Regulation of Actin Filament Length by Muscle Isoforms of Tropomyosin and Cofilin
Abstract
1. Introduction
2. Results
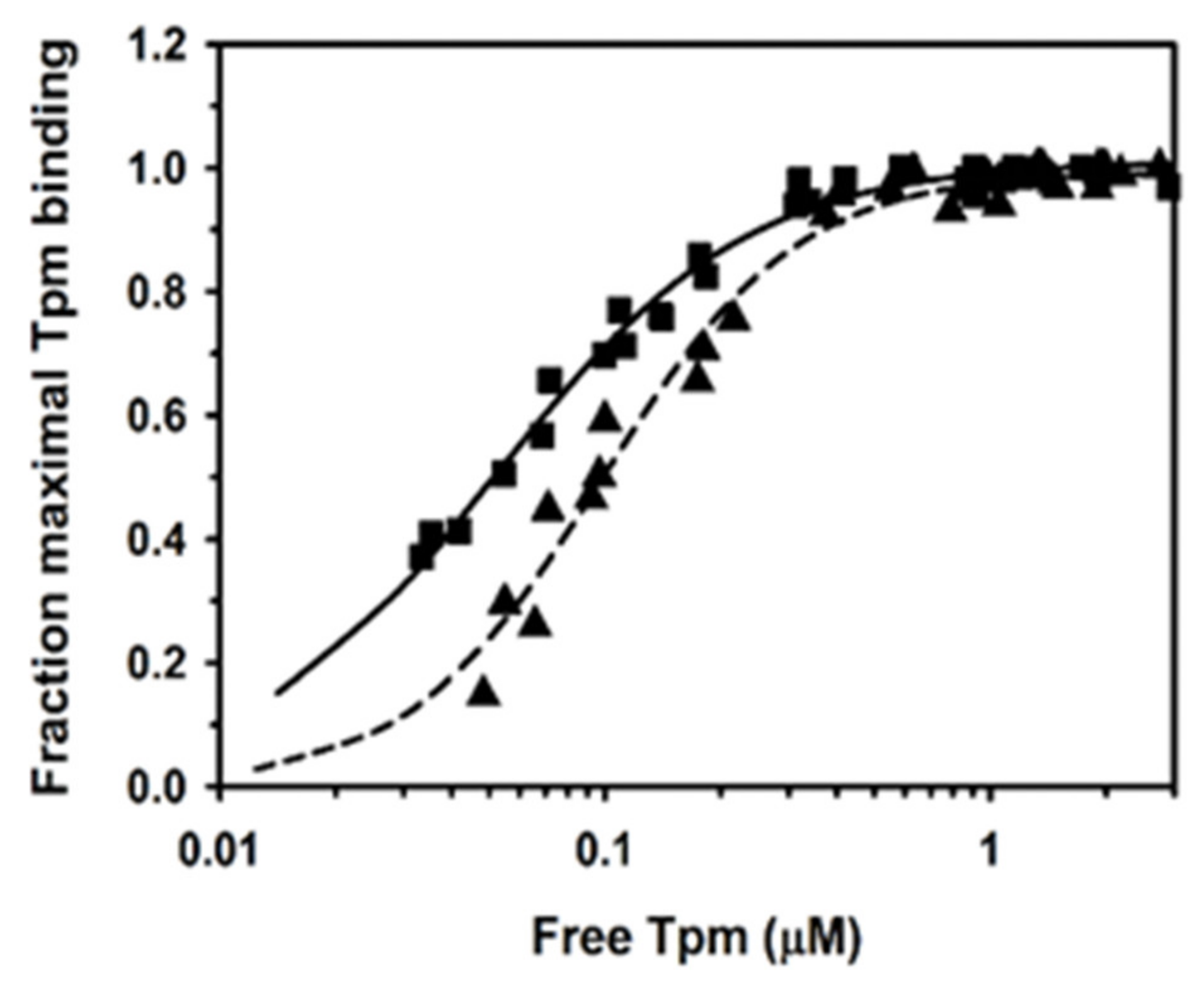
2.1. Binding of Human Tpm1.1 and Tpm3.12 to F-Actin
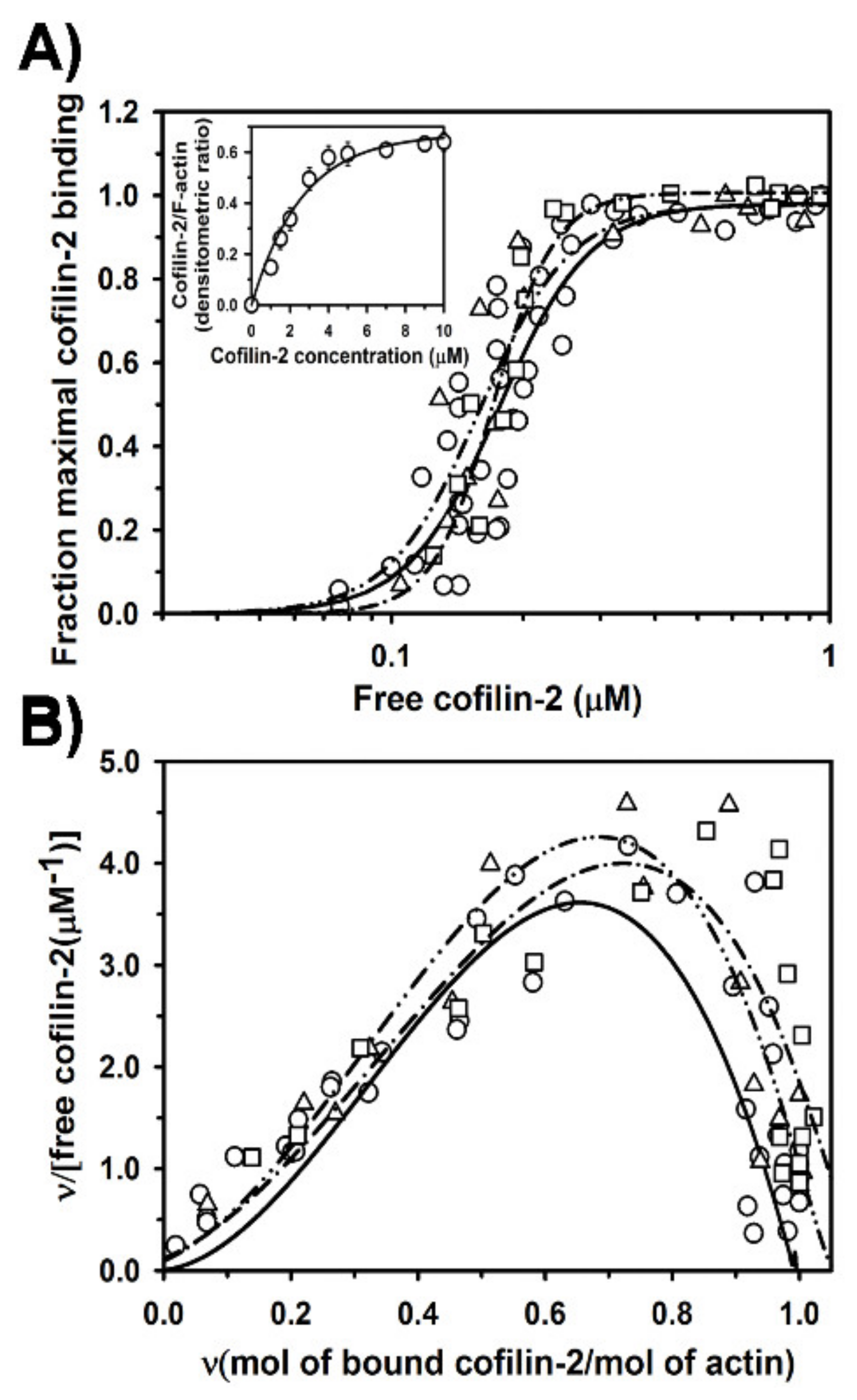
2.2. Regulation of Cofilin-2 Binding to F-Actin by Tpm1.1 and Tpm3.12
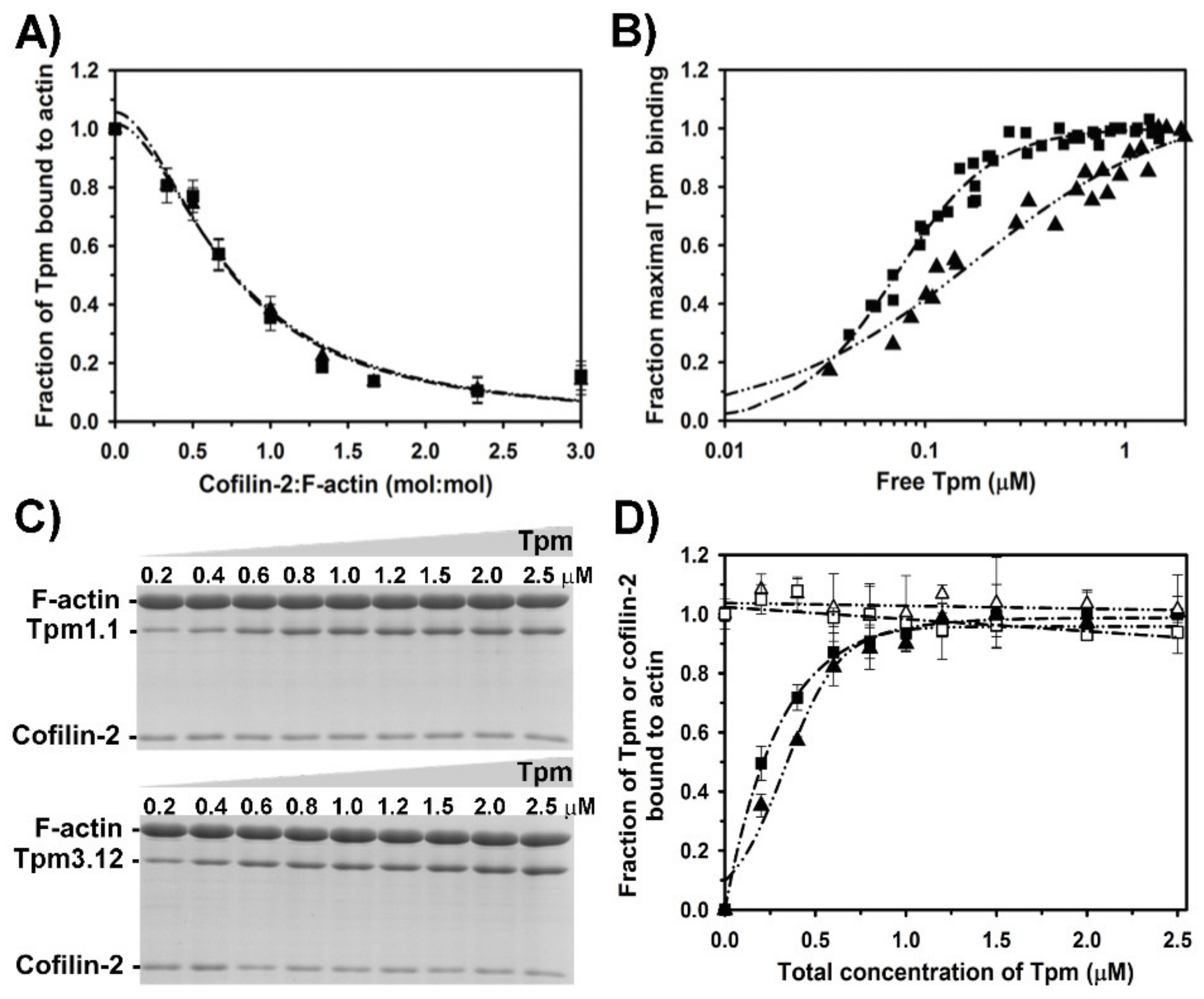
2.3. Effect of Cofilin-2 on Interactions of Tropomyosin Isoforms with F-Actin
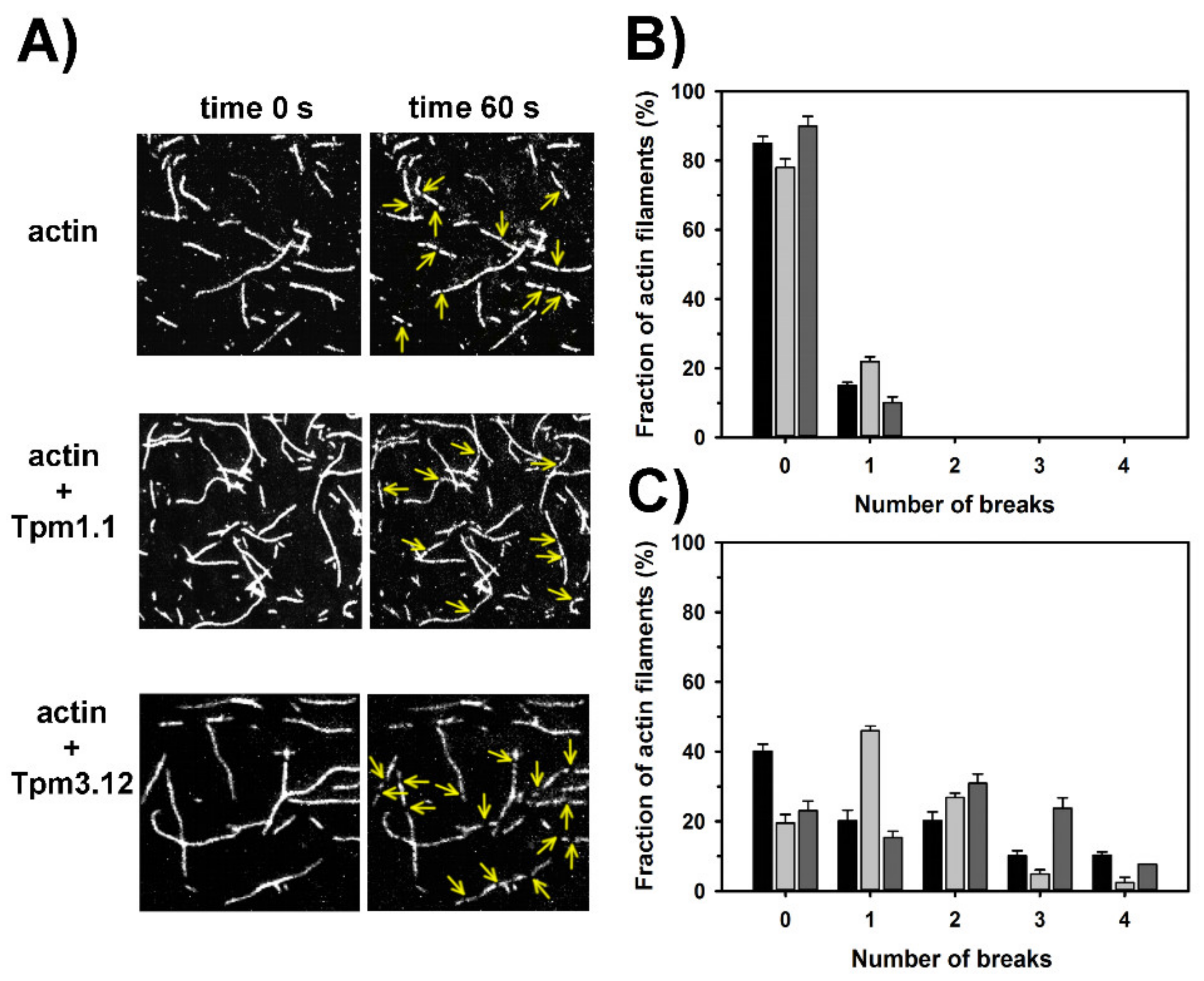
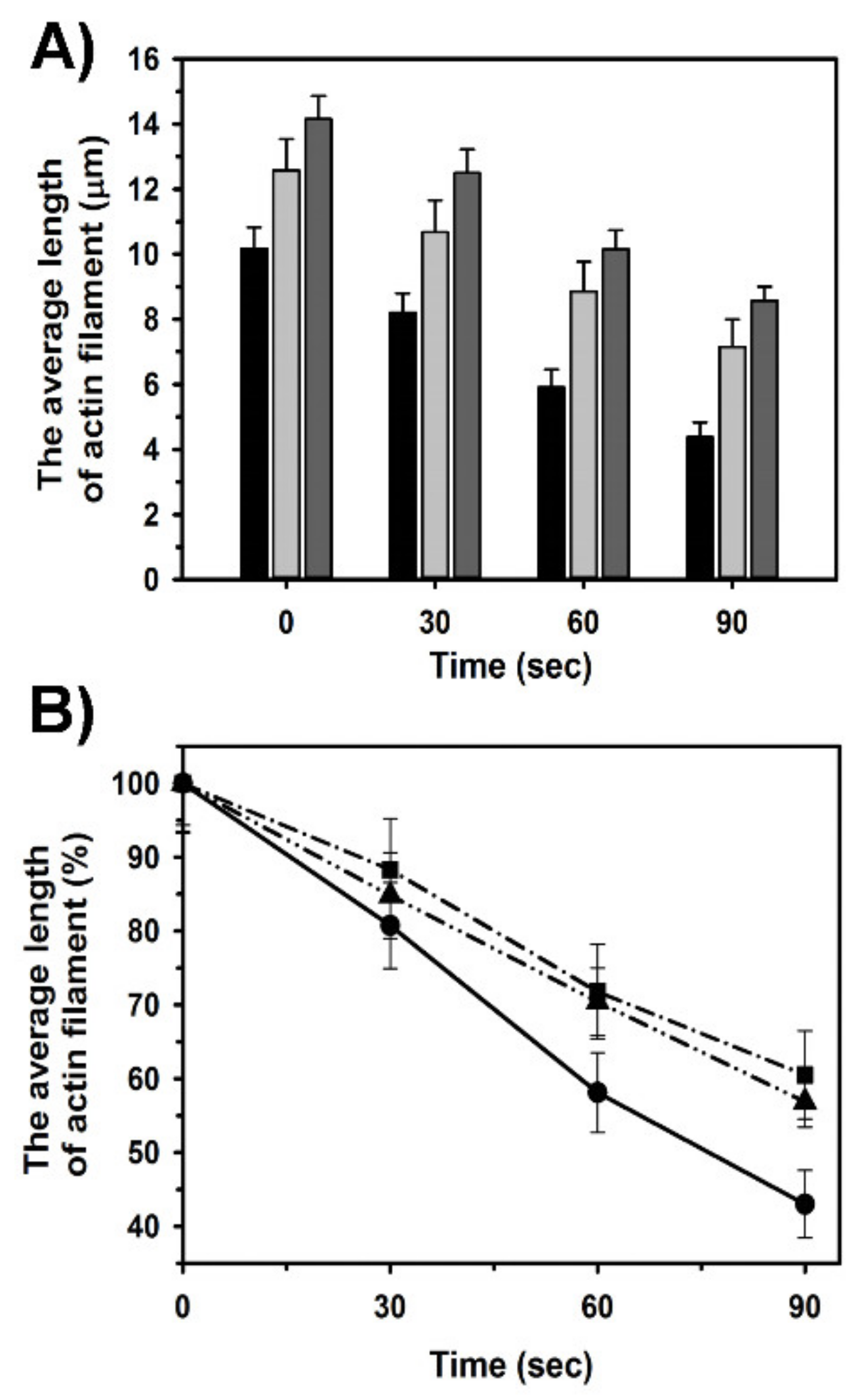
2.4. Effects of Tpm1.1 and Tpm3.12 on Cofilin-2 Induced Fragmentation and Depolymerization of F-Actin
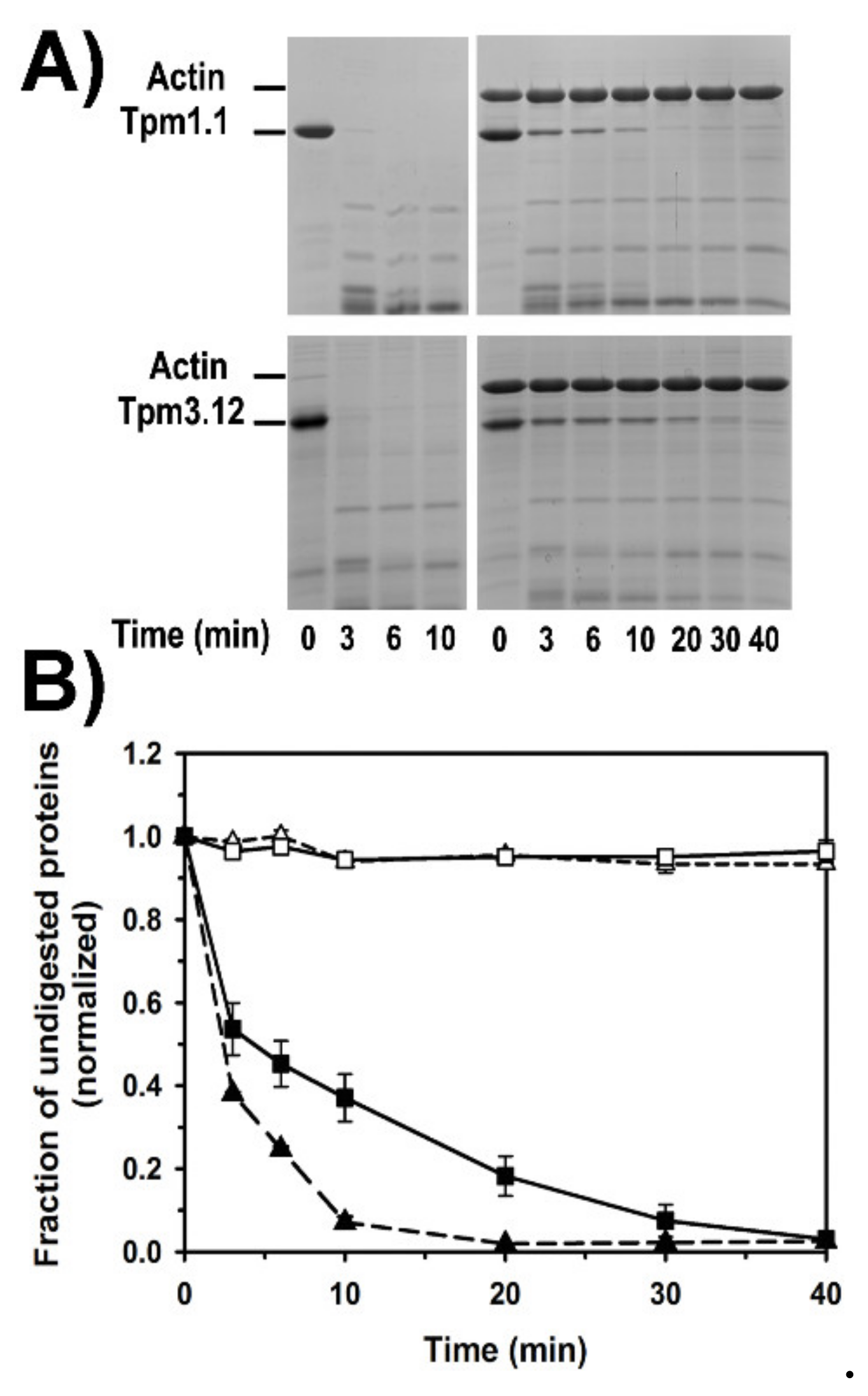
2.5. Dynamics of Tropomyosin Binding to F-Actin
3. Discussion
3.1. Structural Determinants of Tpm1.1 and Tpm3.12 Affinity for Actin
3.2. Tropomyosin-Induced Changes in the Cooperativity of Cofilin-2 Binding to the F-Actin Linear Lattice
3.3. Structural Basis of Tropomyosin Removal from the Filament by Cofilin
3.4. Molecular Bases of the Regulation of Actin Filament Length by Tropomyosin Isoforms
3.5. Roles of Tropomyosin Isoforms in the Regulation of Actin Dynamics at the Pointed End
3.6. Conclusions
4. Materials and Methods
4.1. Protein Preparations from Rabbit Muscle
4.2. Preparation of Recombinant Tropomyosin Isoforms and Cofilin-2
4.3. Fluorescent Labeling of G-Actin with Tetramethylrhodamine Cadaverine
4.4. Co-Sedimentation Assay
4.5. In Vitro Severing/Depolymerization Assay
4.6. Tropomyosin Digestion with Trypsin
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Tpm1.1 | Tropomyosin isoform 1.1 |
| Tpm3.12 | Tropomyosin isoform 3.12 |
| TPM1 | Tropomyosin gene 1 |
| TPM2 | Tropomyosin gene 2 |
| TPM3 | Tropomyosin gene 3 |
| ADF | Actin depolymerizing factor |
| DTT | DL-Dithiothreitol |
| EGTA | Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid |
| TRC | tetramethylrhodamine cadaverine |
| HMM | Heavy meromyosin |
| Hepes | 4-(2-Hydroxyethyl)piperazine−1-ethanesulfonic acid |
| Tris | Tris(hydroksymetylo)aminometan |
| Kapp | Apparent binding constant |
References
- Pette, D.; Staron, R.S. Myosin isoforms, muscle fiber types, and transitions. Microsc. Res. Tech. 2000, 50, 500–509. [Google Scholar] [CrossRef]
- Schiaffino, S. Muscle fiber type diversity revealed by anti-myosin heavy chain antibodies. FEBS J. 2018, 285, 3688–3694. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.M.; Homsher, E.; Regnier, M. Regulation of contraction in striated muscle. Physiol. Rev. 2000, 80, 853–924. [Google Scholar] [CrossRef]
