psychiatry 2019, 24, 1329–1350. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Tanaka, H.; Sugiura, H.; Okamura, K.; Sakaguchi, T.; Tran, U.; Takemiya, T.; Mizoguchi, A.; Yagita, Y.; Sakurai, T.; et al. Activity-induced protocadherin arcadlin regulates dendritic spine number by triggering N-cadherin endocytosis via TAO2 beta and p38 MAP kinases. Neuron 2007, 56, 456–471. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.R.; Tang, Z.; Ma, X.W.; Sun, K.; Fan, L.P.; Fang, J.; Pan, J.P.; Wang, X.J.; An, H.Z.; Zhou, J. TAOK1 negatively regulates IL-17-mediated signaling and inflammation. Cell Mol. Immunol. 2018, 15, 794–802. [Google Scholar] [CrossRef]
- Zhu, L.; Yu, Q.; Gao, P.; Liu, Q.; Luo, X.; Jiang, G.; Ji, R.; Yang, R.; Ma, X.; Xu, J.; et al. TAOK1 positively regulates TLR4-induced inflammatory responses by promoting ERK1/2 activation in macrophages. Mol. Immunol. 2020, 122, 124–131. [Google Scholar] [CrossRef]
- Hammad, H.; Vanderkerken, M.; Pouliot, P.; Deswarte, K.; Toussaint, W.; Vergote, K.; Vandersarren, L.; Janssens, S.; Ramou, I.; Savvides, S.N.; et al. Transitional B cells commit to marginal zone B cell fate by Taok3-mediated surface expression of ADAM10. Nat. Immunol. 2017, 18, 313–320. [Google Scholar] [CrossRef]
- Ormonde, J.V.S.; Li, Z.G.; Stegen, C.; Madrenas, J. TAOK3 Regulates Canonical TCR Signaling by Preventing Early SHP-1-Mediated Inactivation of LCK. J. Immunol. 2018, 201, 3431–3442. [Google Scholar] [CrossRef]
- Molho-Pessach, V.; Ramot, Y.; Mogilevsky, M.; Cohen-Daniel, L.; Eisenstein, E.M.; Abu-Libdeh, A.; Siam, I.; Berger, M.; Karni, R.; Zlotogorski, A. Generalized verrucosis and abnormal T cell activation due to homozygous TAOK2 mutation. J. Dermatol. Sci. 2017, 87, 123–129. [Google Scholar] [CrossRef]
- HuangFu, W.C.; Omori, E.; Akira, S.; Matsumoto, K.; Ninomiya-Tsuji, J. Osmotic stress activates the TAK1-JNK pathway while blocking TAK1-mediated NF-kappa B activation—TAO2 regulates TAK1 pathways. J. Biol. Chem. 2006, 281, 28802–28810. [Google Scholar] [CrossRef]
- Pflanz, R.; Voigt, A.; Yakulov, T.; Jackle, H. Drosophila gene tao-1 encodes proteins with and without a Ste20 kinase domain that affect cytoskeletal architecture and cell migration differently. Open Biol. 2015, 5, 140161. [Google Scholar] [CrossRef]
- Liskovykh, M.; Goncharov, N.V.; Petrov, N.; Aksenova, V.; Pegoraro, G.; Ozbun, L.L.; Reinhold, W.C.; Varma, S.; Dasso, M.; Kumeiko, V.; et al. A novel assay to screen siRNA libraries identifies protein kinases required for chromosome transmission. Genome Res. 2019, 29, 1719–1732. [Google Scholar] [CrossRef] [PubMed]
- Capra, M.; Nuciforo, P.G.; Confalonieri, S.; Quarto, M.; Bianchi, M.; Nebuloni, M.; Boldorini, R.; Pallotti, F.; Viale, G.; Gishizky, M.L.; et al. Frequent alterations in the expression of serine/threonine kinases in human cancers. Cancer Res. 2006, 66, 8147–8154. [Google Scholar] [CrossRef] [PubMed]
- Lai, T.C.; Fang, C.Y.; Jan, Y.H.; Hsieh, H.L.; Yang, Y.F.; Liu, C.Y.; Chang, P.M.H.; Hsiao, M. Kinase shRNA screening reveals that TAOK3 enhances microtubule-targeted drug resistance of breast cancer cells via the NF-κB signaling pathway. Cell Commun. Signal. 2020, in press. [Google Scholar] [CrossRef]
- Schulte, I.; Batty, E.M.; Pole, J.C.; Blood, K.A.; Mo, S.; Cooke, S.L.; Ng, C.; Howe, K.L.; Chin, S.F.; Brenton, J.D.; et al. Structural analysis of the genome of breast cancer cell line ZR-75–30 identifies twelve expressed fusion genes. BMC Genom. 2012, 13, 719. [Google Scholar] [CrossRef] [PubMed]
