Noradrenergic Components of Locomotor Recovery Induced by Intraspinal Grafting of the Embryonic Brainstem in Adult Paraplegic Rats
Abstract
1. Introduction
2. Results
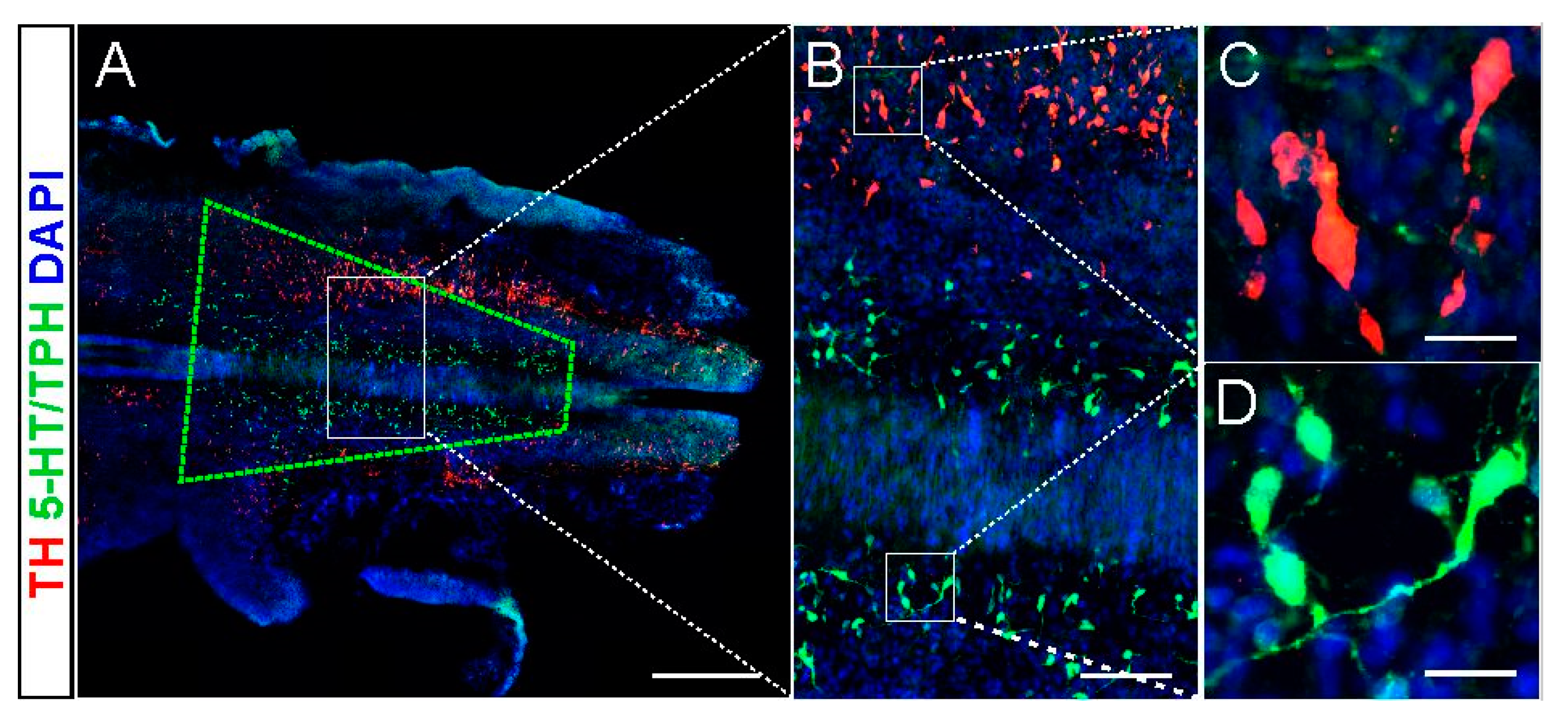
2.1. 5-HT and NA Neurons in the Donor Brainstem
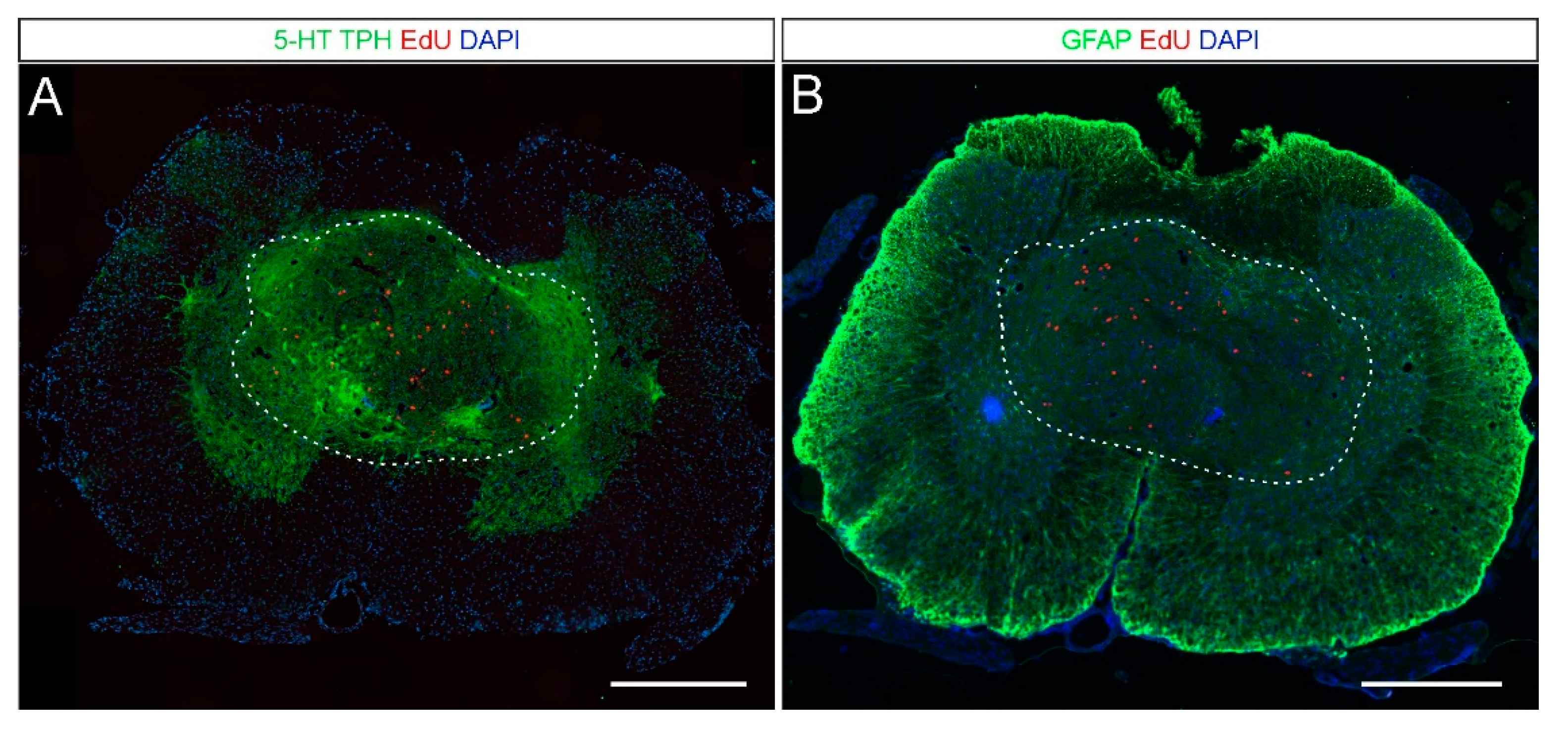
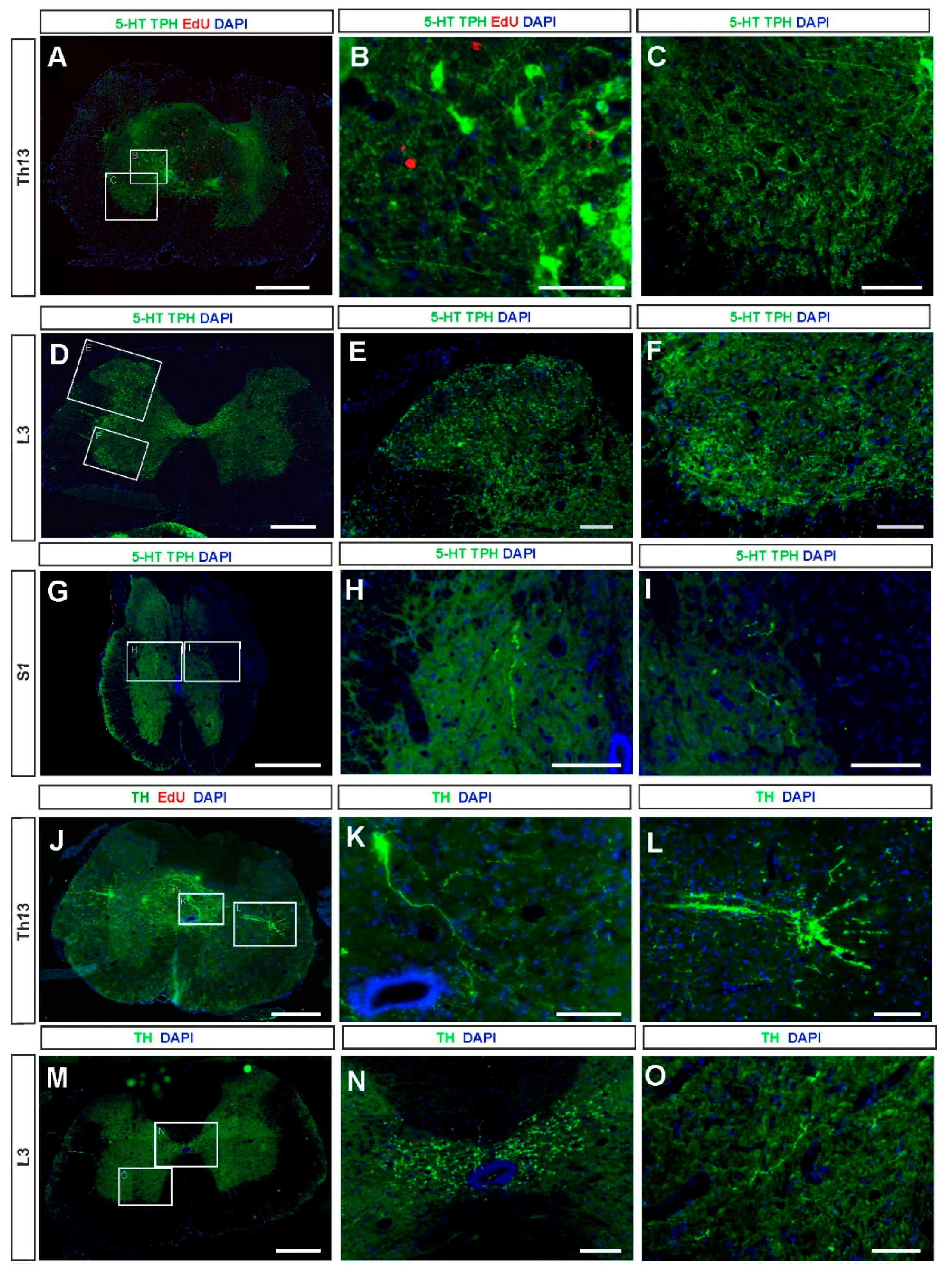
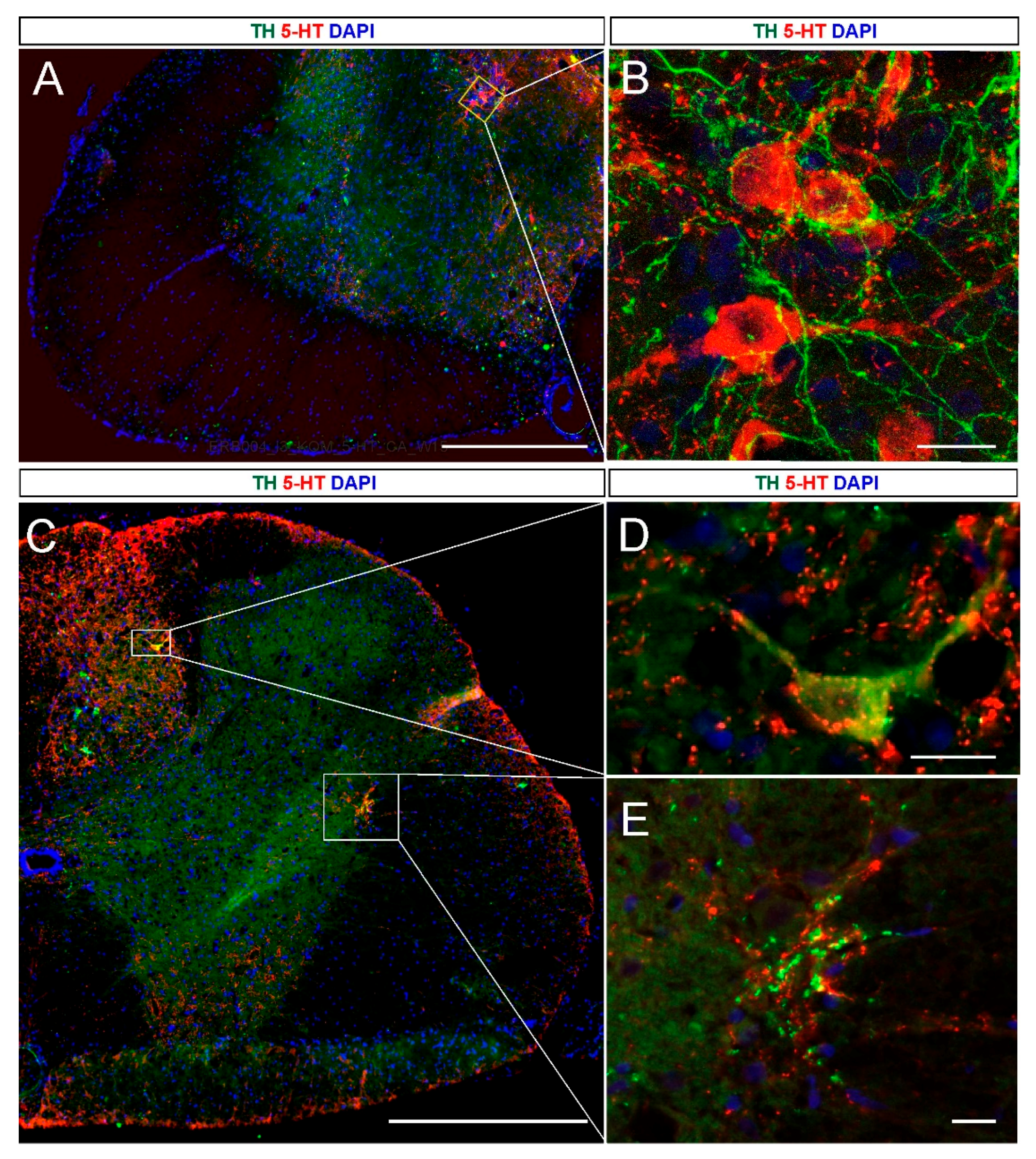
2.2. 5-HT and NA Neurons in the Grafted Spinal Cord
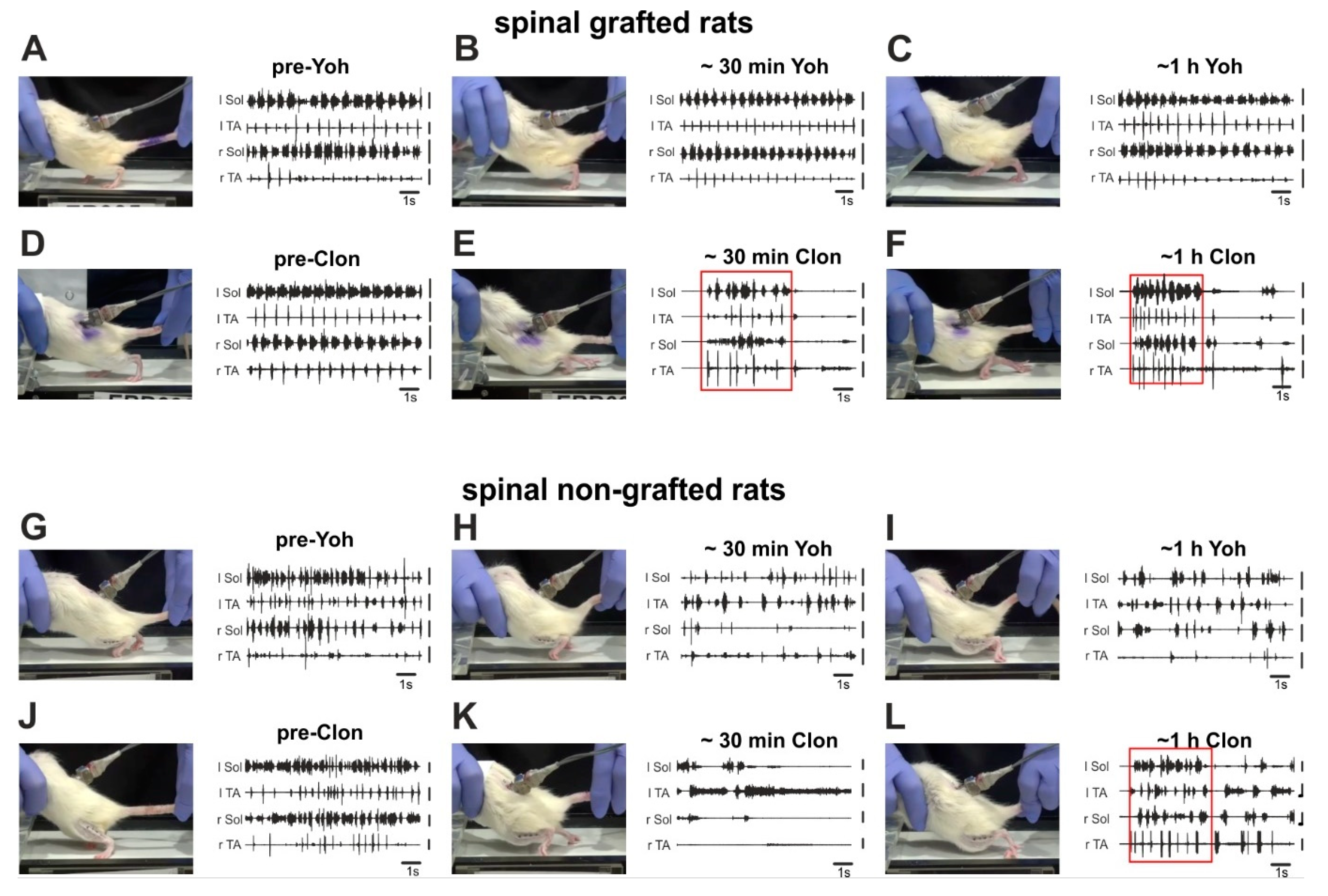
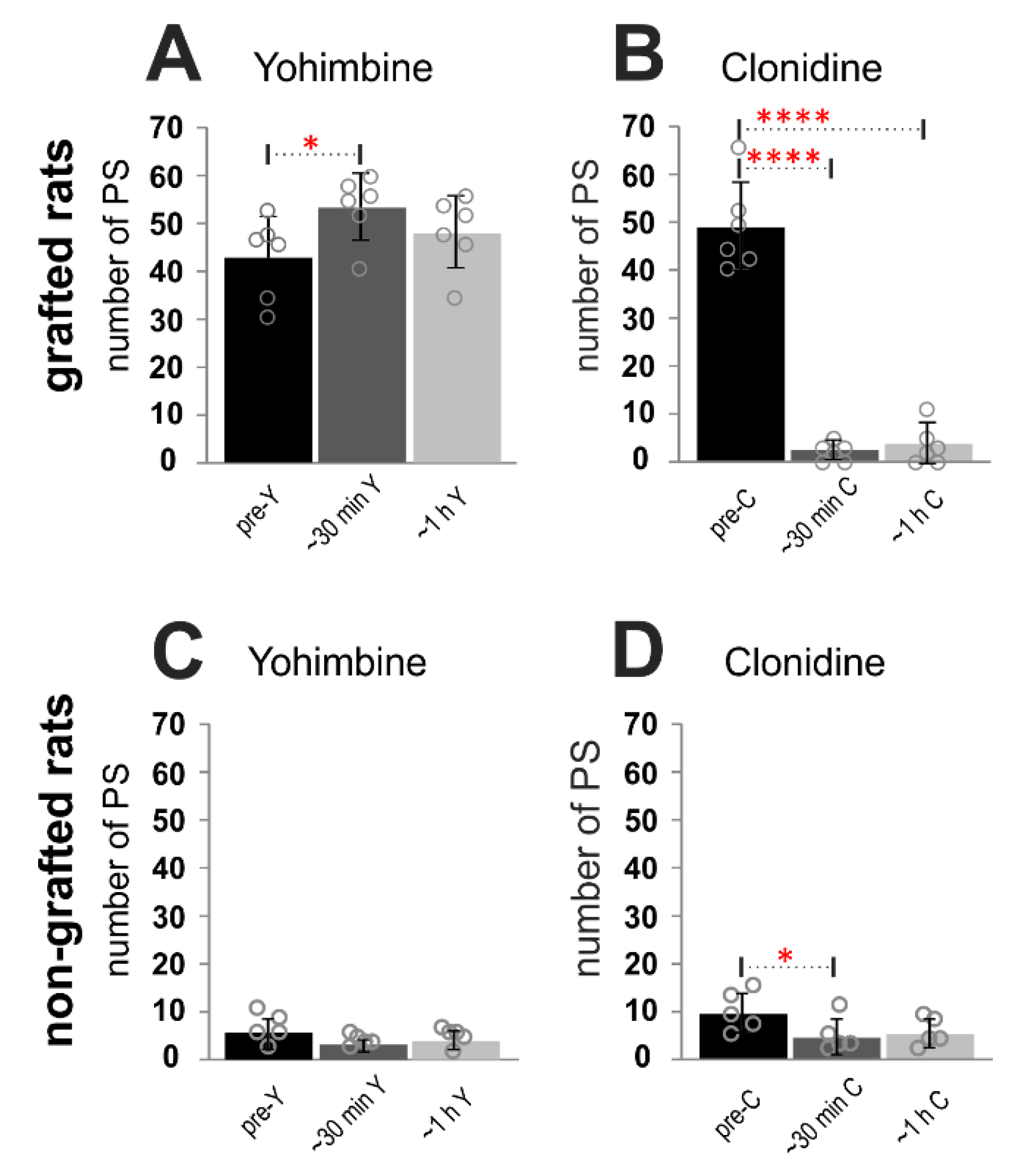
2.3. Adrenergic α2 Receptors in Control of Locomotor Plantar Stepping in Spinal Grafted and non-Grafted Rats—General Considerations
2.4. Adrenergic α2 Receptors in Control of Locomotor Plantar Stepping in Spinal Rats—EMG Activity Analysis
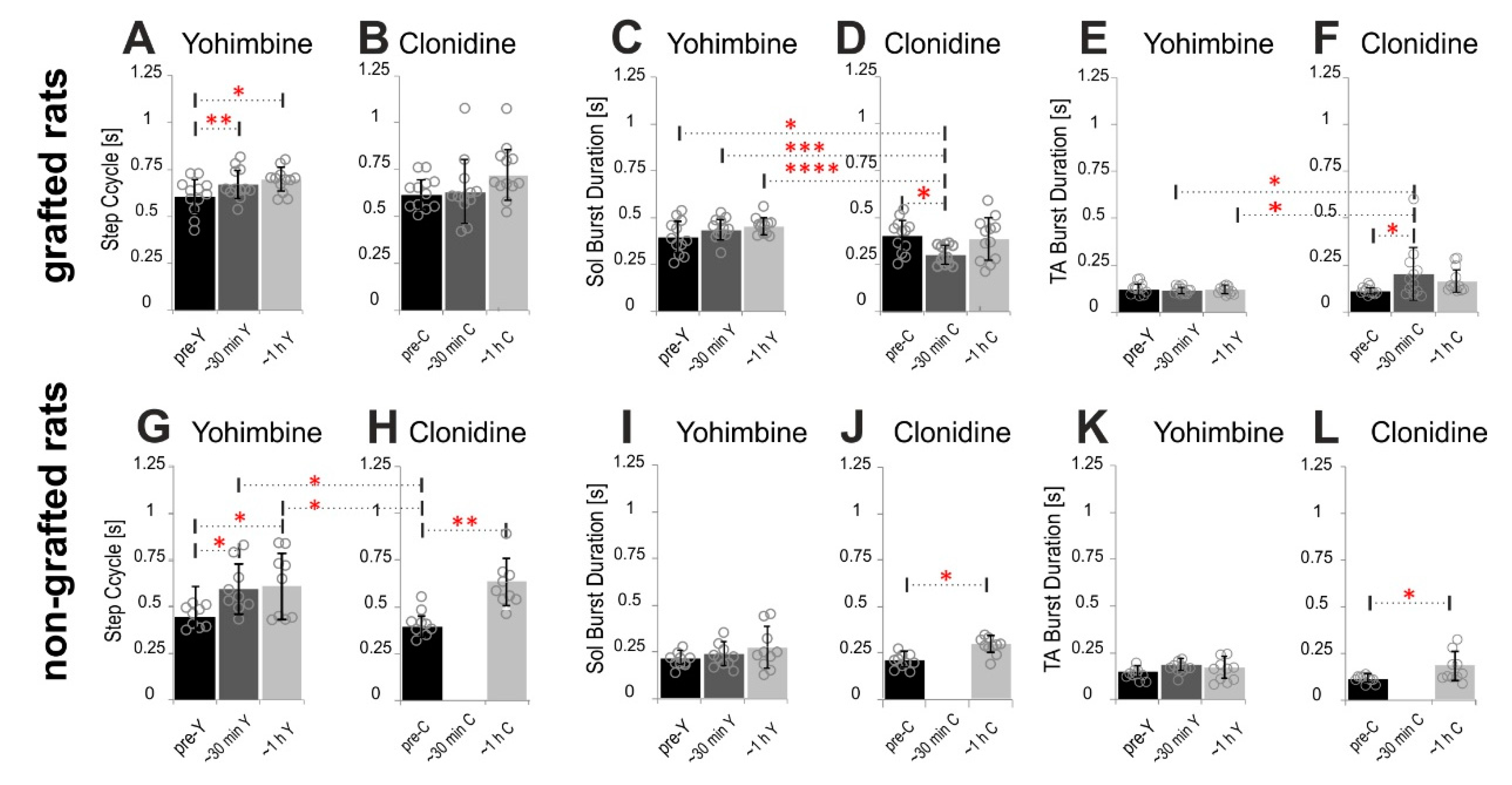
2.4.1. Step Cycle and Burst Durations
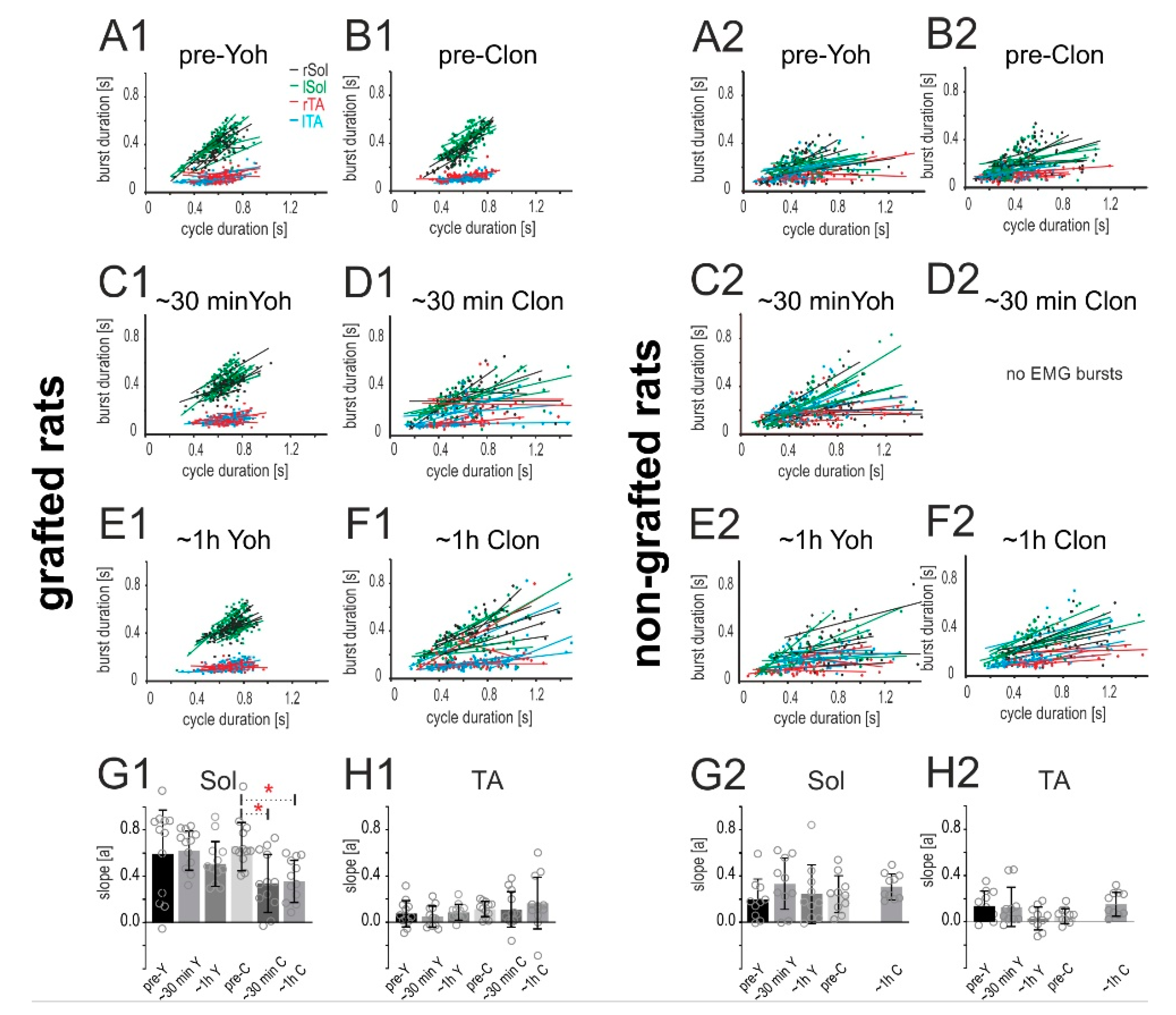
2.4.2. Relationship of the Burst Duration vs. Step Cycle Duration
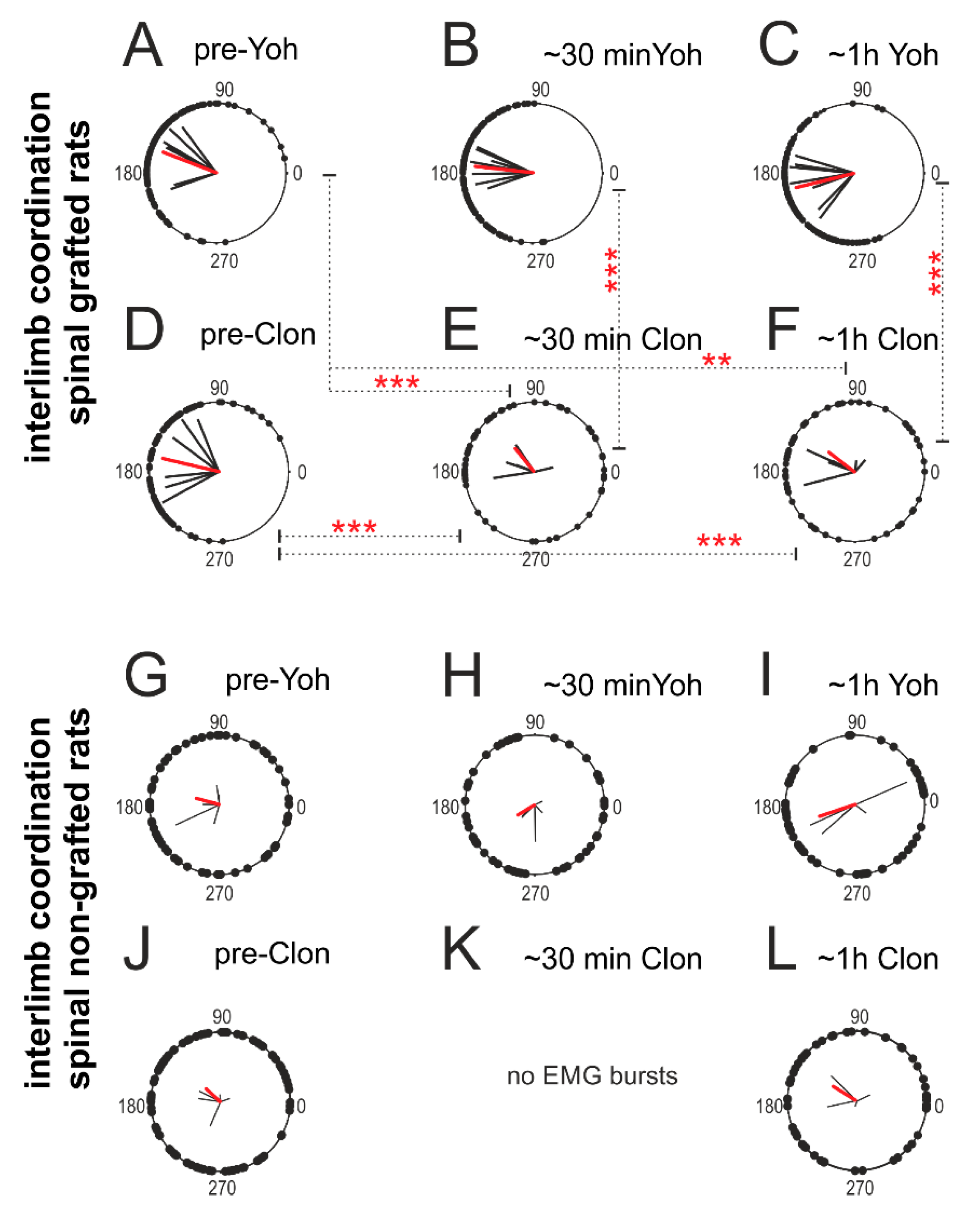
2.4.3. Interlimb Coordination
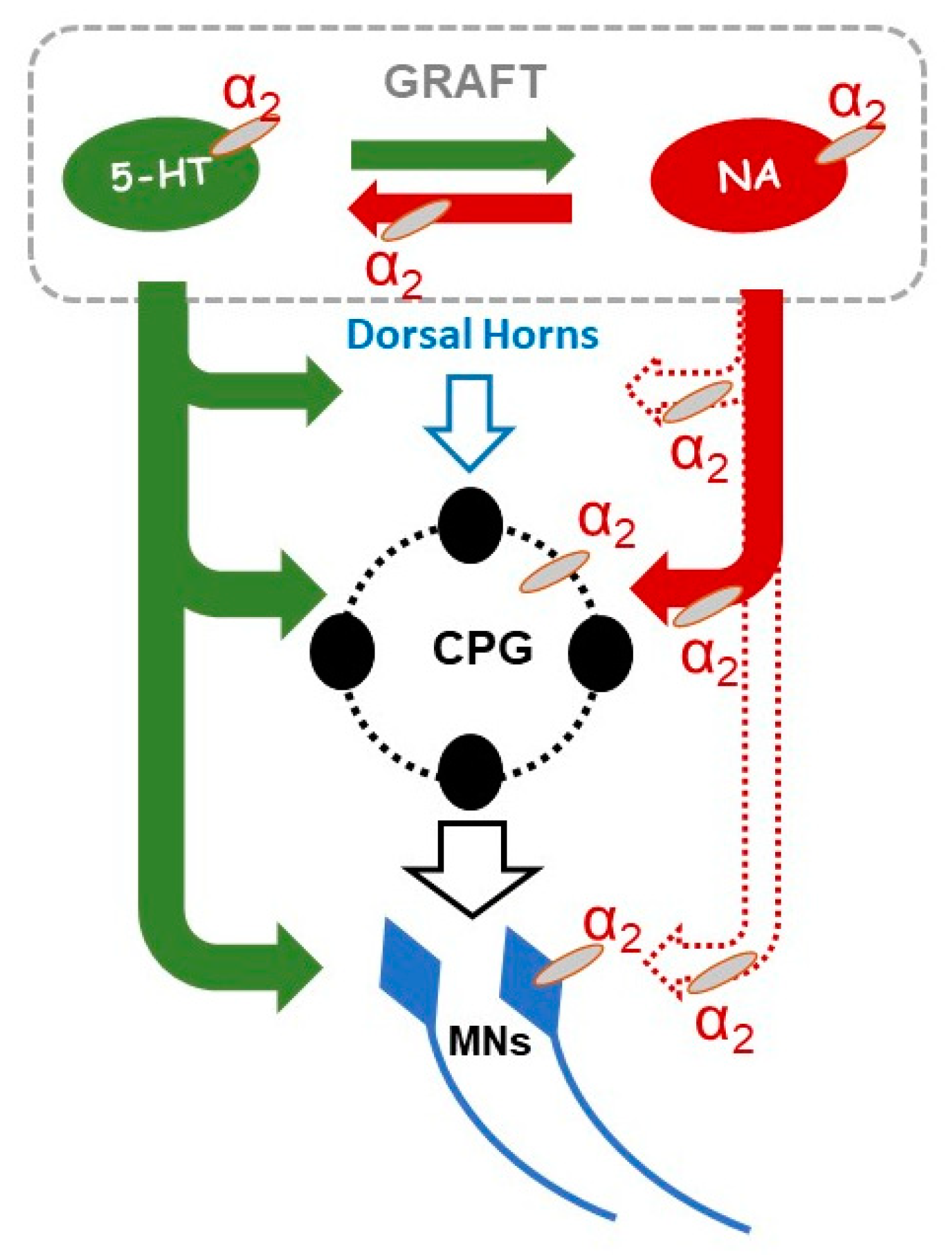
3. Discussion
3.1. Adrenergic α2 Receptors in Motor Control in Normal and in Grafted Rats
3.2. Adrenergic α2 Receptors in Motor Control of Chronic Spinal Rats
4. Material and Methods
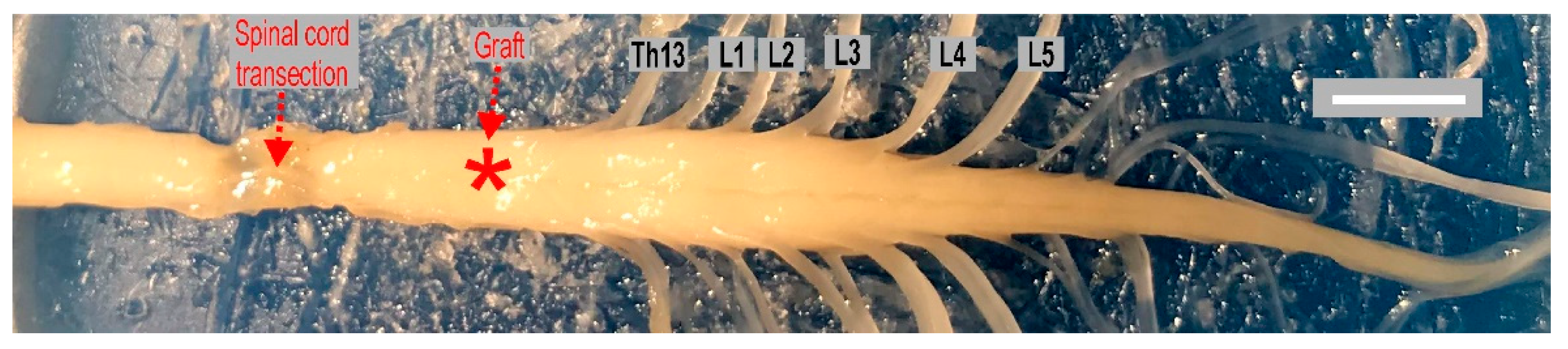
4.1. Complete Spinal Cord Transection
4.2. Intraspinal Grafting of 14-Day Old Rat Embryo Brainstem
4.3. Implantation of EMG Recording Electrodes
4.4. Video and Electromyographic Recordings
4.5. Evaluation of Hindlimb Locomotion
4.6. Drug Tests
4.7. Immunohistochemistry
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jankowska, E.; Jukes, M.G.M.; Lund, S.; Lundberg, A. The effect of Dopa on Spinal Cord. 6. Half-Centre Organization of Interneurones Transmitting Effects from Flexor Reflex Afferents. Acta Physiol. Scand. 1967, 70, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.; Jukes, M.G.M.; Lund, S.; Lundberg, A. The effect of Dopa on Spinal Cord. 5. Reciprocal Organization of Pathways Transmitting Excitatory Action to Alpha Motoneurones of Flexors and Extrnsors. Acta Physiol. Scand. 1967, 70, 369–388. [Google Scholar] [CrossRef] [PubMed]
- Forssberg, H.; Grillner, S. Locomotion of Acute Spinal Cat Injected with Clonidine Iv. Brain Res. 1973, 50, 184–186. [Google Scholar] [CrossRef]
- Barbeau, H.; Rossignol, S. Initiation and modulation of the locomotor pattern in the adult chronic spinal cat by noradrenergic, serotonergic and dopaminergic drugs. Brain Res Bull 1991, 546, 250–260. [Google Scholar] [CrossRef]
