Structural Studies of the Lipopolysaccharide Isolated from Plesiomonas shigelloides O22:H3 (CNCTC 90/89)
Abstract
1. Introduction
2. Results
2.1. P. Shigelloides O22:H3 (Strain CNCTC 90/89) Showed the Prevailing Smooth–Rough Phenotype
2.2. Structures of the O-Specific Polysaccharide and Core Oligosaccharide
2.3. Mass Spectrometry Analysis of the Core OS and OSRU Structures and a Glycine Substituent
3. Discussion
4. Materials and Methods
4.1. Bacteria
4.2. Lipopolysaccharide and Oligosaccharides OS and OSRU
4.3. Partial Acid Hydrolysis
4.4. Analytical Methods
4.5. NMR Spectroscopy
4.6. Mass Spectrometry
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| LPS | lipopolysaccharide |
| OS | oligosaccharide |
| OSRU | core oligosaccharide substituted by one repeating unit of O-specific polysaccharide |
| MALDI-TOF | matrix-assisted laser-desorption/ionisation time-of-flight |
| MS | mass spectrometry |
| NMR | nuclear magnetic resonance |
| COSY | correlated spectroscopy |
| TOCSY | total correlation spectroscopy |
| NOESY | nuclear Overhauser effect spectroscopy |
| HMBC | heteronuclear multiple bond correlation |
| HSQC | heteronuclear single quantum coherence |
| DEPT | distortionless enhancement by polarisation transfer |
References
- Janda, J.M.; Abbott, S.L.; McIver, C.J. Plesiomonas Shigelloides Revisited. Clin. Microbiol. Rev. 2016, 29, 349–374. [Google Scholar] [CrossRef]
- Schuetz, A.N. Emerging Agents of Gastroenteritis: Aeromonas, Plesiomonas, and the Diarrheagenic Pathotypes of Escherichia Coli. Semin. Diagn. Pathol. 2019, 36, 187–192. [Google Scholar] [CrossRef]
- Stock, I. Plesiomonas Shigelloides: An Emerging Pathogen with Unusual Properties. Rev. Med. Microbiol. 2004, 15, 129–139. [Google Scholar] [CrossRef]
- Xia, F.-Q.; Liu, P.-N.; Zhou, Y.-H. Meningoencephalitis Caused by Plesiomonas Shigelloides in a Chinese Neonate: Case Report and Literature Review. Ital. J. Pediatr. 2015, 41, 3. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.S.; McLaughlin, R.W.; Li, J.; Wan, X.; Liu, Y.; Xie, H.; Hao, Y.; Zheng, J. Putative Virulence Factors of Plesiomonas Shigelloides. Anton. Leeuw. Int. J. G. 2019, 112, 1815–1826. [Google Scholar] [CrossRef] [PubMed]
- Gardner, S.E.; Fowlston, S.E.; George, W.L. In Vitro Production of Cholera Toxin-like Activity by Plesiomonas Shigelloides. J. Infect. Dis. 1987, 156, 720–722. [Google Scholar] [CrossRef] [PubMed]
- Sears, C.L.; Kaper, J.B. Enteric Bacterial Toxins: Mechanisms of Action and Linkage to Intestinal Secretion. Microbiol. Rev. 1996, 60, 167–215. [Google Scholar] [CrossRef] [PubMed]
