Oviductal Extracellular Vesicles Improve Post-Thaw Sperm Function in Red Wolves and Cheetahs
Abstract
1. Introduction
2. Results
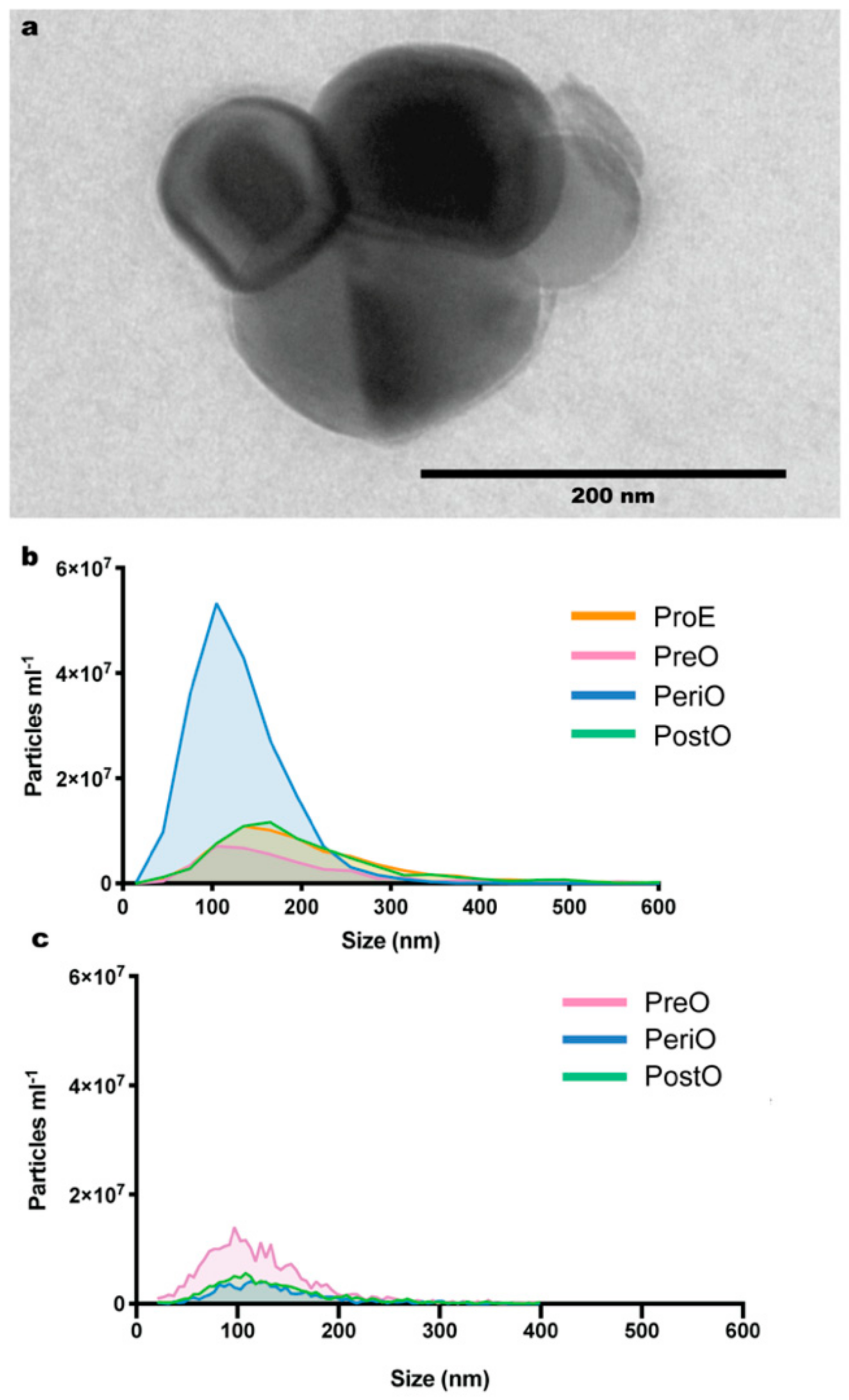
2.1. Isolation and Characterization of Dog and Cat Oviductal Extracellular Vesicles (oEVs)
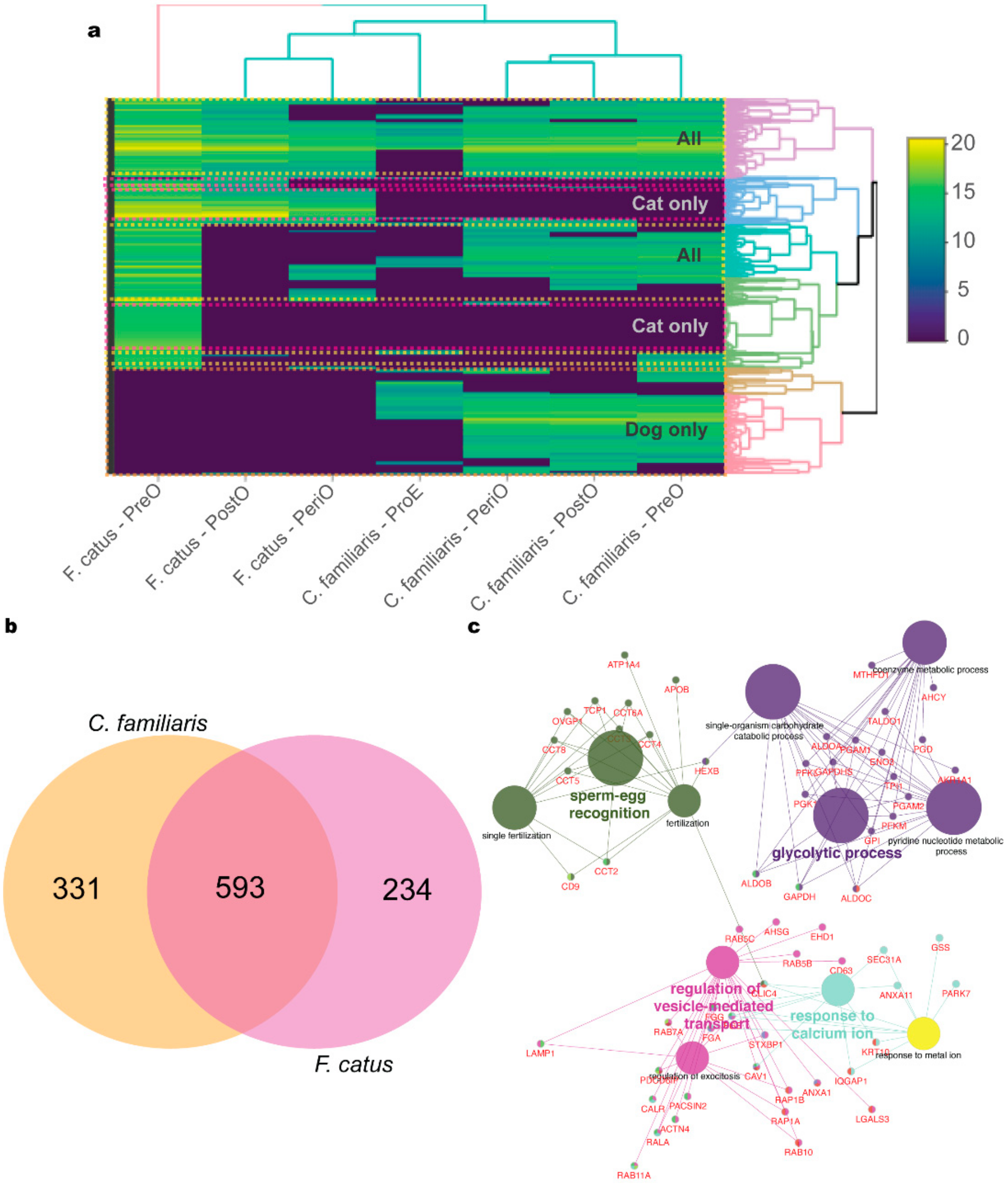
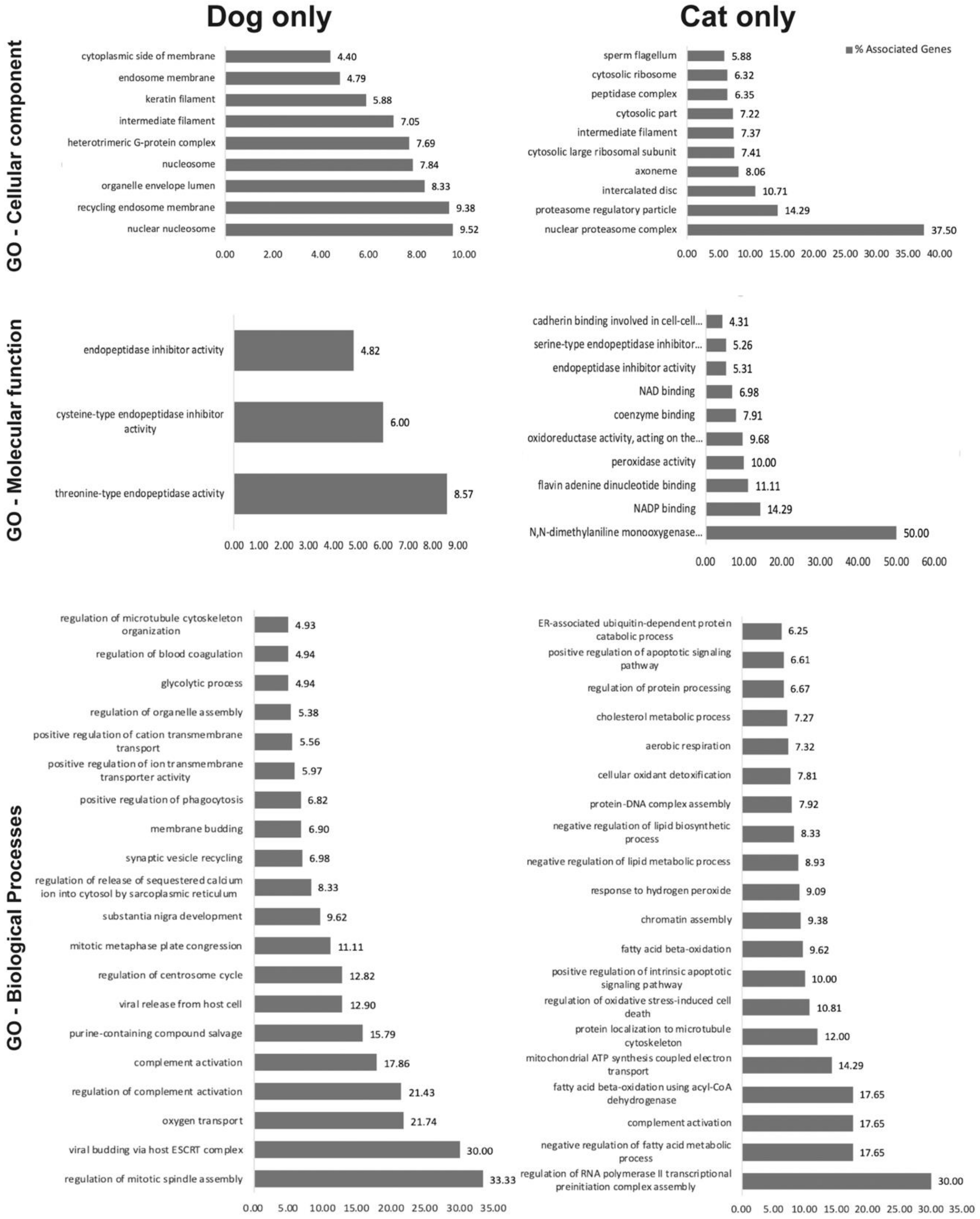
2.2. oEV Protein Content Varies between Dogs and Cats
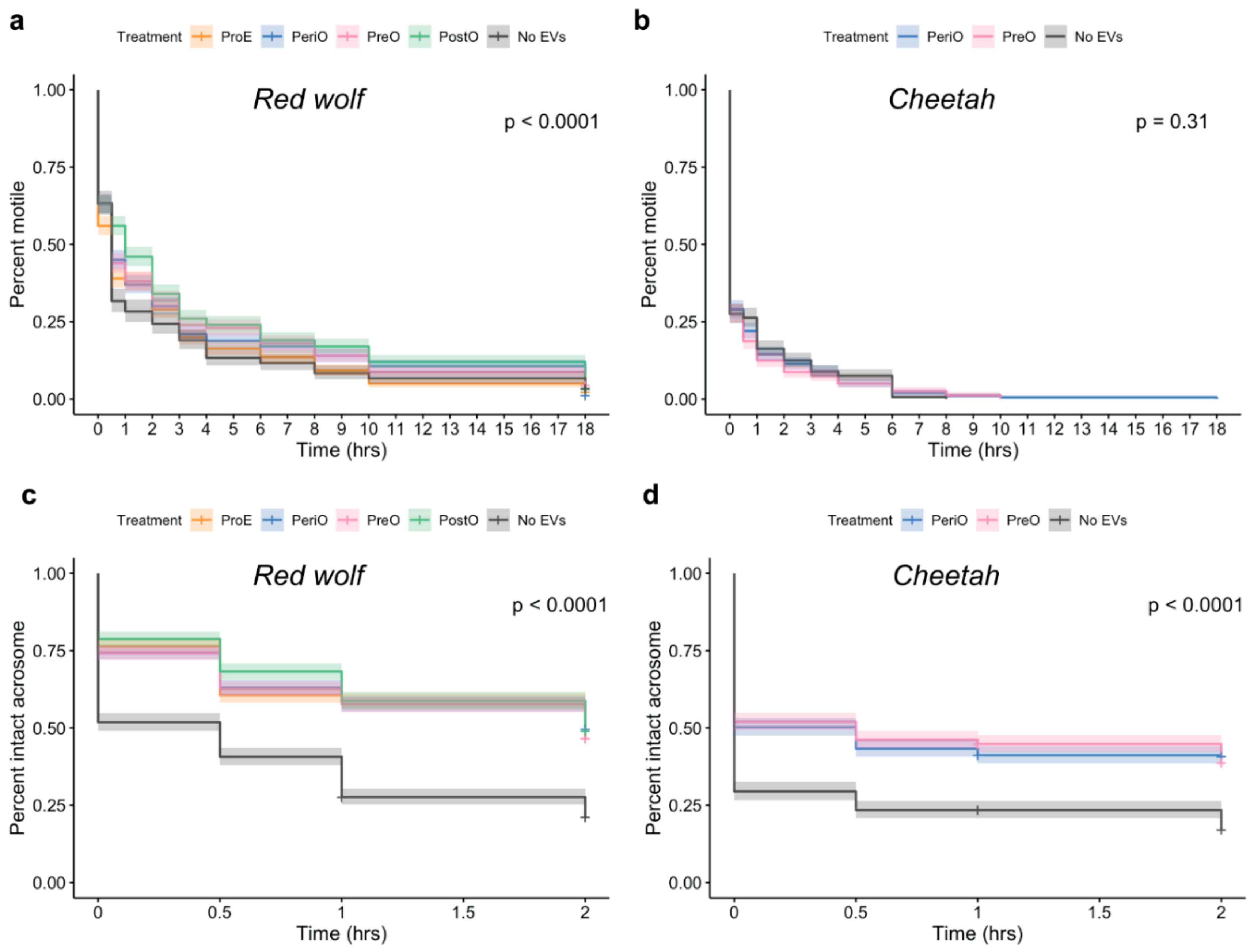
2.3. oEVs Improve Sperm Post-Thaw Motility in the Red Wolf but Not in the Cheetah
2.4. oEVs Prevents Premature Acrosome Reaction of Red Wolf and Cheetah Spermatozoa
3. Discussion
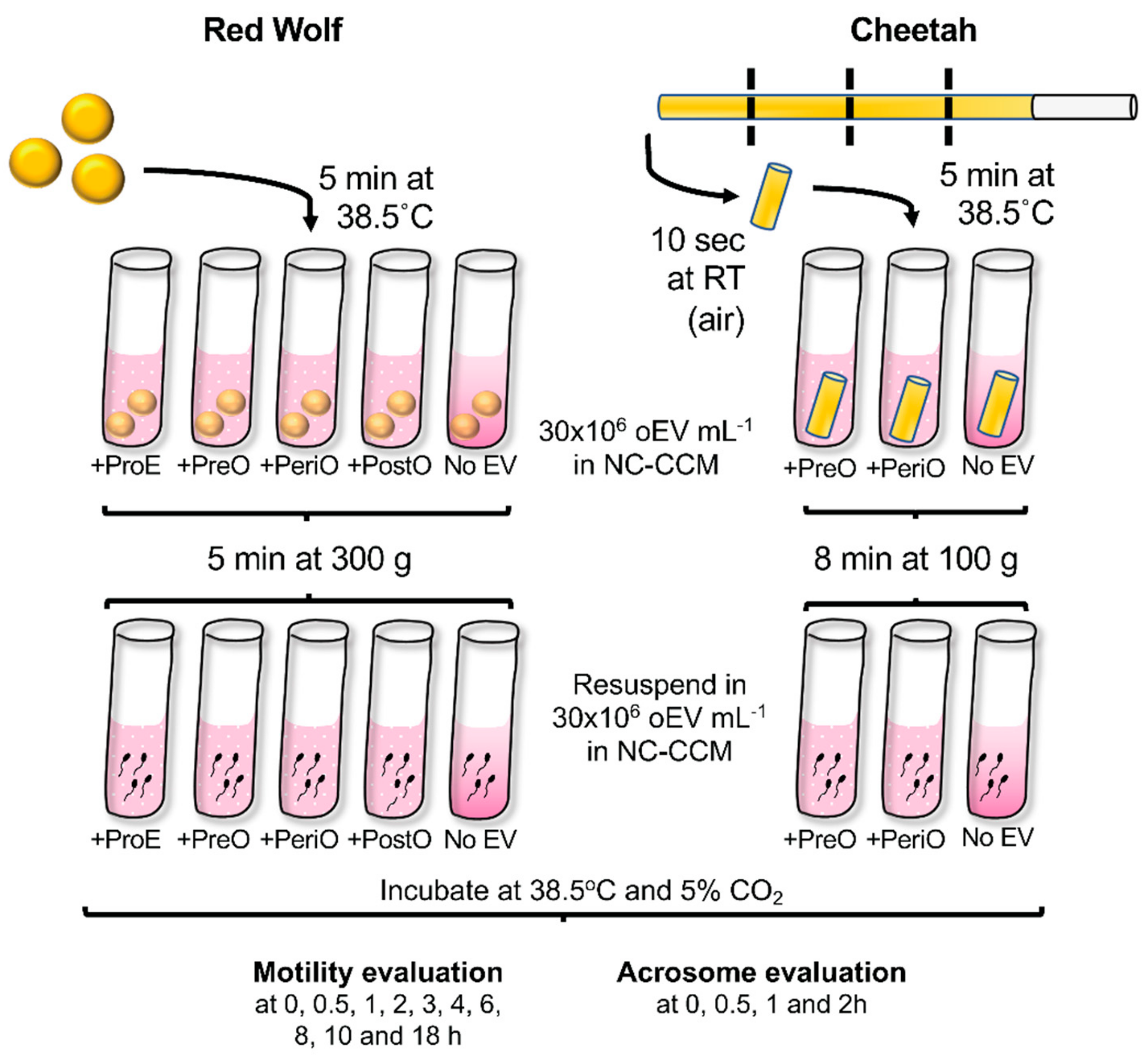
4. Materials and Methods
4.1. Animal Handling and Semen Collection
4.2. Sperm Cryopreservation
4.3. Oviductal EV (oEV) Isolation
4.4. oEVs Quantitation
4.5. oEVs Proteomic Analyses
4.6. Functional GO Clustering
4.7. oEV Transmission Electron Microscopy
4.8. Sperm Thawing
4.9. Sperm Motility and Acrosomal Analyses
4.10. Availability of Data and Materials
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AE | Acrosomal exocytosis |
| AI | Artificial insemination |
| ART | Assisted reproductive technologies |
| EVs | Extracellular vesicles |
| GO | Gene ontology |
| NC-CCM | Non-capacitation canine capacitation medium |
| NTA | Nanoparticle tracking analysis |
| oEVs | Oviductal extracellular vesicles |
| PeriO | Peri-ovulatory |
| PostO | Post-ovulatory |
| PreO | Pre-ovulatory |
| ProE | Proestrus |
| TEM | Transmission electron microscopy |
| TYB | Test yolk buffer |
References
- IUCN 2020. The IUCN Red List of Threatened Species; Version 2020-1; IUCN: Gland, Switzerland, 2020. [Google Scholar]
- Lasher, C. American Red Wolf SAFE Program Action Plan 2019–2022; Association of Zoos & Aquariums: Silver Spring, MD, USA, 2019. [Google Scholar]
- Wildt, D.E.; Rall, W.F.; Critser, J.K.; Monfort, S.L.; Seal, U.S. Living collections for biodiversity conservation. Bioscience 1997, 47, 1–20. [Google Scholar]
- Wildt, D.; Pukazhenthi, B.; Brown, J.; Monfort, S.; Howard, J.; Roth, T. Spermatology for understanding, managing and conserving rare species. Reprod. Fertil. Dev. 1995, 7, 811–24. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’Brien, S.J.; Johnson, W.E.; Driscoll, C.A.; Dobrynin, P.; Marker, L. Conservation genetics of the cheetah: Lessons learned and new opportunities. J. Hered. 2017, 108, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Marker, L.; Johnston, B.; Vannelli, K. International Cheetah (Acinonyx jubatus) Studbook; Cheetah Conservation Fund: Otjiwarongo, Namibia, 2015. [Google Scholar]
- Comizzoli, P.; Crosier, A.E.; Songsasen, N.; Gunther, M.S.; Howard, J.G.; Wildt, D.E. Advances in reproductive science for wild carnivore conservation. Reprod. Domest. Anim. 2009, 44, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.; Faurby, S.; Svenning, J.C. Mammal diversity will take millions of years to recover from the current biodiversity crisis. Proc. Natl. Acad. Sci. USA 2018, 115, 11262–11267. [Google Scholar] [CrossRef]
- Jewgenow, K.; Songsasen, N. Reproduction and advances in reproductive studies in carnivores. In Reproduction and Advances in Reproductive Studies in Carnivores; Holt, W., Brown, J.L., Comizzoli, P., Eds.; Springer US: New York, NY, USA, 2014; ISBN 978-1-4939-0820-2. [Google Scholar]
- Comizzoli, P. Biobanking efforts and new advances in male fertility preservation for rare and endangered species. Asian J. Androl. 2015, 17, 640–645. [Google Scholar] [CrossRef]
- Santymire, R.M.; Livieri, T.M.; Branvold-Faber, H.; Marinari, P.E. The black-footed ferret: on the brink of recovery? Adv. Exp. Med. Biol. 2014, 753, 119–34. [Google Scholar] [CrossRef]
