Bioluminescent Properties of Semi-Synthetic Obelin and Aequorin Activated by Coelenterazine Analogues with Modifications of C-2, C-6, and C-8 Substituents
Abstract
1. Introduction
2. Results and Discussion
2.1. Specific Bioluminescence Activity
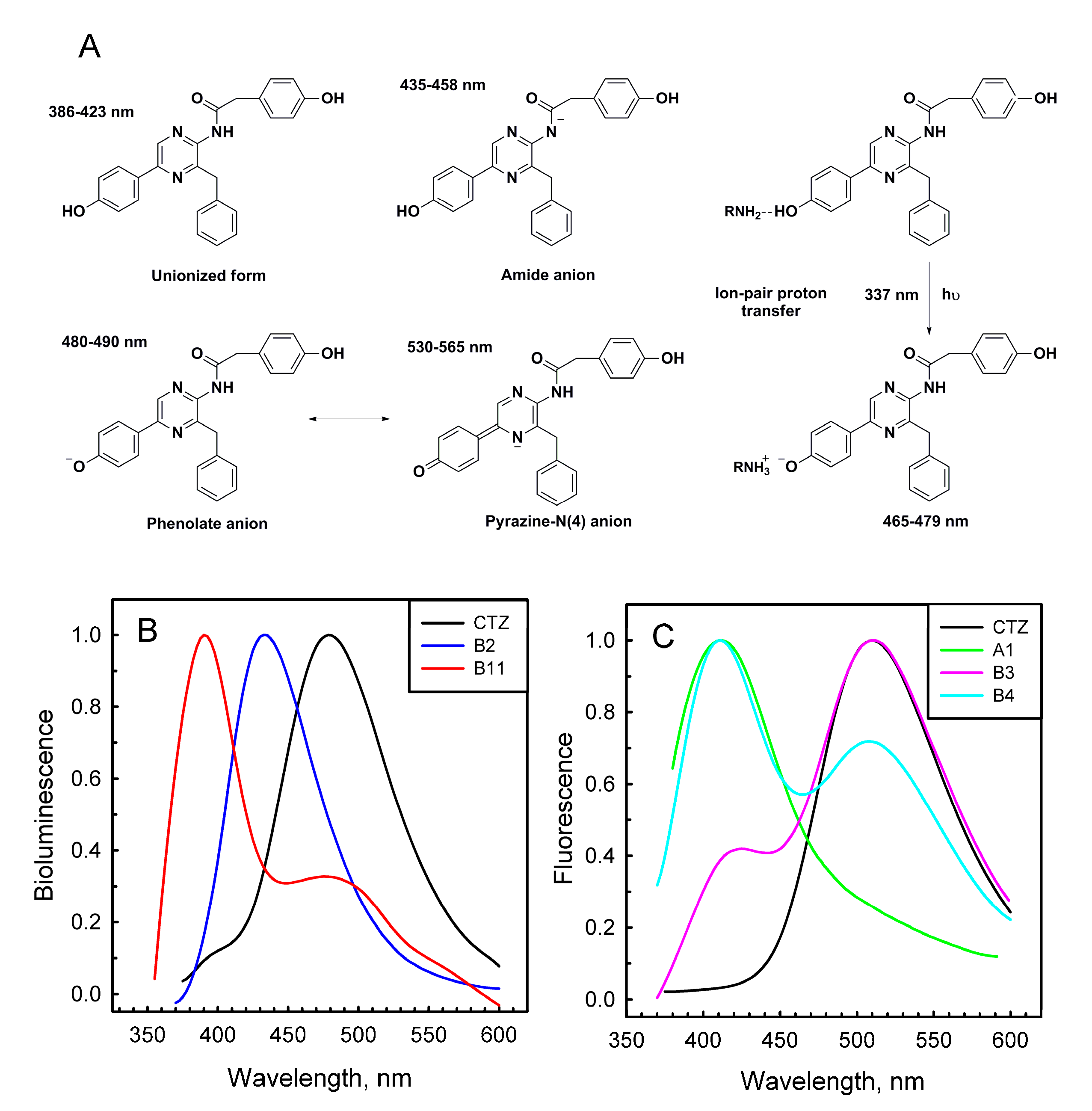
2.2. Bioluminescence and Fluorescence Spectra
2.3. Absorbance Spectra of Coelenterazine Analogues in Ethanol, and Active and Ca2+-Discharged Semi-Synthetic Photoproteins
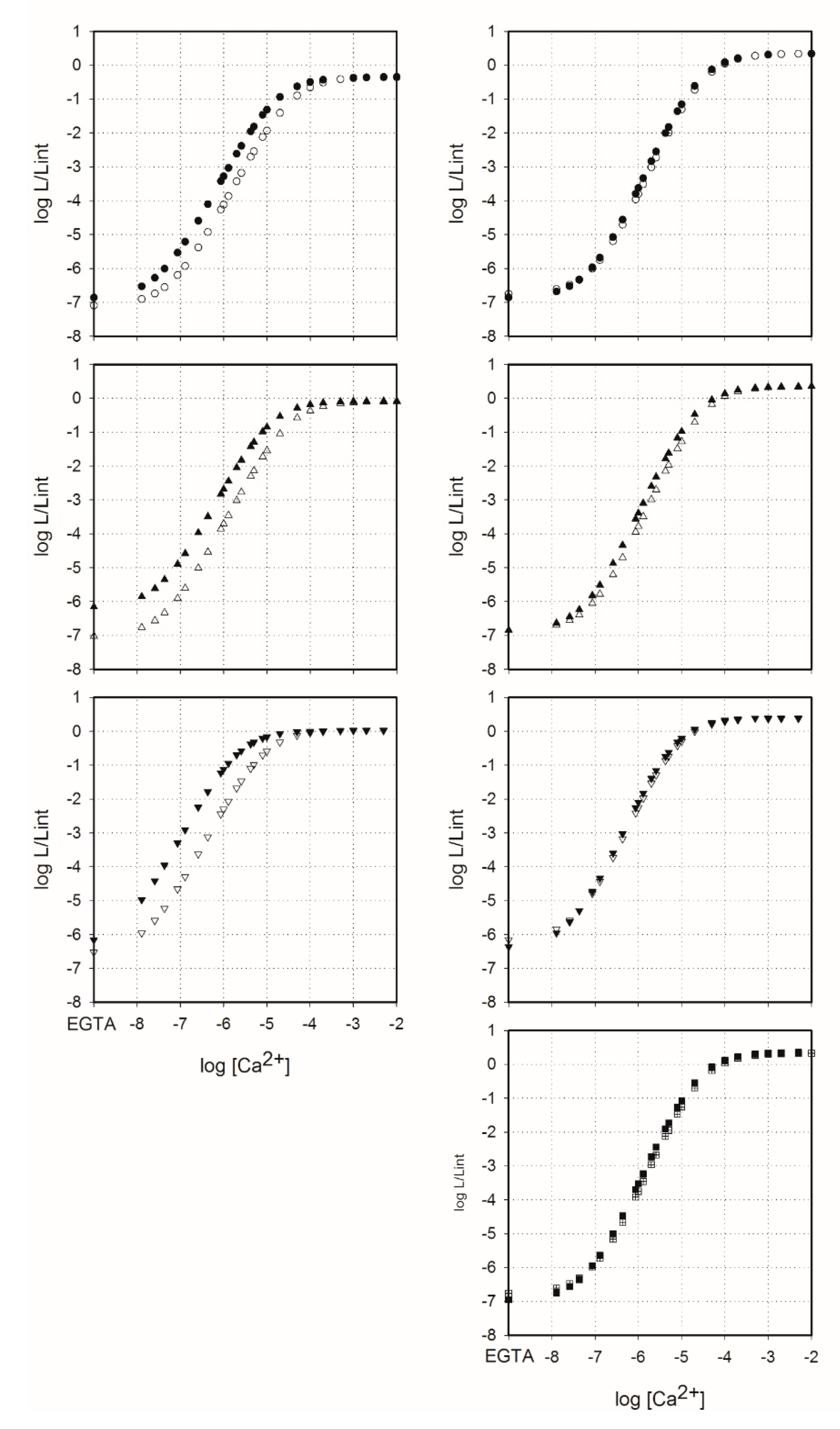
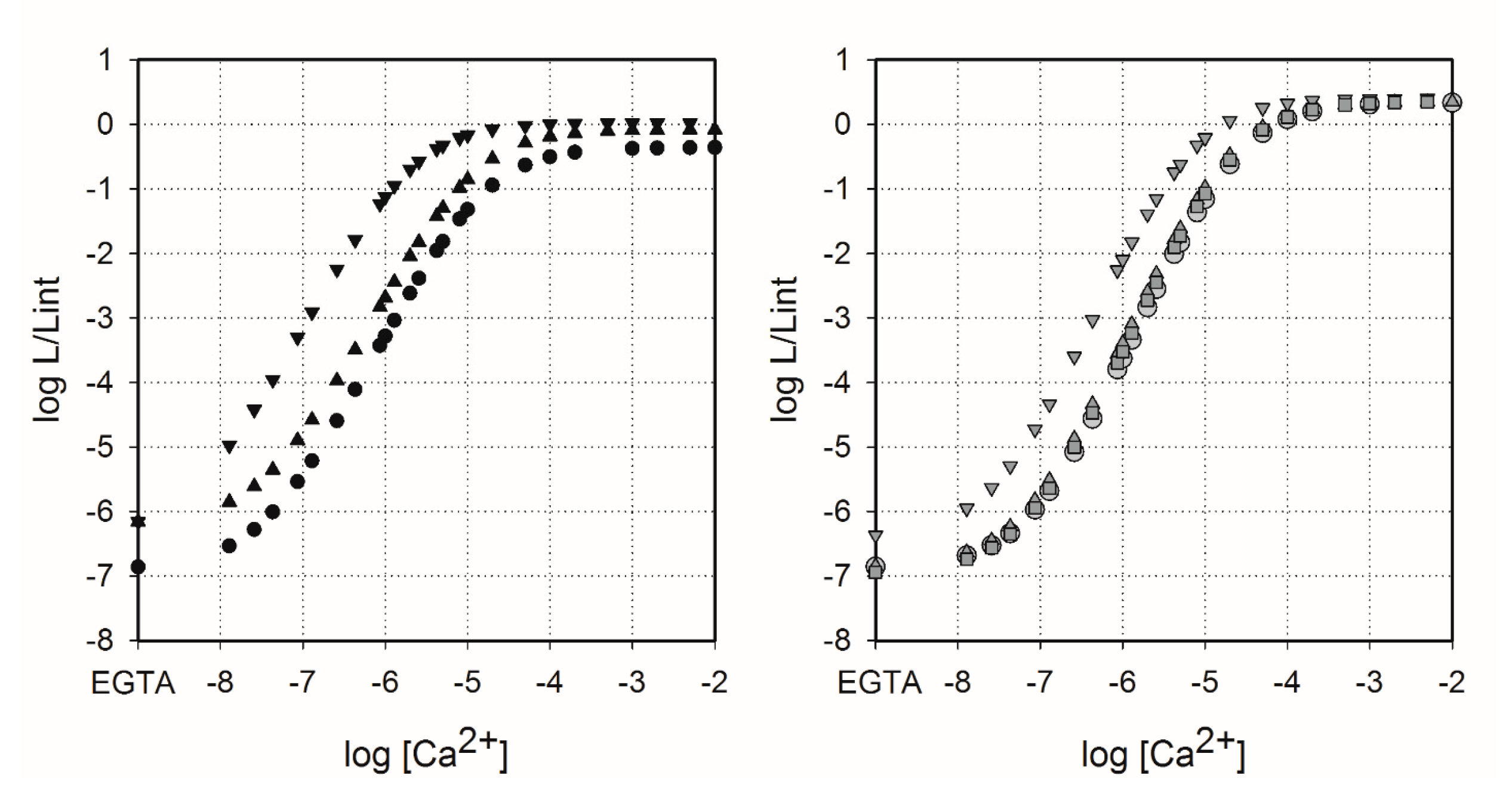
2.4. Sensitivity of Semi-Synthetic Obelins and Aequorins to Calcium
2.5. Rapid-Mixing Kinetics
3. Materials and Methods
3.1. Materials
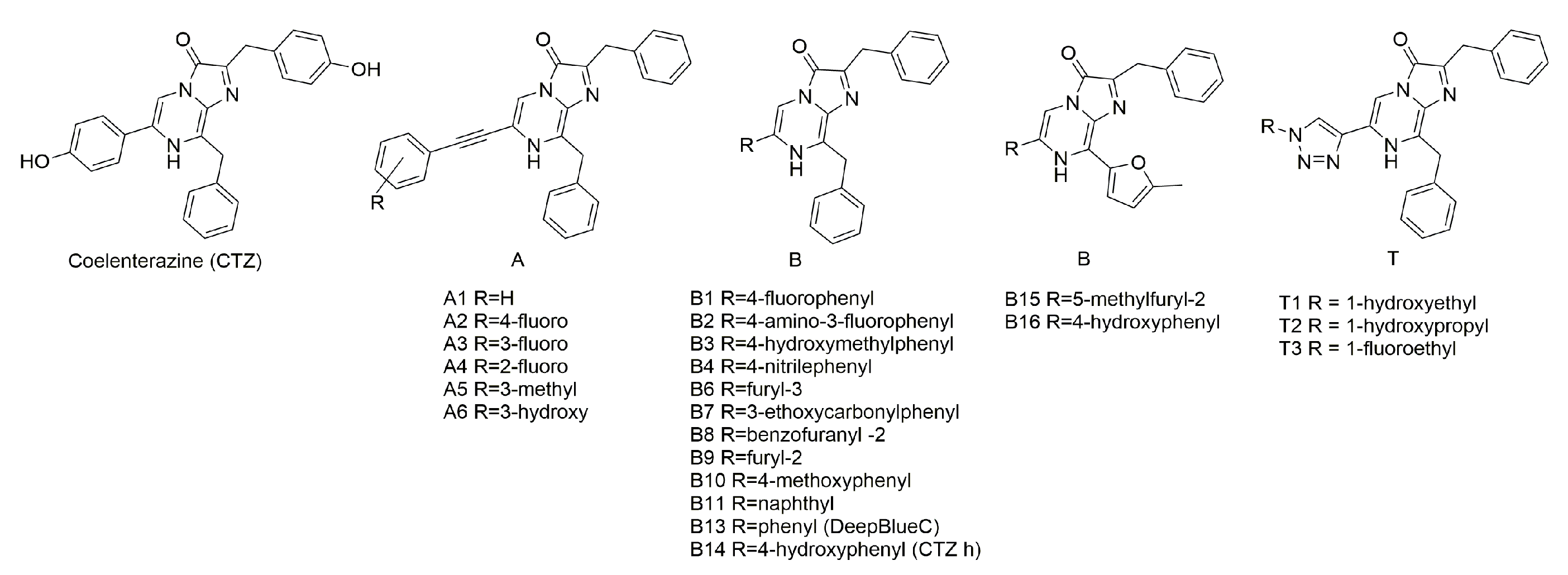
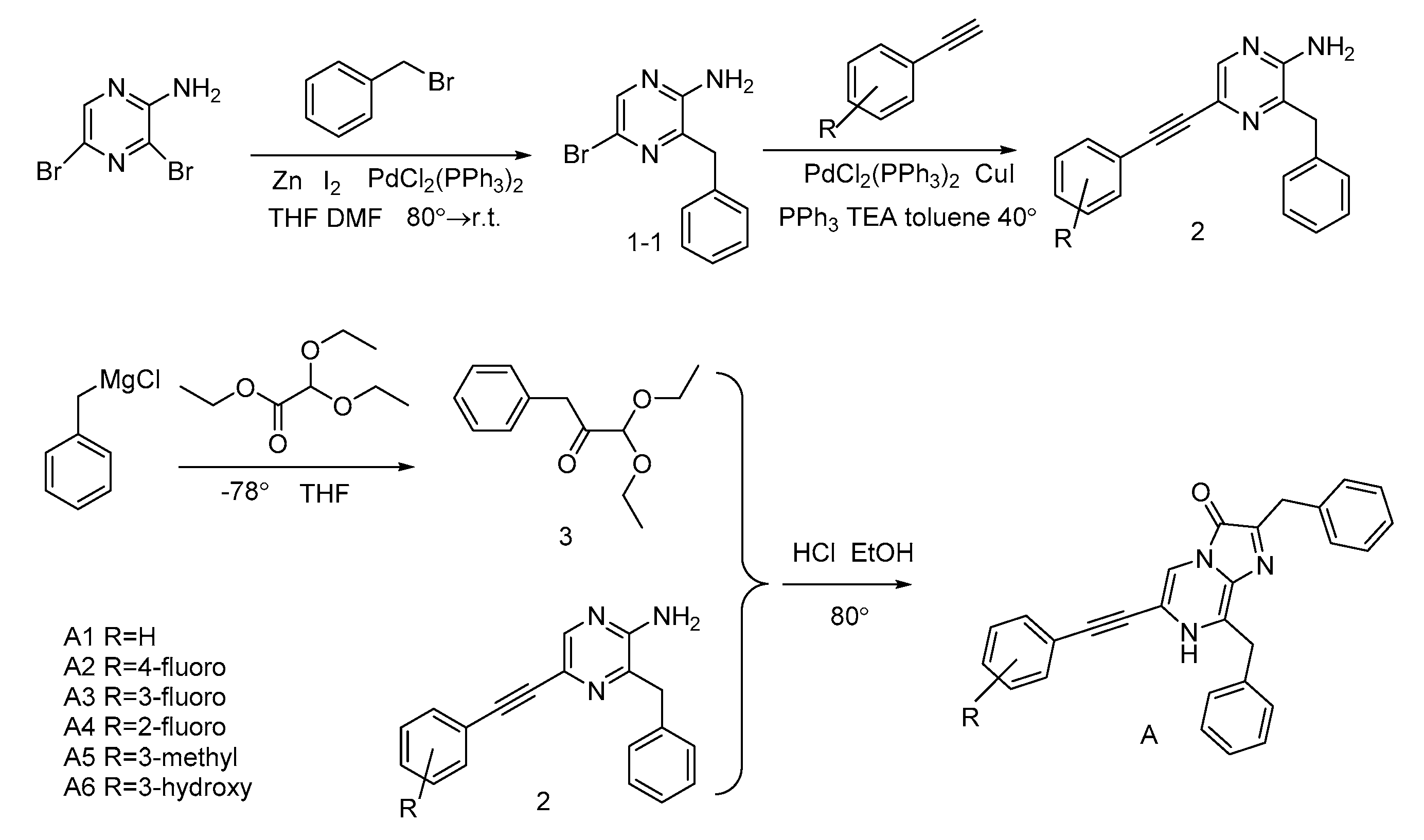
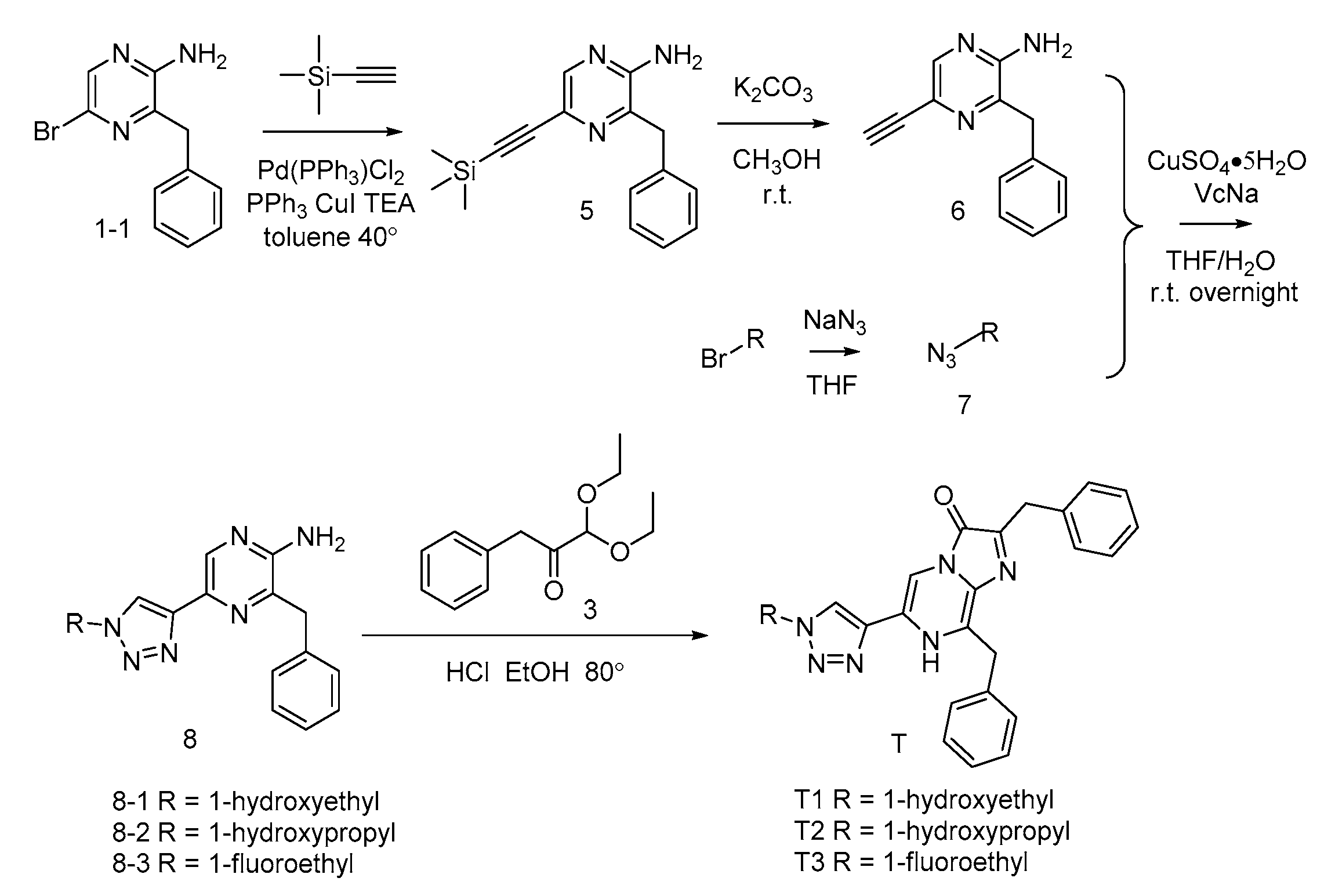
3.2. Synthesis of Coelenterazine Analogues
3.3. Protein Preparation
3.4. Determination of the Specific Bioluminescence Activity
3.5. Spectral Measurements
3.6. EDTA-Free Solutions of Photoproteins
3.7. Stopped-Flow Measurements and Kinetic Analysis
3.8. Calcium Concentration-Effect Curves
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| RLuc | luciferase from Renilla reniformis |
| GLuc | luciferase from Gaussia princeps |
| CTZ | coelenterazine |
| Ol | obelin from Obelia longissima |
| Aq | aequorin from Aequorea victoria |
| Ol_WT | obelin activated by native coelenterazine |
| Ol_A1 | obelin activated by A1 coelenterazine analogue |
| Ol_A2-A5 | obelins activated by A2, A3, A4, A5 analogues |
| Ol_A6 | obelin activated by A6 coelenterazine analogue |
| Ol_B1 | obelin activated by B1 coelenterazine analogue |
| Ol_B2 | obelin activated by B2 coelenterazine analogue |
| Ol_B3 | obelin activated by B3 coelenterazine analogue |
| Ol_B4 | obelin activated by B4 coelenterazine analogue |
| Ol_B11 | obelin activated by B11 coelenterazine analogue |
| Ol_B14 | obelin activated by B14 coelenterazine analogue |
| Aq_WT | aequorin activated by native coelenterazine |
| Aq_A1 | aequorin activated by A1 coelenterazine analogue |
| Aq_A6 | aequorin activated by A6 coelenterazine analogue |
| Aq_B1 | aequorin activated by B1 coelenterazine analogue |
| Aq_B2 | aequorin activated by B2 coelenterazine analogue |
| Aq_B14 | aequorin activated by B14 coelenterazine analogue |
| Aq_B15 | aequorin activated by B15 coelenterazine analogue |
| EDTA | ethylenediaminetetraacetic acid |