- Janda, J.M.; Abbott, S.L. Expression of Hemolytic Activity by Plesiomonas Shigelloides. J. Clin. Microbiol. 1993, 31, 1206–1208. [Google Scholar] [CrossRef]
- Okawa, Y.; Ohtomo, Y.; Tsugawa, H.; Matsuda, Y.; Kobayashi, H.; Tsukamoto, T. Isolation and Characterization of a Cytotoxin Produced by Plesiomonas Shigelloides P-1 Strain. FEMS Microbiol. Lett. 2004, 239, 125–130. [Google Scholar] [CrossRef]
- Theodoropoulos, C.; Wong, T.H.; O’Brien, M.; Stenzel, D. Plesiomonas Shigelloides Enters Polarized Human Intestinal Caco-2 Cells in an in Vitro Model System. Infect. Immun. 2001, 69, 2260–2269. [Google Scholar] [CrossRef]
- Aldová, E.; Shimada, T. New O and H Antigens of the International Antigenic Scheme for Plesiomonas Shigelloides. Folia. Microbiol. (Praha) 2000, 45, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Linnerborg, M.; Widmalm, G.; Weintraub, A.; Albert, M.J. Structural Elucidation of the O-Antigen Lipopolysaccharide from Two Strains of Plesiomonas Shigelloides That Share a Type-Specific Antigen with Shigella Flexneri 6, and the Common Group 1 Antigen with Shigella Flexneri Spp and Shigella Dysenteriae 1. Eur. J. Biochem. 1995, 231, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Säwén, E.; Östervall, J.; Landersjö, C.; Edblad, M.; Weintraub, A.; Ansaruzzaman, M.; Widmalm, G. Structural Studies of the O-Antigenic Polysaccharide from Plesiomonas Shigelloides Strain AM36565. Carbohydr. Res. 2012, 348, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Maciejewska, A.; Lukasiewicz, J.; Niedziela, T.; Szewczuk, Z.; Lugowski, C. Structural Analysis of the O-Specific Polysaccharide Isolated from Plesiomonas Shigelloides O51 Lipopolysaccharide. Carbohydr. Res. 2009, 344, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Maciejewska, A.; Lukasiewicz, J.; Kaszowska, M.; Man-Kupisinska, A.; Jachymek, W.; Lugowski, C. Core Oligosaccharide of Plesiomonas Shigelloides PCM 2231 (Serotype O17) Lipopolysaccharide--Structural and Serological Analysis. Mar. Drugs. 2013, 11, 440–454. [Google Scholar] [CrossRef]
- Kubler-Kielb, J.; Schneerson, R.; Mocca, C.; Vinogradov, E. The Elucidation of the Structure of the Core Part of the LPS from Plesiomonas Shigelloides Serotype O17 Expressing O-Polysaccharide Chain Identical to the Shigella Sonnei O-Chain. Carbohydr. Res. 2008, 343, 3123–3127. [Google Scholar] [CrossRef]
- Pieretti, G.; Corsaro, M.M.; Lanzetta, R.; Parrilli, M.; Canals, R.; Merino, S.; Tomás, J.M. Structural Studies of the O-Chain Polysaccharide from Plesiomonas Shigelloides Strain 302-73 (Serotype O1). Eur. J. Org. Chem. 2008, 18, 3149–3155. [Google Scholar] [CrossRef]
