2.1. Fitting the Models
The effects of five independent variables, namely palm kernel oil ester (PKOE): safflower seed oil (A), lecithin (B), Tween 85:Span 85 (C), glycerol (D), and water (E), on particle size and volume median diameter (VMD) were explored using the D-optimal mixture experimental design (MED) for both DTX- and CCM-loaded nanoemulsions. The upper and lower limits of the independent variables were obtained from previous data collected during the preliminary study [
15], while the software automatically calculated the amount of water. The candidate points referring to the design space and response values for both DTX- and CCM-loaded nanoemulsions are depicted in
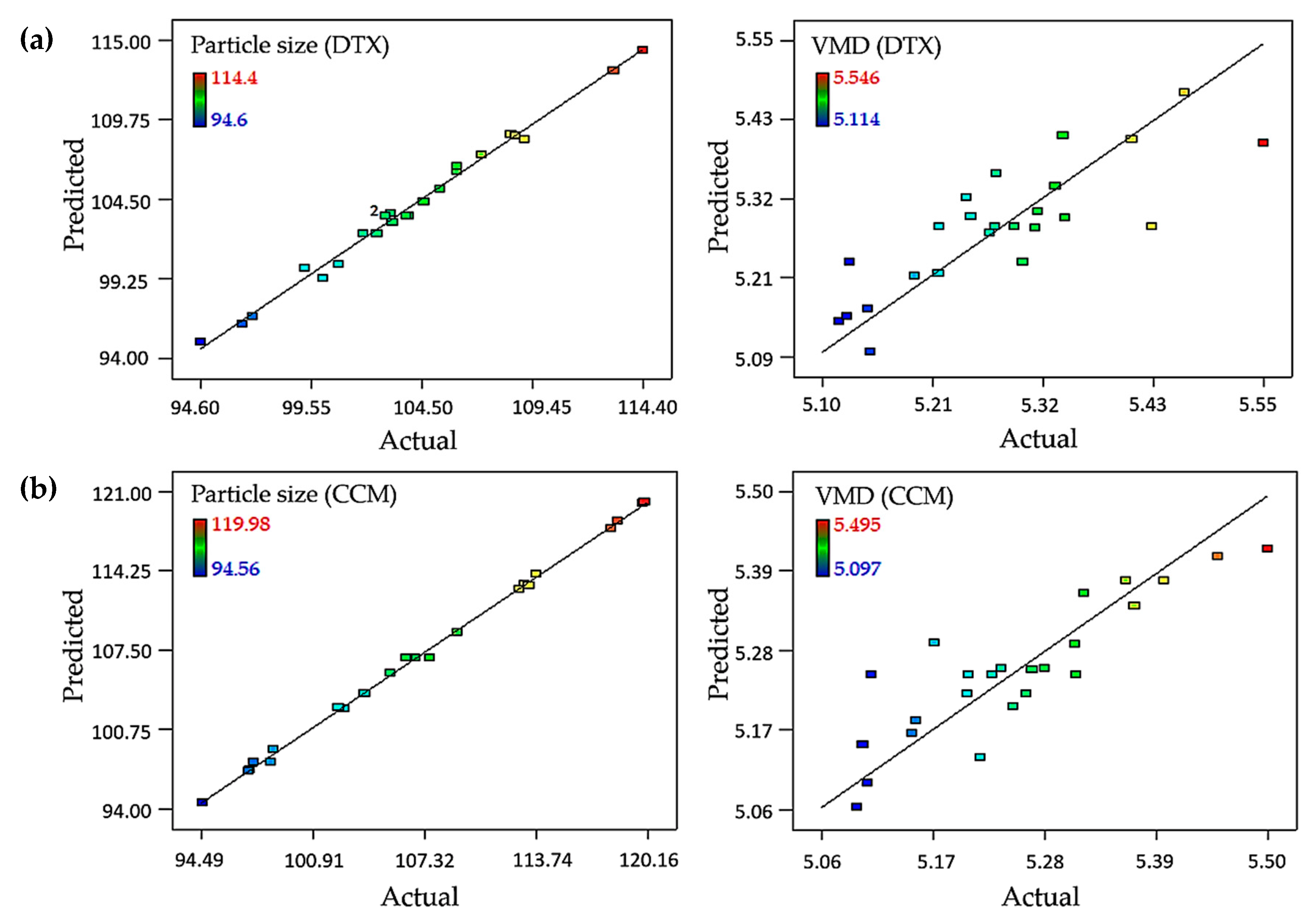
Table 1. The obtained data were fitted into three models, namely linear, quadratic, and special cubic models. Among them, the quadratic polynomial model was found to be the best-fitted to the experimental data of particle size. In contrast, the linear polynomial model matched with the experimental data of volume median diameter (VMD) for both DTX- and CCM-loaded nanoemulsion formulations.
