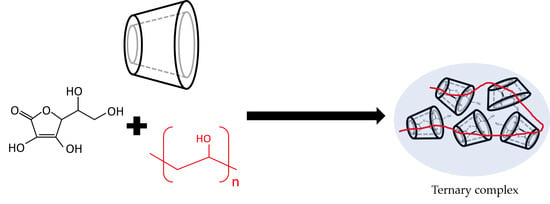
Characterization and Evaluation of Ternary Complexes of Ascorbic Acid with γ-Cyclodextrin and Poly(vinyl Alcohol)
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of Binary and Ternary Complexes
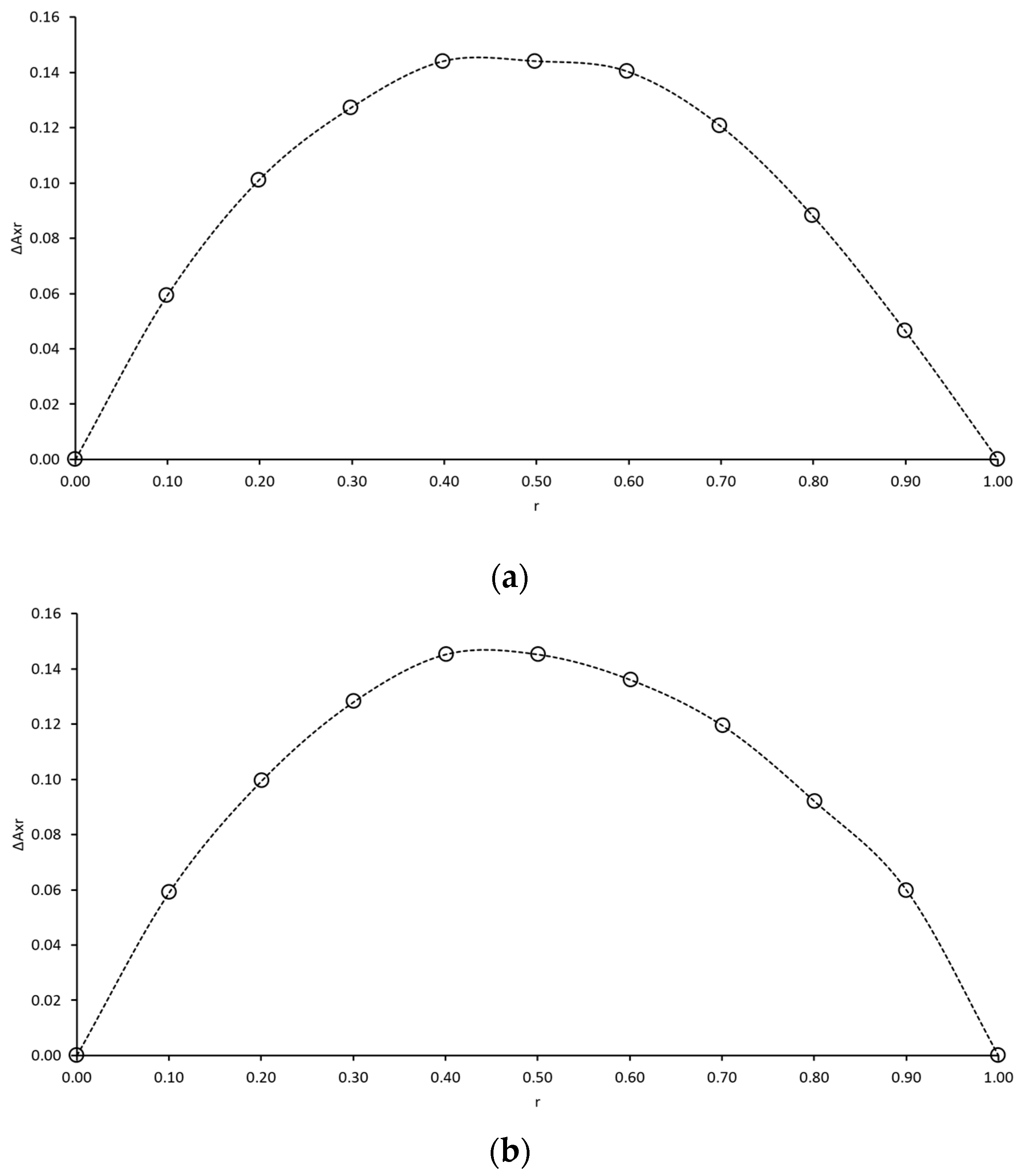
2.1.1. Stoichiometry of Ascorbic Acid Binary Complexes
2.1.2. Binary and Ternary Complexes in Solid State
2.1.3. Binary and Ternary Complexes in Aqueous State
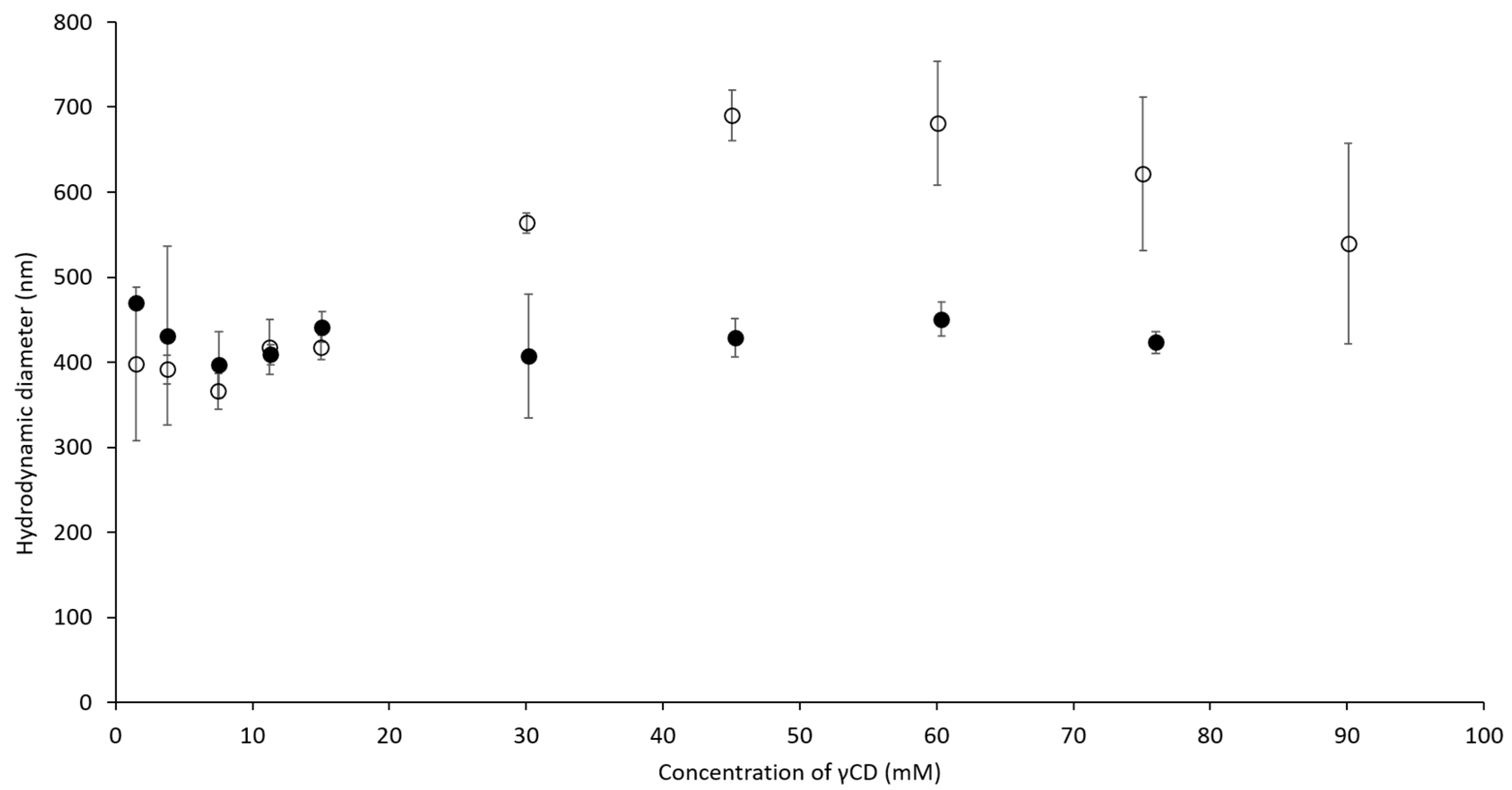
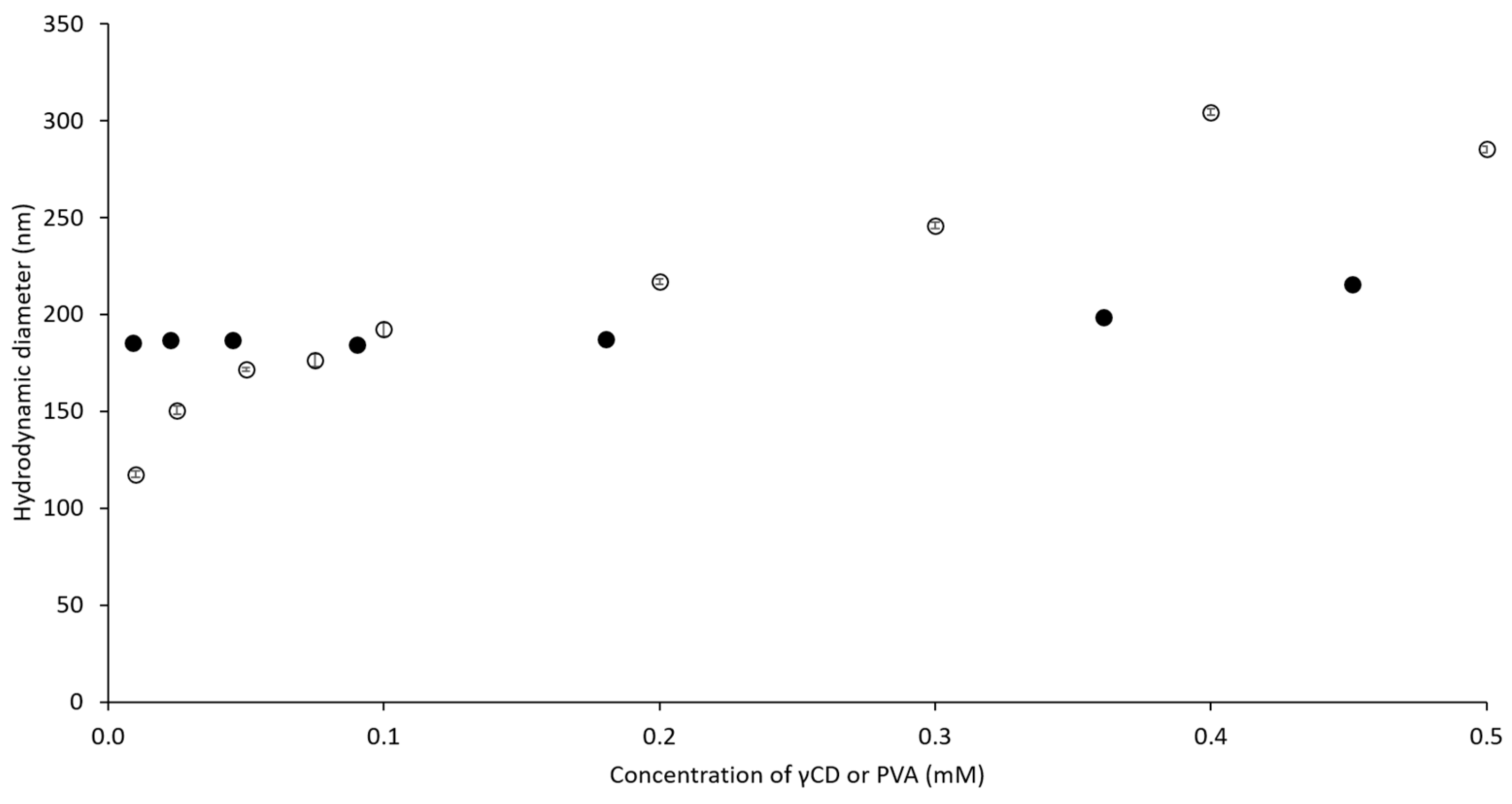
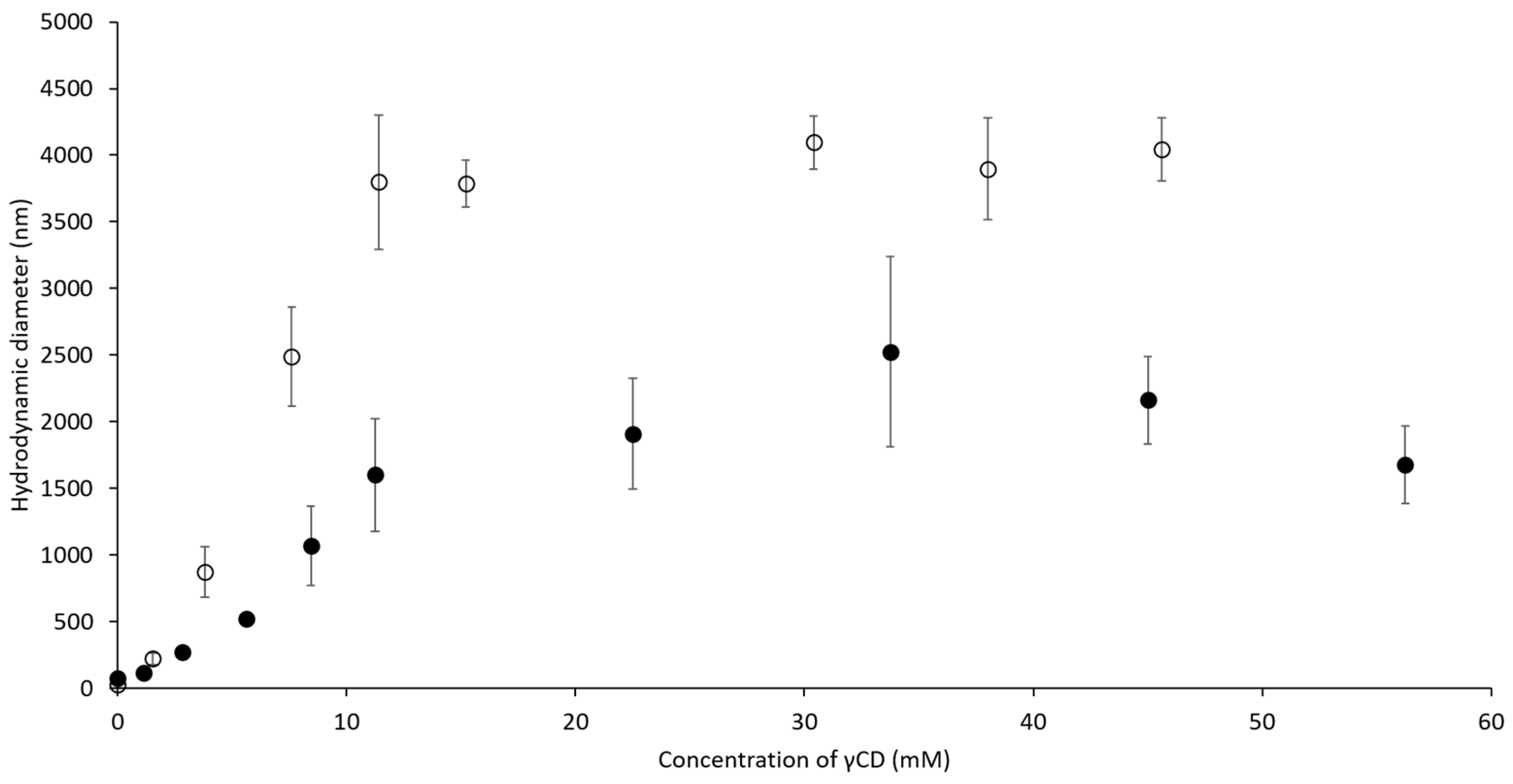
2.2. Hydrodynamic Diameter of Binary and Ternary Complexes
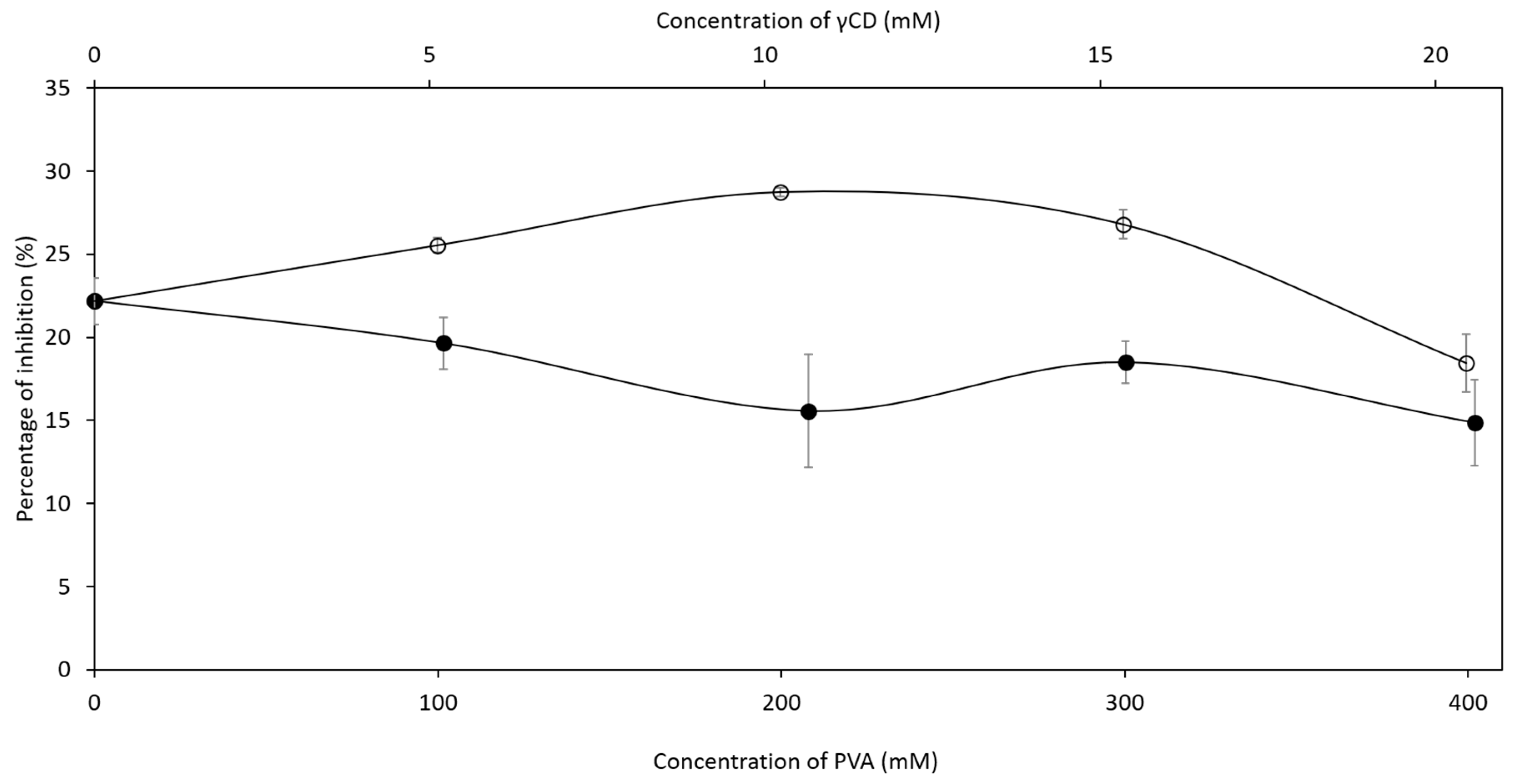
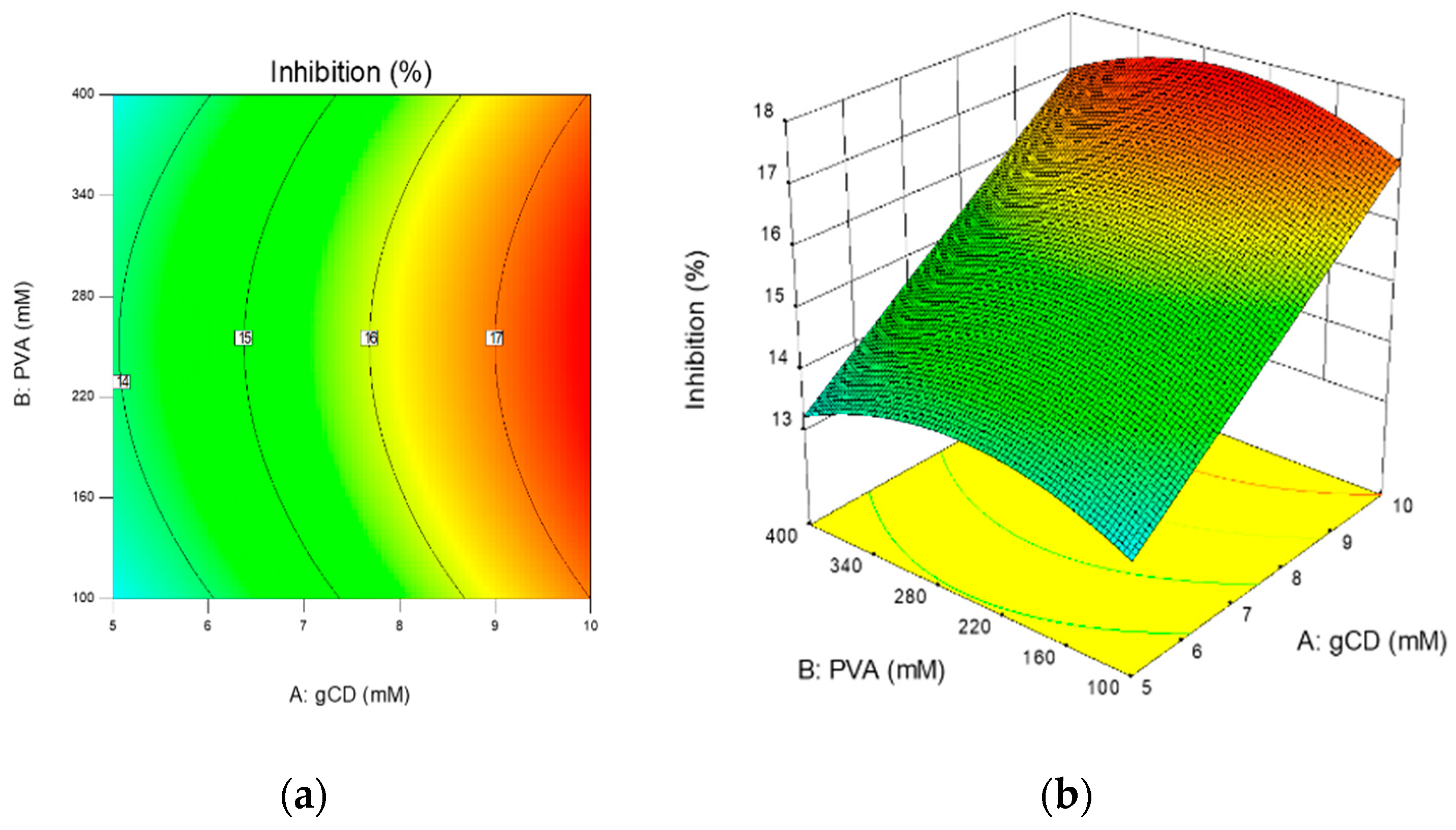
2.3. Inhibition Efficiency of Ascorbic Acid in Ternary Complex
3. Materials and Methods
3.1. Characterization of Binary and Ternary Complexes
3.1.1. Stoichiometry Determination
3.1.2. Fourier Transform Infrared (FTIR) Spectroscopy
3.1.3. Proton Nuclear Magnetic Resonance (1H-NMR) Analysis
3.2. Dynamic Light Scattering (DLS) Analysis
3.3. Antioxidant Capacity Studies
3.4. Experimental Design
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | Ascorbic acid |
| ANOVA | Analysis of variance |
| CCD | Central composite design |
| FT-IR | Fourier-transform infrared spectroscopy |
| γCD | γ-cyclodextrin |
| NMR | Nuclear magnetic resonance |
| PVA | Poly(vinyl alcohol) |
| SD | Standard deviation |
References