- Pieretti, G.; Corsaro, M.M.; Lanzetta, R.; Parrilli, M.; Vilehes, S.; Merino, S.; Tomas, J.M. Structure of the Core Region from the Lipopolysaceharide of Plesiomonas Shigeiloides Strain 302-73 (Serotype Ol). Eur. J. Org. Chem. 2009, 9, 1365–1371. [Google Scholar] [CrossRef]
- Pieretti, G.; Carillo, S.; Lindner, B.; Lanzetta, R.; Parrilli, M.; Jimenez, N.; Regué, M.; Tomás, J.M.; Corsaro, M.M. The Complete Structure of the Core of the LPS from Plesiomonas Shigelloides 302-73 and the Identification of Its O-Antigen Biological Repeating Unit. Carbohydr. Res. 2010, 345, 2523–2528. [Google Scholar] [CrossRef]
- Nestor, G.; Lukasiewicz, J.; Sandström, C. Structural Analysis of the Core Oligosaccharide and the O-Specific Polysaccharide from the Plesiomonas Shigelloides O33:H3 (Strain CNCTC 34/89) Lipopolysaccharide. Eur. J. Org. Chem. 2014, 6, 1241–1252. [Google Scholar] [CrossRef]
- Lundqvist, L.C.E.; Kaszowska, M.; Sandström, C. NMR Study of the O-Specific Polysaccharide and the Core Oligosaccharide from the Lipopolysaccharide Produced by Plesiomonas Shigelloides O24:H8 (Strain CNCTC 92/89). Molecules 2015, 20, 5729–5739. [Google Scholar] [CrossRef] [PubMed]
- Ucieklak, K.; Koj, S.; Pawelczyk, D.; Niedziela, T. Structural Masquerade of Plesiomonas Shigelloides Strain CNCTC 78/89 O-Antigen-High-Resolution Magic Angle Spinning NMR Reveals the Modified d-Galactan I of Klebsiella Pneumoniae. Int. J. Mol. Sci. 2017, 18, 2572. [Google Scholar] [CrossRef] [PubMed]
- Czaja, J.; Jachymek, W.; Niedziela, T.; Lugowski, C.; Aldova, E.; Kenne, L. Structural Studies of the O-Specific Polysaccharide from Plesiomonas Shigelloides Strain CNCTC 113/92. Eur. J. Biochem. 2000, 267, 1672–1679. [Google Scholar] [CrossRef] [PubMed]
- Niedziela, T.; Dag, S.; Lukasiewicz, J.; Dzieciatkowska, M.; Jachymek, W.; Lugowski, C.; Kenne, L. Complete Lipopolysaccharide of Plesiomonas Shigelloides O74:H5 (Strain CNCTC 144/92). 1. Structural Analysis of the Highly Hydrophobic Lipopolysaccharide, Including the O-Antigen, Its Biological Repeating Unit, the Core Oligosaccharide, and the Linkage between Them. Biochemistry 2006, 45, 10422–10433. [Google Scholar] [CrossRef]
- Kaszowska, M.; Jachymek, W.; Lukasiewicz, J.; Niedziela, T.; Kenne, L.; Lugowski, C. The Unique Structure of Complete Lipopolysaccharide Isolated from Semi-Rough Plesiomonas Shigelloides O37 (Strain CNCTC 39/89) Containing (2S)-O-(4-Oxopentanoic Acid)-α-D-Glcp (α-D-Lenose). Carbohydr. Res. 2013, 378, 98–107. [Google Scholar] [CrossRef]
- Kaszowska, M.; Stojkovic, K.; Niedziela, T.; Lugowski, C. The O-Antigen of Plesiomonas Shigelloides Serotype O36 Containing Pseudaminic Acid. Carbohydr. Res. 2016, 434, 1–5. [Google Scholar] [CrossRef]
