Decreased Expression of ZNF554 in Gliomas is Associated with the Activation of Tumor Pathways and Shorter Patient Survival
Abstract
1. Introduction
2. Results
2.1. Brain Tissue Expression Pattern of ZNF554
2.2. ZNF554 Protein Expression in Gliomas
2.3. ZNF554 mRNA Expression in Gliomas
2.4. The Effect of ZNF554 Overexpression in Glioblastoma Cells
3. Discussion
3.1. Principal Findings of This Study:
3.2. ZNFs in the Normal Human Brain
3.3. ZNFs are Double-Edged Swords in Different Cancer Types
3.4. ZNF554 as a Potential Tumor Suppressor in Gliomas
3.5. Potential Diagnostic and Therapeutic Implications of ZNF554 in Gliomas
3.6. Strengths and Limitations
4. Conclusions
5. Materials and Methods
5.1. Analysis of ZNF554 mRNA Expression in Normal Brain
5.2. Analysis of ZNF554 Protein Expression in Gliomas
5.3. Immunohistochemistry for ZNF554
5.4. Evaluation of ZNF554 Immunostainings
5.5. Analysis of ZNF554 mRNA Expression and Patients’ Survival in Gliomas
5.6. Transient Transfection of U87 Glioblastoma Cells with ZNF554
5.7. Total RNA Isolation
5.8. Labeling and Microarray Hybridization
5.9. Quantitative Real-Time PCR
5.10. Proliferation/Viability Assay
5.11. Cell Cycle Assay
5.12. Data Analysis
5.12.1. Allen Brain Atlas Data Analysis
5.12.2. TCGA RNA-Seq Data Analysis
5.12.3. Microarray Data Analysis of Transfected U87 Cells
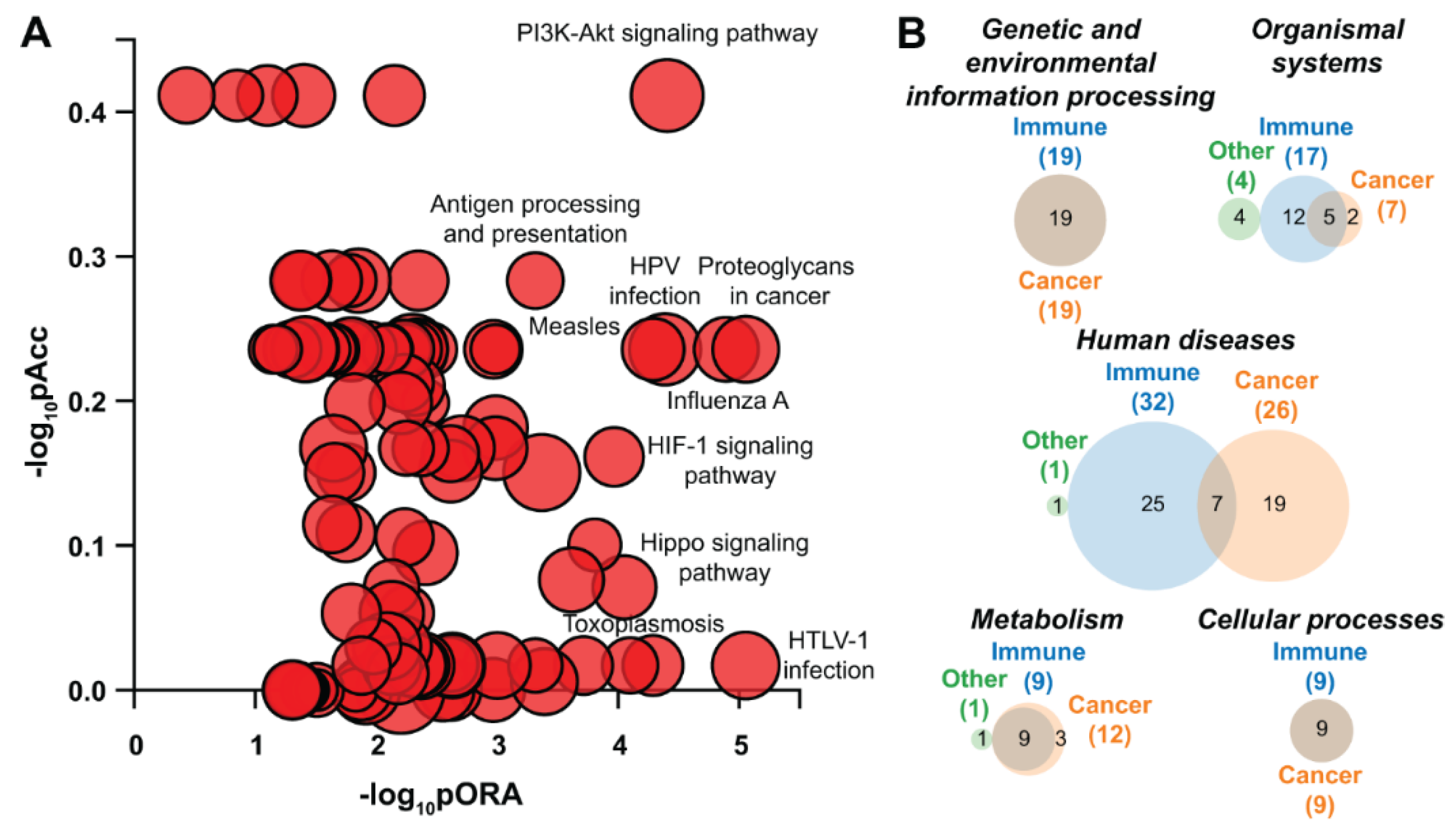
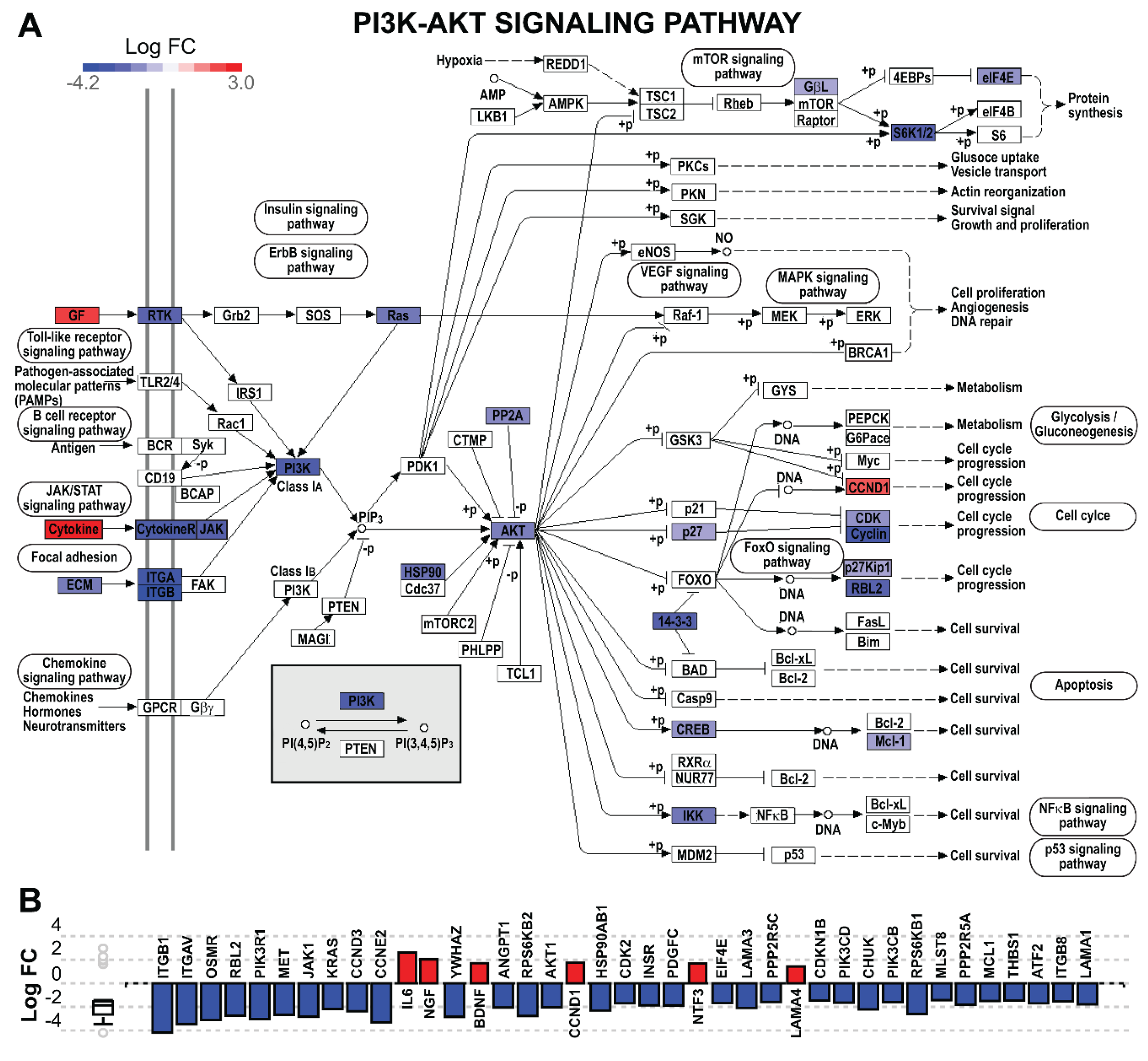
5.12.4. Pathway Analysis
5.12.5. Quantitative RT-PCR Data Analysis
5.12.6. Data Analysis of ZNF554 Immunostainings
5.12.7. Data Analysis of Cell Proliferation/Viability and Cell Cycle
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cassandri, M.; Smirnov, A.; Novelli, F.; Pitolli, C.; Agostini, M.; Malewicz, M.; Melino, G.; Raschella, G. Zinc-finger proteins in health and disease. Cell Death Discov. 2017, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Brayer, K.J.; Kulshreshtha, S.; Segal, D.J. The protein-binding potential of C2H2 zinc finger domains. Cell Biochem. Biophys. 2008, 51, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Font, J.; Mackay, J.P. Beyond DNA: Zinc finger domains as RNA-binding modules. In Engineered Zinc Finger Proteins; Font, J., Mackay, J.P., Eds.; Humana Press: Totowa, NJ, USA, 2010; Volume 649, pp. 479–491. [Google Scholar]