| EGTA | ethyleneglycol-bis(β-aminoethyl ether)tetraacetic acid |
| IPTG | isopropyl β-d-1-thiogalactopyranoside |
| DEAE | diethylaminoethyl (cellulose) |
| DTT | dithiothreitol |
| PIPES | piperazine-1,4-bis(2-ethanesulfonic acid) |
Appendix A
References
- Shimomura, O. Bioluminescence: Chemical Principles and Methods; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2006. [Google Scholar]
- Markova, S.V.; Vysotski, E.S. Coelenterazine-dependent luciferases. Biochemistry 2015, 80, 714–732. [Google Scholar] [PubMed]
- Haddock, S.H.; Rivers, T.J.; Robison, B.H. Can coelenterates make coelenterazine? Dietary requirement for luciferin in cnidarian bioluminescence. Proc. Natl. Acad. Sci. USA 2001, 98, 11148–11151. [Google Scholar] [CrossRef] [PubMed]
- Oba, Y.; Kato, S.; Ojika, M.; Inouye, S. Biosynthesis of coelenterazine in the deep-sea copepod, Metridia Pacifica. Biochem. Biophys. Res. Commun. 2009, 390, 684–688. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, O.; Johnson, F.H. Structure of the light-emitting moiety of aequorin. Biochemistry 1972, 11, 1602–1608. [Google Scholar] [CrossRef] [PubMed]
- Head, J.F.; Inouye, S.; Teranishi, K.; Shimomura, O. The crystal structure of the photoprotein aequorin at 2.3 Å resolution. Nature 2000, 405, 372–376. [Google Scholar] [CrossRef]
- Liu, Z.J.; Vysotski, E.S.; Deng, L.; Lee, J.; Rose, J.; Wang, B.C. Atomic resolution structure of obelin: Soaking with calcium enhances electron density of the second oxygen atom substituted at the C2-position of coelenterazine. Biochem. Biophys. Res. Commun. 2003, 311, 433–439. [Google Scholar] [CrossRef]
- Titushin, M.S.; Feng, Y.; Stepanyuk, G.A.; Li, Y.; Markova, S.V.; Golz, S.; Wang, B.C.; Lee, J.; Wang, J.; Vysotski, E.S.; et al. NMR-derived topology of a GFP-photoprotein energy transfer complex. J. Biol. Chem. 2010, 285, 40891–40900. [Google Scholar] [CrossRef]
- Burakova, L.P.; Natashin, P.V.; Markova, S.V.; Eremeeva, E.V.; Malikova, N.P.; Cheng, C.; Liu, Z.J.; Vysotski, E.S. Mitrocomin from the jellyfish Mitrocoma cellularia with deleted C-terminal tyrosine reveals a higher bioluminescence activity compared to wild type photoprotein. J. Photochem. Photobiol. B 2016, 162, 286–297. [Google Scholar] [CrossRef]
- Shimomura, O.; Johnson, F.H. Regeneration of the photoprotein aequorin. Nature 1975, 256, 236–238. [Google Scholar] [CrossRef]
