1. Introduction
Osteoporosis is a metabolic bone disease associated with a lack of balance between bone resorption and formation that results in low bone mass, structural deterioration of the bone, and fragility fractures (fractures after a small trauma, such as a fall from a low height, or from no identifiable trauma). Osteoporotic fractures (especially in the vertebra or hip) lead to substantial morbidity, mortality, and socioeconomic burdens [
1,
2,
3,
4,
5]. Current pharmacological options target either osteoclasts, by bone resorption inhibition (antiresorptive agents), or osteoblasts, by bone formation stimulation (osteoanabolic agents) [
4,
6,
7,
8,
9]. Most recent pharmacological approaches are typically based on anti-resorptive agents such as selective estrogen receptor modulators (SERMs), bisphosphonates, and denosumab (the receptor activator of the nuclear factor-κB ligand (RANKL) inhibitor). Antiresorptive agents can suppress bone turnover and osteoclastic-mediated bone breakdown, which might lead to unwanted side effects including atypical femur fractures (bisphosphonates, denosumab), osteonecrosis of the jaw, and venous thromboembolism (SERMs). Furthermore, there have been controversies about the effect of SERMS on non-vertebral fractures [
4,
6,
7,
8]. There are currently three US FDA-approved anabolic therapies, namely, teriparatide (recombinant human parathyroid hormone, rhPTH), abaloparatide (PTH-related protein, PTHrP), and romosozumab (anti-human scleroscin antibody) [
6,
7,
8,
9,
10]. Although osteoanabolic agents can reduce the prevalence of nonvertebral and vertebral fractures, there are still concerns about their safety due to a greater risk of osteosarcoma, hypercalcemia (teriparatide, abaloparatide), and cardiovascular events (romosozumab) [
6,
7,
8,
9,
10]. Due to the limitations and undesirable effects associated with current pharmacological treatments, safe and effective alternatives are highly desirable to prevent and treat osteoporosis.
Tauroursodeoxycholic acid (TUDCA) is a taurine conjugate form of ursodeoxycholic acid (UDCA) and is an endogenous hydrophilic tertiary bile acid that is produced in humans at a low concentration. TUDCA is a commercially available bile acid derivative and has been used widely in treating cholestatic liver disease and cholelithiasis. In a multicenter study, the TUDCA was as safe and efficacious as ursodeoxycholic acid (UDCA) in patients with primary biliary cholangitis, and it was better for relieving symptoms than UDCA [
11]. Studies have reported that TUDCA could have a crucial role in preventing apoptosis [
12], suppressing adipogenesis of adipose-derived mesenchymal stem cells (MSCs) [
13], promoting angiogenesis [
14,
15,
16,
17], and enhancing osteogenic differentiation of bone marrow-derived MSCs [
16]. In our previous study on TUDCA in a mouse spinal fusion model, we suggested that TUDCA could induce bone formation and was superior to the recombinant human bone morphogenetic protein-2 (rhBMP-2), in terms of bone quality, because it promoted angiogenesis and increased trabecular bone thickness [
18]. However, to our knowledge, it is not evident whether TUDCA has therapeutic effects on ovariectomy (OVX)-induced osteoporosis.
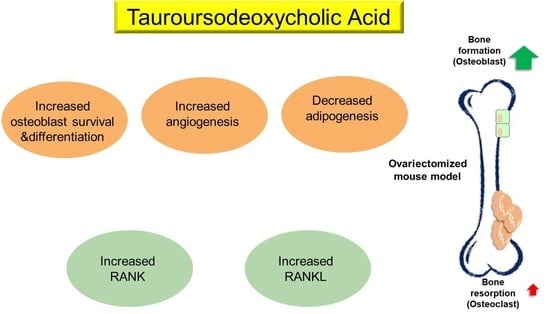
The present study showed that TUDCA has remarkable anti-osteoporotic effects in an OVX-induced osteoporosis mice model. To further make clear the mechanisms underlying the anti-osteoporotic effects of TUDCA, we also investigated the effects of TUDCA on osteoblasts and methylprednisolone (MP)-induced apoptosis in primary cultures of rat osteoblasts.
3. Discussion
Postmenopausal osteoporosis is associated with low bone mass that results from increased resorption of bone and decreased formation of bone, eventually leading to a greater risk of fractures [
1,
2,
3,
4,
5]. Promising pharmacotherapies for postmenopausal osteoporosis have improved health conditions for all patients [
6,
7,
8,
9,
10]. However, certain drugs may not be appropriate for all patients and may cause various side effects in some individuals. Thus, it is still necessary to study novel drugs that are effective and safe for the treatment of postmenopausal osteoporosis. Here, we suggested that TUDCA could promote osteoblast viability and differentiation, protect osteoblasts from MP-induced apoptosis in vitro, and clearly improve OVX-induced osteoporosis in a mouse model. To our knowledge, this is the first study that identifies the anti-osteoporotic effects of TUDCA.
