Identifying the Pressure Points of Acute Cadmium Stress Prior to Acclimation in Arabidopsis thaliana
Abstract
1. Introduction
2. Results
2.1. Cadmium-Induced Growth Responses and Cadmium Accumulation
2.2. Glutathione as Chelator and Anti-Oxidant
2.3. ROS Signalling and Oxidative Challenge under Acute Cd Stress
2.4. Ethylene-Related Signalling and Stress Responses
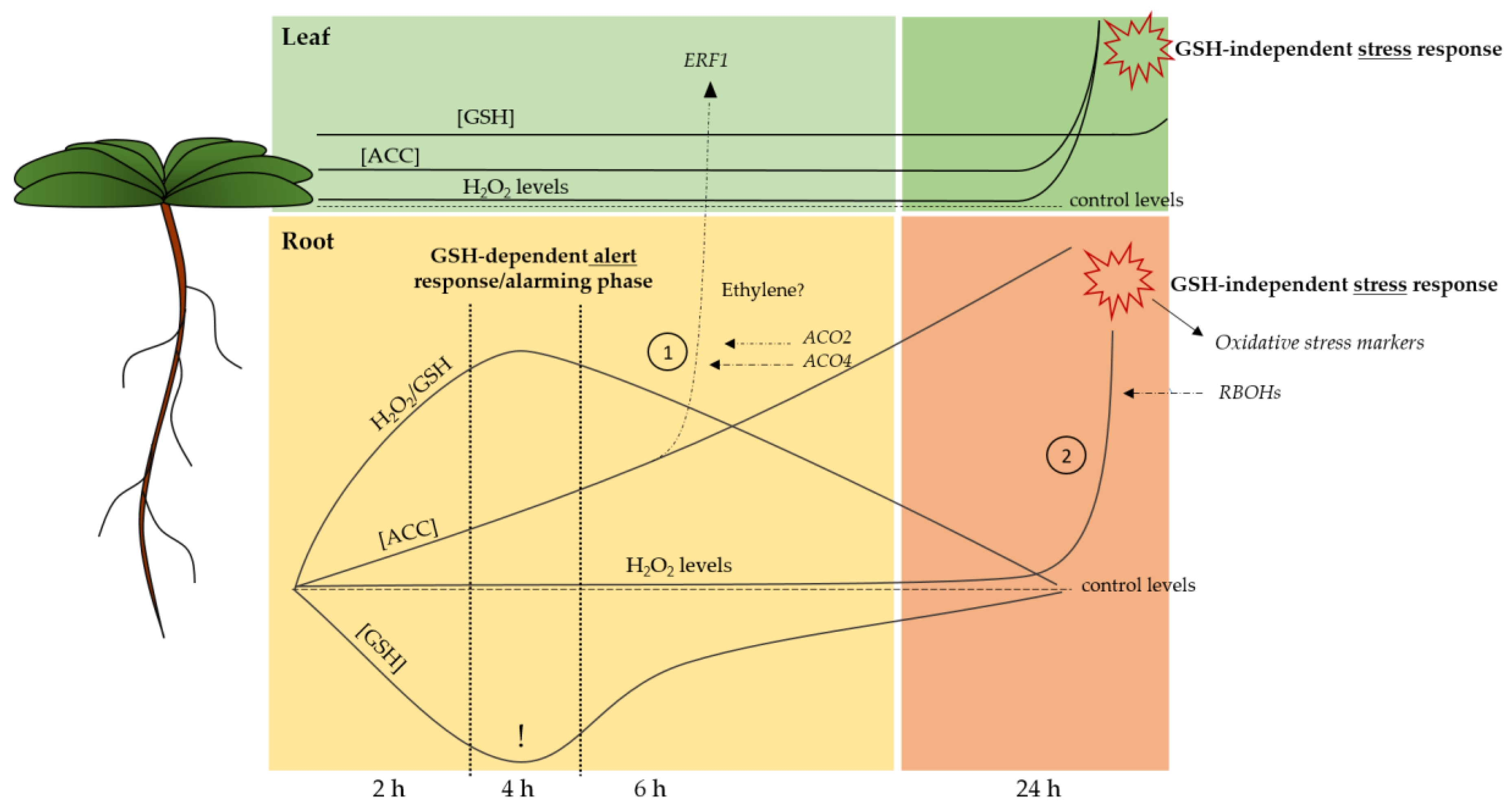
3. Discussion
4. Materials and Methods
4.1. Plant Culture, Cadmium Treatment and Sampling
4.2. Quantification of Root and Rosette Cd Concentrations
4.3. Gene Expression Analysis
4.4. Glutathione Concentration
4.5. Hydrogen Peroxide Measurements
4.6. Determination of Free ACC Content
4.7. Glutathione Reductase Activity Measurements
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACC | 1-aminocyclopropane-1-carboxylic acid |
| ACS | 1-aminocyclopropane-1-carboxylic acid synthase |
| ACO | 1-aminocyclopropane-1-carboxylic acid oxidase |
| AsA | Ascorbate |
| ATP | Adenosine triphosphate |
| Cd | Cadmium |
| eGSH | Extracellular glutathione |
| ERF | Ethylene responsive transcription factor |
| GGT | γ-glutamyl transpeptidase |
| GSH | Glutathione |
| GSH1 | Glutamate cysteine ligase |
| GSH2 | Glutathione synthetase |
| GOI | Gene of interest |
| GR | Glutathione reductase |
| H2O2 | Hydrogen peroxide |
| MPK | Mitogen-activated protein kinase |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| O2•− | Superoxide |
| OXI | Oxidative signal inducible |
| PC | Phytochelatine |
| RBOH | Respiratory burst oxidase homologue |
| ROS | Reactive oxygen species |
| RRTF | Redox-responsive transcription factor |
| SAM | S-adenosylmethionine |
| WRKY | WRKY DNA-binding protein |
| WT | Wild-type |
| ZAT | Zinc finger of Arabidopsis thaliana |
References
- Grierson, C.S.; Barnes, S.R.; Chase, M.W.; Clarke, M.; Grierson, D.; Edwards, K.J.; Jellis, G.J.; Jones, J.D.; Knapp, S.; Oldroyd, G.; et al. One hundred important questions facing plant science research. New Phytol. 2011, 192, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Järup, L.; Berglund, M.; Elinder, C.G.; Nordberg, G.; Vanter, M. Health effects of cadmium exposure–a review of the literature and a risk estimate. Scand. J. Work Health 1998, 24, 1–51. [Google Scholar]
- Verbruggen, N.; Hermans, C.; Schat, H. Mechanisms to cope with arsenic or cadmium excess in plants. Curr. Opin. Plant Biol. 2009, 12, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Di Toppi, L.S.; Gabbrielli, R. Response to cadmium in higher plants. Environ. Exp. Bot. 1999, 41, 105–130. [Google Scholar] [CrossRef]
- Asgher, M.; Khan, M.I.R.; Anjum, N.A.; Khan, N.A. Minimising toxicity of cadmium in plants—Role of plant growth regulators. Protoplasma 2014, 252, 399–413. [Google Scholar] [CrossRef]
- Clemens, S.; Antosiewicz, D.M.; Ward, J.M.; Schachtman, D.P.; Schroeder, J.I. The plant cDNA LCT1 mediates the uptake of calcium and cadmium in yeast. Proc. Natl. Acad. Sci. USA 1998, 95, 12043–12048. [Google Scholar] [CrossRef]
- Cuypers, A.; Plusquin, M.; Remans, T.; Jozefczak, M.; Keunen, E.; Gielen, H.; Opdenakker, K.; Nair, A.R.; Munters, E.; Artois, T.J.; et al. Cadmium stress: An oxidative challenge. Biometals 2010, 23, 927–940. [Google Scholar] [CrossRef]
- Cuypers, A.; Smeets, K.; Ruytinx, J.; Opdenakker, K.; Keunen, E.; Remans, T.; Horemans, N.; Vanhoudt, N.; Van Sanden, S.; Van Belleghem, F.; et al. The cellular redox state as a modulator in cadmium and copper responses in Arabidopsis thaliana seedlings. J. Plant Physiol 2011, 168, 309–316. [Google Scholar] [CrossRef]
- Semane, B.; Cuypers, A.; Smeets, K.; Van Belleghem, F.; Horemans, N.; Schat, H.; Vangronsveld, J. Cadmium responses in Arabidopsis thaliana: Glutathione metabolism and antioxidative defence system. Physiol. Plant. 2007, 129, 519–528. [Google Scholar] [CrossRef]
