The New Frontier in Oxytocin Physiology: The Oxytonic Contraction
Abstract
1. Introduction
2. Oxytocin- and Oxytocin Receptor-Deficient Mice: The Paradox of Normophagic Obesity
3. Thermoregulation after OXT-Neuron Depletion
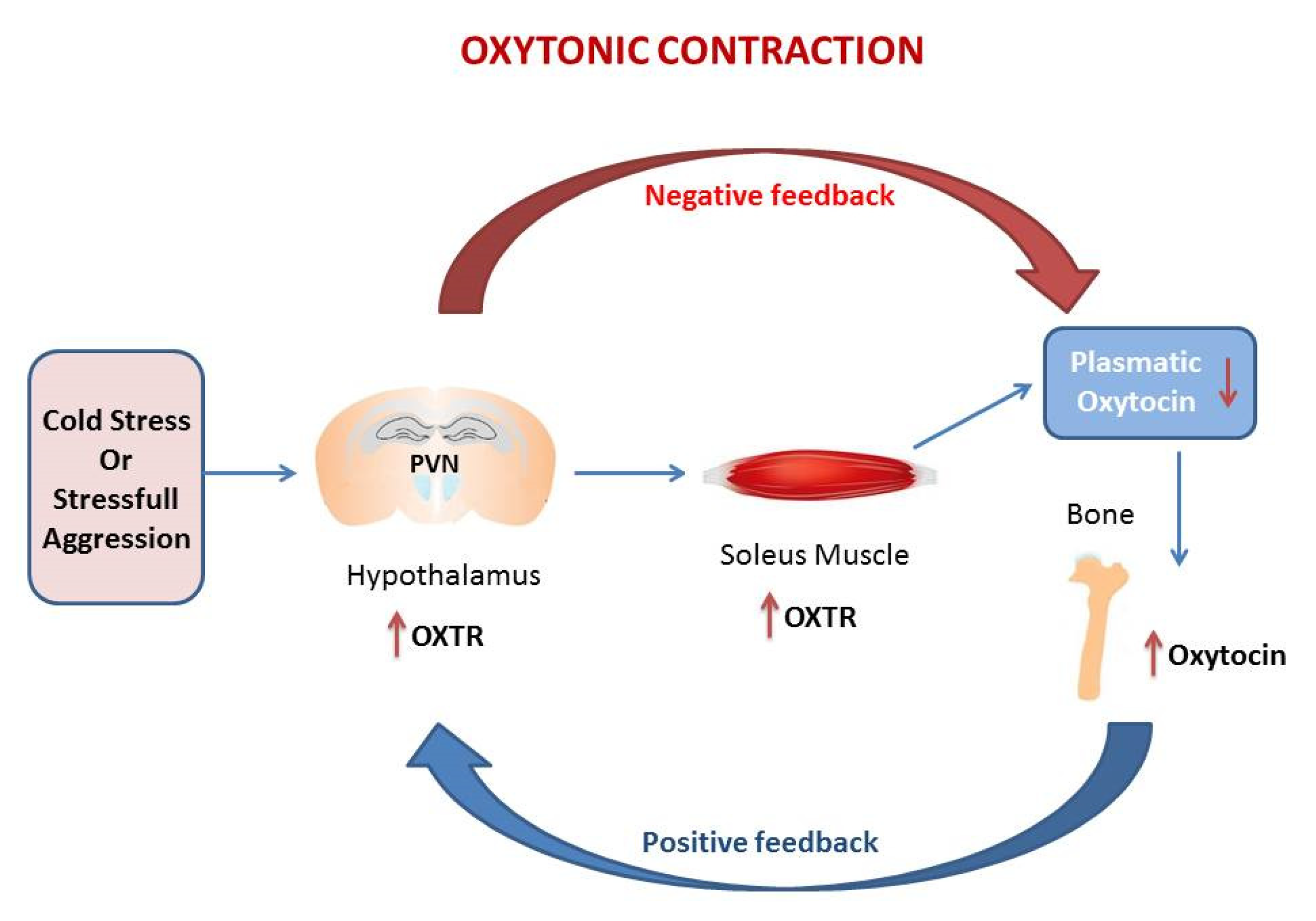
4. Cold Stress Triggers the Oxytonic Contraction
5. A New Hypothesis: Oxytocin Increases Skeletal Muscle Tonicity after Thermogenic Challenge
6. Oxytonic in Skeletal Muscle Physiology
7. Oxytocin Is a Gastrointestinal Hormone: The Tonic Effect
8. Oxytonic in Human Studies
9. Oxytonic in Human Diseases: Prader–Willy Syndrome and Autistic Spectrum Disorder
10. A Genetic Approach: The MAGE Family of Genes
11. Conclusions
Funding
Conflicts of Interest
Abbreviations
| ASD | Autism spectrum disorder |
| BAT | Brown adipose tissue |
| CS | Cold stress |
| CSF | Cerebrospinal fluid |
| HIPP | Hippocampus |
| MyHC | Myosin heavy chain |
| Oxt | Oxytocin |
| Oxtr | Oxytocin receptor |
| PVN | Paraventricular nuclei |
| PWS | Prader–Willy syndrome |
| SON | Supraoptical nuclei |
| T2DM | Type 2 diabetes mellitus |
| TA | Tibialis anterioris |
| TRPV1 | Transient receptor potential cation channel subfamily V member 1 |
References
- Fitch, W.M.; Margoliash, E. Construction of phylogenetic trees. Science 1967, 155, 279–284. [Google Scholar] [CrossRef]
- Fitch, W.M. Distinguishing omologous from analogous proteins. Syst. Biol. 1970, 19, 99–113. [Google Scholar]
- Fitch, W.M. Considerations regarding the regulation of gene transcription and messenger translation. J. Mol. Evol. 1972, 1, 185–207. [Google Scholar] [CrossRef] [PubMed]
- Karsenty, G.; Ferron, M. The contribution of bone to whole-organism physiology. Nature 2012, 481, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Camerino, C.; Conte, E.; Cannone, M.; Caloiero, R.; Fonzino, A.; Tricarico, D. Nerve growth factor, brain-derived neurotrophic factor and osteocalcin gene relationship in energy regulation, bone homeostasis and reproductive organs analyzed by mRNA quantitative evaluation and linear correlation analysis. Front. Physiol. 2016, 7, 456. [Google Scholar] [CrossRef] [PubMed]
- Camerino, C.; Conte, E.; Caloiero, R.; Fonzino, A.; Carratù, M.; Lograno, M.D.; Tricarico, D. Evaluation of short and long term cold stress challenge of nerve grow factor, brain-derived neurotrophic factor, osteocalcin and oxytocin mrna expression in bat, brain, bone and reproductive tissue of male mice using real-time pcr and linear correlation analysis. Front. Physiol. 2018, 8, 1101. [Google Scholar] [PubMed]
- Young, W.F. Netter Atlas of Human Physiopathology and Clinic, 2nd ed.; Elsevier: Philadelphia, PA, USA, 2011. [Google Scholar]
- Yokoyama, Y.; Ueda, T.; Irahara, M.; Aono, T. Releases of oxytocin and prolactin during breast massage and suckling in puerperal women. Eur. J. Obs. Gynecol. Reprod. Biol. 1994, 53, 17–20. [Google Scholar] [CrossRef]
- Eisenberg, Y.; Dugas, L.R.; Akbar, A.; Reddivari, B.; Layden, B.T.; Barengolts, E. Oxytocin is lower in african american men with diabetes and associates with psycho-social and metabolic health factors. PLoS ONE 2018, 13, e0190301. [Google Scholar] [CrossRef]
- Camerino, C.; Conte, E.; Carratù, M.R.; Fonzino, A.; Lograno, M.D.; Tricarico, D. Oxytocin/Osteocalcin/IL-6 and NGF/BDNF mRNA levels in response to cold stress challenge in mice: Possible oxytonic brain-bone-muscle-interaction. Front. Physiol. 2019, 10, 1437. [Google Scholar] [CrossRef]
- Conte, E.; Romano, A.; De Bellis, M.; De Ceglia, M.; Carratù, M.R.; Gaetani, S.; Tricarico, D.; Camerino, C. The effect of oxytocin hits the slow-twitch muscle modulating oxytocin receptor in PVN/SON and plasmatic oxytocin after cold stress challenge in mice. (unpublished; manuscript in preparation).
- Altstein, M.; Gainer, H. Differential biosynthesis and posttranslational processing of vasopressin and oxytocin in rat brain during embryonic and postnatal development. J. Neurosci. 1988, 8, 3967–3977. [Google Scholar] [CrossRef] [PubMed]
