Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium
Abstract
1. Introduction
2. Glucose Transporters in Choroid Plexus Epithelial Cells
2.1. SLC2A Family
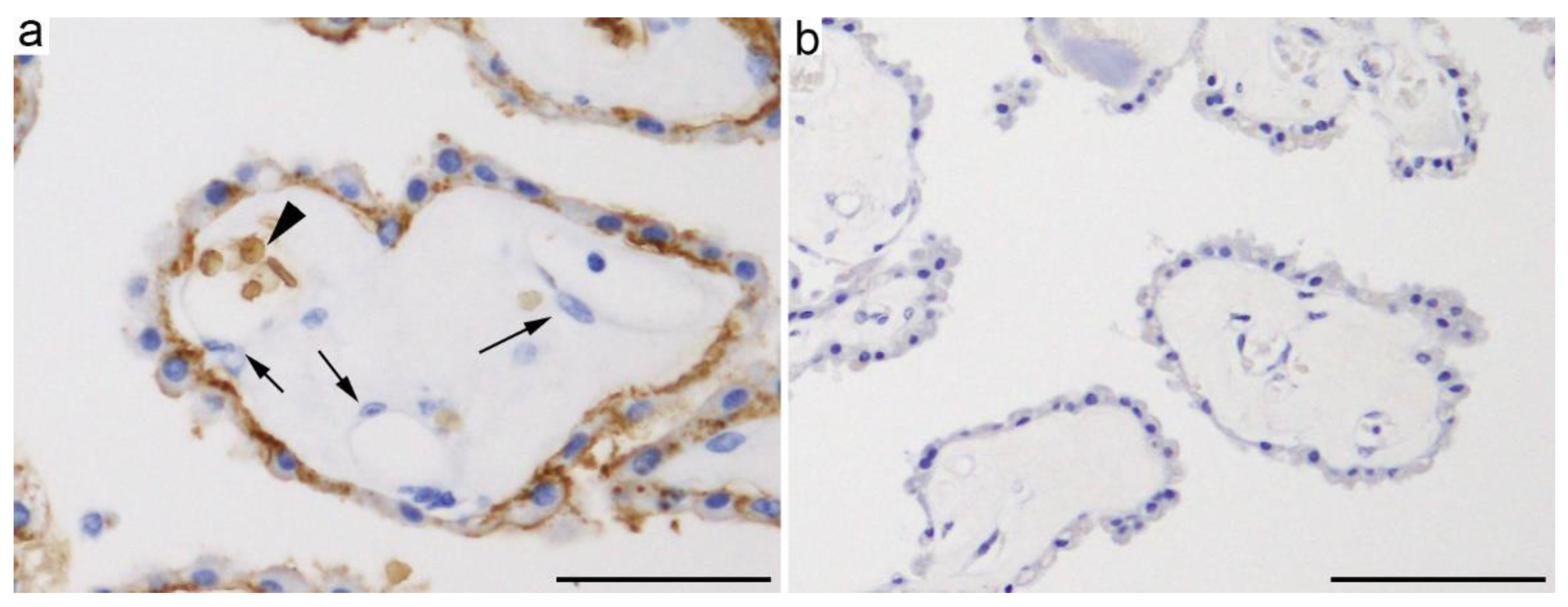
2.1.1. GLUT1 (SLC2A1)
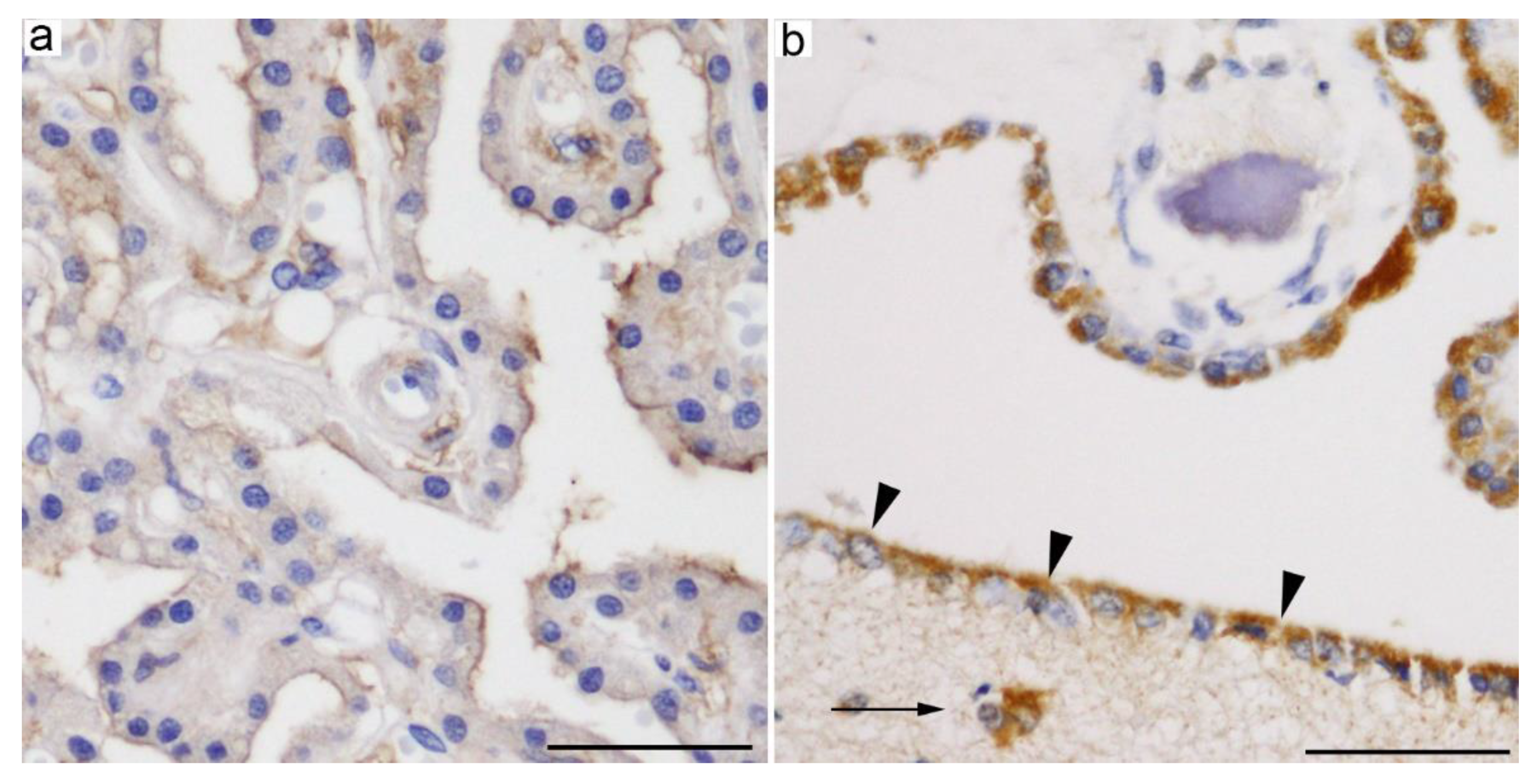
2.1.2. GLUT12 (SLC2A12)
2.1.3. GLUT3 (SLC2A3)
2.1.4. GLUT4 (SLC2A4)
2.2. SLC5A Family
2.2.1. SGLT1 (SLC5A1)
2.2.2. SGLT2 (SLC5A2)
2.3. Physiological and Pathophysiological Considerations of Glucose Transporters in Choroid Plexus Epithelial Cells
3. Fructose Transporters in Choroid Plexus Epithelial Cells
3.1. GLUT5 (SLC2A5)
3.2. GLUT8 (SLC2A8)
3.3. SGLT5 (SLC5A10)
3.4. Physiological and Pathophysiological Considerations of Fructose Transporters in Choroid Plexus Epithelial Cells
4. Urate Transporters in Choroid Plexus Epithelial Cells
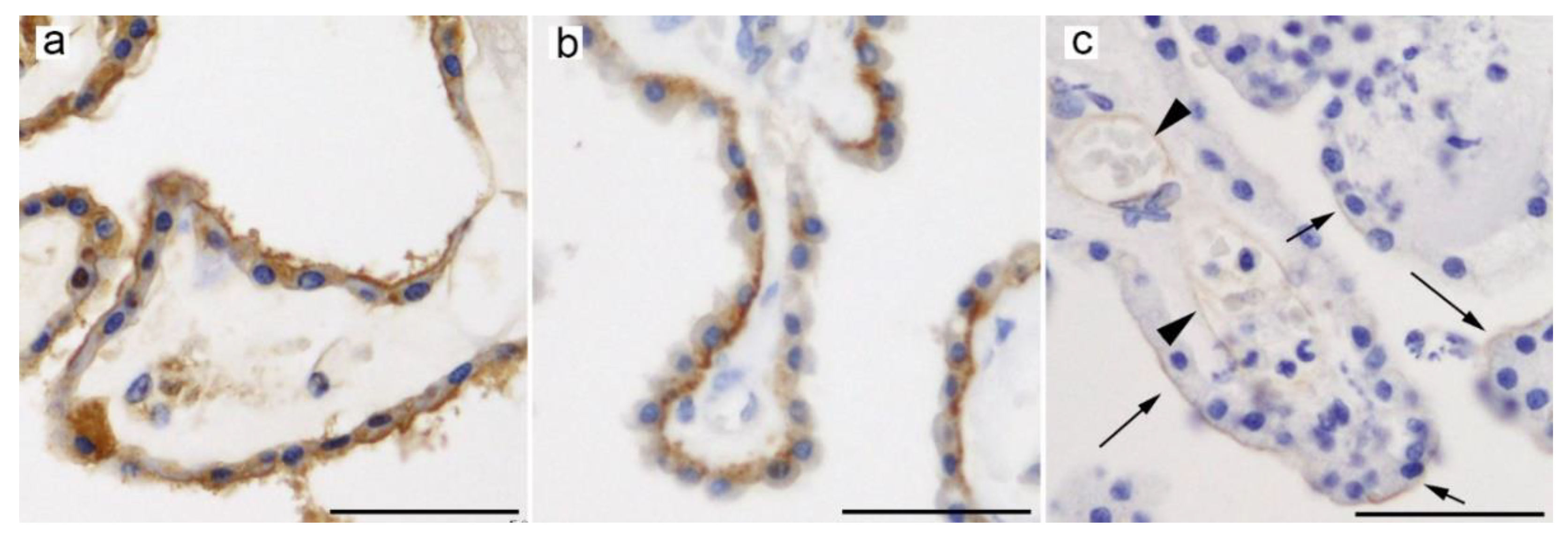
4.1. GLUT9 (SLC2A9)
4.2. URAT1 (SLC22A12)
4.3. BCRP (ABCG2)
4.4. Physiological and Pathophysiological Considerations of Urate Transporters in Choroid Plexus Epithelial Cells
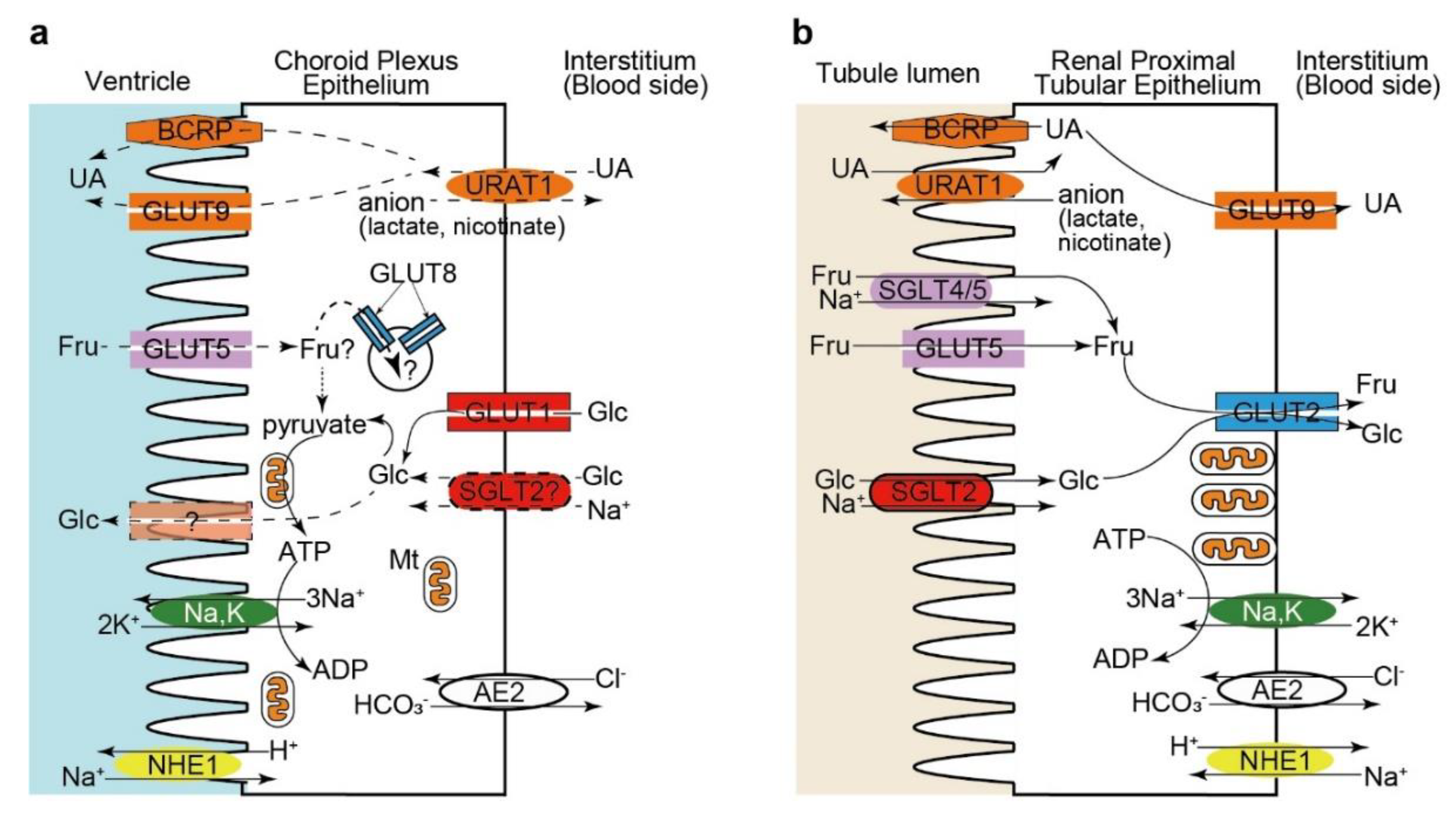
5. Hexose and Urate Transporters in the Choroid Plexus Epithelium: Comparison with the Renal Proximal Tubular Epithelium
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ISF | Interstitial fluid |
| CNS | Central nervous system |
| BBB | Blood-brain barrier |
| BCSFB | Blood-cerebrospinal fluid barrier |
| CP | Choroid plexus |
| CSF | Cerebrospinal fluid |
| Aβ | Amyloid β |
| GLUT | Glucose transporter |
| SGLT | Sodium/glucose cotransporter |
| AD | Alzheimer disease |
| Me-4-FDG | α-methyl-4-deoxy-4-[18F]fluoro-D-glucopyranoside |
| T2DM | Type 2 diabetes mellitus |
| PET | Positron emission tomography |
| PD | Parkinson’s disease |
| ALS | Amyotrophic lateral sclerosis |
| URAT1 | Urate transporter 1 |
| BCRP | Breast cancer resistance protein |
| ABC | ATP-binding cassette |
| FDG | 18F-fluorodeoxyglucose |
References
- Redzic, Z. Molecular biology of the blood-brain and the blood-cerebrospinal fluid barriers: Similarities and differences. Fluids Barriers CNS 2011, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.D.; Zhao, Z.; Montagne, A.; Nelson, A.R.; Zlokovic, B.V. Blood-Brain Barrier: From Physiology to Disease and Back. Physiol. Rev. 2019, 99, 21–78. [Google Scholar] [CrossRef] [PubMed]
- Ueno, M.; Chiba, Y.; Murakami, R.; Matsumoto, K.; Fujihara, R.; Uemura, N.; Yanase, K.; Kamada, M. Disturbance of Intracerebral Fluid Clearance and Blood-Brain Barrier in Vascular Cognitive Impairment. Int. J. Mol. Sci. 2019, 20, 2600. [Google Scholar] [CrossRef]
- Morris, M.E.; Rodriguez-Cruz, V.; Felmlee, M.A. SLC and ABC Transporters: Expression, Localization, and Species Differences at the Blood-Brain and the Blood-Cerebrospinal Fluid Barriers. AAPS J. 2017, 19, 1317–1331. [Google Scholar] [CrossRef] [PubMed]
- Damkier, H.; Praetorius, J. Structure of the Mammalian Chorod Plexus. In Role of the Choroid Plexus in Health and Disease; Praetorius, J., Blazer-Yost, B., Damkier, H., Eds.; Springer: New York, NY, USA, 2020; pp. 1–33. [Google Scholar] [CrossRef]
- Ghersi-Egea, J.F.; Strazielle, N.; Catala, M.; Silva-Vargas, V.; Doetsch, F.; Engelhardt, B. Molecular anatomy and functions of the choroidal blood-cerebrospinal fluid barrier in health and disease. Acta Neuropathol. 2018, 135, 337–361. [Google Scholar] [CrossRef]
- Strazielle, N.; Ghersi-Egea, J.F. Physiology of blood-brain interfaces in relation to brain disposition of small compounds and macromolecules. Mol. Pharm. 2013, 10, 1473–1491. [Google Scholar] [CrossRef]
- Praetorius, J.; Damkier, H.H. Transport across the choroid plexus epithelium. Am. J. Physiol. Cell Physiol. 2017, 312, C673–C686. [Google Scholar] [CrossRef]
- Pardridge, W.M. CSF, blood-brain barrier, and brain drug delivery. Expert Opin. Drug Deliv. 2016, 13, 963–975. [Google Scholar] [CrossRef]
- Johanson, C.E.; Keep, R.F. Blending Established and New Perspectives on Choroid Plexus-CSF Dynamics. In Role of the Choroid Plexus in Health and Disease; Praetorius, J., Blazer-Yost, B., Damkier, H., Eds.; Springer: New York, NY, USA, 2020; pp. 35–81. [Google Scholar] [CrossRef]
- Matsumoto, K.; Chiba, Y.; Fujihara, R.; Kubo, H.; Sakamoto, H.; Ueno, M. Immunohistochemical analysis of transporters related to clearance of amyloid-beta peptides through blood-cerebrospinal fluid barrier in human brain. Histochem. Cell Biol. 2015, 144, 597–611. [Google Scholar] [CrossRef]
- Ueno, M.; Chiba, Y.; Murakami, R.; Matsumoto, K.; Kawauchi, M.; Fujihara, R. Blood-brain barrier and blood-cerebrospinal fluid barrier in normal and pathological conditions. Brain Tumor Pathol. 2016, 33, 89–96. [Google Scholar] [CrossRef]
- Damkier, H.H.; Brown, P.D.; Praetorius, J. Cerebrospinal fluid secretion by the choroid plexus. Physiol. Rev. 2013, 93, 1847–1892. [Google Scholar] [CrossRef] [PubMed]
- Spector, R.; Keep, R.F.; Robert Snodgrass, S.; Smith, Q.R.; Johanson, C.E. A balanced view of choroid plexus structure and function: Focus on adult humans. Exp. Neurol. 2015, 267, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S. Metabolic compartmentalization between astroglia and neurons in physiological and pathophysiological conditions of the neurovascular unit. Neuropathology 2020, 40, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Simpson, I.A.; Carruthers, A.; Vannucci, S.J. Supply and demand in cerebral energy metabolism: The role of nutrient transporters. J. Cereb. Blood Flow Metab. 2007, 27, 1766–1791. [Google Scholar] [CrossRef]