- Giroux, N.; Brustein, E.; Chau, C.; Barbeau, H.; Reader, T.A.; Rossignol, S. Differential effects of the noradrenergic agonist clonidine on the locomotion of intact, partially and completely spinalized adult cats. Ann. NY Acad. Sci. 1998, 860, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Barbeau, H.; Chau, C.; Rossignol, S. Noradrenergic Agonists and Locomotor Training Affect Locomotor Recovery after Cord Transection in Adult Cats. Brain Res. Bull. 1993, 30, 387–393. [Google Scholar] [CrossRef]
- Brustein, E.; Rossignol, S. Recovery of locomotion after ventral and ventrolateral spinal lesions in the cat. II. Effects of noradrenergic and serotoninergic drugs. J. Neurophysiol. 1999, 81, 1513–1530. [Google Scholar] [CrossRef]
- Rajaofetra, N.; Konig, N.; Poulat, P.; Marlier, L.; Sandillon, F.; Drian, M.J.; Geffard, M.; Privat, A. Fate of B1-B2 and B3 rhombencephalic cells transplanted into the transected spinal cord of adult rats: light and electron microscopic studies. Experimental neurology 1992, 117, 59–70. [Google Scholar] [CrossRef]
- Ribotta, M.G.Y.; Provencher, J.; Feraboli-Lohnherr, D.; Rossignol, S.; Privat, A.; Orsal, D. Activation of locomotion in adult chronic spinal rats is achieved by transplantation of embryonic raphe cells reinnervating a precise lumbar level. J. Neurosci. 2000, 20, 5144–5152. [Google Scholar] [CrossRef]
- Sławińska, U.; Majczyński, H.; Djavadian, R. Recovery of hindlimb motor functions after spinal cord transection is enhanced by grafts of the embryonic raphe nuclei. Exp. Brain Res. 2000, 132, 27–38. [Google Scholar] [CrossRef]
- Sławińska, U.; Miazga, K.; Cabaj, A.M.; Leszczyńska, A.N.; Majczyński, H.; Nagy, J.I.; Jordan, L.M. Grafting of fetal brainstem 5-HT neurons into the sublesional spinal cord of paraplegic rats restores coordinated hindlimb locomotion. Experimental neurology 2013, 247, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Majczyński, H.; Maleszak, K.; Cabaj, A.; Sławińska, U. Serotonin-related enhancement of recovery of hind limb motor functions in spinal rats after grafting of embryonic raphe nuclei. J. Neurotrauma 2005, 22, 590–604. [Google Scholar] [CrossRef] [PubMed]
- Yakovleff, A.; Robybrami, A.; Guezard, B.; Mansour, H.; Bussel, B.; Privat, A. Locomotion in Rats Transplanted with Noradrenergic Neurons. Brain Res. Bull. 1989, 22, 115–121. [Google Scholar] [CrossRef]