- Deblon, N.; Veyrat-Durebex, C.; Bourgoin, L.; Caillon, A.; Bussier, A.L.; Petrosino, S.; Piscitelli, F.; Legros, J.J.; Geenen, V.; Foti, M.; et al. Mechanisms of the anti-obesity effects of oxytocin in diet-induced obese rats. PLoS ONE 2011, 6, e25565. [Google Scholar] [CrossRef]
- Gimpl, G.; Fahrenholz, F. The oxytocin receptor system: Structure, function, and regulation. Physiol. Rev. 2001, 81, 629–683. [Google Scholar] [CrossRef]
- Dombret, C.; Nguyen, T.; Schakman, O.; Michaud, J.L.; Hardin-Pouzet, H.; Bertrand, M.J.M.; De Backer, O. Loss of maged1 results in obesity, deficits of social interactions, impaired sexual behavior and severe alteration of mature oxytocin production in the hypothalamu. Hum. Mol. Genet. 2012, 21, 4703–4717. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.; Mikkelsen, J.D. Oxytocin-anatomy and functional assignments: A minireview. Endocr. Regul. 2005, 39, 97–105. [Google Scholar] [PubMed]
- Lee, H.J.; Macbeth, A.H.; Pagani, J.H.; Young, W.S. Oxytocin: The great facilitator of life. Prog. Neurobiol. 2009, 88, 127–151. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Lindenberg, A.; Domes, G.; Kirsch, P.; Heinrichs, M. Oxytocin and vasopressin in the human brain: Social neuropeptides for translational medicine. Nat. Rev. Neurosci. 2011, 12, 524–538. [Google Scholar] [PubMed]
- Fountain, M.D., Jr.; Schaaf, C.P. MAGEL2 and oxytocin-implications in Prader-Willi syndrome and beyond. Biol. Psychiatry 2015, 78, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Feng, M.; Wang, C.; Ye, Y.; Wang, P.S.; Liu, C. Oxytocin receptor expressed on the smooth muscle mediates the excitatory effect of oxytocin on gastric motility in rats. Neurogastroenterol. Motil. 2009, 21, 430–438. [Google Scholar] [CrossRef]
- Elabd, C.; Basillais, A.; Beaupied, H.; Breuil, V.; Wagner, N.; Scheideler, M.; Zaragosi, L.-E.; Massiéra, F.; Lemichez, E.; Trajanoski, Z.; et al. Oxytocin controls differentiation of human mesenchymal stem cells and reverses osteoporosis. Stem. Cells 2008, 26, 2399–2407. [Google Scholar] [CrossRef]
- Yang, X.; Xi, T.F.; Li, Y.X.; Wang, H.H.; Qin, Y.; Zhang, J.P.; Cai, W.T.; Huang, M.T.; Shen, J.Q.; Fan, X.M.; et al. Oxytocin decreases colonic motility of cold water stressed rats via oxytocin receptors. World J. Gastroenterol. 2014, 20, 10886–10894. [Google Scholar] [CrossRef]
- Takayanagi, Y.; Kasahara, Y.; Onaka, T.; Takahashi, N.; Kawada, T.; Nishimori, K. Oxytocin receptor-deficient mice developed late-onset obesity. NeuroReport 2008, 19, 951–955. [Google Scholar] [CrossRef]
- Camerino, C. Low sympathetic tone and obese phenotype in oxytocin-deficient mice. Obesity 2009, 17, 980–984. [Google Scholar] [CrossRef] [PubMed]
- Kublaoui, B.M.; Gemelli, T.; Tolson, K.P.; Wang, Y.; Zinn, A.R. Oxytocin deficiency mediates hyperphagic obesity of sim1 haploinsufficient mice. Mol. Endocrinol. 2008, 22, 1723–1734. [Google Scholar] [CrossRef] [PubMed]
- Qian, S.; Chen, H.; Weingarth, D.; Trumbauer, M.E.; Novi, D.E.; Guan, X.; Yu, H.; Shen, Z.; Feng, Y.; Frazier, E.; et al. Neither agouti-related protein nor neuropeptide y is critically required for the regulation of energy homeostasis in mice. Mol. Cell. Biol. 2002, 22, 5027–5035. [Google Scholar] [CrossRef] [PubMed]
- Lipschitz, D.L.; Crowley, W.R.; Bealer, S.L. Differential sensitivity of intranuclear and systemic oxytocin release to central noradrenergic receptor stimulation during mid-and late gestation in rats. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E523–E528. [Google Scholar] [CrossRef]
- McCormack, S.E.; Blevins, J.E.; Lawson, E.A. Metabolic effects of oxytocin. Endocr. Rev. 2020, 41, 121–145. [Google Scholar] [CrossRef] [PubMed]
- Plante, E.; Menaouar, A.; Danalache, B.A.; Yip, D.; Broderick, T.L.; Chiasson, J.L.; Jankowski, M.; Gutkowska, J. Oxytocin treatment prevents the cardiomyopathy observed in obese diabetic male db/db mice. Endocrinology 2015, 156, 1416–1428. [Google Scholar] [CrossRef]