- Eremeeva, E.V.; Natashin, P.V.; Song, L.; Zhou, Y.; van Berkel, W.J.; Liu, Z.J.; Vysotski, E.S. Oxygen activation of apo-obelin-coelenterazine complex. Chembiochem 2013, 14, 739–745. [Google Scholar] [CrossRef]
- Vysotski, E.S.; Lee, J. Ca2+-regulated photoproteins: Structural insight into the bioluminescence mechanism. Acc. Chem. Res. 2004, 37, 405–415. [Google Scholar] [CrossRef]
- Allen, D.G.; Blinks, J.R.; Prendergast, F.G. Aequorin luminescence: Relation of light emission to calcium concentration—A calcium-independent component. Science 1977, 195, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Du, L.; Li, M. Lighting up bioluminescence with coelenterazine: Strategies and applications. Photochem. Photobiol. Sci. 2016, 15, 466–480. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Yang, X.; Yang, X.; Yuan, M.; Zhang, T.; Zhang, H.; Li, M. Novel bioluminescent coelenterazine derivatives with imidazopyrazinone C-6 extended substitution for Renilla luciferase. Org. Biomol. Chem. 2016, 14, 5272–5281. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Yang, X.; Zhou, Y.; Yampolsky, I.; Du, L.; Li, M. New bioluminescent coelenterazine derivatives with various C-6 substitutions. Org. Biomol. Chem. 2017, 15, 7008–7018. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, O.; Johnson, F.H.; Saiga, Y. Extraction, purification and properties of aequorin, a bioluminescent protein from the luminous hydromedusan, Aequorea. J. Cell. Comp. Physiol. 1962, 59, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Prasher, D.; McCann, R.O.; Cormier, M.J. Cloning and expression of the cDNA coding for aequorin, a bioluminescent calcium-binding protein. Biochem. Biophys. Res. Commun. 1985, 126, 1259–1268. [Google Scholar] [CrossRef]
- Inouye, S.; Noguchi, M.; Sakaki, Y.; Takagi, Y.; Miyata, T.; Iwanaga, S.; Miyata, T.; Tsuji, F.I. Cloning and sequence analysis of cDNA for the luminescent protein aequorin. Proc. Natl. Acad. Sci. USA 1985, 82, 3154–3158. [Google Scholar] [CrossRef]
- Illarionov, B.A.; Bondar, V.S.; Illarionova, V.A.; Vysotski, E.S. Sequence of the cDNA encoding the Ca2+-activated photoprotein obelin from the hydroid polyp Obelia longissima. Gene 1995, 153, 273–274. [Google Scholar] [CrossRef]
- Illarionov, B.A.; Markova, S.V.; Bondar, V.S.; Vysotski, E.S.; Gitelson, J.I. Cloning and expression of cDNA for the Ca2+-activated photoprotein obelin from the hydroid polyp Obelia longissima. Dokl. Akad. Nauk. 1992, 326, 911–913. [Google Scholar]