- Dodziuk, H. Cyclodextrins and Their Complexes: Chemistry, Analytical Methods, Applications; Wiley: Hoboken, NJ, USA, 2006. [Google Scholar]
- Martin, J.; Díaz-Montaña, E.J.; Garcia Asuero, A. Cyclodextrins: Past and Present; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Irie, T.; Uekama, K. Pharmaceutical applications of cyclodextrins. III. Toxicological issues and safety evaluation. J. Pharm. Sci. 1997, 86, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Loftsson, T.; Brewster, M.E. Pharmaceutical applications of cyclodextrins. 1. Drug solubilization and stabilization. J. Pharm. Sci. 1996, 85, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Capelezzo, A.; Mohr, L.; Dalcanton, F.; Mello, J.; Fiori, M. β-Cyclodextrins as Encapsulating Agents of Essential Oils. Cyclodextrin 2018. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Savic, I.M.; Jocic, E.; Nikolic, V.D.; Popsavin, M.M.; Rakic, S.J.; Savic-Gajic, I.M. The effect of complexation with cyclodextrins on the antioxidant and antimicrobial activity of ellagic acid. Pharm. Dev. Technol. 2019, 24, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, S.; Zhu, H.; Wang, S.; Xing, J. Inclusion Complexes of Lycopene and β-Cyclodextrin: Preparation, Characterization, Stability and Antioxidant Activity. Antioxidants 2019, 8, 314. [Google Scholar] [CrossRef]
- Inaki, Y.; Hiratsuka, H. Reversible Photo-Cross-linking of Polyvinyl Alcohol Containing Thymine. J. Photopolym. Sci. Technol. 2000, 13, 739–744. [Google Scholar] [CrossRef][Green Version]
