Exacerbated Imiquimod-Induced Psoriasis-Like Skin Inflammation in IRF5-Deficient Mice
Abstract
1. Introduction
2. Results
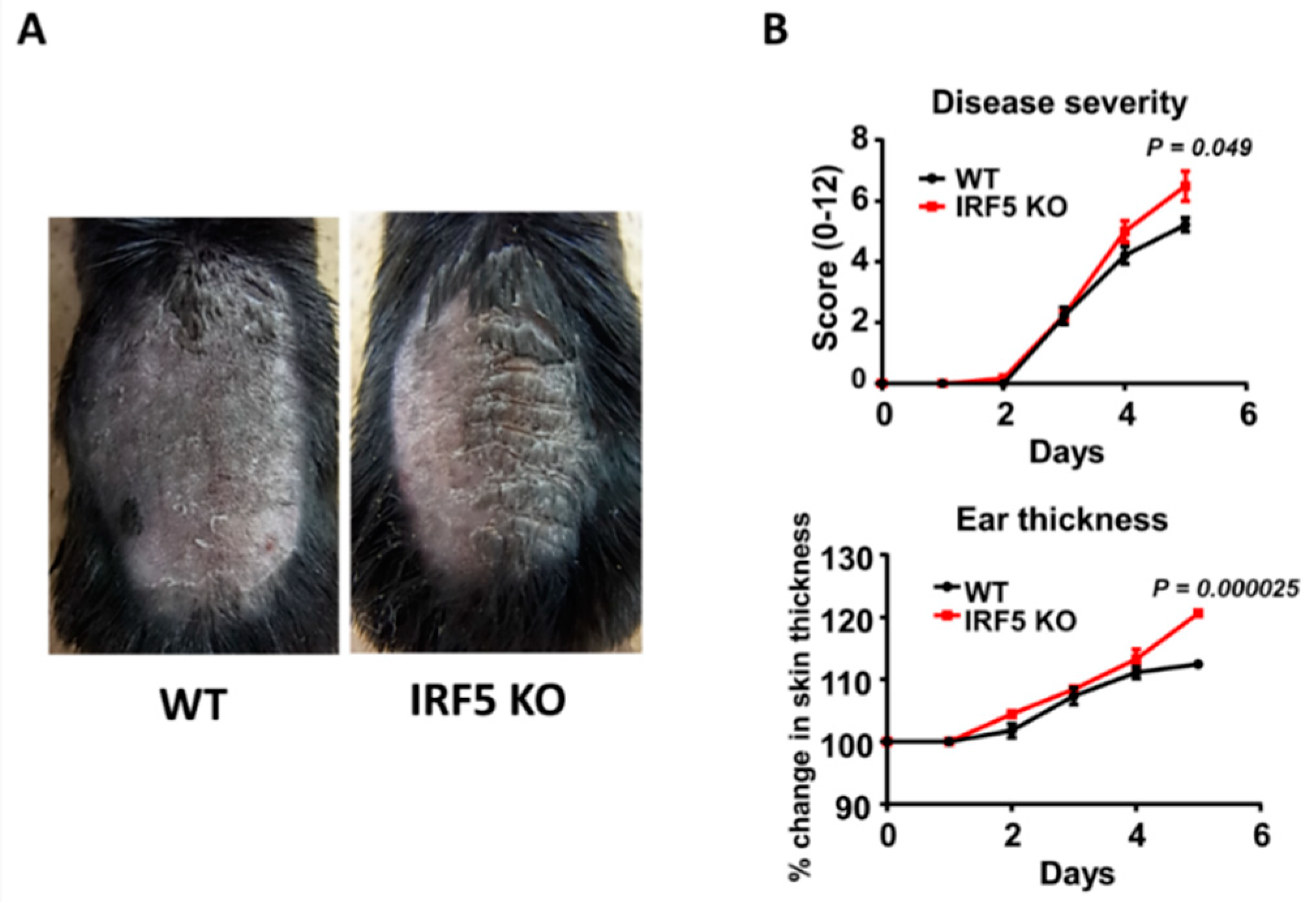
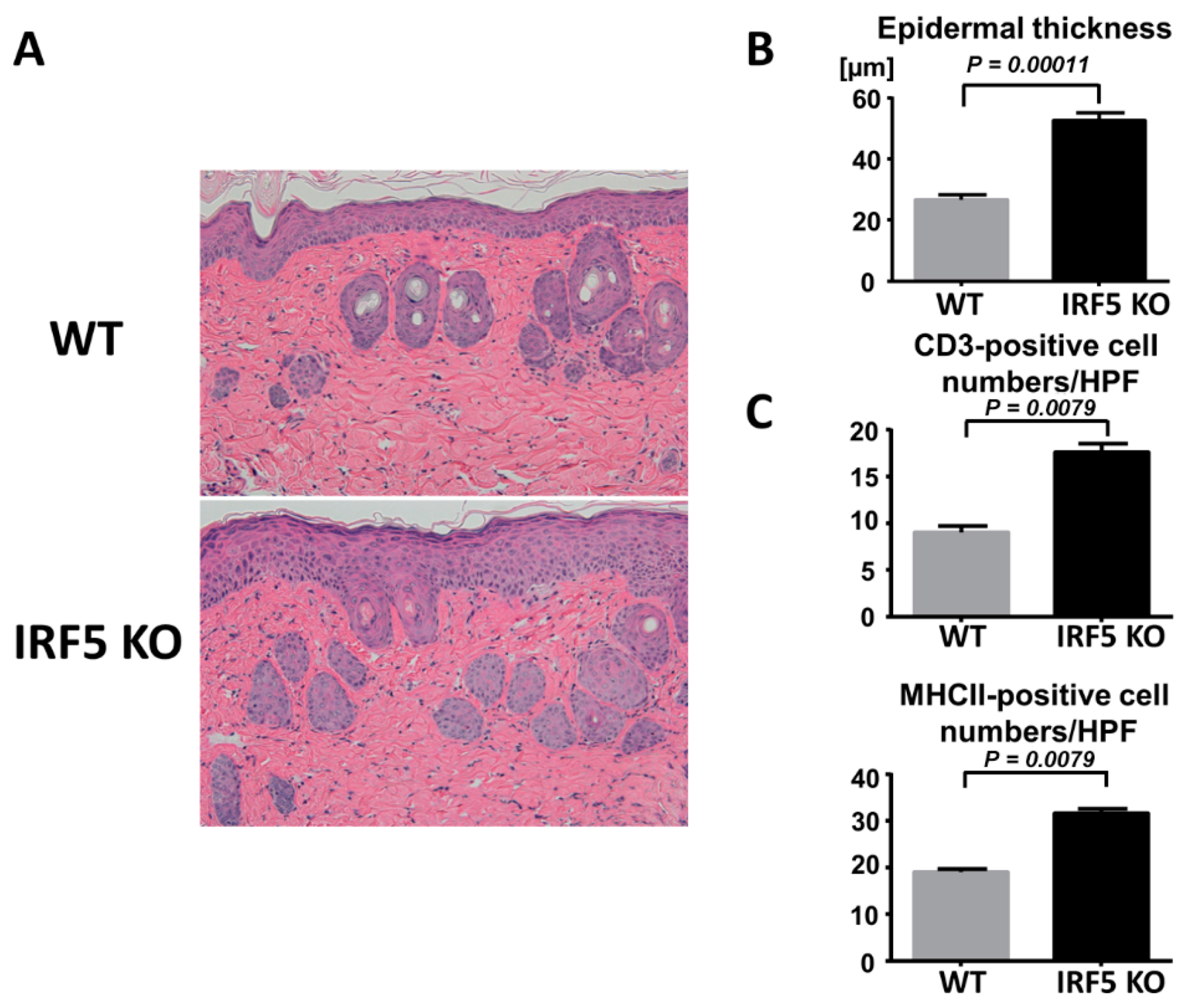
2.1. IRF5 Deficiency Exacerbates Psoriasis-Like Skin Inflammation Induced by Imiquimod Treatment
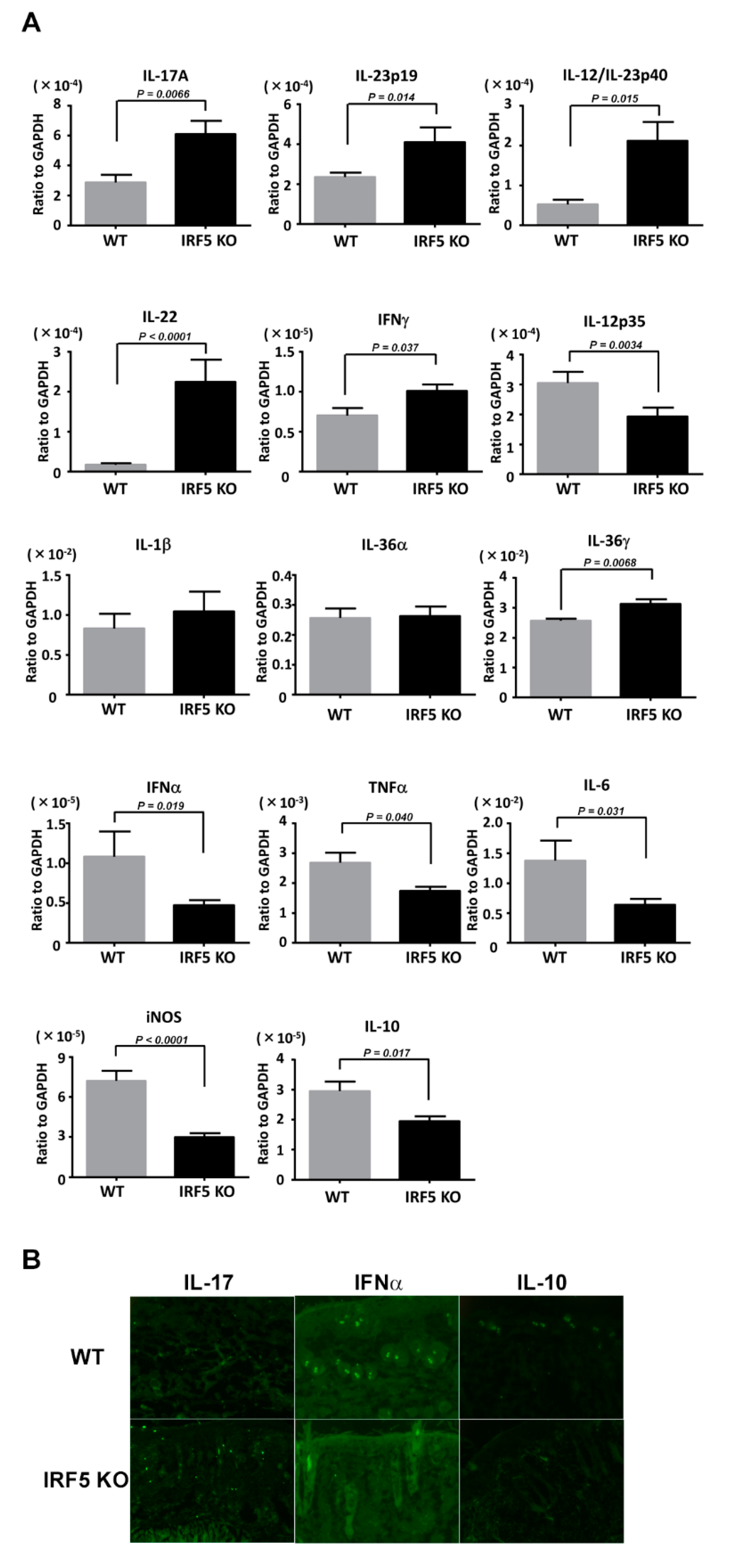
2.2. IRF5 Deficiency Increases Th17 Cytokine mRNA Levels and Decreases IL-10 mRNA Levels
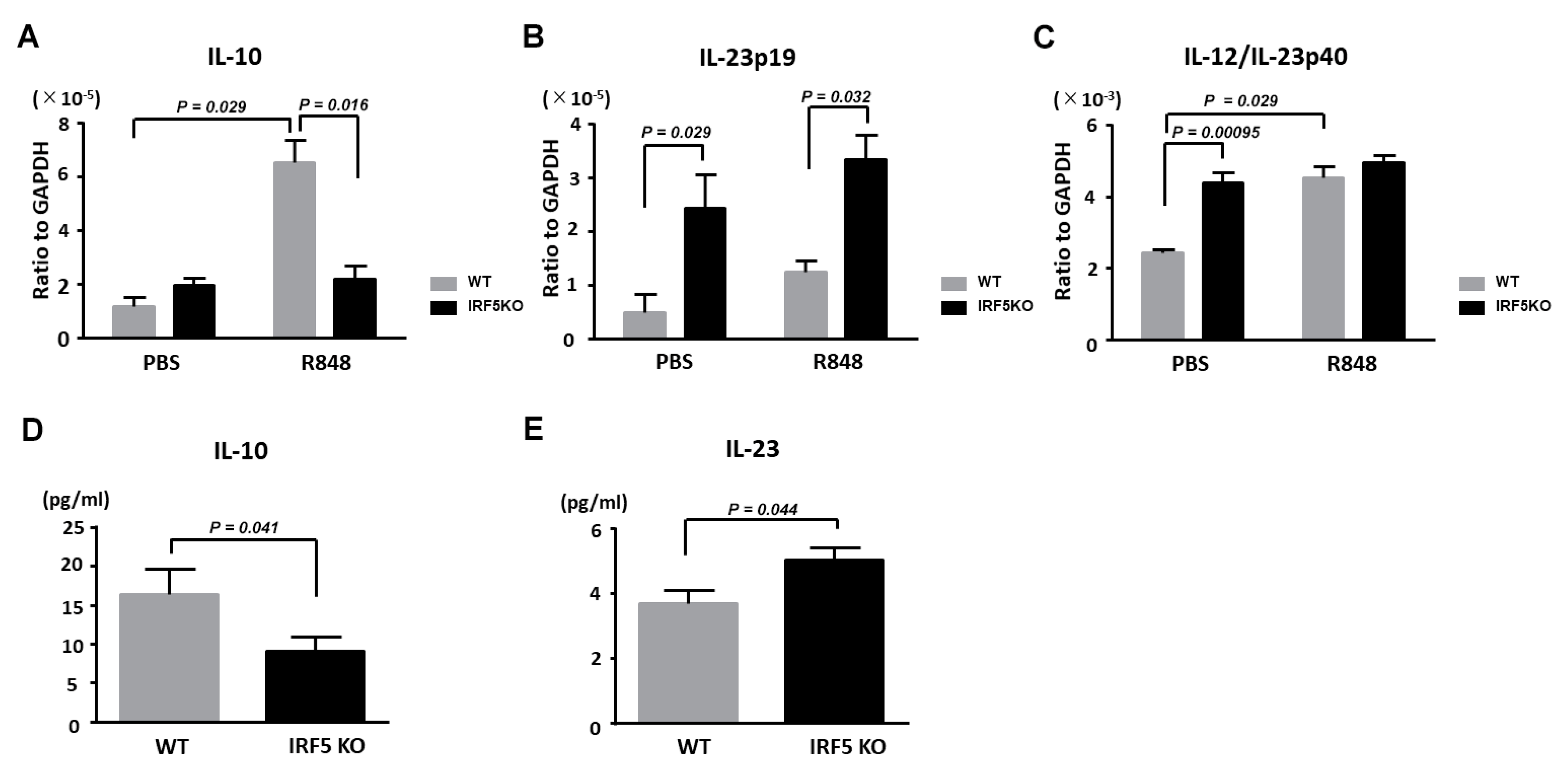
2.3. Decreased IL-10 Expression and Increased IL-23 Expression by Dendritic Cells in IRF5 KO Mice
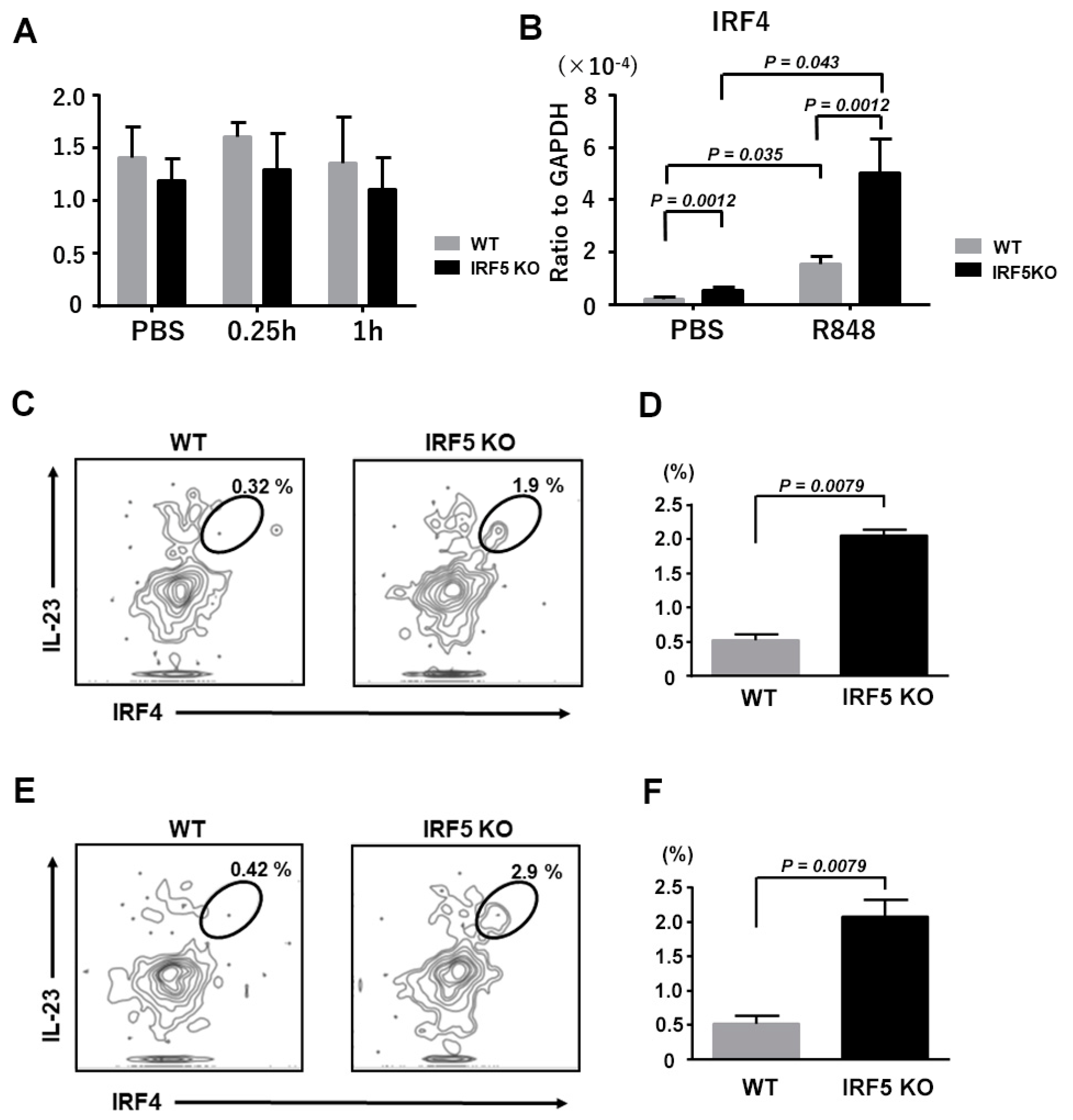
2.4. Upregulated IRF-4 Expression in Dendritic Cells from IRF5 KO Mice
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Induction of Psoriasiform Skin Inflammation by Imiquimod
4.3. Histological and Immunohistochemical Analysis
4.4. RNA Isolation and Quantitative Reverse-Transcription PCR Analysis
4.5. Cell Isolations and DC Cultures
4.6. Intracellular Flow Cytometry
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Di Cesare, A.; Di Meglio, P.; Nestle, F.O. The IL-23/Th17 axis in the immunopathogenesis of psoriasis. J. Investig. Dermatol. 2009, 129, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
- Lowes, M.A.; Bowcock, A.M.; Krueger, J.G. Pathogenesis and therapy of psoriasis. Nature 2007, 445, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Grjibovski, A.M.; Olsen, A.O.; Magnus, P.; Harris, J.R. Psoriasis in Norwegian twins: Contribution of genetic and environmental effects. J. Eur. Acad. Dermatol. Venereol. 2007, 21, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Boehncke, W.H.; Schön, M.P. Psoriasis. Lancet 2015, 386, 983–994. [Google Scholar] [CrossRef]
