Identification of Inhibitors to Trypanosoma cruzi Sirtuins Based on Compounds Developed to Human Enzymes
Abstract
1. Introduction
2. Results
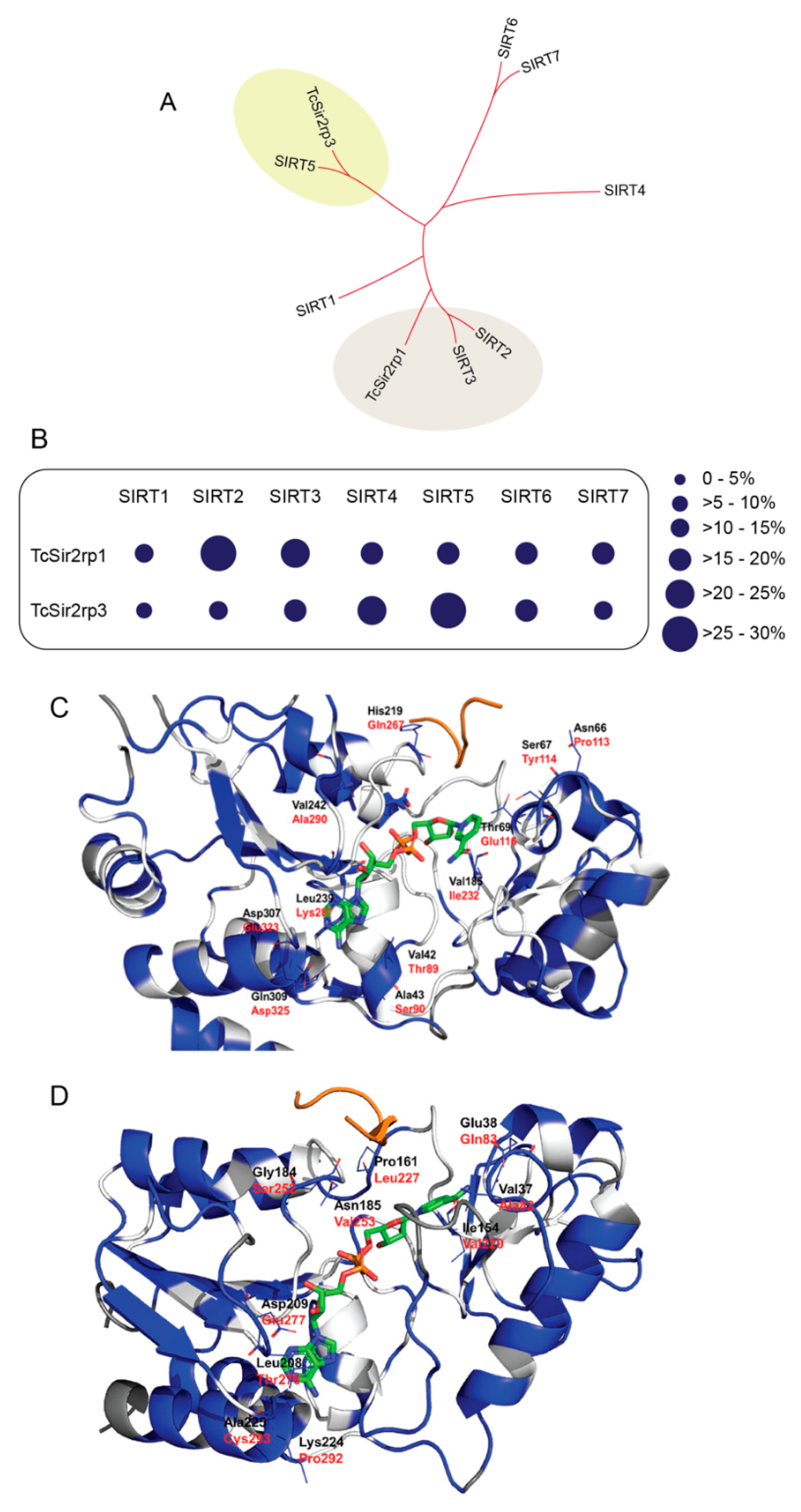
2.1. T. cruzi Has Two Distinct Sirtuins