- Kaszowska, M.; Jachymek, W.; Niedziela, T.; Koj, S.; Kenne, L.; Lugowski, C. The Novel Structure of the Core Oligosaccharide Backbone of the Lipopolysaccharide from the Plesiomonas Shigelloides Strain CNCTC 80/89 (Serotype O13). Carbohydr. Res. 2013, 380, 45–50. [Google Scholar] [CrossRef]
- Niedziela, T.; Lukasiewicz, J.; Jachymek, W.; Dzieciatkowska, M.; Lugowski, C.; Kenne, L. Core Oligosaccharides of Plesiomonas Shigelloides O54:H2 (Strain CNCTC 113/92): Structural and Serological Analysis of the Lipopolysaccharide Core Region, the O-Antigen Biological Repeating Unit, and the Linkage between Them. J. Biol. Chem. 2002, 277, 11653–11663. [Google Scholar] [CrossRef]
- Lukasiewicz, J.; Niedziela, T.; Jachymek, W.; Kenne, L.; Lugowski, C. Structure of the Lipid A-Inner Core Region and Biological Activity of Plesiomonas Shigelloides O54 (Strain CNCTC 113/92) Lipopolysaccharide. Glycobiology 2006, 16, 538–550. [Google Scholar] [CrossRef]
- Lukasiewicz, J.; Dzieciatkowska, M.; Niedziela, T.; Jachymek, W.; Augustyniuk, A.; Kenne, L.; Lugowski, C. Complete Lipopolysaccharide of Plesiomonas Shigelloides O74:H5 (Strain CNCTC 144/92). 2. Lipid A, Its Structural Variability, the Linkage to the Core Oligosaccharide, and the Biological Activity of the Lipopolysaccharide. Biochemistry 2006, 45, 10434–10447. [Google Scholar] [CrossRef]
- Westphal, O.; Jann, K. Bacterial Lipopolysaccharides Extraction with Phenol-Water and Further Applications of the Procedure. Methods Carbohydr. Chem. 1965, 5, 83–91. [Google Scholar]
- Petersson, C.; Niedziela, T.; Jachymek, W.; Kenne, L.; Zarzecki, P.; Lugowski, C. Structural Studies of the O-Specific Polysaccharide of Hafnia Alvei Strain PCM 1206 Lipopolysaccharide Containing D-Allothreonine. Eur. J. Biochem. 1997, 244, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Kubler-Kielb, J.; Vinogradov, E. The Study of the Core Part and Non-Repeating Elements of the O-Antigen of Brucella Lipopolysaccharide. Carbohydr. Res. 2013, 366, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Knirel, Y.A.; Kocharova, N.A.; Shashkov, A.S.; Kochetkov, N.K.; Kholodkova, E.V.; Stanislavsky, E.S. Somatic Antigens of Pseudomonas Aeruginosa. The Structure of O-Specific Polysaccharide Chains of the Lipopolysaccharides from P. Aeruginosa II (Sandvik) and V (IM-1, Verder-Evans). Eur. J. Biochem. 1987, 166, 189–197. [Google Scholar] [CrossRef]
- Shashkov, A.S.; Lipkind, G.M.; Knirel, Y.A.; Kochetkov, N.K. Stereochemical Factors Determining the Effects of Glycosylation on the 13C Chemical Shifts in Carbohydrates. Magn. Reson. Chem. 1988, 26, 735–747. [Google Scholar] [CrossRef]