- Hennig, E.E.; Mikula, M.; Rubel, T.; Dadlez, M.; Ostrowski, J. Comparative kinome analysis to identify putative colon tumor biomarkers. J. Mol. Med. 2012, 90, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, C.L.; Li, W.X.; Zhang, R.X.; Duan, Y. Transcriptome analysis reveals gender-specific differences in overall metabolic response of male and female patients in lung adenocarcinoma. PLoS ONE 2020, 15, e0230796. [Google Scholar] [CrossRef] [PubMed]
- Bian, Y.; Teper, Y.; Mathews Griner, L.A.; Aiken, T.J.; Shukla, V.; Guha, R.; Shinn, P.; Xin, H.W.; Pflicke, H.; Powers, A.S.; et al. Target Deconvolution of a Multikinase Inhibitor with Antimetastatic Properties Identifies TAOK3 as a Key Contributor to a Cancer Stem Cell-Like Phenotype. Mol. Cancer Ther. 2019, 18, 2097–2110. [Google Scholar] [CrossRef]
- Romanuik, T.L.; Wang, G.; Holt, R.A.; Jones, S.J.M.; Marra, M.A.; Sadar, M.D. Identification of novel androgen-responsive genes by sequencing of LongSAGE libraries. BMC Genom. 2009, 10, 476. [Google Scholar] [CrossRef]
- Bii, V.M.; Collins, C.P.; Hocum, J.D.; Trobridge, G.D. Replication-incompetent gammaretroviral and lentiviral vector-based insertional mutagenesis screens identify prostate cancer progression genes. Oncotarget 2018, 9, 15451–15463. [Google Scholar] [CrossRef]
- Sharma, R.; Fedorenko, I.; Spence, P.T.; Sondak, V.K.; Smalley, K.S.M.; Koomen, J.M. Activity-Based Protein Profiling Shows Heterogeneous Signaling Adaptations to BRAF Inhibition. J. Proteome Res. 2016, 15, 4476–4489. [Google Scholar] [CrossRef]
- Coyaud, E.; Struski, S.; Prade, N.; Familiades, J.; Eichner, R.; Quelen, C.; Bousquet, M.; Mugneret, F.; Talmant, P.; Pages, M.P.; et al. Wide diversity of PAX5 alterations in B-ALL: A Groupe Francophone de Cytogenetique Hematologique study. Blood 2010, 115, 3089–3097. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.A.; Shen, Y.P.; Korn, J.M.; Arking, D.E.; Miller, D.T.; Fossdal, R.; Saemundsen, E.; Stefansson, H.; Ferreira, M.A.R.; Green, T.; et al. Association between microdeletion and microduplication at 16p11.2 and autism. N. Engl. J. Med. 2008, 358, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Dulovic-Mahlow, M.; Trinh, J.; Kandaswamy, K.K.; Braathen, G.J.; Di Donato, N.; Rahikkala, E.; Beblo, S.; Werber, M.; Krajka, V.; Busk, O.L.; et al. De Novo Variants in TAOK1 Cause Neurodevelopmental Disorders. Am. J. Hum. Genet. 2019, 105, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Zach, S.; Felk, S.; Gillardon, F. Signal Transduction Protein Array Analysis Links LRRK2 to Ste20 Kinases and PKC Zeta That Modulate Neuronal Plasticity. PLoS ONE 2010, 5, e13191. [Google Scholar] [CrossRef] [PubMed]
- Taymans, J.M.; Cookson, M.R. Mechanisms in dominant parkinsonism: The toxic triangle of LRRK2, alpha-synuclein, and tau. Bioessays 2010, 32, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Li, J.H.; Liu, Z.J.; Wang, L.L.; Xu, H.Y.; Wang, Y.L. Thousand and one kinase 1 protects MCAO-induced cerebral ischemic stroke in rats by decreasing apoptosis and pro-inflammatory factors. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef]
- Li, Z.; Oh, H.; Cung, M.; Marquez, S.J.; Sun, J.; Hammad, H.; Janssens, S.; Pouliot, P.; Lambrecht, B.N.; Yang, Y.S.; et al. TAOK3 is a MAP3K contributing to osteoblast differentiation and skeletal mineralization. Biochem. Biophys. Res. Commun. 2020. [Google Scholar] [CrossRef]
- Yin, R.; Guo, D.; Zhang, S.; Zhang, X. miR-706 inhibits the oxidative stress-induced activation of PKCalpha/TAOK1 in liver fibrogenesis. Sci. Rep. 2016, 6, 37509. [Google Scholar] [CrossRef]