- Vysotski, E.S.; Markova, S.V.; Frank, L.A. Calcium-regulated photoproteins of marine coelenterates. Mol. Biol. 2006, 40, 355–367. [Google Scholar] [CrossRef]
- Eremeeva, E.V.; Vysotski, E.S. Exploring bioluminescence function of the Ca2+-regulated photoproteins with site-directed mutagenesis. Photochem. Photobiol. 2019, 95, 8–23. [Google Scholar] [CrossRef]
- Gealageas, R.; Malikova, N.P.; Picaud, S.; Borgdorff, A.J.; Burakova, L.P.; Brulet, P.; Vysotski, E.S.; Dodd, R.H. Bioluminescent properties of obelin and aequorin with novel coelenterazine analogues. Anal. Bioanal. Chem. 2014, 406, 2695–2707. [Google Scholar] [CrossRef]
- Stepanyuk, G.A.; Golz, S.; Markova, S.V.; Frank, L.A.; Lee, J.; Vysotski, E.S. Interchange of aequorin and obelin bioluminescence color is determined by substitution of one active site residue of each photoprotein. FEBS Lett. 2005, 579, 1008–1014. [Google Scholar] [CrossRef]
- Natashin, P.V.; Markova, S.V.; Lee, J.; Vysotski, E.S.; Liu, Z.J. Crystal structures of the F88Y obelin mutant before and after bioluminescence provide molecular insight into spectral tuning among hydromedusan photoproteins. FEBS J. 2014, 281, 1432–1445. [Google Scholar] [CrossRef]
- Li, Z.S.; Zou, L.Y.; Min, C.G.; Ren, A.M. The effect of micro-environment on luminescence of aequorin: The role of amino acids and explicit water molecules on spectroscopic properties of coelenteramide. J. Photochem. Photobiol. B 2013, 127, 94–99. [Google Scholar] [CrossRef]
- Shimomura, O.; Musicki, B.; Kishi, Y. Semi-synthetic aequorins with improved sensitivity to Ca2+ ions. Biochem. J. 1989, 261, 913–920. [Google Scholar] [CrossRef]
- Malikova, N.P.; Borgdorff, A.J.; Vysotski, E.S. Semisynthetic photoprotein reporters for tracking fast Ca2+ transients. Photochem. Photobiol. Sci. 2015, 14, 2213–2224. [Google Scholar] [CrossRef]
- Shimomura, O.; Teranishi, K. Light-emitters involved in the luminescence of coelenterazine. Luminescence 2000, 15, 51–58. [Google Scholar] [CrossRef]
- Chen, S.F.; Navizet, I.; Roca-Sanjuán, D.; Lindh, R.; Liu, Y.J.; Ferré, N. Chemiluminescence of coelenterazine and fluorescence of coelenteramide: A systematic theoretical study. J. Chem. Theory Comput. 2012, 8, 2796–2807. [Google Scholar] [CrossRef]
- Chen, S.F.; Ferré, N.; Liu, Y.J. QM/MM study on the light emitters of aequorin chemiluminescence, bioluminescence, and fluorescence: A general understanding of the bioluminescence of several marine organisms. Chemistry 2013, 19, 8466–8472. [Google Scholar] [CrossRef]