- Roudet, C.; Ribotta, M.G.Y.; Privat, A.; Feuerstein, C.; Savasta, M. Intraspinal Noradrenergic-Rich Implants Reverse the Increase of Alpha(1) Adrenoceptors Densities Caused by Complete Spinal-Cord Transection or Selective Chemical Denervation - a Quantitative Autoradiographic Study. Brain research 1995, 677, 1–12. [Google Scholar] [CrossRef]
- Feraboli-Lohnherr, D.; Orsal, D.; Yakovleff, A.; Ribotta, M.G.Y.; Privat, A. Recovery of locomotor activity in the adult chronic spinal rat after sublesional transplantation of embryonic nervous cells: Specific role of serotonergic neurons. Exp. Brain Res. 1997, 113, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Antri, M.; Mouffle, C.; Orsal, D.; Barthe, J.Y. 5-HT1A receptors are involved in short- and long-term processes responsible for 5-HT-induced locomotor function recovery in chronic spinal rat. Eur. J. Neurosci. 2003, 18, 1963–1972. [Google Scholar] [CrossRef]
- Antri, M.; Barthe, J.Y.; Mouffle, C.; Orsal, D. Long-lasting recovery of locomotor function in chronic spinal rat following chronic combined pharmacological stimulation of serotonergic receptors with 8-OHDPAT and quipazine. Neurosci. Letters 2005, 384, 162–167. [Google Scholar] [CrossRef]
- Sławińska, U.; Miazga, K.; Jordan, L.M. 5-HT(2) and 5-HT(7) receptor agonists facilitate plantar stepping in chronic spinal rats through actions on different populations of spinal neurons. Front. Neural. Circuits 2014, 8, 95. [Google Scholar] [CrossRef]
- Antri, M.; Orsal, D.; Barthe, J.Y. Locomotor recovery in the chronic spinal rat: effects of long-term treatment with a 5-HT2 agonist. Eur J Neurosci 2002, 16, 467–476. [Google Scholar] [CrossRef]
- Konig, N.; Wilkie, M.B.; Lauder, J.M. Tyrosine-Hydroxylase and Serotonin Containing Cells in Embryonic Rat Rhombencephalon - a Whole-Mount Immunocytochemical Study. J. Neurosci. Res. 1988, 20, 212–223. [Google Scholar] [CrossRef]
- Guyenet, P.G.; Stornetta, R.L.; Riley, T.; Norton, F.R.; Rosin, D.L.; Lynch, K.R. Alpha(2a)-Adrenergic Receptors Are Present in Lower Brain-Stem Catecholaminergic and Serotonergic Neurons Innervating Spinal-Cord. Brain research 1994, 638, 285–294. [Google Scholar] [CrossRef]
- Bejrowska, A.; Miazga, K.; Majczyński, H.; Zawadzka, M.; Sławińska, U. The potent role of catecholaminergic innervation in locomotor recovery induced by intraspinal grafting of embryonic brain stem tissue in adult paraplegic rats. Acta Neurobiol. Exp. (Wars) 2019, 79, P8.9. [Google Scholar]