- Paganoni, S.; Schwarzschild, M.A. Urate as a Marker of Risk and Progression of Neurodegenerative Disease. Neurotherapeutics 2017, 14, 148–153. [Google Scholar] [CrossRef]
- Amaro, S.; Jimenez-Altayo, F.; Chamorro, A. Uric acid therapy for vasculoprotection in acute ischemic stroke. Brain Circ. 2019, 5, 55–61. [Google Scholar] [CrossRef]
- Navale, A.M.; Paranjape, A.N. Glucose transporters: Physiological and pathological roles. Biophys. Rev. 2016, 8, 5–9. [Google Scholar] [CrossRef]
- Mueckler, M.; Thorens, B. The SLC2 (GLUT) family of membrane transporters. Mol. Asp. Med. 2013, 34, 121–138. [Google Scholar] [CrossRef]
- Wright, E.M.; Loo, D.D.; Hirayama, B.A. Biology of human sodium glucose transporters. Physiol. Rev. 2011, 91, 733–794. [Google Scholar] [CrossRef]
- Farrell, C.L.; Pardridge, W.M. Blood-brain barrier glucose transporter is asymmetrically distributed on brain capillary endothelial lumenal and ablumenal membranes: An electron microscopic immunogold study. Proc. Natl. Acad. Sci. USA 1991, 88, 5779–5783. [Google Scholar] [CrossRef]
- Hladky, S.B.; Barrand, M.A. Fluid and ion transfer across the blood-brain and blood-cerebrospinal fluid barriers; a comparative account of mechanisms and roles. Fluids Barriers CNS 2016, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Zhang, Z.; Tachikawa, M.; Terasaki, T. Quantitative targeted absolute proteomics of rat blood-cerebrospinal fluid barrier transporters: Comparison with a human specimen. J. Neurochem. 2015, 134, 1104–1115. [Google Scholar] [CrossRef] [PubMed]
- Saunders, N.R.; Dziegielewska, K.M.; Mollgard, K.; Habgood, M.D.; Wakefield, M.J.; Lindsay, H.; Stratzielle, N.; Ghersi-Egea, J.F.; Liddelow, S.A. Influx mechanisms in the embryonic and adult rat choroid plexus: A transcriptome study. Front. Neurosci. 2015, 9, 123. [Google Scholar] [CrossRef] [PubMed]
- Harik, S.I.; Kalaria, R.N.; Andersson, L.; Lundahl, P.; Perry, G. Immunocytochemical localization of the erythroid glucose transporter: Abundance in tissues with barrier functions. J. Neurosci. 1990, 10, 3862–3872. [Google Scholar] [CrossRef]
- Cornford, E.M.; Hyman, S.; Cornford, M.E.; Damian, R.T. Glut1 glucose transporter in the primate choroid plexus endothelium. J. Neuropathol. Exp. Neurol. 1998, 57, 404–414. [Google Scholar] [CrossRef][Green Version]
- Murakami, R.; Chiba, Y.; Tsuboi, K.; Matsumoto, K.; Kawauchi, M.; Fujihara, R.; Mashima, M.; Kanenishi, K.; Yamamoto, T.; Ueno, M. Immunoreactivity of glucose transporter 8 is localized in the epithelial cells of the choroid plexus and in ependymal cells. Histochem. Cell Biol. 2016, 146, 231–236. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Dziegielewska, K.M.; Ek, C.J.; Habgood, M.D.; Bauer, H.; Bauer, H.C.; Lindsay, H.; Wakefield, M.J.; Strazielle, N.; Kratzer, I.; et al. Mechanisms that determine the internal environment of the developing brain: A transcriptomic, functional and ultrastructural approach. PLoS ONE 2013, 8, e65629. [Google Scholar] [CrossRef]
- Pujol-Gimenez, J.; Martisova, E.; Perez-Mediavilla, A.; Lostao, M.P.; Ramirez, M.J. Expression of the glucose transporter GLUT12 in Alzheimer’s disease patients. J. Alzheimers Dis. 2014, 42, 97–101. [Google Scholar] [CrossRef]
- Gil-Iturbe, E.; Castilla-Madrigal, R.; Barrenetxe, J.; Villaro, A.C.; Lostao, M.P. GLUT12 expression and regulation in murine small intestine and human Caco-2 cells. J. Cell Physiol. 2019, 234, 4396–4408. [Google Scholar] [CrossRef]
- Gil-Iturbe, E.; Arbones-Mainar, J.M.; Moreno-Aliaga, M.J.; Lostao, M.P. GLUT12 and adipose tissue: Expression, regulation and its relation with obesity in mice. Acta Physiol. 2019, 226, e13283. [Google Scholar] [CrossRef]
- Pujol-Gimenez, J.; Perez, A.; Reyes, A.M.; Loo, D.D.; Lostao, M.P. Functional characterization of the human facilitative glucose transporter 12 (GLUT12) by electrophysiological methods. Am. J. Physiol. Cell Physiol. 2015, 308, C1008–C1022. [Google Scholar] [CrossRef]
- Gil-Iturbe, E.; Solas, M.; Cuadrado-Tejedo, M.; Garcia-Osta, A.; Escote, X.; Ramirez, M.J.; Lostao, M.P. GLUT12 Expression in Brain of Mouse Models of Alzheimer’s Disease. Mol. Neurobiol. 2020, 57, 798–805. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.T.; Dahlin, A.; Wang, J. Expression Profiling of Solute Carrier Gene Families at the Blood-CSF Barrier. Front. Pharmacol. 2012, 3, 154. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Freeze, H.H. GLUT14, a duplicon of GLUT3, is specifically expressed in testis as alternative splice forms. Genomics 2002, 80, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Vannucci, S.J. Developmental expression of GLUT1 and GLUT3 glucose transporters in rat brain. J. Neurochem. 1994, 62, 240–246. [Google Scholar] [CrossRef]
- Huang, S.; Czech, M.P. The GLUT4 glucose transporter. Cell Metab. 2007, 5, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, G.; Wu, Z.; Farrell, R.J.; Ryan, T.A. GLUT4 Mobilization Supports Energetic Demands of Active Synapses. Neuron 2017, 93, 606–615. [Google Scholar] [CrossRef]
- McNay, E.C.; Pearson-Leary, J. GluT4: A central player in hippocampal memory and brain insulin resistance. Exp. Neurol. 2020, 323, 113076. [Google Scholar] [CrossRef]
- Vannucci, S.J.; Rutherford, T.; Wilkie, M.B.; Simpson, I.A.; Lauder, J.M. Prenatal expression of the GLUT4 glucose transporter in the mouse. Dev. Neurosci. 2000, 22, 274–282. [Google Scholar] [CrossRef]
- Szablewski, L. Distribution of glucose transporters in renal diseases. J. Biomed. Sci. 2017, 24, 64. [Google Scholar] [CrossRef]