- Howard, J.G.; Lynch, C.; Santymire, R.M.; Marinari, P.E.; Wildt, D.E. Recovery of gene diversity using long-term cryopreserved spermatozoa and artificial insemination in the endangered black-footed ferret. Anim. Conserv. 2016, 19, 102–111. [Google Scholar] [CrossRef]
- Swanson, W.F. Practical application of laparoscopic oviductal artificial insemination for the propagation of domestic cats and wild felids. Reprod. Fertil. Dev. 2019, 31, 27–39. [Google Scholar] [CrossRef]
- Li, D.; Wintle, N.J.P.; Zhang, G.; Wang, C.; Luo, B.; Martin-Wintle, M.S.; Owen, M.A.; Swaisgood, R.R. Analyzing the past to understand the future: Natural mating yields better reproductive rates than artificial insemination in the giant panda. Biol. Conserv. 2017, 216, 10–17. [Google Scholar] [CrossRef]
- Swain, J.E.; Smith, G.D. Advances in embryo culture platforms: Novel approaches to improve preimplantation embryo development through modifications of the microenvironment. Hum. Reprod. Update 2011, 17, 541–557. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, T.B.; Hermes, R.; Saragusty, J.; Potier, R.; Schwammer, H.M.; Balfanz, F.; Vielgrader, H.; Baker, B.; Bartels, P.; Göritz, F. Enriching the captive elephant population genetic pool through artificial insemination with frozen-thawed semen collected in the wild. Theriogenology 2012, 78, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Hermes, R.; Göritz, F.; Saragusty, J.; Sós, E.; Molnar, V.; Reid, C.E.; Schwarzenberger, F.; Hildebrandt, T.B. First successful artificial insemination with frozen-thawed semen in rhinoceros. Theriogenology 2009, 71, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, D.; Zhou, Y.; Zhou, Q.; Li, R.; Wang, C.; Huang, Z.; Hull, V.; Zhang, H. Factors affecting the outcome of artificial insemination using cryopreserved spermatozoa in the giant panda (Ailuropoda melanoleuca). Zoo Biol. 2012, 31, 561–573. [Google Scholar] [CrossRef]
- Drobnis, E.Z.; Yudin, A.I.; Cherr, G.N.; Katz, D.F. Hamster sperm penetration of the zona pellucida: kinematic analysis and mechanical implications. Dev. Biol. 1988, 130, 311–23. [Google Scholar] [CrossRef]
- Cummins, J.M.; Yanagimachi, R. Development of ability to penetrate the cumulus oophorus by hamster spermatozoa capacitated in vitro, in relation to the timing of the acrosome reaction. Gamete Res. 1986, 15, 187–212. [Google Scholar] [CrossRef]
- Yanagimachi, R. Fertility of mammalian spermatozoa: Its development and relativity. Zygote 1994, 2, 371–372. [Google Scholar] [CrossRef]
- Abdillah, D.A.; Setyawan, E.M.N.; Oh, H.J.; Ra, K.; Lee, S.H.; Kim, M.J.; Lee, B.C. Iodixanol supplementation during sperm cryopreservation improves protamine level and reduces reactive oxygen species of canine sperm. J. Vet. Sci. 2019, 20, 79–86. [Google Scholar] [CrossRef]
- Cheuquemán, C.; Sánchez, R.; Risopatrón, J.; Cheuquemán, C. Effect of sperm selection techniques in frozen/thawed cat spermatozoa on sperm motility analyzed by CASA system. Int. J. Morphol. 2017, 35, 1495–1501. [Google Scholar] [CrossRef][Green Version]
- Lockyear, K.M.; Goodrowe, K.L.; Waddell, W.T.; MacDonald, S.E. Comparison of different osmolalities and egg-yolk composition in processing media for the cryopreservation of red wolf (Canis rufus) sperm. Theriogenology 2009, 71, 469–479. [Google Scholar] [CrossRef]