The correlation coefficient (
R2) values for the particle size of the DTX- and CCM-loaded nanoemulsions were 0.9922 and 0.9984, respectively, while the
R2 values for VMD were 0.6894 and 0.7610, respectively. These values specified that the model could be predicted up to 99.22 and 99.84%, respectively, of the particle size observed value, leading to a strong correlation between the predicted data and actual values. The VMD observed value could only be predicted up to 68.94 and 76.10% for the DTX- and CCM-loaded nanoemulsions, respectively. These findings are illustrated by the linearity plot between the predicted and actual values of particle size and the VMD. Good-fitting regression models for particle size were obtained compared to the VMD for both formulations (
Figure 1). The adequate precision for the particle size of the DTX- and CCM-loaded nanoemulsions were 35.58 and 64.87, respectively, whereas the adequate precision for VMD was 11.80 and 13.36, respectively. The signal-to-noise ratio of the models was appropriate because of the high value of adequate precision (>4.00), which indicated adequate model discrimination, as it compared the range of the predicted values at the design points to the average prediction error [
16,
17].
The predicted values for each response in both formulations were derived using regression Equations (1) and (2) for DTX-loaded nanoemulsion and regression Equations (3) and (4) for CCM-loaded nanoemulsion, as suggested by ANOVA. The generated mathematical models were proven to be significant and valid for the responses, as they had small
p-values (
p < 0.0001) and large
F-values, which stipulated a good match between the regression models and experimental data. The models are significant if the
p-value is below 0.05, suggesting that the model is significant at the probability level of 95% [
13,
18]. Other than that, a lack of fitness of the models was not significantly related to pure error (
p > 0.05). Following
p-values of <0.05, the interactions of AB, AC, AD, AE, CD, and CE harbored significant effects on the particle size of a DTX-loaded nanoemulsion, while interactions of AB, AC, AD, AE, BC, BD, BE, CE, and DE showed a significant effect on the particle size of a CCM-loaded nanoemulsion. The ANOVA results for both formulations are summarized in
