Increase of Neutrophil Activation Markers in Venous Thrombosis—Contribution of Circulating Activated Protein C
Abstract
1. Introduction
2. Results
2.1. Clinical Characteristics of the Study Subjects
2.2. Levels of Neutrophil Activation Markers in Patients and Healthy Controls
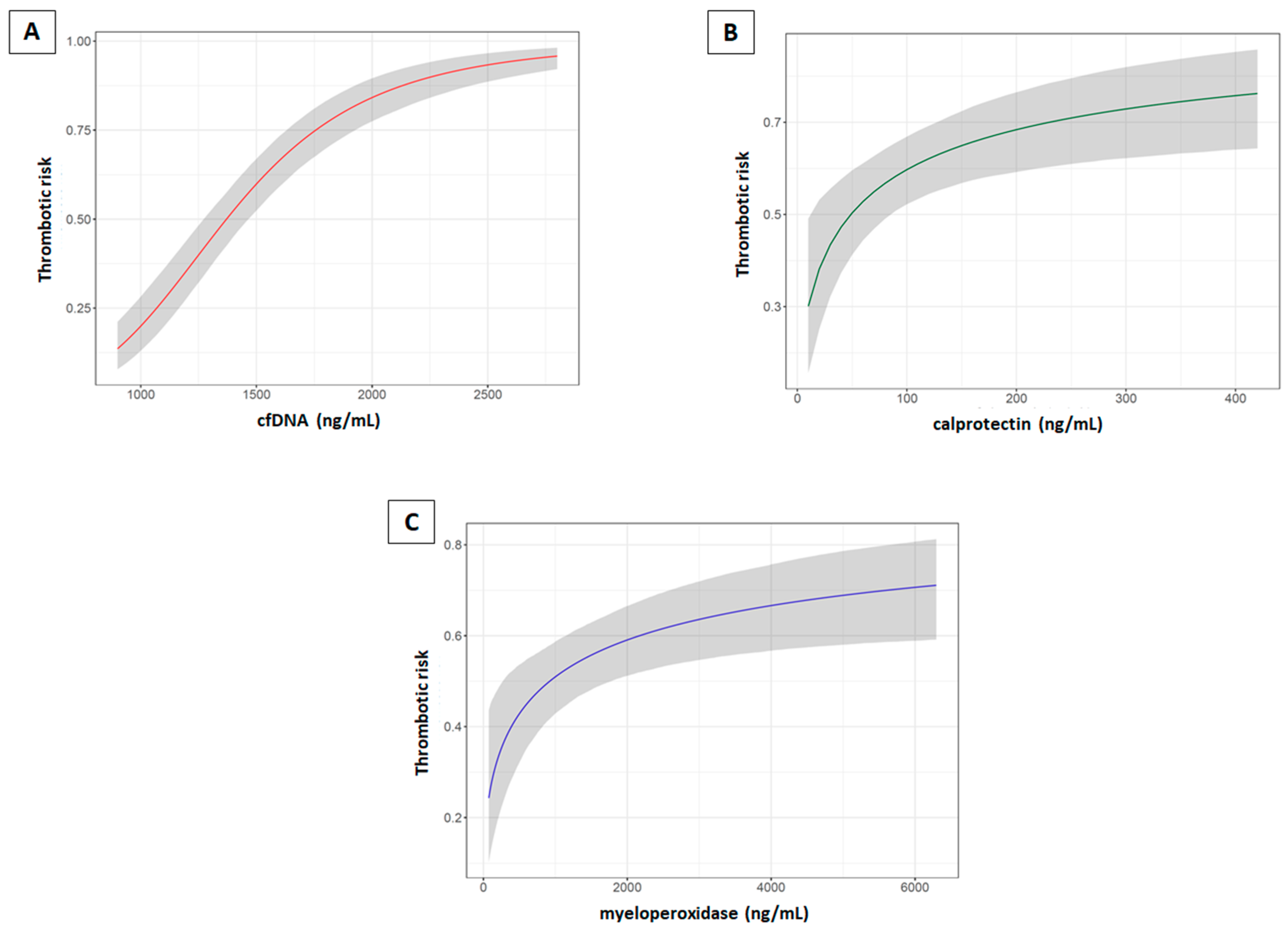
2.3. Neutrophil Activation Markers and Risk of Thrombosis
2.4. Activated Protein C Plasma Levels and Its Genetic Regulation
3. Discussion
4. Materials and Methods
4.1. Study Subjects
4.2. Blood Collection
4.3. Quantification of Neutrophil Activation Markers
4.4. Quantification of Plasma APC, sEPCR, and sTM
4.5. Genotyping of the Genetic Regulators of Plasma APC: PROCR Haplotypes and THBD c.1418C>T Polymorphism
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APC | Activated Protein C |
| cfDNA | Cell-free DNA |
| DVT | Deep vein thrombosis |
| EPCR | Endothelial protein C receptor |
| H | Haplotype |
| MPO | Myeloperoxidase |
| NETs | Neutrophil extracellular traps |
| NLR | Neutrophil-to-lymphocyte ratio |
| PE | Pulmonary embolism |
| PROCR | Gene encoding for the endothelial protein C receptor |
| sEPCR | Soluble endothelial protein C receptor |
| sTM | Soluble thrombomodulin |
| SVT | Splanchnic vein thrombosis |
| THBD | Gene encoding for thrombomodulin |
| TM | Thrombomodulin |
| VTE | Venous thromboembolism |
References
- Engelmann, B.; Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 2013, 13, 34–45. [Google Scholar] [CrossRef] [PubMed]
- von Bruhl, M.L.; Stark, K.; Steinhart, A.; Chandraratne, S.; Konrad, I.; Lorenz, M.; Khandoga, A.; Tirniceriu, A.; Coletti, R.; Kollnberger, M.; et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 2012, 209, 819–835. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, J.M.; Gordon, A.E.; Henke, P.K. Resolution of Deep Venous Thrombosis: Proposed Immune Paradigms. Int. J. Mol. Sci. 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, T.A.; Brill, A.; Wagner, D.D. Neutrophil extracellular trap (NET) impact on deep vein thrombosis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- von Köckritz-Blickwede, M.; Nizet, V. Innate immunity turned inside-out: Antimicrobial defense by phagocyte extracellular traps. J. Mol. Med. 2009, 87, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, T.A.; Brill, A.; Duerschmied, D.; Schatzberg, D.; Monestier, M.; Myers, D.D., Jr.; Wrobleski, S.K.; Wakefield, T.W.; Hartwig, J.H.; Wagner, D.D. Extracellular DNA traps promote thrombosis. Proc. Natl. Acad. Sci. USA 2010, 107, 15880–15885. [Google Scholar] [CrossRef] [PubMed]
- Nickel, K.F.; Long, A.T.; Fuchs, T.A.; Butler, L.M.; Renne, T. Factor XII as a Therapeutic Target in Thromboembolic and Inflammatory Diseases. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 13–20. [Google Scholar] [CrossRef]
- Folco, E.J.; Mawson, T.L.; Vromman, A.; Bernardes-Souza, B.; Franck, G.; Persson, O.; Nakamura, M.; Newton, G.; Luscinskas, F.W.; Libby, P. Neutrophil Extracellular Traps Induce Endothelial Cell Activation and Tissue Factor Production Through Interleukin-1alpha and Cathepsin G. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 1901–1912. [Google Scholar] [CrossRef]
- Massberg, S.; Grahl, L.; von Bruehl, M.L.; Manukyan, D.; Pfeiler, S.; Goosmann, C.; Brinkmann, V.; Lorenz, M.; Bidzhekov, K.; Khandagale, A.B.; et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 2010, 16, 887–896. [Google Scholar] [CrossRef]
