Experimental Models in Neovascular Age Related Macular Degeneration
Abstract
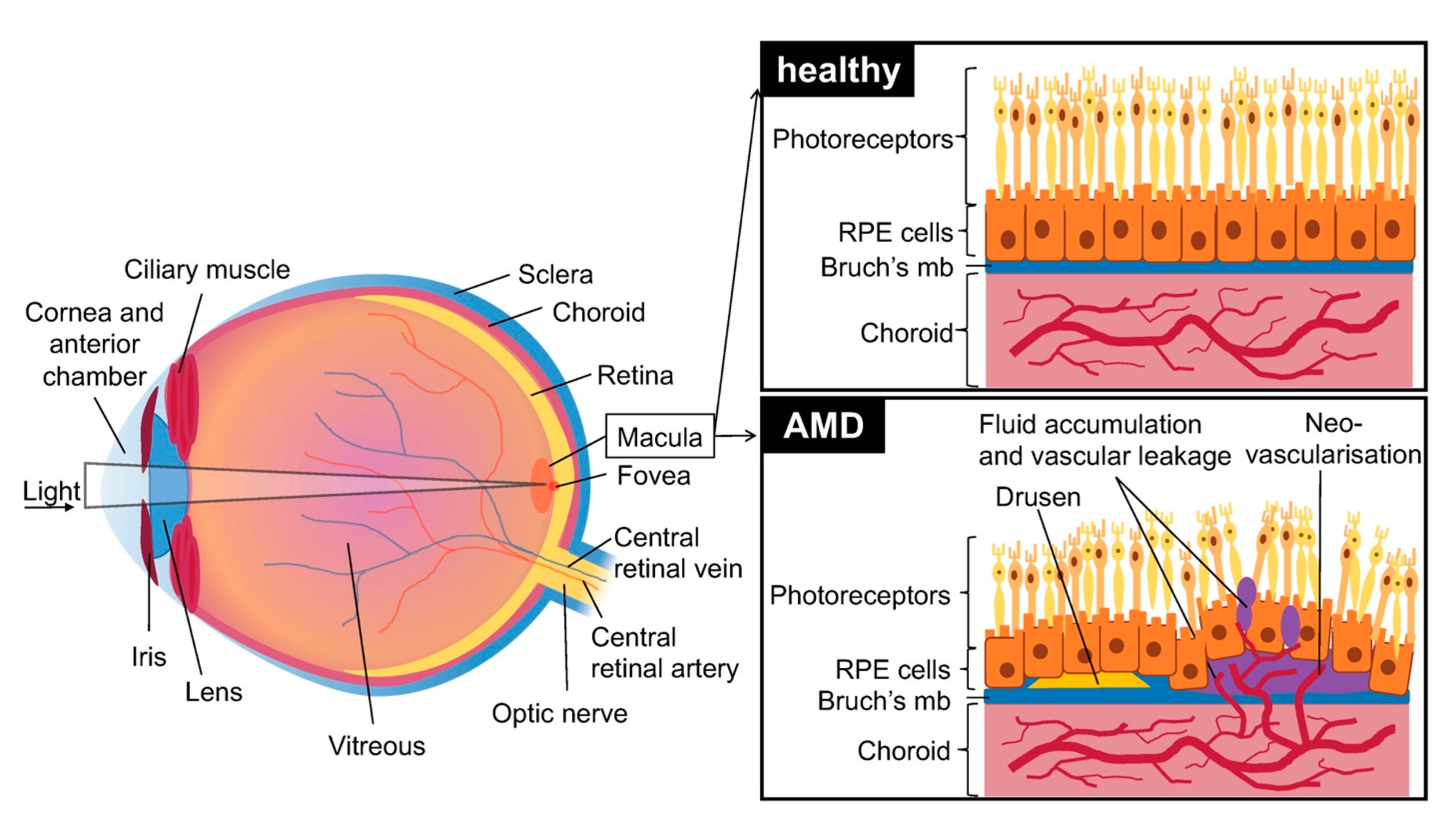
1. Introduction
2. Treatment of vAMD
3. In Vitro Models of vAMD
3.1. Human Primary RPE Cells
3.2. ARPE 19 Cells
3.3. Stem Cell Derived RPE Cells (iPSC-RPE)
3.4. Cell Cocultures and Culture Methods
3.4.1. 2D Models
3.4.2. 3D Models
3.4.3. Cocultures
3.5. In Vitro Stress Models
4. In Vivo Models of Neovascular AMD
4.1. Murine Models
4.1.1. Induced Models
4.1.2. Transgenic Models
4.2. Zebrafish Models
4.2.1. Induced Models
4.2.2. Transgenic Models
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AMD | Age-related macular degeneration |
| CNV | Choroidal neovascularization |
| PlGF | Placenta growth factor |
| RPE | Retinal pigment epithelium |
| vAMD | Neovascular age-related macular degeneration |
| VEGF | Vascular endothelial growth factor A |
References
- Gheorghe, A.; Mahdi, L.; Musat, O. Age-related macular degeneration. Rom. J. Ophthalmol. 2015, 59, 74–77. [Google Scholar] [PubMed]
- Imamura, Y.; Noda, S.; Hashizume, K.; Shinoda, K.; Yamaguchi, M.; Uchiyama, S.; Shimizu, T.; Mizushima, Y.; Shirasawa, T.; Tsubota, K. Drusen, choroidal neovascularization, and retinal pigment epithelium dysfunction in SOD1-deficient mice: A model of age-related macular degeneration. Proc. Natl. Acad. Sci. USA 2006, 103, 11282–11287. [Google Scholar] [CrossRef] [PubMed]
- Kaarniranta, K.; Tokarz, P.; Koskela, A.; Paterno, J.; Blasiak, J. Autophagy regulates death of retinal pigment epithelium cells in age-related macular degeneration. Cell Biol. Toxicol. 2017, 33, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Fleckenstein, M.; Mitchell, P.; Freund, K.B.; Sadda, S.; Holz, F.G.; Brittain, C.; Henry, E.C.; Ferrara, D. The progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmology 2018, 125, 369–390. [Google Scholar] [CrossRef] [PubMed]
- Ferrington, D.A.; Sinha, D.; Kaarniranta, K. Defects in retinal pigment epithelial cell proteolysis and the pathology associated with age-related macular degeneration. Prog. Retin. Eye Res. 2016, 51, 69–89. [Google Scholar] [CrossRef]
- Yu, Y.; Bhangale, T.R.; Fagerness, J.; Ripke, S.; Thorleifsson, G.; Tan, P.L.; Souied, E.H.; Richardson, A.J.; Merriam, J.E.; Buitendijk, G.H.S.; et al. Common variants near FRK/COL10A1 and VEGFA are associated with advanced age-related macular degeneration. Hum. Mol. Genet. 2011, 20, 3699–3709. [Google Scholar] [CrossRef]
- Seddon, J.M.; Yu, Y.; Miller, E.C.; Reynolds, R.; Tan, P.L.; Gowrisankar, S.; Goldstein, J.I.; Triebwasser, M.; Anderson, H.E.; Zerbib, J.; et al. Rare variants in CFI, C3 and C9 are associated with high risk of advanced age-related macular degeneration. Nat. Genet. 2013, 45, 1366–1370. [Google Scholar] [CrossRef]
- van de Ven, J.P.H.; Nilsson, S.C.; Tan, P.L.; Buitendijk, G.H.S.; Ristau, T.; Mohlin, F.C.; Nabuurs, S.B.; Schoenmaker-Koller, F.E.; Smailhodzic, D.; Campochiaro, P.A.; et al. A functional variant in the CFI gene confers a high risk of age-related macular degeneration. Nat. Genet. 2013, 45, 813–817. [Google Scholar] [CrossRef]