- Wright, E.M. Glucose transport families SLC5 and SLC50. Mol. Asp. Med. 2013, 34, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Diez-Sampedro, A.; Hirayama, B.A.; Osswald, C.; Gorboulev, V.; Baumgarten, K.; Volk, C.; Wright, E.M.; Koepsell, H. A glucose sensor hiding in a family of transporters. Proc. Natl. Acad. Sci. USA 2003, 100, 11753–11758. [Google Scholar] [CrossRef]
- Balen, D.; Ljubojevic, M.; Breljak, D.; Brzica, H.; Zlender, V.; Koepsell, H.; Sabolic, I. Revised immunolocalization of the Na+-D-glucose cotransporter SGLT1 in rat organs with an improved antibody. Am. J. Physiol. Cell Physiol. 2008, 295, C475–C489. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E. Sodium-Glucose Co-transporters and Their Inhibition: Clinical Physiology. Cell Metab. 2017, 26, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, C.; Loo, D.D.F.; Wright, E.M. Physiology of renal glucose handling via SGLT1, SGLT2 and GLUT2. Diabetologia 2018, 61, 2087–2097. [Google Scholar] [CrossRef]
- Sabolic, I.; Vrhovac, I.; Eror, D.B.; Gerasimova, M.; Rose, M.; Breljak, D.; Ljubojevic, M.; Brzica, H.; Sebastiani, A.; Thal, S.C.; et al. Expression of Na+-D-glucose cotransporter SGLT2 in rodents is kidney-specific and exhibits sex and species differences. Am. J. Physiol. Cell Physiol. 2012, 302, C1174–C1188. [Google Scholar] [CrossRef]
- Yu, A.S.; Hirayama, B.A.; Timbol, G.; Liu, J.; Basarah, E.; Kepe, V.; Satyamurthy, N.; Huang, S.C.; Wright, E.M.; Barrio, J.R. Functional expression of SGLTs in rat brain. Am. J. Physiol. Cell Physiol. 2010, 299, C1277–C1284. [Google Scholar] [CrossRef]
- Yu, A.S.; Hirayama, B.A.; Timbol, G.; Liu, J.; Diez-Sampedro, A.; Kepe, V.; Satyamurthy, N.; Huang, S.C.; Wright, E.M.; Barrio, J.R. Regional distribution of SGLT activity in rat brain in vivo. Am. J. Physiol. Cell Physiol. 2013, 304, C240–C247. [Google Scholar] [CrossRef]
- Elfeber, K.; Kohler, A.; Lutzenburg, M.; Osswald, C.; Galla, H.J.; Witte, O.W.; Koepsell, H. Localization of the Na+-D-glucose cotransporter SGLT1 in the blood-brain barrier. Histochem. Cell Biol. 2004, 121, 201–207. [Google Scholar] [CrossRef]
- Vemula, S.; Roder, K.E.; Yang, T.; Bhat, G.J.; Thekkumkara, T.J.; Abbruscato, T.J. A functional role for sodium-dependent glucose transport across the blood-brain barrier during oxygen glucose deprivation. J. Pharmacol. Exp. Ther. 2009, 328, 487–495. [Google Scholar] [CrossRef]
- Al-Ahmad, A.J. Comparative study of expression and activity of glucose transporters between stem cell-derived brain microvascular endothelial cells and hCMEC/D3 cells. Am. J. Physiol. Cell Physiol. 2017, 313, C421–C429. [Google Scholar] [CrossRef] [PubMed]
- Sala-Rabanal, M.; Hirayama, B.A.; Ghezzi, C.; Liu, J.; Huang, S.C.; Kepe, V.; Koepsell, H.; Yu, A.; Powell, D.R.; Thorens, B.; et al. Revisiting the physiological roles of SGLTs and GLUTs using positron emission tomography in mice. J. Physiol. 2016, 594, 4425–4438. [Google Scholar] [CrossRef] [PubMed]
- Chiba, Y.; Sugiyama, Y.; Nishi, N.; Nonaka, W.; Murakami, R.; Ueno, M. Sodium/glucose cotransporter 2 is expressed in choroid plexus epithelial cells and ependymal cells in human and mouse brains. Neuropathology 2020. [Google Scholar] [CrossRef] [PubMed]
- Liddelow, S.A.; Temple, S.; Mollgard, K.; Gehwolf, R.; Wagner, A.; Bauer, H.; Bauer, H.C.; Phoenix, T.N.; Dziegielewska, K.M.; Saunders, N.R. Molecular characterisation of transport mechanisms at the developing mouse blood-CSF interface: A transcriptome approach. PLoS ONE 2012, 7, e33554. [Google Scholar] [CrossRef]
- Kanai, Y.; Lee, W.S.; You, G.; Brown, D.; Hediger, M.A. The human kidney low affinity Na+/glucose cotransporter SGLT2. Delineation of the major renal reabsorptive mechanism for D-glucose. J. Clin. Investig. 1994, 93, 397–404. [Google Scholar] [CrossRef]
- Vallon, V.; Platt, K.A.; Cunard, R.; Schroth, J.; Whaley, J.; Thomson, S.C.; Koepsell, H.; Rieg, T. SGLT2 mediates glucose reabsorption in the early proximal tubule. J. Am. Soc. Nephrol. 2011, 22, 104–112. [Google Scholar] [CrossRef]
- Vrhovac, I.; Balen Eror, D.; Klessen, D.; Burger, C.; Breljak, D.; Kraus, O.; Radovic, N.; Jadrijevic, S.; Aleksic, I.; Walles, T.; et al. Localizations of Na(+)-D-glucose cotransporters SGLT1 and SGLT2 in human kidney and of SGLT1 in human small intestine, liver, lung, and heart. Pflugers Arch. 2015, 467, 1881–1898. [Google Scholar] [CrossRef]
- American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019, 42, S90–S102. [Google Scholar] [CrossRef]
- Dandona, P.; Chaudhuri, A. Sodium-glucose co-transporter 2 inhibitors for type 2 diabetes mellitus: An overview for the primary care physician. Int. J. Clin. Pract. 2017, 71, e12937. [Google Scholar] [CrossRef]
- Bonner, C.; Kerr-Conte, J.; Gmyr, V.; Queniat, G.; Moerman, E.; Thevenet, J.; Beaucamps, C.; Delalleau, N.; Popescu, I.; Malaisse, W.J.; et al. Inhibition of the glucose transporter SGLT2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat. Med. 2015, 21, 512–517. [Google Scholar] [CrossRef]
- Enerson, B.E.; Drewes, L.R. The rat blood-brain barrier transcriptome. J. Cereb. Blood Flow Metab. 2006, 26, 959–973. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.M.; Loo, D.D.; Hirayama, B.A.; Turk, E. Surprising versatility of Na+-glucose cotransporters: SLC5. Physiology 2004, 19, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, M.A.; Yusuf, D.; Christy, J.; Solmaz, V.; Erdogan, A.; Taskiran, E.; Erbas, O. Highly selective SGLT2 inhibitor dapagliflozin reduces seizure activity in pentylenetetrazol-induced murine model of epilepsy. BMC Neurol. 2018, 18, 81. [Google Scholar] [CrossRef] [PubMed]
