Immunoregulatory Property of C-Type Lectin-Like Receptors in Fibrosing Interstitial Lung Diseases
Abstract
1. Introduction
2. C-Type Lectin-Like Receptors
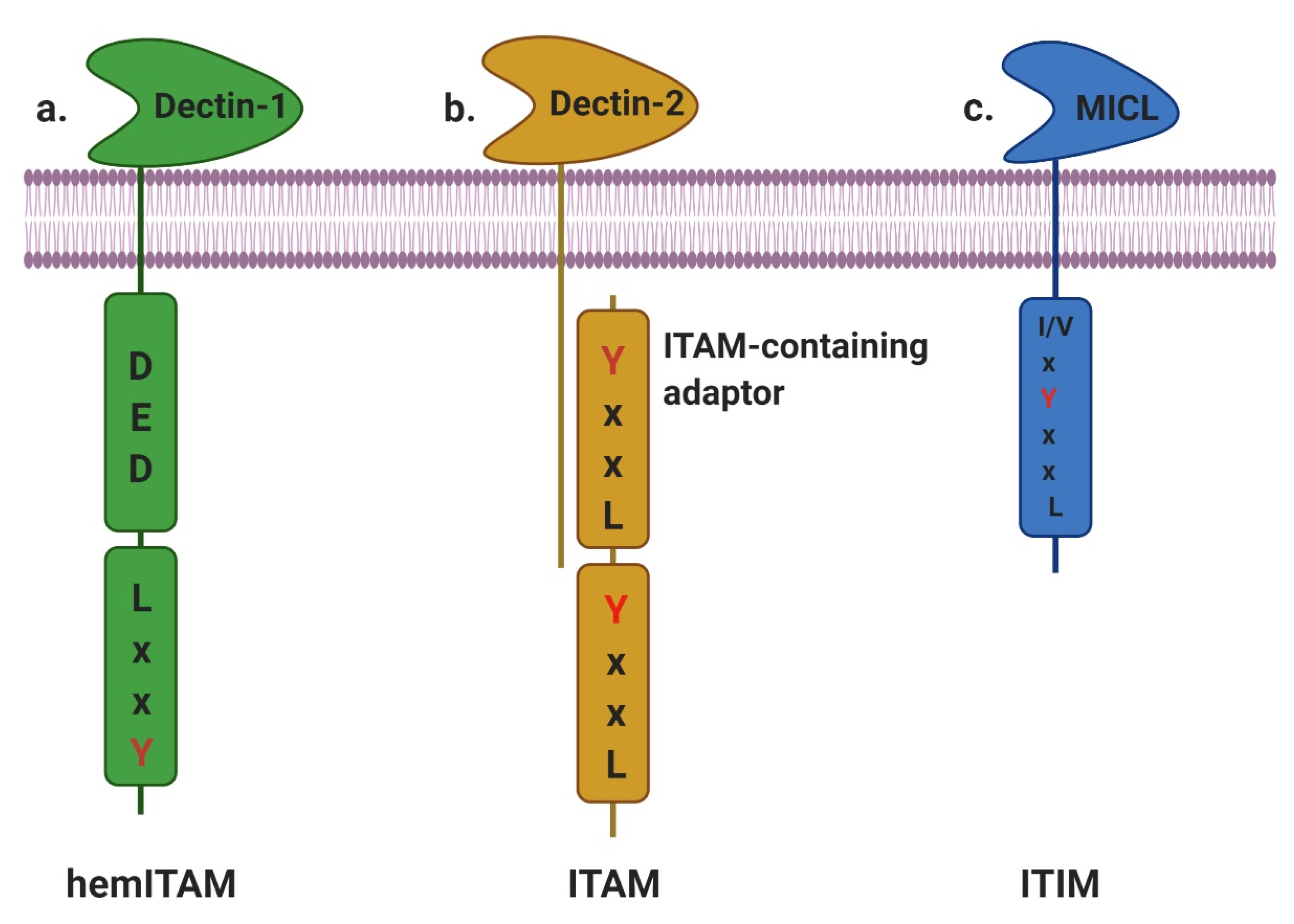
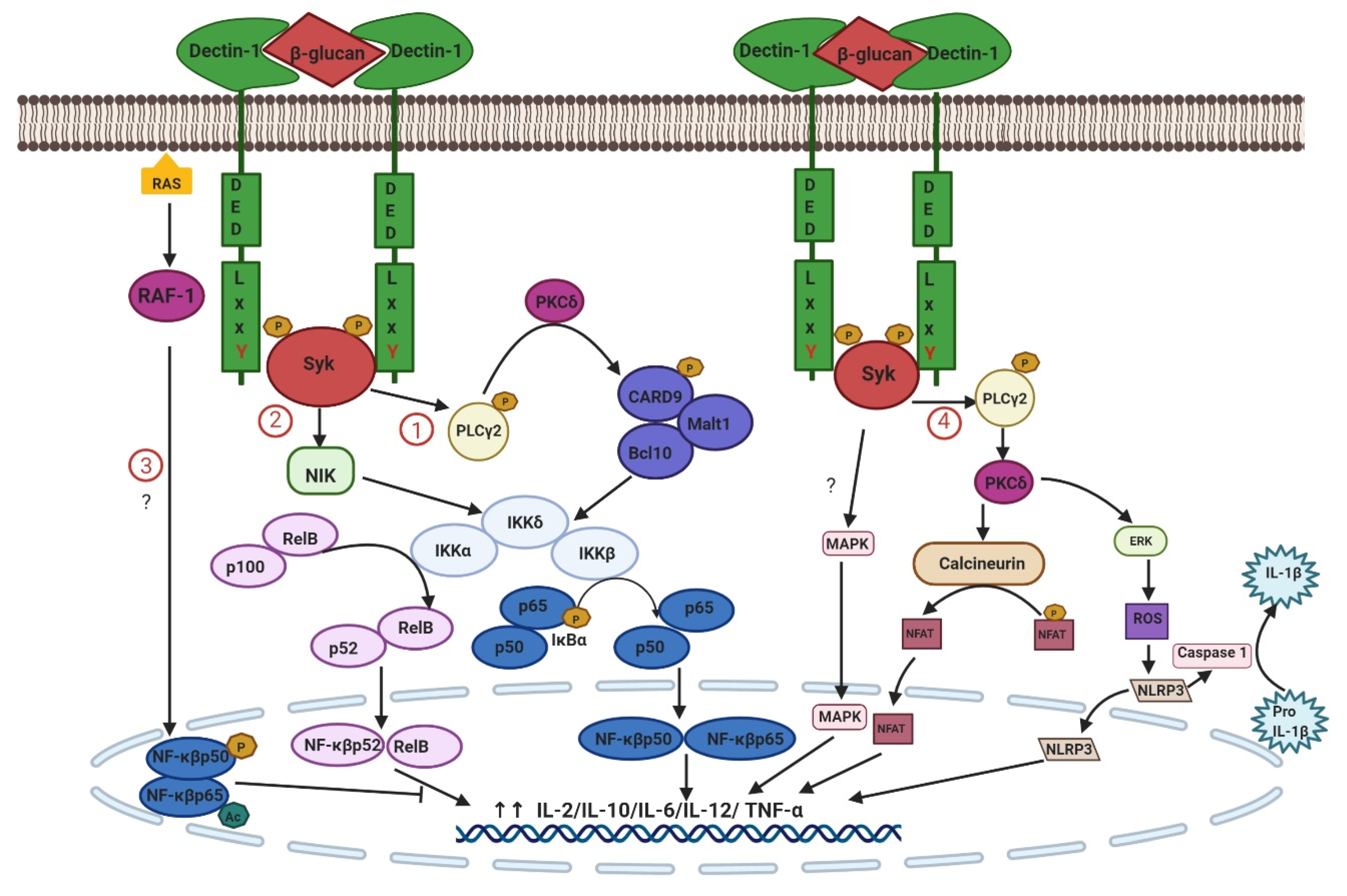
2.1. ITAM- and HemITAM-Bearing CTLRs
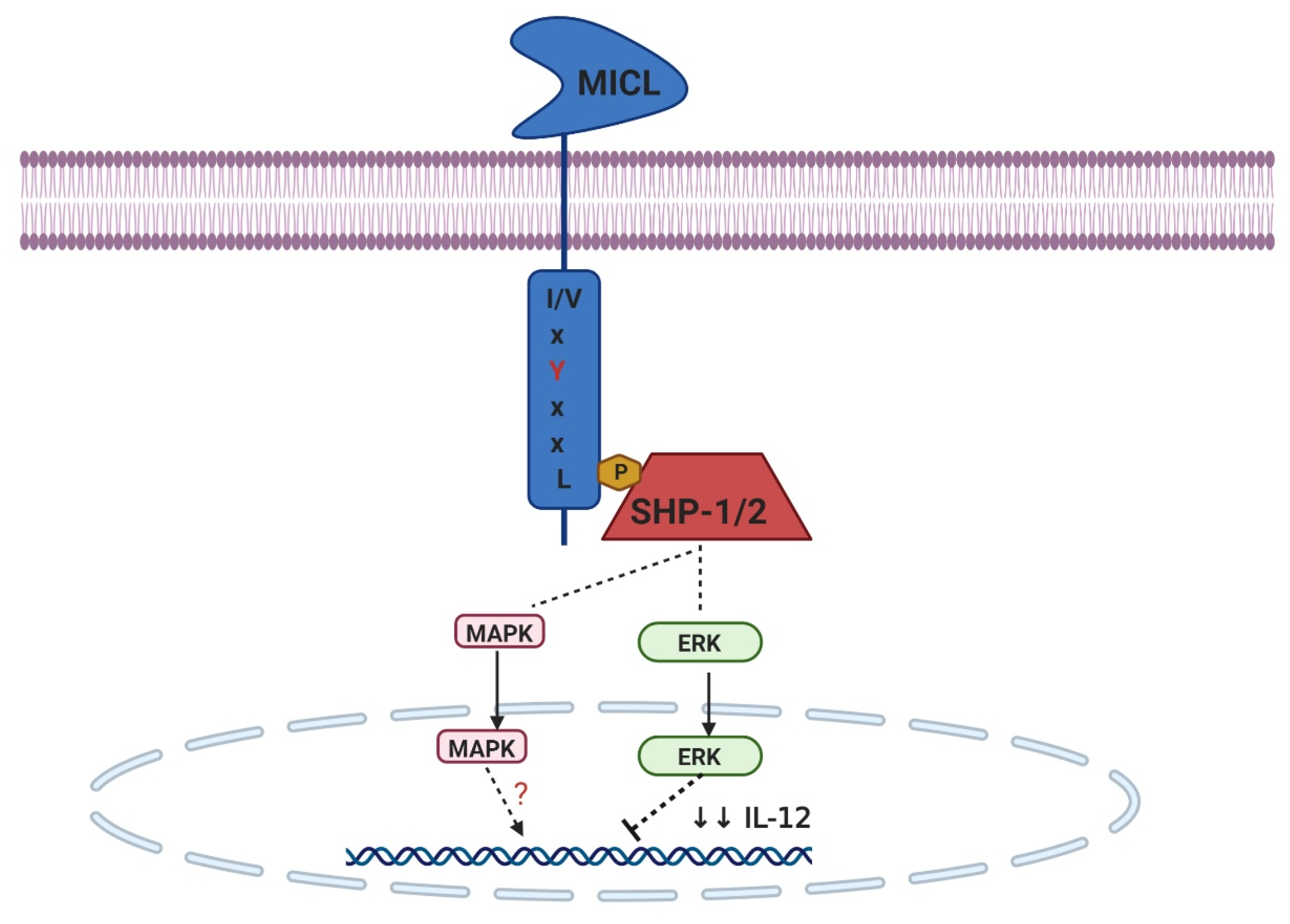
2.2. ITIM-Bearing CTLRs
3. Immune Mechanisms in Fibrosing Interstitial Lung Diseases
4. Immunoregulatory Receptors as A Novel Therapeutic Targets for ILD
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ASC | Apoptosis-associated speck-like protein |
| BCL10 | B cell lymphoma 10 |
| CARD9 | Caspase-recruitment domain family 9 |
| CBM | CARD9-Bcl10-Malt1 |
| CBP | CSK-binding Protein |
| CF | Cystic fibrosis |
| CLRs | C-type lectin receptors |
| CCL18 | CC chemokine ligand 18 |
| CRD | Carbohydrate-recognition domains |
| CSK | Carboxy (C)-terminal Src kinase |
| CTDs | Connective tissue diseases |
| CTLD | C-type lectin-like domain |
| CTLRs | C-type lectin-like receptors |
| CYT | Cytoplasmic Tail |
| DAMPs | Damage-associated molecular patterns |
| DC | Dendritic cell |
| Dectin-1 | Dendritic cell-associated C-type lectin-1 |
| EGR 2 | Early growth response 2 |
| EGR 3 | Early growth response 3 |
| ERK | Extracellular signal-regulated kinase |
| FcRγ | Fc receptor γ-chain |
| GSK3β | Glycogen synthase kinase 3β |
| HP | Hypersensitivity Pneumonitis |
| IFN | Interferon |
| IgG | Immunoglobulin G |
| IKK | Iκβ kinase |
| IPFIs | Invasive pulmonary fungal infections |
| PLCγ | Phospholipase Cγ |
| IL-10 | Interleukin-10 |
| IL-12 | Interleukin-12 |
| IL-17 | Interleukin-17 |
| IL-18 | Interleukin-18 |
| IL-1β | Interleukin-1β |
| IL-2 | Interleukin-2 |
| ILD | Interstitial lung diseases |
| IPF | Idiopathic pulmonary fibrosis |
| ITAMi | Immunoreceptor tyrosine-based activation motif inhibitor |
| ITAMs | Immunoreceptor tyrosine-based activation motifs |
| ITIMa | Immunoreceptor tyrosine-based inhibition motifs activation |
| ITIMs | Immunoreceptor tyrosine-based inhibition motifs |
| LAB | Linker for activation of B cells |
| JAK2 | Janus kinase 2 |
| JNK | JUN- N/amino terminal kinase |
| LAT | Linker for activation of T cells |
| LIME | LCK-interacting membrane protein |
| MALT1 | Mucosa-associated lymphoid tissue lymphoma translocation gene 1 |
| MAPK | Mitogen-activating protein kinase |
| MKP | MAPK-phosphatase |
| M-CSF | Macrophage-colony stimulating factor |
| NFAT | Nuclear factor of activated T cells |
| NF-κβ | Nuclear factor- κβ |
| NIK | NF-κβ-inducing kinase |
| NLRP3 | Nod-like receptor protein 3 |
| NLRs | NOD-like receptors |
| NOD | Nucleotide-binding oligomerization domain |
| NTAL | Non-T-cell activation linker |
| PAG | Protein associated with GEMs |
| PAMPs | Pathogen-associated molecular patterns |
| PH-R | Pleckstrin homology-related domains |
| PTPs | Protein tyrosine phosphatases |
| RIG-1 | Retinoic acid-inducible gene -1 |
| RLRs | RIG-1-like receptors |
| ROS | Reactive oxygen species |
| SH2 | Src homology 2 |
| SH2-C | C-terminal SH2 |
| SH2-N | N-terminal SH2 |
| SHIP-1 | SH2 domain-containing inositol phosphatase 1 |
| SHIP-2 | SH2 domain-containing inositol phosphatase 2 |
| SHP-1 | SH2-domain-containing protein tyrosine phosphatase 1 |
| SHP-2 | SH2-domain-containing protein tyrosine phosphatase 2 |
| SIT | SHP2-interacting TRAP |
| SLP65 | SH2 domain-containing leukocyte protein 65 |
| SLP76 | SH2 domain-containing leukocyte protein 76 |
| TDM | Trehalose dimycolate |
| TLRs | Toll-like receptors |
| TM | Transmembrane |
| TRAPs | Transmembrane adaptor proteins |
| TRIM | TCR-interacting molecule |
| PI3K | Phosphoinositide 3-KINASE |
| UIP | Usual interstitial pneumonia |
References
- Gasteiger, G.; D’Osualdo, A.; Schubert, D.A.; Weber, A.; Bruscia, E.M.; Hartl, D. Cellular Innate Immunity: An Old Game with New Players. J. Innate Immun. 2017, 9, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Amarante-Mendes, G.P.; Adjemian, S.; Branco, L.M.; Zanetti, L.C.; Weinlich, R.; Bortoluci, K.R. Pattern recognition receptors and the host cell death molecular machinery. Front. Immunol. 2018, 9, 2379. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef] [PubMed]