- Crosier, A.E.; Henghali, J.N.; Howard, J.; Pukazhenthi, B.S.; Terrell, K.A.; Marker, L.L.; Wildt, D.E. Improved quality of cryopreserved cheetah (Acinonyx jubatus) spermatozoa after centrifugation through accudenz. J. Androl. 2009, 30, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Thuwanut, P.; Tipkantha, W.; Siriaroonrat, B.; Comizzoli, P.; Chatdarong, K. Beneficial effect of extracellular adenosine 5′-triphosphate treatment on the Indochinese leopard (Panthera pardus delacouri) sperm quality after cryopreservation. Reprod. Domest. Anim. 2017, 52, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Franklin, A.D.; Waddell, W.T.; Goodrowe, K.L. Red wolf (Canis rufus) sperm quality and quantity is affected by semen collection method, extender components, and post-thaw holding temperature. Theriogenology 2018, 116, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Crosier, A.E.; Pukazhenthi, B.S.; Henghali, J.N.; Howard, J.; Dickman, A.J.; Marker, L.; Wildt, D.E. Cryopreservation of spermatozoa from wild-born Namibian cheetahs (Acinonyx jubatus) and influence of glycerol on cryosurvival. Cryobiology 2006, 52, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.J. Regulation of sperm function by oviduct fluid and the epithelium: Insight into the role of glycans. Reprod. Domest. Anim. 2015, 50, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Mammalian sperm interactions with the female reproductive tract. Cell Tissue Res. 2016, 363, 185–194. [Google Scholar] [CrossRef]
- De Ferraz, A.M.M.M.; Carothers, A.; Dahal, R.; Noonan, M.J.; Songsasen, N. Oviductal extracellular vesicles interact with the spermatozoon’s head and mid-piece and improves its motility and fertilizing ability in the domestic cat. Sci. Rep. 2019, 9, 9484. [Google Scholar] [CrossRef]
- Al-Dossary, A.A.; Strehler, E.E.; Martin-DeLeon, P.A. Expression and secretion of plasma membrane Ca2+-ATPase 4a (PMCA4a) during murine estrus: Association with oviductal exosomes and uptake in sperm. PLoS ONE 2013, 8, 1–14. [Google Scholar] [CrossRef]
- Al-Dossary, A.A.; Bathala, P.; Caplan, J.L.; Martin-DeLeon, P.A. Oviductosome-sperm membrane interaction in cargo delivery: Detection of fusion and underlying molecular players using three-dimensional super-resolution structured illumination microscopy (SR-SIM). J. Biol. Chem. 2015, 290, 17710–17723. [Google Scholar] [CrossRef]
- Bathala, P.; Fereshteh, Z.; Li, K.; Al-Dossary, A.A.; Galileo, D.S.; Martin-DeLeon, P.A. Oviductal extracellular vesicles (oviductosomes, OVS) are conserved in humans: murine OVS play a pivotal role in sperm capacitation and fertility. MHR Basic Sci. Reprod. Med. 2018, 24, 143–157. [Google Scholar] [CrossRef]
- Franchi, A.; Moreno-Irusta, A.; Domínguez, E.M.; Adre, A.J.; Giojalas, L.C. Extracellular vesicles from oviductal isthmus and ampulla stimulate the induced acrosome reaction and signaling events associated with capacitation in bovine spermatozoa. J. Cell. Biochem. 2019, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Helwa, I.; Cai, J.; Drewry, M.D.; Zimmerman, A.; Dinkins, M.B.; Khaled, M.L.; Seremwe, M.; Dismuke, W.M.; Bieberich, E.; Stamer, W.D.; et al. A comparative study of serum exosome isolation using differential ultracentrifugation and three commercial reagents. PLoS ONE 2017, 12, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Fridman, W.-H.; Pagès, F.; Trajanoski, Z.; Galon, J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–3. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef]
- Kalra, H.; Drummen, G.; Mathivanan, S. Focus on extracellular vesicles: Introducing the next small big thing. Int. J. Mol. Sci. 2016, 17, 170. [Google Scholar] [CrossRef]
- Xu, R.; Rai, A.; Chen, M.; Suwakulsiri, W.; Greening, D.W.; Simpson, R.J. Extracellular vesicles in cancer - implications for future improvements in cancer care. Nat. Rev. Clin. Oncol. 2018, 15, 617–638. [Google Scholar] [CrossRef]
- Greening, D.W.; Nguyen, H.P.T.; Elgass, K.; Simpson, R.J.; Salamonsen, L.A. Human endometrial exosomes contain hormone-specific cargo modulating trophoblast adhesive capacity: Insights into endometrial-embryo interactions. Biol. Reprod. 2016, 94, 1–15. [Google Scholar] [CrossRef]
