Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
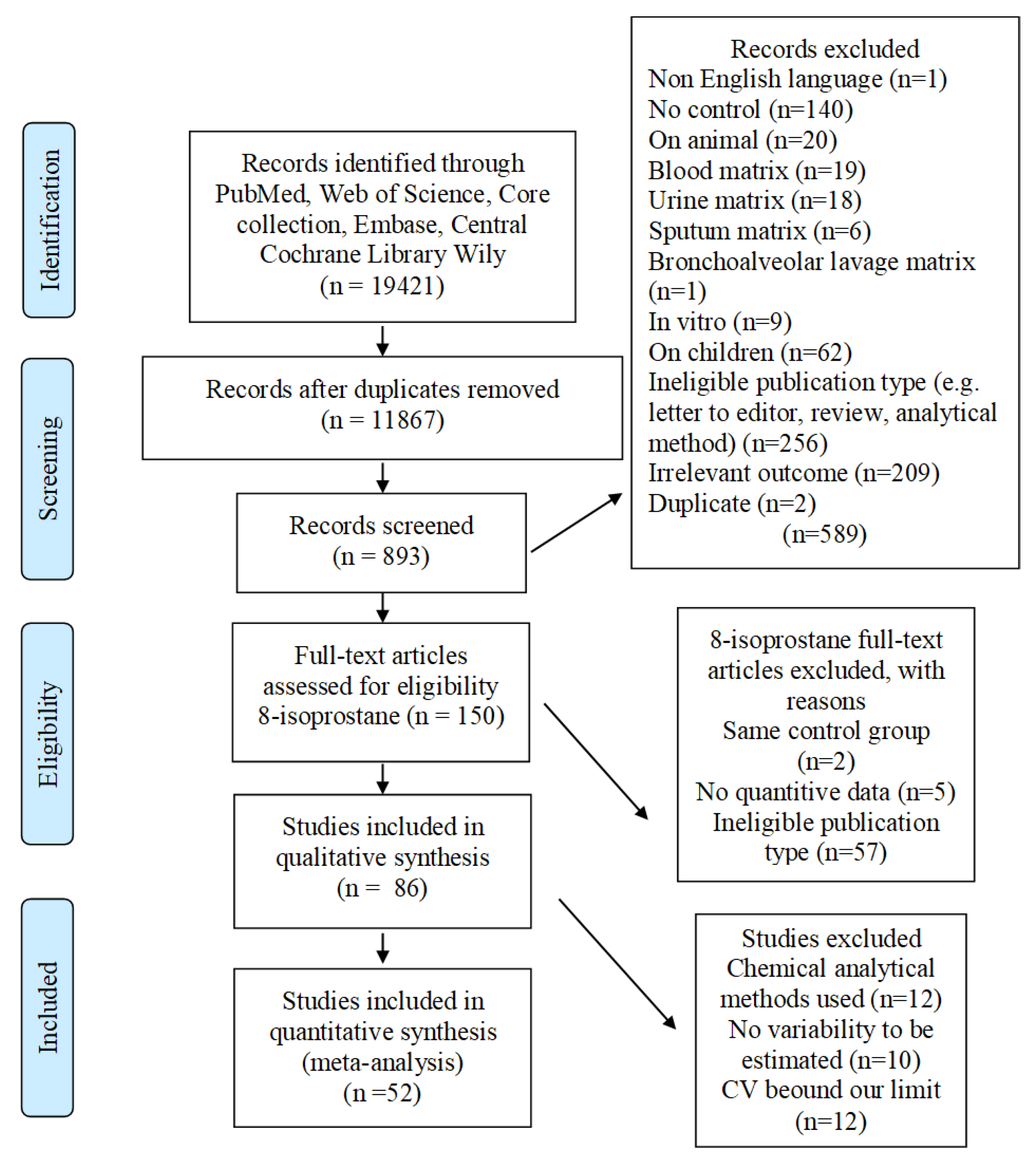
2.1. Study Selection
2.2. Descriptive Analysis
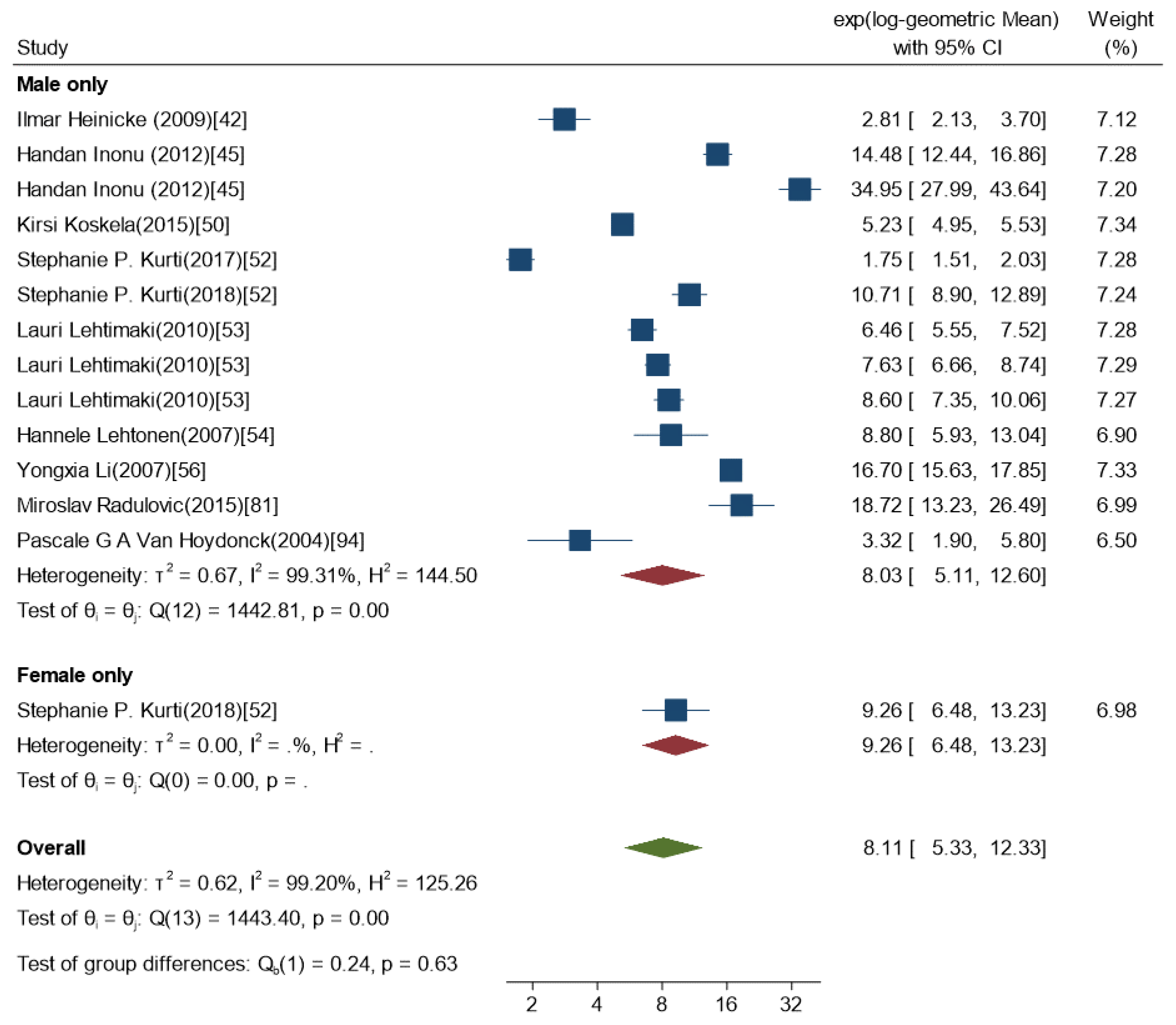
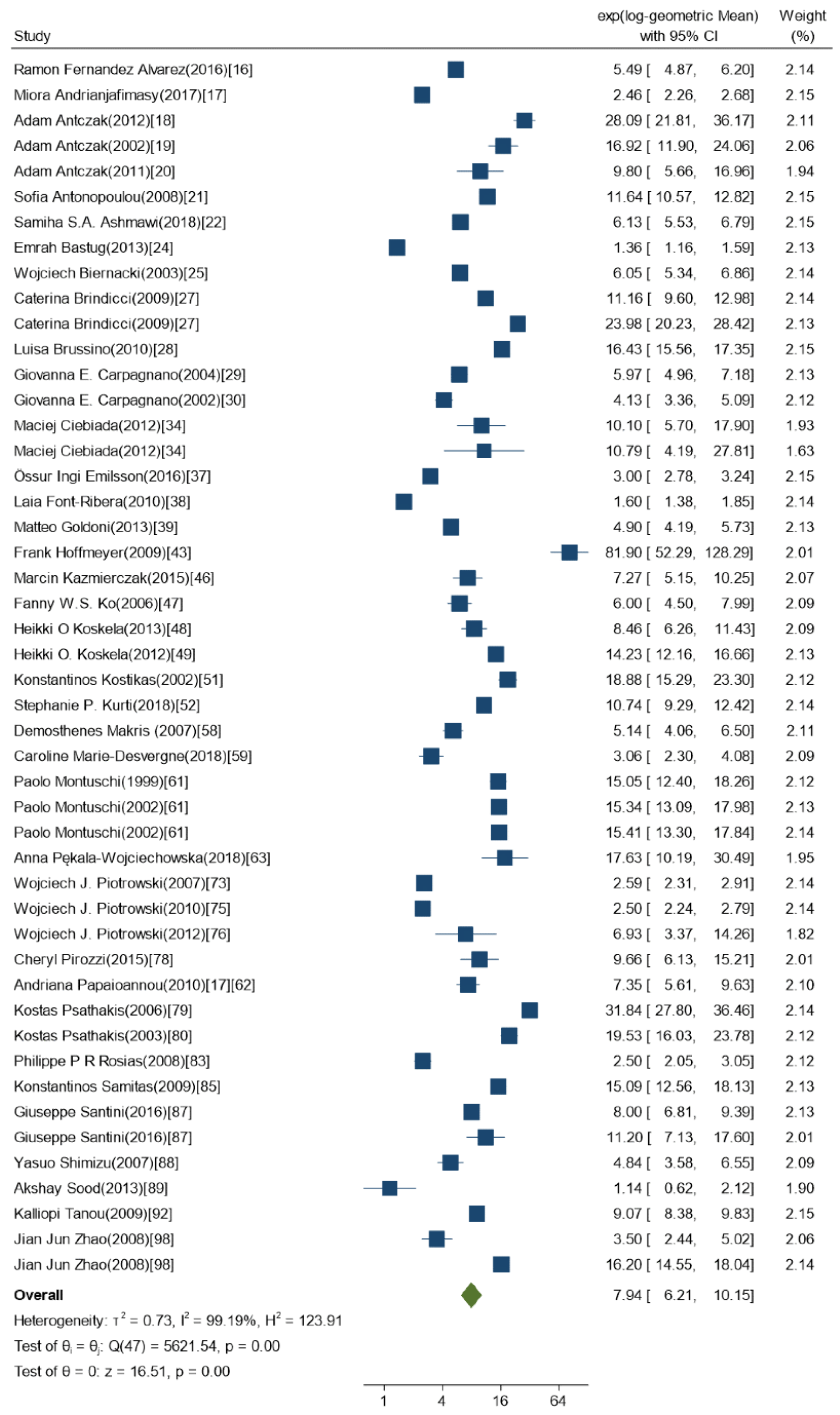
2.3. Meta-Analysis
3. Discussion
3.1. Interpretation of Results
3.1.1. Gender and Individual Characteristics
3.1.2. Factors Related to EBC Sample Collection, Storage and Analysis
3.2. Contribution of the Results to the Currently Available Guidelines for EBC Handling
3.3. Strengths and Limitations
4. Materials and Methods
4.1. Literature Search
4.2. Data Extraction
4.3. Quality Assessment
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CV | Coefficient of variation |
| OS | Oxidative stress |
| EBC | Exhaled Breath Condensate |
| GM | Geometric Mean |
| GSD | Geometric standard deviation |
| BMI | Body Mass Index |
Appendix A
| LogGM | Regression Coefficient | P > |z| | 95%CI |
|---|---|---|---|
| Device | |||
| Ecoscreen | 0 | ||
| NA/others | 0.64 | 0.03 | 0.06–1.21 |
| Rtube | −0.70 | 0.13 | −1.60–0.21 |
| BMI | |||
| Gender | |||
| Males only | 0 | ||
| Males and females | −0.39 | 0.38 | |
| BMI<25 | 0 | ||
| BMI>25 | −0.19 | 0.71 | −1.25–0.85 |
| No BMI reported | −0.08 | 0.87 | −0.88–1.04 |
| Smoking | |||
| Non-smokers | 0 | ||
| Smokers and nonsmokers | −0.24 | 0.43 | −0.86–0.37 |
| Smokers | 0.10 | 0.76 | −0.56–0.76 |
| Mean age | |||
| <40 | 0 | ||
| 40–60 | 0.02 | 0.94 | −0.48–0.52 |
| >60 | 0.02 | 0.96 | −0.82–0.87 |
| Intercept | 2.31 | 0.00 | 1.26–3.36 |
| Between-study standard deviation | 0.56 | 0.25–1.17 | |
| Within-study, between-group standard deviation” | 0.55 | 0.29–1.02 |
| LOGGM | Regression Coefficient | P > |z| | 95%CI |
|---|---|---|---|
| Device | |||
| Ecoscreen | 0 | ||
| NA/Other | 0.47 | 0.113 | 0.11–1.05 |
| Rtube | 0.69 | 0.098 | 1.5–0.12 |
| Temperature | |||
| −20 | −1.31 | 0.219 | −3.40–0.78 |
| −60 | −0.12 | 0.984 | −1.26–1.24 |
| −70 | 0 | ||
| −80 | −0.14 | 0.565 | −0.66–0.36 |
| NA | −2.73 | 0.007 | −4.73–(−0.73) |
| Nose-clip | |||
| No | 0 | ||
| Yes | −0.55 | 0.119 | −1.250.14 |
| Duration | |||
| 10 | 0 | ||
| 10–15 | 0.35 | 0.539 | −0.77–1.48 |
| 15 | −0.07 | 0.767 | −0.55–0.40 |
| 20 | 0.36 | 0.550 | −0.81–1.52 |
| Time | |||
| Afternoon | −0.47 | 0.431 | −1.64–0.70 |
| NA | −0.44 | 0.12 | 0.1–0.12 |
| Morning | 0 |
References
- Halliwell, B. Free radicals and antioxidants: A personal view. Nutr. Rev. 1994, 52, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; Abramov, A.Y. Mechanism of oxidative stress in neurodegeneration. Oxidative Med. Cell. Longev. 2012, 2012, 428010. [Google Scholar] [CrossRef] [PubMed]
- Van’t Erve, T.J.; Kadiiska, M.B.; London, S.J.; Mason, R.P. Classifying oxidative stress by F2-isoprostane levels across human diseases: A meta-analysis. Redox Biol. 2017, 12, 582–599. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P.; Corradi, M.; Ciabattoni, G.; Nightingale, J.; Kharitonov, S.A.; Barnes, P.J. Increased 8-isoprostane, a marker of oxidative stress, in exhaled condensate of asthma patients. Am. J. Respir. Crit. Care Med. 1999, 160, 216–220. [Google Scholar] [CrossRef]
- Horvath, I.; Hunt, J.; Barnes, P.J.; Alving, K.; Antczak, A.; Baraldi, E.; Becher, G.; van Beurden, W.J.; Corradi, M.; Dekhuijzen, R.; et al. Exhaled breath condensate: Methodological recommendations and unresolved questions. Eur. Respir. J. 2005, 26, 523–548. [Google Scholar] [CrossRef]
- Milne, G.L.; Yin, H.Y.; Brooks, J.D.; Sanchez, S.; Roberts, L.J.; Morrow, J.D. Quantification of F2-isoprostanes in biological fluids and tissues as a measure of oxidant stress. Method Enzymol. 2007, 433, 113–126. [Google Scholar] [CrossRef]
- Carpenter, C.T.; Price, P.V.; Christman, B.W. Exhaled breath condensate isoprostanes are elevated in patients with acute lung injury or ARDS. Chest 1998, 114, 1653–1659. [Google Scholar] [CrossRef]
- Carraro, S.; Cogo, P.E.; Isak, I.; Simonato, M.; Corradi, M.; Carnielli, V.P.; Baraldi, E. EIA and GC/MS analysis of 8-isoprostane in EBC of children with problematic asthma. Eur. Respir. J. 2010, 35, 1364–1369. [Google Scholar] [CrossRef]
- Basu, S. F2-isoprostanes in human health and diseases: From molecular mechanisms to clinical implications. Antioxid. Redox Signal. 2008, 10, 1405–1434. [Google Scholar] [CrossRef]
- Koutsokera, A.; Loukides, S.; Gourgoulianis, K.I.; Kostikas, K. Biomarkers in the exhaled breath condensate of healthy adults: Mapping the path towards reference values. Curr. Med. Chem. 2008, 15, 620–630. [Google Scholar] [CrossRef]
- Yuan, Y.; Hunt, R.H. Systematic reviews: The good, the bad, and the ugly. Am. J. Gastroenterol. 2009, 104, 1086–1092. [Google Scholar] [CrossRef] [PubMed]
- Pinchuk, I.; Weber, D.; Kochlik, B.; Stuetz, W.; Toussaint, O.; Debacq-Chainiaux, F.; Dolle, M.E.T.; Jansen, E.H.J.M.; Gonos, E.S.; Sikora, E.; et al. Gender- and age-dependencies of oxidative stress, as detected based on the steady state concentrations of different biomarkers in the MARK-AGE study. Redox Biol. 2019, 24. [Google Scholar] [CrossRef]
- Kander, M.C.; Cui, Y.Q.; Liu, Z.G. Gender difference in oxidative stress: A new look at the mechanisms for cardiovascular diseases. J. Cell Mol. Med. 2017, 21, 1024–1032. [Google Scholar] [CrossRef]