- Weis, W.I.; Taylor, M.E.; Drickamer, K. The C-type lectin superfamily in the immune system. Immunol. Rev. 1998, 163, 19–34. [Google Scholar] [CrossRef]
- Dambuza, I.M.; Brown, G.D. C-type lectins in immunity: Recent developments. Curr. Opin. Immunol. 2015, 32, 21–27. [Google Scholar] [CrossRef]
- Brown, G.D.; Willment, J.A.; Whitehead, L. C-type lectins in immunity and homeostasis. Nat. Rev. Immunol. 2018, 18, 374–389. [Google Scholar] [CrossRef]
- Seifert, L.; Deutsch, M.; Alothman, S.; Alqunaibit, D.; Werba, G.; Pansari, M.; Pergamo, M.; Ochi, A.; Torres-Hernandez, A.; Levie, E.; et al. Dectin-1 Regulates Hepatic Fibrosis and Hepatocarcinogenesis by Suppressing TLR4 Signaling Pathways. Cell Rep. 2015, 13, 1909–1921. [Google Scholar] [CrossRef]
- Lech, M.; Anders, H.J. Macrophages and fibrosis: How resident and infiltrating mononuclear phagocytes orchestrate all phases of tissue injury and repair. Biochim. Biophys. Acta-Mol. Basis Dis. 2013, 1832, 989–997. [Google Scholar] [CrossRef]
- Ricci, M. Immunoregulation in clinical diseases: An overview. Clin. Immunol. Immunopathol. 1989, 50, S3. [Google Scholar] [CrossRef]
- Fehérvari, Z.; Sakaguchi, S. CD4+ Tregs and immune control. J. Clin. Investig. 2004, 114, 1209–1217. [Google Scholar] [CrossRef]
- da Silva, F.P.; Aloulou, M.; Benhamou, M.; Monteiro, R.C. Inhibitory ITAMs: A matter of life and death. Trends Immunol. 2008, 29, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Fei, C.; Pemberton, J.G.; Lillico, D.M.E.; Zwozdesky, M.A.; Stafford, J.L. Biochemical and functional insights into the integrated regulation of innate immune cell responses by teleost leukocyte immune-type receptors. Biology 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Cortes, H.D.; Lillico, D.M.E.; Zwozdesky, M.A.; Pemberton, J.G.; O’Brien, A.; Montgomery, B.C.S.; Wiersma, L.; Chang, J.P.; Stafford, J.L. Induction of phagocytosis and intracellular signaling by an inhibitory channel catfish leukocyte immune-type receptor: Evidence for immunoregulatory receptor functional plasticity in teleosts. J. Innate Immun. 2014, 6, 435–455. [Google Scholar] [CrossRef] [PubMed]
- Hořejší, V.; Zhang, W.; Schraven, B. Transmembrane adaptor proteins: Organizers of immunoreceptor signalling. Nat. Rev. Immunol. 2004, 4, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Zelensky, A.N.; Gready, J.E. The C-type lectin-like domain superfamily. Febs J. 2005, 272, 6179–6217. [Google Scholar] [CrossRef]
- Huysamen, C.; Brown, G.D. The fungal pattern recognition receptor, Dectin-1, and the associated cluster of C-type lectin-like receptors. Fems Microbiol. Lett. 2009, 290, 121–128. [Google Scholar] [CrossRef]
- Chiffoleau, E. C-type lectin-like receptors as emerging orchestrators of sterile inflammation represent potential therapeutic targets. Front. Immunol. 2018, 9, 227. [Google Scholar]
- Plato, A.; Willment, J.A.; Brown, G.D. C-Type lectin-like receptors of the dectin-1 cluster: Ligands and signaling pathways. Int. Rev. Immunol. 2013, 32, 134–156. [Google Scholar] [CrossRef]
- Hamerman, J.A.; Lanier, L.L. Inhibition of immune responses by ITAM-bearing receptors. Sci. Stke 2006, 2006, re1. [Google Scholar] [CrossRef]
- Kane, B.A.; Bryant, K.J.; McNeil, H.P.; Tedla, N.T. Termination of immune activation: An essential component of healthy host immune responses. J. Innate Immun. 2014, 6, 727–738. [Google Scholar] [CrossRef]
- Humphrey, M.B.; Lanier, L.L.; Nakamura, M.C. Role of ITAM-containing adapter proteins and their receptors in the immune system and bone. Immunol. Rev. 2005, 208, 50–65. [Google Scholar] [CrossRef] [PubMed]
- Sancho, D.; Reis e Sousa, C. Signaling by Myeloid C-Type Lectin Receptors in Immunity and Homeostasis. Annu. Rev. Immunol. 2012, 30, 491–529. [Google Scholar] [CrossRef] [PubMed]