- Elder, J.T.; Bruce, A.T.; Gudjonsson, J.E.; Johnston, A.; Stuart, P.E.; Tejasvi, T.; Voorhees, J.J.; Abecasis, G.R.; Nair, R.P. Molecular dissection of psoriasis: Integrating genetics and biology. J. Investig. Dermatol. 2010, 130, 1213–1226. [Google Scholar] [CrossRef]
- Nestle, F.O.; Conrad, C.; Tun-Kyi, A.; Homey, B.; Gombert, M.; Boyman, O.; Burg, G.; Liu, Y.J.; Gilliet, M. Plasmacytoid predendritic cells initiate psoriasis through interferon-alpha production. J. Exp. Med. 2005, 202, 135–143. [Google Scholar] [CrossRef]
- Raimondo, A.; Lembo, S.; Di Caprio, R.; Donnarumma, G.; Monfrecola, G.; Balato, N.; Ayala, F.; Balato, A. Psoriatic cutaneous inflammation promotes human monocyte differentiation into active osteoclasts, facilitating bone damage. Eur. J. Immunol. 2017, 47, 1062–1074. [Google Scholar] [CrossRef]
- Balato, A.; Scala, E.; Balato, N.; Caiazzo, G.; Di Caprio, R.; Monfrecola, G.; Raimondo, A.; Lembo, S.; Ayala, F. Biologics that inhibit the Th17 pathway and related cytokines to treat inflammatory disorders. Expert Opin. Biol. Ther. 2017, 17, 1363–1374. [Google Scholar] [CrossRef]
- Raimondo, A.; Lembo, S.; Caiazzo, G.; Di Caprio, R.; Balato, A. Anti IL-17 therapy rapidly decreases osteoclast activity in psoriatic patients: A novel quality in addition to its efficacy and safety. G. Ital. Dermatol. Venereol. 2018. [Google Scholar] [CrossRef]
- Furue, K.; Ito, T.; Furue, M. Differential efficacy of biologic treatments targeting the TNF-α/IL-23/IL-17 axis in psoriasis and psoriatic arthritis. Cytokine 2018, 111, 182–188. [Google Scholar] [CrossRef]