- Pieples, K.; Wieczorek, D.F. Tropomyosin 3 increases striated muscle isoform diversity. Biochemistry 2000, 39, 8291–8297. [Google Scholar] [CrossRef]
- Corbett, M.A.; Akkari, P.A.; Domazetovska, A.; Cooper, S.T.; North, K.N.; Laing, N.G.; Gunning, P.W.; Hardeman, E.C. An alphaTropomyosin mutation alters dimer preference in nemaline myopathy. Ann. Neurol. 2005, 57, 42–49. [Google Scholar] [CrossRef]
- Pieples, K.; Arteaga, G.; Solaro, R.J.; Grupp, I.; Lorenz, J.N.; Boivin, G.P.; Jagatheesan, G.; Labitzke, E.; DeTombe, P.P.; Konhilas, J.P.; et al. Tropomyosin 3 expression leads to hypercontractility and attenuates myofilament length-dependent Ca(2+) activation. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H1344–H1353. [Google Scholar] [CrossRef]
- Matyushenko, A.M.; Shchepkin, D.V.; Kopylova, G.V.; Bershitsky, S.Y.; Levitsky, D.I. Unique functional properties of slow skeletal muscle tropomyosin. Biochimie 2020, 174, 1–8. [Google Scholar] [CrossRef]
- Moraczewska, J. Thin filament dysfunctions caused by mutations in tropomyosin Tpm3.12 and Tpm1.1. J. Muscle Res. Cell Motil. 2020, 41, 39–53. [Google Scholar] [CrossRef]
- Clarkson, E.; Costa, C.F.; Machesky, L.M. Congenital myopathies: Diseases of the actin cytoskeleton. J. Pathol. 2004, 204, 407–417. [Google Scholar] [CrossRef]
- Clarke, N.F. Skeletal muscle disease due to mutations in tropomyosin, troponin and cofilin. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2008; Volume 642, pp. 40–54. [Google Scholar] [CrossRef]
- Lawlor, M.W.; Dechene, E.T.; Roumm, E.; Geggel, A.S.; Moghadaszadeh, B.; Beggs, A.H. Mutations of tropomyosin 3 (TPM3) are common and associated with type 1 myofiber hypotrophy in congenital fiber type disproportion. Hum. Mutat. 2010, 31, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Phadke, R. Myopathology of Congenital Myopathies: Bridging the Old and the New. Semin. Pediatr. Neurol. 2019, 29, 55–70. [Google Scholar] [CrossRef]
- Schiaffino, S.; Reggiani, C. Molecular diversity of myofibrillar proteins: Gene regulation and functional significance. Physiol. Rev. 1996, 76, 371–423. [Google Scholar] [CrossRef] [PubMed]