- Melo, I.S.; Santos, Y.M.O.; Costa, M.A.; Pacheco, A.L.D.; Silva, N.; Cardoso-Sousa, L.; Pereira, U.P.; Goulart, L.R.; Garcia-Cairasco, N.; Duzzioni, M.; et al. Inhibition of sodium glucose cotransporters following status epilepticus induced by intrahippocampal pilocarpine affects neurodegeneration process in hippocampus. Epilepsy Behav. 2016, 61, 258–268. [Google Scholar] [CrossRef]
- Oerter, S.; Forster, C.; Bohnert, M. Validation of sodium/glucose cotransporter proteins in human brain as a potential marker for temporal narrowing of the trauma formation. Int. J. Legal Med. 2019, 133, 1107–1114. [Google Scholar] [CrossRef]
- Kepe, V.; Scafoglio, C.; Liu, J.; Yong, W.H.; Bergsneider, M.; Huang, S.C.; Barrio, J.R.; Wright, E.M. Positron emission tomography of sodium glucose cotransport activity in high grade astrocytomas. J. Neurooncol. 2018, 138, 557–569. [Google Scholar] [CrossRef]
- Chatard, C.; Sabac, A.; Moreno-Velasquez, L.; Meiller, A.; Marinesco, S. Minimally Invasive Microelectrode Biosensors Based on Platinized Carbon Fibers for in Vivo Brain Monitoring. ACS Cent. Sci. 2018, 4, 1751–1760. [Google Scholar] [CrossRef]
- Deane, R.; Segal, M.B. The transport of sugars across the perfused choroid plexus of the sheep. J. Physiol. 1985, 362, 245–260. [Google Scholar] [CrossRef]
- Steffensen, A.B.; Zeuthen, T. Cotransport of Water in the Choroid Plexus Epithelium: From Amphibians to Mammals. In Role of the Choroid Plexus in Health and Disease; Praetorius, J., Blazer-Yost, B., Damkier, H., Eds.; Springer: New York, NY, USA, 2020; pp. 99–124. [Google Scholar] [CrossRef]
- Sasseville, L.J.; Cuervo, J.E.; Lapointe, J.Y.; Noskov, S.Y. The structural pathway for water permeation through sodium-glucose cotransporters. Biophys. J. 2011, 101, 1887–1895. [Google Scholar] [CrossRef][Green Version]
- Balmaceda-Aguilera, C.; Cortes-Campos, C.; Cifuentes, M.; Peruzzo, B.; Mack, L.; Tapia, J.C.; Oyarce, K.; Garcia, M.A.; Nualart, F. Glucose transporter 1 and monocarboxylate transporters 1, 2, and 4 localization within the glial cells of shark blood-brain-barriers. PLoS ONE 2012, 7, e32409. [Google Scholar] [CrossRef]
- Van Cauwenberghe, C.; Gorle, N.; Vandenbroucke, R.E. Roles of the Choroid Plexus in Aging. In Role of the Choroid Plexus in Health and Disease; Praetorius, J., Blazer-Yost, B., Damkier, H., Eds.; Springer: New York, NY, USA, 2020; pp. 209–232. [Google Scholar] [CrossRef]
- Marques, F.; Sousa, J.C.; Brito, M.A.; Pahnke, J.; Santos, C.; Correia-Neves, M.; Palha, J.A. The choroid plexus in health and in disease: Dialogues into and out of the brain. Neurobiol. Dis. 2017, 107, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Kaur, C.; Rathnasamy, G.; Ling, E.A. The Choroid Plexus in Healthy and Diseased Brain. J. Neuropathol. Exp. Neurol. 2016, 75, 198–213. [Google Scholar] [CrossRef] [PubMed]
- Serot, J.M.; Bene, M.C.; Faure, G.C. Choroid plexus, aging of the brain, and Alzheimer’s disease. Front. Biosci. 2003, 8, s515–s521. [Google Scholar] [CrossRef] [PubMed]
- Balusu, S.; Brkic, M.; Libert, C.; Vandenbroucke, R.E. The choroid plexus-cerebrospinal fluid interface in Alzheimer’s disease: More than just a barrier. Neural Regen. Res. 2016, 11, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Emerich, D.F.; Schneider, P.; Bintz, B.; Hudak, J.; Thanos, C.G. Aging reduces the neuroprotective capacity, VEGF secretion, and metabolic activity of rat choroid plexus epithelial cells. Cell Transplant. 2007, 16, 697–705. [Google Scholar] [CrossRef]
- Cottrell, D.A.; Blakely, E.L.; Johnson, M.A.; Ince, P.G.; Borthwick, G.M.; Turnbull, D.M. Cytochrome c oxidase deficient cells accumulate in the hippocampus and choroid plexus with age. Neurobiol. Aging 2001, 22, 265–272. [Google Scholar] [CrossRef]
- Gorle, N.; Van Cauwenberghe, C.; Libert, C.; Vandenbroucke, R.E. The effect of aging on brain barriers and the consequences for Alzheimer’s disease development. Mamm. Genome 2016, 27, 407–420. [Google Scholar] [CrossRef]
- Emerich, D.F.; Skinner, S.J.; Borlongan, C.V.; Vasconcellos, A.V.; Thanos, C.G. The choroid plexus in the rise, fall and repair of the brain. Bioessays 2005, 27, 262–274. [Google Scholar] [CrossRef]
- Gonzalez-Marrero, I.; Gimenez-Llort, L.; Johanson, C.E.; Carmona-Calero, E.M.; Castaneyra-Ruiz, L.; Brito-Armas, J.M.; Castaneyra-Perdomo, A.; Castro-Fuentes, R. Choroid plexus dysfunction impairs beta-amyloid clearance in a triple transgenic mouse model of Alzheimer’s disease. Front. Cell Neurosci. 2015, 9, 17. [Google Scholar] [CrossRef]
- Brkic, M.; Balusu, S.; Van Wonterghem, E.; Gorle, N.; Benilova, I.; Kremer, A.; Van Hove, I.; Moons, L.; De Strooper, B.; Kanazir, S.; et al. Amyloid beta Oligomers Disrupt Blood-CSF Barrier Integrity by Activating Matrix Metalloproteinases. J. Neurosci. 2015, 35, 12766–12778. [Google Scholar] [CrossRef]
- Daouk, J.; Bouzerar, R.; Chaarani, B.; Zmudka, J.; Meyer, M.E.; Baledent, O. Use of dynamic (18)F-fluorodeoxyglucose positron emission tomography to investigate choroid plexus function in Alzheimer’s disease. Exp. Gerontol. 2016, 77, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Yamamoto, H.; Toda, M.; Hagiwara, S.; Iguchi, K.; Hoshino, M.; Takabayashi, F.; Hasegawa-Ishii, S.; Shimada, A.; Hosokawa, M.; et al. Novel frame-shift mutation in Slc5a2 encoding SGLT2 in a strain of senescence-accelerated mouse SAMP10. Biochem. Biophys. Res. Commun. 2014, 454, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Takagi, Y.; Konishi, T.; Suzuki, M.; Miyake, A.; Kurotaki, T.; Hase, T.; Meguro, S.; Shimada, A.; Hasegawa-Ishii, S.; et al. Mutation in Sodium-Glucose Cotransporter 2 Results in Down-Regulation of Amyloid Beta (A4) Precursor-Like Protein 1 in Young Age, Which May Lead to Poor Memory Retention in Old Age. Int. J. Mol. Sci. 2020, 21, 5579. [Google Scholar] [CrossRef]