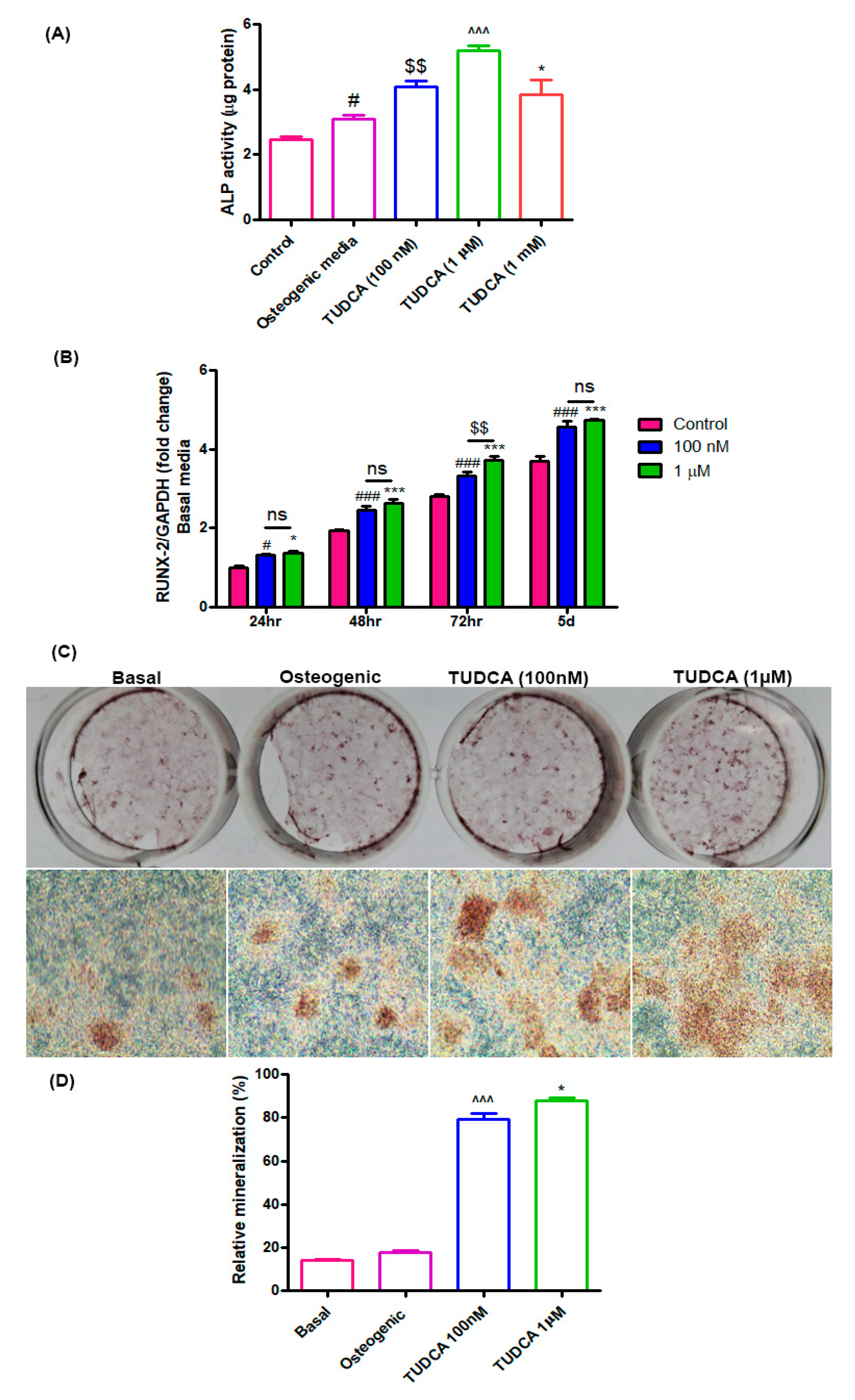
In the current study, we first explored the effects of TUDCA on osteoblast viability and differentiation in vitro. RUNX2 is a critical transcription factor that is presented in the earliest stage of osteogenic differentiation, and ALP is an early marker of osteogenic differentiation [
19]. Therefore, ALP activity was evaluated after 5 days of incubation, and the expression of the important osteoblast-specific gene RUNX2 was measured at 24, 48, and 72 h and 5 days of incubation. On day 5, TUDCA significantly increased ALP activity and RUNX2 expression. In addition, TUDCA at 100 nM and 1 µM concentrations improved bone mineralization and 1 mM TUDCA protected osteoblasts from MP-induced apoptosis. TUDCA has been proven to inhibit endoplasmic reticulum (ER) stress, which is associated with osteoclastogenesis [