- Planel, S.; Salomon, A.; Jalinot, P.; Feige, J.J.; Cherradi, N. A novel concept in antiangiogenic and antitumoral therapy: Multitarget destabilization of short-lived mRNAs by the zinc finger protein ZFP36L1. Oncogene 2010, 29, 5989–6003. [Google Scholar] [CrossRef] [PubMed]
- Jen, J.; Wang, Y.C. Zinc finger proteins in cancer progression. J. Biomed. Sci. 2016, 23, 53. [Google Scholar] [CrossRef] [PubMed]
- Nicolai, S.; Mahen, R.; Raschella, G.; Marini, A.; Pieraccioli, M.; Malewicz, M.; Venkitaraman, A.R.; Melino, G. ZNF281 is recruited on DNA breaks to facilitate DNA repair by non-homologous end joining. Oncogene 2020, 39, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Rowe, H.M.; Jakobsson, J.; Mesnard, D.; Rougemont, J.; Reynard, S.; Aktas, T.; Maillard, P.V.; Layard-Liesching, H.; Verp, S.; Marquis, J.; et al. KAP1 controls endogenous retroviruses in embryonic stem cells. Nature 2010, 463, 237–240. [Google Scholar] [CrossRef]
- Lupo, A.; Cesaro, E.; Montano, G.; Zurlo, D.; Izzo, P.; Costanzo, P. KRAB-Zinc Finger Proteins: A Repressor Family Displaying Multiple Biological Functions. Curr. Genomics. 2013, 14, 268–278. [Google Scholar] [CrossRef]
- Ecco, G.; Imbeault, M.; Trono, D. KRAB zinc finger proteins. Development 2017, 144, 2719–2729. [Google Scholar] [CrossRef]
- O’Geen, H.; Squazzo, S.L.; Iyengar, S.; Blahnik, K.; Rinn, J.L.; Chang, H.Y.; Green, R.; Farnham, P.J. Genome-wide analysis of KAP1 binding suggests autoregulation of KRAB-ZNFs. PLoS Genet. 2007, 3, e89. [Google Scholar] [CrossRef]
- Oleksiewicz, U.; Gladych, M.; Raman, A.T.; Heyn, H.; Mereu, E.; Chlebanowska, P.; Andrzejewska, A.; Sozanska, B.; Samant, N.; Fak, K.; et al. TRIM28 and Interacting KRAB-ZNFs Control Self-Renewal of Human Pluripotent Stem Cells through Epigenetic Repression of Pro-differentiation Genes. Stem Cell Rep. 2017, 9, 2065–2080. [Google Scholar] [CrossRef]
- Vogel, M.J.; Guelen, L.; de Wit, E.; Peric-Hupkes, D.; Loden, M.; Talhout, W.; Feenstra, M.; Abbas, B.; Classen, A.K.; van Steensel, B. Human heterochromatin proteins form large domains containing KRAB-ZNF genes. Genome Res. 2006, 16, 1493–1504. [Google Scholar] [CrossRef] [PubMed]