- Domon, B.; Costello, C.E. A Systematic Nomenclature for Carbohydrate Fragmentations in FAB-MS/MS Spectra of Glycoconjugates. Glycoconj. J. 1988, 5, 397–409. [Google Scholar] [CrossRef]
- Vinogradov, E.; Conlan, W.J.; Gunn, J.S.; Perry, M.B. Characterization of the Lipopolysaccharide O-Antigen of Francisella Novicida (U112). Carbohydr. Res. 2004, 339, 649–654. [Google Scholar] [CrossRef]
- Apicella, M.A.; Post, D.M.B.; Fowler, A.C.; Jones, B.D.; Rasmussen, J.A.; Hunt, J.R.; Imagawa, S.; Choudhury, B.; Inzana, T.J.; Maier, T.M.; et al. Identification, Characterization and Immunogenicity of an O-Antigen Capsular Polysaccharide of Francisella Tularensis. PLoS ONE 2010, 5, e11060. [Google Scholar] [CrossRef]
- Rundlöf, T.; Weintraub, A.; Widmalm, G. Structural Determination of the O-Antigenic Polysaccharide from Escherichia Coli O35 and Cross-Reactivity to Salmonella Arizonae O62. Eur. J. Biochem. 1998, 258, 139–143. [Google Scholar] [CrossRef]
- Parolis, H.; Parolis, L.A.; Olivieri, G. Structural Studies on the Shigella-like Escherichia Coli O121 O-Specific Polysaccharide. Carbohydr. Res. 1997, 303, 319–325. [Google Scholar] [CrossRef]
- Vinogradov, E.V.; Knirel, Y.A.; Shashkov, A.S.; Kochetkov, N.K. Determination of the Degree of Amidation of 2-Deoxy-2-Formamido-D-Galacturonic Acid in O-Specific Polysaccharides of Pseudomonas Aeruginosa O4 and Related Strains. Carbohydr. Res. 1987, 170, 1–4. [Google Scholar] [CrossRef]
- Zdorovenko, E.L.; Varbanets, L.D.; Shashkov, A.S.; Kiprianova, E.A.; Knirel, Y.A. Structure of the O-Polysaccharide of the Lipopolysaccharide of Pseudomonas Chlororaphis Subsp. Aureofaciens UCM B-306. Carbohydr. Res. 2015, 410, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Ciucanu, I.; Kerek, F. A Simple and Rapid Method for the Permethylation of Carbohydrates. Carbohydr. Res. 1984, 131, 209–217. [Google Scholar] [CrossRef]
- Gerwig, G.J.; Kamerling, J.P.; Vliegenthart, J.F.G. Determination of the d and l Configuration of Neutral Monosaccharides by High-Resolution Capillary g.l.c. Carbohydr. Res. 1978, 62, 349–357. [Google Scholar] [CrossRef]
- Gerwig, G.J.; Kamerling, J.P.; Vliegenthart, J.F. Determination of the Absolute Configuration of Mono-Saccharides in Complex Carbohydrates by Capillary G.L.C. Carbohydr. Res. 1979, 77, 1–7. [Google Scholar] [CrossRef]
- Lee, W.; Tonelli, M.; Markley, J.L. NMRFAM-SPARKY: Enhanced Software for Biomolecular NMR Spectroscopy. Bioinformatics 2015, 31, 1325–1327. [Google Scholar] [CrossRef] [PubMed]
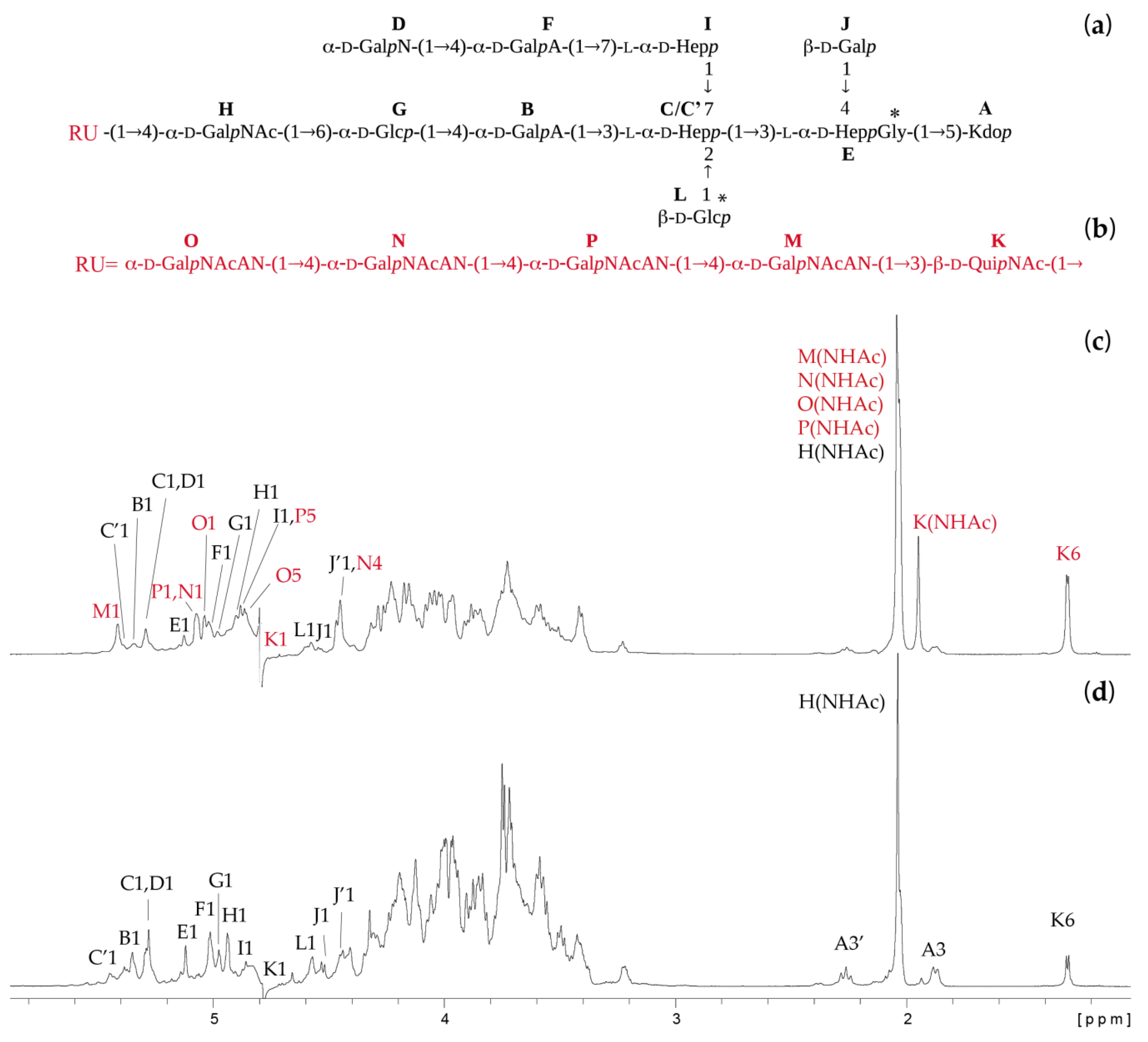
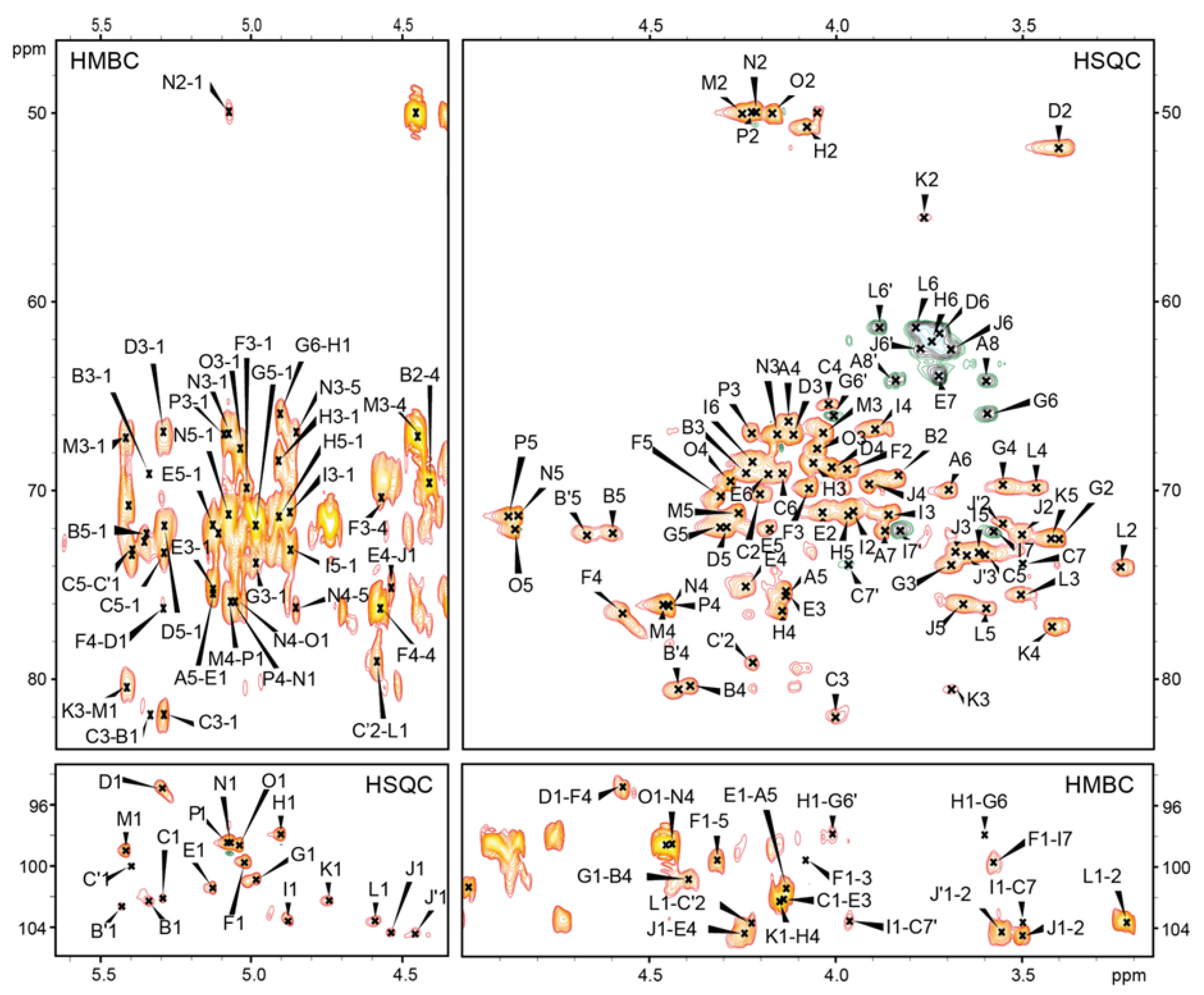