- Al-Zamil, W.; Yassin, S. Recent developments in age-related macular degeneration: A review. Clin. Interv. Aging 2017, 12, 1313–1330. [Google Scholar] [CrossRef]
- Kodjikian, L.; Souied, E.H.; Mimoun, G.; Mauget-Faÿsse, M.; Behar-Cohen, F.; Decullier, E.; Huot, L.; Aulagner, G. Ranibizumab versus bevacizumab for neovascular age-related macular degeneration: Results from the GEFAL noninferiority randomized trial. Ophthalmology 2013, 120, 2300–2309. [Google Scholar] [CrossRef]
- Falavarjani, K.G.; Nguyen, Q.D. Adverse events and complications associated with intravitreal injection of anti-VEGF agents: A review of literature. Eye 2013, 27, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Song, Y.; Xu, G.; Ye, J.; Wu, Z.; Liu, X.; Dong, X.; Zhang, M.; Xing, Y.; Zhu, S.; et al. Conbercept for treatment of neovascular age-related macular degeneration: Results of the randomized phase 3 PHOENIX study. Am. J. Ophthalmol. 2019, 197, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Hubschman, J.P.; Reddy, S.; Schwartz, S.D. Age-related macular degeneration: Current treatments. Clin. Ophthalmol. 2009, 3, 155–166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wei, X.; Zhang, T.; Yao, Y.; Zeng, S.; Li, M.; Xiang, H.; Zhao, C.; Cao, G.; Li, M.; Wan, R.; et al. Efficacy of Lenvatinib, a multitargeted tyrosine kinase inhibitor, on laser-induced CNV mouse model of neovascular AMD. Exp. Eye Res. 2018, 168, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Djigo, A.D.; Bérubé, J.; Landreville, S.; Proulx, S. Characterization of a tissue-engineered choroid. Acta Biomater. 2019, 84, 305–316. [Google Scholar] [CrossRef]
- Adijanto, J.; Philp, N.J. Cultured primary human fetal retinal pigment epithelium (hfRPE) as a model for evaluating RPE metabolism. Exp. Eye Res. 2014, 126, 77–84. [Google Scholar] [CrossRef]
- Gamm, D.M.; Melvan, J.N.; Shearer, R.L.; Pinilla, I.; Sabat, G.; Svendsen, C.N.; Wright, L.S. A novel serum-free method for culturing human prenatal retinal pigment epithelial cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 788–799. [Google Scholar] [CrossRef]
- Blenkinsop, T.A.; Salero, E.; Stern, J.H.; Temple, S. The culture and maintenance of functional retinal pigment epithelial monolayers from adult human eye. Methods Mol. Biol. 2013, 945, 45–65. [Google Scholar] [CrossRef]
- Samuel, W.; Jaworski, C.; Postnikova, O.A.; Kutty, R.K.; Duncan, T.; Tan, L.X.; Poliakov, E.; Lakkaraju, A.; Redmond, T.M. Appropriately differentiated ARPE-19 cells regain phenotype and gene expression profiles similar to those of native RPE cells. Mol. Vis. 2017, 23, 60–89. [Google Scholar]
- Dunn, K.C.; Aotaki-Keen, A.E.; Putkey, F.R.; Hjelmeland, L.M. ARPE-19, A human retinal pigment epithelial cell line with differentiated properties. Exp. Eye Res. 1996, 62, 155–170. [Google Scholar] [CrossRef]
