Application of Mesenchymal Stem Cell Therapy and Inner Ear Regeneration for Hearing Loss: A Review
Abstract
1. Introduction
2. Regeneration of the Inner Ear
3. Potential of Cell Therapy for Sensory Cells Using MSCs
4. Cell Therapy for Inner Ear Spiral Ganglion Neuron Degeneration Using MSCs
5. Three-Dimensional (3D) Organoid Model of the Inner Ear (Cochlea and Vestibular Organ)
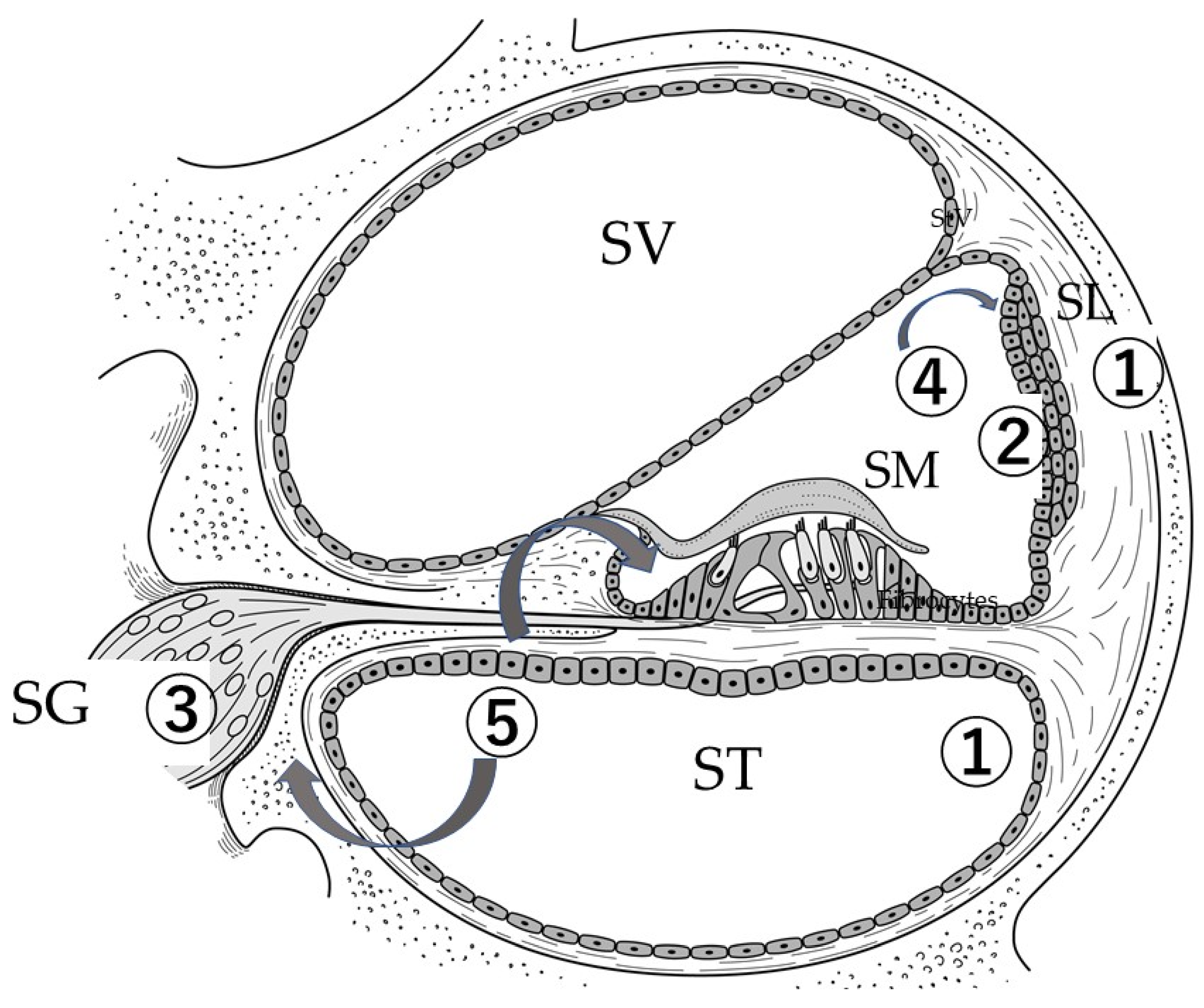
6. Administration of Stem Cells into the Inner Ear
7. Imaging for Tracking Transplanted Cells
8. Auditory Rehabilitation after Stem Cell Transplantation
9. Addressing Clinical Problems Associated with Inner Ear Regenerative Medicine
- Scenario (1): when only hair cells are lost but supporting cells and neurons remain.