- Jozefczak, M.; Keunen, E.; Schat, H.; Bliek, M.; Hernandez, L.E.; Carleer, R.; Remans, T.; Bohler, S.; Vangronsveld, J.; Cuypers, A. Differential response of Arabidopsis leaves and roots to cadmium: Glutathione-related chelating capacity vs. antioxidant capacity. Plant Physiol Biochem 2014, 83, 1–9. [Google Scholar] [CrossRef]
- Jozefczak, M.; Bohler, S.; Schat, H.; Horemans, N.; Guisez, Y.; Remans, T.; Vangronsveld, J.; Cuypers, A. Both the concentration and redox state of glutathione and ascorbate influence the sensitivity of arabidopsis to cadmium. Ann. Bot. 2015, 116, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Hell, R.; Bergmann, L. γ-Glutamylcysteine synthetase in higher plants: Catalytic properties and subcellular localization. Planta 1990, 180, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Law, M.Y.; Halliwell, B. Purification and properties of glutathione synthetase from spinach (Spinacia oleracea) leaves. Plant Sci. 1986, 43, 185–191. [Google Scholar] [CrossRef]
- Hell, R.; Bergmann, L. Glutathione synthetase in tobacco suspension cultures: Catalytic properties and localization. Physiol. Plant. 1988, 72, 70–76. [Google Scholar] [CrossRef]
- Grill, E.; Winnacker, E.-L.; Zenk, M.H. Phytochelatins: The Principal Heavy-Metal Complexing Peptides of Higher Plants. Science 1985, 230, 674–676. [Google Scholar] [CrossRef]
- Groden, D.; Beck, E. H2O2 destruction by ascorbate-dependent systems from chloroplasts. Biochim. Biophys. Acta Bioenerg. 1979, 546, 426–435. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Redox homeostasis and antioxidant signaling: A metabolic interface between stress perception and physiological responses. Plant Cell 2005, 17, 1866–1875. [Google Scholar] [CrossRef]
- Gill, S.S.; Anjum, N.A.; Hasanuzzaman, M.; Gill, R.; Trivedi, D.K.; Ahmad, I.; Pereira, E.; Tuteja, N. Glutathione and glutathione reductase: A boon in disguise for plant abiotic stress defense operations. Plant Physiol. Biochem. 2013, 70, 204–212. [Google Scholar] [CrossRef]
- Mhamdi, A.; Hager, J.; Chaouch, S.; Queval, G.; Han, Y.; Taconnat, L.; Saindrenan, P.; Gouia, H.; Issakidis-Bourguet, E.; Renou, J.P.; et al. Arabidopsis Glutathione Reductase1 plays a crucial role in leaf responses to intracellular hydrogen peroxide and in ensuring appropriate gene expression through both salicylic acid and jasmonic acid signaling pathways. Plant Physiol 2010, 153, 1144–1160. [Google Scholar] [CrossRef]
- Masi, A.; Trentin, A.R.; Agrawal, G.K.; Rakwal, R. Gamma-glutamyl cycle in plants: A bridge connecting the environment to the plant cell? Front. Plant Sci. 2015, 6, 252. [Google Scholar] [CrossRef]
- Tolin, S.; Arrigoni, G.; Trentin, A.R.; Veljovic-Jovanovic, S.; Pivato, M.; Zechman, B.; Masi, A. Biochemical and quantitative proteomics investigations in Arabidopsis ggt1 mutant leaves reveal a role for the gamma-glutamyl cycle in plant’s adaptation to environment. Proteomics 2013, 13, 2031–2045. [Google Scholar] [CrossRef] [PubMed]
- Tamás, L.; Alemayehu, A.; Mistrík, I.; Zelinová, V. Extracellular glutathione recycling by γ-glutamyl transferase in barley root tip exposed to cadmium. Environ. Exp. Bot. 2015, 118, 32–39. [Google Scholar] [CrossRef]
- Destro, T.; Prasad, D.; Martignago, D.; Bernet, I.L.; Trentin, A.R.; Renu, I.K.; Ferretti, M.; Masi, A. Compensatory expression and substrate inducibility of gamma-glutamyl transferase GGT2 isoform in Arabidopsis thaliana. J. Exp. Bot. 2011, 62, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Ohkama-Ohtsu, N.; Radwan, S.; Peterson, A.; Zhao, P.; Badr, A.F.; Xiang, C.; Oliver, D.J. Characterization of the extracellular γ-glutamyl transpeptidases, GGT1 and GGT2, in Arabidopsis. Plant J. 2007, 49, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.N.; Saladores, P.H.; Lambert, E.; Hudson, A.O.; Leustek, T. Localization of members of the gamma-glutamyl transpeptidase family identifies sites of glutathione and glutathione S-conjugate hydrolysis. Plant Physiol 2007, 144, 1715–1732. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, M.; Destro, T.; Tosatto, S.C.E.; La Rocca, N.; Rascio, N.; Masi, A. Gamma-glutamyl transferase in the cell wall participates in extracellular glutathione salvage from the root apoplast. New Phytol. 2009, 181, 115–126. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Suzuki, N.; Miller, G.; Tognetti, V.B.; Vandepoele, K.; Gollery, M.; Shulaev, V.; Van Breusegem, F. ROS signaling: The new wave? Trends Plant Sci 2011, 16, 300–309. [Google Scholar] [CrossRef]