- Glaser, C.B.; Morser, J.; Clarke, J.H.; Blasko, E.; McLean, K.; Kuhn, I.; Chang, R.J.; Lin, J.H.; Vilander, L.; Andrews, W.H.; et al. Oxidation of a specific methionine in thrombomodulin by activated neutrophil products blocks cofactor activity. A potential rapid mechanism for modulation of coagulation. J. Clin. Investig. 1992, 90, 2565–2573. [Google Scholar] [CrossRef]
- Ammollo, C.T.; Semeraro, F.; Xu, J.; Esmon, N.L.; Esmon, C.T. Extracellular histones increase plasma thrombin generation by impairing thrombomodulin-dependent protein C activation. J. Thromb. Haemost. 2011, 9, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, M.A.; Zhao, G.; Zhai, L.; David, G., 3rd; Marcus, S.; Krishnaswamy, S.; Poncz, M. Modulation of protein C activation by histones, platelet factor 4, and heparinoids: New insights into activated protein C formation. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 120–126. [Google Scholar] [CrossRef] [PubMed]
- España, F.; Medina, P.; Navarro, S.; Zorio, E.; Estellés, A.; Aznar, J. The multifunctional protein C system. Curr. Med. Chem. Cardiovasc. Hematol. Agents 2005, 3, 119–131. [Google Scholar] [CrossRef] [PubMed]
- España, F.; Vayá, A.; Mira, Y.; Medina, P.; Estellés, A.; Villa, P.; Falcó, C.; Aznar, J. Low level of circulating activated protein C is a risk factor for venous thromboembolism. Thromb. Haemost. 2001, 86, 1368–1373. [Google Scholar]
- Zorio, E.; Navarro, S.; Medina, P.; Estellés, A.; Osa, A.; Rueda, J.; Cubillo, P.; Aznar, J.; España, F. Circulating activated protein C is reduced in young survivors of myocardial infarction and inversely correlates with the severity of coronary lesions. J. Thromb. Haemost. 2006, 4, 1530–1536. [Google Scholar] [CrossRef]
- Soria, J.M.; Navarro, S.; Medina, P.; Souto, R.; Buil, A.; Estellés, A.; Fontcuberta, J.; España, F. Heritability of plasma concentrations of activated protein C in a Spanish population. Blood Coagul. Fibrinolysis Int. J. Haemost. Thromb. 2009, 20, 17–21. [Google Scholar] [CrossRef]
- Medina, P.; Navarro, S.; Bonet, E.; Martos, L.; Estellés, A.; Bertina, R.M.; Vos, H.L.; España, F. Functional analysis of two haplotypes of the human endothelial protein C receptor gene. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 684–690. [Google Scholar] [CrossRef]
- Navarro, S.; Medina, P.; Bonet, E.; Corral, J.; Martínez-Sales, V.; Martos, L.; Rivera, M.; Roselló-Lletí, E.; Alberca, I.; Roldán, V.; et al. Association of the thrombomodulin gene c.1418C>T polymorphism with thrombomodulin levels and with venous thrombosis risk. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 1435–1440. [Google Scholar] [CrossRef]
- Healy, L.D.; Puy, C.; Fernández, J.A.; Mitrugno, A.; Keshari, R.S.; Taku, N.A.; Chu, T.T.; Xu, X.; Gruber, A.; Lupu, F.; et al. Activated protein C inhibits neutrophil extracellular trap formation in vitro and activation in vivo. J. Biol. Chem. 2017, 292, 8616–8629. [Google Scholar] [CrossRef]
- Brill, A.; Fuchs, T.A.; Savchenko, A.S.; Thomas, G.M.; Martinod, K.; De Meyer, S.F.; Bhandari, A.A.; Wagner, D.D. Neutrophil extracellular traps promote deep vein thrombosis in mice. J. Thromb. Haemost. 2012, 10, 136–144. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, W.; Wang, N.; Tall, A.R.; Tabas, I. Mitochondrial Oxidative Stress Promotes Atherosclerosis and Neutrophil Extracellular Traps in Aged Mice. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e99–e107. [Google Scholar] [CrossRef] [PubMed]
