Exercise-Induced Elevated BDNF Level Does Not Prevent Cognitive Impairment Due to Acute Exposure to Moderate Hypoxia in Well-Trained Athletes
Abstract
1. Introduction
2. Results
2.1. Maximal Workload and Respiratory Variables
2.2. Choice Reaction Time and Number of Correct Reactions
2.3. Brain-Derived Neurotrophic Factor and Selected Biochemical Variables
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Ergocycle Graded Exercise Test
4.3. Psychomotor Performance Determination
4.4. Venous Blood
4.5. Determination of Brain-Derived Neurotrophic Factor, Cortisol and Endothelin-1 Concentrations
4.6. Determination of Catecholamines by HPLC Method
4.7. Determination of Nitrite Concentration
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BDNF | brain-derived neurotrophic factor |
| ET-1 | endothelin-1 |
| 5-HT | serotonin |
| CRT | choice reaction time |
| NCR | number of correct reactions |
| DA | dopamine |
| LA | lactate |
| NA | noradrenaline |
| C | cortisol |
| A | adrenalin |
References
- Bendahan, D.; Badier, M.; Jammes, Y.; Confort-Gouny, S.; Salvan, A.M.; Guilot, C.; Conzzone, P. Metabolic and myoelectrical effects of acute hypoxaemia during isometric contraction of forearm muscles in humans: A combined 31P-magnetic resonance spectroscopy-surface electromyogram (MRS-SEMG) study. Clin. Sci. 1998, 94, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Calbet, J.A.; De Paz, J.A.; Garatachea, N.; Cabeza de Vaca, S.; Chavarren, J. Anaerobic energy provision does not limit Wingate exercise performance in endurance-trained cyclists. J. Appl. Physiol. 2003, 94, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Perrey, S. Decrease in cerebral oxygenation influences central motor output in humans. Acta Physiol. 2009, 196, 279–281. [Google Scholar] [CrossRef] [PubMed]
- Amann, M.; Eldridge, M.W.; Lovering, A.T.; Stickland, M.K.; Pegelow, D.F.; Dempsey, J.A. Arterial oxygenation influences central motor output and exercise performance via effects on peripheral locomotor muscle fatigue in humans. J. Physiol. 2006, 575, 937–952. [Google Scholar] [CrossRef]
- Peltonen, J.E.; Rusko, H.K.; Rantamaki, J.; Sweins, K.; Nittymaki, S.; Vitasalo, J.T. Effects of oxygen fraction in inspired air on force production and electromyogram activity during ergometer rowing. Eur. J. Appl. Physiol. 1997, 76, 495–503. [Google Scholar] [CrossRef]
- Dempsey, J.A.; Wagner, P.D. Exercise-induced arterial hypoxemia. J. Appl. Physiol. 1999, 87, 1997–2006. [Google Scholar] [CrossRef]
- Adams, R.P.; Welch, H.G. Oxygen uptake, acid-base status, and performance with varied inspired oxygen fractions. J. Appl. Physiol. 1980, 49, 863–868. [Google Scholar] [CrossRef]
- Hogan, M.C.; Richardson, R.S.; Haseler, L.J. Human muscle performance and PCr hydrolysis with varied inspired oxygen fraction: A 31P-MRS study. J. Appl. Physiol. 1999, 86, 1367–1373. [Google Scholar] [CrossRef]
- Peltonen, J.E.; Tikkanen, H.O.; Ritola, J.J.; Ahotupa, M.; Rusko, H.K. Oxygen uptake response during maximal cycling in hyperoxia, normoxia and hypoxia. Aviat. Space Environ. Med. 2001, 72, 904–911. [Google Scholar]
- Benoit, H.; Busso, T.; Castells, J.; Geyssant, A.; Denis, C. Decrease in peak heart rate with acute hypoxia in relation to sea level VO(2max). Eur. J. Appl. Physiol. 2003, 90, 514–519. [Google Scholar] [CrossRef]
- Lundby, C.; Moeller, P.; Kanstrup, I.L.; Olsen, N.V. Heart rate response to hypoxic exercise: Role of dopamine D2-receptors and effect of oxygen supplementation. Clin. Sci. 2001, 101, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Kayser, B.; Narici, M.; Binzoni, T.; Grassi, B.; Cerretelli, P. Fatigue and exhaustion in chronic hypobaric hypoxia: Influence of exercising muscle mass. Am. J. Physiol. 1994, 76, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Boushel, R.; Calbet, J.A.; Rådegran, G.; Sondergaard, H.; Wagner, P.D.; Saltin, B. Parasympathetic neural activity accounts for the lowering of exercise heart rate at high altitude. Circulation 2001, 104, 1785–1791. [Google Scholar] [CrossRef] [PubMed]
- Amann, M.; Romer, L.M.; Subudhi, A.W.; Pegelow, D.F.; Dempsey, J.A. Severity of arterial hypoxaemia affects the relative contributions of pripheral muscle fatigue to exercise performance in healthy humans. J. Physiol. 2007, 581, 389–403. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Phys. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef]
- Davis, J.M.; Bailey, S.P. Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exerc. 1997, 29, 45–57. [Google Scholar] [CrossRef]
- Lieberman, P.; Protopapas, A.; Reed, E.; Youngs, J.W.; Kankim, B.G. Cognitive defects at altitude. Nature 1994, 372, 325. [Google Scholar] [CrossRef]
- Blogg, S.L.; Gennser, M. Cerebral blood flow velocity and psychomotor performance during acute hypoxia. Aviat. Space Environ. Med. 2006, 77, 107–113. [Google Scholar]