Table 2.
2.2. Influence of Independent Variables on Particle Size
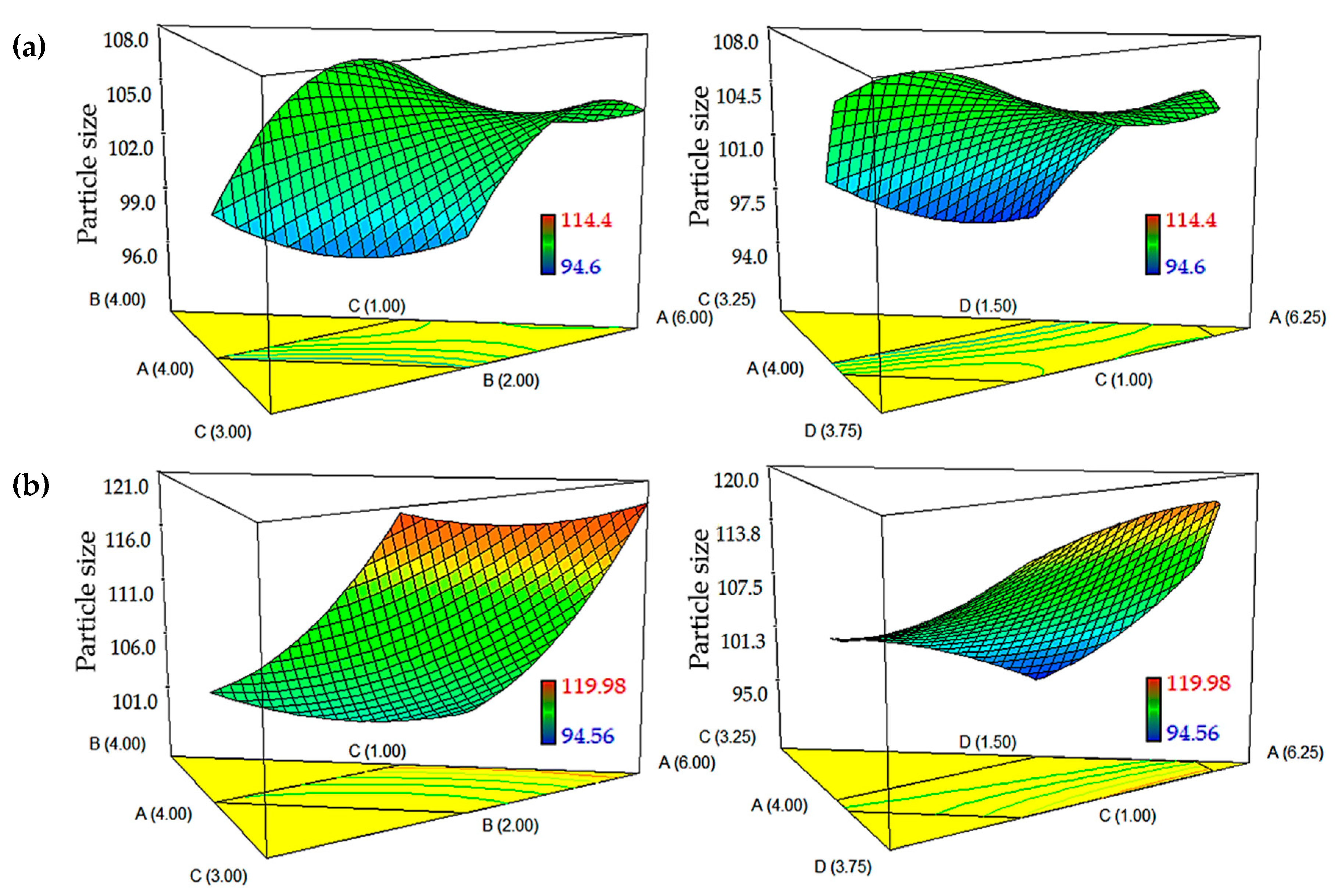
The DTX- and CCM-loaded nanoemulsions showed an increase in the particle size when there was an increase in the amount of oil (
Figure 2). This effect was attributed to the increase of the dispersed phase’s viscosity in conjunction with the increase of oil content, which subsequently resulted in the increase of flow resistance and restriction on the droplet breakup rate [
19]. This effect could be correlated with increased frequency of collision between the nanoemulsion particles, accompanied by an increase in the frequency of coalescence, resulting in a higher chance of coalescence between the particles [
20]. Moreover, adding emulsifiers, including lecithin and Tween 85:Span 85 in the two formulations showed the opposite effect, whereby the particle size decreased when the surfactant and co-surfactant were increased. Other researchers have observed similar results, stating that the decrease of particle size could be due to the availability of free emulsifier to spontaneously enclose the surface of any new oil droplet formed during homogenization [
18,
20,
21]. Moreover, the interfacial tension between water and oil may get decreased, which can reduce the stress for droplet breakup.
The addition of glycerol in the nanoemulsion system could decrease the particle size, as it contained both hydrophilic and lipophilic components in the structure that could invade the surfactant monolayer, resulting in a change in the system’s packing geometry, interfacial tension and surfactant flexibility [
22,
23]. However, if compared with the CCM-loaded nanoemulsion plot, the particle size for the DTX-loaded nanoemulsion was slightly increased initially, as the concentration of emulsifiers and glycerol increased, and the concentration of oil decreased. It started to decline when the concentration of emulsifiers was set to increase, whereby the DTX-loaded nanoemulsion would reach a consistent level of CCM-loaded nanoemulsion in the graphical plot. This finding might be attributed to the higher lipophilicity of DTX (logP
O/W = 4.1) compared to CCM (logP
O/W ~ 3.0), which observed a reduction of emulsification efficiency at low concentration of emulsifiers and high concentration of oil.