- Bidarimath, M.; Khalaj, K.; Kridli, R.T.; Kan, F.W.K.; Koti, M.; Tayade, C. Extracellular vesicle mediated intercellular communication at the porcine maternal-fetal interface: A new paradigm for conceptus-endometrial cross-talk. Sci. Rep. 2017, 7, 40476. [Google Scholar] [CrossRef]
- Burns, G.; Brooks, K.; Wildung, M.; Navakanitworakul, R.; Christenson, L.K.; Spencer, T.E. Extracellular vesicles in luminal fluid of the ovine uterus. PLoS ONE 2014, 9, e90913. [Google Scholar] [CrossRef]
- Almiñana, C.; Corbin, E.; Tsikis, G.; Alcântara-Neto, A.S.; Labas, V.; Reynaud, K.; Galio, L.; Uzbekov, R.; Garanina, A.S.; Druart, X.; et al. Oviduct extracellular vesicles protein content and their role during oviduct–embryo cross-talk. Reproduction 2017, 154, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Burns, G.W.; Brooks, K.E.; Spencer, T.E. Extracellular vesicles originate from the conceptus and uterus during early pregnancy in sheep. Biol. Reprod. 2016, 94, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Saez, F.; Frenette, G.; Sullivan, R. Epididymosomes and prostasomes: their roles in posttesticular maturation of the sperm cells. J. Androl. 2003, 24, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Rowlison, T.; Ottinger, M.A.; Comizzoli, P. Key factors enhancing sperm fertilizing ability are transferred from the epididymis to the spermatozoa via epididymosomes in the domestic cat model. J. Assist. Reprod. Genet. 2018, 35, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Frenette, G.; Sullivan, R. Prostasome-like particles are involved in the transfer of P25b from the bovine epididymal fluid to the sperm surface. Mol. Reprod. Dev. 2001, 59, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Almiñana, C.; Tsikis, G.; Labas, V.; Uzbekov, R.; da Silveira, J.C.; Bauersachs, S.; Mermillod, P. Deciphering the oviductal extracellular vesicles content across the estrous cycle: Implications for the gametes-oviduct interactions and the environment of the potential embryo. BMC Genom. 2018, 19, 1–27. [Google Scholar] [CrossRef]
- Lopera-Vásquez, R.; Hamdi, M.; Fernandez-Fuertes, B.; Maillo, V.V.; Beltrán-Breña, P.; Calle, A.; Redruello, A.; López-Martí, S.; Gutierrez-Adán, A.; Yañez-Mó, M.; et al. Extracellular vesicles from BOEC in in vitro embryo development and quality. PLoS ONE 2016, 11, e0148083. [Google Scholar] [CrossRef]
- Qu, P.; Zhao, Y.; Wang, R.; Zhang, Y.; Li, L.; Fan, J.; Liu, E. Extracellular vesicles derived from donor oviduct fluid improved birth rates after embryo transfer in mice. Reprod. Fertil. Dev. 2018. [Google Scholar] [CrossRef]
- Qamar, A.Y.; Fang, X.; Kim, M.J.; Cho, J. Improved post-thaw quality of canine semen after treatment with exosomes from conditioned medium of adipose-derived mesenchymal stem cells. Animals 2019, 9, 865. [Google Scholar] [CrossRef]
- Mokarizadeh, A.; Rezvanfar, M.-A.; Dorostkar, K.; Abdollahi, M. Mesenchymal stem cell derived microvesicles: Trophic shuttles for enhancement of sperm quality parameters. Reprod. Toxicol. 2013, 42, 78–84. [Google Scholar] [CrossRef]
- Yeste, M. Sperm cryopreservation update: Cryodamage, markers, and factors affecting the sperm freezability in pigs. Theriogenology 2016, 85, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Comizzoli, P.; Songsasen, N.; Hagedorn, M.; Wildt, D.E. Comparative cryobiological traits and requirements for gametes and gonadal tissues collected from wildlife species. Theriogenology 2012, 78, 1666–1681. [Google Scholar] [CrossRef] [PubMed]