- Cuypers, A.; Hendrix, S.; dos Reis, R.A.; De Smet, S.; Deckers, J.; Gielen, H.; Jozefczak, M.; Loix, C.; Vercampt, H.; Vangronsveld, J.; et al. Hydrogen peroxide, signaling in disguise during metal phytotoxicity. Front. Plant Sci. 2016, 7, 470. [Google Scholar] [CrossRef]
- Han, Y.; Chaouch, S.; Mhamdi, A.; Queval, G.; Zechmann, B.; Noctor, G. Functional analysis of Arabidopsis mutants points to novel roles for glutathione in coupling H2O2 to activation of salicylic acid accumulation and signaling. Antioxid. Redox Signal. 2013, 18, 2106–2121. [Google Scholar] [CrossRef]
- Schnaubelt, D.; Queval, G.; Dong, Y.; Diaz-Vivancos, P.; Makgopa, M.E.; Howell, G.; De Simone, A.; Bai, J.; Hannah, M.A.; Foyer, C.H. Low glutathione regulates gene expression and the redox potentials of the nucleus and cytosol in Arabidopsis thaliana. Plant Cell Environ. 2015, 38, 266–279. [Google Scholar] [CrossRef]
- Schellingen, K.; Van Der Straeten, D.; Remans, T.; Vangronsveld, J.; Keunen, E.; Cuypers, A. Ethylene signalling is mediating the early cadmium-induced oxidative challenge in Arabidopsis thaliana. Plant Sci. 2015, 239, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Mersmann, S.; Bourdais, G.; Rietz, S.; Robatzek, S. Ethylene Signaling Regulates Accumulation of the FLS2 Receptor and Is Required for the Oxidative Burst Contributing to Plant Immunity. Plant Physiol. 2010, 154, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Montero-Palmero, M.B.; Martín-Barranco, A.; Escobar, C.; Hernández, L.E. Early transcriptional responses to mercury: A role for ethylene in mercury-induced stress. New Phytol. 2014, 201, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.J.; Huang, C.S.; Huang, G.J.; Chow, T.J.; Lin, Y.H. NADPH oxidase inhibitor diphenyleneiodonium and reduced glutathione mitigate ethephon-mediated leaf senescence, H2O2 elevation and senescence-associated gene expression in sweet potato (Ipomoea batatas). J. Plant Physiol 2013, 170, 1471–1483. [Google Scholar] [CrossRef]
- Schellingen, K.; Van Der Straeten, D.; Vandenbussche, F.; Prinsen, E.; Remans, T.; Vangronsveld, J.; Cuypers, A. Cadmium-induced ethylene production and responses in Arabidopsis thaliana rely on ACS2 and ACS6 gene expression. BMC Plant Biol. 2014, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Argueso, C.T.; Hansen, M.; Kieber, J.J. Regulation of ethylene biosynthesis. J. Plant Growth Regul. 2007, 26, 92–105. [Google Scholar] [CrossRef]
- Lin, Z.; Zhong, S.; Grierson, D. Recent advances in ethylene research. J. Exp. Bot. 2009, 60, 3311–3336. [Google Scholar] [CrossRef]
- Rentel, M.C.; Lecourieux, D.; Ouaked, F.; Usher, S.L.; Petersen, L.; Okamoto, H.; Knight, H.; Peck, S.C.; Grierson, C.S.; Hirt, H.; et al. OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 2004, 427, 858–861. [Google Scholar] [CrossRef]
- Müller, M.; Munné-Bosch, S. Ethylene Response Factors: A Key Regulatory Hub in Hormone and Stress Signaling. Plant Physiol. 2015, 169, 32–41. [Google Scholar] [CrossRef]
- Smeets, K.; Ruytinx, J.; Van Belleghem, F.; Semane, B.; Lin, D.; Vangronsveld, J.; Cuypers, A. Critical evaluation and statistical validation of a hydroponic culture system for Arabidopsis thaliana. Plant Physiol. Biochem. 2008, 46, 212–218. [Google Scholar] [CrossRef]