- Myung, J.; Schmal, C.; Hong, S.; Tsukizawa, Y.; Rose, P.; Zhang, Y.; Holtzman, M.J.; De Schutter, E.; Herzel, H.; Bordyugov, G.; et al. The choroid plexus is an important circadian clock component. Nat. Commun. 2018, 9, 1062. [Google Scholar] [CrossRef] [PubMed]
- Stephan, B.C.; Wells, J.C.; Brayne, C.; Albanese, E.; Siervo, M. Increased fructose intake as a risk factor for dementia. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2010, 65, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Lowette, K.; Roosen, L.; Tack, J.; Vanden Berghe, P. Effects of high-fructose diets on central appetite signaling and cognitive function. Front. Nutr. 2015, 2, 5. [Google Scholar] [CrossRef]
- Dills, W.L., Jr. Protein fructosylation: Fructose and the Maillard reaction. Am. J. Clin. Nutr. 1993, 58, 779S–787S. [Google Scholar] [CrossRef]
- Semchyshyn, H.M.; Miedzobrodzki, J.; Bayliak, M.M.; Lozinska, L.M.; Homza, B.V. Fructose compared with glucose is more a potent glycoxidation agent in vitro, but not under carbohydrate-induced stress in vivo: Potential role of antioxidant and antiglycation enzymes. Carbohydr. Res. 2014, 384, 61–69. [Google Scholar] [CrossRef]
- Lakhan, S.E.; Kirchgessner, A. The emerging role of dietary fructose in obesity and cognitive decline. Nutr. J. 2013, 12, 114. [Google Scholar] [CrossRef]
- Basaranoglu, M.; Basaranoglu, G.; Bugianesi, E. Carbohydrate intake and nonalcoholic fatty liver disease: Fructose as a weapon of mass destruction. Hepatobiliary Surg. Nutr. 2015, 4, 109–116. [Google Scholar] [CrossRef]
- Jimenez-Maldonado, A.; Ying, Z.; Byun, H.R.; Gomez-Pinilla, F. Short-term fructose ingestion affects the brain independently from establishment of metabolic syndrome. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 24–33. [Google Scholar] [CrossRef] [PubMed]
- van der Borght, K.; Kohnke, R.; Goransson, N.; Deierborg, T.; Brundin, P.; Erlanson-Albertsson, C.; Lindqvist, A. Reduced neurogenesis in the rat hippocampus following high fructose consumption. Regul. Pept. 2011, 167, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Cisternas, P.; Salazar, P.; Serrano, F.G.; Montecinos-Oliva, C.; Arredondo, S.B.; Varela-Nallar, L.; Barja, S.; Vio, C.P.; Gomez-Pinilla, F.; Inestrosa, N.C. Fructose consumption reduces hippocampal synaptic plasticity underlying cognitive performance. Biochim. Biophys. Acta 2015, 1852, 2379–2390. [Google Scholar] [CrossRef]
- Agrawal, R.; Noble, E.; Vergnes, L.; Ying, Z.; Reue, K.; Gomez-Pinilla, F. Dietary fructose aggravates the pathobiology of traumatic brain injury by influencing energy homeostasis and plasticity. J. Cereb. Blood Flow Metab. 2016, 36, 941–953. [Google Scholar] [CrossRef]
- Francis, H.; Stevenson, R. The longer-term impacts of Western diet on human cognition and the brain. Appetite 2013, 63, 119–128. [Google Scholar] [CrossRef]
- Ye, X.; Gao, X.; Scott, T.; Tucker, K.L. Habitual sugar intake and cognitive function among middle-aged and older Puerto Ricans without diabetes. Br. J. Nutr. 2011, 106, 1423–1432. [Google Scholar] [CrossRef]
- Reichelt, A.C.; Stoeckel, L.E.; Reagan, L.P.; Winstanley, C.A.; Page, K.A. Dietary influences on cognition. Physiol. Behav. 2018, 192, 118–126. [Google Scholar] [CrossRef]
- Douard, V.; Ferraris, R.P. Regulation of the fructose transporter GLUT5 in health and disease. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E227–E237. [Google Scholar] [CrossRef] [PubMed]
- Mantych, G.J.; James, D.E.; Devaskar, S.U. Jejunal/kidney glucose transporter isoform (Glut-5) is expressed in the human blood-brain barrier. Endocrinology 1993, 132, 35–40. [Google Scholar] [CrossRef]
- Oldendorf, W.H. Brain uptake of radiolabeled amino acids, amines, and hexoses after arterial injection. Am. J. Physiol. 1971, 221, 1629–1639. [Google Scholar] [CrossRef]
- Kayano, T.; Burant, C.F.; Fukumoto, H.; Gould, G.W.; Fan, Y.S.; Eddy, R.L.; Byers, M.G.; Shows, T.B.; Seino, S.; Bell, G.I. Human facilitative glucose transporters. Isolation, functional characterization, and gene localization of cDNAs encoding an isoform (GLUT5) expressed in small intestine, kidney, muscle, and adipose tissue and an unusual glucose transporter pseudogene-like sequence (GLUT6). J. Biol. Chem. 1990, 265, 13276–13282. [Google Scholar] [PubMed]
- Horikoshi, Y.; Sasaki, A.; Taguchi, N.; Maeda, M.; Tsukagoshi, H.; Sato, K.; Yamaguchi, H. Human GLUT5 immunolabeling is useful for evaluating microglial status in neuropathological study using paraffin sections. Acta Neuropathol. 2003, 105, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Kojo, A.; Yamada, K.; Yamamoto, T. Glucose transporter 5 (GLUT5)-like immunoreactivity is localized in subsets of neurons and glia in the rat brain. J. Chem. Neuroanat. 2016, 74, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Oppelt, S.A.; Zhang, W.; Tolan, D.R. Specific regions of the brain are capable of fructose metabolism. Brain Res. 2017, 1657, 312–322. [Google Scholar] [CrossRef]
- Shu, H.J.; Isenberg, K.; Cormier, R.J.; Benz, A.; Zorumski, C.F. Expression of fructose sensitive glucose transporter in the brains of fructose-fed rats. Neuroscience 2006, 140, 889–895. [Google Scholar] [CrossRef]