- Paulson, R.J.; Comizzoli, P. Addressing challenges in developing and implementing successful in vitro fertilization in endangered species: An opportunity for humanity to “give back”. Fertil. Steril. 2018, 109, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.F. The causes of reduced fertility with cryopreserved semen. Anim. Reprod. Sci. 2000, 60–61, 481–492. [Google Scholar] [CrossRef]
- Clulow, J.R.; Evans, G.; Maxwell, W.M.C.; Morris, L.H. Evaluation of the function of fresh and frozen-thawed sex-sorted and non-sorted stallion spermatozoa using a heterologous oocyte binding assay. Reprod. Fertil. Dev. 2010, 22, 710–7. [Google Scholar] [CrossRef]
- Jeong, D.H.; Kim, J.H.; Na, K.J. Characterization and cryopreservation of Amur leopard cats (Prionailurus bengalensis euptilurus) semen collected by urethral catheterization. Theriogenology 2018, 119, 91–95. [Google Scholar] [CrossRef]
- Koderle, M.; Aurich, C.; Schäfer-Somi, S. The influence of cryopreservation and seminal plasma on the chromatin structure of dog spermatozoa. Theriogenology 2009, 72, 1215–1220. [Google Scholar] [CrossRef]
- Cremades, T.; Roca, J.; Rodriguez-Martinez, H.; Abaigar, T.; Vazquez, J.M.; Martinez, E.A. Kinematic changes during the cryopreservation of boar spermatozoa. J. Androl. 2005, 26, 610–8. [Google Scholar] [CrossRef]
- Valcarce, D.G.; Cartón-García, F.; Herráez, M.P.; Robles, V. Effect of cryopreservation on human sperm messenger RNAs crucial for fertilization and early embryo development. Cryobiology 2013, 67, 84–90. [Google Scholar] [CrossRef]
- Sancho, S.; Casas, I.; Ekwall, H.; Saravia, F.; Rodriguez-Martinez, H.; Rodriguez-Gil, J.E.; Flores, E.; Pinart, E.; Briz, M.; Garcia-Gil, N.; et al. Effects of cryopreservation on semen quality and the expression of sperm membrane hexose transporters in the spermatozoa of Iberian pigs. Reproduction 2007, 134, 111–121. [Google Scholar] [CrossRef][Green Version]
- Naresh, S.; Atreja, S.K. The protein tyrosine phosphorylation during in vitro capacitation and cryopreservation of mammalian spermatozoa. Cryobiology 2015, 70, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Kumaresan, A.; Siqueira, A.P.; Hossain, M.S.; Bergqvist, A.S. Cryopreservation-induced alterations in protein tyrosine phosphorylation of spermatozoa from different portions of the boar ejaculate. Cryobiology 2011, 63, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Manee-In, S.; Parmornsupornvichit, S.; Kraiprayoon, S.; Tharasanit, T.; Chanapiwat, P.; Kaeoket, K. L-carnitine supplemented extender improves cryopreserved-thawed cat epididymal sperm motility. Asian-Australas. J. Anim. Sci. 2014. [Google Scholar] [CrossRef] [PubMed]
- Pukazhenthi, B.S.; Wildt, D.E.; Howard, J.G. The phenomenon and significance of teratospermia in felids. J. Reprod. Fertil. Suppl. 2001, 57, 423–33. [Google Scholar] [PubMed]
- Pukazhenthi, B.S.; Neubauer, K.; Jewgenow, K.; Howard, J.; Wildt, D.E. The impact and potential etiology of teratospermia in the domestic cat and its wild relatives. Theriogenology 2006, 66, 112–121. [Google Scholar] [CrossRef]
- Pukazhenthi, B.; Pelican, K.; Wildt, D.; Howard, J. Sensitivity of domestic cat (Felis catus) sperm from normospermic versus teratospermic donors to cold-induced acrosomal damage. Biol. Reprod. 1999, 61, 135–141. [Google Scholar] [CrossRef]
- Dejonge, C.J.; Barratt, C.L.R.R.; Radwanska, E.W.A.; Cooke, I.D.A.N.D.; de Jonge, C.J.; Barratt, C.L.R.R.; Radwanska, E.W.A.; Cooke, I.D.A.N.D. The acrosome reaction-inducing effect of human follicular and oviductal fluid. J. Androl. 1993, 141, 359–365. [Google Scholar] [CrossRef]
- Mcnutt, T.; Killian, G. Influence of bovine follicular and oviduct fluids on sperm capacitation in vitro. J. Androl. 1991, 12, 244–252. [Google Scholar] [CrossRef]