- Elabd, C.; Cousin, W.; Upadhyayula, P.; Chen, R.Y.; Chooljian, M.S.; Li, J.; Kung, S.; Jiang, K.P.; Conboy, I.M. Oxytocin is an age-specific circulating hormone that is necessary for muscle maintenance and regeneration. Nat. Commun. 2014, 5, 4082. [Google Scholar] [CrossRef]
- Ludwig, M. Dendritic release of vasopressin and oxytocin. J. Neuroendocr. 1998, 10, 881–895. [Google Scholar] [CrossRef]
- Knobloch, H.S.; Charlet, A.; Hoffmann, L.C.; Eliava, M.; Khrulev, S.; Cetin, A.H.; Osten, P.; Schwarz, M.K.; Seeburg, P.H.; Stoop, R.; et al. Evoked axonal oxytocin release in the central amygdala attenuates fear response. Neuron 2012, 73, 553–566. [Google Scholar] [CrossRef]
- Balthasar, N.; Dalgaard, L.T.; Lee, C.E.; Yu, J.; Funahashi, H.; Williams, T.; Ferreira, M.; Tang, V.; McGovern, R.A.; Kenny, C.D.; et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 2005, 123, 493–505. [Google Scholar] [CrossRef]
- Song, A.J.; Richard, D.; Palmiter, R.D. Detecting and avoiding problems when using the cre-lox system. Trends Genet. 2018, 34, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Xi, D.; Long, C.; Lai, M.; Casella, A.; O’Lear, L.; Kublaoui, B.; Roizen, J.D. Ablation of oxytocin neurons causes a deficit in cold stress response. J. Endocr. Soc. 2017, 1, 1041–1055. [Google Scholar] [CrossRef] [PubMed]
- Bachman, E.S.; Dhillon, H.; Zhang, C.Y.; Cinti, S.; Bianco, A.C.; Kobilka, B.K.; Lowell, B.B. BetaAR Signaling required for diet-induced thermogenesis and obesity resistance. Science 2002, 297, 843–845. [Google Scholar] [CrossRef]
- Ueta, C.B.; Fernandes, G.W.; Capelo, L.P.; Fonseca, T.L.; D’Angelo Maculan, F.; Gouveia, C.H.A.; Brum, P.C.; Christoffolete, M.A.; Aoki, M.S.; Lancellotti, C.L.; et al. β(1) Adrenergic receptor is key to cold- and diet-induced thermogenesis in mice. J. Endocrinol. 2012, 214, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Xu, Y.; Zhu, Y.; Sutton, A.K.; Zhao, R.; Lowell, B.B.; Olson, D.P.; Tong, Q. An obligate role of oxytocin neurons in diet induced energy expenditure. PLoS ONE 2012, 7, e45167. [Google Scholar] [CrossRef]
- Kasahara, Y.; Takayanagi, Y.; Kawada, T.; Itoi, K.; Nishimori, K. Impaired thermoregulatory ability of oxytocin-deficient mice during cold-exposure. Biosci. Biotechnol. Biochem. 2007, 71, 3122–3126. [Google Scholar] [CrossRef]
- Kasahara, Y.; Sato, K.; Takayanagi, Y.; Mizukami, H.; Ozawa, K.; Hidema, S.; So, K.-H.; Kawada, T.; Inoue, N.; Ikeda, I.; et al. Oxytocin receptor in the hypothalamus is sufficient to rescue normal thermoregulatory function in male oxytocin receptor knockout mice. Endocrinology 2013, 154, 4305–4315. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Li, X.; Cano, G.; Lazarus, M.; Saper, C.B. Parallel preoptic pathways for thermoregulation. J. Neurosci. 2009, 29, 11954–11964. [Google Scholar] [CrossRef]
- Peterson, C.M.; Lecoultre, V.; Frost, E.A.; Simmons, J.; Redman, L.M.; Ravussin, E. The thermogenic responses to overfeeding and cold are differentially regulated. Obesity 2016, 24, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, B.J.; Hansson, S.R.; Mezey, E.; Palkovits, M. Localization and dynamic regulation of biogenic amine transporters in the mammalian central nervous system. Front. Neuroendocr. 1998, 19, 187–231. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P. Origins and early development of the concept that brown adipose tissue thermogenesis is linked to energy balance and obesity. Biochimie 2017, 134, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Camerino, C. Oxytocin thinks globally and acts locally: The oxytocinergic regulation of bone mass. IBMS BoneKEy 2009, 6, 295–300. [Google Scholar] [CrossRef]
- Mosialou, I.; Shikhel, S.; Liu, J.M.; Maurizi, A.; Luo, N.; He, Z.; Huang, Y.; Zong, H.; Friedman, R.A.; Barasch, J.; et al. MC4R-dependent suppression of appetite by bone-derived lipocalin 2. Nature 2017, 543, 385–390. [Google Scholar] [CrossRef]