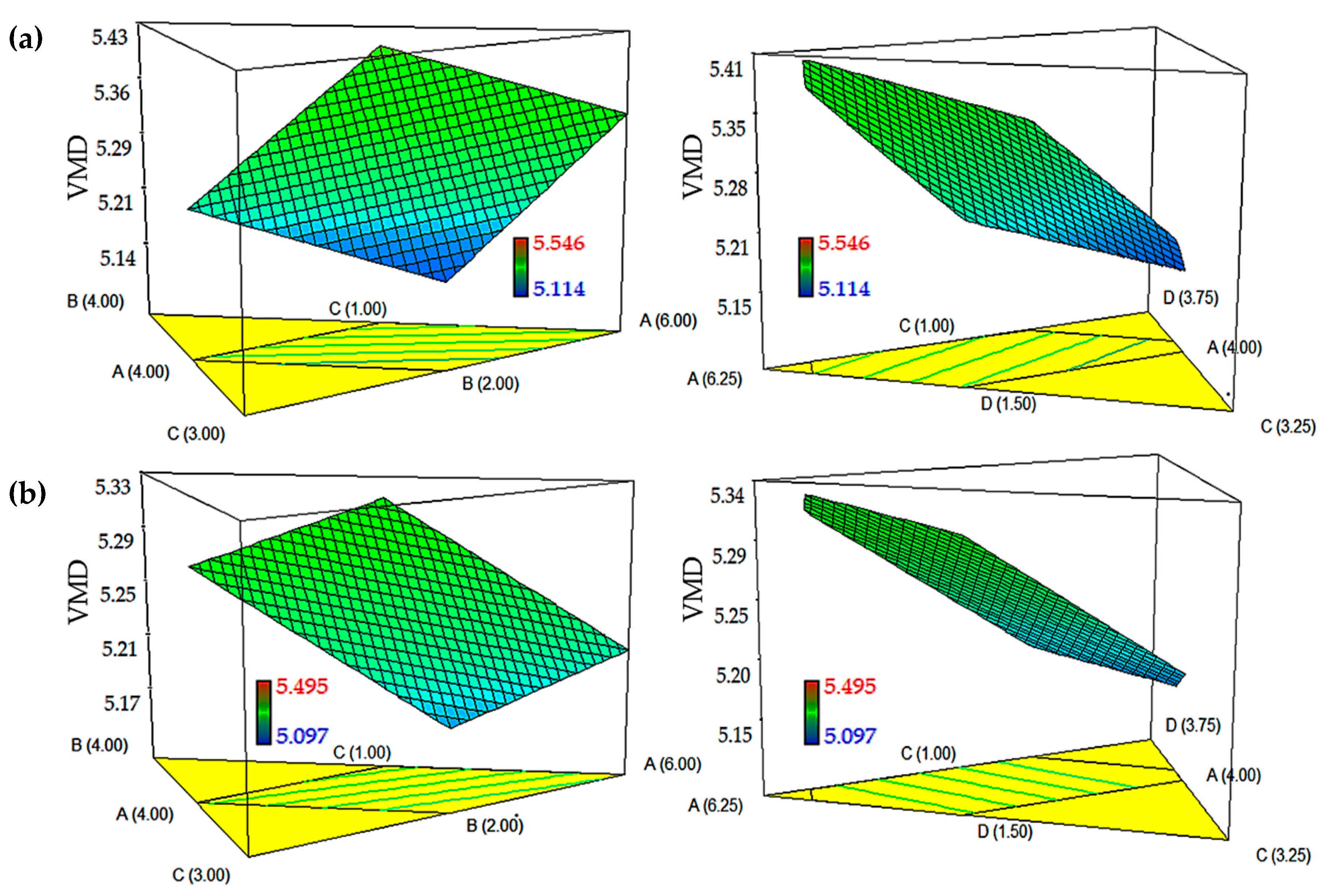
2.3. Influence of Independent Variables on Aerosol Size
The aerodynamic diameter of nanoemulsion aerosols is the determinant of the deposition site in the lungs, as large particles (>6 μm) tend to deposit mainly in the upper airways. The production of aerosol with an aerodynamic diameter between 2 and 6 μm is found to be best-suited to treat central and small airways [
24]. In this work, both models for DTX- and-CCM loaded nanoemulsions showed VMD values in the range of 5.0–5.6 μm. As illustrated in
Figure 3, the VMD value decreased as the amount of Tween 80:Span 80 (9:1,
w/
w) and glycerol increased in both the DTX- and CCM-loaded nanoemulsions. This effect was obtained due to incorporation of surfactant and co-solvent in the system, which reduced the interfacial tension at the interface of oil and water by improving optimal curvature and lowering elasticity of the surfactant monolayer. Consequently, it decreased the viscosity of the system, as well [
22]. However, the VMD value increased as the amount of glycerol increased and the amount of Tween 80:Span 80 (9:1,
w/
w) decreased because of the difficulty in breaking up the oil-water interface without decreasing interfacial tension [
25].