- Hernández, R.; Rusa, M.; Rusa, C.C.; López, D.; Mijangos, C.; Tonelli, A.E. Controlling PVA Hydrogels with γ-Cyclodextrin. Macromolecules 2004, 37, 9620–9625. [Google Scholar] [CrossRef]
- Taleb, M.F.A.; El-Mohdy, H.L.A.; El-Rehim, H.A.A. Radiation preparation of PVA/CMC copolymers and their application in removal of dyes. J. Hazard. Mater. 2009, 168, 68–75. [Google Scholar] [CrossRef]
- Nho, Y.C.; Park, K.R. Preparation and properties of PVA/PVP hydrogels containing chitosan by radiation. J. Appl. Polym. Sci. 2002, 85, 1787–1794. [Google Scholar] [CrossRef]
- Paduraru, O.; Vasile, C.; Cascaval, C.; Stoleriu, A. Phase behaviour of polyvinyl alcohol/β-cyclodextrin blends. J. Optoelectron. Adv. Mater. 2007, 9, 1019–1024. [Google Scholar]
- López-de-Dicastillo, C.; Jordá, M.; Catalá, R.; Gavara, R.; Hernández-Muñoz, P. Development of Active Polyvinyl Alcohol/β-Cyclodextrin Composites to Scavenge Undesirable Food Components. J. Agric. Food Chem. 2011, 59, 11026–11033. [Google Scholar] [CrossRef] [PubMed]
- Chaouat, M.; Le Visage, C.; Baille, W.E.; Escoubet, B.; Chaubet, F.; Mateescu, M.A.; Letourneur, D. A Novel Cross-linked Poly(vinyl alcohol) (PVA) for Vascular Grafts. Adv. Funct. Mater. 2008, 18, 2855–2861. [Google Scholar] [CrossRef]
- Karimi, E.; Raisi, A.; Aroujalian, A. TiO2-induced photo-cross-linked electrospun polyvinyl alcohol nanofibers microfiltration membranes. Polymer 2016, 99, 642–653. [Google Scholar] [CrossRef]