- Hamerman, J.A.; Ni, M.; Killebrew, J.R.; Chu, C.L.; Lowell, C.A. The expanding roles of ITAM adapters FcRγ and DAP12 in myeloid cells. Immunol. Rev. 2009, 232, 42–58. [Google Scholar] [CrossRef] [PubMed]
- Palma, A.S.; Feizi, T.; Zhang, Y.; Stoll, M.S.; Lawson, A.M.; Díaz-Rodríguez, E.; Campanero-Rhodes, M.A.; Costa, J.; Gordon, S.; Brown, G.D.; et al. Ligands for the β-glucan receptor, dectin-1, assigned using “designer” microarrays of oligosaccharide probes (neoglycolipids) generated from glucan polysaccharides. J. Biol. Chem. 2006, 281, 5771–5779. [Google Scholar] [CrossRef] [PubMed]
- Ariizumi, K.; Shen, G.L.; Shikano, S.; Xu, S.; Ritter, R.; Kumamoto, T.; Edelbaum, D.; Morita, A.; Bergstresser, P.R.; Takashima, A. Identification of a novel, dendritic cell-associated molecule, dectin-1, by subtractive cDNA cloning. J. Biol. Chem. 2000, 275, 20157–20167. [Google Scholar] [CrossRef]
- Taylor, P.R.; Brown, G.D.; Reid, D.M.; Willment, J.A.; Martinez-Pomares, L.; Gordon, S.; Wong, S.Y.C. The β-Glucan Receptor, Dectin-1, Is Predominantly Expressed on the Surface of Cells of the Monocyte/Macrophage and Neutrophil Lineages. J. Immunol. 2002, 169, 3876–3882. [Google Scholar] [CrossRef]
- Kato, Y.; Adachi, Y.; Ohno, N. Contribution of N-linked oligosaccharides to the expression and functions of β-glucan receptor, Dectin-1. Biol. Pharm. Bull. 2006, 29, 1580–1586. [Google Scholar] [CrossRef]
- Goodridge, H.S.; Reyes, C.N.; Becker, C.A.; Katsumoto, T.R.; Ma, J.; Wolf, A.J.; Bose, N.; Chan, A.S.H.; Magee, A.S.; Danielson, M.E.; et al. Activation of the innate immune receptor Dectin-1 upon formation of a - Phagocytic synapse-TM. Nature 2011, 472, 471–475. [Google Scholar]
- Bordon, Y. Phagocytosis: A synapse for snaps. Nat. Rev. Immunol. 2011, 11, 371. [Google Scholar]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. Β-Glucan Recognition By the Innate Immune System. Immunol. Rev. 2009, 230, 38–50. [Google Scholar] [CrossRef]
- Camilli, G.; Tabouret, G.; Quintin, J. The Complexity of Fungal β-Glucan in Health and Disease: Effects on the Mononuclear Phagocyte System. Front. Immunol. 2018, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tercelj, M.; Salobir, B.; Rylander, R. β-glucan in lymph nodes in sarcoidosis patients. Eur. Respir. J. 2016, 48, PA828. [Google Scholar]
- Lilly, L.M.; Gessner, M.A.; Dunaway, C.W.; Metz, A.E.; Schwiebert, L.; Weaver, C.T.; Brown, G.D.; Steele, C. The β-Glucan Receptor Dectin-1 Promotes Lung Immunopathology during Fungal Allergy via IL-22. J. Immunol. 2012, 189, 3653–3660. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Dasari, H.; Van Keulen, V.; Egan, A.; Peikert, T.; Carmona, E.M. Microbial Activation of B-cells by Pattern Recognition Receptors Contributes to the Proinflammatory and Profibrotic Milieu in Patients with Idiopathic Pulmonary Fibrosis. J. Immunol. 2019, 202, 190.20 LP-190.20. [Google Scholar]
- Willment, J.A.; Gordon, S.; Brown, G.D. Characterization of the Human β-Glucan Receptor and Its Alternatively Spliced Isoforms. J. Biol. Chem. 2001, 276, 43818–43823. [Google Scholar] [CrossRef]
- Azad, A.K.; Torrelles, J.B.; Schlesinger, L.S. Mutation in the DC-SIGN cytoplasmic triacidic cluster motif markedly attenuates receptor activity for phagocytosis and endocytosis of mannose-containing ligands by human myeloid cells. J. Leukoc. Biol. 2008, 84, 1594–1603. [Google Scholar] [CrossRef]
- Hughes, C.E.; Sinha, U.; Pandey, A.; Eble, J.A.; O’Callaghan, C.A.; Watson, S.P. Critical role for an acidic amino acid region in platelet signaling by the HemITAM (hemi-immunoreceptor tyrosine-based activation motif) containing receptor CLEC-2 (C-type lectin receptor-2). J. Biol. Chem. 2013, 288, 5127–5135. [Google Scholar] [CrossRef]
- Bezbradica, J.S.; Rosenstein, R.K.; Demarco, R.A.; Brodsky, I.; Medzhitov, R. A role for the ITAM signaling module in specifying cytokine-receptor functions. Nat. Immunol. 2014, 15, 333–342. [Google Scholar] [CrossRef]