- Temme, L.A.; Still, D.L.; Acromite, M.T. Hypoxia and flight performance of military instructor pilots in a flight simulator. Aviat. Space Environ. Med. 2010, 81, 654–659. [Google Scholar] [CrossRef]
- McMorris, T.; Hale, B.H.; Barwood, M.; Costello, J.; Corbett, J. Effect of acute hypoxia on cognition: A systematic review and meta-regression analysis. Neurosci. A Biobehav. Rev. 2017, 74, 225–232. [Google Scholar] [CrossRef]
- Lal, C.; Strange, C.; Bachman, D. Neurocognitive impairment in obstructive sleep apnea. Chest 2012, 141, 1601–1610. [Google Scholar] [CrossRef] [PubMed]
- Jensen, A.R. Clocking the Mind: Mental Chronometry and Individual Differences, 1st ed.; Elsevier Science: London, UK, 2006; pp. 43–54. [Google Scholar]
- Salthouse, T.A.; Hedden, T. Interpreting reaction time measures in between-group comparisons. J. Clin. Exp. Neuropsychol. 2002, 24, 858–872. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, H.G.; Coombs, G.B.; Tymko, M.M.; Nowak-Fluck, D.; Ainslie, P.N. Severity-dependent influence of isocapnic hypoxia on reaction time is independent of neurovascular coupling. Physiol. Behav. 2018, 1, 262–269. [Google Scholar] [CrossRef] [PubMed]
- De Aquino-Lemos, V.; Santos, R.V.T.; Antunes, H.K.M.; Lira, F.S.; Bittar, I.G.L.; Caris, L.V.; Tufik, S.; de Mello, M.T. Acute physical exercise under hypoxia improves sleep, mood and reaction time. Physiol. Behav. 2016, 154, 90–99. [Google Scholar] [CrossRef]
- Ochi, G.; Kanazawa, J.; Hyodo, K.; Suwabe, K.; Shimizu, T.; Fukuie, T.; Byun, K.; Soya, H. Hypoxia-induced lowered executive function depends on arterial oxygen desaturation. J. Physiol. Sci. 2018, 68, 847–853. [Google Scholar] [CrossRef]
- Pramsohler, S.; Wimmer, S.; Kopp, M.; Gatterer, H.; Faulhaber, M.; Burtscher, M.; Netzer, N.C. Normobaric hypoxia overnight impairs cognitive reaction time. BMC Neurosi. 2017, 18, 43. [Google Scholar] [CrossRef]
- Hartman, R.E.; Lee, J.M.; Zipfel, G.J.; Wozniak, D.F. Characterizing learning deficits and hippocampal neuron loss following transient global cerebral ischemia in rats. Brain Res. 2005, 1043, 48–56. [Google Scholar] [CrossRef]
- Maiti, P.; Singh, S.B.; Muthuraju, S.; Veleri, S.; Ilavazhagen, G. Hypobaric hypoxia damages the hippocampal pyramidal neurons in the rat brain. Brain Res. 2007, 1175, 1–9. [Google Scholar] [CrossRef]
- Hota, S.K.; Barhwal, K.; Singh, S.B.; Ilavazhagan, G. Chronic hypobaric hypoxia induced apoptosis in CA1 region of hippocampus: A possible role of NMDAR mediated p75NTR upregulation. Exp. Neurol. 2008, 212, 5–13. [Google Scholar] [CrossRef]
- Gale, S.D.; Hopkins, R.O. Effects of hypoxia on the brain: Neuroimaging and neuropsychological findings following carbon monoxide poisoning and obstructive sleep apnea. J. Int. Neuropsychol. Soc. 2004, 10, 60–71. [Google Scholar] [CrossRef]
- Levitt, S.; Gutin, B. Multiple-choice reaction time and movement time during physical exertion. Res. Q. 1971, 42, 406–410. [Google Scholar] [CrossRef]
- Sjöberg, H. Relations between heart rate, reaction speed and subjective effort at different work loads on a bicycle ergometer. J. Hum. Stress 1975, 1, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Chmura, J.; Nazar, K.; Kaciuba-Uscilko, H. Choice reaction time during graded exercise in relation to blood lactate and plasma catecholamine thresholds. Int. J. Sports Med. 1994, 15, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Chmura, J.; Krysztofiak, H.; Ziemba, A.W.; Nazar, K.; Kaciuba-Uscilko, H. Psychomotor performance during prolonged exercise above and below lactate threshold. Eur. J. Appl. Physiol. 1998, 77, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Travlos, A.K.; Marisi, D.Q. Information processing and concentration as a function of fitness level and exercise-induced activation to exhaustion. Percept. Mot. Skills 1995, 80, 15–26. [Google Scholar] [CrossRef]
- Kruk, B.; Chmura, J.; Krzeminski, K.; Ziemba, A.W.; Nazar, K.; Pekkarinen, H.; Kaciuba-Uscilko, H. Influence of caffeine, cold and exercise on multiple choice reaction time. Psychopharmacology 2001, 157, 197–201. [Google Scholar] [CrossRef]
- Bender, V.L.; McGlynn, L. The effect of various levels of strenuous to exhausting exercise on reaction time. Eur. J. Appl. Physiol. 1976, 35, 95–110. [Google Scholar] [CrossRef]
- Rothman, S.M.; Griffioen, K.J.; Wan, R.; Mattson, M.P. Brain-derived neurotrophic factor as a regulator of systemic and brain energy metabolism and cardiovascular health. Ann. N. Y. Acad. Sci. 2012, 1264, 49–63. [Google Scholar] [CrossRef]
- Huang, T.; Larsen, K.T.; Ried-Larsen, M.; Møller, N.C.; Andersen, L.B. The effects of physical activity and exercise on brain-derived neurotrophic factor in healthy humans: A review. Scand. J. Med. Sci. Sports 2014, 24, 1–10. [Google Scholar] [CrossRef]
- Hohn, A.; Leibrock, J.; Bailey, K.; Barde, Y.A. Identyfication and characterization of a novel member of the nerve growth factor/brain-derived neurotrophic factor family. Nature 1990, 344, 339–341. [Google Scholar] [CrossRef]