- Xu, J.; Li, X.; Sun, F.; Cao, P. PVA hydrogels containing beta-cyclodextrin for enhanced loading and sustained release of ocular therapeutics. J. Biomater. Sci. Polym. Ed. 2010, 21, 1023–1038. [Google Scholar] [CrossRef]
- Pooresmaeil, M.; Namazi, H. Preparation and characterization of polyvinyl alcohol/β-cyclodextrin/GO-Ag nanocomposite with improved antibacterial and strength properties. Polym. Adv. Technol. 2019, 30, 447–456. [Google Scholar] [CrossRef]
- Constantin, M.; Fundueanu, G.; Bortolotti, F.; Cortesi, R.; Ascenzi, P.; Menegatti, E. Preparation and characterisation of poly(vinyl alcohol)/cyclodextrin microspheres as matrix for inclusion and separation of drugs. Int. J. Pharm. 2004, 285, 87–96. [Google Scholar] [CrossRef]
- Müller, L.D. Improved extraction methods for avoiding the interference of copper in the LC determination of ascorbic acid in multivitamin-mineral tablets. J. Pharm. Biomed. Anal. 2001, 25, 985–994. [Google Scholar] [CrossRef]
- Saha, S.; Roy, P.M. Encapsulation of Vitamin C into β-Cyclodextrin for Advanced and Regulatory Release; IntechOpen: London, UK, 2017; pp. 129–145. [Google Scholar] [CrossRef]
- Saha, S.; Roy, A.; Roy, K.; Roy, M.N. Study to explore the mechanism to form inclusion complexes of β-cyclodextrin with vitamin molecules. Sci. Rep. 2016, 6, 35764. [Google Scholar] [CrossRef]
- Garnero, C.; Longhi, M. Study of ascorbic acid interaction with hydroxypropyl-β-cyclodextrin and triethanolamine, separately and in combination. J. Pharm. Biomed. Anal. 2007, 45, 536–545. [Google Scholar] [CrossRef]