- Than, N.G.; Romero, R.; Tarca, A.L.; Kekesi, K.A.; Xu, Y.; Xu, Z.; Juhasz, K.; Bhatti, G.; Leavitt, R.J.; Gelencser, Z.; et al. Integrated Systems Biology Approach Identifies Novel Maternal and Placental Pathways of Preeclampsia. Front. Immunol. 2018, 9, 1661. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Ren, F.; Xu, X.; Ren, Y.; Li, H.; Wang, Y.; Zhai, Y.; Chang, Z. ZNF536, a novel zinc finger protein specifically expressed in the brain, negatively regulates neuron differentiation by repressing retinoic acid-induced gene transcription. Mol. Cell Biol. 2009, 29, 3633–3643. [Google Scholar] [CrossRef] [PubMed]
- Nowick, K.; Gernat, T.; Almaas, E.; Stubbs, L. Differences in human and chimpanzee gene expression patterns define an evolving network of transcription factors in brain. Proc. Natl. Acad. Sci. USA 2009, 106, 22358–22363. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, E.J.; Hitomi, M.; Oh, S.Y.; Jin, X.; Jeon, H.M.; Beck, S.; Jin, X.; Kim, J.K.; Park, C.G.; et al. The LIM-only transcription factor LMO2 determines tumorigenic and angiogenic traits in glioma stem cells. Cell Death Differ. 2015, 22, 1517–1525. [Google Scholar] [CrossRef]
- Schwartzbaum, J.A.; Fisher, J.L.; Aldape, K.D.; Wrensch, M. Epidemiology and molecular pathology of glioma. Nat. Clin. Pract. Neurol. 2006, 2, 494–503. [Google Scholar] [CrossRef]
- Louis, D.N.; Ohgaki, H.; Wiestler, O.D.; Cavenee, W.K. WHO Classification of Tumours of the Central Nervous System, Revised, 4th ed.; IARC Press: Lyon, France, 2016; p. 408. [Google Scholar]
- Gittleman, H.; Boscia, A.; Ostrom, Q.T.; Truitt, G.; Fritz, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. Survivorship in adults with malignant brain and other central nervous system tumor from 2000–2014. Neuro Oncol. 2018, 20, vii6–vii16. [Google Scholar] [CrossRef]
- Zhu, Z.; Li, K.; Xu, D.; Liu, Y.; Tang, H.; Xie, Q.; Xie, L.; Liu, J.; Wang, H.; Gong, Y.; et al. ZFX regulates glioma cell proliferation and survival In Vitro and In Vivo. J. Neuro Oncol. 2013, 112, 17–25. [Google Scholar] [CrossRef]
- Fang, X.; Huang, Z.; Zhou, W.; Wu, Q.; Sloan, A.E.; Ouyang, G.; McLendon, R.E.; Yu, J.S.; Rich, J.N.; Bao, S. The zinc finger transcription factor ZFX is required for maintaining the tumorigenic potential of glioblastoma stem cells. Stem Cells 2014, 32, 2033–2047. [Google Scholar] [CrossRef]
- Tatard, V.M.; Xiang, C.; Biegel, J.A.; Dahmane, N. ZNF238 is expressed in postmitotic brain cells and inhibits brain tumor growth. Cancer Res. 2010, 70, 1236–1246. [Google Scholar] [CrossRef]