- Scenario (2): when all the cells in the inner ear, including all the hair cells, the supporting cells, and all the neurons, are lost.
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SL | Spiral ligament |
| StV | Stria vacularis |
| SG | Spiral ganglion |
| SGN | Spiral ganglion neuron |
| MSCs | Mesenchymal stem cells |
| iPSCs | Induced pluripotent stem cells |
| ESCs | Embryonic stem cells |
| OPCs | Otic progenitor cells |
| SPIO | Superparamagnetic iron oxide |
| MRI | Magnetic resonance imaging |
| BM-MNCs | Bone marrow-derived mononuclear cells |
| BM-MSCs | Bone marrow-derived mesenchymal stem cells |
| HSCs | Hematopoietic stem cells |
| ASCs | Adipose tissue-derived stem cells |
| IV | Intravenous injection |
| SV | Scala vestibuli |
| SM | Scala media |
| ST | Scala tympani |
References
- World Health Organization. Deafness and Hearing Loss. Available online: www.who.int/en/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 1 March 2020).
- Liberman, M.; Rowsowski, J.; Lewis, R. Physiology and pathophysiology. In Schuknecht’s Pathology of the Ear, 3rd ed.; Merchant, S., Nadol, J.J., Eds.; PMPH: Shelton, CT, USA, 2010; pp. 98–136. [Google Scholar]
- Goncalves, S.; Bas, E.; Langston, M.; Grobman, A.; Goldstein, B.J.; Angeli, S. Histologic changes of mesenchymal stem cell repair of tympanic membrane perforation. Acta Otolaryngol. 2017, 137, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Maharajan, N.; Cho, G.W.; Jang, C.H. Application of mesenchymal stem cell for tympanic membrane regeneration by tissue engineering approach. Int. J. Pediatr. Otorhinolaryngol. 2020, 133, 109969. [Google Scholar] [CrossRef] [PubMed]
- Nadol, J.J. Disorders of unknown of multiple causes. In Shuknecht’s Pathology of the Ear; Merchant, S., Nadol, J.J., Eds.; PMPH: Shelton, CT, USA, 2010; pp. 572–630. [Google Scholar]
- Bermingham, N.A.; Hassan, B.A.; Price, S.D.; Vollrath, M.A.; Ben-Arie, N.; Eatock, R.A.; Bellen, H.J.; Lysakowski, A.; Zoghbi, H.Y. Math1: An essential gene for the generation of inner ear hair cells. Science 1999, 284, 1837–1841. [Google Scholar] [CrossRef]
- Brodal, P. Structure and Function; Oxford University Press: Oxford, UK, 1998; pp. 281–295. [Google Scholar]
- Lalu, M.M.; McIntyre, L.; Pugliese, C.; Fergusson, D.; Winston, B.W.; Marshall, J.C.; Granton, J.; Stewart, D.J.; Canadian Critical Care Trials Group. Safety of cell therapy with mesenchymal stromal cells (SafeCell): A systematic review and meta-analysis of clinical trials. PLoS ONE 2012, 7, e47559. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, K.; Semi, K.; Yamamoto, T.; Shimizu, M.; Tanaka, A.; Mitsunaga, K.; Okita, K.; Osafune, K.; Arioka, Y.; Maeda, T.; et al. Premature termination of reprogramming in vivo leads to cancer development through altered epigenetic regulation. Cell 2014, 156, 663–677. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Longaker, M.T.; Wu, J.C. Human iPS cell-based therapy: Considerations before clinical applications. Cell Cycle 2010, 9, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Schnabel, L.V.; Abratte, C.M.; Schimenti, J.C.; Felippe, M.J.; Cassano, J.M.; Southard, T.L.; Cross, J.A.; Fortier, L.A. Induced pluripotent stem cells have similar immunogenic and more potent immunomodulatory properties compared with bone marrow-derived stromal cells in vitro. Regen. Med. 2014, 9, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Tappenbeck, N.; Schroder, H.M.; Niebergall-Roth, E.; Hassinger, F.; Dehio, U.; Dieter, K.; Kraft, K.; Kerstan, A.; Esterlechner, J.; Frank, N.Y.; et al. In vivo safety profile and biodistribution of GMP-manufactured human skin-derived ABCB5-positive mesenchymal stromal cells for use in clinical trials. Cytotherapy 2019, 21, 546–560. [Google Scholar] [CrossRef] [PubMed]