- Ribotta, M.G.Y.; Roudet, C.; Sandillon, F.; Privat, A. Transplantation of embryonic noradrenergic neurons in two models of adult rat spinal cord injury: Ultrastructural immunocytochemical study. Brain research 1996, 707, 245–255. [Google Scholar] [CrossRef]
- Sławińska, U.; Miazga, K.; Jordan, L.M. The role of serotonin in the control of locomotor movements and strategies for restoring locomotion after spinal cord injury. Acta Neurobiol. Exp. (Wars) 2014, 74, 172–187. [Google Scholar]
- Giroux, N.; Reader, T.A.; Rossignol, S. Comparison of the effect of intrathecal administration of clonidine and yohimbine on the locomotion of intact and spinal cats. J. Neurophysiol. 2001, 85, 2516–2536. [Google Scholar] [CrossRef]
- Halbertsma, J.M. The Stride Cycle of the Cat - the Modeling of Locomotion by Computerized Analysis of Automatic Recordings. Acta Physiol. Scand. 1983, 1–75. [Google Scholar]
- Majczyński, H.; Maleszak, K.; Górska, T.; Sławińska, U. Comparison of two methods for quantitative assessment of unrestrained locomotion in the rat. J. Neurosci. Methods 2007, 163, 197–207. [Google Scholar] [CrossRef]
- Frigon, A.; Gossard, J.P. Asymmetric control of cycle period by the spinal locomotor rhythm generator in the adult cat. J. Physiol. 2009, 587, 4617–4628. [Google Scholar] [CrossRef]
- Sławińska, U.; Majczyński, H.; Dai, Y.; Jordan, L.M. The upright posture improves plantar stepping and alters responses to serotonergic drugs in spinal rats. J. Physiol. 2012, 590, 1721–1736. [Google Scholar] [CrossRef]
- Fernandez, P.; Moolman-Smook, J.; Brink, P.; Corfield, V. A gene locus for progressive familial heart block type II (PFHBII) maps to chromosome 1q32.2-q32.3. Hum. Genet. 2005, 118, 133–137. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Wu, Q.; Huang, H.P.; Li, S. Effects of alpha 2A Adrenoceptors on Norepinephrine Secretion from the Locus Coeruleus during Chronic Stress-Induced Depression. Front. Neurosci. 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Dietl, M.; Arluison, M.; Mouchet, P.; Feuerstein, C.; Manier, M.; Thibault, J. Immunohistochemical Demonstration of Catecholaminergic Cell-Bodies in the Spinal-Cord of the Rat—Preliminary Note. Histochemistry 1985, 82, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Takeoka, A.; Kubasak, M.D.; Zhong, H.; Kaplan, J.; Roy, R.R.; Phelps, P.E. Noradrenergic innervation of the rat spinal cord caudal to a complete spinal cord transection: Effects of olfactory ensheathing glia. Experimental neurology 2010, 222, 59–69. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mouchet, P.; Manier, M.; Dietl, M.; Feuerstein, C.; Berod, A.; Arluison, M.; Denoroy, L.; Thibault, J. Immunohistochemical study of catecholaminergic cell bodies in the rat spinal cord. Brain Res Bull 1986, 16, 341–353. [Google Scholar] [CrossRef]
- Svensson, T.H.; Bunney, B.S.; Aghajanian, G.K. Inhibition of both noradrenergic and serotonergic neurons in brain by the alpha-adrenergic agonist clonidine. Brain research 1975, 92, 291–306. [Google Scholar] [CrossRef]
- Rosin, D.L.; Zeng, D.; Stornetta, R.L.; Norton, F.R.; Riley, T.; Okusa, M.D.; Guyenet, P.G.; Lynch, K.R. Immunohistochemical Localization of Alpha-2a-Adrenergic Receptors in Catecholaminergic and Other Brain-Stem Neurons in the Rat. Neuroscience 1993, 56, 139–155. [Google Scholar] [CrossRef]
- Hopwood, S.E.; Stamford, J.A. Noradrenergic median modulation of serotonin release in rat dorsal and raphe nuclei via alpha(1) and alpha(2) adrenoceptors. Neuropharmacol. 2001, 41, 433–442. [Google Scholar] [CrossRef]
- Hopwood, S.E.; Stamford, J.A. Multiple 5-HT1 autoreceptor subtypes govern serotonin release in dorsal and median raphe nuclei. Neuropharmacol. 2001, 40, 508–519. [Google Scholar] [CrossRef]
- Hammar, I.; Stecina, K.; Jankowska, E. Differential modulation by monoamine membrane receptor agonists of reticulospinal input to lamina VIII feline spinal commissural interneurons. Eur. J. Neurosci. 2007, 26, 1205–1212. [Google Scholar] [CrossRef]
- Rank, M.M.; Murray, K.C.; Stephens, M.J.; D’Amico, J.; Gorassini, M.A.; Bennett, D.J. Adrenergic Receptors Modulate Motoneuron Excitability, Sensory Synaptic Transmission and Muscle Spasms After Chronic Spinal Cord Injury. J. Neurophysiol. 2011, 105, 410–422. [Google Scholar] [CrossRef]
- Jankowska, E.; Riddell, J.S.; Skoog, B.; Noga, B.R. Gating of Transmission to Motoneurons by Stimuli Applied in the Locus-Ceruleus and Raphe Nuclei of the Cat. J. Physiol. (London) 1993, 461, 705–722. [Google Scholar] [CrossRef] [PubMed]
- Rekling, J.C.; Funk, G.D.; Bayliss, D.A.; Dong, X.W.; Feldman, J.L. Synaptic control of motoneuronal excitability. Physiol. Rev. 2000, 80, 767–852. [Google Scholar] [CrossRef] [PubMed]