Characteristics of the nanoemulsion system like viscosity and the overall surface tension would notably affect the system’s aerosolization efficiency. Increasing the viscosity or surface tension would need higher energy to atomize the nanoemulsion into aerosols, leading to an increase in aerosol size [
26,
27]. Arbain et al. and Shah et al. report nearly the same observations wherein decreasing the surface tension and viscosity of the nebulized nanoemulsion resulted in effective aerosolization using the Omron nebulizer [
11,
28]. Remarkably, the VMD value increased as the amount of oil and lecithin increased in DTX-loaded nanoemulsion. In contrast, the VMD value decreased as the amount of oil increased, and the amount of lecithin increased in the CCM-loaded nanoemulsion. This phenomenon might be due to lower lipophilicity and molecular weight of CCM compared to DTX, which permits a minimum packing geometry and requires less surfactant (lecithin) to reduce the surface tension. Besides, a higher molecular weight of lipophilic drugs has been reported to increase the viscosity of the solution and generate a larger-size of aerosols [
29].
As shown in
Table 3, the experimental response values for randomized and optimized formulations, as suggested by the MED, were juxtaposed with the predicted values to verify the generated models. The results showed that predicted values of all formulations in both the DTX- and CCM-loaded nanoemulsions resembled the experimental values closely. Moreover, the percentage of residual standard error (RSE) values were small with less than 2.2% for particle size and less than 3.8% for the VMD. These results were correlated reasonably well with the models’ regression correlation between the predicted and actual values, as higher
R2 values were obtained for particle size compared to that of the VMD. The RSE values for both responses fell within the acceptable limits of model validation. It could be concluded that the postulated models were acceptable for use in predicting the response accurately.
2.4. Physicochemical and Aerodynamic Properties of Optimized Nanoemulsions
It is widely understood that nanoemulsion systems with a high quantity of oil, surfactants, and co-solvent may increase the safety issues in pulmonary application [
30]. Therefore, it is necessary to use a minimal and optimal concentration of these components in the nanoemulsion formulation to allow the formation of a stable, safe, and fine nanoemulsion. In this study, the optimal formulation for a DTX-loaded nanoemulsion (DNE
Opt) and CCM-loaded nanoemulsion (CNE
Opt) were selected based on their minimum value of response variables at a minimal amount of nanoemulsion compositions for maximizing the effectiveness on the delivery and treatment of inhalable drugs. The optimized formulations for both DNE
Opt and CNE
Opt could be attained with a 6.0 wt% mixture of PKOE and safflower seed oils (1:1), 2.5 wt% of lecithin, 2.0 wt% mixture of Tween 85 and Span 85 (9:1), and 2.5 wt% of glycerol in the aqueous phase as suggested by MED.
Table 4 summarizes the overview of the physicochemical properties of the optimized formulations. The mean particle size of DNE
Opt and CNE
Opt was found to be 95.80 and 96.94 nm, respectively, with an RSE of less than 4%. The obtained particle size of the optimized formulations was further ascertained by the TEM micrographs, as depicted in
Figure 4. The particles of the nanoemulsions were spherical, and the average particle size measured using TEM was in line with the average size determined from the photon correlation spectroscopy. The stability of the nanoemulsion system would increase when the polydispersity index (PDI) value was minimized, whereas the Ostwald ripening due to repulsive interaction between the particles during storage could be reduced when the
ζ-potential value was maximized [
31]. The PDI and
ζ-potential values for both optimized formulations were in the range 0.22–0.25 and greater than −30 mV, respectively. These results suggest that the systems are slightly polydisperse but still adequate for pulmonary administration and they possess a sufficiently high negative surface charge to maintain stability due to the presence of anionic groups of fatty acids and glycol in the oils and lecithin [
32,
33].