- Ide, T.; Tsutsui, H.; Ohashi, N.; Hayashidani, S.; Suematsu, N.; Tsuchihashi, M.; Tamai, H.; Takeshita, A. Greater oxidative stress in healthy young men compared with premenopausal women. Arter. Thromb. Vasc. Biol. 2002, 22, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Emerson, S.R.; Smith, J.R.; Rosenkranz, S.K.; Alexander, S.A.; Lovoy, G.M.; Harms, C.A. Older women exhibit greater airway 8-isoprostane responses to strenuous exercise compared with older men and younger controls. Appl. Physiol. Nutr. Metab. 2018, 43, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Alfaro, M.F.; Walby, W.F.; Adams, W.C.; Schelegle, E.S. Breath condensate levels of 8-isoprostane and leukotriene B4 after ozone inhalation are greater in sensitive versus nonsensitive subjects. Exp. Lung Res. 2007, 33, 115–133. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, R.F.; Cuadrado, G.R.; Arias, R.A.; Hernandez, J.A.C.; Antequera, B.P.; Urrutia, M.I.; Clara, P.C. Snoring as a Determinant Factor of Oxidative Stress in the Airway of Patients with Obstructive Sleep Apnea. Lung 2016, 194, 469–473. [Google Scholar] [CrossRef]
- Andrianjafimasy, M.; Zerimech, F.; Akiki, Z.; Huyvaert, H.; Le Moual, N.; Siroux, V.; Matran, R.; Dumas, O.; Nadif, R. Oxidative stress biomarkers and asthma characteristics in adults of the EGEA study. Eur. Respir. J. 2017, 50. [Google Scholar] [CrossRef]
- Antczak, A.; Ciebiada, M.; Pietras, T.; Piotrowski, W.J.; Kurmanowska, Z.; Gorski, P. Exhaled eicosanoids and biomarkers of oxidative stress in exacerbation of chronic obstructive pulmonary disease. Arch. Med. Sci. 2012, 8, 277–285. [Google Scholar] [CrossRef]
- Antczak, A.; Montuschi, P.; Kharitonov, S.; Gorski, P.; Barnes, P.J. Increased exhaled cysteinyl-leukotrienes and 8-isoprostane in aspirin-induced asthma. Am. J. Respir. Crit. Care Med. 2002, 166, 301–306. [Google Scholar] [CrossRef]
- Antczak, A.; Piotrowski, W.; Marczak, J.; Ciebiada, M.; Gorski, P.; Barnes, P.J. Correlation between eicosanoids in bronchoalveolar lavage fluid and in exhaled breath condensate. Dis. Markers 2011, 30, 213–220. [Google Scholar] [CrossRef]
- Antonopoulou, S.; Loukides, S.; Papatheodorou, G.; Roussos, C.; Alchanatis, M. Airway inflammation in obstructive sleep apnea: Is leptin the missing link? Respir. Med. 2008, 102, 1399–1405. [Google Scholar] [CrossRef][Green Version]
- Ashmawi, S.S.A.; Dewdar, I.A.; Mohamed, N.A.; Elhefny, A.A. Measurement of 8-isoprostane in exhaled breath condensate of patients with chronic obstructive pulmonary disease. Egypt. J. Chest Dis. Tuberc. 2018, 67, 226–230. [Google Scholar] [CrossRef]
- Barreto, M.; Villa, M.P.; Corradi, M.; Barberi, S.; Monaco, G.; Martella, S.; Bohmerova, Z.; Sabatino, G.; Ronchetti, R. Non-invasive assessment of airway inflammation in ship-engine workers. Int. J. Immunopathol. Pharmacol. 2006, 19, 601–608. [Google Scholar] [CrossRef]
- Bastug, E.; Tasliyurt, T.; Kutluturk, F.; Sahin, S.; Yilmaz, A.; Sivgin, H.; Yelken, B.M.; Ozturk, B.; Yilmaz, A.; Sahin, S. Evaluation of oxidative status with exhaled breath 8-isoprostane levels in patients with hyperthyroidism. Endocr. Metab. Immune Disord. Drug Targets 2013, 13, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Biernacki, W.A.; Kharitonov, S.A.; Barnes, P.J. Increased leukotriene B4 and 8-isoprostane in exhaled breath condensate of patients with exacerbations of COPD. Thorax 2003, 58, 294–298. [Google Scholar] [CrossRef]
- Borrill, Z.L.; Roy, K.; Vessey, R.S.; Woodcock, A.A.; Singh, D. Non-invasive biomarkers and pulmonary function in smokers. Int. J. Chronic Obstr. Pulm. Dis. 2008, 3, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Brindicci, C.; Ito, K.; Torre, O.; Barnes, P.J.; Kharitonov, S.A. Effects of aminoguanidine, an inhibitor of inducible nitric oxide synthase, on nitric oxide production and its metabolites in healthy control subjects, healthy smokers, and COPD patients. Chest 2009, 135, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Brussino, L.; Badiu, I.; Sciascia, S.; Bugiani, M.; Heffler, E.; Guida, G.; Malinovschi, A.; Bucca, C.; Rolla, G. Oxidative stress and airway inflammation after allergen challenge evaluated by exhaled breath condensate analysis. Clin. Exp. Allergy 2010, 40, 1642–1647. [Google Scholar] [CrossRef]
- Carpagnano, G.E.; Kharitonov, S.A.; Foschino-Barbaro, M.P.; Resta, O.; Gramiccioni, E.; Barnes, P.J. Supplementary oxygen in healthy subjects and those with COPD increases oxidative stress and airway inflammation. Thorax 2004, 59, 1016–1019. [Google Scholar] [CrossRef][Green Version]
- Carpagnano, G.E.; Kharitonov, S.A.; Resta, O.; Foschino-Barbaro, M.P.; Gramiccioni, E.; Barnes, P.J. Increased 8-isoprostane and interleukin-6 in breath condensate of obstructive sleep apnea patients. Chest 2002, 122, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.P.; Tran, V.; Lewis, C.; Thomas, P.S. Elevated levels of oxidative stress markers in exhaled breath condensate. J. Thorac. Oncol. 2009, 4, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.; Campbell, C.; Sandrini, A.; Thomas, P.S.; Johnson, A.R.; Yates, D.H. Exhaled breath condensate biomarkers in asbestos-related lung disorders. Respir. Med. 2009, 103, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.; Thomas, P.S.; Malouf, M.; Yates, D.H. Exhaled breath condensate (EBC) biomarkers in pulmonary fibrosis. J. Breath Res. 2012, 6, 16004. [Google Scholar] [CrossRef]
- Ciebiada, M.; Gorski, P.; Antczak, A. Eicosanoids in exhaled breath condensate and bronchoalveolar lavage fluid of patients with primary lung cancer. Dis. Markers 2012, 32, 329–335. [Google Scholar] [CrossRef][Green Version]
- Cruz, M.J.; Sanchez-Vidaurre, S.; Romero, P.V.; Morell, F.; Munoz, X. Impact Of Age on pH, 8-Isoprostane, and Nitrogen Oxides in Exhaled Breath Condensate. Chest 2009, 135, 462–467. [Google Scholar] [CrossRef]
- Do, R.; Bartlett, K.H.; Dimich-Ward, H.; Chu, W.; Kennedy, S.M. Biomarkers of airway acidity and oxidative stress in exhaled breath condensate from grain workers. Am. J. Respir. Crit. Care Med. 2008, 178, 1048–1054. [Google Scholar] [CrossRef]