- Maruotti, J.; Sripathi, S.R.; Bharti, K.; Fuller, J.; Wahlin, K.J.; Ranganathan, V.; Sluch, V.M.; Berlinicke, C.A.; Davis, J.; Kim, C.; et al. Small-molecule–directed, efficient generation of retinal pigment epithelium from human pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2015, 112, 10950–10955. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, D.E.; Pennington, B.O.; Croze, R.H.; Hinman, C.R.; Coffey, P.J.; Clegg, D.O. Rapid and efficient directed differentiation of human pluripotent stem cells into retinal pigmented epithelium. Stem Cells Transl. Med. 2013, 2, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.N.; D’Antonio-Chronowska, A.; Greenwald, W.W.; Borja, V.; Aguiar, L.R.; Pogue, R.; Matsui, H.; Benaglio, P.; Borooah, S.; D’Antonio, M.; et al. Human iPSC-derived retinal pigment epithelium: A model system for prioritizing and functionally characterizing causal variants at AMD risk loci. Stem Cell Rep. 2019, 12, 1342–1353. [Google Scholar] [CrossRef] [PubMed]
- Golestaneh, N.; Chu, Y.; Cheng, S.K.; Cao, H.; Poliakov, E.; Berinstein, D.M. Repressed SIRT1/PGC-1α pathway and mitochondrial disintegration in iPSC-derived RPE disease model of age-related macular degeneration. J. Transl. Med. 2016, 14, 344. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Cai, H.; Noggle, S.; Paull, D.; Rizzolo, L.J.; Del Priore, L.V.; Fields, M.A.; NYSCF Global Stem Cell Array Team. Stem cell-derived retinal pigment epithelium from patients with age-related macular degeneration exhibit reduced metabolism and matrix interactions. Stem Cells Transl. Med. 2020, 9, 364–376. [Google Scholar] [CrossRef]
- Murphy, A.R.; Truong, Y.B.; O’Brien, C.M.; Glattauer, V. Bio-inspired human in vitro outer retinal models: Bruch’s membrane and its cellular interactions. Acta Biomater. 2020, 104, 1–16. [Google Scholar] [CrossRef]
- Malek, G.; Johnson, L.V.; Mace, B.E.; Saloupis, P.; Schmechel, D.E.; Rickman, D.W.; Toth, C.A.; Sullivan, P.M.; Rickman, C.B. Apolipoprotein E allele-dependent pathogenesis: A model for age-related retinal degeneration. Proc. Natl. Acad. Sci. USA 2005, 102, 11900–11905. [Google Scholar] [CrossRef] [PubMed]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Vemuganti, G.; Singhal, S. Primary adult human retinal pigment epithelial cell cultures on human amniotic membranes. Indian J. Ophthalmol. 2005, 53, 109. [Google Scholar] [CrossRef]
- Xiang, P.; Wu, K.-C.; Zhu, Y.; Xiang, L.; Li, C.; Chen, D.-L.; Chen, F.; Xu, G.; Wang, A.; Li, M.; et al. A novel Bruch’s membrane-mimetic electrospun substrate scaffold for human retinal pigment epithelium cells. Biomaterials 2014, 35, 9777–9788. [Google Scholar] [CrossRef]
- De Cillà, S.; Farruggio, S.; Cocomazzi, G.; Mary, D.; Alkabes, M.; Rossetti, L.; Vujosevic, S.; Grossini, E. Aflibercept and Ranibizumab Modulate Retinal Pigment Epithelial Cells Function by Acting on Their Cross Talk with Vascular Endothelial Cells. Cell Physiol. Biochem. 2020, 54, 161–179. [Google Scholar] [CrossRef]
- Jeong, H.-S.; Yun, J.-H.; Lee, D.-H.; Lee, E.H.; Cho, C.-H. Retinal pigment epithelium-derived transforming growth factor-β2 inhibits the angiogenic response of endothelial cells by decreasing vascular endothelial growth factor receptor-2 expression. J. Cell Physiol. 2019, 234, 3837–3849. [Google Scholar] [CrossRef] [PubMed]
- Leclaire, M.D.; Nettels-Hackert, G.; König, J.; Höhn, A.; Grune, T.; Uhlig, C.E.; Hansen, U.; Eter, N.; Heiduschka, P. Lipofuscin-dependent stimulation of microglial cells. Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 931–952. [Google Scholar] [CrossRef]