- Hakkim, A.; Furnrohr, B.G.; Amann, K.; Laube, B.; Abed, U.A.; Brinkmann, V.; Herrmann, M.; Voll, R.E.; Zychlinsky, A. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc. Natl. Acad. Sci. USA 2010, 107, 9813–9818. [Google Scholar] [CrossRef] [PubMed]
- Demers, M.; Wagner, D.D. NETosis: A new factor in tumor progression and cancer-associated thrombosis. Semin. Thromb. Hemost. 2014, 40, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Alcázar, M.; Napirei, M.; Panda, R.; Kohler, E.C.; Kremer Hovinga, J.A.; Mannherz, H.G.; Peine, S.; Renne, T.; Lammle, B.; Fuchs, T.A. Impaired DNase1-mediated degradation of neutrophil extracellular traps is associated with acute thrombotic microangiopathies. J. Thromb. Haemost. 2015, 13, 732–742. [Google Scholar] [CrossRef] [PubMed]
- van Montfoort, M.L.; Stephan, F.; Lauw, M.N.; Hutten, B.A.; Van Mierlo, G.J.; Solati, S.; Middeldorp, S.; Meijers, J.C.; Zeerleder, S. Circulating nucleosomes and neutrophil activation as risk factors for deep vein thrombosis. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 147–151. [Google Scholar] [CrossRef]
- Díaz, J.A.; Fuchs, T.A.; Jackson, T.O.; Kremer Hovinga, J.A.; Lammle, B.; Henke, P.K.; Myers, D.D., Jr.; Wagner, D.D.; Wakefield, T.W.; for the Michigan Research Venous, G. Plasma DNA is Elevated in Patients with Deep Vein Thrombosis. J. Vasc. Surg. Venous Lymphat. Disord. 2013, 1, 341–348. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Kremer Hovinga, J.A.; Schatzberg, D.; Wagner, D.D.; Lämmle, B. Circulating DNA and myeloperoxidase indicate disease activity in patients with thrombotic microangiopathies. Blood 2012, 120, 1157–1164. [Google Scholar] [CrossRef]
- Vallés, J.; Lago, A.; Santos, M.T.; Latorre, A.M.; Tembl, J.I.; Salom, J.B.; Nieves, C.; Moscardó, A. Neutrophil extracellular traps are increased in patients with acute ischemic stroke: Prognostic significance. Thromb. Haemost. 2017, 117, 1919–1929. [Google Scholar] [CrossRef]
- Mauracher, L.M.; Posch, F.; Martinod, K.; Grilz, E.; Daullary, T.; Hell, L.; Brostjan, C.; Zielinski, C.; Ay, C.; Wagner, D.D.; et al. Citrullinated histone H3, a biomarker of neutrophil extracellular trap formation, predicts the risk of venous thromboembolism in cancer patients. J. Thromb. Haemost. 2018, 16, 508–518. [Google Scholar] [CrossRef]
- Arnalich, F.; Maldifassi, M.C.; Ciria, E.; Codoceo, R.; Renart, J.; Fernández-Capitán, C.; Herruzo, R.; García-Rio, F.; López-Collazo, E.; Montiel, C. Plasma levels of mitochondrial and nuclear DNA in patients with massive pulmonary embolism in the emergency department: A prospective cohort study. Crit. Care 2013, 17, R90. [Google Scholar] [CrossRef]
- Pertiwi, K.R.; van der Wal, A.C.; Pabittei, D.R.; Mackaaij, C.; van Leeuwen, M.B.; Li, X.; de Boer, O.J. Neutrophil Extracellular Traps Participate in All Different Types of Thrombotic and Haemorrhagic Complications of Coronary Atherosclerosis. Thromb. Haemost. 2018, 118, 1078–1087. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yuan, K.; Zhu, Q.; Lu, Q.; Jiang, H.; Zhu, M.; Huang, G.; Xu, A. Andrographolide Ameliorates Rheumatoid Arthritis by Regulating the Apoptosis-NETosis Balance of Neutrophils. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Oto, J.; Navarro, S.; Larsen, A.C.; Solmoirago, M.J.; Plana, E.; Hervás, D.; Fernández-Pardo, A.; España, F.; Kristensen, S.R.; Thorlacius-Ussing, O.; et al. microRNAs and neutrophil activation markers predict venous thrombosis in pancreatic ductal adenocarcinoma and distal extrahepatic cholangiocarcinoma. Int. J. Mol. Sci. 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Oto, J.; Plana, E.; Solmoirago, M.J.; Fernández-Pardo, A.; Hervás, D.; Cana, F.; España, F.; Artoni, A.; Bucciarelli, P.; Carrabba, G.; et al. microRNAs and markers of neutrophil activation as predictors of early incidental post-surgical pulmonary embolism in patients with intracranial tumors. Cancers 2020, 12. [Google Scholar] [CrossRef]
- Jimenez-Alcázar, M.; Limacher, A.; Panda, R.; Méan, M.; Bitterling, J.; Peine, S.; Renné, T.; Beer, J.H.; Aujesky, D.; Lämmle, B.; et al. Circulating extracellular DNA is an independent predictor of mortality in elderly patients with venous thromboembolism. PLoS ONE 2018, 13, e0191150. [Google Scholar] [CrossRef]
- Brinkmann, V.; Goosmann, C.; Kuhn, L.I.; Zychlinsky, A. Automatic quantification of in vitro NET formation. Front. Immunol. 2012, 3, 413. [Google Scholar] [CrossRef]
- Brinkmann, V.; Abu Abed, U.; Goosmann, C.; Zychlinsky, A. Immunodetection of NETs in Paraffin-Embedded Tissue. Front. Immunol. 2016, 7, 513. [Google Scholar] [CrossRef]
- Noubouossie, D.F.; Whelihan, M.F.; Yu, Y.B.; Sparkenbaugh, E.; Pawlinski, R.; Monroe, D.M.; Key, N.S. In vitro activation of coagulation by human neutrophil DNA and histone proteins but not neutrophil extracellular traps. Blood 2017, 129, 1021–1029. [Google Scholar] [CrossRef]
- Fagerhol, M.K. Calprotectin, a faecal marker of organic gastrointestinal abnormality. Lancet 2000, 356, 1783–1784. [Google Scholar] [CrossRef]
- Ricciuto, A.; Griffiths, A.M. Clinical value of fecal calprotectin. Crit. Rev. Clin. Lab. Sci. 2019, 56, 307–320. [Google Scholar] [CrossRef]
- Brun, J.G.; Jonsson, R.; Haga, H.J. Measurement of plasma calprotectin as an indicator of arthritis and disease activity in patients with inflammatory rheumatic diseases. J. Rheumatol. 1994, 21, 733–738. [Google Scholar] [PubMed]
- Tibble, J.A.; Sigthorsson, G.; Bridger, S.; Fagerhol, M.K.; Bjarnason, I. Surrogate markers of intestinal inflammation are predictive of relapse in patients with inflammatory bowel disease. Gastroenterology 2000, 119, 15–22. [Google Scholar] [CrossRef]
- Tibble, J.A.; Sigthorsson, G.; Foster, R.; Scott, D.; Fagerhol, M.K.; Roseth, A.; Bjarnason, I. High prevalence of NSAID enteropathy as shown by a simple faecal test. Gut 1999, 45, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Tibble, J.; Teahon, K.; Thjodleifsson, B.; Roseth, A.; Sigthorsson, G.; Bridger, S.; Foster, R.; Sherwood, R.; Fagerhol, M.; Bjarnason, I. A simple method for assessing intestinal inflammation in Crohn’s disease. Gut 2000, 47, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Kronborg, O.; Ugstad, M.; Fuglerud, P.; Johne, B.; Hardcastle, J.; Scholefield, J.H.; Vellacott, K.; Moshakis, V.; Reynolds, J.R. Faecal calprotectin levels in a high risk population for colorectal neoplasia. Gut 2000, 46, 795–800. [Google Scholar] [CrossRef]
- Mortensen, O.H.; Nielsen, A.R.; Erikstrup, C.; Plomgaard, P.; Fischer, C.P.; Krogh-Madsen, R.; Lindegaard, B.; Petersen, A.M.; Taudorf, S.; Pedersen, B.K. Calprotectin—A novel marker of obesity. PLoS ONE 2009, 4, e7419. [Google Scholar] [CrossRef]