- Rogers, N.C.; Slack, E.C.; Edwards, A.D.; Nolte, M.A.; Schulz, O.; Schweighoffer, E.; Williams, D.L.; Gordon, S.; Tybulewicz, V.L.; Brown, G.D.; et al. Syk-dependent cytokine induction by dectin-1 reveals a novel pattern recognition pathway for C type lectins. Immunity 2005, 22, 507–517. [Google Scholar] [CrossRef]
- Schorey, J.S.; Lawrence, C. The pattern recognition receptor Dectin-1: From fungi to mycobacteria. Curr. Drug Targets 2008, 9, 123–129. [Google Scholar] [CrossRef]
- Tsang, E.; Giannetti, A.M.; Shaw, D.; Dinh, M.; Tse, J.K.Y.; Gandhi, S.; Ho, A.; Wang, S.; Papp, E.; Bradshaw, J.M. Molecular mechanism of the Syk activation switch. J. Biol. Chem. 2008, 283, 32650–32659. [Google Scholar] [CrossRef] [PubMed]
- Grädler, U.; Schwarz, D.; Dresing, V.; Musil, D.; Bomke, J.; Frech, M.; Greiner, H.; Jäkel, S.; Rysiok, T.; Müller-Pompalla, D.; et al. Structural and biophysical characterization of the Syk activation switch. J. Mol. Biol. 2013, 425, 309–333. [Google Scholar] [CrossRef] [PubMed]
- Sada, K.; Takano, T.; Yanagi, S.; Yamamura, H. Structure and function of Syk protein-tyrosine kinase. J. Biochem. 2001, 130, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Mócsai, A.; Ruland, J.; Tybulewicz, V.L.J. The SYK tyrosine kinase: A crucial player in diverse biological functions. Nat. Rev. Immunol. 2010, 10, 387–402. [Google Scholar] [CrossRef]
- Gringhuis, S.I.; den Dunnen, J.; Litjens, M.; van der Vlist, M.; Wevers, B.; Bruijns, S.C.M.; Geijtenbeek, T.B.H. Dectin-1 directs T helper cell differentiation by controlling noncanonical NF-κB activation through Raf-1 and Syk. Nat. Immunol. 2009, 10, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Kingeter, L.M.; Lin, X. C-type lectin receptor-induced NF-κB activation in innate immune and inflammatory responses. Cell. Mol. Immunol. 2012, 9, 105–112. [Google Scholar] [CrossRef]
- Gross, O.; Gewies, A.; Finger, K.; Schäfer, M.; Sparwasser, T.; Peschel, C.; Förster, I.; Ruland, J. Card9 controls a non-TLR signalling pathway for innate anti-fungal immunity. Nature 2006, 442, 651–656. [Google Scholar] [CrossRef]
- Shih, V.F.S.; Tsui, R.; Caldwell, A.; Hoffmann, A. A single NFκB system for both canonical and non-canonical signaling. Cell Res. 2011, 21, 86–102. [Google Scholar] [CrossRef]
- Osorio, F.; LeibundGut-Landmann, S.; Lochner, M.; Lahl, K.; Sparwaser, T.; Eberl, G.; Reis e Sousa, C. DC activated via dectin-1 convert Treg into IL-17 producers. Eur. J. Immunol. 2008, 38, 3274–3281. [Google Scholar] [CrossRef]
- LeibundGut-Landmann, S.; Osorio, F.; Brown, G.D.; Sousa, C.R.E. Stimulation of dendritic cells via the dectin-1/Syk pathway allows priming of cytotoxic T-cell responses. Blood 2008, 112, 4971–4980. [Google Scholar] [CrossRef]
- Loures, F.V.; Araújo, E.F.; Feriotti, C.; Bazan, S.B.; Calich, V.L.G. TLR-4 cooperates with Dectin-1 and mannose receptor to expand Th17 and Tc17 cells induced by Paracoccidioides brasiliensis stimulated dendritic cells. Front. Microbiol. 2015, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Quintin, J.; Saeed, S.; Martens, J.H.A.; Giamarellos-Bourboulis, E.J.; Ifrim, D.C.; Logie, C.; Jacobs, L.; Jansen, T.; Kullberg, B.J.; Wijmenga, C.; et al. Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe 2012, 12, 223–232. [Google Scholar] [CrossRef]
- Jia, X.M.; Tang, B.; Zhu, L.L.; Liu, Y.H.; Zhao, X.Q.; Gorjestani, S.; Hsu, Y.M.S.; Yang, L.; Guan, J.H.; Xu, G.T.; et al. CARD9 mediates Dectin-1-induced ERK activation by linking Ras-GRF1 to H-Ras for antifungal immunity. J. Exp. Med. 2014, 211, 2307–2321. [Google Scholar] [CrossRef] [PubMed]
- LeibundGut-Landmann, S.; Groß, O.; Robinson, M.J.; Osorio, F.; Slack, E.C.; Tsoni, S.V.; Schweighoffer, E.; Tybulewicz, V.; Brown, G.D.; Ruland, J.; et al. Syk- and CARD9-dependent coupling of innate immunity to the induction of T helper cells that produce interleukin 17. Nat. Immunol. 2007, 8, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, H.S.; Simmons, R.M.; Underhill, D.M. Dectin-1 Stimulation by Candida albicans Yeast or Zymosan Triggers NFAT Activation in Macrophages and Dendritic Cells. J. Immunol. 2007, 178, 3107–3115. [Google Scholar] [CrossRef] [PubMed]