- Binder, D.K.; Scharfman, H.E. Brain-derived neurotrophic factor. Growth Factors 2004, 22, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Małczyńska, P.; Piotrowicz, Z.; Drabarek, D.; Langfort, J.; Chalimoniuk, M. Rola mózgowego czynnika neurotroficznego (BDNF) w procesach neurodegeneracji oraz w mechanizmach neuroregeneracji wywołanej wzmożoną aktywnością fizyczną. Postępy Biochem. 2019, 65, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Tyler, W.J.; Alonso, M.; Bramham, C.R.; Pozzo-Miller, L.D. From acquisition to consolidation: On the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learn. Mem. 2002, 9, 224–237. [Google Scholar] [CrossRef]
- Knaepen, K.; Goekint, M.; Heyman, E.M.; Meeusen, R. Neuroplasticity-exercise-induced response of peripheral brain-derived neurotrophic factor: A systematic review of experimental studies in human subjects. Sports Med. 2010, 40, 765–801. [Google Scholar] [CrossRef] [PubMed]
- Zoladz, J.A.; Pilc, A.; Majerczak, J.; Grandys, M.; Zapart-Bukowska, J.; Duda, K. Endurance training increases plasma brain-derived neurotrophic factor concentration in young healthy men. J. Physiol. Pharmacol. 2008, 59, 119–132. [Google Scholar] [PubMed]
- Correia, P.R.; Scorza, F.A.; Gomes da Silva, S.; Pansani, A.; Toscano-Silva, M.; de Almeida, A.C.; Arida, R.M. Increased basal plasma brain-derived neurotrophic factor levels in sprint runners. Neurosci. Bull. 2011, 27, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.; Wang, S.; Cai, J.; Rao, M.S.; Mattson, M.P. Nitric oxide acts in a positive feedback loop with BDNF to regulate neural progenitor cell proliferation and differentiation in the mammalian brain. Dev. Biol. 2003, 258, 319–333. [Google Scholar] [CrossRef]
- Chen, M.J.; Ivy, A.S.; Russo-Neustadt, A.A. Nitric oxide synthesis is required for exercise-induced increases in hippocampal BDNF and phosphatidylinositol 3′ kinase expression. Brain Res. Bull. 2006, 68, 257–268. [Google Scholar] [CrossRef]
- Zajac, A.; Poprzecki, S.; Zebrowska, A.; Chalimoniuk, M.; Langfort, J. Arginine and ornithine supplementation increases growth hormone and insulin-like growth factor-1 serum levels after heavy-resistance exercise in strength-trained athletes. J. Strength Cond. Res. 2010, 24, 1082–1090. [Google Scholar] [CrossRef]
- Dessypris, A.; Wägar, G.; Fyhrquist, F.; Mäkinen, T.; Welin, M.G.; Lamberg, B.A. Marathon run: Effects on blood cortisol-ACTH, iodothyronines-TSH and vasopressin. Acta Endocrinol. (Copenh.) 1980, 95, 151–157. [Google Scholar] [CrossRef]
- Yau, S.Y.; Lau, B.W.; Zhang, E.D.; Lee, J.C.; Li, A.; Lee, T.M.; Ching, Y.P.; Xu, A.M.; So, K.F. Effects of voluntary running on plasma levels of neurotrophins, hippocampal cell proliferation and learning and memory in stressed rats. Neuroscience 2012, 222, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Robinet, C.; Pellerin, L. Brain-derived neurotrophic factor enhances the expression of the monocarboxylate transporter 2 through translational activation in mouse cultured cortical neurons. J. Cereb. Blood Flow Metab. 2010, 30, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.K.S.; Ho, C.S.H.; Tam, W.W.S.; Kua, E.H.; Ho, R.C. Decreased Serum Brain-Derived Neurotrophic Factor (BDNF) Levels in Patients with Alzheimer’s Disease (AD): A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 257. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.Z.; Nusslock, R. Exercise-Mediated Neurogenesis in the Hippocampus via BDNF. Front. Neurosci. 2018, 12, 52. [Google Scholar] [CrossRef]
- Pan, W.; Banks, W.A.; Fasold, M.B.; Bluth, J.; Kastin, A.J. Transport of brain-derived neurotrophic factor across the blood-brain barrier. Neuropharmacology 1998, 37, 1553–1561. [Google Scholar] [CrossRef]
- Ward, R.; Abdul, Y.; Ergul, A. Endothelin-1 inhibition improves the mBDNF/proBDNF ratio in endothelial cells and HT22 neurons under high glucose/palmitate growth conditions. Physiol. Res. 2018, 67 (Suppl. 1), S237–S246. [Google Scholar] [CrossRef]
- Hyman, C.; Hofer, M.; Barde, Y.A.; Juhasz, M.; Yancopoulos, G.D.; Squinto, S.P.; Lindsay, R.M. BDNF is a neurotrophic factor for dopaminergic neurons of the substantia nigra. Nature 1991, 350, 230–232. [Google Scholar] [CrossRef]
- Popova, N.K.; Naumenko, V.S. Neuronal and behavioral plasticity: The role of serotonin and BDNF systems tandem. Expert Opin. Ther. Targets 2019, 23, 227–239. [Google Scholar] [CrossRef]
- Venezia, A.C.; Quinian, E.; Roth, S.M. A single bout of exercise increase hippocampal Bdnf: Influence of chronic exercise and noradrenaline. Genes Brain Behav. 2017, 16, 800–811. [Google Scholar]
- Cooke, M.; Cruttenden, R.; Mellor, A.; Lumb, A.; Pattman, S.; Burnett, A.; Boot, C.; Burnip, L.; Boos, C.; O’Hara, J.; et al. A pilot investigation into the effects of acute normobaric hypoxia, high altitude exposure and exercise on serum angiotensin-converting enzyme, aldosterone and cortisol. J. Renin Angiotensin Aldosterone Syst. 2018, 19, 1470320318782782. [Google Scholar] [CrossRef]