- Jung, S.Y.; Park, Y.B.; Ha, Y.J.; Lee, K.H.; Lee, S.K. Serum calprotectin as a marker for disease activity and severity in adult-onset Still’s disease. J. Rheumatol. 2010, 37, 1029–1034. [Google Scholar] [CrossRef]
- Andreásson, K.; Scheja, A.; Saxne, T.; Ohlsson, B.; Hesselstrand, R. Faecal calprotectin: A biomarker of gastrointestinal disease in systemic sclerosis. J. Intern. Med. 2011, 270, 50–57. [Google Scholar] [CrossRef]
- Bianchi, M.; Niemiec, M.J.; Siler, U.; Urban, C.F.; Reichenbach, J. Restoration of anti-Aspergillus defense by neutrophil extracellular traps in human chronic granulomatous disease after gene therapy is calprotectin-dependent. J. Allergy Clin. Immunol. 2011, 127, 1243–1252.e1247. [Google Scholar] [CrossRef] [PubMed]
- Holmgaard, D.B.; Mygind, L.H.; Titlestad, I.; Madsen, H.; Pedersen, S.S.; Mortensen, O.H.; Pedersen, C. Calprotectin—A marker of mortality in COPD? Results from a prospective cohort study. COPD 2013, 10, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, L.; Nybo, M.; Poulsen, M.K.; Henriksen, J.E.; Dahl, J.; Rasmussen, L.M. Plasma calprotectin and its association with cardiovascular disease manifestations, obesity and the metabolic syndrome in type 2 diabetes mellitus patients. BMC Cardiovasc. Disord. 2014, 14, 196. [Google Scholar] [CrossRef] [PubMed]
- Malham, M.; Carlsen, K.; Riis, L.; Paerregaard, A.; Vind, I.; Fenger, M.; Wewer, V. Plasma calprotectin is superior to serum calprotectin as a biomarker of intestinal inflammation in ulcerative Colitis. Scand. J. Gastroenterol. 2019, 54, 1214–1219. [Google Scholar] [CrossRef] [PubMed]
- Urban, C.F.; Ermert, D.; Schmid, M.; Abu-Abed, U.; Goosmann, C.; Nacken, W.; Brinkmann, V.; Jungblut, P.R.; Zychlinsky, A. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog. 2009, 5, e1000639. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, A.B.; de los Reyes-García, A.M.; Rivera-Caravaca, J.M.; Valledor, P.; García-Barberá, N.; Roldan, V.; Vicente, V.; Martínez, C.; González-Conejero, R. miR-146a regulates neutrophil extracellular trap formation that predicts adverse cardiovascular events in patients with atrial fibrillation. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 892–902. [Google Scholar] [CrossRef]
- Vincent, D.; Klinke, M.; Eschenburg, G.; Trochimiuk, M.; Appl, B.; Tiemann, B.; Bergholz, R.; Reinshagen, K.; Boettcher, M. NEC is likely a NETs dependent process and markers of NETosis are predictive of NEC in mice and humans. Sci. Rep. 2018, 8, 12612. [Google Scholar] [CrossRef]
- Du, P. Neutrophil-Lymphocyte Ratio is a Predictor of Venous Thromboembolism in Gastric Cancer Patients. Clin. Lab. 2019, 65. [Google Scholar] [CrossRef]
- Seo, W.W.; Park, M.S.; Kim, S.E.; Lee, J.H.; Park, D.G.; Han, K.R.; Oh, D.J.; Hyon, M.S. Neutrophil-Lymphocyte Ratio as a Predictor of Venous Thromboembolism after Total Knee Replacement. J. Knee Surg. 2019. [Google Scholar] [CrossRef]
- Kuplay, H.; Erdogan, S.B.; Bastopcu, M.; Arslanhan, G.; Baykan, D.B.; Orhan, G. The neutrophil-lymphocyte ratio and the platelet-lymphocyte ratio correlate with thrombus burden in deep venous thrombosis. J. Vasc. Surg. Venous Lymphat. Disord. 2020, 8, 360–364. [Google Scholar] [CrossRef]