- Jing, H.; Zhang, X.Y.; Luo, K.; Luo, Q.C.; Yin, M.; Wang, W.; Zhu, Z.Q.; Zheng, J.H.; He, X.M. miR-381-abundant small extracellular vesicles derived from kartogenin-preconditioned mesenchymal stem cells promote chondrogenesis of MSCs by targeting TAOK1. Biomaterials 2020, 231. [Google Scholar] [CrossRef]
- Patel, M.; Cai, Q.F.; Ding, D.L.; Salvi, R.; Hu, Z.H.; Hu, B.H. The miR-183/Taok1 Target Pair Is Implicated in Cochlear Responses to Acoustic Trauma. PLoS ONE 2013, 8, e58471. [Google Scholar] [CrossRef]
- Balashanmugam, M.V.; Shivanandappa, T.B.; Nagarethinam, S.; Vastrad, B.; Vastrad, C. Analysis of Differentially Expressed Genes in Coronary Artery Disease by Integrated Microarray Analysis. Biomolecules 2019, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, V.R.; Binatti, A.; Coppe, A.; Teramo, A.; Vicenzetto, C.; Calabretto, G.; Barila, G.; Barizza, A.; Giussani, E.; Facco, M.; et al. A high definition picture of somatic mutations in chronic lymphoproliferative disorder of natural killer cells. Blood Cancer J. 2020, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Quereda, J.J.; Morel, C.; Lopez-Montero, N.; Ziveri, J.; Rolland, S.; Grenier, T.; Aulner, N.; Danckaert, A.; Charbit, A.; Enninga, J.; et al. A role for Taok2 in Listeria monocytogenes vacuolar escape. J. Infect. Dis. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cook-Sather, S.D.; Li, J.; Goebel, T.K.; Sussman, E.M.; Rehman, M.A.; Hakonarson, H. TAOK3, a novel genome-wide association study locus associated with morphine requirement and postoperative pain in a retrospective pediatric day surgery population. Pain 2014, 155, 1773–1783. [Google Scholar] [CrossRef] [PubMed]
- Gutteridge, T.; Kumaran, M.; Ghosh, S.; Fainsinger, R.; Klepstad, P.; Tarumi, Y.; Damaraju, S.; Baracos, V.E. Single-Nucleotide Polymorphisms in TAOK3 Are Associated With High Opioid Requirement for Pain Management in Patients With Advanced Cancer Admitted to a Tertiary Palliative Care Unit. J. Pain Symptom Manag. 2018, 56, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Sadhasivam, S.; Zhang, X.; Prows, C.A.; Kaufman, K.M.; Martin, L.J. Challenges and cautions with small and retrospective postoperative pain genome-wide association studies with TAOK3. Pain 2014, 155, 2434–2435. [Google Scholar] [CrossRef]
- Matic, M.; van den Bosch, G.E.; de Wildt, S.N.; Tibboel, D.; van Schaik, R.H.N. Pain versus analgesia: TAOK3 as a pharmacogene Reply. Pain 2017, 158, 1622–1623. [Google Scholar] [CrossRef]
- Kapfhamer, D.; Taylor, S.; Zou, M.E.; Lim, J.P.; Kharazia, V.; Heberlein, U. Taok2 controls behavioral response to ethanol in mice. Genes Brain Behav. 2013, 12, 87–97. [Google Scholar] [CrossRef]
- Huang, R.C.; Garratt, E.S.; Pan, H.; Wu, Y.; Davis, E.A.; Barton, S.J.; Burdge, G.C.; Godfrey, K.M.; Holbrook, J.D.; Lillycrop, K.A. Genome-wide methylation analysis identifies differentially methylated CpG loci associated with severe obesity in childhood. Epigenetics Us 2015, 10, 995–1005. [Google Scholar] [CrossRef]
- Komazin-Meredith, G.; Cardinale, S.C.; Comeau, K.; Magalhaes, K.J.; Hartline, C.B.; Williams, J.D.; Opperman, T.J.; Prichard, M.N.; Bowlin, T.L. TAOK3 phosphorylates the methylenecyclopropane nucleoside MBX 2168 to its monophosphate. Antivir. Res. 2015, 119, 23–27. [Google Scholar] [CrossRef]