20]. An inducer of ER stress (thapsigargin) was demonstrated to decrease osteoclast formation from RAW264.7 macrophages [
21]. The inhibition of ER stress could induce the downregulation of monocyte chemotactic protein-1 (MCP-1)-mediated osteoclast precursor differentiation [
22]. In a recent report, Wang et al. demonstrated that the inhibition of ER stress by TUDCA significantly decreased the formation of osteoclasts medicated by TiAl6V4 alloy particles (
TiPs) in vitro and in vivo [
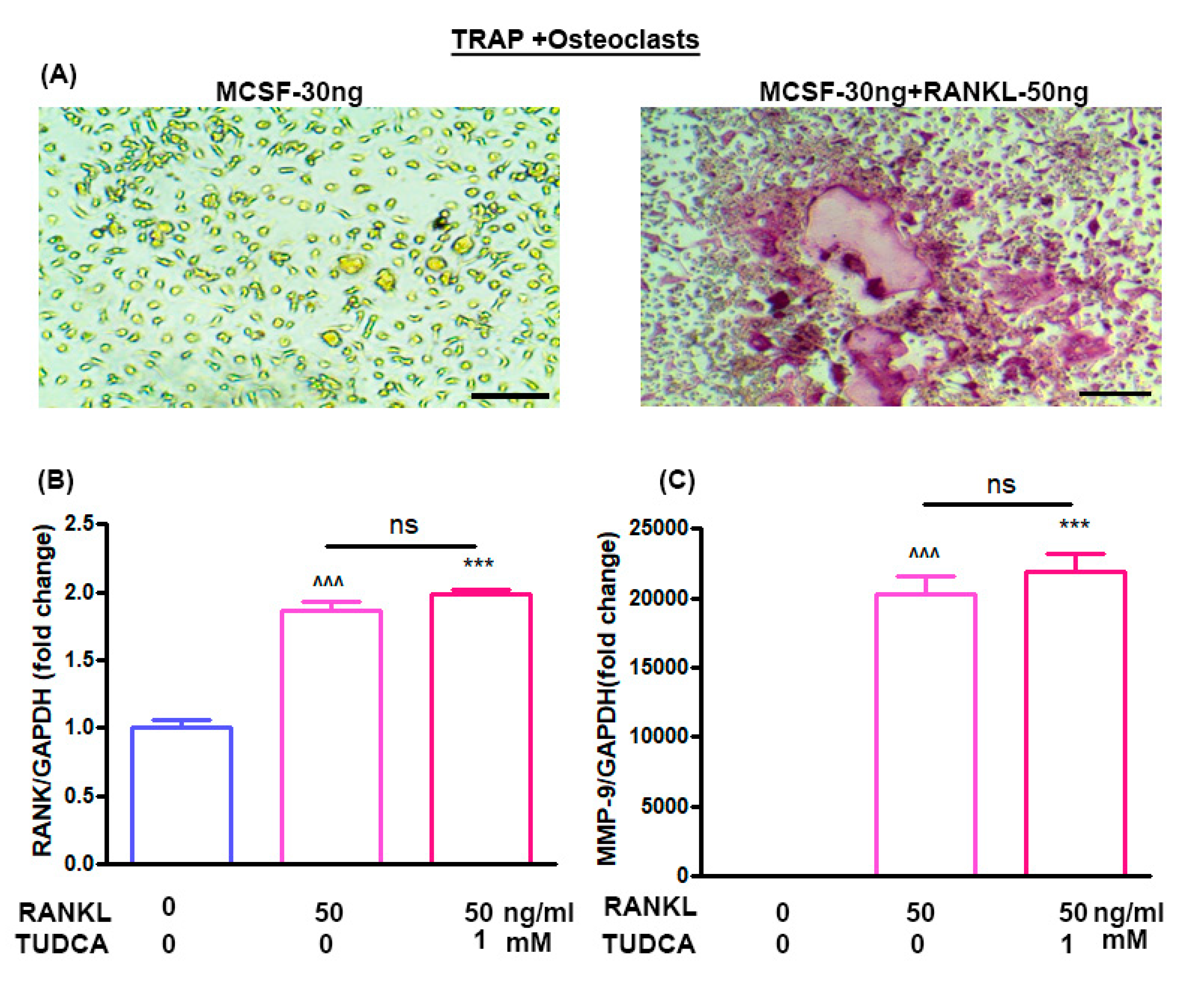
23]. Thus, in our study, we also analyzed the effects of TUDCA on osteoclasts and found that TUDCA did not significantly inhibit the expression of osteoclast-specific genes, such as MMP-9 and RANK. All of these in vitro results suggested that TUDCA could play an important role in promoting osteoblast viability and differentiation as well as in preventing glucocorticoid-induced osteoblast apoptosis.