- Daëron, M.; Jaeger, S.; Du Pasquier, L.; Vivier, E. Immunoreceptor tyrosine-based inhibition motifs: A quest in the past and future. Immunol. Rev. 2008, 224, 11–43. [Google Scholar] [CrossRef] [PubMed]
- Billadeau, D.D.; Leibson, P.J. ITAMs versus ITIMs: Striking a balance during cell regulation. J. Clin. Invest. 2002, 109, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Ravetch, J.V.; Lanier, L.L. Immune inhibitory receptors. Science 2000, 290, 84–89. [Google Scholar] [CrossRef]
- Waterman, P.M.; Cambier, J.C. The conundrum of inhibitory signaling by ITAM-containing immunoreceptors: Potential molecular mechanisms. Febs Lett. 2010, 584, 4878–4882. [Google Scholar] [CrossRef]
- Coxon, C.H.; Geer, M.J.; Senis, Y.A. ITIM receptors: More than just inhibitors of platelet activation. Blood 2017, 129, 3407–3418. [Google Scholar] [CrossRef]
- Pauls, S.D.; Marshall, A.J. Regulation of immune cell signaling by SHIP1: A phosphatase, scaffold protein, and potential therapeutic target. Eur. J. Immunol. 2017, 47, 932–945. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, L.; Song, X.; Mo, Y.; Komma, C.; Bellamy, H.D.; Zhao, Z.J.; Zhou, G.W. Crystal structure of human protein tyrosine phosphatase SHP-1 in the open conformation. J. Cell. Biochem. 2011, 112, 2062–2071. [Google Scholar] [CrossRef] [PubMed]
- Pauls, S.D.; Ray, A.; Hou, S.; Vaughan, A.T.; Cragg, M.S.; Marshall, A.J. FcγRIIB-Independent Mechanisms Controlling Membrane Localization of the Inhibitory Phosphatase SHIP in Human B Cells. J. Immunol. 2016, 197, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Bounab, Y.; Getahun, A.; Cambier, J.C.; Daëron, M. Phosphatase regulation of immunoreceptor signaling in T cells, B cells and mast cells. Curr. Opin. Immunol. 2013, 25, 313–320. [Google Scholar] [CrossRef]
- Chong, Z.Z.; Maiese, K. The Src homology 2 domain tyrosine phosphatases SHP-1 and SHP-2: Diversified control of cell growth, inflammation, and injury. Histol. Histopathol. 2007, 22, 1251–1267. [Google Scholar]
- Yi, T.; Mui, A.L.; Krystal, G.; Ihle, J.N. Hematopoietic cell phosphatase associates with the interleukin-3 (IL-3) receptor beta chain and down-regulates IL-3-induced tyrosine phosphorylation and mitogenesis. Mol. Cell. Biol. 1993, 13, 7577–7586. [Google Scholar] [CrossRef]
- Pani, G.; Kozlowski, M.; Cambier, J.C.; Mills, G.B.; Siminovitch, K.A. Identification of the tyrosine phosphatase PTPlC as a B cell antigen receptor-associated protein involved in the regulation of B cell signaling. J. Exp. Med. 1995, 181, 2077–2084. [Google Scholar] [CrossRef]
- Marshall, A.S.J.; Willmen, J.A.; Lin, H.H.; Williams, D.L.; Gordon, S.; Brown, G.D. Identification and Characterization of a Novel Human Myeloid Inhibitory C-type Lectin-like Receptor (MICL) That Is Predominantly Expressed on Granulocytes and Monocytes. J. Biol. Chem. 2004, 279, 14792–14802. [Google Scholar] [CrossRef]
- Marshall, A.S.J.; Willment, J.A.; Pyz, E.; Dennehy, K.M.; Reid, D.M.; Dri, P.; Gordon, S.; Wong, S.Y.C.; Brown, G.D. Human MICL (CLEC12A) is differentially glycosylated and is down-regulated following cellular activation. Eur. J. Immunol. 2006, 36, 2159–2169. [Google Scholar] [CrossRef]
- Foster, A.J.; Bird, J.H.; Timmer, M.S.M.; Stocker, B.L. The ligands of C-type lectins. In C-Type Lectin Receptors in Immunity; Springer: Tokyo, Japan, 2016; pp. 191–215. ISBN 9784431560159. [Google Scholar]
- Pyz, E.; Huysamen, C.; Marshall, A.S.J.; Gordon, S.; Taylor, P.R.; Brown, G.D. Characterisation of murine MICL (CLEC12A) and evidence for an endogenous ligand. Eur. J. Immunol. 2008, 38, 1157–1163. [Google Scholar] [CrossRef]
- Chen, C.H.; Floyd, H.; Olson, N.E.; Magaletti, D.; Li, C.; Draves, K.; Clark, E.A. Dendritic-cell-associated C-type lectin 2 (DCAL-2) alters dendritic-cell maturation and cytokine production. Blood 2006, 107, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Geijtenbeek, T.B.H.; Gringhuis, S.I. Signalling through C-type lectin receptors: Shaping immune responses. Nat. Rev. Immunol. 2009, 9, 465–479. [Google Scholar] [CrossRef] [PubMed]