- Emilsson, O.I.; Benediktsdottir, B.; Olafsson, I.; Cook, E.; Juliusson, S.; Bjornsson, E.S.; Guethlaugsdottir, S.; Guethmundsdottir, A.S.; Mirgorodskaya, E.; Ljungstrom, E.; et al. Respiratory symptoms, sleep-disordered breathing and biomarkers in nocturnal gastroesophageal reflux. Respir. Res. 2016, 17, 115. [Google Scholar] [CrossRef]
- Font-Ribera, L.; Kogevinas, M.; Zock, J.P.; Gomez, F.P.; Barreiro, E.; Nieuwenhuijsen, M.J.; Fernandez, P.; Lourencetti, C.; Perez-Olabarria, M.; Bustamante, M.; et al. Short-Term Changes in Respiratory Biomarkers after Swimming in a Chlorinated Pool. Environ. Health Perspect. 2010, 118, 1538–1544. [Google Scholar] [CrossRef]
- Goldoni, M.; Corradi, M.; Mozzoni, P.; Folesani, G.; Alinovi, R.; Pinelli, S.; Andreoli, R.; Pigini, D.; Tillo, R.; Filetti, A.; et al. Concentration of exhaled breath condensate biomarkers after fractionated collection based on exhaled CO2 signal. J. Breath Res. 2013, 7. [Google Scholar] [CrossRef]
- Gratziou, C.; Rovina, N.; Makris, M.; Simoes, D.C.M.; Papapetropoulos, A.; Roussos, C. Breath Markers of Oxidative Stress and Airway Inflammation in Seasonal Allergic Rhinitis. Int. J. Immunopathol. Pharmacol. 2008, 21, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Hakim, F.; Hellou, E.; Goldbart, A.; Katz, R.; Bentur, Y.; Bentur, L. The Acute Effects of Water-Pipe Smoking on the Cardiorespiratory System. Chest 2011, 139, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Heinicke, I.; Boehler, A.; Rechsteiner, T.; Bogdanova, A.; Jelkmann, W.; Hofer, M.; Rawlings, P.; Araneda, O.F.; Behn, C.; Gassmann, M.; et al. Moderate altitude but not additional endurance training increases markers of oxidative stress in exhaled breath condensate. Eur. J. Appl. Physiol. 2009, 106, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Hoffmeyer, F.; Raulf-Heimsoth, M.; Harth, V.; Bunger, J.; Bruning, T. Comparative analysis of selected exhaled breath biomarkers obtained with two different temperature-controlled devices. BMC Pulm. Med. 2009, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Hoffmeyer, F.; Raulf-Heimsoth, M.; Lehnert, M.; Kendzia, B.; Bernard, S.; Berresheim, H.; Duser, M.; Henry, J.; Weiss, T.; Koch, H.M.; et al. Impact of Different Welding Techniques on Biological Effect Markers in Exhaled Breath Condensate of 58 Mild Steel Welders. J. Toxicol. Environ. Health Part A 2012, 75, 525–532. [Google Scholar] [CrossRef]
- Inonu, H.; Doruk, S.; Sahin, S.; Erkorkmaz, U.; Celik, D.; Celikel, S.; Seyfikli, Z. Oxidative stress levels in exhaled breath condensate associated with COPD and smoking. Respir. Care 2012, 57, 413–419. [Google Scholar] [CrossRef]
- Kazmierczak, M.; Ciebiada, M.; Pekala-Wojciechowska, A.; Pawlowski, M.; Pietras, T.; Antczak, A. Correlation of inflammatory markers with echocardiographic parameters of left and right ventricular function in patients with chronic obstructive pulmonary disease and cardiovascular diseases. Pol. Arch. Med. Wewn. 2014, 124, 290–297. [Google Scholar] [CrossRef]
- Ko, F.W.; Lau, C.Y.; Leung, T.F.; Wong, G.W.; Lam, C.W.; Hui, D.S. Exhaled breath condensate levels of 8-isoprostane, growth related oncogene alpha and monocyte chemoattractant protein-1 in patients with chronic obstructive pulmonary disease. Respir. Med. 2006, 100, 630–638. [Google Scholar] [CrossRef]
- Koskela, H.O.; Purokivi, M.K. Airway oxidative stress in chronic cough. Cough 2013, 9, 26. [Google Scholar] [CrossRef]
- Koskela, H.O.; Purokivi, M.K.; Nieminen, R.M.; Moilanen, E. Asthmatic cough and airway oxidative stress. Respir. Physiol. Neurobiol. 2012, 181, 346–350. [Google Scholar] [CrossRef]
- Koskela, K.; Oksa, P.; Sauni, R.; Linnainmaa, M.; Toivio, P.; Lehtimaki, L.; Moilanen, E.; Nieminen, R.; Luukkonen, R.; Uitti, J. Pulmonary inflammation in foundry workers. J. Occup. Environ. Med. 2015, 57, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Kostikas, K.; Papatheodorou, G.; Ganas, K.; Psathakis, K.; Panagou, P.; Loukides, S. pH in expired breath condensate of patients with inflammatory airway diseases. Am. J. Respir. Crit. Care Med. 2002, 165, 1364–1370. [Google Scholar] [CrossRef] [PubMed]
- Kurti, S.P.; Emerson, S.R.; Rosenkranz, S.K.; Teeman, C.S.; Emerson, E.M.; Cull, B.J.; Smith, J.R.; Harms, C.A. Post-prandial systemic 8-isoprostane increases after consumption of moderate and high-fat meals in insufficiently active males. Nutr. Res. 2017, 39, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Lehtimaki, L.; Oksa, P.; Jarvenpaa, R.; Vierikko, T.; Nieminen, R.; Kankaanranta, H.; Uitti, J.; Moilanen, E. Pulmonary inflammation in asbestos-exposed subjects with borderline parenchymal changes on HRCT. Respir. Med. 2010, 104, 1042–1049. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lehtonen, H.; Oksa, P.; Lehtimaki, L.; Sepponen, A.; Nieminen, R.; Kankaanranta, H.; Saarelainen, S.; Jarvenpaa, R.; Uitti, J.; Moilanen, E. Increased alveolar nitric oxide concentration and high levels of leukotriene B-4 and 8-isoprostane in exhaled breath condensate in patients with asbestosis. Thorax 2007, 62, 602–607. [Google Scholar] [CrossRef]
- Leung, T.F.; Li, C.Y.; Yung, E.; Liu, E.K.; Lam, C.W.; Wong, G.W. Clinical and technical factors affecting pH and other biomarkers in exhaled breath condensate. Pediatr. Pulmonol. 2006, 41, 87–94. [Google Scholar] [CrossRef]
- Li, Y.X.; Chongsuvivatwong, V.; Geater, A.; Liu, A. Exhaled breath condensate cytokine level as a diagnostic tool for obstructive sleep apnea syndrome. Sleep Med. 2009, 10, 95–103. [Google Scholar] [CrossRef]