- Nebel, C.; Aslanidis, A.; Rashid, K.; Langmann, T. Activated microglia trigger inflammasome activation and lysosomal destabilization in human RPE cells. Biochem. Biophys. Res. Commun. 2017, 484, 681–686. [Google Scholar] [CrossRef]
- Ma, W.; Zhao, L.; Fontainhas, A.M.; Fariss, R.N.; Wong, W.T. Microglia in the mouse retina alter the structure and function of retinal pigmented epithelial cells: A potential cellular interaction relevant to AMD. PLoS ONE 2009, 4, e7945. [Google Scholar] [CrossRef]
- Jensen, C.; Teng, Y. Is it time to start transitioning from 2D to 3D cell culture? Front. Mol. Biosci. 2020, 7, 33. [Google Scholar] [CrossRef]
- Shokoohmand, A.; Jeon, J.E.; Theodoropoulos, C.; Baldwin, J.G.; Hutmacher, D.W.; Feigl, B. A novel 3D cultured model for studying early changes in age-related macular degeneration. Macromol. Biosci. 2017, 17, 1700221. [Google Scholar] [CrossRef]
- Giddabasappa, A.; Lalwani, K.; Norberg, R.; Gukasyan, H.J.; Paterson, D.; Schachar, R.A.; Rittenhouse, K.; Klamerus, K.; Mosyak, L.; Eswaraka, J. Axitinib inhibits retinal and choroidal neovascularization in in vitro and in vivo models. Exp. Eye Res. 2016, 145, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Kernt, M.; Neubauer, A.S.; Liegl, R.G.; Hirneiss, C.; Alge, C.S.; Wolf, A.; Ulbig, M.W.; Kampik, A. Sorafenib prevents human retinal pigment epithelium cells from light-induced overexpression of VEGF, PDGF and PlGF. Br. J. Ophthalmol. 2010, 94, 1533–1539. [Google Scholar] [CrossRef]
- Shah, R.S.; Soetikno, B.T.; Lajko, M.; Fawzi, A.A. A mouse model for laser-induced choroidal neovascularization. JoVE 2015, 53502. [Google Scholar] [CrossRef] [PubMed]
- Pennesi, M.E.; Neuringer, M.; Courtney, R.J. Animal models of age related macular degeneration. Mol. Asp. Med. 2012, 33, 487–509. [Google Scholar] [CrossRef]
- Kim, K.; Park, S.W.; Kim, J.H.; Lee, S.H.; Kim, D.; Koo, T.; Kim, K.; Kim, J.H.; Kim, J.-S. Genome surgery using Cas9 ribonucleoproteins for the treatment of age-related macular degeneration. Genome Res. 2017, 27, 419–426. [Google Scholar] [CrossRef]
- Koo, T.; Park, S.W.; Jo, D.H.; Kim, D.; Kim, J.H.; Cho, H.-Y.; Kim, J.; Kim, J.H.; Kim, J.-S. CRISPR-LbCpf1 prevents choroidal neovascularization in a mouse model of age-related macular degeneration. Nat. Commun. 2018, 9, 1855. [Google Scholar] [CrossRef] [PubMed]
- Luhmann, U.F.O.; Robbie, S.; Munro, P.M.G.; Barker, S.E.; Duran, Y.; Luong, V.; Fitzke, F.W.; Bainbridge, J.W.B.; Ali, R.R.; MacLaren, R.E. The drusenlike phenotype in aging Ccl2 -knockout mice Is caused by an accelerated accumulation of swollen autofluorescent subretinal macrophages. Investig. Ophthalmol. Vis. Sci. 2009, 50, 5934–5943. [Google Scholar] [CrossRef] [PubMed]