- Raimondo, A.; Balato, A.; Megna, M.; Balato, N. Limitations of current monoclonal antibodies for plaque-type psoriasis and an outlook for the future. Expert Opin. Biol. Ther. 2018, 18, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Taniguchi, T. IRFs: Master regulators of signalling by Toll-like receptors and cytosolic pattern-recognition receptors. Nat. Rev. Immunol. 2006, 6, 644–658. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Kitagawa, M.; Tanaka, N.; Yamamoto, H.; Harada, K.; Ishihara, M.; Taniguchi, T. Anti-oncogenic and oncogenic potentials of interferon regulatory factors-1 and-2. Science 1993, 259, 971–974. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.; Howie, S.E.; Weller, R.; Sabin, E.; Hunter, J.A.; McKenzie, R.C. Psoriatic keratinocytes show reduced IRF-1 and STAT-1alpha activation in response to gamma-IFN. FASEB J. 1999, 13, 495–502. [Google Scholar] [CrossRef] [PubMed]
- van der Fits, L.; van der Wel, L.I.; Laman, J.D.; Prens, E.P.; Verschuren, M.C. Psoriatic lesional skin exhibits an aberrant expression pattern of interferon regulatory factor-2 (IRF-2). J. Pathol. 2003, 199, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, J.; Charon, C.; Baker, B.S.; Powles, A.V.; Rogers, S.; Caird, A.; Smedley, D.; Halford, S.; Fry, L.; McCarthy, M.I. Variation at the IRF2 gene and susceptibility to psoriasis in chromosome 4q-linked families. J. Investig. Dermatol. 2004, 122, 640–643. [Google Scholar] [CrossRef]
- Van Der Fits, L.; Kant, M.; Van Der Wel, L.I.; Prens, E.P. Polymorphisms in the interferon regulatory factor-1 promoter are not associated with psoriasis and do not influence IFN-alpha-induced Th1 polarization. J. Interferon Cytokine Res. 2007, 27, 841–846. [Google Scholar] [CrossRef]
- Hida, S.; Ogasawara, K.; Sato, K.; Abe, M.; Takayanagi, H.; Yokochi, T.; Sato, T.; Hirose, S.; Shirai, T.; Taki, S.; et al. CD8(+) T cell-mediated skin disease in mice lacking IRF-2, the transcriptional attenuator of interferon-alpha/beta signaling. Immunity 2000, 13, 643–655. [Google Scholar] [CrossRef]