In the present study, we did not evaluate the effect of TUDCA on inflammatory reaction. It is well known that TUDCA has a powerful anti-inflammatory effect [
24]. Human and animal experiments showed pro-inflammatory cytokines as primary mediators of the accelerated bone loss at post-menopause including interleukin-1, tumor necrosis factor-alpha, and interleukin-6. These pro-inflammatory cytokines are associated with osteoclastic bone resorption in various situations [
25]. Therefore, the anti-inflammatory effect of TUDCA may be one of its anti-osteoporotic effects. It should be evaluated in a future study.
Bilateral ovariectomy was performed to establish an OVX mouse model of osteoporosis. Ovariectomy-induced osteoporosis in rodents produces skeletal responses that are similar to those in post-menopausal women [
26]. Both structural and mechanical investigations can be easily performed on the distal femur of rodents due to the geometry of the bone and the greater amount of tissue available for sampling. Hence, in our OVX-induced osteoporosis mice model, the distal femur was chosen to analyze the effect of TUDCA on OVX-induced osteoporosis. BMD is considered an important indicator that reflects bone metabolism status and is widely used to analyze changes in bone mass and predict fracture risk [
1,
2,
3,
4,
18,
24]. In addition, the levels of PINP and CTX are commonly used as biochemical markers for bone resorption and formation, respectively [
1,
2,
3,
4,
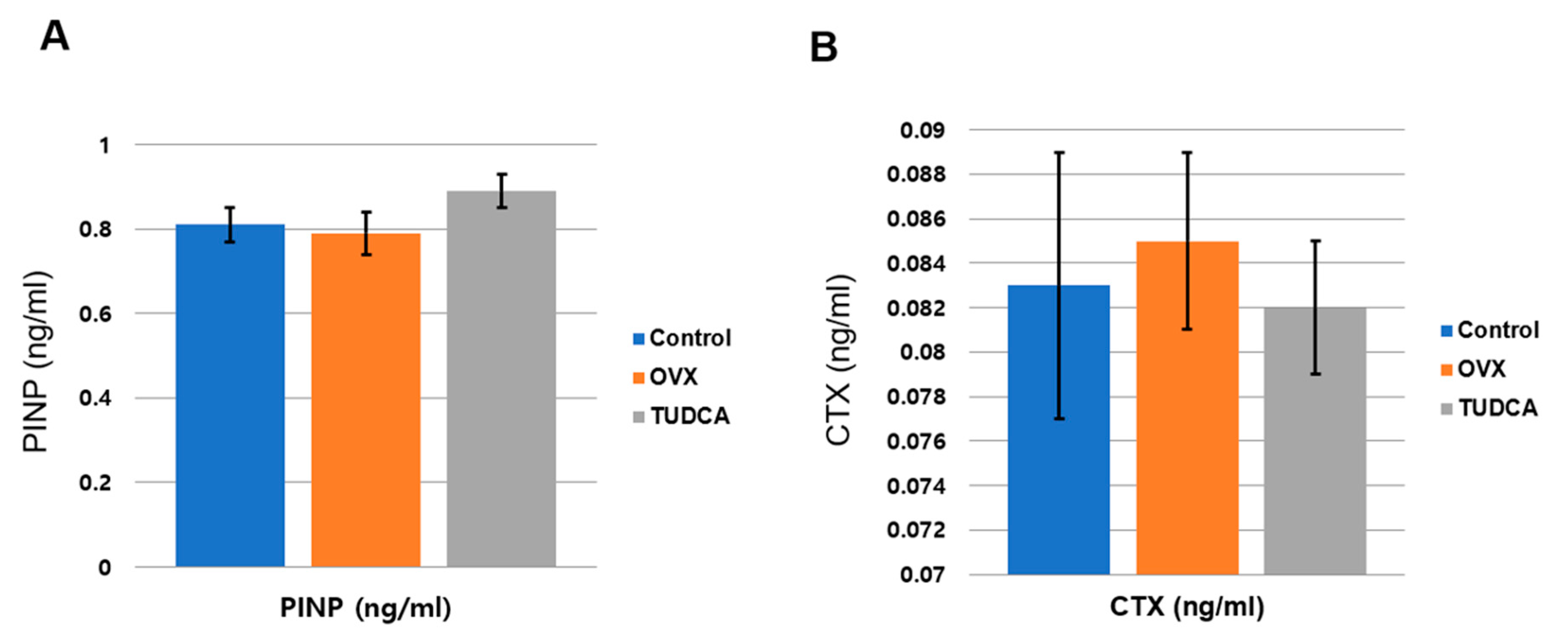
27]. Thus, we analyzed the effect of TUDCA on the levels of PINP and CTX. The primary findings of our in vivo study are as follows: (i) TUDCA remarkably reversed the ovariectomy-induced reduction in BMD; (ii) TUDCA not only elevated the PINP levels but also reduced the CTX levels in an OVX-induced osteoporosis model, even though this did not result in a statistical difference due to small number of animals (8/each group); (iii) TUDCA treatment clearly preserved trabecular microstructures in vivo. Taken together, our results indicate that TUDCA could alleviate OVX-induced osteoporosis in a mice model.