- Maher, T.M.; Wuyts, W. Management of Fibrosing Interstitial Lung Diseases. Adv. Ther 2019, 36, 1518–1531. [Google Scholar] [CrossRef] [PubMed]
- Olson, A.L.; Swigris, J.J. Idiopathic Pulmonary Fibrosis: Diagnosis and Epidemiology. Clin. Chest Med. 2012, 33, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Cottin, V.; Wollin, L.; Fischer, A.; Quaresma, M.; Stowasser, S.; Harari, S. Fibrosing interstitial lung diseases: Knowns and unknowns. Eur. Respir. Rev. 2019, 28, 180100. [Google Scholar] [CrossRef]
- Plantier, L.; Cazes, A.; Dinh-Xuan, A.T.; Bancal, C.; Marchand-Adam, S.; Crestani, B. Physiology of the lung in idiopathic pulmonary fibrosis. Eur. Respir. Rev. 2018, 27, 170062. [Google Scholar] [CrossRef]
- Hügle, T. Immunology of fibrotic lung disease: Managing infections whilst preventing autoimmunity? J. Inflamm. Res. 2011, 4, 21–27. [Google Scholar]
- Kolahian, S.; Fernandez, I.E.; Eickelberg, O.; Hartl, D. Immune mechanisms in pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2016, 55, 309–322. [Google Scholar] [CrossRef]
- Desai, O.; Winkler, J.; Minasyan, M.; Herzog, E.L. The Role of Immune and Inflammatory Cells in Idiopathic Pulmonary Fibrosis. Front. Med. 2018, 5, 43. [Google Scholar] [CrossRef]
- Sivokozov, I.; Lovacheva, O.; Shmelev, E.; Larionova, E. Fungal colonization in interstitial lung diseases. Eur. Respir. J. 2011, 38, 3749. [Google Scholar]
- Ge, H.F.; Liu, X.Q.; Zhu, Y.Q.; Chen, H.Q.; Chen, G.Z. Invasive pulmonary fungal infections in patients with connective tissue disease: A retrospective study from northern China. Braz. J. Med. Biol. Res. 2016, 49. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.L.; Metz, A.E.; Horn, D.; Schoeb, T.R.; Hewitt, M.M.; Schwiebert, L.M.; Faro-Trindade, I.; Brown, G.D.; Steele, C. Requisite Role for the Dectin-1 β-Glucan Receptor in Pulmonary Defense against Aspergillus fumigatus. J. Immunol. 2009, 182, 4938–4946. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.L.; Steele, C. Innate Receptors and Cellular Defense against Pulmonary Infections. J. Immunol. 2014, 193, 3842–3850. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Collard, H.R.; Egan, J.J.; Martinez, F.J.; Behr, J.; Brown, K.K.; Colby, T.V.; Cordier, J.F.; Flaherty, K.R.; Lasky, J.A.; et al. An Official ATS/ERS/JRS/ALAT Statement: Idiopathic pulmonary fibrosis: Evidence-based guidelines for diagnosis and management. Am. J. Respir. Crit. Care Med. 2011, 183, 788–824. [Google Scholar] [CrossRef]
- Bouros, D.; Antoniou, K.M. Current and future therapeutic approaches in idiopathic pulmonary fibrosis. Eur. Respir. J. 2005, 26, 693–702. [Google Scholar] [CrossRef]
- Ramani, K.; Biswas, P.S. Interleukin-17: Friend or foe in organ fibrosis. Cytokine 2019, 120, 282–288. [Google Scholar] [CrossRef]
- Weng, D.; Chen, X.Q.; Li, H.P.; Ge, B.X. IL-17 And MIG Contribute To Fungi-Induced Acute Exacerbation Of Idiopathic Pulmonary Fibrosis Via A Dectin-1 Dependent Mechanism. In Proceedings of the A61. LUNG FIBROSIS: ANIMAL MODELS II; American Thoracic Society: New York, NY, USA, 2014; p. A1983. [Google Scholar]
- Hasan, S.A.; Eksteen, B.; Reid, D.; Paine, H.V.; Alansary, A.; Johannson, K.; Gwozd, C.; Goring, K.A.R.; Vo, T.; Proud, D.; et al. Role of IL-17A and neutrophils in fibrosis in experimental hypersensitivity pneumonitis. J. Allergy Clin. Immunol. 2013, 131, 1663–1673.e5. [Google Scholar] [CrossRef]
- Higashino-Kameda, M.; Yabe-Wada, T.; Matsuba, S.; Takeda, K.; Anzawa, K.; Mochizuki, T.; Makimura, K.; Saijo, S.; Iwakura, Y.; Toga, H.; et al. A critical role of Dectin-1 in hypersensitivity pneumonitis. Inflamm. Res. 2016, 65, 235–244. [Google Scholar] [CrossRef]
- Ralhan, A.; Frey, N.; Schroth, C.; Singh, A.; Hector, A.; Hartl, D. Regulation of dectin-1 in cystic fibrosis bronchial epithelial cells during host pathogen interactions. Eur. Respir. J. 2016, 48, PA4858. [Google Scholar]