- Shah, K.; Desilva, S.; Abbruscato, T. The role of glucose transporters in brain disease: Diabetes and Alzheimer’s Disease. Int. J. Mol. Sci. 2012, 13, 12629–12655. [Google Scholar] [CrossRef]
- Ueno, M.; Nishi, N.; Nakagawa, T.; Chiba, Y.; Tsukamoto, I.; Kusaka, T.; Miki, T.; Sakamoto, H.; Yamaguchi, F.; Tokuda, M. Immunoreactivity of glucose transporter 5 is located in epithelial cells of the choroid plexus and ependymal cells. Neuroscience 2014, 260, 149–157. [Google Scholar] [CrossRef]
- Mashima, M.; Chiba, Y.; Murakami, R.; Uemura, N.; Matsumoto, K.; Kawauchi, M.; Kanenishi, K.; Hata, T.; Ueno, M. Glucose transporter 8 immunoreactivity in astrocytic and microglial cells in subependymal areas of human brains. Neurosci. Lett. 2017, 636, 90–94. [Google Scholar] [CrossRef]
- Schmidt, S.; Joost, H.G.; Schurmann, A. GLUT8, the enigmatic intracellular hexose transporter. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E614–E618. [Google Scholar] [CrossRef]
- Mayer, A.L.; Higgins, C.B.; Heitmeier, M.R.; Kraft, T.E.; Qian, X.; Crowley, J.R.; Hyrc, K.L.; Beatty, W.L.; Yarasheski, K.E.; Hruz, P.W.; et al. SLC2A8 (GLUT8) is a mammalian trehalose transporter required for trehalose-induced autophagy. Sci. Rep. 2016, 6, 38586. [Google Scholar] [CrossRef] [PubMed]
- Narita, H.; Tanji, K.; Miki, Y.; Mori, F.; Wakabayashi, K. Trehalose intake and exercise upregulate a glucose transporter, GLUT8, in the brain. Biochem. Biophys. Res. Commun. 2019, 514, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Reagan, L.P.; Rosell, D.R.; Alves, S.E.; Hoskin, E.K.; McCall, A.L.; Charron, M.J.; McEwen, B.S. GLUT8 glucose transporter is localized to excitatory and inhibitory neurons in the rat hippocampus. Brain Res. 2002, 932, 129–134. [Google Scholar] [CrossRef]
- Schmidt, S.; Gawlik, V.; Holter, S.M.; Augustin, R.; Scheepers, A.; Behrens, M.; Wurst, W.; Gailus-Durner, V.; Fuchs, H.; Hrabe de Angelis, M.; et al. Deletion of glucose transporter GLUT8 in mice increases locomotor activity. Behav. Genet. 2008, 38, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Membrez, M.; Hummler, E.; Beermann, F.; Haefliger, J.A.; Savioz, R.; Pedrazzini, T.; Thorens, B. GLUT8 is dispensable for embryonic development but influences hippocampal neurogenesis and heart function. Mol. Cell Biol. 2006, 26, 4268–4276. [Google Scholar] [CrossRef] [PubMed]
- Grempler, R.; Augustin, R.; Froehner, S.; Hildebrandt, T.; Simon, E.; Mark, M.; Eickelmann, P. Functional characterisation of human SGLT-5 as a novel kidney-specific sodium-dependent sugar transporter. FEBS Lett. 2012, 586, 248–253. [Google Scholar] [CrossRef]
- Gonzalez-Vicente, A.; Cabral, P.D.; Hong, N.J.; Asirwatham, J.; Saez, F.; Garvin, J.L. Fructose reabsorption by rat proximal tubules: Role of Na(+)-linked cotransporters and the effect of dietary fructose. Am. J. Physiol. Renal Physiol. 2019, 316, F473–F480. [Google Scholar] [CrossRef]
- Kawasaki, T.; Akanuma, H.; Yamanouchi, T. Increased fructose concentrations in blood and urine in patients with diabetes. Diabetes Care 2002, 25, 353–357. [Google Scholar] [CrossRef]
- Lutz, N.W.; Viola, A.; Malikova, I.; Confort-Gouny, S.; Audoin, B.; Ranjeva, J.P.; Pelletier, J.; Cozzone, P.J. Inflammatory multiple-sclerosis plaques generate characteristic metabolic profiles in cerebrospinal fluid. PLoS ONE 2007, 2, e595. [Google Scholar] [CrossRef]
- Hwang, J.J.; Johnson, A.; Cline, G.; Belfort-DeAguiar, R.; Snegovskikh, D.; Khokhar, B.; Han, C.S.; Sherwin, R.S. Fructose levels are markedly elevated in cerebrospinal fluid compared to plasma in pregnant women. PLoS ONE 2015, 10, e0128582. [Google Scholar] [CrossRef]
- Hwang, J.J.; Jiang, L.; Hamza, M.; Dai, F.; Belfort-DeAguiar, R.; Cline, G.; Rothman, D.L.; Mason, G.; Sherwin, R.S. The human brain produces fructose from glucose. JCI Insight 2017, 2, e90508. [Google Scholar] [CrossRef]
- Miyamoto, T.; Slone, J.; Song, X.; Amrein, H. A fructose receptor functions as a nutrient sensor in the Drosophila brain. Cell 2012, 151, 1113–1125. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.H.; Wolfgang, M.; Tokutake, Y.; Chohnan, S.; Lane, M.D. Differential effects of central fructose and glucose on hypothalamic malonyl-CoA and food intake. Proc. Natl. Acad. Sci. USA 2008, 105, 16871–16875. [Google Scholar] [CrossRef]
- Funari, V.A.; Crandall, J.E.; Tolan, D.R. Fructose metabolism in the cerebellum. Cerebellum 2007, 6, 130–140. [Google Scholar] [CrossRef]
- Rouillard, A.D.; Gundersen, G.W.; Fernandez, N.F.; Wang, Z.; Monteiro, C.D.; McDermott, M.G.; Ma’ayan, A. The harmonizome: A collection of processed datasets gathered to serve and mine knowledge about genes and proteins. Database 2016, 2016, baw100. [Google Scholar] [CrossRef] [PubMed]
- Cortese, F.; Giordano, P.; Scicchitano, P.; Faienza, M.F.; De Pergola, G.; Calculli, G.; Meliota, G.; Ciccone, M.M. Uric acid: From a biological advantage to a potential danger. A focus on cardiovascular effects. Vascul. Pharmacol. 2019, 120, 106565. [Google Scholar] [CrossRef] [PubMed]
- Keenan, R.T. The biology of urate. Semin. Arthritis Rheum. 2020, 50, S2–S10. [Google Scholar] [CrossRef]
- El Ridi, R.; Tallima, H. Physiological functions and pathogenic potential of uric acid: A review. J. Adv. Res. 2017, 8, 487–493. [Google Scholar] [CrossRef]