Although the underlying mechanisms implicated in the anti-osteoporotic effects of TUDCA remain unclear, we suggest several possibilities based on previous studies and our current results: (i) promotion of osteoblast survival and inhibition of osteoblast apoptosis, (ii) suppression of adipogenesis in adipose tissue-derived MSCs by modulating endoplasmic reticulum (ER) stress, (iii) promotion of angiogenesis by recruiting vasculogenic progenitor cells, (iv) promotion of osteogenic differentiation from MSCs, and (v) its osteogenic potential by inducing new bone formation [
15,
16,
17]. Currently available bone-forming agents such as teriparatide, abaloparatide, and romosozumab have different effects on bone surfaces [
6,
7]. Teriparatide and abaloparatide improve bone mass by inducing greater bone formation than resorption [
28]. Teriparatide stimulates osteoblast number and activity, and can stimulate RANKL production from the osteoblasts, leading to bone resorption [
16,
17,
18]. Abaloparatide could cause greater net bone formation than resorption compared to teriparatide [
9]. In contrast, romosozumab acts primarily by activating bone formation while inhibiting bone resorption [
6]. In this study, we found no evidence that TUDCA inhibited the expression of osteoclast-specific genes, including MMP-9 and RANK. However, we observed that the TUDCA-treated group had higher levels of MMP-9 and RANK, even though there was no statistical significance. These findings raise the possibility that TUDCA could stimulate an increase in osteoblast number and activity as well as RANKL production from the osteoblasts, possibly leading to bone resorption. However, TUDCA induced greater net bone formation than resorption, similar to abaloparatide.
4. Materials and Methods
4.1. Drugs
The drugs used in the current study were tauroursodeoxycholic acid (TUDCA; Tokyo Chemical Industry Co., Tokyo, Japan) and methylprednisolone (MP; corticosteroid).
4.2. Isolation and Culture of Osteoblasts from Rat Calvaria
Rat calvaria were collected from 3-day-old Sprague-Dawley rats. All procedures involving animals were performed following the protocol approved by the Institutional Animal Care and Use Committee (IACUC) of CHA University (IACUC 190076, March, 2019). After removal of sutures and adherent mesenchymal tissues and surgical isolation from the skull, the calvaria were introduced with 5 sequential (10–15 min) digestions at 37 °C in a solution containing 0.1% dispase and 0.1% collagenase P. Cells released from the second to the fifth digestions were centrifuged, collected, and resuspended. Those cells were cultured and incubated in proliferation medium (DMEM (Gibco BRL) supplemented with 10% (v/v) fetal bovine serum (FBS, Gibco BRL) and 100 units/mL penicillin (GibcoBRL)) or osteogenic medium (DMEM supplemented with 10% (v/v) FBS, 0.2 mM ascorbic acid (Sigma, St. Louis, MO, USA)), 1% GlutaMAX (GibcoBRL), 10 mM glycerol 2-phosphate (Sigma), and 100 units/mL penicillin (GibcoBRL) in humidified air with 5% (v/v) CO2 at 37 °C until the cells reached confluency.