- Bosnyak, E.; Michelhaugh, S.K.; Klinger, N.V.; Kamson, D.O.; Barger, G.R.; Mittal, S.; Juhasz, C. Prognostic Molecular and Imaging Biomarkers in Primary Glioblastoma. Clin. Nucl. Med. 2017, 42, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Marosvari, D.; Nagy, N.; Kriston, C.; Deak, B.; Hajdu, M.; Bodor, C.; Csala, I.; Bago, A.G.; Szallasi, Z.; Sebestyen, A.; et al. Discrepancy Between Low Levels of mTOR Activity and High Levels of P-S6 in Primary Central Nervous System Lymphoma May Be Explained by PAS Domain-Containing Serine/Threonine-Protein Kinase-Mediated Phosphorylation. J. Neuropathol. Exp. Neurol. 2018, 77, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 2008, 455, 1061–1068. [Google Scholar] [CrossRef]
- Huntley, S.; Baggott, D.M.; Hamilton, A.T.; Tran-Gyamfi, M.; Yang, S.; Kim, J.; Gordon, L.; Branscomb, E.; Stubbs, L. A comprehensive catalog of human KRAB-associated zinc finger genes: Insights into the evolutionary history of a large family of transcriptional repressors. Genome Res. 2006, 16, 669–677. [Google Scholar] [CrossRef]
- Imbeault, M.; Helleboid, P.Y.; Trono, D. KRAB zinc-finger proteins contribute to the evolution of gene regulatory networks. Nature 2017, 543, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Nowick, K.; Fields, C.; Gernat, T.; Caetano-Anolles, D.; Kholina, N.; Stubbs, L. Gain, loss and divergence in primate zinc-finger genes: A rich resource for evolution of gene regulatory differences between species. PLoS ONE 2011, 6, e21553. [Google Scholar] [CrossRef]
- Farmiloe, G.; Lodewijk, G.A.; Robben, S.F.; van Bree, E.J.; Jacobs, F.M.J. Widespread correlation of KRAB zinc finger protein binding with brain-developmental gene expression patterns. Philos. Trans. R. Soc. B 2020, 375, 20190333. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, J.; Jiang, H.; Xie, T.; Zheng, J.; Tian, Y.; Li, R.; Wang, B.; Lin, J.; Xu, A.; et al. The Tumor Suppressor Role of Zinc Finger Protein 671 (ZNF671) in Multiple Tumors Based on Cancer Single-Cell Sequencing. Front. Oncol. 2019, 9, 1214. [Google Scholar] [CrossRef]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjostedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef]