- Ambati, J.; Anand, A.; Fernandez, S.; Sakurai, E.; Lynn, B.C.; Kuziel, W.A.; Rollins, B.J.; Ambati, B.K. An animal model of age-related macular degeneration in senescent Ccl-2- or Ccr-2-deficient mice. Nat. Med. 2003, 9, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Combadière, C.; Feumi, C.; Raoul, W.; Keller, N.; Rodéro, M.; Pézard, A.; Lavalette, S.; Houssier, M.; Jonet, L.; Picard, E.; et al. CX3CR1-dependent subretinal microglia cell accumulation is associated with cardinal features of age-related macular degeneration. J. Clin. Investig. 2007, 117, 2920–2928. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Liu, Y.; Wang, L.; Li, W. Age-related macular degeneration phenotypes are associated with increased tumor necrosis-alpha and subretinal immune cells in aged Cxcr5 knockout mice. PLoS ONE 2017, 12, e0173716. [Google Scholar] [CrossRef]
- Lennikov, A.; Saddala, M.S.; Mukwaya, A.; Tang, S.; Huang, H. Autoimmune-mediated retinopathy in CXCR5-deficient mice as the result of age-related macular degeneration associated proteins accumulation. Front. Immunol. 2019, 10, 1903. [Google Scholar] [CrossRef]
- Picard, E.; Houssier, M.; Bujold, K.; Sapieha, P.; Lubell, W.; Dorfman, A.; Racine, J.; Hardy, P.; Febbraio, M.; Lachapelle, P.; et al. CD36 plays an important role in the clearance of oxLDL and associated age-dependent sub-retinal deposits. Aging 2010, 2, 981–989. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, Y.; Wang, J.; Sternberg, P.; Freeman, M.L.; Grossniklaus, H.E.; Cai, J. Age-related retinopathy in NRF2-deficient mice. PLoS ONE 2011, 6, e19456. [Google Scholar] [CrossRef]
- Huang, H.; Lennikov, A. CXCR5/NRF2 double knockout mice develop retinal degeneration phenotype at early adult age. Exp. Eye Res. 2020, 196, 108061. [Google Scholar] [CrossRef] [PubMed]
- Mattapallil, M.J.; Wawrousek, E.F.; Chan, C.-C.; Zhao, H.; Roychoudhury, J.; Ferguson, T.A.; Caspi, R.R. The Rd8 mutation of the Crb1 gene is present in vendor lines of C57BL/6N mice and embryonic stem cells, and confounds ocular induced mutant phenotypes. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2921–2927. [Google Scholar] [CrossRef]
- Vascotto, S.G.; Beckham, Y.; Kelly, G.M. The zebrafish’s swim to fame as an experimental model in biology. Biochem. Cell Biol. 1997, 75, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.; Mukwaya, A.; Biesemeier, A.; Ntzouni, M.; Ramsköld, D.; Giatrellis, S.; Mammadzada, P.; Cao, R.; Lennikov, A.; Marass, M.; et al. Intussusceptive vascular remodeling precedes pathological neovascularization. ATVB 2019, 39, 1402–1418. [Google Scholar] [CrossRef] [PubMed]
- Link, B.A.; Collery, R.F. Zebrafish models of retinal disease. Annu. Rev. Vis. Sci. 2015, 1, 125–153. [Google Scholar] [CrossRef]
- Li, L.; Zhu, M.; Wu, W.; Qin, B.; Gu, J.; Tu, Y.; Chen, J.; Liu, D.; Shi, Y.; Liu, X.; et al. Brivanib, a multitargeted small-molecule tyrosine kinase inhibitor, suppresses laser-induced CNV in a mouse model of neovascular AMD. J. Cell Physiol. 2020, 235, 1259–1273. [Google Scholar] [CrossRef]
- Cao, R.; Jensen, L.D.E.; Söll, I.; Hauptmann, G.; Cao, Y. Hypoxia-induced retinal angiogenesis in Zebrafish as a model to study retinopathy. PLoS ONE 2008, 3, e2748. [Google Scholar] [CrossRef]
- Hanovice, N.J.; Leach, L.L.; Slater, K.; Gabriel, A.E.; Romanovicz, D.; Shao, E.; Collery, R.; Burton, E.A.; Lathrop, K.L.; Link, B.A.; et al. Regeneration of the zebrafish retinal pigment epithelium after widespread genetic ablation. PLoS Genet. 2019, 15, e1007939. [Google Scholar] [CrossRef]