| Residue (a) | Fraction (b) | Chemical Shifts (ppm) (c) | Selected Inter-Residue NOE and 3JH,C Connectivities | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OS | OSRU | H1 C1 | H2, (H3′) C2 | H3 C3 | H4 C4 | H5 C5 | H6, H6′ C6 | H7, H7′ C7 | H8, H8′ C8 | H1/C1 Connectivities to | Inter-Residue Atom/Residue | ||
| A | →5)-α-d-Kdop | * | * | (2.26) | 1.87 34.3 | 4.13 66.5 | 4.13 75.5 | 3.70 70.0 | 3.87 72.1 | 3.60, 3.84 64.2 | |||
| B | →4)-α-d-GalpA-(1→ | * | * | 5.34 102.2 | 3.84 69.2 | 4.25 69.2 | 4.39 80.4 | 4.60 72.3 | 174.6 | 4.01 82.0 | H3 of C | ||
| B′ | →4)-α-d-GalpA-(1→ | * | * | 5.43 102.6 | 3.85 69.1 | 4.25 69.1 | 4.42 80.6 | 4.67 72.4 | 174.3 | ||||
| C | →3,7)-l-α-d-Hepp-(1→ | * | * | 5.29 102.2 | 4.20 70.7 | 4.01 82.0 | 4.02 65.5 | 3.61 73.4 | 4.15 69.1 | 3.50, 3.97 73.9 | 4.14 | H3 of E | |
| C′ | →2,3,7)-l-α-d-Hepp-(1→ (d) | * | * | 5.39 100.0 | 4.23 79.1 | nd | nd | nd | nd | nd | |||
| D | α-d-GalpN-(1→ | * | * | 5.29 94.9 | 3.40 51.9 | 4.11 67.1 | 4.01 68.8 | 4.30 72.0 | 3.72 61.7 | 4.57 76.5 | H4 of F | ||
| E | →3,4)-l-α-d-Hepp-(1→ | * | * | 5.13 101.5 | 4.04 71.2 | 4.14 75.6 | 4.24 75.2 | 4.18 72.0 | 4.19 69.1 | 3.73 63.9 | 4.13 75.5 | H5 of A | |
| F | →4)-α-d-GalpA-(1→ | * | * | 5.02 99.7 | 3.97 68.9 | 4.08 69.9 | 4.57 76.5 | 4.31 70.3 | 176.8 | 3.58 | H7 of I | ||
| G | →6)-α-d-Glcp-(1→ | * | * | 4.98 100.9 | 3.40 72.6 | 3.69 74.0 | 3.55 69.7 | 4.31 72.0 | 3.60, 4.01 66.0 | 4.39 80.4 | H4 of B | ||
| H | →4)-α-d-GalpNAc-(1→ | (*) | * | 4.90 98.0 | 4.08 50.8 | 4.06 68.5 | 4.15 76.4 | 3.97 71.4 | 3.74 62.1 | 3.60 66.0 | H6 of G | ||
| H | α-d-GalpNAc-(1→ | * | 4.95 98.1 | 4.18 50.8 | 3.94 68.6 | 3.99 69.3 | 3.96 71.9 | 3.75 61.9 | 3.60 66.0 | H6 of G | |||
| I | →7)-l-α-d-Hepp-(1→ | * | * | 4.88 103.6 | 3.96 71.1 | 3.86 71.3 | 3.90 66.8 | 3.61 73.3 | 4.23 68.5 | 3.58, 3.83 72.1 | 3.50 | H7 of C | |
| J | β-d-Galp-(1→ | * | * | 4.54 104.5 | 3.50 72.3 | 3.68 73.2 | 3.91 69.7 | 3.66 76.0 | 3.69, 3.77 62.5 | 4.24 75.2 | H4 of E | ||
| K | →3)-β-d-QuipNAc-(1→ | * | 4.74 102.3 | 3.76 55.6 | 3.70 80.6 | 3.42 77.2 | 3.42 72.5 | 1.30 17.3 | 4.15 | H4 of H | |||
| K | β-d-QuipNAc-(1→ | (*) | 4.72 102.5 | 3.72 56.8 | 3.50 74.6 | 3.22 76.1 | 3.45 72.6 | 1.31 17.5 | 4.15 | H4 of H | |||
| L | β-d-Glcp-(1→ | * | * | 4.59 103.7 | 3.23 74.1 | 3.50 75.5 | 3.47 69.9 | 3.59 76.2 | 3.79, 3.89 61.4 | 4.23 79.1 | H2 of C′ | ||
| M | →4)-α-d-GalpNAcAN-(1→ | * | 5.41 99.0 | 4.24 50.0 | 4.03 67.0 | 4.45 76.1 | 4.26 71.2 | 174.4 | 3.70 80.6 | H3 of K | |||
| N | →4)-α-d-GalpNAcAN-(1→ | * | 5.07 98.4 | 4.22 50.0 | 4.17 67.1 | 4.44 76.3 | 4.85 71.4 | 174.5 | 4.46 | H4 of P | |||
| O | α-d-GalpNAcAN-(1→ | * | 5.04 98.6 | 4.16 50.1 | 4.05 67.8 | 4.28 69.6 | 4.86 72.1 | 175.1 | 4.44 | H4 of N | |||