- Farah, R.; Nseir, W.; Kagansky, D.; Khamisy-Farah, R. The role of neutrophil-lymphocyte ratio, and mean platelet volume in detecting patients with acute venous thromboembolism. J. Clin. Lab. Anal. 2020, 34, e23010. [Google Scholar] [CrossRef]
- Zhao, L.; Dai, Q.; Chen, X.; Li, S.; Shi, R.; Yu, S.; Yang, F.; Xiong, Y.; Zhang, R. Neutrophil-to-Lymphocyte Ratio Predicts Length of Stay and Acute Hospital Cost in Patients with Acute Ischemic Stroke. J. Stroke Cerebrovasc. Dis. 2016, 25, 739–744. [Google Scholar] [CrossRef]
- Xue, J.; Huang, W.; Chen, X.; Li, Q.; Cai, Z.; Yu, T.; Shao, B. Neutrophil-to-Lymphocyte Ratio Is a Prognostic Marker in Acute Ischemic Stroke. J. Stroke Cerebrovasc. Dis. 2017, 26, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, A.; Montecucco, F.; Dallegri, F.; Carbone, F.; Luscher, T.F.; Camici, G.G.; Liberale, L. Novel findings in neutrophil biology and their impact on cardiovascular disease. Cardiovasc. Res. 2019, 115, 1266–1285. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, A.; Carbone, F.; Liberale, L.; Mach, F.; Roth, A.; Burger, F.; Pende, A.; Vecchie, A.; Bertolotto, M.; Spinella, G.; et al. Platelet-to-lymphocyte ratio at the time of carotid endarterectomy is associated with acute coronary syndrome occurrence. J. Cardiovasc. Med. 2020, 21, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Naess, I.A.; Christiansen, S.C.; Romundstad, P.; Cannegieter, S.C.; Rosendaal, F.R.; Hammerstrom, J. Incidence and mortality of venous thrombosis: A population-based study. J. Thromb. Haemost. 2007, 5, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Ducroux, C.; Di Meglio, L.; Loyau, S.; Delbosc, S.; Boisseau, W.; Deschildre, C.; Ben Maacha, M.; Blanc, R.; Redjem, H.; Ciccio, G.; et al. Thrombus Neutrophil Extracellular Traps Content Impair tPA-Induced Thrombolysis in Acute Ischemic Stroke. Stroke 2018, 49, 754–757. [Google Scholar] [CrossRef] [PubMed]
- Laridan, E.; Denorme, F.; Desender, L.; Francois, O.; Andersson, T.; Deckmyn, H.; Vanhoorelbeke, K.; De Meyer, S.F. Neutrophil extracellular traps in ischemic stroke thrombi. Ann. Neurol. 2017, 82, 223–232. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, X.; Pelayo, R.; Monestier, M.; Ammollo, C.T.; Semeraro, F.; Taylor, F.B.; Esmon, N.L.; Lupu, F.; Esmon, C.T. Extracellular histones are major mediators of death in sepsis. Nat. Med. 2009, 15, 1318–1321. [Google Scholar] [CrossRef]
- Allam, R.; Kumar, S.V.; Darisipudi, M.N.; Anders, H.J. Extracellular histones in tissue injury and inflammation. J. Mol. Med. 2014, 92, 465–472. [Google Scholar] [CrossRef]
- Mezzano, D.; España, F.; Panes, O.; Medina, P.; Pais, E.; Marshall, G.; Tagle, R.; Downey, P.; Cáceres, S.; González, F.; et al. Increased activation of protein C, but lower plasma levels of free, activated protein C in uraemic patients: Relationship with systemic inflammation and haemostatic activation. Br. J. Haematol. 2001, 113, 905–910. [Google Scholar] [CrossRef]
- Cattaneo, M.; Franchi, F.; Zighetti, M.L.; Martinelli, I.; Asti, D.; Mannucci, P.M. Plasma levels of activated protein C in healthy subjects and patients with previous venous thromboembolism: Relationships with plasma homocysteine levels. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1371–1375. [Google Scholar] [CrossRef]