4.3. Assay for Osteoblast Proliferation and Viability
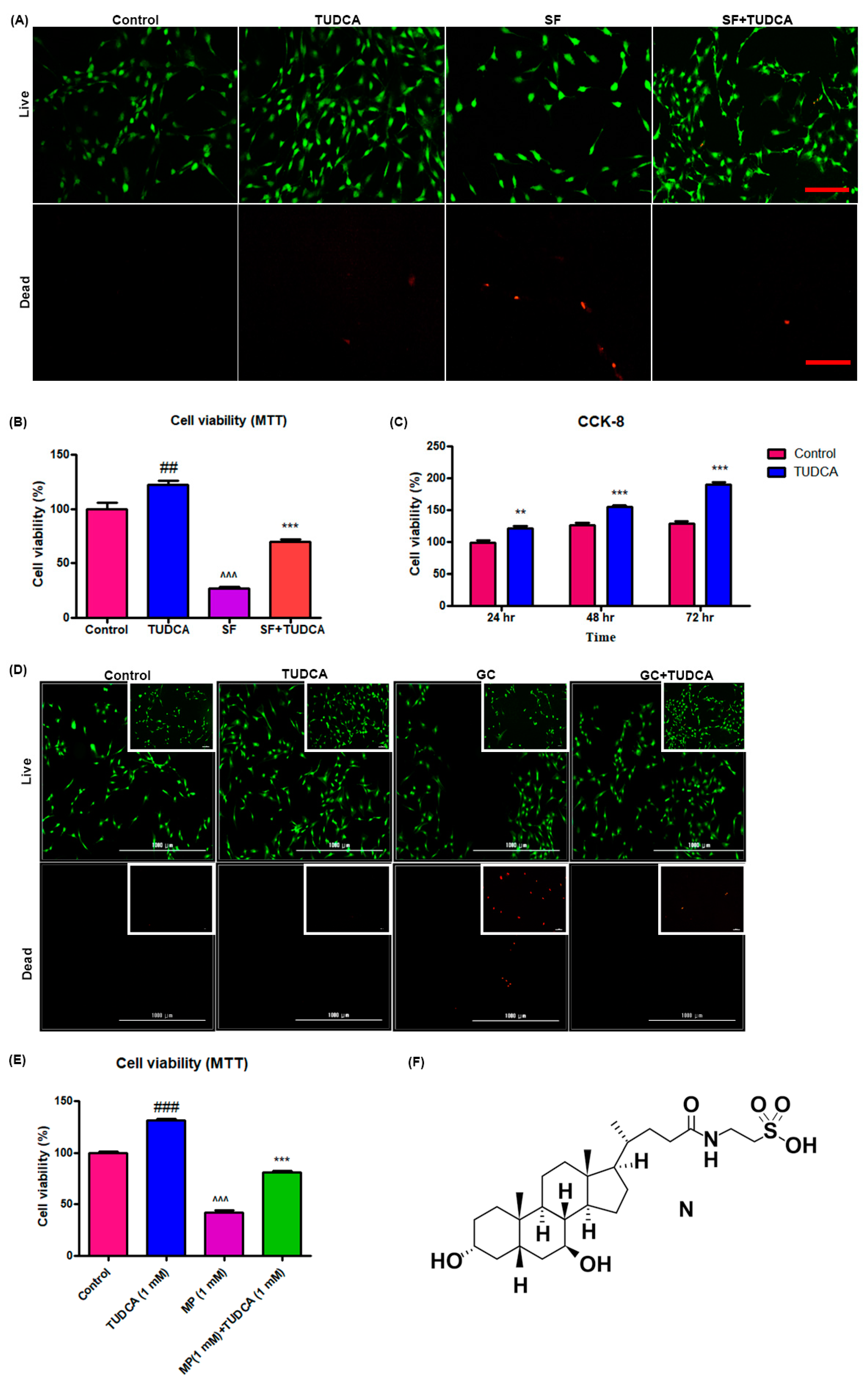
To determine the effect of 1 mM TUDCA on the proliferation and viability of osteoblasts, the cells were seeded at a density of 1 × 103 cells/cm2 on cell culture plates and incubated for 24 h under the following four conditions: (i) DMEM + 10% fetal calf serum (FCS; control), (ii) DMEM + 10% FCS with 1 mM TUDCA, (iii) DMEM only (serum-free), and (iv) DMEM only (serum-free) with 1 mM TUDCA. After 24 h of TUDCA treatment (Calbiochem, San Diego, CA), the survival of the cells in each group was visualized using the Zeiss LSM510 Meta laser scanning confocal microscope (Carl Zeiss Microimaging Inc., Göttingen, Germany) equipped with an argon laser light source after staining with fluorescein diacetate/ethidium bromide (Sigma). Cell viability was determined using a Cell Counting Kit-8 (CCK-8, Dojindo, Kumamoto, Japan) following the manufacturer’s guidance. The CCK-8 assay was used on cells cultured for different durations (72, 48, and 24 h) and the cell viability was presented as a percentage of control cells.
Cell proliferation and cell viability were analyzed using an MTT assay at 3 days of cell culture under four different conditions (control, serum-free media with TUDCA, serum-free media, and basal media with TUDCA). In addition, the effect of TUDCA on MP-induced cell apoptosis was evaluated. Osteoblasts were incubated in 96-well plates, maintained in growth media at 37 °C, and treated with 1 mM MP as well as different concentrations of TUDCA (1 μm, 10 μm, 100 μm, and 1 mM) for 72 h. Absorbance at an emission wavelength of 450 nm was measured by a microplate reader (Synergy H1 Hybrid Reader, BioTek, Winooski, VT, USA). Cell viability was presented as a percentage of the control cells or the ratio of the optical densities.