| P | →4)-α-d-GalpNAcAN-(1→ | * | 5.08 98.4 | 4.23 50.0 | 4.22 67.0 | 4.46 76.0 | 4.88 71.4 | 174.0 | 4.45 | H4 of M | |||
| Gly | * | * | 3.98 40.9 | 169.0 | |||||||||
| Oligosaccharide Structure a | Calculated Mass (Da) | Observed Ion (m/z) | Calculated Ion (m/z) | Interpretation of the Ion |
|---|---|---|---|---|
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo·Gly | 3125.03 | 3147.91 | 3148.02 | [M+H, Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo·Gly | 3125.03 | 3129.91 | 3130.01 | [M-H2O+H, Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo·Gly | 3125.03 | 3107.92 | 3108.03 | [M-H2O+H]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 3068.01 | 3090.93 | 3091.00 | [M+H, Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 3068.01 | 3068.95 | 3069.02 | [M+H]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 3068.01 | 3050.94 | 3051.00 | [M-H2O+H]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 2962.98 | 3007.91 | 3007.95 | [M+H, 2Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 2962.98 | 2989.88 | 2989.94 | [M-H2O+H, 2Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 2962.98 | 2967.92 | 2967.96 | [M-H2O+H, Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 2962.98 | 2945.92 | 2945.97 | [M-H2O+H]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 2905.96 | 2928.92 | 2928.95 | [M+H, Na]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 2905.96 | 2906.93 | 2906.97 | [M+H]+ |
| GalNAcAN4·QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 2905.96 | 2888.92 | 2888.95 | [M-H2O+H]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo·Gly | 2260.73 | 2261.52 | 2261.74 | [M+H]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2203.71 | 2226.47 | 2226.70 | [M+H, Na]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2203.71 | 2204.47 | 2204.72 | [M+H]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2203.71 | 2186.44 | 2186.71 | [M-H2O+H]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 2098.68 | 2099.38 | 2099.69 | [M+H]+ |
| QuiNAc·GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 2041.66 | 2064.33 | 2064.65 | [M+H, Na]+ |
| GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2016.63 | 2039.28 | 2039.62 | [M+H, Na]+ |
| GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2016.63 | 2017.28 | 2017.64 | [M+H]+ |
| GalNAc·GalN·GalA2·Hex3·Hep3·Kdo | 2016.63 | 1999.26 | 1999.62 | [M-H2O+H]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 1911.60 | 1934.20 | 1934.59 | [M+H, Na]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 1911.60 | 1912.20 | 1912.61 | [M+H]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo·Gly | 1911.60 | 1894.18 | 1894.59 | [M-H2O+H]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 1854.58 | 1877.16 | 1877.57 | [M+H, Na]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 1854.58 | 1855.16 | 1855.58 | [M+H]+ |