- Martos, L.; Ramón, L.A.; Oto, J.; Fernández-Pardo, A.; Bonanad, S.; Cid, A.R.; Gruber, A.; Griffin, J.H.; España, F.; Navarro, S.; et al. α2-Macroglobulin is a significant in vivo inhibitor of activated protein C and low APC:α2M levels are associated with venous thromboembolism. Thromb. Haemost. 2018, 118, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Vecht, L.; Zuurbier, S.M.; Meijers, J.C.M.; Coutinho, J.M. Elevated factor VIII increases the risk of cerebral venous thrombosis: A case-control study. J. Neurol. 2018, 265, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Kessenbrock, K.; Krumbholz, M.; Schonermarck, U.; Back, W.; Gross, W.L.; Werb, Z.; Grone, H.J.; Brinkmann, V.; Jenne, D.E. Netting neutrophils in autoimmune small-vessel vasculitis. Nat. Med. 2009, 15, 623–625. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, A.; Vecchié, A.; Abbate, A.; Montecucco, F. Neutrophil Extracellular Traps and Cardiovascular Diseases: An Update. Cells 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Medina, P.; Navarro, S.; Estellés, A.; Vayá, A.; Bertina, R.M.; España, F. Influence of the 4600A/G and 4678G/C polymorphisms in the endothelial protein C receptor (EPCR) gene on the risk of venous thromboembolism in carriers of factor V Leiden. Thromb. Haemost. 2005, 94, 389–394. [Google Scholar] [CrossRef] [PubMed]

| Clinical Characteristics | Patients | Controls | ||
|---|---|---|---|---|
| Lower Extremity VTE | SVT | Total | ||
| N (% of total) | 192 (68.6) | 61 (21.8) | 253 | 249 |
| Age, y | 45 (36, 55) | 58 (46, 66) | 47 (37, 60) | 41 (32, 55) |
| Age at first onset, y | 42 (33, 51) | 53 (38, 61) | 42 (34, 53) | ---- |
| Male sex, N (%) | 106 (55.2) | 47 (77.0) | 153 (60.5) | 117 (47.0) |
| Recurrent thrombosis, N (%) | 57 (29.7) | 6 (9.8) | 63 (24.9) | ---- |
| Familial thrombosis, N (%) | 69 (35.9) | 20 (32.8) | 89 (35.2) | --- |
| Spontaneous thrombosis, N (%) | 48 (25.0) | 3 (4.9) | 51 (20.2) | ---- |
| Leukocytes, × 109/L | 6.4 (5.3, 7.4) | 4.6 (3.6, 5.5) | 5.9 (4.8, 7.1) | 6.4 (5.5, 7.9) |
| Neutrophils, × 109/L | 3.5 (2.9, 4.3) | 2.6 (2.1, 3.3) | 3.2 (2.5, 4.1) | 3.7 (3.0, 4.6) |
| Monocytes, × 109/L | 0.5 (0.4, 0.6) | 0.5 (0.4, 0.7) | 0.5 (0.4, 0.6) | 0.5 (0.4, 0.6) |
| Eosinophils, × 109/L | 0.15 (0.10, 0.22) | 0.13 (0.08, 0.24) | 0.15 (0.10, 0.23) | 0.18 (0.10, 0.21) |
| Basophils, × 109/L | 0.01 (0.00, 0.03) | 0.01 (0.00, 0.02) | 0.01 (0.00, 0.03) | 0 (0.00, 0.02) |
| Lymphocytes, × 109/L | 1.90 (1.53, 2.34) | 1.12 (0.78, 1.53) | 1.71 (1.23, 2.20) | 2.10 (1.71, 2.60) |
| Platelets, × 109/L | 223 (188, 257) | 97 (67, 153) | 205 (143, 248) | 238 (204, 276) |
| Neutrophil-to- lymphocyte ratio | 1.81 (1.44, 2.35) | 2.26 (1.64, 3.41) | 1.90 (1.46, 2.52) | 1.68 (1.33, 2.24) |
| FV Leiden, N (%) | ||||
| -/- | 166 (86.5) | 59 (96.7) | 225 (88.9) | 244 (98.0) |
| +/- & +/+ | 25 & 1 (13.5) | 2 & 0 (3.3) | 27 & 1 (10.0) | 4 & 1 (2.0) |
| PT g.20210G>A, N (%) | ||||
| GG | 176 (91.7) | 57 (93.4) | 233 (92.1) | 236 (94.8) |
| GA & AA | 16 & 0 (8.9) | 4 & 0 (6.6) | 20 & 0 (7.9) | 13 & 0 (5.2) |
| Genotype | APC (ng/mL) | sEPCR (ng/mL) | sTM (ng/mL) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Patients | n | Controls | M-W Test p | n | Patients | n | Controls | M-W Test p | n | Patients | n | Controls | M-W Test p | |
| All | 98 | 1.05 (0.83–1.23) | 153 | 1.25 (1.08–1.60) | <0.0001 | 94 | 102.0 (84.0–122.0) | 153 | 100.0 (82.0–130.0) | 0.7720 | 53 | 4.16 (3.69–4.73) | 129 | 3.77 (3.33–4.44) | 0.0205 |