- Eshraghi, A.A.; Ocak, E.; Zhu, A.; Mittal, J.; Davies, C.; Shahal, D.; Bulut, E.; Sinha, R.; Shah, V.; Perdomo, M.M.; et al. Biocompatibility of Bone Marrow-Derived Mesenchymal Stem Cells in the Rat Inner Ear following Trans-Tympanic Administration. J. Clin. Med. 2020, 9, 1711. [Google Scholar] [CrossRef] [PubMed]
- Izumikawa, M.; Minoda, R.; Kawamoto, K.; Abrashkin, K.A.; Swiderski, D.L.; Dolan, D.F.; Brough, D.E.; Raphael, Y. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat. Med. 2005, 11, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Hertzano, R.; Montcouquiol, M.; Rashi-Elkeles, S.; Elkon, R.; Yucel, R.; Frankel, W.N.; Rechavi, G.; Moroy, T.; Friedman, T.B.; Kelley, M.W.; et al. Transcription profiling of inner ears from Pou4f3(ddl/ddl) identifies Gfi1 as a target of the Pou4f3 deafness gene. Hum. Mol. Genet. 2004, 13, 2143–2153. [Google Scholar] [CrossRef] [PubMed]
- Wallis, D.; Hamblen, M.; Zhou, Y.; Venken, K.J.; Schumacher, A.; Grimes, H.L.; Zoghbi, H.Y.; Orkin, S.H.; Bellen, H.J. The zinc finger transcription factor Gfi1, implicated in lymphomagenesis, is required for inner ear hair cell differentiation and survival. Development 2003, 130, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Menendez, L.; Trecek, T.; Gopalakrishnan, S.; Tao, L.; Markowitz, A.L.; Yu, H.V.; Wang, X.; Llamas, J.; Huang, C.; Lee, J.; et al. Generation of inner ear hair cells by direct lineage conversion of primary somatic cells. Elife 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Mizutari, K.; Fujioka, M.; Hosoya, M.; Bramhall, N.; Okano, H.J.; Okano, H.; Edge, A.S. Notch inhibition induces cochlear hair cell regeneration and recovery of hearing after acoustic trauma. Neuron 2013, 77, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Kloosterman, W.; Fekete, D.M. MicroRNA-183 family members regulate sensorineural fates in the inner ear. J. Neurosci. 2010, 30, 3254–3263. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zou, S.; Wu, F.; He, Z.; Kong, W. Role of microRNA in inner ear stem cells and related research progress. Am. J. Stem. Cells 2020, 9, 16–24. [Google Scholar] [PubMed]
- Chang, S.Y.; Carpena, N.T.; Mun, S.; Jung, J.Y.; Chung, P.S.; Shim, H.; Han, K.; Ahn, J.C.; Lee, M.Y. Enhanced Inner-Ear Organoid Formation from Mouse Embryonic Stem Cells by Photobiomodulation. Mol. Ther. Methods Clin. Dev. 2020, 17, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Ito, J.; Kojima, K.; Kawaguchi, S. Survival of neural stem cells in the cochlea. Acta Otolaryngol. 2001, 121, 140–142. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, S. Gene Delivery into the Inner Ear and Its Clinical Implications for Hearing and Balance. Molecules 2018, 23, 2507. [Google Scholar] [CrossRef] [PubMed]