- Guan, C.; He, L.; Chang, Z.; Gu, X.; Liang, J.; Liu, R. ZNF774 is a potent suppressor of hepatocarcinogenesis through dampening the NOTCH2 signaling. Oncogene 2020, 39, 1665–1680. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, X.; Yu, L.; Zhang, C.; Ye, L.; Ren, D.; Li, Y.; Sun, X.; Yu, L.; Ouyang, Y.; et al. Zinc finger protein 367 promotes metastasis by inhibiting the Hippo pathway in breast cancer. Oncogene 2020, 39, 2568–2582. [Google Scholar] [CrossRef] [PubMed]
- Murat, A.; Migliavacca, E.; Hussain, S.F.; Heimberger, A.B.; Desbaillets, I.; Hamou, M.F.; Ruegg, C.; Stupp, R.; Delorenzi, M.; Hegi, M.E. Modulation of angiogenic and inflammatory response in glioblastoma by hypoxia. PLoS ONE 2009, 4, e5947. [Google Scholar] [CrossRef] [PubMed]
- Pang, F.; Zha, R.; Zhao, Y.; Wang, Q.; Chen, D.; Zhang, Z.; Chen, T.; Yao, M.; Gu, J.; He, X. MiR-525-3p enhances the migration and invasion of liver cancer cells by downregulating ZNF395. PLoS ONE 2014, 9, e90867. [Google Scholar] [CrossRef] [PubMed]
- Nakada, M.; Kita, D.; Watanabe, T.; Hayashi, Y.; Teng, L.; Pyko, I.V.; Hamada, J. Aberrant signaling pathways in glioma. Cancers 2011, 3, 3242–3278. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.; Maiello, M.R.; D’Alessio, A.; Pergameno, M.; Normanno, N. The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: Role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin. Ther. Targets 2012, 16, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, B.A.; Restuccia, D.F. PI3K-PKB/Akt pathway. Cold Spring Harb. Perspect. Biol. 2012, 4, a011189. [Google Scholar] [CrossRef] [PubMed]
- Alqurashi, N.; Hashimi, S.M.; Wei, M.Q. Chemical Inhibitors and microRNAs (miRNA) Targeting the Mammalian Target of Rapamycin (mTOR) Pathway: Potential for Novel Anticancer Therapeutics. Int. J. Mol. Sci. 2013, 14, 3874–3900. [Google Scholar] [CrossRef]
- Park, H.K.; Hong, J.H.; Oh, Y.T.; Kim, S.S.; Yin, J.; Lee, A.J.; Chae, Y.C.; Kim, J.H.; Park, S.H.; Park, C.K.; et al. Interplay between TRAP1 and Sirtuin-3 Modulates Mitochondrial Respiration and Oxidative Stress to Maintain Stemness of Glioma Stem Cells. Cancer Res. 2019, 79, 1369–1382. [Google Scholar] [CrossRef]
- Szarvas, T.; Csizmarik, A.; Szucs, M.; Nyiradi, P. Molecular subtypes and perspectives of targeted therapies in prostate cancer. Orv. Hetil. 2019, 7, 252–263. [Google Scholar] [CrossRef]
- Bottyan, K.; Kemeny, L.; Csoma, Z.R. Successful treatment of facial angiofibromas with local sirolimus in childhood in Bourneville-Pringle disease. Orv. Hetil. 2019, 13, 516–520. [Google Scholar]