- Siemankowski, R.F.; Wiseman, M.O.; White, H.D. ADP dissociation from actomyosin subfragment 1 is sufficiently slow to limit the unloaded shortening velocity in vertebrate muscle. Proc. Natl. Acad. Sci. USA 1985, 82, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Nyitrai, M.; Rossi, R.; Adamek, N.; Pellegrino, M.A.; Bottinelli, R.; Geeves, M.A. What limits the velocity of fast-skeletal muscle contraction in mammals? J. Mol. Biol. 2006, 355, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Iorga, B.; Adamek, N.; Geeves, M.A. The slow skeletal muscle isoform of myosin shows kinetic features common to smooth and non-muscle myosins. J. Biol. Chem. 2007, 282, 3559–3570. [Google Scholar] [CrossRef] [PubMed]
- Bloemink, M.J.; Adamek, N.; Reggiani, C.; Geeves, M.A. Kinetic analysis of the slow skeletal myosin MHC-1 isoform from bovine masseter muscle. J. Mol. Biol. 2007, 373, 1184–1197. [Google Scholar] [CrossRef]
- Huxley, H.E. Fifty years of muscle and the sliding filament hypothesis. Eur. J. Biochem. 2004, 271, 1403–1415. [Google Scholar] [CrossRef]
- Gokhin, D.S.; Kim, N.E.; Lewis, S.A.; Hoenecke, H.R.; D’Lima, D.D.; Fowler, V.M. Thin-filament length correlates with fiber type in human skeletal muscle. Am. J. Physiol. Cell Physiol. 2012, 302, C555–C565. [Google Scholar] [CrossRef]
- Granzier, H.L.; Akster, H.A.; Ter Keurs, H.E. Effect of thin filament length on the force-sarcomere length relation of skeletal muscle. Am. J. Physiol. 1991, 260, C1060–C1070. [Google Scholar] [CrossRef]
- Gregorio, C.C.; Weber, A.; Bondad, M.; Pennise, C.R.; Fowler, V.M. Requirement of pointed-end capping by tropomodulin to maintain actin filament length in embryonic chick cardiac myocytes. Nature 1995, 377, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Littlefield, R.S.; Fowler, V.M. Thin filament length regulation in striated muscle sarcomeres: Pointed-end dynamics go beyond a nebulin ruler. Semin. Cell Dev. Biol. 2008, 19, 511–519. [Google Scholar] [CrossRef]
- Tsukada, T.; Pappas, C.T.; Moroz, N.; Antin, P.B.; Kostyukova, A.S.; Gregorio, C.C. Leiomodin-2 is an antagonist of tropomodulin-1 at the pointed end of the thin filaments in cardiac muscle. J. Cell Sci. 2010, 123, 3136–3145. [Google Scholar] [CrossRef] [PubMed]
- Kremneva, E.; Makkonen, M.H.; Skwarek-Maruszewska, A.; Gateva, G.; Michelot, A.; Dominguez, R.; Lappalainen, P. Cofilin-2 controls actin filament length in muscle sarcomeres. Dev. Cell 2014, 31, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.B.; Joshi, M.; Savic, T.; Chen, Z.; Beggs, A.H. Normal myofibrillar development followed by progressive sarcomeric disruption with actin accumulations in a mouse Cfl2 knockout demonstrates requirement of cofilin-2 for muscle maintenance. Hum. Mol. Genet. 2012, 21, 2341–2356. [Google Scholar] [CrossRef]