- Griffiths, J.S.; Thompson, A.; Stott, M.; Benny, A.; Lewis, N.A.; Taylor, P.R.; Forton, J.; Herrick, S.; Orr, S.J.; McGreal, E.P. Differential susceptibility of Dectin-1 isoforms to functional inactivation by neutrophil and fungal proteases. Faseb J. 2018, 32, 3385–3397. [Google Scholar] [CrossRef]
- De Rose, V.; Molloy, K.; Gohy, S.; Pilette, C.; Greene, C.M. Airway epithelium dysfunction in cystic fibrosis and COPD. Mediat. Inflamm. 2018, 2018, 1309746. [Google Scholar] [CrossRef] [PubMed]
- Ramaprakash, H.; Ito, T.; Standiford, T.J.; Kunkel, S.L.; Hogaboam, C.M. Toll-like receptor 9 modulates immune responses to Aspergillus fumigatus conidia in immunodeficient and allergic mice. Infect. Immun. 2009, 77, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Prasse, A.; Pechkovsky, D.V.; Toews, G.B.; Schäfer, M.; Eggeling, S.; Ludwig, C.; Germann, M.; Kollert, F.; Zissel, G.; Müller-Quernheim, J. CCL18 as an indicator of pulmonary fibrotic activity in idiopathic interstitial pneumonias and systemic sclerosis. Arthritis Rheum. 2007, 56, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Niu, R.; Li, X.; Zhang, Y.; Wang, H.; Wang, Y.; Zhang, Y.; Wang, W. (No Title). Int. J. Clin. Exp. Pathol. 2016, 9, 8922–8932. [Google Scholar]
- Hoffmann-Vold, A.M.; Tennøe, A.H.; Garen, T.; Midtvedt, Ø.; Abraityte, A.; Aaløkken, T.M.; Lund, M.B.; Brunborg, C.; Aukrust, P.; Ueland, T.; et al. High Level of Chemokine CCL18 Is Associated With Pulmonary Function Deterioration, Lung Fibrosis Progression, and Reduced Survival in Systemic Sclerosis. Chest 2016, 150, 299–306. [Google Scholar] [CrossRef]
- Wermuth, P.J.; Jimenez, S.A. The significance of macrophage polarization subtypes for animal models of tissue fibrosis and human fibrotic diseases. Clin. Transl. Med. 2015, 4, 1–19. [Google Scholar] [CrossRef]
- de Gaetano, M.; Crean, D.; Barry, M.; Belton, O. M1- and M2-Type Macrophage Responses Are Predictive of Adverse Outcomes in Human Atherosclerosis. Front. Immunol. 2016, 7, 275. [Google Scholar] [CrossRef]
- El Taweel, M.; Walker, H.; Mekhael, O.; Vierhout, M.; Padwal, M.; Naiel, S.; Patel, H.; Parthasarathy, P.; Imani, J.; Ayaub, E.; et al. Dectin-1 Is a Marker of Alternatively Activated Macrophages and a Therapeutic Target in Interstitial Fibrotic Lung Diseases. In Proceedings of the American Thoracic Society International Conference Meetings Abstracts American Thoracic Society International Conference Meetings Abstracts; American Thoracic Society: New York, NY, USA, 2019; p. A5403. [Google Scholar]
- Kaminski, N.; Belperio, J.A.; Bitterman, P.B.; Chen, L.; Chensue, S.W.; Choi, A.M.K.; Dacic, S.; Dauber, J.H.; Bois, R.M.; Enghild, J.J.; et al. Idiopathic Pulmonary Fibrosis. Proceedings of the 1st Annual Pittsburgh International Lung Conference. October 2002. Am. J. Respir. Cell Mol. Biol. 2003, 29, S1–S105. [Google Scholar] [CrossRef]
- Grimminger, F.; Günther, A.; Vancheri, C. The role of tyrosine kinases in the pathogenesis of idiopathic pulmonary fibrosis. Eur. Respir. J. 2015, 45, 1426–1433. [Google Scholar] [CrossRef]
- Pamuk, O.N.; Can, G.; Ayvaz, S.; Karaca, T.; Pamuk, G.E.; Demirtas, S.; Tsokos, G.C. Spleen tyrosine kinase (Syk) inhibitor fostamatinib limits tissue damage and fibrosis in a bleomycin-induced scleroderma mouse model. Clin. Exp. Rheumatol. 2015, 33, 15–22. [Google Scholar]
- Poeck, H.; Ruland, J. SYK kinase signaling and the NLRP3 inflammasome in antifungal immunity. J. Mol. Med. 2010, 88, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Huang, D.-Y.; Wang, J.-S.; Lin, Y.-L.; Hsieh, S.-L.; Huang, K.-C.; Lin, W.-W. Syk is involved in NLRP3 inflammasome-mediated caspase-1 activation through adaptor ASC phosphorylation and enhanced oligomerization. J. Leukoc. Biol. 2015, 97, 825–835. [Google Scholar] [CrossRef]