- Li, X.; Wu, C.; Chen, N.; Gu, H.; Yen, A.; Cao, L.; Wang, E.; Wang, L. PI3K/Akt/mTOR signaling pathway and targeted therapy for glioblastoma. Oncotarget 2016, 7, 33440–33450. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.F.; Wang, J.; Shao, W.; Wu, C.P.; Chen, Z.P.; To, S.T.; Li, W.P. Recent advances in the use of PI3K inhibitors for glioblastoma multiforme: Current preclinical and clinical development. Mol. Cancer 2017, 16, 100. [Google Scholar] [CrossRef] [PubMed]
- Juric, V.; Murphy, B. Cyclin-dependent kinase inhibitors in brain cancer: Current state and future directions. Cancer Drug Resist. 2020, 3, 48–62. [Google Scholar] [CrossRef]
- Jean-Quartier, C.; Jeanquartier, F.; Holzinger, A. Open Data for Differential Network Analysis in Glioma. Int. J. Mol. Sci. 2020, 21, 547. [Google Scholar] [CrossRef]
- Wang, L.; Wei, B.; Hu, G.; Wang, L.; Bi, M.; Sun, Z.; Jin, Y. Screening of differentially expressed genes associated with human glioblastoma and functional analysis using a DNA microarray. Mol. Med. Rep. 2015, 12, 1991–1996. [Google Scholar] [CrossRef]
- Huang, H.; Hara, A.; Homma, T.; Yonekawa, Y.; Ohgaki, H. Altered expression of immune defense genes in pilocytic astrocytomas. J. Neuropathol. Exp. Neurol. 2005, 64, 891–901. [Google Scholar] [CrossRef]
- Zagzag, D.; Salnikow, K.; Chiriboga, L.; Yee, H.; Lan, L.; Ali, M.A.; Garcia, R.; Demaria, S.; Newcomb, E.W. Downregulation of major histocompatibility complex antigens in invading glioma cells: Stealth invasion of the brain. Lab. Investig. 2005, 85, 328–341. [Google Scholar] [CrossRef]
- Caffery, B.; Lee, J.S.; Alexander-Bryant, A.A. Vectors for Glioblastoma Gene Therapy: Viral & Non-Viral Delivery Strategies. Nanomaterials 2019, 9, 105. [Google Scholar] [CrossRef]
- Lang, F.F.; Bruner, J.M.; Fuller, G.N.; Aldape, K.; Prados, M.D.; Chang, S.; Berger, M.S.; McDermott, M.W.; Kunwar, S.M.; Junck, L.R.; et al. Phase I trial of adenovirus-mediated p53 gene therapy for recurrent glioma: Biological and clinical results. J. Clin. Oncol. 2003, 21, 2508–2518. [Google Scholar] [CrossRef]
- Hong, Y.K.; Joe, Y.A.; Yang, Y.J.; Lee, K.S.; Son, B.C.; Jeun, S.S.; Chung, D.S.; Cho, K.K.; Park, C.K.; Kim, M.C.; et al. Potentials and limitations of adenovirus-p53 gene therapy for brain tumors. J. Korean Med. Sci. 2000, 15, 315–322. [Google Scholar] [CrossRef]