- Gurniak, C.B.; Chevessier, F.; Jokwitz, M.; Jonsson, F.; Perlas, E.; Richter, H.; Matern, G.; Boyl, P.P.; Chaponnier, C.; Furst, D.; et al. Severe protein aggregate myopathy in a knockout mouse model points to an essential role of cofilin2 in sarcomeric actin exchange and muscle maintenance. Eur. J. Cell Biol. 2014, 93, 252–266. [Google Scholar] [CrossRef] [PubMed]
- Ono, S.; Minami, N.; Abe, H.; Obinata, T. Characterization of a novel cofilin isoform that is predominantly expressed in mammalian skeletal muscle. J. Biol. Chem. 1994, 269, 15280–15286. [Google Scholar]
- Vartiainen, M.K.; Mustonen, T.; Mattila, P.K.; Ojala, P.J.; Thesleff, I.; Partanen, J.; Lappalainen, P. The three mouse actin-depolymerizing factor/cofilins evolved to fulfill cell-type-specific requirements for actin dynamics. Mol. Biol. Cell 2002, 13, 183–194. [Google Scholar] [CrossRef]
- Carlier, M.F.; Laurent, V.; Santolini, J.; Melki, R.; Didry, D.; Xia, G.X.; Hong, Y.; Chua, N.H.; Pantaloni, D. Actin depolymerizing factor (ADF/cofilin) enhances the rate of filament turnover: Implication in actin-based motility. J. Cell Biol. 1997, 136, 1307–1322. [Google Scholar] [CrossRef]
- Michelot, A.; Berro, J.; Guerin, C.; Boujemaa-Paterski, R.; Staiger, C.J.; Martiel, J.L.; Blanchoin, L. Actin-filament stochastic dynamics mediated by ADF/cofilin. Curr. Biol. 2007, 17, 825–833. [Google Scholar] [CrossRef]
- McGough, A.; Pope, B.; Chiu, W.; Weeds, A. Cofilin changes the twist of F-actin: Implications for actin filament dynamics and cellular function. J. Cell Biol. 1997, 138, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Andrianantoandro, E.; Pollard, T.D. Mechanism of actin filament turnover by severing and nucleation at different concentrations of ADF/cofilin. Mol. Cell 2006, 24, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Robaszkiewicz, K.; Ostrowska, Z.; Marchlewicz, K.; Moraczewska, J. Tropomyosin isoforms differentially modulate the regulation of actin filament polymerization and depolymerization by cofilins. FEBS J. 2016, 283, 723–737. [Google Scholar] [CrossRef]