- Kelly, B.J.; Diefenbach, E.; Fraefel, C.; Diefenbach, R.J. Identification of host cell proteins which interact with herpes simplex virus type 1 tegument protein pUL37. Biochem. Bioph. Res. Commun. 2012, 417, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.J.; Sun, L.G.; Gao, Y.; Goldsmith, E.J. Crystal structure of the MAP3K TAO2 kinase domain bound by an inhibitor staurosporine. Acta Biochimica et Biophysica Sinica 2006, 38, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Anand, R.; Maksimoska, J.; Pagano, N.; Wong, E.Y.; Gimotty, P.A.; Diamond, S.L.; Meggers, E.; Marmorstein, R. Toward the Development of a Potent and Selective Organoruthenium Mammalian Sterile 20 Kinase Inhibitor. J. Med. Chem. 2009, 52, 1602–1611. [Google Scholar] [CrossRef] [PubMed]
- Piala, A.T.; Akella, R.; Potts, M.B.; Dudics-Giagnocavo, S.A.; He, H.X.; Wei, S.G.; White, M.A.; Posner, B.A.; Goldsmith, E.J. Discovery of novel TAOK2 inhibitor scaffolds from high-throughput screening. Bioorganic Med. Chem. Lett. 2016, 26, 3923–3927. [Google Scholar] [CrossRef]
- Chen, X.; Liu, H.; Xie, W.; Yang, Y.; Wang, Y.; Fan, Y.; Hua, Y.; Zhu, L.; Zhao, J.; Lu, T.; et al. Investigation of crystal structures in structure-based virtual screening for protein kinase inhibitors. J. Chem. Inf. Model. 2019, 59, 5244–5262. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wen, Y.; Gao, L.; Gao, L.; He, F.; Zhou, J.; Wang, J.; Dai, R.; Chen, X.; Kang, D.; et al. Design, synthesis and biological evaluation of novel 1h-1,2,4-triazole, benzothiazole and indazole-based derivatives as potent fgfr1 inhibitors viafragment-based virtual screening. J. Enzyme Inhib. Med. Chem. 2020, 35, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Raman, M.; Gao, Y.; Earnest, S.; Chen, Z.; Machius, M.; Cobb, M.H.; Goldsmith, E.J. Crystal structure of the tao2 kinase domain: Activation and specificity of a ste20p map3k. Structure 2004, 12, 1891–1900. [Google Scholar] [CrossRef]
| Compound Name | IUPAC Name | Cell-Based Inhibition Assay (IC50) (Assay Methodology) * | Kinase Inhibition Assay (IC50) (Assay Methodology) * | Reference |
|---|---|---|---|---|
| Compound 43 | N-[2-oxo-2-(1,2,3,4-tetrahydro-naphthalen-1-ylamino)ethyl]biphenyl-4-carboxamide | NA/showed growth inhibition to SK-BR-3 but not MCF-10A cells | TAOK1: 11 ± 0.44 nmol/L (MBPp) TAOK2: 15 ± 1.63 nmol/L (MBPp) Selectively inhibits TAOK1, 2, 3 while showing low inhibition for other 19 kinases | [33] |
| Compound 63 | N-{3-[(2-{[6-methoxy-1,3-benzothiazol-2yl]amino}-2-oxoethyl)amino]-3-oxo-1-phenylpropyl}benzamide | NA | TAOK1: 19 ± 1.87 nmol/L (MBPp) TAOK2: 39 ± 6.43 nmol/L (MBPp) Selectively inhibits TAOK1, 2, 3 while showing low inhibition for other 19 kinases | [33] |
| NCGC00188382 | N-[5-[(3,3-dimethylbutan-2-ylamino)methyl]-1-(2-hydroxy-2-methylpropyl)benzimidazol-2-yl]-5-(1H-pyrazol-4-yl)thiophene-2-carboxamide | 25 to 300 nmol/L in 24 pancreatic cancer cell lines (FA6 cell: ~25 nmol/L; MDA-Panc-28: ~300 nmol/L) (Cytotoxicity assay) | NA/ showed inhibition to the activity of TAOK3, aurora B kinase, and cyclin-dependent kinase 7 in pancreatic cancer cells | [57] |
| SW034538 | N-(2-((2,5-dimethoxyphenyl)amino)-4’-methyl-[4,5’-bithiazol]-2’-yl)propionamide | NA | TAOK2: 300 nmol/L (MBPp) | [84] |
| SW083688 | N-((2,3-dihydrobenzo[b][1,4]dioxin-2-yl)methyl)-3-(3-ethoxypropyl)-4-oxo-2-thioxo-1,2,3,4-tetrahydroquinazoline-7-carboxamide | NA | TAOK2: 1.3 μmol/L (MBPp) | [84] |
| Disease | Sample and Experimental Approach * | Results | Reference |
|---|---|---|---|
| Cancers | |||
| Breast cancer | Biopsy/ISH; cell line/KD and pathway assay | Upregulation of TAOK1 in tumor tissue; TAOK3 enhances microtubule-targeted drug resistance | [52,53] |
| Colorectal cancer | Biopsy/ISH and kinome profiling | Upregulation of TAOK1 in tumor tissue; downregulation of TAOK3 in adenocarcinoma | [52,55] |