4.4. Alkaline Phosphatase Activity Assay
The alkaline phosphatase (ALP) activity of the cells grown with different concentrations of TUDCA was measured after 5 days. The cells were washed with PBS and lysed with 0.2 mL 0.5% Triton X-100 in PBS. The lysed cells were sonicated for 60 s and then centrifuged at 15,000× g for 10 min at 4 °C to remove insoluble material. The supernatant was mixed with p-nitrophenol phosphate disodium (p-NPP) substrate and incubated at 25 °C for 60 min. The optical density was spectrophotometrically determined at 405 nm using a μQuant microplate reader (μQuant, Bio-Tek, Winooski, VT, USA). The ALP activity was recorded as per μg total protein for each sample.
4.5. Alizarin Red-S Staining
To analyze mineralized nodule formation, rat osteoblasts were grown under the following four conditions: (i) basal media, (ii) osteogenic media, (iii) basal media with 100 nM TUDCA, and (iv) basal media with 1 µM TUDCA. On incubation day 14, the cells were fixed with 4% paraformaldehyde (PFA) at room temperature and stained with Alizarin Red-S (Sigma) at room temperature for 30 min. The mineralized nodules were examined under a microscope (magnification, ×100) and the relative value of mineralization was ascertained by determining the relative intensity of staining with image processing and analysis software (ImageJ version 1.8.0; National Institutes of Health, Bethesda, MD, USA).
4.6. Mouse Bone Marrow Cell Isolation
Eight-week-old Balb/c male mice were obtained from Orient Bio (Orient Bio, Inc., Seongnam, Korea). The femurs and tibias were harvested into α-Minimal Essential Medium (α-MEM; Sigma) containing 10% FBS, 100 ug/mL streptomycin, and 100 IU/mL penicillin G (culture medium) after the removal of excessive tissue. Bone marrow cells were collected from the cancellous bone marrow by flushing with culture medium using a 26-gauge needle. The bone marrow cells (2 × 105) were cultured in 1 mL of culture medium with a macrophage colony-stimulating factor (M-CSF, 30 ng/mL) and RANKL (50 ng/mL) without or with TUDCA. Cultures were maintained at 37 °C in a humidified atmosphere of 5% CO2 in air for 6 days. Adherent cells were defined as bone marrow-derived macrophages. After 6 days, real-time polymerase chain reaction (PCR) was done to evaluate the gene markers RANK and matrix metalloproteinase-9 (MMP-9).
4.7. Real Time PCR
Whole RNA was extracted from transfected cells using TRIzol (Invitrogen), and 2 μg of total RNA was used for cDNA synthesis with RT-PreMix (Bioneer, Daejeon, Korea). PCR was done with PCR-PreMix (Bioneer) under typical PCR conditions. The PCR cycles consisted of an initial denaturation step at 94 °C for 5 min, followed by 32 amplification cycles consisting of 30 s of denaturation at 94 °C, 30 s of annealing at 62 °C, and 1 min of extension at 72 °C. A final extension was performed at 72 °C for 10 min. PCR products were evaluated by UV irradiation on a 1.2% agarose gel stained with ethidium bromide. For quantitative real-time PCR analysis, gene-specific primers were designed to amplify runt-related transcription factor 2 (RUNX2), glyceraldehyde 3-phosphate dehydrogenase (GAPDH), RANK, and MMP-9. Primer pairs were as follows: GAPDH (5′-ACA TCG CTC AGA CAC CAT G-3, 5-TGT AGT TGA GGT CAA TGA AGG G-3), RANK (5-AAA CCT TGG ACC AAC TGC AC-3, 5-ACC ATC TTC TCC TCC CHA GT-3), and MMP-9 (5-CGA CTT TTG TGG TCT TCC CC-3, 5-TGA AGG TTT GGA ATC GAC CC-3). All amplifications were done in a final reaction mixture (20 μL) containing one final concentration of SYBR supermix (Thermo Fischer Scientific, Waltham, MA, USA), 500 nmol/L of gene-specific primers, and 1 mL of template, using the following conditions: an initial denaturation at 95 °C for 1 min, followed by 45 cycles of 56 °C for 15 s, 95 °C for 15 s, and 72 °C for 15 s, with a final extension at 72°C for 5 min. After amplification, the threshold and baseline levels for each reaction were decided using a software package provided with the PCR system (Exicycler 96; Bioneer, Daejeon, Korea). To validate the PCR results, the amplified products were separated on 1% agarose gels and visualized by ethidium bromide staining [
1,
2,
3,
5,
6].
4.8. Experimental Animals
Three-month-old female C57BL/6 mice (n = 24) were obtained from Orient Bio Inc. (Seongnam, Korea). All animal experiments were undertaken according to a protocol approved by the IACUC of our institute (IACUC170005) and the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Bethesda, MD, USA). All mice were maintained at 20 °C on a 12-h light/12-h dark cycle with free access to food and water in a pathogen-free ventilated cage.