- Ostrowska, Z.; Robaszkiewicz, K.; Moraczewska, J. Regulation of actin filament turnover by cofilin-1 and cytoplasmic tropomyosin isoforms. Biochim. Biophys. Acta Proteins Proteom. 2017, 1865, 88–98. [Google Scholar] [CrossRef]
- Monteiro, P.B.; Lataro, R.C.; Ferro, J.A.; Reinach Fde, C. Functional alpha-tropomyosin produced in Escherichia coli. A dipeptide extension can substitute the amino-terminal acetyl group. J. Biol. Chem. 1994, 269, 10461–10466. [Google Scholar]
- Robaszkiewicz, K.; Dudek, E.; Kasprzak, A.A.; Moraczewska, J. Functional effects of congenital myopathy-related mutations in gamma-tropomyosin gene. Biochim. Biophys. Acta 2012, 1822, 1562–1569. [Google Scholar] [CrossRef]
- Moraczewska, J.; Robaszkiewicz, K.; Śliwinska, M.; Czajkowska, M.; Ly, T.; Kostyukova, A.; Wen, H.; Zheng, W. Congenital myopathy-related mutations in tropomyosin disrupt regulatory function through altered actin affinity and tropomodulin binding. FEBS J. 2019. [Google Scholar] [CrossRef]
- Landis, C.A.; Bobkova, A.; Homsher, E.; Tobacman, L.S. The active state of the thin filament is destabilized by an internal deletion in tropomyosin. J. Biol. Chem. 1997, 272, 14051–14056. [Google Scholar] [CrossRef]
- Sliwinska, M.; Robaszkiewicz, K.; Czajkowska, M.; Zheng, W.; Moraczewska, J. Functional effects of substitutions I92T and V95A in actin-binding period 3 of tropomyosin. Biochim. Biophys. Acta 2018, 1866, 558–568. [Google Scholar] [CrossRef]
- Nakashima, K.; Sato, N.; Nakagaki, T.; Abe, H.; Ono, S.; Obinata, T. Two mouse cofilin isoforms, muscle-type (MCF) and non-muscle type (NMCF), interact with F-actin with different efficiencies. J. Biochem. 2005, 138, 519–526. [Google Scholar] [CrossRef]
- De La Cruz, E.M. Cofilin binding to muscle and non-muscle actin filaments: Isoform-dependent cooperative interactions. J. Mol. Biol. 2005, 346, 557–564. [Google Scholar] [CrossRef]
- De La Cruz, E.M. How cofilin severs an actin filament. Biophys. Rev. 2009, 1, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Mossakowska, M.; Moraczewska, J.; Khaitlina, S.; Strzelecka-Golaszewska, H. Proteolytic removal of three C-terminal residues of actin alters the monomer-monomer interactions. Biochem. J. 1993, 289 Pt 3, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Khaitlina, S.; Fitz, H.; Hinssen, H. The interaction of gelsolin with tropomyosin modulates actin dynamics. FEBS J. 2013, 280, 4600–4611. [Google Scholar] [CrossRef] [PubMed]