- Bjornstad, P.; Lanaspa, M.A.; Ishimoto, T.; Kosugi, T.; Kume, S.; Jalal, D.; Maahs, D.M.; Snell-Bergeon, J.K.; Johnson, R.J.; Nakagawa, T. Fructose and uric acid in diabetic nephropathy. Diabetologia 2015, 58, 1993–2002. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Maiuolo, J.; Oppedisano, F.; Gratteri, S.; Muscoli, C.; Mollace, V. Regulation of uric acid metabolism and excretion. Int. J. Cardiol. 2016, 213, 8–14. [Google Scholar] [CrossRef]
- Engel, B.; Gomm, W.; Broich, K.; Maier, W.; Weckbecker, K.; Haenisch, B. Hyperuricemia and dementia—A case-control study. BMC Neurol. 2018, 18, 131. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wei, X.; Yang, W.; Reynolds, G.P. Agricultural work and reduced circulating uric acid are both associated with initial hospital admission for Parkinson’s disease. J. Neural Transm. 2020, 127, 779–783. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hu, W.; Wang, J.; Qian, W.; Xiao, H. Low serum uric acid levels in patients with multiple sclerosis and neuromyelitis optica: An updated meta-analysis. Mult. Scler. Relat. Disord. 2016, 9, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Bakshi, R.; Xu, Y.; Mueller, K.A.; Chen, X.; Granucci, E.; Paganoni, S.; Sadri-Vakili, G.; Schwarzschild, M.A. Urate mitigates oxidative stress and motor neuron toxicity of astrocytes derived from ALS-linked SOD1(G93A) mutant mice. Mol. Cell Neurosci. 2018, 92, 12–16. [Google Scholar] [CrossRef]
- Afsar, B.; Elsurer, R.; Covic, A.; Johnson, R.J.; Kanbay, M. Relationship between uric acid and subtle cognitive dysfunction in chronic kidney disease. Am. J. Nephrol. 2011, 34, 49–54. [Google Scholar] [CrossRef]
- Verhaaren, B.F.; Vernooij, M.W.; Dehghan, A.; Vrooman, H.A.; de Boer, R.; Hofman, A.; Witteman, J.C.; Niessen, W.J.; Breteler, M.M.; van der Lugt, A.; et al. The relation of uric acid to brain atrophy and cognition: The Rotterdam Scan Study. Neuroepidemiology 2013, 41, 29–34. [Google Scholar] [CrossRef]
- Shao, X.; Lu, W.; Gao, F.; Li, D.; Hu, J.; Li, Y.; Zuo, Z.; Jie, H.; Zhao, Y.; Cen, X. Uric Acid Induces Cognitive Dysfunction through Hippocampal Inflammation in Rodents and Humans. J. Neurosci. 2016, 36, 10990–11005. [Google Scholar] [CrossRef]
- Uemura, N.; Murakami, R.; Chiba, Y.; Yanase, K.; Fujihara, R.; Mashima, M.; Matsumoto, K.; Kawauchi, M.; Shirakami, G.; Ueno, M. Immunoreactivity of urate transporters, GLUT9 and URAT1, is located in epithelial cells of the choroid plexus of human brains. Neurosci. Lett. 2017, 659, 99–103. [Google Scholar] [CrossRef]
- Tomioka, N.H.; Tamura, Y.; Takada, T.; Shibata, S.; Suzuki, H.; Uchida, S.; Hosoyamada, M. Immunohistochemical and in situ hybridization study of urate transporters GLUT9/URATv1, ABCG2, and URAT1 in the murine brain. Fluids Barriers CNS 2016, 13, 22. [Google Scholar] [CrossRef]
- Tomioka, N.H.; Nakamura, M.; Doshi, M.; Deguchi, Y.; Ichida, K.; Morisaki, T.; Hosoyamada, M. Ependymal cells of the mouse brain express urate transporter 1 (URAT1). Fluids Barriers CNS 2013, 10, 31. [Google Scholar] [CrossRef]
- Vitart, V.; Rudan, I.; Hayward, C.; Gray, N.K.; Floyd, J.; Palmer, C.N.; Knott, S.A.; Kolcic, I.; Polasek, O.; Graessler, J.; et al. SLC2A9 is a newly identified urate transporter influencing serum urate concentration, urate excretion and gout. Nat. Genet. 2008, 40, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Anzai, N.; Ichida, K.; Jutabha, P.; Kimura, T.; Babu, E.; Jin, C.J.; Srivastava, S.; Kitamura, K.; Hisatome, I.; Endou, H.; et al. Plasma urate level is directly regulated by a voltage-driven urate efflux transporter URATv1 (SLC2A9) in humans. J. Biol. Chem. 2008, 283, 26834–26838. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.K.; Mount, D.B. The molecular physiology of uric acid homeostasis. Annu. Rev. Physiol. 2015, 77, 323–345. [Google Scholar] [CrossRef] [PubMed]
- Döring, A.; Gieger, C.; Mehta, D.; Gohlke, H.; Prokisch, H.; Coassin, S.; Fischer, G.; Henke, K.; Klopp, N.; Kronenberg, F.; et al. SLC2A9 influences uric acid concentrations with pronounced sex-specific effects. Nat. Genet. 2008, 40, 430–436. [Google Scholar] [CrossRef]
- Bi, M.; Jiao, Q.; Du, X.; Jiang, H. Glut9-mediated Urate Uptake Is Responsible for Its Protective Effects on Dopaminergic Neurons in Parkinson’s Disease Models. Front. Mol. Neurosci. 2018, 11, 21. [Google Scholar] [CrossRef]
- Li, J.; Badve, S.V.; Zhou, Z.; Rodgers, A.; Day, R.; Oh, R.; Lee, M.; Perkovic, V.; de Zeeuw, D.; Mahaffey, K.W.; et al. The effects of canagliflozin on gout in type 2 diabetes: A post-hoc analysis of the CANVAS Program. Lancet Rheumatol. 2019, 1, e220–e228. [Google Scholar] [CrossRef]
- Bailey, C.J. Uric acid and the cardio-renal effects of SGLT2 inhibitors. Diabetes Obes. Metab. 2019, 21, 1291–1298. [Google Scholar] [CrossRef]
- Chino, Y.; Samukawa, Y.; Sakai, S.; Nakai, Y.; Yamaguchi, J.; Nakanishi, T.; Tamai, I. SGLT2 inhibitor lowers serum uric acid through alteration of uric acid transport activity in renal tubule by increased glycosuria. Biopharm. Drug Dispos. 2014, 35, 391–404. [Google Scholar] [CrossRef]
- Nigam, S.K. The SLC22 Transporter Family: A Paradigm for the Impact of Drug Transporters on Metabolic Pathways, Signaling, and Disease. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 663–687. [Google Scholar] [CrossRef]
- Enomoto, A.; Kimura, H.; Chairoungdua, A.; Shigeta, Y.; Jutabha, P.; Cha, S.H.; Hosoyamada, M.; Takeda, M.; Sekine, T.; Igarashi, T.; et al. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 2002, 417, 447–452. [Google Scholar] [CrossRef]