- Biehlmaier, O.; Neuhauss, S.C.F.; Kohler, K. Double cone dystrophy and RPE degeneration in the retina of the Zebrafish gnn Mutant. Investig. Ophthalmol. Vis. Sci. 2003, 44, 1287–1298. [Google Scholar] [CrossRef]
- Viringipurampeer, I.A.; Shan, X.; Gregory-Evans, K.; Zhang, J.P.; Mohammadi, Z.; Gregory-Evans, C.Y. Rip3 knockdown rescues photoreceptor cell death in blind pde6c zebrafish. Cell Death Differ. 2014, 21, 665–675. [Google Scholar] [CrossRef]
- van Rooijen, E.; Voest, E.E.; Logister, I.; Bussmann, J.; Korving, J.; van Eeden, F.J.; Giles, R.H.; Schulte-Merker, S. von Hippel-Lindau tumor suppressor mutants faithfully model pathological hypoxia-driven angiogenesis and vascular retinopathies in zebrafish. Dis. Models Mech. 2010, 3, 343–353. [Google Scholar] [CrossRef]
- Gerri, C.; Marín-Juez, R.; Marass, M.; Marks, A.; Maischein, H.-M.; Stainier, D.Y.R. Hif-1α regulates macrophage-endothelial interactions during blood vessel development in zebrafish. Nat. Commun. 2017, 8, 15492. [Google Scholar] [CrossRef]
- Saidi, E.A.; Davey, P.G.; Cameron, D.J. The effect of Zeaxanthin on the visual acuity of Zebrafish. PLoS ONE 2015, 10, e0135211. [Google Scholar] [CrossRef]
| Models | Advantages | Disadvantages |
|---|---|---|
| In Vitro | ||
| Cell Lines | Reduced donor-to-donor variability. Defined models with structured experimental conditions and good reproducibility. | A single cell type will not reproduce systemic defects. Does not reproduce the complexity of interactions in a living model. Cells grow unnaturally fast and gene and protein expression are often vastly different than in vivo. |
| PrimaryRPE cells | Human cells with natural differentiation. Mimic perfectly human pathology. | Can be used for only a few passages. Need to have access to this type of sample and the related authorizations |
| ARPE-19 cells | Immortalized, rapid cell growth. Exhibits similar morphology and genetic makeup to primary RPE cells. | Incomplete polarization of the cells compared to primary RPEs. |
| IPSC-RPE | Exhibits similar morphology and genetic makeup to primary RPE cells. Patient derived iPSC-RPE cells could be used in autologous cell replacement therapy. | Differentiation of the cells is time consuming and requires growth factors. |
| Cocultures | Enables cell-to-cell interactions and cross-talks, and modulation of cytokine production. More cytokines are usually produced in cocultures. | |
| 2D models | Easier to put in place than 3D models, better for long-term cultures. | Lack of sophistication, cells grow in monolayers at the same speed. Drugs are up-taken more easily than they would in vivo which is less accurate. |
| 3D models | Multi layered, the cells can self-organize. The most accurate in vitro representation of the choroid, Bruch’s membrane and RPE cells. Cells develop vascular networks and can migrate. More representative of drug exposure. Gene and protein levels closer to those in vivo. Better cell junctions. | More resource intensive, electrospun scaffolds require specialized equipment, in addition to more time and expertise. Can be difficult to replicate. |