- Vrhovski, B.; Theze, N.; Thiebaud, P. Structure and evolution of tropomyosin genes. Adv. Exp. Med. Biol. 2008, 644, 6–26. [Google Scholar] [CrossRef]
- Hitchcock-DeGregori, S.E.; Singh, A. What makes tropomyosin an actin binding protein? A perspective. J. Struct. Biol. 2010, 170, 319–324. [Google Scholar] [CrossRef][Green Version]
- Barua, B.; Pamula, M.C.; Hitchcock-DeGregori, S.E. Evolutionarily conserved surface residues constitute actin binding sites of tropomyosin. Proc. Natl. Acad. Sci. USA 2011, 108, 10150–10155. [Google Scholar] [CrossRef]
- Marston, S.; Memo, M.; Messer, A.; Papadaki, M.; Nowak, K.; McNamara, E.; Ong, R.; El-Mezgueldi, M.; Li, X.; Lehman, W. Mutations in repeating structural motifs of tropomyosin cause gain of function in skeletal muscle myopathy patients. Hum. Mol. Genet. 2013, 22, 4978–4987. [Google Scholar] [CrossRef]
- McGhee, J.D.; von Hippel, P.H. Theoretical aspects of DNA-protein interactions: Co-operative and non-co-operative binding of large ligands to a one-dimensional homogeneous lattice. J. Mol. Biol. 1974, 86, 469–489. [Google Scholar] [CrossRef]
- Bobkov, A.A.; Muhlrad, A.; Pavlov, D.A.; Kokabi, K.; Yilmaz, A.; Reisler, E. Cooperative effects of cofilin (ADF) on actin structure suggest allosteric mechanism of cofilin function. J. Mol. Biol. 2006, 356, 325–334. [Google Scholar] [CrossRef]
- Galkin, V.E.; Orlova, A.; Kudryashov, D.S.; Solodukhin, A.; Reisler, E.; Schroder, G.F.; Egelman, E.H. Remodeling of actin filaments by ADF/cofilin proteins. Proc. Natl. Acad. Sci. USA 2011, 108, 20568–20572. [Google Scholar] [CrossRef] [PubMed]
- McCullough, B.R.; Grintsevich, E.E.; Chen, C.K.; Kang, H.; Hutchison, A.L.; Henn, A.; Cao, W.; Suarez, C.; Martiel, J.L.; Blanchoin, L.; et al. Cofilin-linked changes in actin filament flexibility promote severing. Biophys. J. 2011, 101, 151–159. [Google Scholar] [CrossRef]
- Kang, H.; Bradley, M.J.; Cao, W.; Zhou, K.; Grintsevich, E.E.; Michelot, A.; Sindelar, C.V.; Hochstrasser, M.; De La Cruz, E.M. Site-specific cation release drives actin filament severing by vertebrate cofilin. Proc. Natl. Acad. Sci. USA 2014, 111, 17821–17826. [Google Scholar] [CrossRef] [PubMed]
- Huehn, A.R.; Bibeau, J.P.; Schramm, A.C.; Cao, W.; De La Cruz, E.M.; Sindelar, C.V. Structures of cofilin-induced structural changes reveal local and asymmetric perturbations of actin filaments. Proc. Natl. Acad. Sci. USA 2020, 117, 1478–1484. [Google Scholar] [CrossRef]