- Schneider, D.A.; McGuiggin, M.E.; Kamimorim, G.H. A comparison of the blood lactate and plasma catecholamine thresholds in untrained male subjects. Int. J. Sports Med. 1992, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Miguel, P.M.; Deniz, B.F.; Deckmann, I.; Confortim, H.D.; Diaz, R.; Diaz, R.; Laureano, D.P.; Silveira, P.P.; Pereira, L.O. Prefrontal Cortex Dysfunction in Hypoxic-Ischaemic Encephalopathy Contributes to Executive Function Impairments in Rats: Potential Contribution for Attention-Deficit/Hyperactivity Disorder. World J. Biol. Psychiatry 2018, 19, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, E.A.; Acworth, I.N.; Blomstrand, E. Amino acids, brain neurotransmitters and a functional link between muscle brain that is important in sustained exercise. In Advances in Biochemistry; Benzi, G., Ed.; John Libby Eurotext: London, UK, 1987; pp. 127–138. [Google Scholar]
- Wang, G.J.; Volkow, N.D.; Fowler, J.S.; Franceschi, D.; Logan, J.; Pappas, N.R.; Wong, C.T.; Netusilm, N. PET studies of the effects of aerobic exercise on human striatal dopamine release. J. Nucl. Med. 2000, 41, 1352–1356. [Google Scholar] [PubMed]
- El-Khodor, B.F.; Boksa, P. Long-term reciprocal changes in dopamine levels in prefrontal cortex versus nucleus accumbens in rats born by Caesarean section compared to vaginal birth. Exp. Neurol. 1997, 145, 118–129. [Google Scholar] [CrossRef]
- Piotrowicz, ż.; Chalimoniuk, M.; Ploszczyca, K.; Czuba, M.; Langfort, J. Acute normobaric hypoxia does not affect the simultaneous exercise-induced increase in circulating BDNF and GDNF in young healthy men: A feasibility study. PLoS ONE 2019, 14, e0224207. [Google Scholar] [CrossRef]
- Wang, H.; Niu, F.; Fan, W.; Shi, J.; Zhang, J.; Li, B. Modulating effects of predconditioning exercise in the expression of ET-1 and BNP via HIF-1a in ischemically injured brain. Metab. Brain Dis. 2019, 34, 1299–1311. [Google Scholar] [CrossRef]
- Cotman, C.W.; Engesser-Cesar, C. Exercise Enhances and Protects Brain Function. Exerc. Sport Sci. Rev. 2002, 30, 75–79. [Google Scholar] [CrossRef]
- Ludyga, S.; Gerber, M.; Brand, S.; Holsboer-Trachsler, E.; Puhse, U. Acute Effects of Moderate Aerobic Exercise on Specific Aspects of Executive Function in Different Age and Fitness Groups: A Meta-Analysis. Psychophysiology 2016, 53, 1611–1626. [Google Scholar] [CrossRef]
- Czuba, M.; Wilk, R.; Karpiński, J.; Chalimoniuk, M.; Zajac, A.; Langfort, J. Intermittent hypoxic training improves anaerobic performance in competitive swimmers when implemented into a direct competition mesocycle. PLoS ONE 2017, 12, e0180380. [Google Scholar] [CrossRef]
- Czuba, M.; Bril, G.; Płoszczyca, K.; Piotrowicz, Z.; Chalimoniuk, M.; Roczniok, R.; Zembron-Lacny, A.; Gerasimuk, D.; Langfort, J. Intermittent hypoxic training at lactate threshold intensity improves aiming performance in well-trained biathletes with little change of cardiovascular variables. Biomed. Res. Int. 2019, 2019, 1287506. [Google Scholar] [CrossRef]
- Millet, G.P.; Roels, B.; Schmitt, L.; Woorons, X.; Richalet, J.P. Combining hypoxic methods for peak performance. Sports Med. 2010, 40, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Dobashi, S.; Horiuchi, M.; Endo, J.; Kiuchi, M.; Koyama, K. Cognitive Function and Cerebral Oxygenation during Prolonged Exercise under Hypoxia in Healthy Young Males. High Alt. Med. Biol. 2016, 17, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, G.; You, H.Y.; Zheng, R.; Gao, Y.Q. Training-dependent cognitive advantage is suppressed at high altitude. Physiol. Behav. 2012, 106, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef]
- Archer, T.; Josefsson, T.; Lindwall, M. Effects of Physical Exercise on Depressive Symptoms and Biomarkers in Depression. CNS Neurol. Disord. Drug Targets 2014, 13, 1640–1653. [Google Scholar] [CrossRef]
- Campos, C.; Rocha, N.B.; Lattari, E.; Nardi, A.E.; Machado, S. Exercise Induced Neuroplasticity to Enhance Therapeutic Outcomes of Cognitive Remediation in Schizophrenia: Analyzing the Role of Brai Nderived Neurotrophic Factor. CNS Neurol. Disord. Drug Targets 2017, 16, 638–651. [Google Scholar] [CrossRef]
- Das, A.K.; Dhar, P.; Sharma, V.K.; Barhwal, K.; Hota, S.K.; Norboo, T.; Singh, S.B. High Altitude With Monotonous Environment Has Significant Impact on Mood and Cognitive Performance of Acclimatized Lowlanders: Possible Role of Altered Serum BDNF and Plasma Homocysteine Level. J. Affect. Disord. 2018, 237, 94–103. [Google Scholar] [CrossRef]
- Wang, W.H.; He, G.P.; Xiau, P.; Gu, C.; Chen, H.Y. Relationship Between Brain-Derived Neurotrophic Factor and Cognitive Function of Obstructive Sleep Apnea/Hypopnea Syndrome Patients. Asian Pac. J. Trop. Med. 2012, 5, 906–910. [Google Scholar] [CrossRef]