- Liou, S.H.; Wu, W.T.; Liao, H.Y.; Chen, C.Y.; Tsai, C.Y.; Jung, W.T.; Lee, H.L. Global DNA methylation and oxidative stress biomarkers in workers exposed to metal oxide nanoparticles. J. Hazard. Mater. 2017, 331, 329–335. [Google Scholar] [CrossRef]
- Makris, D.; Paraskakis, E.; Korakas, P.; Karagiannakis, E.; Sourvinos, G.; Siafakas, N.M.; Tzanakis, N. Exhaled breath condensate 8-isoprostane, clinical parameters, radiological indices and airway inflammation in COPD. Respiration 2008, 75, 138–144. [Google Scholar] [CrossRef]
- Marie-Desvergne, C.; Dubosson, M.; Mossuz, V.C. Evaluation of a new method for the collection and measurement of 8-isoprostane in exhaled breath for future application in nanoparticle exposure biomonitoring. J. Breath Res. 2018, 12. [Google Scholar] [CrossRef]
- Mazur, W.; Stark, H.; Sovijarvi, A.; Myllarniemi, M.; Kinnula, V.L. Comparison of 8-Isoprostane and Interleukin-8 in Induced Sputum and Exhaled Breath Condensate from Asymptomatic and Symptomatic Smokers. Respiration 2009, 78, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P.; Nightingale, J.A.; Kharitonov, S.A.; Barnes, P.J. Ozone-induced increase in exhaled 8-isoprostane in healthy subjects is resistant to inhaled budesonide. Free Radic. Biol. Med. 2002, 33, 1403–1408. [Google Scholar] [CrossRef]
- Papaioannou, A.I.; Koutsokera, A.; Tanou, K.; Kiropoulos, T.S.; Tsilioni, I.; Oikonomidi, S.; Liadaki, K.; Pournaras, S.; Gourgoulianis, K.I.; Kostikas, K. The acute effect of smoking in healthy and asthmatic smokers. Eur. J. Clin. Investig. 2010, 40, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Pekala-Wojciechowska, A.; Poznanski, M.; Szyszow, K.; Antczak, A. Concentration of 8-isoprostanes in the exhaled breath condensate as a marker of oxidative stress in patients with type 1 diabetes. Adv. Respir. Med. 2018, 86, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Fenclova, Z.; Kacer, P.; Kuzma, M.; Navratil, T.; Lebedova, J. Increased 8-isoprostane, a marker of oxidative stress in exhaled breath condensate in subjects with asbestos exposure. Ind. Health 2008, 46, 484–489. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Kacer, P.; Navratil, T.; Kuzma, M.; Lebedova, J.; Klusackova, P. 8-isoprostane and leukotrienes in exhaled breath condensate in Czech subjects with silicosis. Ind. Health 2007, 45, 766–774. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Navratil, T.; Vlckova, S.; Syslova, K.; Kuzma, M.; Zdimal, V.; Schwarz, J.; Pusman, J.; Zikova, N.; et al. Markers of oxidative stress in exhaled breath condensate are significantly increased in workers exposed to aerosol containing TiO2 nanoparticles. Toxicol. Lett. 2014, 229, S12. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Syslova, K.; Vlckova, S.; Lebedova, J.; Pecha, O.; Belacek, J.; Navratil, T.; Kuzma, M.; Kacer, P. Oxidative Stress Markers in Exhaled Breath Condensate in Lung Fibroses Are Not Significantly Affected by Systemic Diseases. Ind. Health 2011, 49, 746–754. [Google Scholar] [CrossRef]
- Pelclova, D.; Fenclova, Z.; Vlckova, S.; Klusackova, P.; Lebedova, J.; Syslova, K.; Belacek, J.; Kuzma, M.; Navratil, T.; Zakharov, S.; et al. Occupational asthma follow-up--which markers are elevated in exhaled breath condensate and plasma? Int. J. Occup. Med. Environ. Health 2014, 27, 206–215. [Google Scholar] [CrossRef]
- Pelclova, D.; Navratil, T.; Vlckova, S.; Fenclova, Z.; Pelcl, T.; Kacerova, T.; Kacer, P. Exhaled breath condensate biomarkers reflect systemic changes in patients with chronic dioxin intoxication. Monatsh. Chem. 2018, 149, 1579–1586. [Google Scholar] [CrossRef]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Fenclova, Z.; Vlckova, S.; Syslova, K.; Navratil, T.; Schwarz, J.; Zikova, N.; Barosova, H.; et al. Oxidative stress markers are elevated in exhaled breath condensate of workers exposed to nanoparticles during iron oxide pigment production. J. Breath Res. 2016, 10. [Google Scholar] [CrossRef]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Komarc, M.; Fenclova, Z.; Vlckova, S.; Zikova, N.; Schwarz, J.; Makes, O.; Navratil, T.; et al. Markers of lipid oxidative damage among office workers exposed intermittently to air pollutants including nanoTiO(2) particles. Rev. Environ. Health 2017, 32, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Zdimal, V.; Kacer, P.; Zikova, N.; Komarc, M.; Fenclova, Z.; Vlckova, S.; Schwarz, J.; Makes, O.; Syslova, K.; et al. Markers of lipid oxidative damage in the exhaled breath condensate of nano TiO2 production workers. Nanotoxicology 2017, 11, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Piotrowski, W.J.; Antczak, A.; Marczak, J.; Nawrocka, A.; Kurmanowska, Z.; Gorski, P. Eicosanoids in exhaled breath condensate and BAL fluid of patients with sarcoidosis. Chest 2007, 132, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Piotrowski, W.J.; Kurmanowska, Z.; Antczak, A.; Marczak, J.; Ciebiada, M.; Gorski, P. Exhaled 8-isoprostane in sarcoidosis: Relation to superoxide anion production by bronchoalveolar lavage cells. Inflamm. Res. 2010, 59, 1027–1032. [Google Scholar] [CrossRef] [PubMed]
- Piotrowski, W.J.; Kurmanowska, Z.; Antczak, A.; Marczak, J.; Gorski, P. Exhaled 8-isoprostane as a prognostic marker in sarcoidosis. A short term follow-up. BMC Pulm. Med. 2010, 10, 23. [Google Scholar] [CrossRef]
- Piotrowski, W.J.; Majewski, S.; Marczak, J.; Kurmanowska, Z.; Gorski, P.; Antczak, A. Exhaled breath 8-isoprostane as a marker of asthma severity. Arch. Med. Sci. 2012, 8, 515–520. [Google Scholar] [CrossRef]