- Fagerberg, L.; Hallstrom, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Kusuhara, H.; Adachi-Akahane, S.; Hasegawa, M.; Morita, N.; Endou, H.; Sugiyama, Y. The renal-specific transporter mediates facilitative transport of organic anions at the brush border membrane of mouse renal tubules. J. Am. Soc. Nephrol. 2004, 15, 2012–2022. [Google Scholar] [CrossRef] [PubMed]
- Do, T.M.; Noel-Hudson, M.S.; Ribes, S.; Besengez, C.; Smirnova, M.; Cisternino, S.; Buyse, M.; Calon, F.; Chimini, G.; Chacun, H.; et al. ABCG2- and ABCG4-mediated efflux of amyloid-beta peptide 1-40 at the mouse blood-brain barrier. J. Alzheimers Dis. 2012, 30, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, Y.; Uchida, Y.; Tachikawa, M.; Inoue, T.; Ohtsuki, S.; Terasaki, T. Quantitative atlas of blood-brain barrier transporters, receptors, and tight junction proteins in rats and common marmoset. J. Pharm. Sci. 2013, 102, 3343–3355. [Google Scholar] [CrossRef]
- Tachikawa, M.; Watanabe, M.; Hori, S.; Fukaya, M.; Ohtsuki, S.; Asashima, T.; Terasaki, T. Distinct spatio-temporal expression of ABCA and ABCG transporters in the developing and adult mouse brain. J. Neurochem. 2005, 95, 294–304. [Google Scholar] [CrossRef]
- Cooray, H.C.; Blackmore, C.G.; Maskell, L.; Barrand, M.A. Localisation of breast cancer resistance protein in microvessel endothelium of human brain. Neuroreport 2002, 13, 2059–2063. [Google Scholar] [CrossRef]
- Ito, K.; Uchida, Y.; Ohtsuki, S.; Aizawa, S.; Kawakami, H.; Katsukura, Y.; Kamiie, J.; Terasaki, T. Quantitative membrane protein expression at the blood-brain barrier of adult and younger cynomolgus monkeys. J. Pharm. Sci. 2011, 100, 3939–3950. [Google Scholar] [CrossRef]
- Uchida, Y.; Ohtsuki, S.; Katsukura, Y.; Ikeda, C.; Suzuki, T.; Kamiie, J.; Terasaki, T. Quantitative targeted absolute proteomics of human blood-brain barrier transporters and receptors. J. Neurochem. 2011, 117, 333–345. [Google Scholar] [CrossRef]
- Ghersi-Egea, J.F.; Vasiljevic, A.; Blondel, S.; Strazielle, N. Neuroprotective Mechanisms at the Blood-CSF Barrier of the Developing and Adult Brain. In Role of the Choroid Plexus in Health and Disease; Praetorius, J., Blazer-Yost, B., Damkier, H., Eds.; Springer: New York, NY, USA, 2020; pp. 193–207. [Google Scholar] [CrossRef]
- Mollgard, K.; Dziegielewska, K.M.; Holst, C.B.; Habgood, M.D.; Saunders, N.R. Brain barriers and functional interfaces with sequential appearance of ABC efflux transporters during human development. Sci. Rep. 2017, 7, 11603. [Google Scholar] [CrossRef]
- Bowman, G.L.; Shannon, J.; Frei, B.; Kaye, J.A.; Quinn, J.F. Uric acid as a CNS antioxidant. J. Alzheimers Dis. 2010, 19, 1331–1336. [Google Scholar] [CrossRef]
- Shu, Y.; Li, H.; Zhang, L.; Wang, Y.; Long, Y.; Li, R.; Qiu, W.; Lu, Z.; Hu, X.; Peng, F. Elevated cerebrospinal fluid uric acid during relapse of neuromyelitis optica spectrum disorders. Brain Behav. 2017, 7, e00584. [Google Scholar] [CrossRef] [PubMed]
- Walter, G.A.; Phillis, J.W.; O’Reagan, M.H. Determination of rat cerebrospinal fluid concentrations of adenosine, inosine, hypoxanthine, xanthine and uric acid by high performance liquid chromatography. J. Pharm. Pharmacol. 1988, 40, 140–142. [Google Scholar] [CrossRef] [PubMed]
- De Luca, M.A.; Cauli, O.; Morelli, M.; Simola, N. Elevation of striatal urate in experimental models of Parkinson’s disease: A compensatory mechanism triggered by dopaminergic nigrostriatal degeneration? J. Neurochem. 2014, 131, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Pavelka, M.; Roth, J. Basal Labyrinth. In Functional Ultrastructure; Springer: Vienna, Austria, 2010; pp. 178–179. [Google Scholar] [CrossRef]
- Spector, R.; Johanson, C. Micronutrient and urate transport in choroid plexus and kidney: Implications for drug therapy. Pharm. Res. 2006, 23, 2515–2524. [Google Scholar] [CrossRef]
- Christensen, I.B.; Mogensen, E.N.; Damkier, H.H.; Praetorius, J. Choroid plexus epithelial cells express the adhesion protein P-cadherin at cell-cell contacts and syntaxin-4 in the luminal membrane domain. Am. J. Physiol. Cell Physiol. 2018, 314, C519–C533. [Google Scholar] [CrossRef]
- Huls, M.; Brown, C.D.; Windass, A.S.; Sayer, R.; van den Heuvel, J.J.; Heemskerk, S.; Russel, F.G.; Masereeuw, R. The breast cancer resistance protein transporter ABCG2 is expressed in the human kidney proximal tubule apical membrane. Kidney Int. 2008, 73, 220–225. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiba, Y.; Murakami, R.; Matsumoto, K.; Wakamatsu, K.; Nonaka, W.; Uemura, N.; Yanase, K.; Kamada, M.; Ueno, M. Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium. Int. J. Mol. Sci. 2020, 21, 7230. https://doi.org/10.3390/ijms21197230
Chiba Y, Murakami R, Matsumoto K, Wakamatsu K, Nonaka W, Uemura N, Yanase K, Kamada M, Ueno M. Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium. International Journal of Molecular Sciences. 2020; 21(19):7230. https://doi.org/10.3390/ijms21197230
Chicago/Turabian StyleChiba, Yoichi, Ryuta Murakami, Koichi Matsumoto, Keiji Wakamatsu, Wakako Nonaka, Naoya Uemura, Ken Yanase, Masaki Kamada, and Masaki Ueno. 2020. "Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium" International Journal of Molecular Sciences 21, no. 19: 7230. https://doi.org/10.3390/ijms21197230
APA StyleChiba, Y., Murakami, R., Matsumoto, K., Wakamatsu, K., Nonaka, W., Uemura, N., Yanase, K., Kamada, M., & Ueno, M. (2020). Glucose, Fructose, and Urate Transporters in the Choroid Plexus Epithelium. International Journal of Molecular Sciences, 21(19), 7230. https://doi.org/10.3390/ijms21197230