- Kawaguchi, M.; Oka, T.; Sugaya, M.; Suga, H.; Kimura, T.; Morimura, S.; Fujita, H.; Sato, S. IRF-2 haploinsufficiency causes enhanced imiquimod-induced psoriasis-like skin inflammation. J. Dermatol. Sci. 2018, 90, 35–45. [Google Scholar] [CrossRef]
- Takaoka, A.; Yanai, H.; Kondo, S.; Duncan, G.; Negishi, H.; Mizutani, T.; Kano, S.; Honda, K.; Ohba, Y.; Mak, T.W.; et al. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature 2005, 434, 243–249. [Google Scholar] [CrossRef]
- Tang, L.; Chen, B.; Ma, B.; Nie, S. Association between IRF5 polymorphisms and autoimmune diseases: A meta-analysis. Genet. Mol. Res. 2014, 13, 4473–4485. [Google Scholar] [CrossRef] [PubMed]
- Dideberg, V.; Kristjansdottir, G.; Milani, L.; Libioulle, C.; Sigurdsson, S.; Louis, E.; Wiman, A.C.; Vermeire, S.; Rutgeerts, P.; Belaiche, J.; et al. An insertion-deletion polymorphism in the interferon regulatory Factor 5 (IRF5) gene confers risk of inflammatory bowel diseases. Hum. Mol. Genet. 2007, 16, 3008–3016. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, A.; Inoue, N.; Ajimi, C.; Sada, K.E.; Kobayashi, S.; Yamada, H.; Furukawa, H.; Sumida, T.; Tohma, S.; Miyasaka, N.; et al. Association of IRF5 polymorphism with MPO-ANCA-positive vasculitis in a Japanese population. Genes Immun. 2013, 14, 527–529. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Rose-Zerilli, M.J.; Koppelman, G.H.; Sandling, J.K.; Holloway, J.W.; Postma, D.S.; Holgate, S.T.; Bours, V.; Syvänen, A.C.; Dideberg, V. Evidence of association between interferon regulatory factor 5 gene polymorphisms and asthma. Gene 2012, 504, 220–225. [Google Scholar] [CrossRef]