- Pirozzi, C.; Sturrock, A.; Carey, P.; Whipple, S.; Haymond, H.; Baker, J.; Weng, H.; Greene, M.; Scholand, M.; Kanner, R.; et al. Respiratory effects of particulate air pollution episodes in former smokers with and without chronic obstructive pulmonary disease: A panel study. COPD Res. Pract. 2015, 1, 122. [Google Scholar] [CrossRef]
- Pirozzi, C.; Sturrock, A.; Weng, H.Y.; Greene, T.; Scholand, M.B.; Kanner, R.; Paine, R., 3rd. Effect of naturally occurring ozone air pollution episodes on pulmonary oxidative stress and inflammation. Int. J. Environ. Res. Public Health 2015, 12, 5061–5075. [Google Scholar] [CrossRef]
- Psathakis, K.; Mermigkis, D.; Papatheodorou, G.; Loukides, S.; Panagou, P.; Polychronopoulos, V.; Siafakas, N.M.; Bouros, D. Exhaled markers of oxidative stress in idiopathic pulmonary fibrosis. Eur. J. Clin. Investig. 2006, 36, 362–367. [Google Scholar] [CrossRef]
- Psathakis, K.; Papatheodorou, G.; Plataki, M.; Panagou, P.; Loukides, S.; Siafakas, N.M.; Bouros, D. 8-Isoprostane, a marker of oxidative stress, is increased in the expired breath condensate of patients with pulmonary sarcoidosis. Chest 2004, 125, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Radulovic, M.; Bauman, W.A.; Wecht, J.M.; LaFountaine, M.; Kahn, N.; Hobson, J.; Singh, K.; Renzi, C.; Yen, C.; Schilero, G.J. Biomarkers of inflammation in persons with chronic tetraplegia. J. Breath Res. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Romero, P.V.; Rodriguez, B.; Martinez, S.; Canizares, R.; Sepulveda, D.; Manresa, F. Analysis of oxidative stress in exhaled breath condensate from patients with severe pulmonary infections. Arch. Bronconeumol. 2006, 42, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Rosias, P.P.; Robroeks, C.M.; Kester, A.; den Hartog, G.J.; Wodzig, W.K.; Rilkers, G.T.; Zimmermann, L.J.; van Schayck, C.P.; Joebsis, Q.; Dompeling, E. Biomarker reproducibility in exhaled breath condensate collected with different condensers. Eur. Respir. J. 2008, 31, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Rosias, P.P.; Robroeks, C.M.; Niemarkt, H.J.; Kester, A.D.; Vernooy, J.H.; Suykerbuyk, J.; Teunissen, J.; Heynens, J.; Hendriks, H.J.; Jobsis, Q.; et al. Breath condenser coatings affect measurement of biomarkers in exhaled breath condensate. Eur. Respir. J. 2006, 28, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- Samitas, K.; Chorianopoulos, D.; Vittorakis, S.; Zervas, E.; Economidou, E.; Papatheodorou, G.; Loukides, S.; Gaga, M. Exhaled cysteinyl-leukotrienes and 8-isoprostane in patients with asthma and their relation to clinical severity. Respir. Med. 2009, 103, 750–756. [Google Scholar] [CrossRef]
- Sanak, M.; Gielicz, A.; Bochenek, G.; Kaszuba, M.; Nizankowska-Mogilnicka, E.; Szczeklik, A. Targeted eicosanoid lipidomics of exhaled breath condensate provide a distinct pattern in the aspirin-intolerant asthma phenotype. J. Allergy Clin. Immunol. 2011, 127, 1141–1147. [Google Scholar] [CrossRef]
- Santini, G.; Mores, N.; Shohreh, R.; Valente, S.; Dabrowska, M.; Trove, A.; Zini, G.; Cattani, P.; Fuso, L.; Mautone, A.; et al. Exhaled and non-exhaled non-invasive markers for assessment of respiratory inflammation in patients with stable COPD and healthy smokers. J. Breath Res. 2016, 10, 017102. [Google Scholar] [CrossRef]
- Shimizu, Y.; Dobashi, K.; Zhao, J.J.; Kawata, T.; Ono, A.; Yanagitani, N.; Kaira, K.; Utsugi, M.; Hisada, T.; Ishizuka, T.; et al. Proton pump inhibitor improves breath marker in moderate asthma with gastroesophageal reflux disease. Respiration 2007, 74, 558–564. [Google Scholar] [CrossRef]
- Sood, A.; Qualls, C.; Seagrave, J.; McDonald, J.; Shohreh, R.; Chiavaroli, A.; Schuyler, M. Effect of Allergen Inhalation on Airway Oxidant Stress, Using Exhaled Breath Condensate 8-Isoprostane, in Mild Asthma. J. Asthma 2013, 50, 449–456. [Google Scholar] [CrossRef]
- Syslova, K.; Kacer, P.; Kuzma, M.; Najmanova, V.; Fenclova, Z.; Vlckova, S.; Lebedova, J.; Pelclova, D. Rapid and easy method for monitoring oxidative stress markers in body fluids of patients with asbestos or silica-induced lung diseases. J. Chromatogr. B 2009, 877, 2477–2486. [Google Scholar] [CrossRef] [PubMed]
- Syslova, K.; Kacer, P.; Kuzma, M.; Pankracova, A.; Fenclova, Z.; Vlckova, S.; Lebedova, J.; Pelclova, D. LC-ESI-MS/MS method for oxidative stress multimarker screening in the exhaled breath condensate of asbestosis/silicosis patients. J. Breath Res. 2010, 4. [Google Scholar] [CrossRef] [PubMed]
- Tanou, K.; Koutsokera, A.; Kiropoulos, T.S.; Maniati, M.; Papaioannou, A.I.; Georga, K.; Zarogiannis, S.; Gourgoulianis, K.I.; Kostikas, K. Inflammatory and oxidative stress biomarkers in allergic rhinitis: The effect of smoking. Clin. Exp. Allergy 2009, 39, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Tufvesson, E.; Bozovic, G.; Hesselstrand, R.; Bjermer, L.; Scheja, A.; Wuttge, D.M. Increased cysteinyl-leukotrienes and 8-isoprostane in exhaled breath condensate from systemic sclerosis patients. Rheumatology 2010, 49, 2322–2326. [Google Scholar] [CrossRef] [PubMed]
- Van Hoydonck, P.G.; Wuyts, W.A.; Vanaudenaerde, B.M.; Schouten, E.G.; Dupont, L.J.; Temme, E.H. Quantitative analysis of 8-isoprostane and hydrogen peroxide in exhaled breath condensate. Eur. Respir. J. 2004, 23, 189–192. [Google Scholar] [CrossRef]
- Vizcaya, D.; Mirabelli, M.C.; Orriols, R.; Anto, J.M.; Barreiro, E.; Burgos, F.; Arjona, L.; Gomez, F.; Zock, J.P. Functional and biological characteristics of asthma in cleaning workers. Respir. Med. 2013, 107, 673–683. [Google Scholar] [CrossRef]