- Jones, R.C. Changes occurring in the head of boar spermatozoa: vesiculation or vacuolation of the acrosome? J. Reprod. Fertil. 1973, 33, 113–118. [Google Scholar] [CrossRef][Green Version]
- Okada, A.; Igarashi, H.; Kuroda, M.; Terao, K.; Yoshikawa, Y.; Sankai, T. Cryopreservation-induced acrosomal vesiculation in live spermatozoa from cynomolgus monkeys (Macaca fascicularis). Hum. Reprod. 2001, 16, 2139–2147. [Google Scholar] [CrossRef][Green Version]
- Songsasen, N.; Wildt, D.E. Oocyte biology and challenges in developing in vitro maturation systems in the domestic dog. Anim. Reprod. Sci. 2007, 98, 2–22. [Google Scholar] [CrossRef] [PubMed]
- Karre, I.; Meyer-Lindenberg, A.; Urhausen, C.; Beineke, A.; Meinecke, B.; Piechotta, M.; Beyerbach, M.; Günzel-Apel, A.-R. Distribution and viability of spermatozoa in the canine female genital tract during post-ovulatory oocyte maturation. Acta Vet. Scand. 2012, 54, 49. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, T.; Takahashi, F.; Hori, T.; Kawakami, E.; Concannon, P.W.P. Prolonged duration of fertility of dog ova. Reprod. Domest. Anim. 2009, 44, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Concannon, P.W.; Castracane, V.D.; Temple, M.; Montanez, A. Endocrine control of ovarian function in dogs and other carnivores. Anim. Reprod. 2009, 6, 172–193. [Google Scholar]
- Tajaddini Mahani, S.; Behnam, B.; Abbassi, M.; Asgari, H.; Nazmara, Z.; Shirinbayan, P.; Joghataei, M.; Koruji, M. Tsga10 expression correlates with sperm profiles in the adult formalin-exposed mice. Andrologia 2016, 48, 1092–1099. [Google Scholar] [CrossRef]
- Restrepo, G.; Rojano, B.; Usuga, A. Relationship of cysteine-rich secretory protein-3 gene and protein with semen quality in stallions. Reprod. Domest. Anim. 2019, 54, 39–45. [Google Scholar] [CrossRef]
- Jobim, M.I.M.; Trein, C.; Zirkler, H.; Gregory, R.M.; Sieme, H.; Mattos, R.C. Two-dimensional polyacrylamide gel electrophoresis of equine seminal plasma proteins and their relation with semen freezability. Theriogenology 2011, 76, 765–771. [Google Scholar] [CrossRef]
- Lim, S.; Kierzek, M.; O’Connor, A.E.; Brenker, C.; Merriner, D.J.; Okuda, H.; Volpert, M.; Gaikwad, A.; Bianco, D.; Potter, D.; et al. CRISP2 is a regulator of multiple aspects of sperm function and male fertility. Endocrinology 2019, 160, 915–924. [Google Scholar] [CrossRef]
- Schuh, K.; Cartwright, E.J.; Jankevics, E.; Bundschu, K.; Liebermann, J.; Williams, J.C.; Armesilla, A.L.; Emerson, M.; Oceandy, D.; Knobeloch, K.-P.; et al. Plasma membrane Ca2+ ATPase 4 is required for sperm motility and male fertility. J. Biol. Chem. 2004, 279, 28220–28226. [Google Scholar] [CrossRef]
- Holt, W.V.; Del Valle, I.; Fazeli, A. Heat shock protein A8 stabilizes the bull sperm plasma membrane during cryopreservation: Effects of breed, protein concentration, and mode of use. Theriogenology 2015, 84, 693–701. [Google Scholar] [CrossRef]
- Georgiou, A.S.; Snijders, A.P.L.; Sostaric, E.; Aflatoonian, R.; Vazquez, J.L.; Vazquez, J.M.; Roca, J.; Martinez, E.A.; Wright, P.C.; Fazeli, A. Modulation of the oviductal environment by gametes. J. Proteome Res. 2007, 6, 4656–4666. [Google Scholar] [CrossRef] [PubMed]
- Reynaud, K.; Saint-Dizier, M.; Tahir, M.Z.; Havard, T.; Thoumire, S.; Fontbonne, A.; Harichaux, G.; Labas, V.; Thoumire, S.; Fontbonne, A.; et al. Progesterone plays a critical role in canine oocyte maturation and fertilization1. Biol. Reprod. 2015, 93, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Elliott, R.M.A.; Lloyd, R.E.; Fazeli, A.; Sostaric, E.; Georgiou, A.S.; Satake, N.; Watson, P.F.; Holt, W.V. Effects of HSPA8, an evolutionarily conserved oviductal protein, on boar and bull spermatozoa. Reproduction 2009, 137, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, R.E.; Elliott, R.M.A.; Fazeli, A.; Watson, P.F.; Holt, W.V. Effects of oviductal proteins, including heat shock 70 kDa protein 8, on survival of ram spermatozoa over 48 h in vitro. Reprod. Fertil. Dev. 2009, 21, 408–418. [Google Scholar] [CrossRef]