- Sánchez, F.O.; Linga Reddy, M.V.; Mallbris, L.; Sakuraba, K.; Ståhle, M.; Alarcón-Riquelme, M.E. IFN-regulatory factor 5 gene variants interact with the class I MHC locus in the Swedish psoriasis population. J. Investig. Dermatol. 2008, 128, 1704–1709. [Google Scholar] [CrossRef]
- Barnes, B.J.; Richards, J.; Mancl, M.; Hanash, S.; Beretta, L.; Pitha, P.M. Global and distinct targets of IRF-5 and IRF-7 during innate response to viral infection. J. Biol. Chem. 2004, 279, 45194–45207. [Google Scholar] [CrossRef]
- Izaguirre, A.; Barnes, B.J.; Amrute, S.; Yeow, W.S.; Megjugorac, N.; Dai, J.; Feng, D.; Chung, E.; Pitha, P.M.; Fitzgerald-Bocarsly, P. Comparative analysis of IRF and IFN-alpha expression in human plasmacytoid and monocyte-derived dendritic cells. J. Leukoc. Biol. 2003, 74, 1125–1138. [Google Scholar] [CrossRef]
- Watkins, A.A.; Yasuda, K.; Wilson, G.E.; Aprahamian, T.; Xie, Y.; Maganto-Garcia, E.; Shukla, P.; Oberlander, L.; Laskow, B.; Menn-Josephy, H.; et al. IRF5 deficiency ameliorates lupus but promotes atherosclerosis and metabolic dysfunction in a mouse model of lupus-associated atherosclerosis. J. Immunol. 2015, 194, 1467–1479. [Google Scholar] [CrossRef]
- Stein, T.; Wollschlegel, A.; Te, H.; Weiss, J.; Joshi, K.; Kinzel, B.; Billich, A.; Guntermann, C.; Lehmann, J.C.U. Interferon regulatory factor 5 and nuclear factor kappa-B exhibit cooperating but also divergent roles in the regulation of pro-inflammatory cytokines important for the development of TH1 and TH17 responses. FEBS J. 2018, 285, 3097–3113. [Google Scholar] [CrossRef]