- Terekhova, I.; Obukhova, N. Calorimetric and spectrophotometric study on the interaction of hydroxypropyl-β-cyclodextrin with ascorbic acid. Mendeleev Commun. 2005, 15, 38–40. [Google Scholar] [CrossRef]
- Terekhova, I.V.; Kulikov, O.V. Thermodynamics of the interactions of ascorbic acid with α- and β-cyclodextrins in aqueous solutions. Mendeleev Commun. 2002, 12, 111–112. [Google Scholar] [CrossRef]
- Terekhova, I.V.; Obukhova, N.A.; Agafonov, A.V.; Kurochkina, G.I.; Syrtsev, A.N.; Gratchev, M.K. Thermodynamics of the effects of substituent, degree of substitution, and pH on complex formation of hydroxypropyl-α- and hydroxypropyl-β-cyclodextrins with ascorbic acid. Russ. Chem. Bull. 2005, 54, 1883–1886. [Google Scholar] [CrossRef]
- Inoue, Y.; Horage, M.; Suzuki, R.; Niiyama, D.; Urano, R.; Ando, S.; Kikuchi, J.; Murata, I.; Kanamoto, I. Study on complexation of ascorbic acid derivatives with γ-cyclodextrin. Int. J. Pharm. 2017, 7, 9–21. [Google Scholar]
- Bastianini, M.; Sisani, M.; Petracci, A. Ascorbyl Tetraisopalmitate Inclusion into Υ-Cyclodextrin and Mesoporous SBA-15: Preparation, Characterization and In Vitro Release Study. Cosmetics 2017, 4, 21. [Google Scholar] [CrossRef]
- Terekhova, I.V.; Kulikov, O.V.; Kumeev, R.S.; Nikiforov, M.Y.; Al’per, G.A. 1H NMR study of complexation of α- and β-cyclodextrins with some biologically active acids. Russ. J. Coord. Chem. 2005, 31, 218–220. [Google Scholar] [CrossRef]