- Okura, H.; Smith, C.A.; Rutka, J.T. Gene therapy for malignant glioma. Mol. Cell. Ther. 2014, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- Mees, C.; Nemunaitis, J.; Senzer, N. Transcription factors: Their potential as targets for an individualized therapeutic approach to cancer. Cancer Gene Ther. 2009, 16, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Hawrylycz, M.J.; Lein, E.S.; Guillozet-Bongaarts, A.L.; Shen, E.H.; Ng, L.; Miller, J.A.; van de Lagemaat, L.N.; Smith, K.A.; Ebbert, A.; Riley, Z.L.; et al. An anatomically comprehensive atlas of the adult human brain transcriptome. Nature 2012, 489, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Wesseling, P.; Capper, D. WHO 2016 Classification of gliomas. Neuropathol. Appl. Neurobiol. 2018, 44, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, M.; Barthel, F.P.; Malta, T.M.; Sabedot, T.S.; Salama, S.R.; Murray, B.A.; Morozova, O.; Newton, Y.; Radenbaugh, A.; Pagnotta, S.M.; et al. Molecular Profiling Reveals Biologically Discrete Subsets and Pathways of Progression in Diffuse Glioma. Cell 2016, 164, 550–563. [Google Scholar] [CrossRef]
- Goldman, M.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. The UCSC Xena platform for public and private cancer genomics data visualization and interpretation. BioRxiv 2019, 326470. [Google Scholar] [CrossRef]
- Sarkozy, M.; Zvara, A.; Gyemant, N.; Fekete, V.; Kocsis, G.F.; Pipis, J.; Szucs, G.; Csonka, C.; Puskas, L.G.; Ferdinandy, P.; et al. Metabolic syndrome influences cardiac gene expression pattern at the transcript level in male ZDF rats. Cardiovasc. Diabetol. 2013, 12, 16. [Google Scholar] [CrossRef]
- Kauffmann, A.; Gentleman, R.; Huber, W. arrayQualityMetrics—A bioconductor package for quality assessment of microarray data. Bioinformatics 2009, 25, 415–416. [Google Scholar] [CrossRef]
- Benjamini, Y.; Drai, D.; Elmer, G.; Kafkafi, N.; Golani, I. Controlling the false discovery rate in behavior genetics research. Behav. Brain Res. 2001, 125, 279–284. [Google Scholar] [CrossRef]
- Draghici, S.; Khatri, P.; Tarca, A.L.; Amin, K.; Done, A.; Voichita, C.; Georgescu, C.; Romero, R. A systems biology approach for pathway level analysis. Genome Res. 2007, 17, 1537–1545. [Google Scholar] [CrossRef]
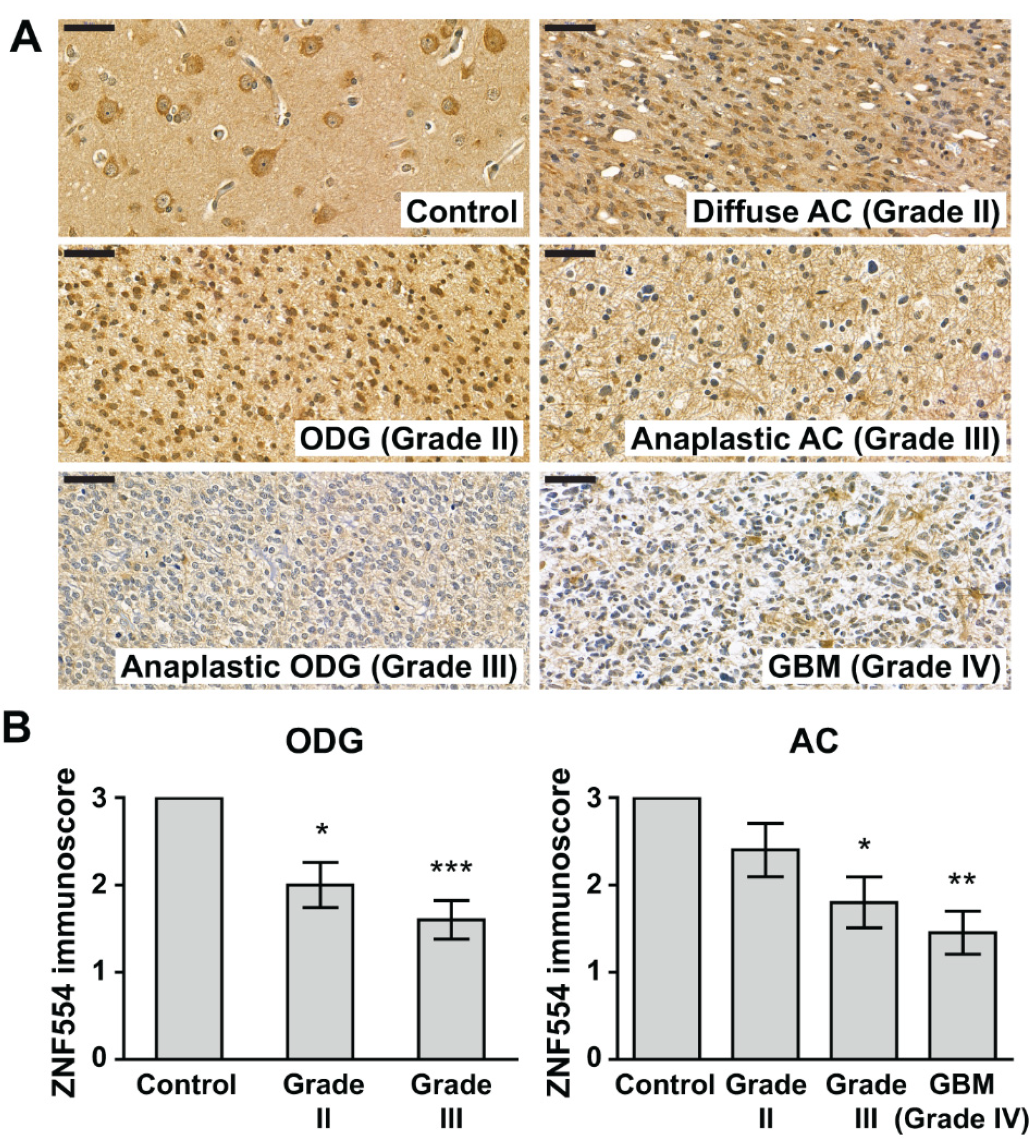
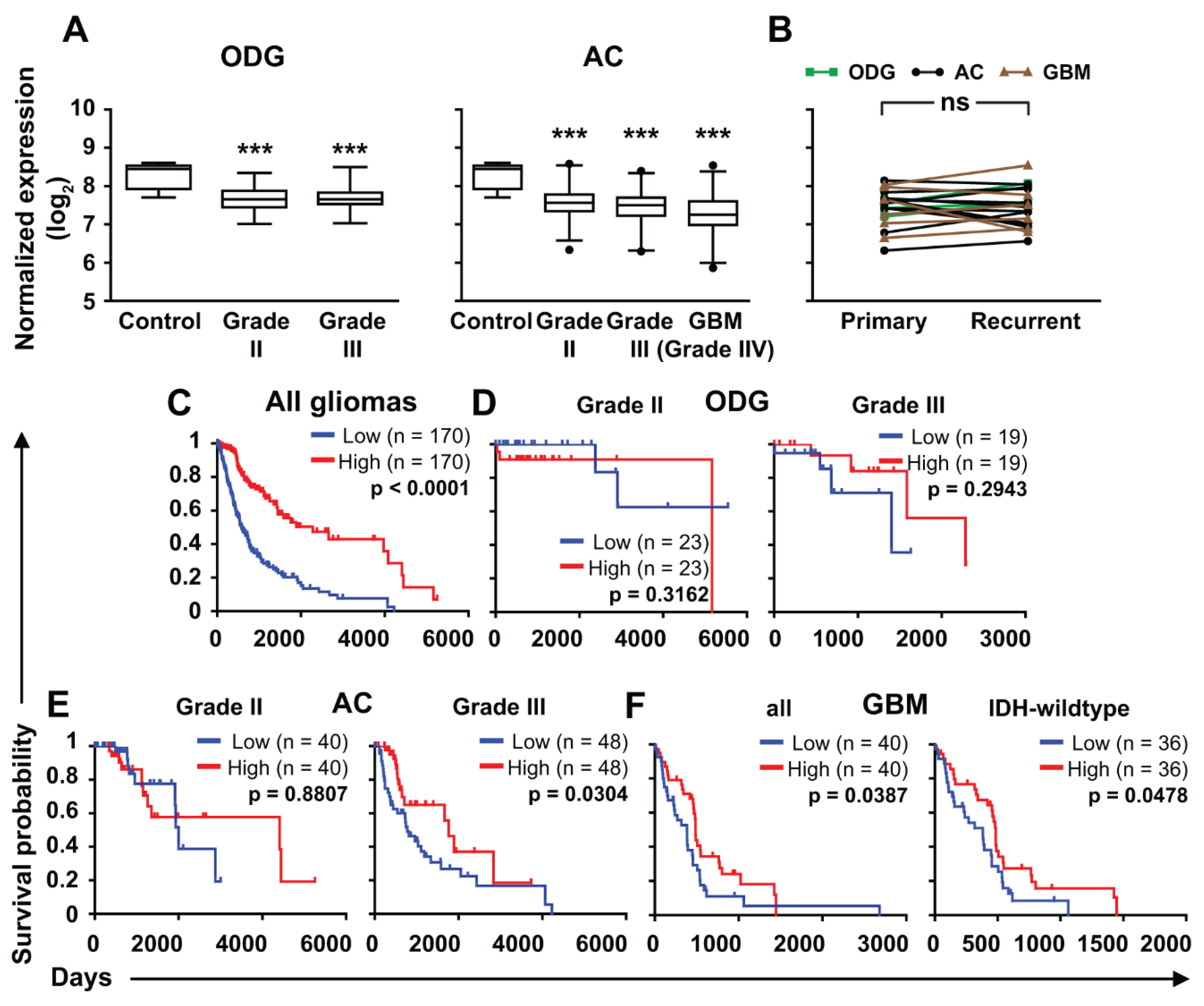
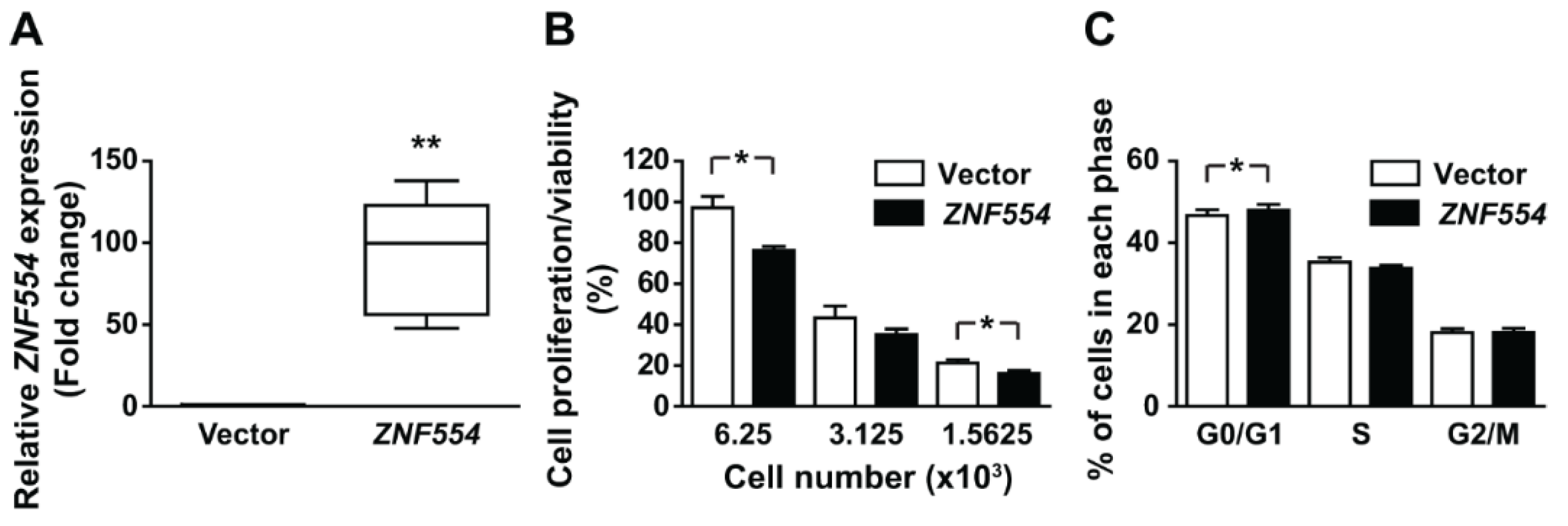