- Zhang, G.; Bai, H.; Zhang, H.; Dean, C.; Wu, Q.; Li, J.; Guariglia, S.; Meng, Q.; Cai, D. Neuropeptide exocytosis involving synaptotagmin-4 and oxytocin in hypothalamic programming of body weight and energy balance. Neuron 2011, 69, 523–535. [Google Scholar] [CrossRef]
- Arletti, R.; Benelli, A.; Bertolini, A. Influence of oxytocin on feeding behavior in the rat. Peptides 1989, 10, 89–93. [Google Scholar] [CrossRef]
- Stock, S.; Granström, L.; Backman, L.; Matthiesen, A.S.; Uvnäs-Moberg, K. Elevated plasma levels of oxytocin in obese subjects before and after gastric banding. Int. J. Obes. 1989, 13, 213–222. [Google Scholar]
- Lawson, E.A.; Donoho, D.A.; Blum, J.I.; Meenaghan, E.M.; Misra, M.; Herzog, D.B.; Sluss, P.M.; Miller, K.K.; Klibanski, A. Decreased nocturnal oxytocin levels in anorexia nervosa are associated with low bone mineral density and fat mass. J. Clin. Psychiatry 2011, 72, 1546–1551. [Google Scholar] [CrossRef]
- Monod, J.; Jacob, F. Teleonomic mechanisms in cellular metabolism, growth, and differentiation. Cold Spring Harb. Symp. Quant. Biol. 1961, 26, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Abboud, F.M. The Walter, B. Cannon Memorial Award Lecture. Physiology in perspective: The wisdom of the body. In search of autonomic balance: The good, the bad, and the ugly. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1449–R1467. [Google Scholar] [CrossRef] [PubMed]
- Pette, D.; Staron, R.S. Cellular and molecular diversities of mammalian skeletal muscle fibers. Rev. Physiol. Biochem. Pharm. 1990, 116, 1–76. [Google Scholar]
- Mizunoya, W.; Iwamoto, Y.; Sato, Y.; Tatsumi, R.; Ikeuchi, Y. Cold exposure increases slow-type myosin heavy chain 1 (MyHC1) composition of soleus muscle in rats. Anim. Sci. J. 2014, 85, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F.; Clegg, D.J. Non-shivering thermogenesis as a mechanism to facilitate sustainable weight loss. Obes. Rev. 2017, 18, 819–831. [Google Scholar] [CrossRef] [PubMed]
- Blondin, D.P.; Haman, F. Shivering and nonshivering thermogenesis in skeletal muscles. Handb. Clin. Neurol. 2018, 156, 153–173. [Google Scholar]
- Zierath, J.R.; Hawley, J.A. Skeletal muscle fiber type: Influence on contractile and metabolic properties. PLoS Biol. 2004, 2, e348. [Google Scholar] [CrossRef]
- Termin, A.; Staron, R.S.; Pette, D. Myosin heavy chain isoforms in histochemically defined fiber types of rat muscle. Histochemistry 1989, 92, 453–457. [Google Scholar] [CrossRef]
- Rivero, J.L.; Talmadge, R.J.; Edgerton, V.R. Fibre size and metabolic properties of myosin heavy chain-based fibre types in rat skeletal muscle. J. Muscle. Res. Cell. Motil. 1998, 19, 733–742. [Google Scholar] [CrossRef]
- Rivero, J.L.; Serrano, A.L.; Barrey, E.; Valette, J.P.; Jouglin, M. Analysis of myosin heavy chains at the protein level in horse skeletal muscle. J. Muscle. Res. Cell. Motil. 1999, 20, 211–221. [Google Scholar] [CrossRef]
- De Jager, N.; Hudson, N.J.; Reverter, A.; Wang, Y.H.; Nagaraj, S.H.; Cafe, L.M.; Greenwood, P.L.; Barnard, R.T.; Kongsuwan, K.P.; Dalrymple, B.P. Chronic exposure to anabolic steroids induces the muscle expression of oxytocin and a more than fiftyfold increase in circulating oxytocin in cattle. Physiol. Genom. 2011, 43, 467–478. [Google Scholar] [CrossRef][Green Version]
- Arrowsmith, S.; Quenby, S.; Weeks, A.; Burdyga, T.; Wray, S. Poor spontaneous and oxytocin-stimulated contractility in human myometrium from postdates pregnancies. PLoS ONE 2012, 7, e36787. [Google Scholar] [CrossRef]
- Xu, B.; Lang, L.M.; Lian, S.; Guo, J.R.; Wang, J.F.; Liu, J.; Yang, H.M.; Li, S.Z. Neuroinflammation induced by secretion of acetylated HMGB1 from activated microglia in hippocampi of mice following chronic cold exposure. Brain Res. 2020, 1726, 146495. [Google Scholar] [CrossRef]