- Giroux, N.; Rossignol, S.; Reader, T.A. Autoradiographic study of alpha(1)- and alpha(2)-noradrenergic and serotonin(1A) receptors in the spinal cord of normal and chronically transected cats. J. Comp. Neurol. 1999, 406, 402–414. [Google Scholar] [CrossRef]
- Roudet, C.; Mouchet, P.; Feuerstein, C.; Savasta, M. Normal-Distribution of Alpha-2-Adrenoceptors in the Rat Spinal-Cord and Its Modification after Noradrenergic Denervation - a Quantitative Autoradiographic Study. J. Neurosci. Res. 1994, 39, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Majczyński, H.; Cabaj, A.; Sławińska, U.; Górska, T. Intrathecal administration of yohimbine impairs locomotion in intact rats. Behav. Brain Res. 2006, 175, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Jordan, L.M.; McCrea, D.A. Analysis of Effects of Para-Methoxy-Phenylethylamine on Spinal-Cord Neurons. Brit J Pharmacol 1976, 57, 191–199. [Google Scholar] [CrossRef]
- Hochman, J.; Garraway, S.M.; Machacek, D.W.; Shay, B.L. 5-HT receptors and the neuromodulatory control of spinal cord function. In Motor Neurobiology of the Spinal Cord; Cope, T.C., Ed.; CRC Press: New York, NY, USA, 2001; pp. 48–87. [Google Scholar]
- Jankowska, E.; Edgley, S.A. Functional subdivision of feline spinal interneurons in reflex pathways from group Ib and II muscle afferents; an update. Eur. J. Neurosci. 2010, 32, 881–893. [Google Scholar] [CrossRef]
- Rossignol, S.; Dubuc, R.J.; Gossard, J.P. Dynamic sensorimotor interactions in locomotion. Physiol. Rev. 2006, 86, 89–154. [Google Scholar] [CrossRef]
- Rossignol, S. Plasticity of connections underlying locomotor recovery after central and/or peripheral lesions in the adult mammals. Philos. Trans. R. Soc. B 2006, 361, 1647–1671. [Google Scholar] [CrossRef]
- McCrea, D.A. Spinal circuitry of sensorimotor control of locomotion. J. Physiol. (London) 2001, 533, 41–50. [Google Scholar] [CrossRef]
- Cabaj, A.M.; Majczyński, H.; Couto, E.; Gardiner, P.F.; Stecina, K.; Sławińska, U.; Jordan, L.M. Serotonin controls initiation of locomotion and afferent modulation of coordination via 5-HT7 receptors in adult rats. J. Physiol. 2017, 595, 301–320. [Google Scholar] [CrossRef] [PubMed]
- Chau, C.; Barbeau, H.; Rossignol, S. Effects of intrathecal alpha(1)- and alpha(2)-noradrenergic agonists and norepinephrine on locomotion in chronic spinal cats. J. Neurophysiol. 1998, 79, 2941–2963. [Google Scholar] [CrossRef] [PubMed]