- Karim, A.A.; Tan, E.; Loh, X.J. Encapsulation of Vitamin C with its Protection from Oxidation by Poly(Vinyl Alcohol). J. Mol. Eng. Mater. 2017, 5, 1750013. [Google Scholar] [CrossRef]
- Yohannan Panicker, C.; Tresa Varghese, H.; Philip, D. FT-IR, FT-Raman and SERS spectra of Vitamin C. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2006, 65, 802–804. [Google Scholar] [CrossRef]
- Reis, E.F.D.; Campos, F.S.; Lage, A.P.; Leite, R.C.; Heneine, L.G.; Vasconcelos, W.L.; Lobato, Z.I.P.; Mansur, H.S. Synthesis and characterization of poly (vinyl alcohol) hydrogels and hybrids for rMPB70 protein adsorption. Mater. Res. 2006, 9, 185–191. [Google Scholar] [CrossRef]
- Saokham, P.; Do, T.T.; Van den Mooter, G.; Loftsson, T. Inclusion complexes of p-hydroxybenzoic acid esters and γ-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2018, 90, 111–122. [Google Scholar] [CrossRef]
- Saokham, P.; Loftsson, T. A new approach for quantitative determination of γ-cyclodextrin in aqueous solutions: Application in aggregate determinations and solubility in hydrocortisone/γ-cyclodextrin inclusion complex. J. Pharm. Sci. 2015, 104, 3925–3933. [Google Scholar] [CrossRef] [PubMed]
- Matei, N.; Birghila, S.; Popescu, V.; Dobrinas, S.; Soceanu, A.; Oprea, C.; Magearu, V. Kinetic study of vitamin C degradation from pharmaceutical products. Rom. Rep. Phys. 2008, 53, 343–351. [Google Scholar]
- Huang, C.Y. Determination of binding stoichiometry by the continuous variation method: The Job plot. Methods Enzymol. 1982, 87, 509–525. [Google Scholar] [CrossRef] [PubMed]