- Yang, C.; He, D.; Yin, C.; Tan, J. Inhibition of Interferon Regulatory Factor 4 Suppresses Th1 and Th17 Cell Differentiation and Ameliorates Experimental Autoimmune Encephalomyelitis. Scand. J. Immunol. 2015, 82, 345–351. [Google Scholar]
- Savitsky, D.A.; Yanai, H.; Tamura, T.; Taniguchi, T.; Honda, K. Contribution of IRF5 in B cells to the development of murine SLE-like disease through its transcriptional control of the IgG2a locus. Proc. Natl. Acad. Sci. USA 2010, 107, 10154–10159. [Google Scholar] [CrossRef] [PubMed]
- Richez, C.; Yasuda, K.; Bonegio, R.G.; Watkins, A.A.; Aprahamian, T.; Busto, P.; Richards, R.J.; Liu, C.L.; Cheung, R.; Utz, P.J.; et al. IFN regulatory factor 5 is required for disease development in the FcgammaRIIB-/-Yaa and FcgammaRIIB-/- mouse models of systemic lupus erythematosus. J. Immunol. 2010, 184, 796–806. [Google Scholar] [CrossRef] [PubMed]
- Tada, Y.; Kondo, S.; Aoki, S.; Koarada, S.; Inoue, H.; Suematsu, R.; Ohta, A.; Mak, T.W.; Nagasawa, K. Interferon regulatory factor 5 is critical for the development of lupus in MRL/lpr mice. Arthritis Rheum. 2011, 63, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Duffau, P.; Menn-Josephy, H.; Cuda, C.M.; Dominguez, S.; Aprahamian, T.R.; Watkins, A.A.; Yasuda, K.; Monach, P.; Lafyatis, R.; Rice, L.M.; et al. Promotion of Inflammatory Arthritis by Interferon Regulatory Factor 5 in a Mouse Model. Arthritis Rheumatol. 2015, 67, 3146–3157. [Google Scholar] [CrossRef] [PubMed]
- Ueyama, A.; Yamamoto, M.; Tsujii, K.; Furue, Y.; Imura, C.; Shichijo, M.; Yasui, K. Mechanism of pathogenesis of imiquimod-induced skin inflammation in the mouse: A role for interferon-alpha in dendritic cell activation by imiquimod. J. Dermatol. 2014, 41, 135–143. [Google Scholar] [CrossRef]