- Xu, B.; Lian, S.; Guo, J.R.; Wang, J.F.; Zhang, L.P.; Li, S.Z.; Yang, H.M. Activation of the MAPK signaling pathway induces upregulation of pro-apoptotic proteins in the hippocampi of cold stressed adolescent mice. Neurosci. Lett. 2019, 699, 97–102. [Google Scholar] [CrossRef]
- Dumitru, A.; Radu, B.M.; Radu, M.; Cretoiu, S.M. Muscle changes during atrophy. Adv. Exp. Med. Biol. 2018, 1088, 73–92. [Google Scholar] [PubMed]
- Gajdosechova, L.; Krskova, K.; Olszanecki, R.; Zorad, S. Differential regulation of oxytocin receptor in various adipose tissue depots and skeletal muscle types in obese zucker rats. Horm. Metab. Res. 2015, 47, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Nersesyan, Y.; Demirkhanyan, L.; Cabezas-Bratesco, D.; Oakes, V.; Kusuda, R.; Dawson, T.; Sun, X.; Cao, C.; Cohen, A.M.; Chelluboina, B.; et al. Oxytocin modulates nociception as an agonist of pain-sensing TRPV1. Cell Rep. 2017, 21, 1681–1691. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Hernandez, A.; Charlet, A. Oxytocin, GABA, and TRPV1, the analgesic triad? Front. Mol. Neurosci. 2018, 11, 398. [Google Scholar] [CrossRef]
- Yuan, Z.; McCauley, R.; Chen-Scarabelli, C.; Abounit, K.; Stephanou, A.; Barry, S.P.; Knight, R.; Saravolatz, S.F.; Saravolatz, L.D.; Ulgen, B.O.; et al. Activation of src protein tyrosine kinase plays an essential role in urocortin-mediated cardioprotection. Mol. Cell. Endocrinol. 2010, 325, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Breton, C.; Haenggeli, C.; Barberis, C.; Heitz, F.; Bader, C.R.; Bernheim, L.; Tribollet, E. Presence of functional oxytocin receptors in cultured human myoblasts. J. Clin. Endocrinol. Metab. 2002, 87, 1415–1418. [Google Scholar] [CrossRef]
- Lee, E.S.; Uhm, K.O.; Lee, Y.M.; Kwon, J.; Park, S.H.; Kim Soo, K.H. Oxytocin stimulates glucose uptake in skeletal muscle cells through the calcium-CaMKK-AMPK pathway. Regul. Pept. 2008, 151, 71–74. [Google Scholar] [CrossRef]
- Gajdosechova, L.; Krskova, K.; Segarra, A.B.; Spolcova, A.; Suski, M.; Olszanecki, R.; Zorad, S. Hypooxytocinaemia in obese zucker rats relates to oxytocin degradation in liver and adipose tissue. J. Endocrinol. 2014, 220, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Işeri, S.O.; Sener, G.; Sağlam, B.; Gedik, N.; Ercan, F.; Yeğen, B.C. Oxytocin ameliorates oxidative colonic inflammation by a neutrophil-dependent mechanism. Peptides 2005, 26, 483–491. [Google Scholar] [CrossRef]
- Monstein, H.J.; Grahn, N.; Truedsson, M.; Ohlsson, B. Oxytocin and oxytocin-receptor mRNA expression in the human gastrointestinal tract: A polymerase chain reaction study. Regul. Pept. 2004, 119, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, B.; Truedsson, M.; Djerf, P.; Sundler, F. oxytocin is expressed throughout the human gastrointestinal tract. Regul. Pept. 2006, 135, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, B.; Truedsson, M.; Bengtsson, M.; Torstenson, R.; Sjölund, K.; Björnsson, E.S.; Simrèn, M. Effects of long-term treatment with oxytocin in chronic constipation; a double blind, placebo-controlled pilot trial. Neurogastroenterol. Motil. 2005, 17, 697–704. [Google Scholar] [CrossRef]
- Ohlsson, B.; Björgell, O.; Ekberg, O.; Darwiche, G. The oxytocin/vasopressin receptor antagonist atosiban delays the gastric emptying of a semisolid meal compared to saline in human. BMC Gastroenterol. 2006, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Hashmonai, M.; Torem, S.; Argov, S.; Barzilai, A.; Schramek, A. Prolonged post-vagotomy gastric atony treated by oxytocin. Br. J. Surg. 1979, 66, 550–551. [Google Scholar] [CrossRef]
- Petring, O.U. The effect of oxytocin on basal and pethidine-induced delayed gastric emptying. Br. J. Clin. Pharm. 1989, 28, 329–332. [Google Scholar] [CrossRef]
- Wu, C.L.; Hung, C.R.; Chang, F.Y.; Pau, K.Y.; Wang, J.L.; Wang, P.S. Involvement of cholecystokinin receptor in the inhibition of gastric emptying by oxytocin in male rats. Pflug. Arch. 2002, 445, 187–193. [Google Scholar] [CrossRef]