- Ireland, J.F.; Wyatt, P.A.H. Acid-base properties of electronically excited states of organic molecules. In Advances in Physical Organic Chemistry; Gold, V., Ed.; Academic Press: Cambridge, MA, USA, 1976; Volume 12, pp. 131–221. [Google Scholar]
- van Oort, B.; Eremeeva, E.V.; Koehorst, R.B.; Laptenok, S.P.; van Amerongen, H.; van Berkel, W.J.; Malikova, N.P.; Markova, S.V.; Vysotski, E.S.; Visser, A.J.; et al. Picosecond fluorescence relaxation spectroscopy of the calcium-discharged photoproteins aequorin and obelin. Biochemistry 2009, 48, 10486–10491. [Google Scholar] [CrossRef]
- Burakova, L.P.; Vysotski, E.S. Recombinant Ca2+-regulated photoproteins of ctenophores: Current knowledge and application prospects. Appl. Microbiol. Biotechnol. 2019, 103, 5929–5946. [Google Scholar] [CrossRef]
- Ridgway, E.B.; Ashley, C.C. Calcium transients in single muscle fibers. Biochem. Biophys. Res. Commun. 1967, 29, 229–234. [Google Scholar] [CrossRef]
- Ottolini, D.; Calì, T.; Brini, M. Measurements of Ca2+ concentration with recombinant targeted luminescent probes. In Calcium Signaling Protocols; Lambert, D.G., Rainbow, R.D., Eds.; Humana Press: Totowa, NJ, USA, 2013; pp. 273–291. [Google Scholar]
- Shimomura, O.; Musicki, B.; Kishi, Y.; Inouye, S. Light-emitting properties of recombinant semi-synthetic aequorins and recombinant fluorescein-conjugated aequorin for measuring cellular calcium. Cell Calcium 1993, 14, 373–378. [Google Scholar] [CrossRef]
- Markova, S.V.; Vysotski, E.S.; Blinks, J.R.; Burakova, L.P.; Wang, B.C.; Lee, J. Obelin from the bioluminescent marine hydroid Obelia geniculata: Cloning, expression, and comparison of some properties with those of other Ca2+-regulated photoproteins. Biochemistry 2002, 41, 2227–22236. [Google Scholar] [CrossRef]
- Romani, A.M.P. Cellular magnesium homeostasis. Arch. Biochem. Biophys. 2011, 512, 1–23. [Google Scholar] [CrossRef]
- Malikova, N.P.; Burakova, L.P.; Markova, S.V.; Vysotski, E.S. Characterization of hydromedusan Ca2+-regulated photoproteins as a tool for measurement of Ca2+ concentration. Anal. Bioanal. Chem. 2014, 406, 5715–5726. [Google Scholar] [CrossRef]
- Sabatini, B.L.; Oertner, T.G.; Svoboda, K. The life cycle of Ca2+ ions in dendritic spines. Neuron 2002, 33, 439–452. [Google Scholar] [CrossRef]