- Halievski, K.; Nath, S.; Katsuno, M.; Adachi, H.; Sobue, G.; Breedlove, S.; Lieberman, A.; Jordan, C. Disease Affects Bdnf Expression in Synaptic and Extrasynaptic Regions of Skeletal Muscle of Three SBMA Mouse Models. Int. J. Mol. Sci. 2019, 20, 1314. [Google Scholar] [CrossRef]
- Satriotomo, I.; Nichols, N.I.; Dale, E.A.; Emery, A.T.; Dahlberg, J.M.; Mitcell, G.S. Repetitive Acute Intermittent Hypoxia Increases Growth/Neurotrophic Factor Expression in Non-Respiratory Motor Neurons. Neuroscience 2016, 332, 449–488. [Google Scholar] [CrossRef]
- Karege, F.; Schwald, M.; Cisse, M. Postnatal developmental profile of brain-derived neurotrophic factor in rat brain and platelets. Neurosci. Lett. 2002, 328, 261–264. [Google Scholar] [CrossRef]
- Rasmussen, P.; Brassard, P.; Adser, H.; Pedersen, M.V.; Leick, L.; Hart, E.; Secher, N.H.; Pedersen, B.K.; Pilegaard, H. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 2009, 94, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Voss, M.W.; Prakas, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M. Exercise Training Increases Size of Hippocampus and Improves Memory. Proc. Natl. Acad. Sci. USA 2011, 108, 3017–3322. [Google Scholar] [CrossRef] [PubMed]
- Lipski, R.H.; Marini, A.M. Neurotrophic factor in neuronal survival and behavior-related plasticity. Ann. N. Y. Acad. Sci. 2007, 1122, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Huang, E.J.; Reichardt, L.E. Neurotrophins: Roles in neuronal development and function. Annu. Rev. Neurosci. 2009, 24, 677–736. [Google Scholar] [CrossRef]
- Ferris, L.T.; Williams, J.S.; Shen, C.L. The Effect of Acute Exercise on Serum Brain-Derived Neurotrophic Factor Levels and Cognitive Function. Med. Sci. Sports Exerc. 2007, 39, 728–734. [Google Scholar] [CrossRef]
- Tsai, C.L.; Chen, F.C.; Pan, C.Y.; Wang, C.H.; Huang, T.H.; Chen, T.C. Impact of Acute Aerobic Exercise and Cardiorespiratory Fitness on Visuospatial Attention Performance and Serum BDNF Levels. Psychoneuroedocrinology 2014, 41, 121–131. [Google Scholar] [CrossRef]
- Slusher, A.L.; Patterson, V.T.; Schwartz, C.S.; Acevedo, E.O. Impact of high intensity exercise on execrative function and brain derived neurotrophic factor in healthy college aged males. Phsiol. Behav. 2018, 191, 116–122. [Google Scholar] [CrossRef]
- Sasi, M.; Vignoli, B.; Cannossa, M.; Blum, R. Neurobiology of Local and Intercellular BDNF Signaling. Plugers Arch. Eur. J. Physiol. 2017, 469, 593–610. [Google Scholar] [CrossRef]
- Fowler, C.; Gervitz, L.; Hamilton, E.; Walker, J.A. Systemic Hypoxia and the Depression of Synaptic Transmission in Rat Hippocampus after Carotid Artery Occlusion. J. Physiol. 2003, 550, 961–972. [Google Scholar] [CrossRef]
- Lanigan, S.; Corcoran, A.E.; Wall, A.; Mukandala, G.; O’Connor, J.J. Acute Hypoxic Exposure and Prolyl-Hydroxylase Inhibition Improves Synaptic Transmission Recovery Time from a Subsequent Hypoxic Insult in Rat Hippocampus. Brain Res. 2018, 1701, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Kline, D.D.; Ramirez-Navarro, A.; Kunze, D.L. Adaptive Depression in Synaptic Transmission in the Nucleus of the Solitary Tract after In Vivo Chronic Intermittent Hypoxia: Evidence for Homeostatic Plasticity. J. Neurosci. 2007, 27, 4663–4673. [Google Scholar] [CrossRef] [PubMed]
- Jonas, E.; Hickman, J.A.; Hardwick, J.M.; Kaczmarek, L.K. Exposure to Hypoxia Rapidly Induces Mitochondrial Channel Activity within a Living Synapse. J. Biol. Chem. 2005, 280, 4491–4497. [Google Scholar] [CrossRef] [PubMed]
- Muthuraju, S.; Maiti, P.; Solanki, P.; Sharma, A.K.; Amitabh; Singh, S.B.; Prasad, D.; Ilavazhagan, G. Acetylcholinesterase Inhibitors Enhance Cognitive Functions in Rats Following Hypobaric Hypoxia. Behav. Brain Res. 2009, 203, 1–14. [Google Scholar] [CrossRef]
- Echouffo-Tcheugui, J.B.; Conner, S.C.; Himali, J.J.; Maillard, P.; DeCarli, C.S.; Beiser, A.S.; Vasan, R.S.; Seshadri, S. Circulating Cortisol and Cognitive and Structural Brain Measures: The Framingham Heart Study. Neurology 2018, 91, e1961–e1970. [Google Scholar] [CrossRef]
- Sroykham, W.; Wongsawat, Y. Effects of Brain Activity, Morning Salivary Cortisol, and Emotion Regulation on Cognitive Impairment in Elderly People. Medicine 2019, 98, e16114. [Google Scholar] [CrossRef]
- Stomby, A.; Boraxbekk, C.J.; Lundquist, A.; Nordin, A.; Nilsson, L.G.; Adolfsson, R.; Nyberg, L.; Olsson, T. Higher Diurnal Salivary Cortisol Levels Are Related to Smaller Prefrontal Cortex Surface Area in Elderly Men and Women. Eur. J. Endocrinol. 2016, 175, 117–126. [Google Scholar] [CrossRef]
- Dominguez, G.; Henkous, N.; Prevot, T.; David, V.; Guillou, J.L.; Belzung, C.; Mons, N.; Béracochéa, D. Sustained Corticosterone Rise in the Prefrontal Cortex Is a Key Factor for Chronic Stress-Induced Working Memory Deficits in Mice. Neurobiol. Stress 2019, 10, 100161. [Google Scholar] [CrossRef]