- Wu, C.L.; Hung, C.R.; Chang, F.Y.; Pau, K.Y.; Wang, P.S. Pharmacological effects of oxytocin on gastric emptying and intestinal transit of a non-nutritive liquid meal in female rats. Naunyn Schmiedebergs Arch. Pharmacol. 2003, 367, 406–413. [Google Scholar] [CrossRef]
- Louvel, D.; Delvaux, M.; Felez, A.; Fioramonti, J.; Bueno, L.; Lazorthes, Y.; Frexinos, J. Oxytocin increases thresholds of colonic visceral perception in patients with irritable bowel syndrome. Gut 1996, 39, 741–747. [Google Scholar] [CrossRef]
- Spiller, R. Pharmacotherapy: Non-serotonergic mechanisms. Gut 2002, 51 (Suppl. 1), i87–i90. [Google Scholar] [CrossRef]
- Welch, M.G.; Tamir, H.; Gross, K.J.; Chen, J.; Anwar, M.; Gershon, M.D. Expression and developmental regulation of oxytocin (OT) and oxytocin receptors (OTR) in the enteric nervous system (ENS) and intestinal epithelium. J. Comp. Neurol. 2009, 512, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Che, T.; Sun, H.; Li, J.; Yu, X.; Zhu, D.; Xue, B.; Liu, K.; Zhang, M.; Kunze, W.; Liu, C. Oxytocin hyperpolarizes cultured duodenum myenteric intrinsic primary afferent neurons by opening BK(Ca) channels through IP₃ pathway. J. Neurochem. 2012, 121, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Chen, L.; Liu, C.; Liu, K. The inhibitory effects of oxytocin on distal colonic contractile activity in rabbits are enhanced by ovarian steroids. Acta Physiol. 2006, 186, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Qin, J.; Wang, C.; Ye, Y.; Wang, S.; Xie, D.; Wang, P.S.; Liu, C. Estradiol upregulates the expression of oxytocin receptor in colon in rats. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E1059–E1066. [Google Scholar] [CrossRef] [PubMed]
- Blevins, J.E.; Ho, J.M. Role of oxytocin signaling in the regulation of body weight. Rev. Endocr. Metab. Disord. 2013, 14, 311–329. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Zhu, T.; Tang, B.; Yu, S.; Hu, H.; Sun, W.; Pan, R.; Wang, J.; Wang, D.; Yang, L.; et al. Decreased circulating levels of oxytocin in obesity and newly diagnosed type 2 diabetic patients. J. Clin. Endocrinol. Metab. 2014, 99, 4683–4689. [Google Scholar] [CrossRef] [PubMed]
- Binay, C.; Paketçi, C.; Güzel, S.; Samancı, N. Serum Irisin and oxytocin levels as predictors of metabolic parameters in obese children. J. Clin. Res. Pediatr. Endocrinol. 2017, 9, 124–131. [Google Scholar] [CrossRef]
- Lawson, E.A.; Ackerman, K.E.; Slattery, M.; Marengi, D.A.; Clarke, H.; Misra, M. Oxytocin secretion is related to measures of energy homeostasis in young amenorrheic athletes. J. Clin. Endocrinol. Metab. 2014, 99, E881–E885. [Google Scholar] [CrossRef] [PubMed]
- Aulinas, A.; Pulumo, R.L.; Asanza, E.; Mancuso, C.J.; Slattery, M.; Tolley, C.; Plessow, F.; Thomas, J.J.; Eddy, K.T.; Miller, K.K.; et al. Endogenous oxytocin levels in relation to food intake, menstrual phase, and age in females. J. Clin. Endocrinol. Metab. 2019, 104, 1348–1356. [Google Scholar] [CrossRef] [PubMed]
- Lawson, E.A.; Ackerman, K.E.; Estella, N.M.; Guereca, G.; Pierce, L.; Sluss, P.M.; Bouxsein, M.L.; Klibanski, A.; Misra, M. Nocturnal oxytocin secretion is lower in amenorrheic athletes than nonathletes and associated with bone microarchitecture and finite element analysis parameters. Eur. J. Endocrinol. 2013, 168, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Camerino, C. Oxytocin inhibits bone formation through the activation of the sympathetic tone: A new candidate in the central regulation of bone formation. J. Bone Min. Res. 2008, 23 (Suppl. 1), S56. [Google Scholar]
- Hew-Butler, T.; Jordaan, E.; Stuempfle, K.J.; Speedy, D.B.; Siegel, A.J.; Noakes, T.D.; Soldin, S.J.; Verbalis, J.G. Osmotic and nonosmotic regulation of arginine vasopressin during prolonged endurance exercise. J. Clin. Endocrinol. Metab. 2008, 93, 2072–2078. [Google Scholar] [CrossRef] [PubMed]