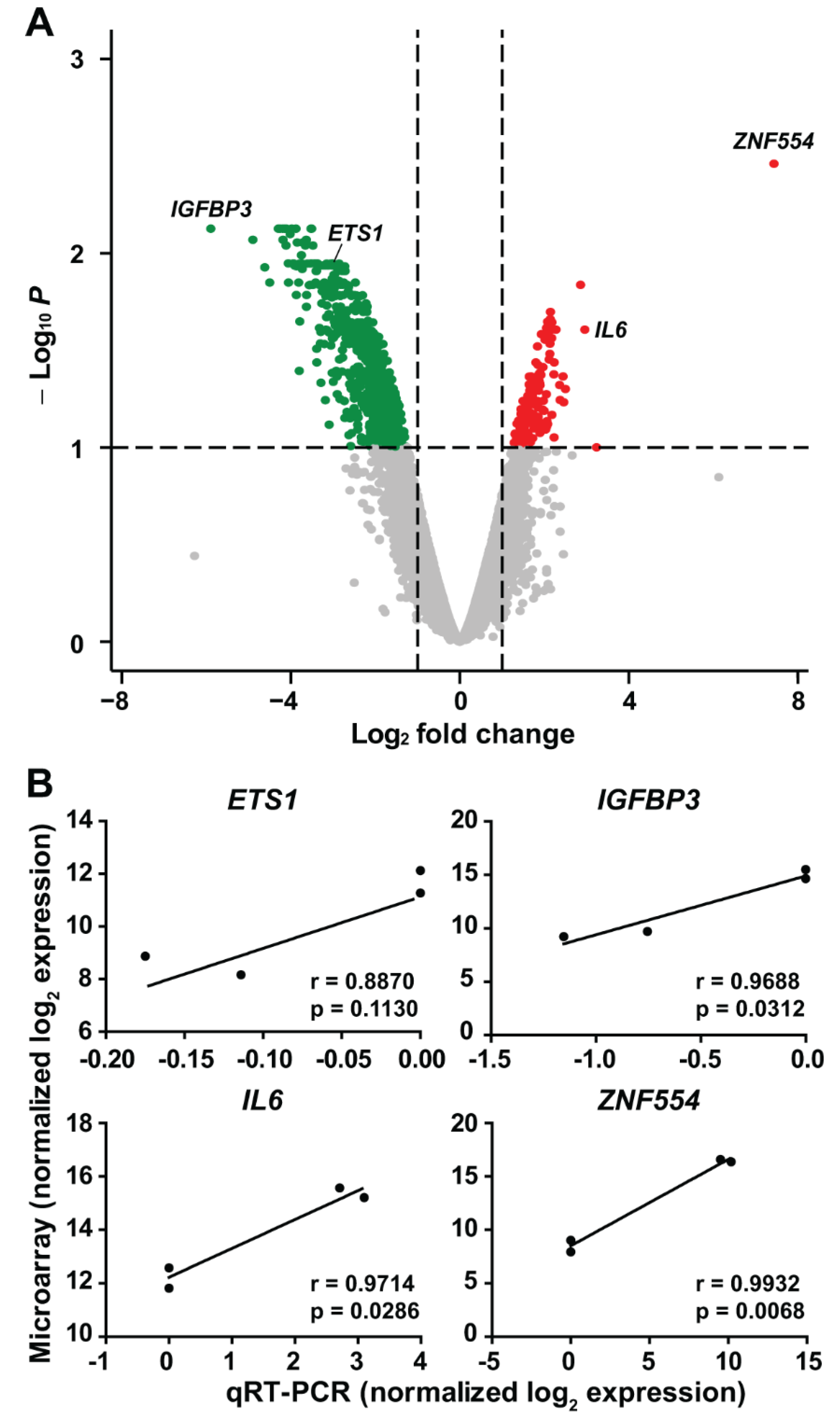
| Groups | Grade | na | Age (Years) at Diagnosis a | Male/Female b |
|---|---|---|---|---|
| Control (epilepsy) | - | 11 | 38.0 (14–64) | 5/6 |
| Oligodendroglioma | II | 10 | 51.5 (29–65) | 3/7 |
| Anaplastic oligondendroglioma | III | 10 | 47.0 (30–67) | 3/7 |
| Diffuse astrocytoma | II | 10 | 47.0 (29–69) | 4/6 |
| Anaplastic astrocytoma | III | 10 | 41.5 (31–78) | 6/4 |
| Glioblastoma multiforme | IV | 11 | 64.0 (24–73) | 6/5 |
| Gene Symbol | Gene Name | Assay ID |
|---|---|---|
| ZNF554 | Zinc finger protein 554 | Hs00171072_m1 |
| IL6 | Interleukin 6 | Hs00174131_m1 |
| IGFBP3 | Insulin-like growth factor-binding protein 3 | Hs00181211_m1 |
| RPLP0 | Ribosomal protein lateral stalk subunit P0 | Hs99999902_m1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balogh, A.; Reiniger, L.; Hetey, S.; Kiraly, P.; Toth, E.; Karaszi, K.; Juhasz, K.; Gelencser, Z.; Zvara, A.; Szilagyi, A.; et al. Decreased Expression of ZNF554 in Gliomas is Associated with the Activation of Tumor Pathways and Shorter Patient Survival. Int. J. Mol. Sci. 2020, 21, 5762. https://doi.org/10.3390/ijms21165762
Balogh A, Reiniger L, Hetey S, Kiraly P, Toth E, Karaszi K, Juhasz K, Gelencser Z, Zvara A, Szilagyi A, et al. Decreased Expression of ZNF554 in Gliomas is Associated with the Activation of Tumor Pathways and Shorter Patient Survival. International Journal of Molecular Sciences. 2020; 21(16):5762. https://doi.org/10.3390/ijms21165762
Chicago/Turabian StyleBalogh, Andrea, Lilla Reiniger, Szabolcs Hetey, Peter Kiraly, Eszter Toth, Katalin Karaszi, Kata Juhasz, Zsolt Gelencser, Agnes Zvara, Andras Szilagyi, and et al. 2020. "Decreased Expression of ZNF554 in Gliomas is Associated with the Activation of Tumor Pathways and Shorter Patient Survival" International Journal of Molecular Sciences 21, no. 16: 5762. https://doi.org/10.3390/ijms21165762
APA StyleBalogh, A., Reiniger, L., Hetey, S., Kiraly, P., Toth, E., Karaszi, K., Juhasz, K., Gelencser, Z., Zvara, A., Szilagyi, A., Puskas, L. G., Matko, J., Papp, Z., Kovalszky, I., Juhasz, C., & Than, N. G. (2020). Decreased Expression of ZNF554 in Gliomas is Associated with the Activation of Tumor Pathways and Shorter Patient Survival. International Journal of Molecular Sciences, 21(16), 5762. https://doi.org/10.3390/ijms21165762