- Nishida, A.; Takahashi, M.; Tanihara, H.; Nakano, I.; Takahashi, J.B.; Mizoguchi, A.; Ide, C.; Honda, Y. Incorporation and differentiation of hippocampus-derived neural stem cells transplanted in injured adult rat retina. Investig. Ophthalmol. Vis. Sci. 2000, 41, 4268–4274. [Google Scholar] [PubMed]
- Tateya, I.; Nakagawa, T.; Iguchi, F.; Kim, T.S.; Endo, T.; Yamada, S.; Kageyama, R.; Naito, Y.; Ito, J. Fate of neural stem cells grafted into injured inner ears of mice. Neuroreport 2003, 14, 1677–1681. [Google Scholar] [CrossRef] [PubMed]
- Oshima, K.; Shin, K.; Diensthuber, M.; Peng, A.W.; Ricci, A.J.; Heller, S. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell 2010, 141, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Hosoya, M.; Fujioka, M.; Sone, T.; Okamoto, S.; Akamatsu, W.; Ukai, H.; Ueda, H.R.; Ogawa, K.; Matsunaga, T.; Okano, H. Cochlear Cell Modeling Using Disease-Specific iPSCs Unveils a Degenerative Phenotype and Suggests Treatments for Congenital Progressive Hearing Loss. Cell Rep. 2017, 18, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Alford, R.L.; Friedman, T.B.; Keats, B.J.; Kimberling, W.J.; Proud, V.K.; Smith, R.J.; Arnos, K.S.; Korf, B.R.; Rehm, H.L.; Toriello, H.V. Early childhood hearing loss: Clinical and molecular genetics. An educational slide set of the American College of Medical Genetics. Genet. Med. 2003, 5, 338–341. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kim, W.J.; Gong, J.S.; Park, K.H. Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients. J. Audiol. Otol. 2018, 22, 105–109. [Google Scholar] [CrossRef] [PubMed]
- McLean, W.J.; McLean, D.T.; Eatock, R.A.; Edge, A.S. Distinct capacity for differentiation to inner ear cell types by progenitor cells of the cochlea and vestibular organs. Development 2016, 143, 4381–4393. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, H.; Heller, S. Pluripotent stem cells from the adult mouse inner ear. Nat. Med. 2003, 9, 1293–1299. [Google Scholar] [CrossRef] [PubMed]
- Yoo, T.J.; Du, X.; Zhou, B. The paracrine effect of mesenchymal human stem cells restored hearing in beta-tubulin induced autoimmune sensorineural hearing loss. Hear. Res. 2015, 330, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.; Yang, Y.; Omelchenko, I.; Nuttall, A.L.; Kachelmeier, A.; Xiu, R.; Shi, X. Bone marrow cell recruitment mediated by inducible nitric oxide synthase/stromal cell-derived factor-1alpha signaling repairs the acoustically damaged cochlear blood-labyrinth barrier. Am. J. Pathol. 2010, 177, 3089–3099. [Google Scholar] [CrossRef] [PubMed]
- Naito, Y.; Nakamura, T.; Nakagawa, T.; Iguchi, F.; Endo, T.; Fujino, K.; Kim, T.S.; Hiratsuka, Y.; Tamura, T.; Kanemaru, S.; et al. Transplantation of bone marrow stromal cells into the cochlea of chinchillas. Neuroreport 2004, 15, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Watada, Y.; Yamashita, D.; Toyoda, M.; Tsuchiya, K.; Hida, N.; Tanimoto, A.; Ogawa, K.; Kanzaki, S.; Umezawa, A. Magnetic resonance monitoring of superparamagnetic iron oxide (SPIO)-labeled stem cells transplanted into the inner ear. Neurosci. Res. 2015, 95, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Kada, S.; Hamaguchi, K.; Ito, J.; Omori, K.; Nakagawa, T. Bone Marrow Stromal Cells Accelerate Hearing Recovery via Regeneration or Maintenance of Cochlear Fibrocytes in Mouse Spiral Ligaments. Anat. Rec. 2020, 303, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Lang, H.; Ebihara, Y.; Schmiedt, R.A.; Minamiguchi, H.; Zhou, D.; Smythe, N.; Liu, L.; Ogawa, M.; Schulte, B.A. Contribution of bone marrow hematopoietic stem cells to adult mouse inner ear: Mesenchymal cells and fibrocytes. J. Comp. Neurol. 