- Hew-Butler, T.; Noakes, T.D.; Soldin, S.J.; Verbalis, J.G. Acute changes in endocrine and fluid balance markers during high-intensity, steady-state, and prolonged endurance running: Unexpected increases in oxytocin and brain natriuretic peptide during exercise. Eur. J. Endocrinol. 2008, 159, 729–737. [Google Scholar] [CrossRef]
- Chicharro, J.L.; Hoyos, J.; Bandrés, F.; Gómez Gallego, F.; Pérez, M.; Lucía, A. Plasma oxytocin during intense exercise in professional cyclists. Horm. Res. 2001, 55, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Bittel, D.C.; Butler, M.G. Prader-Willi syndrome: Clinical genetics, cytogenetics and molecular biology. Expert Rev. Mol. Med. 2005, 7, 1–20. [Google Scholar] [CrossRef]
- Johnson, L.; Manzardo, A.M.; Miller, J.L.; Driscoll, D.J.; Butler, M.G. Elevated plasma oxytocin levels in children with Prader-Willi syndrome compared with healthy unrelated siblings. Am. J. Med. Genet. A 2016, 170, 594–601. [Google Scholar] [CrossRef]
- Swaab, D.F.; Purba, J.S.; Hofman, M.A. Alterations in the hypothalamic paraventricular nucleus and its oxytocin neurons (putative satiety cells) in Prader-Willi syndrome: A study of five cases. J. Clin. Endocrinol. Metab. 1995, 80, 573–579. [Google Scholar]
- Butler, M.G. Prader-Willi syndrome: Obesity due to genomic imprinting. Curr. Genom. 2011, 12, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.G. Prader-Willi syndrome: Current understanding of cause and diagnosis. Am. J. Med. Genet. 1990, 35, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Carrel, A.L.; Moerchen, V.; Myers, S.E.; Bekx, M.T.; Whitman, B.Y.; Allen, D.B. Growth hormone improves mobility and body composition in infants and toddlers with Prader-Willi syndrome. J. Pediatr. 2004, 145, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.G. Management of obesity in Prader-Willi syndrome. Nat. Clin. Pr. Endocrinol. Metab. 2006, 2, 592–593. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; State, M.; Anderson, G.M.; Kaye, W.M.; Hanchett, J.M.; McConaha, C.W.; North, W.G.; Leckman, J.F. Cerebrospinal fluid levels of oxytocin in Prader-Willi syndrome: A preliminary report. Biol. Psychiatry 1998, 44, 1349–1352. [Google Scholar] [CrossRef]
- Miller, M.; Bales, K.L.; Taylor, S.L.; Yoon, J.; Hostetler, C.M.; Carter, C.S.; Solomon, M. Oxytocin and vasopressin in children and adolescents with autism spectrum disorders: Sex differences and associations with symptoms. Autism Res. 2013, 6, 91–102. [Google Scholar] [CrossRef]
- Carson, D.S.; Berquist, S.W.; Trujillo, T.H.; Garner, J.P.; Hannah, S.L.; Hyde, S.A.; Sumiyoshi, R.D.; Jackson, L.P.; Moss, J.K.; Strehlow, M.C.; et al. Cerebrospinal fluid and plasma oxytocin concentrations are positively correlated and negatively predict anxiety in children. Mol. Psychiatry 2015, 20, 1085–1090. [Google Scholar] [CrossRef]
- Rice, L.J.; Einfeld, S.L.; Hu, N.; Carter, C.S. A review of clinical trials of oxytocin in Prader-Willi syndrome. Curr. Opin. Psychiatry 2018, 31, 123–127. [Google Scholar] [CrossRef]
- Muscatelli, F.; Abrous, D.N.; Massacrier, A.; Boccaccio, I.; Le Moal, M.; Cau, P.; Cremer, H. Disruption of the mouse necdin gene results in hypothalamic and behavioral alterations reminiscent of the human Prader-Willi syndrome. Hum. Mol. Genet. 2000, 9, 3101–3110. [Google Scholar] [CrossRef]
- Schaller, F.; Watrin, F.; Sturny, R.; Massacrier, A.; Szepetowski, P.; Muscatelli, F. A single postnatal injection of oxytocin rescues the lethal feeding behaviour in mouse newborns deficient for the imprinted magel2 gene. Hum. Mol. Genet. 2010, 19, 4895–4905. [Google Scholar] [CrossRef]
- Burnett, L.C.; LeDuc, C.A.; Sulsona, C.R.; Paull, D.; Rausch, R.; Eddiry, S.; Carli, J.F.M.; Morabito, M.V.; Skowronski, A.A.; Hubner, G.; et al. Deficiency in prohormone convertase pc1 impairs prohormone processing in Prader-Willi syndrome. J. Clin. Investig. 2017, 127, 293–305. [Google Scholar] [CrossRef]