- Cheng, H.; Lederer, W.J.; Cannell, M.B. Calcium sparks: Elementary events underlying excitation-contraction coupling in heart muscle. Science 1993, 262, 740–744. [Google Scholar] [CrossRef]
- Eremeeva, E.V.; Bartsev, S.I.; van Berkel, W.J.; Vysotski, E.S. Unanimous model for describing the fast bioluminescence kinetics of Ca2+-regulated photoproteins of different organisms. Photochem. Photobiol. 2017, 93, 495–502. [Google Scholar] [CrossRef]
- Markova, S.V.; Vysotski, E.S.; Lee, J. Obelin hyperexpression in E. coli, purification and characterization. In Bioluminescence and Chemiluminescence; Case, J.F., Herring, P.J., Robison, B.H., Haddock, S.H.D., Kricka, L.J., Stanley, P.E., Eds.; World Scientific Publishing Co.: Singapore, 2001; pp. 115–119. [Google Scholar]
- Deng, L.; Vysotski, E.S.; Markova, S.V.; Liu, Z.J.; Lee, J.; Rose, J.; Wang, B.C. All three Ca2+-binding loops of photoproteins bind calcium ions: The crystal structures of calcium-loaded apo-aequorin and apo-obelin. Protein Sci. 2005, 14, 663–675. [Google Scholar] [CrossRef]
- Illarionov, B.A.; Frank, L.A.; Illarionova, V.A.; Bondar, V.S.; Vysotski, E.S.; Blinks, J.R. Recombinant obelin: Cloning and expression of cDNA purification, and characterization as a calcium indicator. Methods Enzymol. 2000, 305, 223–249. [Google Scholar]
- Edelhoch, H. Spectroscopic determination of tryptophan and tyrosine in proteins. Biochemistry 1967, 6, 1948–1954. [Google Scholar] [CrossRef]
- Eremeeva, E.V.; Markova, S.V.; Frank, L.A.; Visser, A.J.; van Berkel, W.J.; Vysotski, E.S. Bioluminescent and spectroscopic properties of His-Trp-Tyr triad mutants of obelin and aequorin. Photochem. Photobiol. Sci. 2013, 12, 1016–1024. [Google Scholar] [CrossRef]
- Klabusay, M.; Blinks, J.R. Some commonly overlooked properties of calcium buffer systems: A simple method for detecting and correcting stoichiometric imbalance in CaEGTA stock solutions. Cell Calcium 1996, 20, 227–234. [Google Scholar] [CrossRef]

| CTZ | Aequorin | Obelin | ||||
|---|---|---|---|---|---|---|
| Activity % | Bioluminescence λmax (nm) | Fluorescence λmax (nm) | Activity % | Bioluminescence λmax (nm) | Fluorescence λmax (nm) | |
| native | 100.00 | 473 | 470 | 100.00 | 390/482 | 505 |
| A1 | 0.40 | 468 | 407 | 8.2 | 390/475 | 410 |
| A2 | 0.20 | 446 | 441 | 0.02 | 434 | 410/510 |
| A3 | 0.04 | 458 | 407 | 0.01 | 432 | 407/500 |
| A4 | 0.03 | 466 | 407 | 0.01 | 444 | 404/500 |