- Von der Ecken, J.; Heissler, S.M.; Pathan-Chhatbar, S.; Manstein, D.J.; Raunser, S. Cryo-EM structure of a human cytoplasmic actomyosin complex at near-atomic resolution. Nature 2016, 534, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Ngo, K.X.; Umeki, N.; Kijima, S.T.; Kodera, N.; Ueno, H.; Furutani-Umezu, N.; Nakajima, J.; Noguchi, T.Q.; Nagasaki, A.; Tokuraku, K.; et al. Allosteric regulation by cooperative conformational changes of actin filaments drives mutually exclusive binding with cofilin and myosin. Sci. Rep. 2016, 6, 35449. [Google Scholar] [CrossRef]
- Ono, S.; Ono, K. Tropomyosin inhibits ADF/cofilin-dependent actin filament dynamics. J. Cell Biol. 2002, 156, 1065–1076. [Google Scholar] [CrossRef]
- Tanaka, K.; Takeda, S.; Mitsuoka, K.; Oda, T.; Kimura-Sakiyama, C.; Maeda, Y.; Narita, A. Structural basis for cofilin binding and actin filament disassembly. Nat. Commun. 2018, 9, 1860. [Google Scholar] [CrossRef]
- Moraczewska, J.; Gruszczynska-Biegala, J.; Redowicz, M.J.; Khaitlina, S.Y.; Strzelecka-Golaszewska, H. The DNase-I binding loop of actin may play a role in the regulation of actin-myosin interaction by tropomyosin/troponin. J. Biol. Chem. 2004, 279, 31197–31204. [Google Scholar] [CrossRef]
- Lehman, W.; Li, X.E.; Orzechowski, M.; Fischer, S. The structural dynamics of alpha-tropomyosin on F-actin shape the overlap complex between adjacent tropomyosin molecules. Arch. Biochem. Biophys. 2014, 552–553, 68–73. [Google Scholar] [CrossRef][Green Version]
- Hitchcock-DeGregori, S.E.; Sampath, P.; Pollard, T.D. Tropomyosin inhibits the rate of actin polymerization by stabilizing actin filaments. Biochemistry 1988, 27, 9182–9185. [Google Scholar] [CrossRef] [PubMed]
- Lal, A.A.; Korn, E.D. Effect of muscle tropomyosin on the kinetics of polymerization of muscle actin. Biochemistry 1986, 25, 1154–1158. [Google Scholar] [CrossRef] [PubMed]
- Ostrowska-Podhorodecka, Z.; Sliwinska, M.; Reisler, E.; Moraczewska, J. Tropomyosin isoforms regulate cofilin 1 activity by modulating actin filament conformation. Arch. Biochem. Biophys. 2020, 682, 108280. [Google Scholar] [CrossRef] [PubMed]
- Ly, T.; Pappas, C.T.; Johnson, D.; Schlecht, W.; Colpan, M.; Galkin, V.E.; Gregorio, C.C.; Dong, W.J.; Kostyukova, A.S. Effects of cardiomyopathy-linked mutations K15N and R21H in tropomyosin on thin-filament regulation and pointed-end dynamics. Mol. Biol. Cell 2019, 30, 268–281. [Google Scholar] [CrossRef]