- El Malki, K.; Karbach, S.H.; Huppert, J.; Zayoud, M.; Reissig, S.; Schüler, R.; Nikolaev, A.; Karram, K.; Münzel, T.; Kuhlmann, C.R.; et al. An alternative pathway of imiquimod-induced psoriasis-like skin inflammation in the absence of interleukin-17 receptor a signaling. J. Investig. Dermatol. 2013, 144, 441–451. [Google Scholar] [CrossRef]
- Xu, D.; Meyer, F.; Ehlers, E.; Blasnitz, L.; Zhang, L. Interferon regulatory factor 4 (IRF-4) targets IRF-5 to regulate Epstein-Barr virus transformation. J. Biol. Chem. 2011, 286, 18261–18267. [Google Scholar] [CrossRef]
- De, S.; Zhang, B.; Shih, T.; Singh, S.; Winkler, A.; Donnelly, R.; Barnes, B.J. B Cell-Intrinsic Role for IRF5 in TLR9/BCR-Induced Human B Cell Activation, Proliferation, and Plasmablast Differentiation. Front. Immunol. 2018, 8, 1938. [Google Scholar] [CrossRef]
- Schlitzer, A.; McGovern, N.; Teo, P.; Zelante, T.; Atarashi, K.; Low, D.; Ho, A.W.; See, P.; Shin, A.; Wasan, P.S.; et al. IRF4 transcription factor-dependent CD11b+ dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 2013, 38, 970–983. [Google Scholar] [CrossRef]
- Gelderblom, M.; Gallizioli, M.; Ludewig, P.; Thom, V.; Arunachalam, P.; Rissiek, B.; Bernreuther, C.; Glatzel, M.; Korn, T.; Arumugam, T.V.; et al. IL-23 (Interleukin-23)-Producing Conventional Dendritic Cells Control the Detrimental IL-17 (Interleukin-17) Response in Stroke. Stroke 2018, 49, 155–164. [Google Scholar] [CrossRef]
- Bajaña, S.; Roach, K.; Turner, S.; Paul, J.; Kovats, S. IRF4 promotes cutaneous dendritic cell migration to lymph nodes during homeostasis and inflammation. J. Immunol. 2012, 189, 3368–3377. [Google Scholar] [CrossRef] [PubMed]