| HxHx | 20 | 0.91 (0.68–1.07) | 32 | 1.10 (0.90–1.53) | 0.0229 | 19 | 80.0 (70.0–100.0) | 32 | 87.0 (73.5–106.5) | 0.4770 | 12 | 4.17 (3.62–4.55) | 28 | 3.92 (3.50–4.70) | 0.7455 |
| HxH1 | 40 | 1.09 (0.95–1.23) | 60 | 1.26 (1.15–1.52) | <0.0001 | 38 | 94.5 (85.5–112.0) | 60 | 96.0 (81.0–112.5) | 0.8354 | 25 | 4.08 (3.37–4.42) | 52 | 3.76 (3.23–4.41) | 0.2534 |
| H1H1 | 20 | 1.23 (1.15–1.44) | 32 | 1.31 (1.11–1.72) | 0.5472 | 20 | 100.5 (86.5–116.0) | 32 | 90.5 (80.5–111.5) | 0.4806 | 8 | 4.29 (3.95–4.84) | 26 | 3.72 3.07–4.34) | 0.0490 |
| K-W test p | 0.0004 | 0.0482 | 0.0171 | 0.2222 | 0.5129 | 0.4800 | |||||||||
| H1H3 | 7 | 0.97 (0.89–1.03) | 20 | 1.29 (1.10–1.72) | 0.0026 | 6 | 260.0 (234.0–294.0) | 20 | 251.5 (208.5–281.0) | 0.5838 | 5 | 5.90 (3.59–6.25) | 17 | 3.84 (3.43–4.54) | 0.1367 |
| HxHx | 20 | 0.91 (0.68–1.07) | 32 | 1.10 (0.90–1.53) | 0.0229 | 19 | 80.0 (70.0–100.0) | 32 | 87.0 (73.5–106.5) | 0.4770 | 12 | 4.17 (3.62–4.55) | 28 | 3.92 (3.49–4.70) | 0.7455 |
| HxH3 | 7 | 0.79 (0.77–0.88) | 9 | 1.30 (1.09–1.83) | 0.0002 | 7 | 261.0 (234.0–297.0) | 9 | 215.0 (205.0–307.5) | 0.6806 | 1 | 3.73 | 6 | 3.74 (3.47–4.23) | -- |
| H3H3 | 4 | 0.45 (0.20–0.75) | 0 | -- | -- | 4 | 498.5 (488.5–543.0) | 0 | -- | -- | 2 | 5.16 | 0 | -- | -- |
| K-W test p | 0.0008 | -- | <0.0001 | -- | 0.3450 | -- | |||||||||
| c.1418CC | 45 | 1.03 (0.83–1.28) | 99 | 1.24 (1.07–1.60) | 0.0002 | 43 | 101.0 (80.0–122.0) | 99 | 101.0 (82.0–160.0) | 0.3928 | 43 | 4.27 (3.85–4.82) | 87 | 3.82 (3.51–4.50) | 0.0495 |
| c.1418CT | 10 | 1.05 (0.95–1.37) | 43 | 1.29 (1.09–1.62) | 0.0673 | 9 | 109.0 (93.0–175.5) | 43 | 100.0 (82.0–115.0) | 0.2818 | 8 | 4.00 (3.44–4.27) | 33 | 3.80 (3.32–4.48) | 0.8051 |
| c.1418TT | 1 | 1.23 | 10 | 1.29 (1.16–1.66) | -- | 1 | 81.0 | 10 | 92.5 (68.0–154.0) | -- | 1 | 2.47 | 9 | 3.00 (2.60–3.45) | -- |
| K-W test p | 0.6866 | 0.7600 | 0.4530 | 0.5174 | 0.1338 | 0.0032 | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martos, L.; Oto, J.; Fernández-Pardo, Á.; Plana, E.; Solmoirago, M.J.; Cana, F.; Hervás, D.; Bonanad, S.; Ferrando, F.; España, F.; et al. Increase of Neutrophil Activation Markers in Venous Thrombosis—Contribution of Circulating Activated Protein C. Int. J. Mol. Sci. 2020, 21, 5651. https://doi.org/10.3390/ijms21165651
Martos L, Oto J, Fernández-Pardo Á, Plana E, Solmoirago MJ, Cana F, Hervás D, Bonanad S, Ferrando F, España F, et al. Increase of Neutrophil Activation Markers in Venous Thrombosis—Contribution of Circulating Activated Protein C. International Journal of Molecular Sciences. 2020; 21(16):5651. https://doi.org/10.3390/ijms21165651
Chicago/Turabian StyleMartos, Laura, Julia Oto, Álvaro Fernández-Pardo, Emma Plana, María José Solmoirago, Fernando Cana, David Hervás, Santiago Bonanad, Fernando Ferrando, Francisco España, and et al. 2020. "Increase of Neutrophil Activation Markers in Venous Thrombosis—Contribution of Circulating Activated Protein C" International Journal of Molecular Sciences 21, no. 16: 5651. https://doi.org/10.3390/ijms21165651
APA StyleMartos, L., Oto, J., Fernández-Pardo, Á., Plana, E., Solmoirago, M. J., Cana, F., Hervás, D., Bonanad, S., Ferrando, F., España, F., Navarro, S., & Medina, P. (2020). Increase of Neutrophil Activation Markers in Venous Thrombosis—Contribution of Circulating Activated Protein C. International Journal of Molecular Sciences, 21(16), 5651. https://doi.org/10.3390/ijms21165651