- Keunen, E.; Truyens, S.; Bruckers, L.; Remans, T.; Vangronsveld, J.; Cuypers, A. Survival of Cd-exposed Arabidopsis thaliana: Are these plants reproductively challenged? Plant Physiol. Biochem. 2011, 49, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.K.E.; Cobbett, C.S. HMA P-type ATPases are the major mechanism for root-to-shoot Cd translocation in Arabidopsis thaliana. New Phytol. 2009, 181, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Gadjev, I.; Vanderauwera, S.; Gechev, T.S.; Laloi, C.; Minkov, I.N.; Shulaev, V.; Apel, K.; Inze, D.; Mittler, R.; Van Breusegem, F. Transcriptomic footprints disclose specificity of reactive oxygen species signaling in Arabidopsis. Plant Physiol. 2006, 141, 436–445. [Google Scholar] [CrossRef]
- Remans, T.; Opdenakker, K.; Smeets, K.; Mathijsen, D.; Vangronsveld, J.; Cuypers, A. Metal-specific and NADPH oxidase dependent changes in lipoxygenase and NADPH oxidase gene expression in Arabidopsis thaliana exposed to cadmium or excess copper. Funct. Plant Biol. 2010, 37, 532–544. [Google Scholar] [CrossRef]
- Foyer, C.H.; Lopez-Delgado, H.; Dat, J.F.; Scott, I.M. Hydrogen peroxide- and glutathione-associated mechanisms of acclimatory stress tolerance and signalling. Physiol. Plant. 1997, 100, 241–254. [Google Scholar] [CrossRef]
- Keunen, E.; Schellingen, K.; Vangronsveld, J.; Cuypers, A. Ethylene and metal stress: Small molecule, big impact. Front. Plant Sci. 2016, 7, 23. [Google Scholar] [CrossRef]
- Van de Poel, B.; Van Der Straeten, D. 1-aminocyclopropane-1-carboxylic acid (ACC) in plants: More than just the precursor of ethylene! Front. Plant Sci. 2014, 5, 640. [Google Scholar] [CrossRef]
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant Physiological Ecology, 1st ed.; Springer: New York, NY, USA, 1998; pp. 1–540. [Google Scholar]
- Tausz, M.; Šircelj, H.; Grill, D. The glutathione system as a stress marker in plant ecophysiology: Is a stress-response concept valid? J. Exp. Bot. 2004, 55, 1955–1962. [Google Scholar] [CrossRef]
- Zagorchev, L.; Seal, C.E.; Kranner, I.; Odjakova, M. A central role for thiols in plant tolerance to abiotic stress. Int. J. Mol. Sci. 2013, 14, 7405–7432. [Google Scholar] [CrossRef]
- Li, Y.; Dhankher, O.P.; Carreira, L.; Balish, R.S.; Meagher, R.B. Arsenic and mercury tolerance and cadmium sensitivity in Arabidopsis plants expressing bacterial γ-glutamylcysteine synthetase. Environ. Toxicol. Chem. 2005, 24, 1376–1386. [Google Scholar] [CrossRef]
- Xiang, C.; Werner, B.L.; Christensen, E.M.; Oliver, D.J. The biological functions of glutathione revisited in Arabidopsis transgenic plants with altered glutathione levels. Plant Physiol. 2001, 126, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Zlobin, I.E.; Kartashov, A.V.; Shpakovski, G.V. Different roles of glutathione in copper and zinc chelation in Brassica napus roots. Plant Physiol. Biochem. 2017, 118, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Bankaji, I.; Caçador, I.; Sleimi, N. Physiological and biochemical responses of Suaeda fruticosa to cadmium and copper stresses: Growth, nutrient uptake, antioxidant enzymes, phytochelatin, and glutathione levels. Environ. Sci. Pollut. Res. 2015, 22, 13058–13069. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.; Oliver, D.J. Glutathione Metabolic Genes Coordinately Respond to Heavy Metals and Jasmonic Acid in Arabidopsis. Plant Cell 1998, 10, 1539–1550. [Google Scholar] [CrossRef]
- Chen, J.; Yang, L.; Yan, X.; Liu, Y.; Wang, R.; Fan, T.; Ren, Y.; Tang, X.; Xiao, F.; Liu, Y.; et al. Zinc-Finger Transcription Factor ZAT6 Positively Regulates Cadmium Tolerance through the Glutathione-Dependent Pathway in Arabidopsis. Plant Physiol. 2016, 171, 707–719. [Google Scholar] [CrossRef]
- Jozefczak, M.; Remans, T.; Vangronsveld, J.; Cuypers, A. Glutathione is a key player in metal-induced oxidative stress defenses. Int. J. Mol. Sci. 2012, 13, 3145–3175. [Google Scholar] [CrossRef]
- Masi, A.; Destro, T.; Turetta, L.; Varotto, S.; Caporale, G.; Ferretti, M. Localization of gamma-glutamyl transferase activity and protein in Zea mays organs and tissues. J. Plant Physiol. 2007, 164, 1527–1535. [Google Scholar] [CrossRef]