2006, 496, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Hakuba, N.; Morizane, I.; Fujita, K.; Cao, F.; Zhu, P.; Uchida, N.; Kameda, K.; Sakanaka, M.; Gyo, K.; et al. Hematopoietic stem cells prevent hair cell death after transient cochlear ischemia through paracrine effects. Neuroscience 2007, 145, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, K.; Fujinami, Y.; Hoya, N.; Okamoto, Y.; Kouike, H.; Komatsuzaki, R.; Kusano, R.; Nakagawa, S.; Satoh, H.; Fujii, M.; et al. Mesenchymal stem cell transplantation accelerates hearing recovery through the repair of injured cochlear fibrocytes. Am. J. Pathol. 2007, 171, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Kasagi, H.; Kuhara, T.; Okada, H.; Sueyoshi, N.; Kurihara, H. Mesenchymal stem cell transplantation to the mouse cochlea as a treatment for childhood sensorineural hearing loss. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.Y.; Song, J.J.; Chang, S.O.; Kim, S.U.; Oh, S.H. Intravenous administration of human mesenchymal stem cells after noise- or drug-induced hearing loss in rats. Acta Otolaryngol. 2012, 132 (Suppl. 1), S94–S102. [Google Scholar] [CrossRef] [PubMed]
- Bas, E.; Van De Water, T.R.; Lumbreras, V.; Rajguru, S.; Goss, G.; Hare, J.M.; Goldstein, B.J. Adult human nasal mesenchymal-like stem cells restore cochlear spiral ganglion neurons after experimental lesion. Stem Cells Dev. 2014, 23, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Schack, L.; Budde, S.; Lenarz, T.; Krettek, C.; Gross, G.; Windhagen, H.; Hoffmann, A.; Warnecke, A. Induction of neuronal-like phenotype in human mesenchymal stem cells by overexpression of Neurogenin1 and treatment with neurotrophins. Tissue Cell 2016, 48, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Roemer, A.; Kohl, U.; Majdani, O.; Kloss, S.; Falk, C.; Haumann, S.; Lenarz, T.; Kral, A.; Warnecke, A. Biohybrid cochlear implants in human neurosensory restoration. Stem Cell Res. Ther. 2016, 7, 148. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Nakagawa, T.; Kita, T.; Kada, S.; Yoshimoto, M.; Nakahata, T.; Ito, J. Bone marrow-derived cells expressing Iba1 are constitutively present as resident tissue macrophages in the mouse cochlea. J. Neurosci. Res. 2008, 86, 1758–1767. [Google Scholar] [CrossRef] [PubMed]
- Seyyedi, M.; Viana, L.M.; Nadol, J.B., Jr. Within-subject comparison of word recognition and spiral ganglion cell count in bilateral cochlear implant recipients. Otol. Neurotol. 2014, 35, 1446–1450. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, S.; Stover, T.; Kawamoto, K.; Prieskorn, D.M.; Altschuler, R.A.; Miller, J.M.; Raphael, Y. Glial cell line-derived neurotrophic factor and chronic electrical stimulation prevent VIII cranial nerve degeneration following denervation. J. Comp. Neurol. 2002, 454, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Leake, P.A.; Stakhovskaya, O.; Hetherington, A.; Rebscher, S.J.; Bonham, B. Effects of brain-derived neurotrophic factor (BDNF) and electrical stimulation on survival and function of cochlear spiral ganglion neurons in deafened, developing cats. J. Assoc. Res. Otolaryngol. 