- Bittel, D.C.; Kibiryeva, N.; Sell, S.M.; Strong, T.V.; Butler, M.G. Whole genome microarray analysis of gene expression in Prader-Willi syndrome. Am. J. Med. Genet. A 2007, 143A, 430–442. [Google Scholar] [CrossRef]
- Miller, J.L.; Tamura, R.; Butler, M.G.; Kimonis, V.; Sulsona, C.; Gold, J.A.; Driscoll, D.J. Oxytocin treatment in children with Prader-Willi syndrome: A double-blind, placebo-controlled, crossover study. Am. J. Med. Genet. A 2017, 173, 1243–1250. [Google Scholar] [CrossRef]
- Huang, H.; Michetti, C.; Busnelli, M.; Managò, F.; Sannino, S.; Scheggia, D.; Giancardo, L.; Sona, D.; Murino, V.; Chini, B.; et al. Chronic and acute intranasal oxytocin produce divergent social effects in mice. Neuropsychopharmacology 2014, 39, 1102–1114. [Google Scholar] [CrossRef] [PubMed]
- Passoni, I.; Leonzino, M.; Gigliucci, V.; Chini, B.; Busnelli, M. Carbetocin is a functional selective Gq agonist that does not promote oxytocin receptor recycling after inducing β-arrestin-independent internalisation. J. Neuroendocr. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Green, L.; Fein, D.; Modahl, C.; Feinstein, C.; Waterhouse, L.; Morris, M. Oxytocin and autistic disorder: Alterations in peptide forms. Biol. Psychiatry 2001, 50, 609–613. [Google Scholar] [CrossRef]
- Hollander, E. Social synchrony and oxytocin: From behavior to genes to therapeutics. Am. J. Psychiatry 2013, 170, 1086–1089. [Google Scholar] [CrossRef]
- Kabasakalian, A.; Ferretti, C.J.; Hollander, E. Oxytocin and Prader-Willi syndrome. Curr. Top. Behav. Neurosci. 2018, 35, 529–557. [Google Scholar] [PubMed]
- Levy, S.E.; Mandell, D.S.; Schultz, R.T. Autism. Lancet 2009, 374, 1627–1638. [Google Scholar] [CrossRef]
- Boccaccio, I.; Glatt-Deeley, H.; Watrin, F.; Roëckel, N.; Lalande, M.; Muscatelli, F. The human MAGEL2 gene and its mouse homologue are paternally expressed and mapped to the Prader-Willi region. Hum. Mol. Genet. 1999, 8, 2497–2505. [Google Scholar] [CrossRef] [PubMed]
- Jay, P.; Rougeulle, C.; Massacrier, A.; Moncla, A.; Mattei, M.G.; Malzac, P.; Roëckel, N.; Taviaux, S.; Lefranc, J.L.; Cau, P.; et al. The human necdin gene, NDN, is maternally imprinted and located in the Prader-Willi syndrome chromosomal region. Nat. Genet. 1997, 17, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Gao, J.; Yang, M. When MAGE meets RING: Insights into biological functions of MAGE proteins. Protein Cell 2011, 2, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Chomez, P.; De Backer, O.; Bertrand, M.; De Plaen, E.; Boon, T.; Lucas, S. An Overview of the MAGE gene family with the identification of all human members of the family. Cancer Res. 2001, 61, 5544–5551. [Google Scholar] [PubMed]
- Bertrand, M.; Huijbers, I.; Chomez, P.; De Backer, O. Comparative expression analysis of the MAGED genes during embryogenesis and brain development. Dev. Dyn. 2004, 230, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, B.S.; Geschwind, D.H. Advances in autism genetics: On the threshold of a new neurobiology. Nat. Rev. Genet. 2008, 9, 341–355. [Google Scholar] [CrossRef]
- Mercer, R.E.; Wevrick, R. Loss of magel2, a candidate gene for features of Prader-Willi syndrome, impairs reproductive function in mice. PLoS ONE 2009, 4, e4291. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camerino, C. The New Frontier in Oxytocin Physiology: The Oxytonic Contraction. Int. J. Mol. Sci. 2020, 21, 5144. https://doi.org/10.3390/ijms21145144
Camerino C. The New Frontier in Oxytocin Physiology: The Oxytonic Contraction. International Journal of Molecular Sciences. 2020; 21(14):5144. https://doi.org/10.3390/ijms21145144
Chicago/Turabian StyleCamerino, Claudia. 2020. "The New Frontier in Oxytocin Physiology: The Oxytonic Contraction" International Journal of Molecular Sciences 21, no. 14: 5144. https://doi.org/10.3390/ijms21145144
APA StyleCamerino, C. (2020). The New Frontier in Oxytocin Physiology: The Oxytonic Contraction. International Journal of Molecular Sciences, 21(14), 5144. https://doi.org/10.3390/ijms21145144