- Spudich, J.A.; Watt, S. The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J. Biol. Chem. 1971, 246, 4866–4871. [Google Scholar]
- Margossian, S.S.; Lowey, S. Preparation of myosin and its subfragments from rabbit skeletal muscle. Methods Enzymol. 1982, 85 Pt B, 5571–5727. [Google Scholar] [CrossRef]
- Muhlrad, A.; Kudryashov, D.; Michael Peyser, Y.; Bobkov, A.A.; Almo, S.C.; Reisler, E. Cofilin induced conformational changes in F-actin expose subdomain 2 to proteolysis. J. Mol. Biol. 2004, 342, 1559–1567. [Google Scholar] [CrossRef]
- Skorzewski, R.; Sliwinska, M.; Borys, D.; Sobieszek, A.; Moraczewska, J. Effect of actin C-terminal modification on tropomyosin isoforms binding and thin filament regulation. Biochim. Biophys. Acta 2009, 1794, 237–243. [Google Scholar] [CrossRef]

| − Cofilin-2 | + Cofilin-2 | |||
|---|---|---|---|---|
| Kapp (µM−1) | αH | Kapp (µM−1) | αH | |
| Tpm1.1 | 10.2 ± 0.9 a,b | 1.7 ± 0.3 b | 5.6 ± 0.9 a,b | 0.9 ± 0.1 a,b |
| Tpm3.12 | 19.1 ± 1.6 a,b | 1.3 ± 0.2 | 13.9 ± 0.8 a,b | 1.8 ± 0.2 a |
| Kapp (µM−1) | Κ0 (µM−1) | ω | |
|---|---|---|---|
| Cofilin-2 | 5.7 ± 0.2 | 0.01 | 50 |
| Cofilin-2 + Tpm1.1 | 6.3 ± 0.4 | 0.17 | 12 |
| Cofilin-2 + Tpm3.12 | 5.8 ± 0.2 | 0.21 | 10 |
| 50% Dissociation of Tropomyosin (Cofilin-2/Actin) | αH | |
|---|---|---|
| Tpm1.1 | 0.75 ± 0.04 | 1.9 ± 0.3 |
| Tpm3.12 | 0.71 ± 0.03 | 1.9 ± 0.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robaszkiewicz, K.; Śliwinska, M.; Moraczewska, J. Regulation of Actin Filament Length by Muscle Isoforms of Tropomyosin and Cofilin. Int. J. Mol. Sci. 2020, 21, 4285. https://doi.org/10.3390/ijms21124285
Robaszkiewicz K, Śliwinska M, Moraczewska J. Regulation of Actin Filament Length by Muscle Isoforms of Tropomyosin and Cofilin. International Journal of Molecular Sciences. 2020; 21(12):4285. https://doi.org/10.3390/ijms21124285
Chicago/Turabian StyleRobaszkiewicz, Katarzyna, Małgorzata Śliwinska, and Joanna Moraczewska. 2020. "Regulation of Actin Filament Length by Muscle Isoforms of Tropomyosin and Cofilin" International Journal of Molecular Sciences 21, no. 12: 4285. https://doi.org/10.3390/ijms21124285
APA StyleRobaszkiewicz, K., Śliwinska, M., & Moraczewska, J. (2020). Regulation of Actin Filament Length by Muscle Isoforms of Tropomyosin and Cofilin. International Journal of Molecular Sciences, 21(12), 4285. https://doi.org/10.3390/ijms21124285