4.9. Surgical Procedure and Experimental Design
After 2 weeks of stabilization, 16 mice were subjected to OVX procedure. Anesthesia was initiated in a 5% gaseous isoflurane-filled holding chamber and was maintained with 3%–4% gaseous isoflurane through a nose cone. The ventral surface of the mouse was shaved and disinfected using an alcohol and betadine solution. Buprenorphine 0.05 mg/kg was given subcutaneously. An incision was made between the thorax and the hind limb on the left flank of each mouse. Blunt forceps were then used to extract the left ovary along with the surrounding fat pad. The uterine artery and oviduct were tied using 4-0 vicryl sutures (Ethicon), and the fat pad was replaced after hemostasis. The fascia and skin were closed using 4-0 vicryl sutures (Ethicon) and a skin clip, respectively. Immediately afterward, the same procedure was performed on the right flank to extract the right ovary18.
The mice were divided randomly into three different groups: (i) control group without any operation or drug administration (n = 8); (ii) untreated OVX group (with vehicle (receiving phosphate-buffered saline [PBS]) treatment) (n = 8); and (iii) OVX with TUDCA treatment group (n = 8). Six weeks after the OVX surgery, the untreated OVX group was intraperitoneally injected with 1 mL of PBS and the OVX-TUDCA group was intraperitoneally injected with 1 mL of PBS and 200 mg/kg TUDCA. All intraperitoneal injections were conducted every other day for a total of 4 weeks. Serum analyses were performed for the biomarkers of bone resorption and bone formation at 11 weeks post-surgery. Animals were sacrificed with a CO2 overdose 1 week after the final injection. The right distal femurs were harvested and the effect of TUDCA on bone formation was evaluated by micro-computed tomography (CT) and histologic analyses.
4.10. Histomorphometric Analysis Using Micro-Computed Tomography
After TUDCA injections for a total of 4 weeks, the mice were sacrificed 1 week after the final injection. The right femur bones were harvested and the samples were fixed in 4% PFA for 48 h then stored in 70% ethanol for scanning by micro-CT. The metaphysis region of the right distal femur was scanned using a high-resolution micro-CT (SkyScan 1173, Bruker MicroCT N.V., Kontich, Belgium) at an image resolution of 9 µm (55 kV and 181 mA radiation source; 0.5 mm aluminum filter). A global threshold of 60 (1.01573 g/cm3) was applied to all scans to obtain a physiologically accurate representation of the trabecular bone. Morphometric parameters were then determined from binarized images using direct 3D techniques (marching cubes and sphere-fitting methods). The computed parameters were the bone mineral density (BMD, g/cm3), percent bone volume/tissue volume (BV/TV, %), trabecular number (Tb.N, mm−1), trabecular thickness (Tb.Th, mm), and trabecular separation (Tb.Sp, mm).
4.11. Analysis of Bone Formation and Resorption Markers
Blood samples were obtained in a serum separator tube from the direct cardiac puncture when the mice were sacrificed. Sera were stored at −83 °C until analyses. Serum type I procollagen N-terminal propeptides (PINP, bone formation marker) were evaluated using a mouse enzyme immunoassay (EIA) kit (Immunodiagnostic Systems Ltd., Boldon, UK). Type I collagen crosslinked C-telopeptide (CTX, bone resorption marker) levels in the serum were measured using the Serum CrossLaps® (CTX-I) ELISA kit (Immunodiagnostic Systems Ltd., Boldon, UK).
4.12. Histological Examinations
After micro-CT scanning, the femurs were decalcified by immersion in a decalcification solution (National Diagnostics, Atlanta, GA, USA). The tissues were placed in a dehydrated graded series of ethanol and xylene and embedded in paraffin. Axial sections (4 µum thickness) were then obtained. The sections were stained with hematoxylin and eosin (H&E) staining and Masson’s trichrome stain to analyze new bone formation. All images were examined using an Olympus BX51 microscope (Olympus Corporation, Japan) and photomicrographs were obtained using a MicroFire digital camera with PictureFrame software (Optronics, Fremont, CA, USA).
4.13. Statistical Analysis
Data were collected and statistically presented as mean ± standard error (S.E). One-way analysis of variance (ANOVA) was used for the analysis of quantitative values, and Tukey’s honestly significant difference post-hoc test was used for all pair-wise comparisons among the groups. The Mann–Whitney U test was also used to compare the mean values in two groups. The Kruskal–Wallis test (with Bonferroni post-hoc tests) was used to test the significance of data to compare more than two groups. The SPSS for Windows Version 18.0 statistical software (SPSS, Chicago, IL, USA) was used for all statistical analyses. Statistical significance was determined at the p < 0.05 level.