- Moein-Vaziri, N.; Phillips, I.; Smith, S.; Almiňana, C.; Maside, C.; Gil, M.A.; Roca, J.; Martinez, E.A.; Holt, W.V.; Pockley, A.G.; et al. Heat-shock protein A8 restores sperm membrane integrity by increasing plasma membrane fluidity. Reproduction 2014, 147, 719–732. [Google Scholar] [CrossRef]
- Alvarez-Rodríguez, M.; Alvarez, M.; Borragan, S.; Martinez-Pastor, F.; Holt, W.V.; Fazeli, A.; de Paz, P.; Anel, L. The addition of heat shock protein HSPA8 to cryoprotective media improves the survival of brown bear (Ursus arctos) spermatozoa during chilling and after cryopreservation. Theriogenology 2013, 79, 541–550. [Google Scholar] [CrossRef]
- Lee, J.-S.; Kwon, W.-S.; Rahman, M.S.; Yoon, S.-J.; Park, Y.-J.; Pang, M.-G. Actin-related protein 2/3 complex-based actin polymerization is critical for male fertility. Andrology 2015, 3, 937–946. [Google Scholar] [CrossRef]
- Skotland, T.; Hessvik, N.P.; Sandvig, K.; Llorente, A. Exosomal lipid composition and the role of ether lipids and phosphoinositides in exosome biology. J. Lipid Res. 2019, 60, 9–18. [Google Scholar] [CrossRef]
- Piehl, L.L.; Cisale, H.; Torres, N.; Capani, F.; Sterin-Speziale, N.; Hager, A. Biochemical characterization and membrane fluidity of membranous vesicles isolated from boar seminal plasma. Anim. Reprod. Sci. 2006, 92, 401–410. [Google Scholar] [CrossRef]
- Arienti, G.; Polci, A.; De Cosmo, A.; Saccardi, C.; Carlini, E.; Palmerini, C.A. Lipid fatty acid and protein pattern of equine prostasome-like vesicles. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 128, 661–666. [Google Scholar] [CrossRef]
- Arienti, G.; Carlini, E.; Polci, A.; Cosmi, E.V.; Palmerini, C.A. Fatty acid pattern of human prostasome lipid. Arch. Biochem. Biophys. 1998, 358, 391–395. [Google Scholar] [CrossRef]
- Salmon, V.M.; Leclerc, P.; Bailey, J.L. Cholesterol-loaded cyclodextrin increases the cholesterol content of goat sperm to improve cold and osmotic resistance and maintain sperm function after cryopreservation1. Biol. Reprod. 2016, 94, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mocé, E.; Blanch, E.; Tomás, C.; Graham, J. Use of Cholesterol in sperm cryopreservation: Present moment and perspectives to future. Reprod. Domest. Anim. 2010, 45, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Yadav, H.P.; Kumar, A.; Shah, N.; Chauhan, D.S.; Lone, S.A.; Swain, D.K.; Saxena, A. Effect of cholesterol-loaded cyclodextrin on membrane and acrosome status of Hariana bull sperm during cryopreservation. Cryo Lett. 2018, 39, 386–390. [Google Scholar] [PubMed]
- Koester, D.C.; Freeman, E.W.; Brown, J.L.; Wildt, D.E.; Terrell, K.A.; Franklin, A.D.; Crosier, A.E. Motile sperm output by male cheetahs (Acinonyx jubatus) managed ex situ is influenced by public exposure and number of care-givers. PLoS ONE 2015. [Google Scholar] [CrossRef]
- Concannon, P.W. Reproductive cycles of the domestic bitch. Anim. Reprod. Sci. 2011, 124, 200–210. [Google Scholar] [CrossRef]
- Reynaud, K.; Fontbonne, A.; Saint-Dizier, M.; Thoumire, S.; Marnier, C.; Tahir, M.; Meylheuc, T.; Chastant-Maillard, S. Folliculogenesis, ovulation and endocrine control of oocytes and embryos in the Dog. Reprod. Domest. Anim. 2012, 47, 66–69. [Google Scholar] [CrossRef]
- Malandain, E.; Rault, D.; Froment, E.; Baudon, S.; Desquilbet, L.; Begon, D.; Chastant-Maillard, S. Follicular growth monitoring in the female cat during estrus. Theriogenology 2011, 76, 1337–1346. [Google Scholar] [CrossRef]