- Wu, C.M.; Adetona, A.; Song, C.C.; Naeher, L.; Adetona, O. Measuring acute pulmonary responses to occupational wildland fire smoke exposure using exhaled breath condensate. Arch. Environ. Occup. Health 2020, 75, 65–69. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, T.; Kipen, H.; Wang, G.; Huang, W.; Rich, D.; Zhu, P.; Wang, Y.; Lu, S.E.; Ohman-Strickland, P.; et al. Cardiorespiratory biomarker responses in healthy young adults to drastic air quality changes surrounding the 2008 Beijing Olympics. Res. Rep. Health Eff. Inst. 2013, 174, 5–174. [Google Scholar]
- Zhao, J.J.; Shimizu, Y.; Dobashi, K.; Kawata, T.; Ono, A.; Yanagitani, N.; Kaira, K.; Utsugi, M.; Hisada, T.; Ishizuka, T.; et al. The relationship between oxidative stress and acid stress in adult patients with mild asthma. J. Investig. Allergol. Clin. Immunol. 2008, 18, 41–45. [Google Scholar]
- Peel, A.M.; Crossman-Barnes, C.J.; Tang, J.; Fowler, S.J.; Davies, G.A.; Wilson, A.M.; Loke, Y.K. Biomarkers in adult asthma: A systematic review of 8-isoprostane in exhaled breath condensate. J. Breath Res. 2017, 11, 016011. [Google Scholar] [CrossRef][Green Version]
- Brooks, S.M.; Haight, R.R.; Gordon, R.L. Age does not affect airway pH and ammonia as determined by exhaled breath measurements. Lung 2006, 184, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Kullmann, T.; Barta, I.; Lazar, Z.; Szili, B.; Barat, E.; Valyon, M.; Kollai, M.; Horvath, I. Exhaled breath condensate pH standardised for CO2 partial pressure. Eur. Respir. J. 2007, 29, 496–501. [Google Scholar] [CrossRef]
- Montuschi, P.; Collins, J.V.; Ciabattoni, G.; Lazzeri, N.; Corradi, M.; Kharitonov, S.A.; Barnes, P.J. Exhaled 8-isoprostane as an in vivo biomarker of lung oxidative stress in patients with COPD and healthy smokers. Am. J. Respir. Crit. Care 2000, 162, 1175–1177. [Google Scholar] [CrossRef] [PubMed]
- Ahmadzai, H.; Huang, S.; Hettiarachchi, R.; Lin, J.L.; Thomas, P.S.; Zhang, Q. Exhaled breath condensate: A comprehensive update. Clin. Chem. Lab. Med. 2013, 51, 1343–1361. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, P.; Knobloch, H. Exhaled breath condensate: Lessons learned from veterinary medicine. J. Breath Res. 2010, 4, 017001. [Google Scholar] [CrossRef]
- Rosias, P. Methodological aspects of exhaled breath condensate collection and analysis. J. Breath Res. 2012, 6, 027102. [Google Scholar] [CrossRef]
- Janicka, M.; Kot-Wasik, A.; Kot, J.; Namiesnik, J. Isoprostanes-Biomarkers of Lipid Peroxidation: Their Utility in Evaluating Oxidative Stress and Analysis. Int. J. Mol. Sci. 2010, 11, 4631–4659. [Google Scholar] [CrossRef]
- Grob, N.M.; Aytekin, M.; Dweik, R.A. Biomarkers in exhaled breath condensate: A review of collection, processing and analysis. J. Breath Res. 2008, 2, 037004. [Google Scholar] [CrossRef]
- Horvath, I.; Barnes, P.J.; Loukides, S.; Sterk, P.J.; Hogman, M.; Olin, A.C.; Amann, A.; Antus, B.; Baraldi, E.; Bikov, A.; et al. A European Respiratory Society technical standard: Exhaled biomarkers in lung disease. Eur. Respir. J. 2017, 49. [Google Scholar] [CrossRef]
- Hemmendinger, M.; Graille, M.; Wild, P.; Hopf, N.; Guseva-Canu, I. Baseline Values, Between and Within Subject Variability of Non-Invasive Biomarkers of Inflammation and Oxidative Stress: A systematic Literature Review and Meta-Analysis; IUMSP Research Data Repository; Center for Primary Care and Public Health (Unisanté), University of Lausanne: Lausanne, Switzerland, 2019. [Google Scholar] [CrossRef]
- Hemmendinger, M.; Wild, P.; Shoman, Y.; Graille, M.; Bergamaschi, E.; Hopf, N.; Guseva Canu, I. Reference ranges of oxidative stress biomarkers selected for non-invasive biological surveillance of nanotechnology workers: Study protocol and meta-analysis results for 8-OHdG in exhaled breath condensate. Toxicol. Lett. 2020, 327, 41–47. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; for the PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [PubMed]
- Graille, M.; Wild, P.; Sauvain, J.-J.; Hemmendinger, M.; Guseva-Canu, I.; Hopf, N.B. Urinary 8-isoprostane as a biomarker for oxidative stress. A systematic review and meta-analysis. Toxicol. Lett. 2020, in press. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schunemann, H.J.; GRADE Working Group. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef]
- Deeks, J.; Higgins, J.; Altman, D. Analysing data and undertaking meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; Green, J.P.H.A.S., Ed.; The Cochrane Collaboration: London, UK, 2017. [Google Scholar]
| Author, Year, Country, [Ref]* | Study Objectives | Population Studied and Number of Participants | Control Population and Sub-Population | Method of EBC Collection and Analysis | Main Findings | Quality Score |
|---|---|---|---|---|---|---|
| Antonopoulou,2008, Greece, Ref [21]* | To assess airway inflammation by measuring the levels of 8-isoprostane, interleukin-6 (IL-6), Tumor Necrosis Factor-alpha (TNF-a), and pH in EBC and study their plausible relation with plasma levels of leptin. | 112 consecutive patients referred with symptoms suggestive of OSA. After a full night diagnostic polysomnography, 45 OSA patients (37 males, age 52 ± 12 years, BMI 33.5 ± 7, 28 smokers) finally formed the patients group. Patients with Apnea/Hypopnea Index (AHI) 10 were included in the study. | 25 healthy subjects non-randomly selected, matched for age, gender, and BMI, (18 males, age 51 ± 7 years, BMI 31 ± 3, 15 smokers). They were mainly recruited from a population used as healthy subjects in other studies of this research group.§ | EBC was collected by using a condenser (EcoScreen; Jaeger, Wurzburg, Germany). The condensate was stored at −70 °C. | Increased levels of leptin were not associated with the observed airway inflammation in OSA. The observed airway inflammation seemed to be independent of smoking habit with limited association with disease severity. | 20 |