| Lung cancer | Biopsy/ISH and transcriptome assay | Upregulation of TAOK1 in tumor tissue; downregulation of TAOK2 in tumor tissue | [52,56] |
| Pancreatic cancer | Cell line/OE and KD, mouse xenograft model | TAOK3 supports the stemness traits and growth of tumor spheroids | [57] |
| Prostate cancer | Cell line/transcriptome; MGS-PCR | TAOK3 is a prostate cancer progression gene and its expression can predict the risk of recurrence after androgen deprivation therapy | [58,59] |
| Melanoma | Cell line/activity-based protein profiling | TAOK2 activates JNK and contributes to the BRAF inhibitor (vemurafenib) resistance | [60] |
| Larynx cancer | Biopsy/ISH | Upregulation of TAOK1 in tumor tissue | [52] |
| B cell leukemia | Cell line/rolling-circle amplification of cDNA ends | PAX5–TAOK1 fusion protein may be a competitive inhibitor of wild-type PAX5 | [61] |
| Neurodegenerative disease | |||
| Autism | Cell line; mouse model; patient DNA sample/KD and ectopic expression; TAOK2-KO mouse; genotyping | Downregulation of TAOK1/2 activity during neuron development results in cognitive abnormalities and autism | [34,42,62,63] |
| Alzheimer’s disease | Cell line; mouse model; human biopsy/ OE in cell; IHC; inhibitor assay | TAOK1/2 dysregulate tau phosphorylation and participate in the development of dementia and AD | [32,37] |
| Parkinson’s disease | Cell line/protein array assay | TAOK3 is a novel LRRK2 substrate and involved in LRRK2-induced PD | [64] |
| Cerebral ischemic stroke | Mouse model/induced cerebral ischemic stroke | TAOK1 ameliorates induced cerebral ischemic stroke by decreasing pro-inflammatory factors and reducing apoptosis | [66] |
| Inflammation | |||
| IL-17-associated | Cell line/KD and OE; TAOK1 KO-mouse model | TAOK1 inhibits IL-17-mediated signal transduction and inflammation | [44] |
| LPS-induced | KO-mouse model of TAOK1 in myeloid cells | TAOK1 enhances LPS-induced activation of ERK1/2 and positively regulates the TLR4-induced inflammatory response | [45] |
| Other diseases | |||
| Liver fibrosis | Mouse model of liver fibrosis | miR-706 inhibits PKCα and TAOK1 expression, thus prevents liver fibrosis | [68] |
| Listeria infection | Bacteria and cell line/siRNA microscopy screening | TAOK2 regulates vacuolar rupture and cytoplasmic access of Listeria | [73] |
| Coronary artery disease | Expression database analysis | TAOK1 highly expressed in patients with coronary artery disease | [71] |
| Sensorineural hearing loss | Noise-traumatized rat model/ miRNA expression analysis | Downregulation of miRNA-183 and upregulation of TAOK1 may be involved in sensorineural hearing loss | [70] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, C.-Y.; Lai, T.-C.; Hsiao, M.; Chang, Y.-C. The Diverse Roles of TAO Kinases in Health and Diseases. Int. J. Mol. Sci. 2020, 21, 7463. https://doi.org/10.3390/ijms21207463
Fang C-Y, Lai T-C, Hsiao M, Chang Y-C. The Diverse Roles of TAO Kinases in Health and Diseases. International Journal of Molecular Sciences. 2020; 21(20):7463. https://doi.org/10.3390/ijms21207463
Chicago/Turabian StyleFang, Chih-Yeu, Tsung-Ching Lai, Michael Hsiao, and Yu-Chan Chang. 2020. "The Diverse Roles of TAO Kinases in Health and Diseases" International Journal of Molecular Sciences 21, no. 20: 7463. https://doi.org/10.3390/ijms21207463
APA StyleFang, C.-Y., Lai, T.-C., Hsiao, M., & Chang, Y.-C. (2020). The Diverse Roles of TAO Kinases in Health and Diseases. International Journal of Molecular Sciences, 21(20), 7463. https://doi.org/10.3390/ijms21207463