- Xi, H.; Xu, H.; Xu, W.; He, Z.; Xu, W.; Ma, M. A SAL1 Loss-of-Function Arabidopsis Mutant Exhibits Enhanced Cadmium Tolerance in Association with Alleviation of Endoplasmic Reticulum Stress. Plant Cell Physiol. 2016. [Google Scholar] [CrossRef]
- Uzilday, B.; Ozgur, R.; Sekmen, A.H.; Turkan, I. Endoplasmic reticulum stress regulates glutathione metabolism and activities of glutathione related enzymes in Arabidopsis. Funct. Plant Biol. 2018, 45, 284–296. [Google Scholar] [CrossRef]
- Matsuo, M.; Johnson, J.M.; Hieno, A.; Tokizawa, M.; Nomoto, M.; Tada, Y.; Godfrey, R.; Obokata, J.; Sherameti, I.; Yamamoto, Y.Y.; et al. High REDOX RESPONSIVE TRANSCRIPTION FACTOR1 Levels Result in Accumulation of Reactive Oxygen Species in Arabidopsis thaliana Shoots and Roots. Mol. Plant 2015, 8, 1253–1273. [Google Scholar] [CrossRef]
- Joo, S.; Liu, Y.; Lueth, A.; Zhang, S. MAPK phosphorylation-induced stabilization of ACS6 protein is mediated by the non-catalytic C-terminal domain, which also contains the cis-determinant for rapid degradation by the 26S proteasome pathway. Plant J. 2008, 54, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Meng, X.; Wang, R.; Mao, G.; Han, L.; Liu, Y.; Zhang, S. Dual-Level Regulation of ACC Synthase Activity by MPK3/MPK6 Cascade and Its Downstream WRKY Transcription Factor during Ethylene Induction in Arabidopsis. PLoS Genet. 2012, 8, e1002767. [Google Scholar] [CrossRef] [PubMed]
- Skottke, K.R.; Yoon, G.M.; Kieber, J.J.; DeLong, A. Protein Phosphatase 2A Controls Ethylene Biosynthesis by Differentially Regulating the Turnover of ACC Synthase Isoforms. PLoS Genet. 2011, 7, e1001370. [Google Scholar] [CrossRef]
- Remans, T.; Smeets, K.; Opdenakker, K.; Mathijsen, D.; Vangronsveld, J.; Cuypers, A. Normalisation of real-time RT-PCR gene expression measurements in Arabidopsis thaliana exposed to increased metal concentrations. Planta 2008, 227, 1343–1349. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Queval, G.; Noctor, G. A plate reader method for the measurement of NAD, NADP, glutathione, and ascorbate in tissue extracts: Application to redox profiling during Arabidopsis rosette development. Anal. Biochem. 2007, 363, 58–69. [Google Scholar] [CrossRef]
- Smets, R.; Claes, V.; Van Onckelen, H.A.; Prinsen, E. Extraction and quantitative analysis of 1-aminocyclopropane-1-carboxylic acid in plant tissue by gas chromatography coupled to mass spectrometry. J. Chromatogr. A 2003, 993, 79–87. [Google Scholar] [CrossRef]
- Cuypers, A.; Vangronsveld, J.; Clijsters, H. Biphasic effect of copper on the ascorbate-glutathione pathway in primary leaves of Phaseolus vulgaris seedlings during the early stages of metal assimilation. Physiol. Plant. 2000, 110, 512–517. [Google Scholar] [CrossRef]


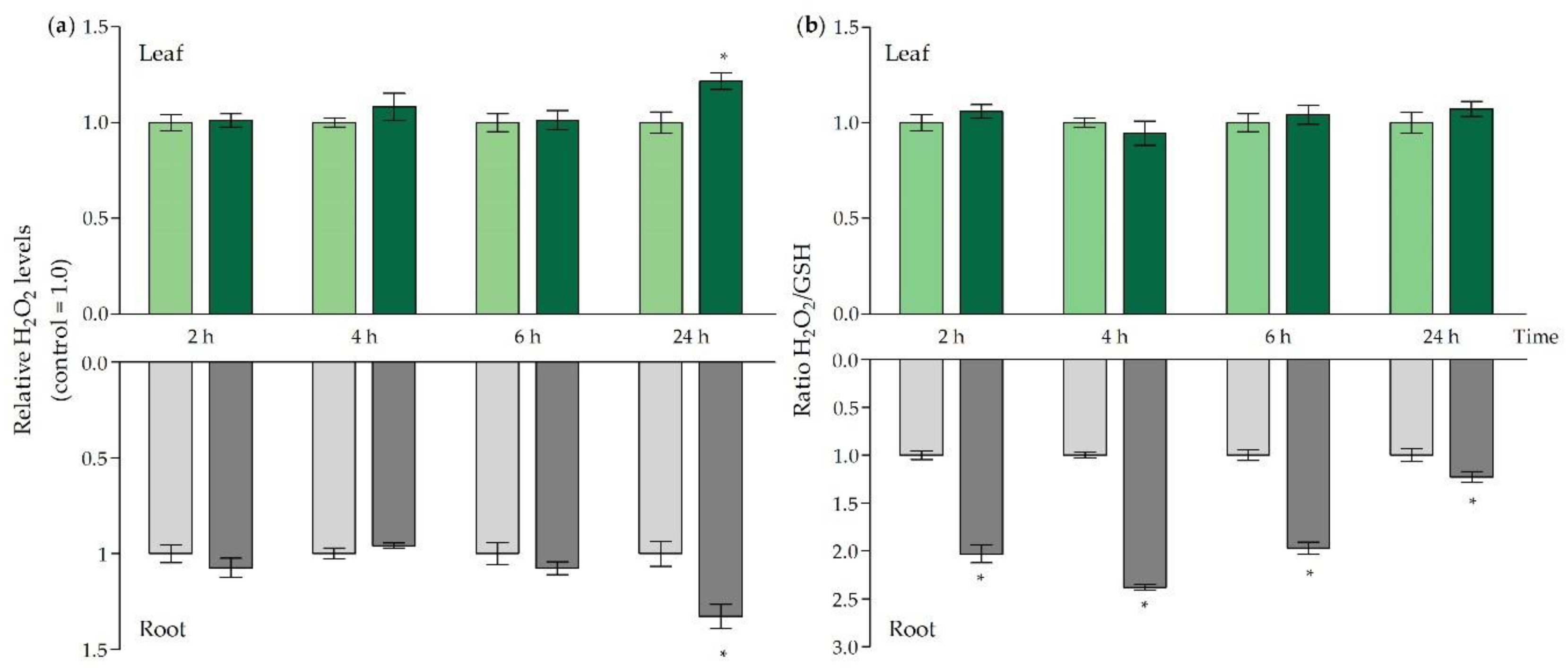

| Cd Concentration (mg kg−1 DW) | |||||
| Organ | [CdSO4] | 2 h | 4 h | 6 h | 24 h |
| Leaf | 0 µM | 0.68 ± 0.12 | 0.62 ± 0.16 | 0.65 ± 0.09 | 0.90 ± 0.17 |
| 5 µM | 6.86 ± 0.17 a | 61.01 ± 2.73 b | 152.97 ± 4.84 c | 840.24 ± 11.71 d | |
| Root | 0 µM | LOD | LOD | LOD | LOD |
| 5 µM | 536.44 ± 14.24 a | 620.39 ± 13.40 b | 730.78 ± 14.25 c | 1788.57 ± 65.40 d* | |
| Translocation Factor | |||||
| [CdSO4] | 2 h | 4 h | 6 h | 24 h | |
| 5 µM | 1.28 ± 0.04 a | 9.86 ± 0.53 b | 21.00 ± 0.97 c | 47.24 ± 1.88 d | |
 ). GSH1: γ-glutamylcysteine synthetase; GSH2: GSH synthetase; GR1: glutathione reductase; GGT1: γ-glutamyl transpeptidase 1; ZAT6: zinc finger of Arabidopsis thaliana 6.
). GSH1: γ-glutamylcysteine synthetase; GSH2: GSH synthetase; GR1: glutathione reductase; GGT1: γ-glutamyl transpeptidase 1; ZAT6: zinc finger of Arabidopsis thaliana 6.
 ). GSH1: γ-glutamylcysteine synthetase; GSH2: GSH synthetase; GR1: glutathione reductase; GGT1: γ-glutamyl transpeptidase 1; ZAT6: zinc finger of Arabidopsis thaliana 6.