- Langer, S.Z. Presynaptic Receptors and Their Role in Regulation of Transmitter Release. Brit. J. Pharmacol. 1977, 60, 481–497. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K.C. Catecholamines and their actions in the spinal cord. In Handbook of Spinal Cord; Davidoff, R.A., Ed.; Marcel Dekker: New York, NY, USA, 1983; pp. 275–328. [Google Scholar]
- Kehne, J.H.; Gallager, D.W.; Davis, M. Spinalization Unmasks Clonidines Alpha-1-Adrenergic Mediated Excitation of the Flexor Reflex in Rats. J. Neurosci. 1985, 5, 1583–1590. [Google Scholar] [CrossRef]
- Lapointe, N.P.; Ung, R.V.; Rouleau, P.; Guertin, P.A. Tail pinching-induced hindlimb movements are suppressed by clonidine in spinal cord injured mice. Behav. Neurosci. 2008, 122, 576–588. [Google Scholar] [CrossRef]
- Lapointe, N.P.; Ung, R.V.; Rouleau, P.; Guertin, P.A. Effects of spinal alpha(2)-adrenoceptor and I-1-imidazoline receptor activation on hindlimb movement induction in spinal cord-injured mice. J. Pharmacol. Exp. Ther. 2008, 325, 994–1006. [Google Scholar] [CrossRef]
- Batschelet, E. Circular Statistics in Biology; Academic Press: New York, NY, USA, 1981. [Google Scholar]
- Kjaerulff, O.; Kiehn, O. Distribution of networks generating and coordinating locomotor activity in the neonatal rat spinal cord in vitro: a lesion study. J. Neurosci. 1996, 16, 5777–5794. [Google Scholar] [CrossRef]
- Zar, J.H. Circular distribution. In Biostatistical Analysis, 5th ed.; McElroy, W.D., Swanson, C.P., Eds.; Prentice Hall: Upper Saddle River, NJ, USA, 2010; pp. 605–668. [Google Scholar]
- Cowley, K.C.; Zaporozhets, E.; Maclean, J.N.; Schmidt, B.J. Is NMDA receptor activation essential for the production of locomotor-like activity in the neonatal rat spinal cord? J. Neurophysiol. 2005, 94, 3805–3814. [Google Scholar] [CrossRef]
- Majczyński, H.; Cabaj, A.M.; Jordan, L.M.; Sławińska, U. Contribution of 5-HT2 Receptors to the Control of the Spinal Locomotor System in Intact Rats. Front. Neural Circuits 2020, 14. [Google Scholar] [CrossRef]
- Lev-Tov, A.; Etlin, A.; Blivis, D. Sensory-induced activation of pattern generators in the absence of supraspinal control. Ann. N. Y. Acad. Sci. 2010, 1198, 54–62. [Google Scholar] [CrossRef]
- Meisel, R.L.; Rakerd, B. Induction of Hindlimb Stepping Movements in Rats Spinally Transected as Adults or as Neonates. Brain research 1982, 240, 353–356. [Google Scholar] [CrossRef]
- Pearson, K.G.; Rossignol, S. Fictive Motor Patterns in Chronic Spinal Cats. J. Neurophysiol. 1991, 66, 1874–1887. [Google Scholar] [CrossRef] [PubMed]