- Vuolteenaho, O.; Koistinen, P.; Martikkala, V.; Takala, T. Leppaluoto. Effect of physical exercise in hypobaric conditions o atrial natriuretic peptide secretion. Am. J. Physiol. 1992, 263, 647–652. [Google Scholar]
- Rikki, R.E.; Edwards, M.E. Effects of a three-year exercise program on motor function and cognitive processing speed I older woe. Res. Qart. Exerc. Sport 1991, 62, 61–67. [Google Scholar]
- Bouissou, P.; Peronnet, F.; Brisson, G.; Helie, R.; Ledoux, M. Metabolic and endocrine responses to graded exercise under acute hypoxia. Eur. J. Appl. Physiol. 1986, 55, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Bouissou, P.; Peronnet, F.; Brisson, G.; Helie, R.; Ledoux, M. Fluid-electrolyte shift and renin-aldosterone responses to exercise under hypoxia. Horm. Met. Res. 1987, 19, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Rostrup, M. Catecholamines, hypoxia and high altitude. Acta Phsiol. Scand. 1988, 11, 389–399. [Google Scholar] [CrossRef]
- Weber, D.A.; Reynolds, C.R. Clinical Perspectives on Neurobiological Effects of Psychological Trauma. Neuropsychol. Rev. 2004, 14, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Reader, T.A. Distribution of Catecholamines and Serotonin in the Rat Cerebral Cortex: Absolute Levels and Relative Proportions. J. Neural Transm. 1981, 50, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Miguel, P.M.; Pereira, L.O.; Barth, B.; Filho, E.J.M.; Pokhvisneva, I.; Nguyen, T.T.T.; Garg, E.; Razzolini, B.R.; Koh, D.X.P. Prefrontal Cortex Dopamine Transporter Gene Network Moderates the Effect of Perinatal Hypoxic-Ischemic Conditions on Cognitive Flexibility and Brain Gray Matter Density in Children. Biol. Psychiatry 2019, 86, 621–630. [Google Scholar] [CrossRef]
- Ogh, S.; Tsukamoto, H.; Hirasawa, A.; Hasegawa, H.; Hirose, N.; Hashimoto, T. The Effect of Changes in Cerebral Blood Flow on Cognitive Function during Exercise. Physiol. Rep. 2014, 2, e12163. [Google Scholar] [CrossRef]
- Adachi, M.; Yang, Y.; Furiuchi, Y.; Miiyamoto, C. Cloning and characterization of cDNA encoding human A-type endothelin receptor. Biochem. Biophys. Res. Commun. 1991, 180, 1265–1272. [Google Scholar] [CrossRef]
- Palmer, R.M.; Ferrige, A.G.; Moncada, S. Nitric Oxide Release Accounts for the Biological Activity of Endothelium-Derived Relaxing Factor. Nature 1987, 327, 524–526. [Google Scholar] [CrossRef]
- Garthwaite, J. Nitric oxide as a multimodal transmitter in the brain: Discovery and current status. Br. J. Pharmacol. 2019, 176, 197–211. [Google Scholar] [CrossRef]
- Gilchrist, M.; Winyard, P.G.; Fulford, J.; Anning, C.; Shore, A.C.; Benjamin, N. Dietary nitrate supplementation improves reaction time in type 2 diabetes: Development and application of a novel nitrate-depleted beetroot juice placebo. Nitric Oxide 2014, 19, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Lefferts, W.K.; Hughes, W.E.; White, C.N.; Brutsaert, T.D.; Heffernan, K.S. Effect of acute nitrate supplementation on neurovascular coupling and cognitive performance in hypoxia. Appl. Physiol. Nutr. Metab. 2016, 41, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Shannon, O.M.; Duckworth, L.; Barlow, M.J.; Deighton, K.; Matu, J.; Williams, E.L.; Woods, D.; Xie, L.; Stephan, B.C.M.; Siervo, M.; et al. Effects of Dietary Nitrate Supplementation on Physiological Responses, Cognitive Function, and Exercise Performance at Moderate and Very-High Simulated Altitude. Front. Physiol. 2017, 8, 401. [Google Scholar] [CrossRef] [PubMed]
- Basso, J.C.; Suzuki, W.A. The Effects of Acute Exercise on Mood, Cognition, Neurophysiology, and Neurochemical Pathways: A Review. Brain Plast. 2017, 2, 127–152. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.; Lopez-Ojen, M.; Funcasta-Calderon, R.; Ameneiros-Rodríguez, E.; Donapetry-García, C.; Vila-Altesor, M.; Rodríguez-Seijas, J. Comprehensive review on Lactate Metabolism in Human Health. Mitochondrion 2014, 17, 76–100. [Google Scholar] [CrossRef]
- Pellerin, L.; Pellegri, G.; Bittar, P.G.; Charnay, Y.; Bouras, C.; Martinm, J.L.; Stella, N.; Magistretti, P.J. Evidence supporting the existence of an activity-dependent astrocyte neuron lactate shuttle. Dev. Neurosci. 1998, 20, 291–299. [Google Scholar] [CrossRef]
- Carrard, A.; Elsayed, M.; Margineanu, M.; Boury-Jamot, B.; Fragnière, L.; Meylan, E.M.; Petit, J.M.; Fiumelli, H.; Magistretti, P.J.; Martin, J.L. Peripheral administration of lactate produces antidepressant-like effects. Mol. Psychiatry 2018, 23, 392–399. [Google Scholar] [CrossRef]
- Liu, C.; Wu, J.; Zhu, J.; Kuei, C.; Yu, J.; Shelton, J.; Sutton, S.W.; Li, X.; Yun, S.J.; Mirzadegan, T. Lactate inhibits lipolysis in fat cells through activation of an orphan G-protein-coupled receptor, GPR81. J. Biol. Chem. 2009, 284, 2811–2822. [Google Scholar] [CrossRef]
- Urbaniak, G.C.; Plous, S. Research Randomizer (Version 4.0) [Computer Software]. 2013. Available online: http://www.randomizer.org/ (accessed on 25 March 2017).