). GSH1: γ-glutamylcysteine synthetase; GSH2: GSH synthetase; GR1: glutathione reductase; GGT1: γ-glutamyl transpeptidase 1; ZAT6: zinc finger of Arabidopsis thaliana 6.| GSH-Related Genes | |||||
|---|---|---|---|---|---|
| Gene | [CdSO4] | 2 h | 4 h | 6 h | 24 h |
| Leaf | |||||
| GSH1 | 0 µM | 1.00 ± 0.16 | 1.00 ± 0.06 | 1.00 ± 0.13 | 1.00 ± 0.04 |
| 5 µM | 0.75 ± 0.13 | 0.91 ± 0.05 | 0.79 ± 0.12 | 1.77 ± 0.19 | |
| GSH2 | 0 µM | 1.00 ± 0.06 | 1.00 ± 0.04 | 1.00 ± 0.06 | 1.00 ± 0.04 |
| 5 µM | 0.77 ± 0.11 | 0.91 ± 0.10 | 0.90 ± 0.06 | 2.74 ± 0.14 | |
| GR1 | 0 µM | 1.00 ± 0.04 | 1.00 ± 0.07 | 1.00 ± 0.20 | 1.00 ± 0.10 |
| 5 µM | 0.81 ± 0.08 | 1.05 ± 0.08 | 0.87 ± 0.13 | 3.15 ± 0.52 | |
| GGT1 | 0 µM | 1.00 ± 0.26 | 1.00 ± 0.06 | 1.00 ± 0.27 | 1.00 ± 0.10 |
| 5 µM | 0.82 ± 0.12 | 1.34 ± 0.19 | 1.47 ± 0.43 | 4.60 ± 0.85 | |
| ZAT6 | 0 µM | 1.00 ± 0.20 | 1.00 ± 0.22 | 1.00 ± 0.08 | 1.00 ± 0.30 |
| 5 µM | 0.93 ± 0.14 | 1.14 ± 0.28 | 1.55 ± 0.09 | 15.06 ± 0.55 | |
| Root | |||||
| GSH1 | 0 µM | 1.00 ± 0.22 | 1.00 ± 0.16 | 1.00 ± 0.10 | 1.00 ± 0.17 |
| 5 µM | 2.08 ± 0.45 | 1.35 ± 0.05 | 1.63 ± 0.06 | 4.67 ± 0.28 | |
| GSH2 | 0 µM | 1.00 ± 0.15 | 1.00 ± 0.11 | 1.00 ± 0.01 | 1.00 ± 0.09 |
| 5 µM | 1.67 ± 0.01 | 1.31 ± 0.11 | 1.44 ± 0.08 | 2.85 ± 0.20 | |
| GR1 | 0 µM | 1.00 ± 0.13 | 1.00 ± 0.02 | 1.00 ± 0.04 | 1.00 ± 0.04 |
| 5 µM | 1.01 ± 0.09 | 0.88 ± 0.07 | 1.36 ± 0.03 | 2.06 ± 0.19 | |
| GGT1 | 0 µM | 1.00 ± 0.16 | 1.00 ± 0.14 | 1.00 ± 0.07 | 1.00 ± 0.13 |
| 5 µM | 1.72 ± 0.11 | 1.67 ± 0.12 | 2.00 ± 0.26 | 5.07 ± 0.02 | |
| ZAT6 | 0 µM | 1.00 ± 0.16 | 1.00 ± 0.13 | 1.00 ± 0.16 | 1.00 ± 0.15 |
| 5 µM | 1.10 ± 0.19 | 1.29 ± 0.21 | 1.94 ± 0.02 | 2.89 ± 0.55 | |
 , increased:
, increased:  ).
).
 , increased:
, increased:  ).
).| GR activity (mU g−1 FW) | ||||
|---|---|---|---|---|
| [CdSO4] | 2 h | 4 h | 6 h | 24 h |
| Leaf | ||||
| 0 µM | 64.46 ± 1.75 | 68.08 ± 0.08 | 65.33 ± 0.93 | 57.02 ± 1.55 |
| 5 µM | 64.52 ± 0.86 | 65.70 ± 1.94 | 70.48 ± 1.53 | 69.70 ± 4.62 |
| Root | ||||
| 0 µM | 33.82 ± 1.01 | 32.23 ± 1.67 | 31.50 ± 1.26 | 30.56 ± 0.11 |
| 5 µM | 30.16 ± 0.86 | 29.60 ± 0.74 | 30.91 ± 0.57 | 39.05 ± 0.93 |
 , downregulated:
, downregulated:  ). RBOH: respiratory burst oxidase homologue; ZAT12: zinc finger of Arabidopsis thaliana 12; RRTF1: redox-responsive transcription factor 1.
). RBOH: respiratory burst oxidase homologue; ZAT12: zinc finger of Arabidopsis thaliana 12; RRTF1: redox-responsive transcription factor 1.
 , downregulated:
, downregulated:  ). RBOH: respiratory burst oxidase homologue; ZAT12: zinc finger of Arabidopsis thaliana 12; RRTF1: redox-responsive transcription factor 1.
). RBOH: respiratory burst oxidase homologue; ZAT12: zinc finger of Arabidopsis thaliana 12; RRTF1: redox-responsive transcription factor 1.| NADPH Oxidases | |||||
|---|---|---|---|---|---|
| Gene | [CdSO4] | 2 h | 4 h | 6 h | 24 h |
| Leaf | |||||
| RBOHC | 0 µM | 1.00 ± 0.05 | 1.00 ± 0.06 | 1.00 ± 0.14 | 1.00 ± 0.25 |
| 5 µM | 1.01 ± 0.26 | 10.33 ± 4.58 | 3.35 ± 1.93 | 56.48 ± 8.23 | |
| RBOHD | 0 µM | 1.00 ± 0.25 | 1.00 ± 0.08 | 1.00 ± 0.12 | 1.00 ± 0.07 |
| 5 µM | 0.62 ± 0.02 | 1.29 ± 0.22 | 1.60 ± 0.28 | 1.61 ± 0.13 | |
| RBOHF | 0 µM | 1.00 ± 0.11 | 1.00 ± 0.10 | 1.00 ± 0.18 | 1.00 ± 0.08 |