- Rajaofetra, N.; Ridet, J.L.; Poulat, P.; Marlier, L.; Sandillon, F.; Geffard, M.; Privat, A. Immunocytochemical Mapping of Noradrenergic Projections to the Rat Spinal-Cord with an Antiserum against Noradrenaline. J. Neurocytol. 1992, 21, 481–494. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Togashi, H.; Yoshioka, M.; Hirokami, M.; Tochihara, M.; Ikeda, T.; Smith, C.B.; Saito, H. Inhibitory Effects of Clonidine on Serotonergic Neuronal-Activity as Measured by Cerebrospinal-Fluid Serotonin and Its Metabolite in Anesthetized Rats. Life Sci. 1993, 53, 615–620. [Google Scholar] [CrossRef]
- Ortega, J.E.; Mendiguren, A.; Pineda, J.; Meana, J.J. Regulation of central noradrenergic activity by 5-HT3 receptors located in the locus coeruleus of the rat. Neuropharmacology 2012, 62, 2472–2479. [Google Scholar] [CrossRef]
- Pudovkina, O.L.; Cremers, T.I.F.H.; Westerink, B.H.C. The interaction between the locus coeruleus and dorsal raphe nucleus studied with dual-probe microdialysis. Eur. J. Pharmacol. 2002, 445, 37–42. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwaśniewska, A.; Miazga, K.; Majczyński, H.; Jordan, L.M.; Zawadzka, M.; Sławińska, U. Noradrenergic Components of Locomotor Recovery Induced by Intraspinal Grafting of the Embryonic Brainstem in Adult Paraplegic Rats. Int. J. Mol. Sci. 2020, 21, 5520. https://doi.org/10.3390/ijms21155520
Kwaśniewska A, Miazga K, Majczyński H, Jordan LM, Zawadzka M, Sławińska U. Noradrenergic Components of Locomotor Recovery Induced by Intraspinal Grafting of the Embryonic Brainstem in Adult Paraplegic Rats. International Journal of Molecular Sciences. 2020; 21(15):5520. https://doi.org/10.3390/ijms21155520
Chicago/Turabian StyleKwaśniewska, Anna, Krzysztof Miazga, Henryk Majczyński, Larry M. Jordan, Małgorzata Zawadzka, and Urszula Sławińska. 2020. "Noradrenergic Components of Locomotor Recovery Induced by Intraspinal Grafting of the Embryonic Brainstem in Adult Paraplegic Rats" International Journal of Molecular Sciences 21, no. 15: 5520. https://doi.org/10.3390/ijms21155520
APA StyleKwaśniewska, A., Miazga, K., Majczyński, H., Jordan, L. M., Zawadzka, M., & Sławińska, U. (2020). Noradrenergic Components of Locomotor Recovery Induced by Intraspinal Grafting of the Embryonic Brainstem in Adult Paraplegic Rats. International Journal of Molecular Sciences, 21(15), 5520. https://doi.org/10.3390/ijms21155520