- Kuipers, H.; Verstappen, F.T.J.; Keizer, H.A.; Guerten, P.; van Kranenburg, G. Variability of aerobic performance in the laboratory and its physiological correlates. Int. J. Sports Med. 1985, 6, 197–201. [Google Scholar] [CrossRef]

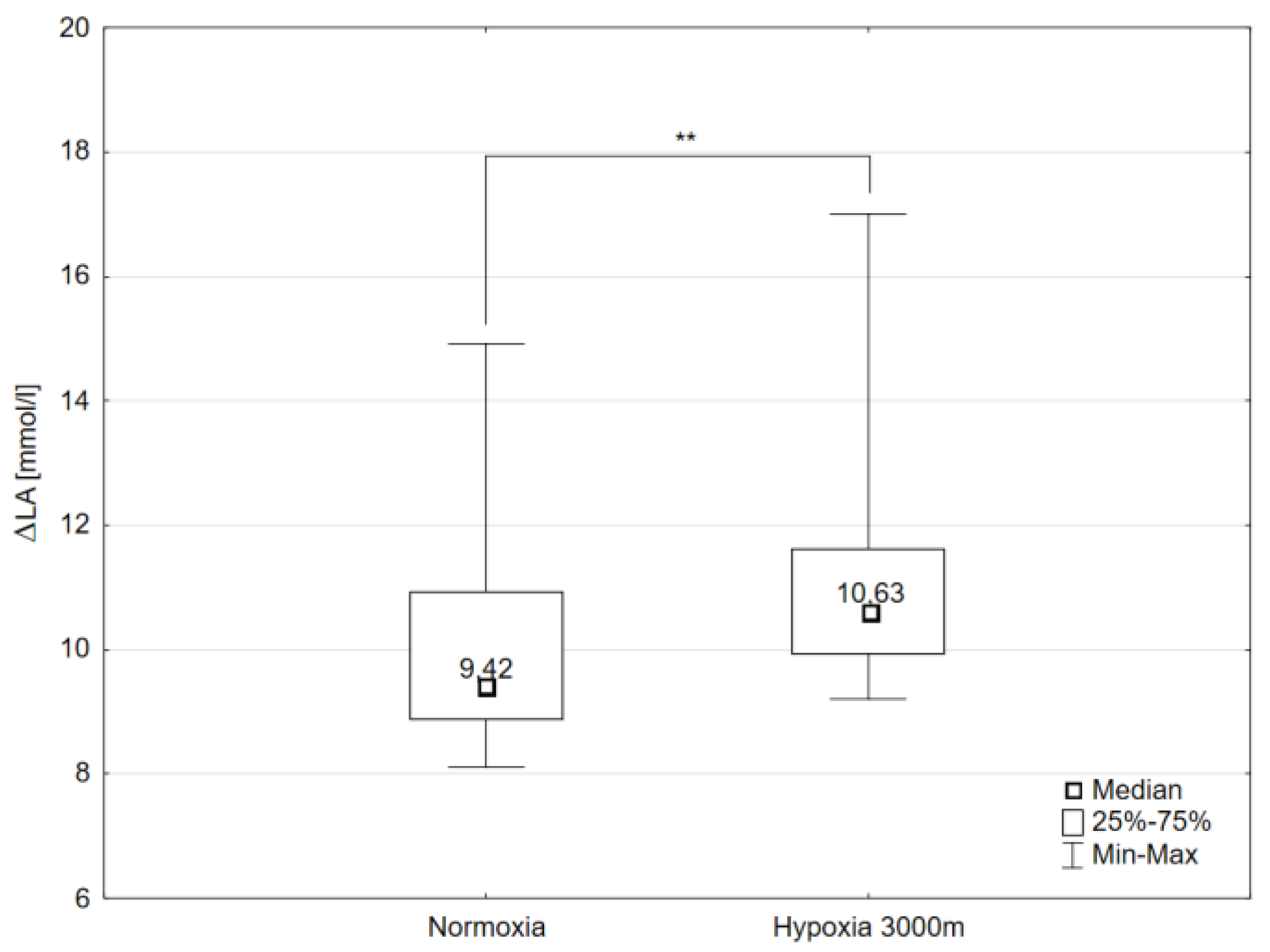


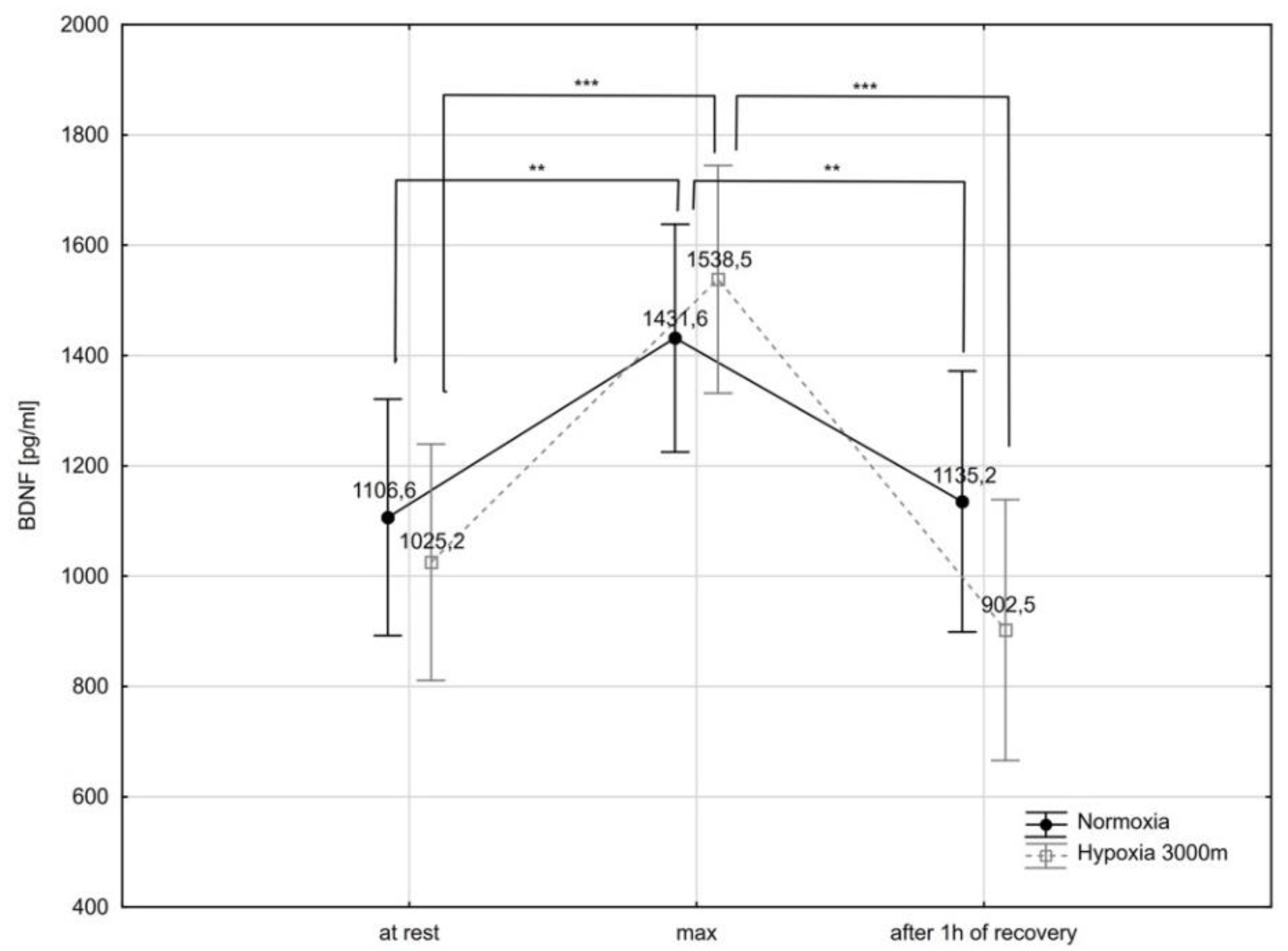
| Variables | Normoxia (N) | Hypoxia 3000 m (H3) | Significance of Differences (* p < 0.05; ** p < 0.01) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| at Rest (1) | Max (2) | after 1 h (3) | at Rest (4) | Max (5) | after 1 h (6) | ||||||||
| x ± SD | Me | x ± SD | Me | x ± SD | Me | x ± SD | Me | x ± SD | Me | x ± SD | Me | ||
| ET-1 (pg/mL) | 2.5 ± 0.8 | 2.5 | 2.4 ± 1.7 | 2.3 | 2.0 ± 1.1 | 1.8 | 2.3 ± 0.8 | 2.5 | 2.3 ± 1.0 | 2.61 | 2.7 ± 1.2 | 2.5 | |
| NO2− (pg/mL) | 28.7 ± 12.1 | 23.2 | 47.6 ± 21.2 | 38.8 | 30.2 ± 12.7 | 25.6 | 33.8 ± 16.3 | 39.1 | 52.3 ± 16.3 | 49.8 | 29.5 ± 9.3 | 26.3 | N: X2 = 13.81; p = 0.001 1–2 *; 2–3 * H3: X2 = 16.9; p = 0.002 4–5 *; 5–6 * |
| C (pg/mL) | 8.7 ± 4.1 | 7.1 | 11.5 ± 3.8 | 11.1 | 12.4 ± 1041 | 9.8 | 7.2 ± 2.7 | 6.7 | 8.2 ± 2.48 | 8.8 | 9.4 ± 5.9 | 7.2 | N vs. H3: 2–5 * (U = 29.0; p = 0.041) |
| NA (pg/L) | 338.9 ± 213.4 | 337.2 | 555.6 ± 25.6 | 575.6 | 406.8 ± 28.4 | 329.3 | 334.6 ± 153.2 | 238.7 | 1109.2 ± 1045.4 | 348.5 | 575.2 ± 359 | 435.2 | |
| A (pg/L) | 53.9 ± 29.4 | 48.8 | 479 ± 358.1 | 369 | 112.5 ± 102.3 | 117.1 | 206.9 ± 70.7 | 228.5 | 1436.1 ± 622.9 | 1384.7 | 827.4 ± 263.7 | 742.7 | N: X2 = 16.9; p = 0.002 1–2 *; 2–3 * H3: X2 = 18.2; p = 0.001 4–5 *; 4–6 * N vs. H3: 1–4 ** (U = 1.0; p = 0.001) 2–5 ** (U = 9.0; p = 0.001) 3–6 ** (U = 0.0; p = 0.001) |