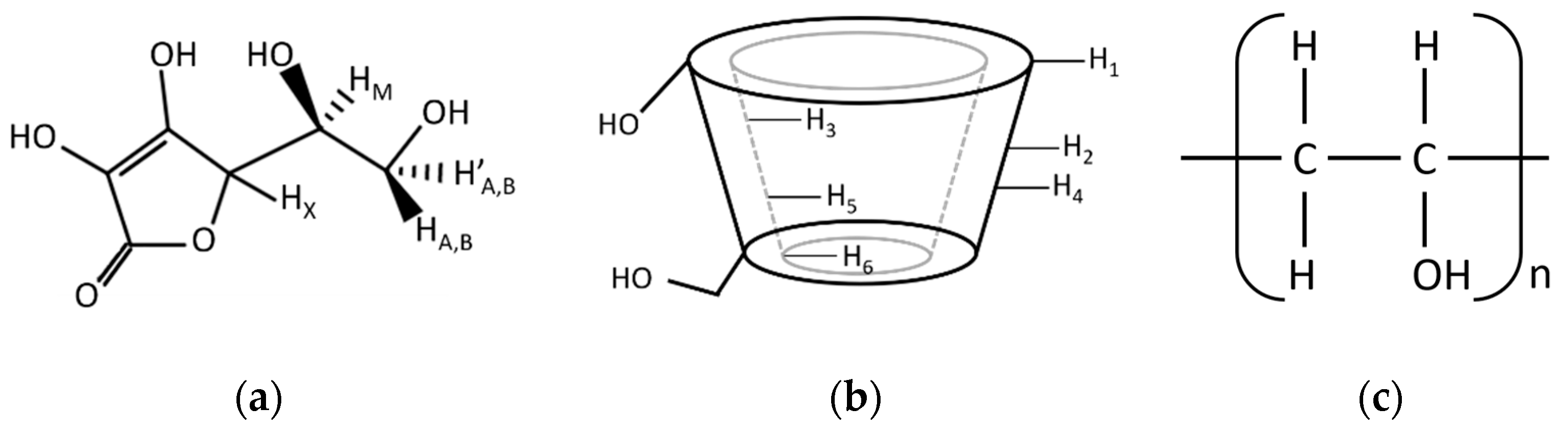
| Assignment | AA | γCD | PVA | Δcm−1 | |||
|---|---|---|---|---|---|---|---|
| AA/γCD | AA/PVA | γCD/PVA | AA/γCD/PVA | ||||
| O-H stretching | - | 3260.53 | - | 45.52 | - | 60.97 | 46.15 |
| C=O stretching | 1750.80 | - | - | 10.83 | 2.69 | - | 7.76 |
| C=C stretching | 1651.54 | - | - | 37.48 | 0.63 | - | 37.46 |
| C-O stretching | - | - | 1078.72 | - | 7.90 | 1.40 | 0.77 |
| Assignment | δ (ppm) | Δδ (ppm) | |||
|---|---|---|---|---|---|
| AA/γCD | AA/PVA | γCD/PVA | AA/γCD/PVA | ||
| HX | 4.8658 | −0.0073 | −0.0084 | - | −0.0144 |
| HM | 3.9977 | −0.0027 | −0.0013 | - | −0.0028 |
| HA,B | 3.6667 | −0.0018 | −0.0005 | - | −0.0013 |
| H3 | 3.8598 | −0.0030 | - | −0.0023 | −0.0022 |
| H5 | 3.7970 | −0.0027 | - | −0.0019 | −0.0018 |
| HCH2 | 1.6291 | - | 0.0003 | −0.0007 | −0.0001 |
| Experimental No. | Concentration of γCD (mM) | Concentration of PVA (mM) |
|---|---|---|
| 1 | 5 | 400 |
| 2 | 5 | 100 |
| 3 | 7.5 | 50 |
| 4 | 3 | 250 |
| 5 | 12 | 250 |
| 6 | 7.5 | 250 |
| 7 | 7.5 | 450 |
| 8 | 10 | 100 |
| 9 | 7.5 | 250 |
| 10 | 7.5 | 250 |
| 11 | 7.5 | 250 |
| 12 | 10 | 400 |
| 13 | 7.5 | 250 |
| Quadratic Model (R2 = 0.8124) | Two-Factor Model (R2 = 0.6685) | Linear Model (R2 = 0.6507) | ||||
|---|---|---|---|---|---|---|
| Coefficient | p-Value | Coefficient | p-Value | Coefficient | p-Value | |
| Model | - | 0.0175 * | - | 0.0153 * | - | 0.0052 * |
| γCD | 1.94 | 0.0015 * | 1.89 | 0.0023 * | 1.89 | 0.0015 * |