| A5 | 0.03 | 466 | 407 | 0.01 | 444 | 404/500 |
| A6 | 79.0 | 468 | 476 | 70.0 | 390/478 | 512 |
| B1 | 0.20 | 470 | 422 | 48.0 | 390/476 | 420/512 |
| B2 | 0.50 | 452 | 488 | 5.3 | 432 | 437 |
| B3 | 0.84 | 468 | 422 | 22.7 | 390/476 | 420/512 |
| B4 | 0.25 | 478 | 416 | 6.6 | 390/477 | 410/508 |
| B6 | 0.16 | 475 | 420 | 0.2 | 390/475 | 416 |
| B7 | 0.02 | ND | 422/505 | 0.25 | 390/477 | 420/505 |
| B8 | 0.02 | ND | 430 | 0.06 | ND | 423 |
| B9 | 0.06 | ND | 420 | 0.10 | 390/477 | 428 |
| B10 | 0.06 | ND | 430 | 0.06 | ND | 428 |
| B11 | 0.07 | 473 | 430/510 | 0.05 | 390/482 | 411/518 |
| B13 | 0.01 | ND | 414 | 0.04 | ND | 409/510 |
| B14 | 50.0 | 470 | 400/464 | 76.1 | 482 | 410/501 |
| B15 | 30.0 | 470 | 400/480 | 2.5 | 390/478 | 410/500 |
| B16 | 0.70 | 473 | 424/484 | 0.09 | 474 | 440–490 |
| T1 | 0.08 | 473 | 416 | 0.08 | 390/480 | 416 |
| T2 | 0.09 | 470 | 413 | 0.02 | ND | 413 |
| T3 | 0.02 | ND | 416 | 0.01 | ND | 416 |
| Protein | Bioluminescence Activity, % | ||
|---|---|---|---|
| Native CTZ | B1 | B15 | |
| Obelin WT | 100 | 48.0 | 2.5 |
| Obelin F88Y | 100 | 16.0 | 19.0 |
| Aequorin WT | 100 | 0.2 | 30.0 |
| Aequorin Y82F | 100 | 45.0 | 1.2 |
| CTZ | Absorbance (λmax, nm) | ||||
|---|---|---|---|---|---|
| In Ethanol | Aequorin | Obelin | |||
| Active | Ca2+-Discharged | Active | Ca2+-Discharged | ||
| native | 350/434 | 460 | 335 | 460 | 335 |
| A1 | 344/405 | 344 | 344 | 460 | 335 |
| A2 | 386/434 | 344 | 344 | ND | ND |
| A3 | 393/434 | 344 | 344 | ND | ND |
| A4 | 390/434 | 344 | 344 | 344 | 344 |
| A5 | 386/434 | 350 | 350 | 350 | 350 |
| A6 | 386/434 | 460 | 335 | 460 | 344 |
| B1 | 358/434 | 338/405 | 338/405 | 344/460 | 344 |
| B2 | 434 | 350/400 | 340/430 | 350/430 | 350/430 |
| B3 | 358/434 | 350/410 | 350/410 | 340/420 | 345/420 |
| B4 | 334/394 | 336 | 336 | 336/445 | 336/445 |
| B6 | 330/434 | 322 | 322 | 360/445 | 360/445 |
| B7 | 344/422 | 314/400 | 425 | 346 | 346 |
| B8 | 395 | 350/400 | 350/400 | 350 | 350 |
| B9 | 350/418 | 350/400 | 350/400 | 360/425 | 360/425 |
| B10 | 354/437 | 352/400 | 352/400 | 360 | 360 |
| B11 | 354/426 | 354/426 | 354/426 | 335/365 | 433 |
| B13 | 354/430 | 330 | 330 | 334 | 334/427 |
| B14 | 344/434 | 344/434 | 344 | 344/434 | 344 |
| B15 | 380/450 | 400 | 320/417 | 377 | 320/387 |
| B16 | 390 | 370 | 370 | 380 | 380 |
| T1 | 349/428 | 334/400 | 334/400 | 334/400 | 334/400 |
| T2 | 344/426 | 334/400 | 334/400 | 334/400 | 334/400 |