| Bastug, 2013, Turkey, Ref [24]* | To measure oxidative stress in Hyperthyrod patients in EBC through measuring the levels of 8-isoprostane. | 42 Hyperthyrodism patients (12 males, 30 females). | 42 (12 males, 30 females, non-smokers) age and BMI matched healthy control subjects.§ | EBC was collected using a condenser (EcoScreen). Subjects were asked to breath tidally for 15 min using a noseclip. Condensates were stored immediately at −70 °C. | 8-isoprostane levels in EBC of hyperthyroid patients were found to be significantly higher than that in healthy control group. | 20 |
| Chow, 2009, Australia, Ref [32] | To assess lung oxidative stress and inflammation in vivo in subjects with asbestos-related disorders and compare them with age matched controls. | All subjects (n = 60) had a confirmed history of workplace asbestos exposure other than controls (n = 26) and were classified into three groups (asbestosis, diffuse pleural thickening (DPT) and pleural plaques (PPs). Smokers were excluded. | Age and sex-matched controls (n = 26). All control subjects were never or exsmokers without any evidence of asbestos-related or other lung disease after screening. | EBC was collected using Ecoscreen, subjects breathed tidally with nose-clip on. Condensate was collected after 10 min, the cooled condensate was immediately stored at −80 °C. | In asbestos-related disorders, markers of inflammation and oxidative stress are significantly elevated in subjects with asbestosis compared with healthy individuals but not in pleural diseases. | 20 |
| Chow, 2012, Australia, Ref [33] | To investigate whether levels of Several reactive oxygen species (ROS) and Several reactive nitrogen species (RNS) in EBC of patients with PF differed significantly from age- and sex-matched controls, and whether these correlated with lung function. | 20 subjects had pulmonary fibrosis (PF). | 20 were normal controls (16 male, mean age ± SD 55.3 ± 13.4). | EBC was collected using Ecoscreen, subjects breathed tidally with nose-clip on. Condensate was collected after 10 min, the cooled condensate was immediately stored at 80 °C. | Inflammatory and oxidative stress biomarkers are raised in patients with PF compared with controls. EBC may be useful for detecting and monitoring lung inflammation in PF. | 21 |
| Emilsson, 2016, Iceland, Ref [37]* | To investigate the association between nocturnal gastroesophageal reflux (nGER) and respiratory symptoms, exacerbations of respiratory symptoms, lung function and Sleep-disordered breathing (SDB). | This study is based on a 20 years prospective, population-based cohort study in Iceland. Among the 522 subjects contacted, a total of 455 participated, or 87% of those invited. Of the 455, 82 had symptoms suggestive of nGER. These 82 subjects were invited for a second visit in 2013, of which 71 (87%) participated. | Age and gender paired controls without any nGER symptoms (participation rate 78%, n = 42, Female 48%, mean age ± SD 56.4 ± 7.0).§ | EBC samples were collected with ECoScreen II. Participants wore a nose-clip and used tidal breathing for 15 min. The samples were immediately frozen at −20 °C, and within four hours moved to −80 °C for storage. | In a general population sample, nGER is associated with symptoms of asthma and bronchitis, as well as exacerbations of respiratory symptoms. In addition, nGER is associated with increased respiratory effort during sleep. | 21 |
| Hoffmeyer, 2012, Germany, Ref [44] | To evaluate subclinical changes in otherwise healthy current welders with the majority practicing this profession for decades. | 58 welders (all male, 27 smokers) from the cross-sectional study WELDOX were examined. Welders were processing mild steel applying gas metal arc welding with solid wire (GMAW) or flux cored wire (FCAW). | NA | EBC was collected after shift with the commercially available temperature-controlled device ECoScreen2. The collection time was exactly 10 min. | An enhanced irritative effect in the lower airways of mild steel welders due to the application of FCAW compared to GMAW, most likely associated with a higher emission of welding fumes. | 21 |
| Inonu, 2012,Turkey, Ref [45]* | To evaluate the differences in the burden of oxidative stress in patients with COPD, smokers, and non-smokers by measuring H2O2, MDA, and 8-isoprostane levels in the EBC samples. | The subjects in Group I (n = 25) had COPD (all ex-smokers). | Group II (n = 26) were healthy smokers (mean age ± SD 61.2 ± 6 y, all males) and Group III (n = 29) were healthy nonsmokers (mean age ± SD 60 ± 8 y, all males).§ | EBCs were collected using a condenser (EcoScreen). The subjects were asked to breathe while wearing a nose clip, for a period of 15 min. The samples were immediately stored at 70 °C. All EBC samples were collected between 2 PM to 4 PM. | Even if respiratory function tests are within normal limits, oxidant burden in lungs of smokers is equivalent to that in COPD patients. 8-isoprostane could be useful in assessing symptom severity and health status of COPD patients. | 23 |