| DA (pg/L) | 7.6 ± 10.9 | 4.3 | 8.9 ± 5.8 | 7.8 | 5.6 ± 1.4 | 5.7 | 7.3 ± 2.5 | 6.7 | 12.5 ± 2.7 | 13.2 | 12.1 ± 5.5 | 13.7 | N: X2 = 10.1; p = 0.006 2–3 * H3: X2 = 10.4; p = 0.005 4–5 *; 4–6 * N vs. H3: 2–5 ** (U = 17.0; p = 0.008) 3–6 ** (U = 16.0; p = 0.006) |
| 5-HT (pg/L) | 155.1 ± 104.2 | 135.4 | 212.6 ± 148.1 | 150.3 | 192.1 ± 90.1 | 190.7 | 136.5 ± 67.1 | 156.7 | 205.9 ± 124.3 | 150.4 | 156.4 ± 82.1 | 148.3 | |
| BH (cm) | BM (kg) | FAT (%) |
|---|---|---|
| 180.5 ± 6.5 | 70.3 ± 6.8 | 9.4 ± 3.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piotrowicz, Z.; Chalimoniuk, M.; Płoszczyca, K.; Czuba, M.; Langfort, J. Exercise-Induced Elevated BDNF Level Does Not Prevent Cognitive Impairment Due to Acute Exposure to Moderate Hypoxia in Well-Trained Athletes. Int. J. Mol. Sci. 2020, 21, 5569. https://doi.org/10.3390/ijms21155569
Piotrowicz Z, Chalimoniuk M, Płoszczyca K, Czuba M, Langfort J. Exercise-Induced Elevated BDNF Level Does Not Prevent Cognitive Impairment Due to Acute Exposure to Moderate Hypoxia in Well-Trained Athletes. International Journal of Molecular Sciences. 2020; 21(15):5569. https://doi.org/10.3390/ijms21155569
Chicago/Turabian StylePiotrowicz, Zofia, Małgorzata Chalimoniuk, Kamila Płoszczyca, Miłosz Czuba, and Józef Langfort. 2020. "Exercise-Induced Elevated BDNF Level Does Not Prevent Cognitive Impairment Due to Acute Exposure to Moderate Hypoxia in Well-Trained Athletes" International Journal of Molecular Sciences 21, no. 15: 5569. https://doi.org/10.3390/ijms21155569
APA StylePiotrowicz, Z., Chalimoniuk, M., Płoszczyca, K., Czuba, M., & Langfort, J. (2020). Exercise-Induced Elevated BDNF Level Does Not Prevent Cognitive Impairment Due to Acute Exposure to Moderate Hypoxia in Well-Trained Athletes. International Journal of Molecular Sciences, 21(15), 5569. https://doi.org/10.3390/ijms21155569