| In Vivo | ||
| Murine Models | Most retinal degeneration genes in mice have a corresponding gene in humans, many human gene orthologs in the mice genome. They have short lifespans which enables us to see the ageing process. Protocols for genetic studies are well established. | Mice do not possess a macula. They do not produce drusen that are similar in location and composition to human drusen. Pathogenesis can differ. Late onset genetic models can lead to waiting over a year. |
| Laser induced CNV | Replicates the neovascularization in neovascular AMD, is low cost, and the CNV develops rapidly. | It is an acute injury rather than a chronic one, and thus has the inability to reproduce the complex events that lead to AMD. Risk of cataract and fibrosis if the procedure is not performed correctly. |
| Injection induced CNV | Simulates the exudative deposits and lesions in neovascular AMD. | Lower incidence of CNV than laser induced. Injections can cause tears in Bruch’s membrane. |
| Injection of adenovirus | Injection of vectors expressing VEGF have high incidence and long-term capability to induce CNV. These models work well on transgenic models. | |
| Transgenic models | Increased sensitivity to laser induced CNV, some models develop CNV with age. They are more complex models than acute induced CNV. | Most transgenic models exhibit dry AMD symptoms, such as drusen formation and retinal degeneration, and therefore will need injection or laser induced CNV to simulate neovascular AMD, making the experiment more costly and time consuming. |
| Zebrafish Models | Common features in retinal vasculature, many human orthologs in the zebrafish genome. Cost effective. Accessibility of screening and study of vascular patterning. Easy to treat. | Fewer models that replicate AMD, as zebrafish are less used than murine models. Pathogenesis can differ. |
| Hypoxia induced | Non-invasive induction of angiogenesis, easily reproducible and low cost. Can be induced in transgenic fish. | |
| Transgenic models | VHL knockout models exhibit high vascularization. These models are easy to work with as treatments are easy to deliver. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rastoin, O.; Pagès, G.; Dufies, M. Experimental Models in Neovascular Age Related Macular Degeneration. Int. J. Mol. Sci. 2020, 21, 4627. https://doi.org/10.3390/ijms21134627
Rastoin O, Pagès G, Dufies M. Experimental Models in Neovascular Age Related Macular Degeneration. International Journal of Molecular Sciences. 2020; 21(13):4627. https://doi.org/10.3390/ijms21134627
Chicago/Turabian StyleRastoin, Olivia, Gilles Pagès, and Maeva Dufies. 2020. "Experimental Models in Neovascular Age Related Macular Degeneration" International Journal of Molecular Sciences 21, no. 13: 4627. https://doi.org/10.3390/ijms21134627
APA StyleRastoin, O., Pagès, G., & Dufies, M. (2020). Experimental Models in Neovascular Age Related Macular Degeneration. International Journal of Molecular Sciences, 21(13), 4627. https://doi.org/10.3390/ijms21134627