| 5 µM | 1.40 ± 0.02 | 1.04 ± 0.13 | 1.18 ± 0.30 | 2.53 ± 0.55 | |
| Root | |||||
| RBOHC | 0 µM | 1.00 ± 0.10 | 1.00 ± 0.11 | 1.00 ± 0.14 | 1.00 ± 0.23 |
| 5 µM | 1.41 ± 0.11 | 1.65 ± 0.07 | 1.48 ± 0.09 | 0.72 ± 0.08 | |
| RBOHD | 0 µM | 1.00 ± 0.06 | 1.00 ± 0.14 | 1.00 ± 0.10 | 1.00 ± 0.14 |
| 5 µM | 1.63 ± 0.09 | 1.74 ± 0.13 | 1.60 ± 0.05 | 2.48 ± 0.35 | |
| RBOHF | 0 µM | 1.00 ± 0.12 | 1.00 ± 0.10 | 1.00 ± 0.05 | 1.00 ± 0.17 |
| 5 µM | 1.74 ± 0.09 | 1.54 ± 0.09 | 1.34 ± 0.07 | 2.53 ± 0.14 | |
| Oxidative Stress Markers and Related Genes | |||||
| Leaf | |||||
| AT1G05340 | 0 µM | 1.00 ± 0.05 | 1.00 ± 0.04 | 1.00 ± 0.15 | 1.00 ± 0.18 |
| 5 µM | 1.67 ± 0.73 | 1.95 ± 0.17 | 2.18 ± 0.68 | 53.23 ± 12.52 | |
| AT1G19020 | 0 µM | 1.00 ± 0.30 | 1.00 ± 0.08 | 1.00 ± 0.13 | 1.00 ± 0.18 |
| 5 µM | 1.19 ± 0.57 | 5.22 ± 1.97 | 3.20 ± 0.32 | 36.20 ± 7.13 | |
| AT1G57630 | 0 µM | 1.00 ± 0.07 | 1.00 ± 0.07 | 1.00 ± 0.08 | 1.00 ± 0.13 |
| 5 µM | 1.78 ± 0.97 | 10.67 ± 5.73 | 2.45 ± 1.31 | 30.33 ± 3.42 | |
| AT2G21640 | 0 µM | 1.00 ± 0.05 | 1.00 ± 0.07 | 1.00 ± 0.09 | 1.00 ± 0.21 |
| 5 µM | 0.83 ± 0.06 | 1.09 ± 0.17 | 0.61 ± 0.11 | 8.42 ± 1.33 | |
| AT2G43510 | 0 µM | 1.00 ± 0.02 | 1.00 ± 0.08 | 1.00 ± 0.20 | 1.00 ± 0.12 |
| 5 µM | 0.64 ± 0.14 | 1.25 ± 0.33 | 0.88 ± 0.14 | 26.36 ± 4.00 | |
| ZAT12 | 0 µM | 1.00 ± 0.32 | 1.00 ± 0.17 | 1.00 ± 0.15 | 1.00 ± 0.40 |
| 5 µM | 0.82 ± 0.33 | 3.26 ± 1.40 | 0.66 ± 0.15 | 18.33 ± 1.17 | |
| RRTF1 | 0 µM | 1.00 ± 0.41 | 1.00 ± 0.48 | 1.00 ± 0.30 | 1.00 ± 0.53 |
| 5 µM | 0.25 ± 0.07 | 0.55 ± 0.14 | 3.54 ± 2.25 | 4.59 ± 1.11 | |
| Root | |||||
| AT1G05340 | 0 µM | 1.00 ± 0.35 | 1.00 ± 0.12 | 1.00 ± 0.35 | 1.00 ± 0.12 |
| 5 µM | 1.11 ± 0.18 | 1.33 ± 0.25 | 0.47 ± 0.07 | 1.50 ± 0.31 | |
| AT1G19020 | 0 µM | 1.00 ± 0.27 | 1.00 ± 0.14 | 1.00 ± 0.27 | 1.00 ± 0.31 |
| 5 µM | 2.20 ± 0.52 | 1.88 ± 0.35 | 0.52 ± 0.09 | 3.56 ± 0.75 | |
| AT1G57630 | 0 µM | 1.00 ± 0.10 | 1.00 ± 0.18 | 1.00 ± 0.01 | 1.00 ± 0.32 |
| 5 µM | 1.20 ± 0.03 | 1.43 ± 0.26 | 0.46 ± 0.07 | 2.57 ± 0.76 | |
| AT2G21640 | 0 µM | 1.00 ± 0.07 | 1.00 ± 0.19 | 1.00 ± 0.00 | 1.00 ± 0.11 |
| 5 µM | 1.23 ± 0.05 | 0.95 ± 0.07 | 0.76 ± 0.10 | 1.32 ± 0.29 | |
| AT2G43510 | 0 µM | 1.00 ± 0.69 | 1.00 ± 0.46 | 1.00 ± 0.32 | 1.00 ± 0.26 |
| 5 µM | 0.60 ± 0.20 | 0.31 ± 0.04 | 0.42 ± 0.07 | 2.96 ± 1.38 | |
| ZAT12 | 0 µM | 1.00 ± 0.04 | 1.00 ± 0.21 | 1.00 ± 0.22 | 1.00 ± 0.21 |
| 5 µM | 1.35 ± 0.43 | 1.24 ± 0.31 | 1.09 ± 0.14 | 10.17 ± 2.24 | |
| RRTF1 | 0 µM | 1.00 ± 0.11 | 1.00 ± 0.46 | 1.00 ± 0.69 | 1.00 ± 0.43 |
| 5 µM | 1.27 ± 0.19 | 0.64 ± 0.13 | 1.20 ± 0.50 | 8.58 ± 0.66 | |
 , downregulated:
, downregulated:  ). ACS: ACC synthase; ACO: ACC oxidase; ERF1: ethylene responsive factor 1; OXI1: oxidative signal inducible 1; MPK: mitogen-activated protein kinase; WRKY33: WRKY DNA-binding protein 33.
). ACS: ACC synthase; ACO: ACC oxidase; ERF1: ethylene responsive factor 1; OXI1: oxidative signal inducible 1; MPK: mitogen-activated protein kinase; WRKY33: WRKY DNA-binding protein 33.
 , downregulated:
, downregulated:  ). ACS: ACC synthase; ACO: ACC oxidase; ERF1: ethylene responsive factor 1; OXI1: oxidative signal inducible 1; MPK: mitogen-activated protein kinase; WRKY33: WRKY DNA-binding protein 33.