| Lehtimaki, 2010, Finland, Ref [53]* | To find out if borderline parenchymal changes on HRCT in subjects with moderate to heavy asbestos exposure are related to the degree of pulmonary inflammation. | Of the 104 asbestos-exposed men recruited,33 were excluded based on the exclusion criteria. 35 subjects had normal parenchymal findings on HRCT and 31 subjects had borderline parenchymal changes. | 41 healthy men (mean age 63) not exposed to asbestos or other harmful agents.§ | EBC was collected during 15 min of tidal breathing with Ecoscreen condenser while wearing noseclips. The samples were stored at −70 °C. | Borderline parenchymal changes on HRCT in asbestos-exposed subjects are associated with increased markers of pulmonary inflammation. Such borderline parenchymal changes are likely a mild or early form of the same pathological process that leads to asbestosis. | 20 |
| Pelclova, 2007, Czech Republic, Ref [65] | To measure 8-isoprostane, leukotrienes B4, C4, D4, and E4 in exhaled breath condensate in patients with silicosis. | Patients with silicosis (n = 60, 58 men and 2 women). | The control group was composed of 25 subjects (23 men and 2 women), previously working as office employees and safety inspectors, never occupationally exposed to fibrogenic dusts. | EBC samples were collected using the EcoScreen. Each subject was asked to breathe through the collection kit for 15 min with more than 2 mL of EBC collected. Samples were immediately frozen after collection (−80 °C) | No significant effect of smoking or alcohol consumption on the markers examined was seen. This is the first study using exhaled breath condensate analysis in patients with silicosis. | 20 |
| Pelclova, 2008, Czech Republic, Ref [64] | To investigate the hypothesis that oxidative stress due to asbestos is the main cause of increased 8-isoprostane in EBC. | 92 asbestos-exposed subjects were examined (46 women and 46 men). | The control group was represented by 46 subjects (23 men and 23 women), employed as hospital technical workers (gatekeepers, adjuncts and helpers, hospital mailmen, etc.) without occupational exposure. | EBC samples were collected using the EcoScreen. Each subject was asked to breathe through the collection kit for 15 min with more than 2 mL of EBC collected. Samples were immediately frozen after collection (−80 °C) | Measurement of 8-isoprostane in EBC is a promising non-invasive means for assessing the activity of asbestos-induced diseases. | 20 |
| Sood, 2013, USA, Ref [89]* | To evaluate EBC 8-isoprostane concentrations following allergen-induced bronchoprovocation in asthma. | Eight mild atopic asthmatics (5 women) | Six healthy controls(four women): the majority of enrolled subjects were premenopausal overweight women(age mean ± SD 39.9 ± 9.7)§ | EBC was collected using an R-tube and condensate was collected during a period of 20–30 min. EBC was stored at −70 °C. | EBC 8-isoprostane concentrations do not acutely change following bronchoprovocation in subjects with mild asthma. | 20 |
| Vizcaya, 2013, Spain, Ref [95] | To evaluate associations of domestic and occupational use of cleaning products with asthma and biomarkers of respiratory health. | 42 cleaners with a history of asthma and/or recent respiratory symptoms (participation rate 60%). | 53 symptom-free controls (participation rate 44%) | EBC was collected using an EcoScreen condenser. Collection was performed from 09:00 to 10:00 in the morning. Each subject was asked to breathe into the device for 10 min while wearing a nose clip. The samples were stored at –70 °C. | Asthma in cleaning workers is characterized by non-reversible lung function decrement and increased total IgE. | 21 |
| Zhao, 2008, Japan, Ref [98]* | the relationship between the pH of EBC and the concentration in EBC of a marker of oxidative stress, 8-isoprostane, was investigated. The relationship between these markers and lung function was also studied. | Adults aged 18 years or over with asthma were recruited (n = 44, 20 females, nonsmokers)§ | Sex-matched and age-matched healthy volunteers without respiratory disease were recruited as control subjects (n = 20, 8 females, nonsmokers). | EBC was collected using an EcoScreen condenser. Collection was performed from 09:00 to 10:00 in the morning. Each subject was asked to breathe into the device for 10 min while wearing a nose clip. The samples were stored at –70 °C. | Stress and oxidative stress assessed by pH and 8-isoprostane concentration, respectively, in EBC did not show parallel changes associated with asthma and were not correlated with lung function in asthma patients. | 20 |
| EBC Device | Males Only | Males and Females | All Population |
|---|---|---|---|
| Rtube | 6.23(1.75–10.71), (n = 2) | 6.36(2.46–10.74), (n = 6) | 9.26(2.46–10.71), (n = 9) |
| Ecoscreen | 18.7(8.6–23.0), (n = 17) | 8.00(4.13–14.23), (n = 35) | 9.44(5.73–19.15), (n = 52) |
| NA/Other | 2.81(2.81–2.81), (n = 1) | 15.41(6.12–19.52), (n = 11) | 15.37(5.63–19.20), (n = 12) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shoman, Y.; Wild, P.; Hemmendinger, M.; Graille, M.; Sauvain, J.-J.; B. Hopf, N.; Guseva Canu, I. Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 3822. https://doi.org/10.3390/ijms21113822
Shoman Y, Wild P, Hemmendinger M, Graille M, Sauvain J-J, B. Hopf N, Guseva Canu I. Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2020; 21(11):3822. https://doi.org/10.3390/ijms21113822
Chicago/Turabian StyleShoman, Yara, Pascal Wild, Maud Hemmendinger, Melanie Graille, Jean-Jacques Sauvain, Nancy B. Hopf, and Irina Guseva Canu. 2020. "Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 21, no. 11: 3822. https://doi.org/10.3390/ijms21113822
APA StyleShoman, Y., Wild, P., Hemmendinger, M., Graille, M., Sauvain, J.-J., B. Hopf, N., & Guseva Canu, I. (2020). Reference Ranges of 8-Isoprostane Concentrations in Exhaled Breath Condensate (EBC): A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 21(11), 3822. https://doi.org/10.3390/ijms21113822