Administration of too acidic or basic and hypo- or hyperosmotic formulations via the pulmonary route is known to cause cough and irritation of the lung linings, which can increase drug loss and reduce the carrier efficiency [
34,
35]. The formulation’s pH for liquid preparation intended for pulmonary administration should resemble closely with the pH of lung fluid at around 7.0 and 8.0 [
36], while for human red blood cells, the normal osmotic pressure in the range of 280–320 mOsm/kg was measured at pH 7.40 [
37]. The pH and osmolality values of the optimized formulations fell within the acceptable range for pulmonary delivery. On top of that, both formulations exhibited low viscosity values between 2.05 and 2.22 cP, which serve as a key parameter in influencing the effective delivery of drugs via the pulmonary route. The aerosolization efficiency of the formulations will be reduced at high viscosity as it contributes to an increase in aerodynamic diameter [
26,
27].
The laser diffraction method was used during the stage of aerosolization to assess the quality of the aerosol, while the Anderson cascade impaction (ACI) method was applied for summative aerosolization for inhalation. As given in
Table 5, the optimized formulations had produced aerosol size within the respirable range, with the VMD (DV
50) and mass median aerodynamic diameter (MMAD) falling within the optimal respirable range (2–6 µm). However, the size of the aerosol from the analysis of laser diffraction was relatively greater than that of the cascade impaction method, which was similar to the results obtained from other findings [
28,
38]. This effect was attributed to the loss of small particles during aerosolization through evaporation during the laser diffraction analysis, which led to a significant shift in the particle size distribution, causing a larger aerosol size than in the ACI method. Both DNE
Opt and CNE
Opt exhibited relatively narrow size distribution (<2.00) with span values of 1.64 and 1.69 and geometric standard deviation (GSD) values of 1.71 and 1.67, respectively.
For both the optimized formulations, the values of percent inhaled (PI) and percent dispersed (PD) were determined to be high since drug inhalation and emission into the lungs were more than 70%, while the rest of the drug may settle on the sampling apparatus. Similarly, a high percentage of fine particle fraction (FPF > 75%) was obtained, which indicated that the majority of the aerosol particles could be predominated in the deep lung region. The results appeared to conform with the in vitro deposition of optimized formulations on the ACI (
Figure 5a). It was found that most of the nebulized aerosols were deposited on stages 3, 4, 5, and 6, which represented lower parts of respiratory airways with the aerodynamic cut-off diameters of 3.3–4.70, 2.10–3.3, 1.10–2.10, and 0.65–1.10 µm, respectively. Additionally, the in vitro drug release profile (
Figure 5b) showed that the optimized formulations were able to release the DTX and CCM up to 48 h steadily. This was due to the additional time needed for the drug to permeate from the core of the oil droplet through the interfacial layer of surfactants before it could reach into the dissolution medium. The sustained release of encapsulated DTX and CCM can offer prolonged exposure at the target site and further enhance the efficacy of the pulmonary application.
Moreover, the drug release profiles of the encapsulated drugs were found to be relatively faster compared to the free drug solubilized in an aqueous solution, as reported by several studies [
39,
40,
41], due to their poor water solubility. The drug release was lower in simulated lung fluid (SLF) at pH 7.4 compared to pH 6.5. These results were in agreement with previous studies on DTX and CCM, where the release was accelerated significantly under an intracellular tumor environment (pH~6.5) than an intracellular healthy environment (pH 7.4) [
42,
43]. The DNE
Opt was able to release the drug faster compared to CNE
Opt due to a lower degree of ionization in the CCM compared to DTX when tested at pH 6.5 and 7.4. According to the passive diffusion barrier concept, the drug in nonionized form could passively be diffused through the barriers effectively [
44]. In contrast, the drug at a high degree of ionization state was deemed to be difficult in passing through the membranes, as it carried charge [
45]. The result of optimized nanoemulsions with appropriate physicochemical and aerodynamic properties for inhalation and a sustained drug release manner might suggest the nanoemulsions great potential in pulmonary application.