- De Nardo, D.; De Nardo, C.M.; Latz, E. New insights into mechanisms controlling the NLRP3 inflammasome and its role in lung disease. Am. J. Pathol. 2014, 184, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Artlett, C.M. The Role of the NLRP3 Inflammasome in Fibrosis. Open Rheumatol. J. 2012, 6, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.C.; Wong, M.H. Caught in the Akt: Regulation of WNT signaling in the intestine. Gastroenterology 2010, 139, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Langhammer, T.-S.; Roolf, C.; Krohn, S.; Kretzschmar, C.; Huebner, R.; Rolfs, A.; Freund, M.; Junghanss, C. PI3K/Akt Signaling Interacts With Wnt/β-Catenin Signaling But Does Not Induce An Accumulation Of β-Catenin In The Nucleus Of Acute Lymphoblastic Leukemia Cell Lines. Blood 2013, 122, 4886. [Google Scholar] [CrossRef]
- Chen, Z.; Oh, S.Y.; Fang, P.; Li, Q.; Zhang, F.; Tang, H.; Homer, R.; Zhang, X.; Kaminski, N.; Zheng, T.; et al. Tyrosine Phosphatase SHP-1 Regulates Pulmonary Fibrosis Through Inhibition of ?-Catenin Activation in Lung Epithelial Cells. In Proceedings of the B107. MECHANISTIC ADVANCES IN PULMONARY FIBROSIS; American Thoracic Society: New York, NY, USA, 2018; p. A4364. [Google Scholar]
- Park, J.; Jeong, J.; Kim, Y.; Oh, S.; Oh, M.; Wu, F.; Zheng, T.; Zhu, Z. Increased Susceptibility to Bleomycin-Induced Pulmonary Injury and Fibrosis in Mice Deficient in Protein Tyrosine Phosphatase SHP-1. J. Allergy Clin. Immunol. 2009, 123, 729. [Google Scholar] [CrossRef]
- Lam, A.P.; Gottardi, C.J. β-catenin signaling: A novel mediator of fibrosis and potential therapeutic target. Curr. Opin. Rheumatol. 2011, 23, 562–567. [Google Scholar] [CrossRef]
- Piersma, B.; Bank, R.A.; Boersema, M. Signaling in fibrosis: TGF-β, WNT, and YAP/TAZ converge. Front. Med. 2015, 2, 59. [Google Scholar] [CrossRef]
- Vallée, A.; Lecarpentier, Y.; Guillevin, R.; Vallée, J.N. Interactions between TGF-β1, canonical WNT/β-catenin pathway and PPAR γ in radiation-induced fibrosis. Oncotarget 2017, 8, 90579–90604. [Google Scholar] [CrossRef]
- Wynn, T.A. Integrating mechanisms of pulmonary fibrosis. J. Exp. Med. 2011, 208, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Wang, C.; Chen, X.; Hou, J.; Xiang, Z.; Shen, Y.; Han, X. Inhibition of Wnt/β-catenin signaling suppresses myofibroblast differentiation of lung resident mesenchymal stem cells and pulmonary fibrosis. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Kim, S.H.; Seo, J.Y.; Chung, H.; Kwak, H.J.; Lee, S.K.; Yoon, H.J.; Shin, D.H.; Park, S.S.; Sohn, J.W. Blockade of the Wnt/β-catenin pathway attenuates bleomycin-induced pulmonary fibrosis. Tohoku J. Exp. Med. 2011, 223, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Tzouvelekis, A.; Karampitsakos, T.; Min, K.; Xylourgidis, N.; Yu, G.; Herazo-Maya, J.; Bizenhofer, L.; Bennett, A.; Kaminski, N. Role of Mitogen activated-kinase (MAPK)-phosphatase (MKP)-5 in pulmonary fibrosis. Eur. Respir. J. 2018, 52, LSC-1111. [Google Scholar]
- Madala, S.K.; Schmidt, S.; Davidson, C.; Ikegami, M.; Wert, S.; Hardie, W.D. MEK-ERK pathway modulation ameliorates pulmonary fibrosis associated with epidermal growth factor receptor activation. Am. J. Respir. Cell Mol. Biol. 2012, 46, 380–388. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Effendi, W.I.; Nagano, T.; Hasan, H.; Yudhawati, R. Immunoregulatory Property of C-Type Lectin-Like Receptors in Fibrosing Interstitial Lung Diseases. Int. J. Mol. Sci. 2020, 21, 3665. https://doi.org/10.3390/ijms21103665
Effendi WI, Nagano T, Hasan H, Yudhawati R. Immunoregulatory Property of C-Type Lectin-Like Receptors in Fibrosing Interstitial Lung Diseases. International Journal of Molecular Sciences. 2020; 21(10):3665. https://doi.org/10.3390/ijms21103665
Chicago/Turabian StyleEffendi, Wiwin Is, Tatsuya Nagano, Helmia Hasan, and Resti Yudhawati. 2020. "Immunoregulatory Property of C-Type Lectin-Like Receptors in Fibrosing Interstitial Lung Diseases" International Journal of Molecular Sciences 21, no. 10: 3665. https://doi.org/10.3390/ijms21103665
APA StyleEffendi, W. I., Nagano, T., Hasan, H., & Yudhawati, R. (2020). Immunoregulatory Property of C-Type Lectin-Like Receptors in Fibrosing Interstitial Lung Diseases. International Journal of Molecular Sciences, 21(10), 3665. https://doi.org/10.3390/ijms21103665