- Ni, A.; Chen, H.; Wu, Y.; Li, W.; Chen, S.; Li, J. Expression of IRF-4 and IBP in skin lesions of patients with psoriasis vulgaris. J. Huazhong Univ. Sci. Technol. Med. Sci. 2012, 32, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.S.; Gaida, M.M.; Ogawa, Y.; Kolios, A.G.; Lasitschka, F.; Ashwell, J.D. Counter-regulation of T cell effector function by differentially activated p38. J. Exp. Med. 2014, 211, 1257–1270. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.G.; Kim, S.H.; Lee, M.G. The Origin of Skin Dendritic Cell Network and Its Role in Psoriasis. Int. J. Mol. Sci. 2017, 19, 42. [Google Scholar] [CrossRef]
| Cytokines Increased in IRF5 KO Mice | IL-17A, IL-23p19, IL-12/23p40, IL-22, IFN-γ, IL-36γ |
| Cytokines Decreased in IRF5 KO Mice | IFN-α, TNF-α, IL-6, iNOS, IL-10, IL-12p35 |
| Cytokines not Changed in IRF5 KO Mice | IL-1β, IL-36α |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakao, M.; Miyagaki, T.; Sugaya, M.; Sato, S. Exacerbated Imiquimod-Induced Psoriasis-Like Skin Inflammation in IRF5-Deficient Mice. Int. J. Mol. Sci. 2020, 21, 3681. https://doi.org/10.3390/ijms21103681
Nakao M, Miyagaki T, Sugaya M, Sato S. Exacerbated Imiquimod-Induced Psoriasis-Like Skin Inflammation in IRF5-Deficient Mice. International Journal of Molecular Sciences. 2020; 21(10):3681. https://doi.org/10.3390/ijms21103681
Chicago/Turabian StyleNakao, Momoko, Tomomitsu Miyagaki, Makoto Sugaya, and Shinichi Sato. 2020. "Exacerbated Imiquimod-Induced Psoriasis-Like Skin Inflammation in IRF5-Deficient Mice" International Journal of Molecular Sciences 21, no. 10: 3681. https://doi.org/10.3390/ijms21103681
APA StyleNakao, M., Miyagaki, T., Sugaya, M., & Sato, S. (2020). Exacerbated Imiquimod-Induced Psoriasis-Like Skin Inflammation in IRF5-Deficient Mice. International Journal of Molecular Sciences, 21(10), 3681. https://doi.org/10.3390/ijms21103681