). ACS: ACC synthase; ACO: ACC oxidase; ERF1: ethylene responsive factor 1; OXI1: oxidative signal inducible 1; MPK: mitogen-activated protein kinase; WRKY33: WRKY DNA-binding protein 33.| Ethylene-Related Genes | |||||
|---|---|---|---|---|---|
| Gene | CdSO4 | 2 h | 4 h | 6 h | 24 h |
| Leaf | |||||
| ACS2 | 0 µM | 1.00 ± 0.08 | 1.00 ± 0.33 | 1.00 ± 0.27 | 1.00 ± 0.18 |
| 5 µM | 1.17± 0.56 | 2.01 ± 0.82 | 1.57 ± 0.21 | 366.12 ± 25.94 | |
| ACS6 | 0 µM | 1.00 ± 0.06 | 1.00 ± 0.09 | 1.00 ± 0.02 | 1.00 ± 0.12 |
| 5 µM | 0.91 ± 0.08 | 2.03 ± 0.61 | 3.73 ± 1.07 | 9.36 ± 1.76 | |
| ACO2 | 0 µM | 1.00 ± 0.08 | 1.00 ± 0.08 | 1.00 ± 0.23 | 1.00 ± 0.10 |
| 5 µM | 0.74 ± 0.09 | 1.08 ± 0.08 | 1.00 ± 0.25 | 5.48 ± 0.19 | |
| ACO4 | 0 µM | 1.00 ± 0.06 | 1.00 ± 0.02 | 1.00 ± 0.23 | 1.00 ± 0.08 |
| 5 µM | 0.95 ± 0.22 | 1.41 ± 0.05 | 1.76 ± 0.44 | 9.36 ± 2.24 | |
| ERF1 | 0 µM | 1.00 ± 0.27 | 1.00 ± 0.24 | 1.00 ± 0.10 | 1.00 ± 0.10 |
| 5 µM | 0.83 ± 0.35 | 2.59 ± 1.33 | 9.00 ± 1.09 | 142.90 ± 21.59 | |
| OXI1 | 0 µM | 1.00 ± 0.35 | 1.00 ± 0.41 | 1.00 ± 0.27 | 1.00 ± 0.51 |
| 5 µM | 1.07 ± 0.34 | 2.07 ± 0.33 | 10.66 ± 1.41 | 58.15 ± 18.54 | |
| MPK3 | 0 µM | 1.00 ± 0.02 | 1.00 ± 0.19 | 1.00 ± 0.08 | 1.00 ± 0.06 |
| 5 µM | 1.13 ± 0.13 | 1.53 ± 0.38 | 1.85 ± 0.13 | 4.02 ± 0.43 | |
| MPK6 | 0 µM | 1.00 ± 0.04 | 1.00 ± 0.09 | 1.00 ± 0.05 | 1.00 ± 0.02 |
| 5 µM | 1.39 ± 0.01 | 1.45 ± 0.09 | 1.00 ± 0.07 | 2.04 ± 0.14 | |
| WRKY33 | 0 µM | 1.00 ± 0.02 | 1.00 ± 0.20 | 1.00 ± 0.07 | 1.00 ± 0.16 |
| 5 µM | 1.30 ± 0.27 | 2.65 ± 0.78 | 3.87 ± 1.43 | 13.43 ± 0.93 | |
| Root | |||||
| ACS2 | 0 µM | 1.00 ± 0.12 | 1.00 ± 0.07 | 1.00 ± 0.22 | 1.00 ± 0.11 |
| 5 µM | 1.13 ± 0.11 | 0.78 ± 0.11 | 0.75 ± 0.08 | 3.76 ± 1.22 | |
| ACS6 | 0 µM | 1.00 ± 0.02 | 1.00 ± 0.07 | 1.00 ± 0.14 | 1.00 ± 0.09 |
| 5 µM | 1.61 ± 0.16 | 1.72 ± 0.17 | 1.60 ± 0.17 | 5.25 ± 1.83 | |
| ACO2 | 0 µM | 1.00 ± 0.21 | 1.00 ± 0.13 | 1.00 ± 0.09 | 1.00 ± 0.31 |
| 5 µM | 1.11 ± 0.17 | 1.15 ± 0.07 | 2.23 ± 0.11 | 5.05 ± 0.34 | |
| ACO4 | 0 µM | 1.00 ± 0.17 | 1.00 ± 0.11 | 1.00 ± 0.15 | 1.00 ± 0.11 |
| 5 µM | 1.18 ± 0.10 | 1.51 ± 0.14 | 1.95 ± 0.12 | 7.39 ± 1.23 | |
| ERF1 | 0 µM | 1.00 ± 0.37 | 1.00 ± 0.33 | 1.00 ± 0.17 | 1.00 ± 0.02 |
| 5 µM | 1.27 ± 0.13 | 1.39 ± 0.20 | 3.01 ± 0.50 | 22.01 ± 2.75 | |
| OXI1 | 0 µM | 1.00 ± 0.08 | 1.00 ± 0.07 | 1.00 ± 0.12 | 1.00 ± 0.14 |
| 5 µM | 1.27 ± 0.13 | 1.70 ± 0.11 | 1.22 ± 0.10 | 1.20 ± 0.37 | |
| MPK3 | 0 µM | 1.00 ± 0.12 | 1.00 ± 0.02 | 1.00 ± 0.07 | 1.00 ± 0.12 |
| 5 µM | 1.68 ± 0.18 | 1.51 ± 0.10 | 1.31 ± 0.07 | 2.89 ± 0.36 | |
| MPK6 | 0 µM | 1.00 ± 0.14 | 1.00 ± 0.06 | 1.00 ± 0.07 | 1.00 ± 0.02 |
| 5 µM | 1.63 ± 0.13 | 1.11 ± 0.02 | 2.93 ± 0.26 | 2.36 ± 0.21 | |
| WRKY33 | 0 µM | 1.00 ± 0.30 | 1.00 ± 0.09 | 1.00 ± 0.23 | 1.00 ± 0.16 |
| 5 µM | 1.15 ± 0.09 | 1.93 ± 0.02 | 0.97 ± 0.04 | 1.74 ± 0.44 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deckers, J.; Hendrix, S.; Prinsen, E.; Vangronsveld, J.; Cuypers, A. Identifying the Pressure Points of Acute Cadmium Stress Prior to Acclimation in Arabidopsis thaliana. Int. J. Mol. Sci. 2020, 21, 6232. https://doi.org/10.3390/ijms21176232
Deckers J, Hendrix S, Prinsen E, Vangronsveld J, Cuypers A. Identifying the Pressure Points of Acute Cadmium Stress Prior to Acclimation in Arabidopsis thaliana. International Journal of Molecular Sciences. 2020; 21(17):6232. https://doi.org/10.3390/ijms21176232
Chicago/Turabian StyleDeckers, Jana, Sophie Hendrix, Els Prinsen, Jaco Vangronsveld, and Ann Cuypers. 2020. "Identifying the Pressure Points of Acute Cadmium Stress Prior to Acclimation in Arabidopsis thaliana" International Journal of Molecular Sciences 21, no. 17: 6232. https://doi.org/10.3390/ijms21176232
APA StyleDeckers, J., Hendrix, S., Prinsen, E., Vangronsveld, J., & Cuypers, A. (2020). Identifying the Pressure Points of Acute Cadmium Stress Prior to Acclimation in Arabidopsis thaliana. International Journal of Molecular Sciences, 21(17), 6232. https://doi.org/10.3390/ijms21176232