| T3 | 349/430 | 334/400 | 334/400 | 334/400 | 334/400 |
| Photoprotein | [Ca2+]limit, nM | Kd, nM | ||
|---|---|---|---|---|
| Without Mg2+ | With 1 mM Mg2+ | Without Mg2+ | With 1 mM Mg2+ | |
| Ol_WT | 52 ± 4 | 57 ± 5 | 87 ± 11 | 112 ± 9 |
| Ol_A6 | 40 ± 5 | 48 ± 4 | 71 ± 8 | 100 ± 8 |
| Ol_B1 | 43 ± 5 | 63 ± 6 | 74 ± 9 | 105 ± 10 |
| Ol_B14 | 21 ± 2 | 23 ± 2 | 33 ± 2 | 45 ± 5 |
| Aq_WT | 19 ± 2 | 53 ± 5 | 41 ± 5 | 100 ± 9 |
| Aq_A6 | 27 ± 4 | 46 ± 5 | 38 ± 3 | 83 ± 6 |
| Aq_B14 | 5.6 ± 1 | 12 ± 1 | 6.3 ± 3 | 33 ± 4 |
| Photoprotein | krise, s−1 | kdecay | |
|---|---|---|---|
| k1, s−1 | k2, s−1 | ||
| Ol_WT | 510.0 ± 5.0 | 40.0 ± 1.75 (0.66) | 4.8 ± 0.01 (0.34) |
| Ol_A6 | 599.3 ± 7.7 | 38.1 ± 0.14 (0.62) | 5.9 ± 0.01 (0.38) |
| Ol_B1 | 677.1 ± 14.2 | 39.2 ± 0.29 (0.44) | 5.0 ± 0.01 (0.56) |
| Ol_B14 | 577.6 ± 30.5 | 38.5 ± 0.14 (0.60) | 5.5 ± 0.01 (0.40) |
| Aq_WT | 123.0 ± 1.0 | 0.81 ± 0.01 | - |
| Aq_A6 | 302.9 ± 3.4 | 8.11 ± 0.03 (0.37) | 0.86 ± 0.01 (0.63) |
| Aq_B14 | 160.3 ± 1.7 | 1.17 ± 0.01 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eremeeva, E.V.; Jiang, T.; Malikova, N.P.; Li, M.; Vysotski, E.S. Bioluminescent Properties of Semi-Synthetic Obelin and Aequorin Activated by Coelenterazine Analogues with Modifications of C-2, C-6, and C-8 Substituents. Int. J. Mol. Sci. 2020, 21, 5446. https://doi.org/10.3390/ijms21155446
Eremeeva EV, Jiang T, Malikova NP, Li M, Vysotski ES. Bioluminescent Properties of Semi-Synthetic Obelin and Aequorin Activated by Coelenterazine Analogues with Modifications of C-2, C-6, and C-8 Substituents. International Journal of Molecular Sciences. 2020; 21(15):5446. https://doi.org/10.3390/ijms21155446
Chicago/Turabian StyleEremeeva, Elena V., Tianyu Jiang, Natalia P. Malikova, Minyong Li, and Eugene S. Vysotski. 2020. "Bioluminescent Properties of Semi-Synthetic Obelin and Aequorin Activated by Coelenterazine Analogues with Modifications of C-2, C-6, and C-8 Substituents" International Journal of Molecular Sciences 21, no. 15: 5446. https://doi.org/10.3390/ijms21155446
APA StyleEremeeva, E. V., Jiang, T., Malikova, N. P., Li, M., & Vysotski, E. S. (2020). Bioluminescent Properties of Semi-Synthetic Obelin and Aequorin Activated by Coelenterazine Analogues with Modifications of C-2, C-6, and C-8 Substituents. International Journal of Molecular Sciences, 21(15), 5446. https://doi.org/10.3390/ijms21155446