| PVA | 0.0277 | 0.9465 | 0.0167 | 0.9723 | 0.0069 | 0.9882 |
| γCD*PVA | −0.4462 | 0.4440 | −0.4482 | 0.5047 | - | - |
| (γCD)2 | −0.5900 | 0.1744 | - | - | - | - |
| (PVA)2 | −0.8360 | 0.0914 ** | - | - | - | - |
| Lack of Fit | - | 0.4871 | - | 0.3241 | - | 0.3660 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saokham, P.; Burapapadh, K.; Praphanwittaya, P.; Loftsson, T. Characterization and Evaluation of Ternary Complexes of Ascorbic Acid with γ-Cyclodextrin and Poly(vinyl Alcohol). Int. J. Mol. Sci. 2020, 21, 4399. https://doi.org/10.3390/ijms21124399
Saokham P, Burapapadh K, Praphanwittaya P, Loftsson T. Characterization and Evaluation of Ternary Complexes of Ascorbic Acid with γ-Cyclodextrin and Poly(vinyl Alcohol). International Journal of Molecular Sciences. 2020; 21(12):4399. https://doi.org/10.3390/ijms21124399
Chicago/Turabian StyleSaokham, Phennapha, Kanokporn Burapapadh, Pitsiree Praphanwittaya, and Thorsteinn Loftsson. 2020. "Characterization and Evaluation of Ternary Complexes of Ascorbic Acid with γ-Cyclodextrin and Poly(vinyl Alcohol)" International Journal of Molecular Sciences 21, no. 12: 4399. https://doi.org/10.3390/ijms21124399
APA StyleSaokham, P., Burapapadh, K., Praphanwittaya, P., & Loftsson, T. (2020). Characterization and Evaluation of Ternary Complexes of Ascorbic Acid with γ-Cyclodextrin and Poly(vinyl Alcohol). International Journal of Molecular Sciences, 21(12), 4399. https://doi.org/10.3390/ijms21124399