- Hughes, C.S.; Moggridge, S.; Müller, T.; Sorensen, P.H.; Morin, G.B.; Krijgsveld, J. Single-pot, solid-phase-enhanced sample preparation for proteomics experiments. Nat. Protoc. 2019, 14, 68–85. [Google Scholar] [CrossRef]
- Bjornson, R.D.; Carriero, N.J.; Colangelo, C.; Shifman, M.; Cheung, K.; Miller, P.L.; Williams, K. X!!Tandem, an improved method for running X!Tandem in parallel on collections of commodity computers. J. Proteome Res. 2008, 7, 293–299. [Google Scholar] [CrossRef]
- Maclean, B.; Eng, J.K.; Beavis, R.C.; Mcintosh, M. General framework for developing and evaluating database scoring algorithms using the TANDEM search engine. Bioinformatics 2006, 22, 2830–2832. [Google Scholar] [CrossRef] [PubMed]
- Geer, L.Y.; Markey, S.P.; Kowalak, J.A.; Wagner, L.; Xu, M.; Maynard, D.M.; Yang, X.; Shi, W.; Bryant, S.H. Open mass spectrometry search algorithm. J. Proteome Res. 2004, 3, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Sturm, M.; Bertsch, A.; Gröpl, C.; Hildebrandt, A.; Hussong, R.; Lange, E.; Pfeifer, N.; Schulz-Trieglaff, O.; Zerck, A.; Reinert, K.; et al. OpenMS – An open-source software framework for mass spectrometry. BMC Bioinform. 2008, 9, 163. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.J. rentrez: An R package for the NCBI eUtils API. R J. 2017, 9, 520–526. [Google Scholar] [CrossRef]
- Dieterle, F.; Ross, A.; Schlotterbeck, G.; Senn, H. Probabilistic quotient normalization as robust method to account for dilution of complex niological mixtures. Application in 1 H NMR metabonomics. Anal. Chem. 2006, 78, 4281–4290. [Google Scholar] [CrossRef]
- Noonan, M.J.; Tinnesand, H.V.; Buesching, C.D. Normalizing gas-chromatography-mass spectrometry data: Method choice can alter biological inference. BioEssays 2018, 40, 1700210. [Google Scholar] [CrossRef]
- Tarazona, S.; García, F.; Ferrer, A.; Dopazo, J.; Conesa, A. NOIseq: a RNA-seq differential expression method robust for sequencing depth biases. EMBnet. J. 2012, 17, 18. [Google Scholar] [CrossRef]
- Mahi, C.A.; Yanagimachi, R. Capacitation, acrosome reaction, and egg penetration by canine spermatozoa in a simple defined medium. Gamete Res. 1978, 1, 101–109. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric estimation from incomplete observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Therneau, T.M.; Lumley, T. Package ‘Survival’ 2014; CRAN: Vienna, Austria, 2014. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Almeida Monteiro Melo Ferraz, M.; Nagashima, J.B.; Noonan, M.J.; Crosier, A.E.; Songsasen, N. Oviductal Extracellular Vesicles Improve Post-Thaw Sperm Function in Red Wolves and Cheetahs. Int. J. Mol. Sci. 2020, 21, 3733. https://doi.org/10.3390/ijms21103733
de Almeida Monteiro Melo Ferraz M, Nagashima JB, Noonan MJ, Crosier AE, Songsasen N. Oviductal Extracellular Vesicles Improve Post-Thaw Sperm Function in Red Wolves and Cheetahs. International Journal of Molecular Sciences. 2020; 21(10):3733. https://doi.org/10.3390/ijms21103733
Chicago/Turabian Stylede Almeida Monteiro Melo Ferraz, Marcia, Jennifer Beth Nagashima, Michael James Noonan, Adrienne E. Crosier, and Nucharin Songsasen. 2020. "Oviductal Extracellular Vesicles Improve Post-Thaw Sperm Function in Red Wolves and Cheetahs" International Journal of Molecular Sciences 21, no. 10: 3733. https://doi.org/10.3390/ijms21103733
APA Stylede Almeida Monteiro Melo Ferraz, M., Nagashima, J. B., Noonan, M. J., Crosier, A. E., & Songsasen, N. (2020). Oviductal Extracellular Vesicles Improve Post-Thaw Sperm Function in Red Wolves and Cheetahs. International Journal of Molecular Sciences, 21(10), 3733. https://doi.org/10.3390/ijms21103733