| GalNAc·GalN·GalA2·Hex2·Hep3·Kdo | 1854.58 | 1837.14 | 1837.57 | [M-H2O+H]+ |
| Oligosaccharide Structure | Calculated Mass (Da) | Observed Ion (m/z) | Calculated Ion (m/z) | Interpretation of the Ion |
|---|---|---|---|---|
| GalNAcAN4·QuiNAc | 1069.39 | 1074.39 | 1074.36 | [M-H2O+H, Na]+ |
| GalNAcAN5·QuiNAc | 1285.47 | 1290.47 | 1290.43 | [M-H2O+H, Na]+ |
| GalNAcAN6·QuiNAc | 1501.54 | 1506.56 | 1506.51 | [M-H2O+H, Na]+ |
| GalNAcAN6·QuiNAc | 1501.54 | 1525.57 | 1525.54 | [M+H, Na]+ |
| GalNAcAN7·QuiNAc | 1717.62 | 1722.61 | 1722.58 | [M-H2O+H, Na]+ |
| GalNAcAN7·QuiNAc | 1717.62 | 1740.65 | 1740.61 | [M+H, Na]+ |
| GalNAcAN8·QuiNAc2 | 2120.78 | 2125.79 | 2125.74 | [M-H2O+H, Na]+ |
| GalNAcAN9·QuiNAc2 | 2336.85 | 2341.87 | 2341.82 | [M-H2O +H, Na]+ |
| GalNAcAN10·QuiNAc2 | 2552.93 | 2557.91 | 2557.89 | [M-H2O+H, Na]+ |
| GalNAcAN10·QuiNAc2 | 2552.93 | 2576.93 | 2576.92 | [M+H, Na]+ |
| GalNAcAN11·QuiNAc2 | 2769.00 | 2792.02 | 2791.99 | [M+H, Na]+ |
| GalNAcAN12·QuiNAc3 | 3172.16 | 3195.20 | 3195.14 | [M+H, Na]+ |
| GalNAcAN13·QuiNAc3 | 3388.23 | 3411.31 | 3411.22 | [M+H, Na]+ |
| GalNAcAN14·QuiNAc3 | 3604.31 | 3627.43 | 3627.30 | [M+H, Na]+ |
| GalNAcAN15·QuiNAc3 | 3820.38 | 3843.58 | 3843.37 | [M+H, Na]+ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maciejewska, A.; Bednarczyk, B.; Lugowski, C.; Lukasiewicz, J. Structural Studies of the Lipopolysaccharide Isolated from Plesiomonas shigelloides O22:H3 (CNCTC 90/89). Int. J. Mol. Sci. 2020, 21, 6788. https://doi.org/10.3390/ijms21186788
Maciejewska A, Bednarczyk B, Lugowski C, Lukasiewicz J. Structural Studies of the Lipopolysaccharide Isolated from Plesiomonas shigelloides O22:H3 (CNCTC 90/89). International Journal of Molecular Sciences. 2020; 21(18):6788. https://doi.org/10.3390/ijms21186788
Chicago/Turabian StyleMaciejewska, Anna, Brygida Bednarczyk, Czeslaw Lugowski, and Jolanta Lukasiewicz. 2020. "Structural Studies of the Lipopolysaccharide Isolated from Plesiomonas shigelloides O22:H3 (CNCTC 90/89)" International Journal of Molecular Sciences 21, no. 18: 6788. https://doi.org/10.3390/ijms21186788
APA StyleMaciejewska, A., Bednarczyk, B., Lugowski, C., & Lukasiewicz, J. (2020). Structural Studies of the Lipopolysaccharide Isolated from Plesiomonas shigelloides O22:H3 (CNCTC 90/89). International Journal of Molecular Sciences, 21(18), 6788. https://doi.org/10.3390/ijms21186788