2013, 14, 187–211. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, A.J.; Kondo, T.; Miyamoto, R.T.; Hashino, E. Enhanced survival of bone-marrow-derived pluripotent stem cells in an animal model of auditory neuropathy. Laryngoscope 2007, 117, 1629–1635. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Jongkamonwiwat, N.; Abbas, L.; Eshtan, S.J.; Johnson, S.L.; Kuhn, S.; Milo, M.; Thurlow, J.K.; Andrews, P.W.; Marcotti, W.; et al. Restoration of auditory evoked responses by human ES-cell-derived otic progenitors. Nature 2012, 490, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, F.; Nakagawa, T.; Tateya, I.; Kim, T.S.; Endo, T.; Taniguchi, Z.; Naito, Y.; Ito, J. Trophic support of mouse inner ear by neural stem cell transplantation. Neuroreport 2003, 14, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, S.A.; Higashi, A.Y.; Loomis, B.; Schrepfer, T.; Wan, G.; Corfas, G.; Dressler, G.R.; Duncan, R.K. From Otic Induction to Hair Cell Production: Pax2(EGFP) Cell Line Illuminates Key Stages of Development in Mouse Inner Ear Organoid Model. Stem Cells Dev. 2018, 27, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Hartman, B.H.; Bscke, R.; Ellwanger, D.C.; Keymeulen, S.; Scheibinger, M.; Heller, S. Fbxo2(VHC) mouse and embryonic stem cell reporter lines delineate in vitro-generated inner ear sensory epithelia cells and enable otic lineage selection and Cre-recombination. Dev. Biol. 2018, 443, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Perny, M.; Ting, C.C.; Kleinlogel, S.; Senn, P.; Roccio, M. Generation of Otic Sensory Neurons from Mouse Embryonic Stem Cells in 3D Culture. Front. Cell Neurosci. 2017, 11, 409. [Google Scholar] [CrossRef] [PubMed]
- Koehler, K.R.; Hashino, E. 3D mouse embryonic stem cell culture for generating inner ear organoids. Nat. Protoc. 2014, 9, 1229–1244. [Google Scholar] [CrossRef] [PubMed]
- de Groot, S.C.; Sliedregt, K.; van Benthem, P.P.G.; Rivolta, M.N.; Huisman, M.A. Building an Artificial Stem Cell Niche: Prerequisites for Future 3D-Formation of Inner Ear Structures—Toward 3D Inner Ear Biotechnology. Anat. Rec. 2020, 303, 408–426. [Google Scholar] [CrossRef] [PubMed]
- Lenz, D.R.; Gunewardene, N.; Abdul-Aziz, D.E.; Wang, Q.; Gibson, T.M.; Edge, A.S.B. Applications of Lgr5-Positive Cochlear Progenitors (LCPs) to the Study of Hair Cell Differentiation. Front. Cell Dev. Biol. 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Jeong, M.; O’Reilly, M.; Kirkwood, N.K.; Al-Aama, J.; Lako, M.; Kros, C.J.; Armstrong, L. Generating inner ear organoids containing putative cochlear hair cells from human pluripotent stem cells. Cell Death Dis. 2018, 9, 922. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.C.; Hashino, E.; Nelson, R.F. Progress in Modeling and Targeting Inner Ear Disorders with Pluripotent Stem Cells. Stem Cell Rep. 2020, 14, 996–1008. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.C.; Alex, A.L.; Nie, J.; Lee, J.; Roth, A.A.; Booth, K.T.; Koehler, K.R.; Hashino, E.; Nelson, R.F. Defective Tmprss3-Associated Hair Cell Degeneration in Inner Ear Organoids. Stem Cell Rep. 2019, 13, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, S.; Kawamoto, K.; Oh, S.H.; Stover, T.; Suzuki, M.; Ishimoto, S.; Yagi, M.; Miller, J.M.; Lomax, M.I.; Raphael, Y. From gene identification to gene therapy. Audiol. Neurootol. 2002, 7, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Prieskorn, D.M.; Miller, J.M. Technical report: Chronic and acute intracochlear infusion in rodents. Hear. Res. 2000, 140, 212–215. [Google Scholar] [CrossRef]
- Slepecky, N.B. Spinger Handbook Structure of the Mammalian Cochlea. In The Cochlea; Dallos, P., Fay, R.R., Eds.; Springer: New York, NY, USA, 1996; pp. 44–129. [Google Scholar]
- Yamasoba, T.; Yagi, M.; Roessler, B.J.; Miller, J.M.; Raphael, Y. Inner ear transgene expression after adenoviral vector inoculation in the endolymphatic sac. Hum. Gene Ther. 1999, 10, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Kitahara, T.; Kubo, T.; Okumura, S.; Kitahara, M. Effects of endolymphatic sac drainage with steroids for intractable Meniere’s disease: A long-term follow-up and randomized controlled study. Laryngoscope 2008, 118, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, S.; Shiotani, A.; Inoue, M.; Hasegawa, M.; Ogawa, K. Sendai virus vector-mediated transgene expression in the cochlea in vivo. Audiol. Neurootol. 2007, 12, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Mittal, R.; Ocak, E.; Zhu, A.; Perdomo, M.M.; Pena, S.A.; Mittal, J.; Bohorquez, J.; Eshraghi, A.A. Effect of Bone Marrow-Derived Mesenchymal Stem Cells on Cochlear Function in an Experimental Rat Model. Anat. Rec. 2020, 303, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Aazh, H.; Moore, B.C. Audiological Rehabilitation for Facilitating Hearing Aid Use: A Review. J. Am. Acad. Audiol. 2017, 28, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Hassepass, F.; Arndt, S.; Aschendorff, A.; Laszig, R.; Wesarg, T. Cochlear implantation for hearing rehabilitation in single-sided deafness after translabyrinthine vestibular schwannoma surgery. Eur. Arch. Otorhinolaryngol. 2016, 273, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- McKay, C.M. Brain Plasticity and Rehabilitation with a Cochlear Implant. Adv. Otorhinolaryngol. 2018, 81, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Naples, J.G.; Ruckenstein, M.J. Cochlear Implant. Otolaryngol. Clin. N. Am. 2020, 53, 87–102. [Google Scholar] [CrossRef] [PubMed]
|
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanzaki, S.; Toyoda, M.; Umezawa, A.; Ogawa, K. Application of Mesenchymal Stem Cell Therapy and Inner Ear Regeneration for Hearing Loss: A Review. Int. J. Mol. Sci. 2020, 21, 5764. https://doi.org/10.3390/ijms21165764
Kanzaki S, Toyoda M, Umezawa A, Ogawa K. Application of Mesenchymal Stem Cell Therapy and Inner Ear Regeneration for Hearing Loss: A Review. International Journal of Molecular Sciences. 2020; 21(16):5764. https://doi.org/10.3390/ijms21165764
Chicago/Turabian StyleKanzaki, Sho, Masashi Toyoda, Akihiro Umezawa, and Kaoru Ogawa. 2020. "Application of Mesenchymal Stem Cell Therapy and Inner Ear Regeneration for Hearing Loss: A Review" International Journal of Molecular Sciences 21, no. 16: 5764. https://doi.org/10.3390/ijms21165764
APA StyleKanzaki, S., Toyoda, M., Umezawa, A., & Ogawa, K. (2020). Application of Mesenchymal Stem Cell Therapy and Inner Ear Regeneration for Hearing Loss: A Review. International Journal of Molecular Sciences, 21(16), 5764. https://doi.org/10.3390/ijms21165764