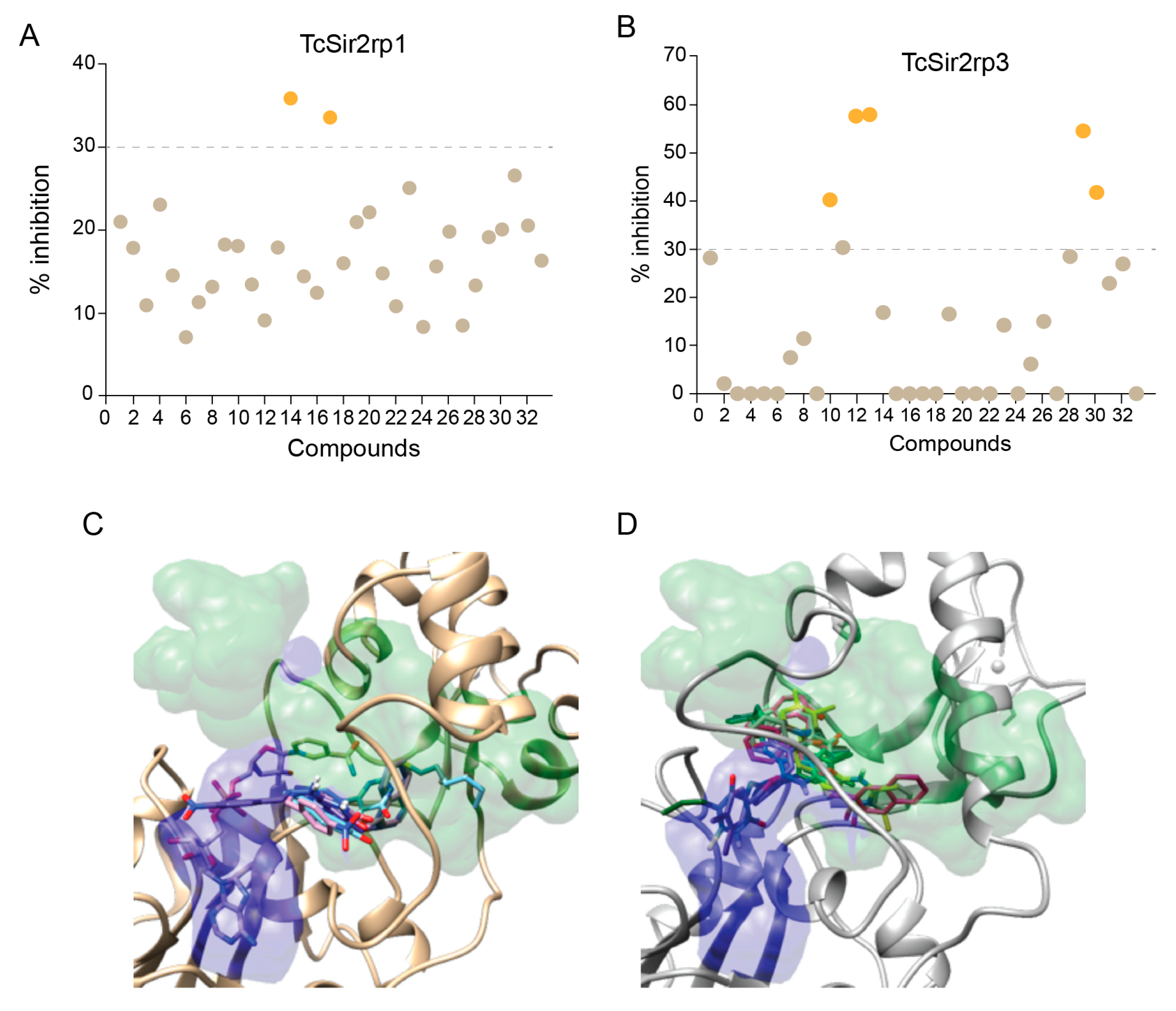
2.2. Effect of Inhibitors on TcSir2rp1 and rp3 Deacetylase Activity
2.3. Binding Mode of SIRTi with Effect on T. cruzi Sirtuins
2.4. Effect of SIRTi on T. cruzi Infection In Vitro
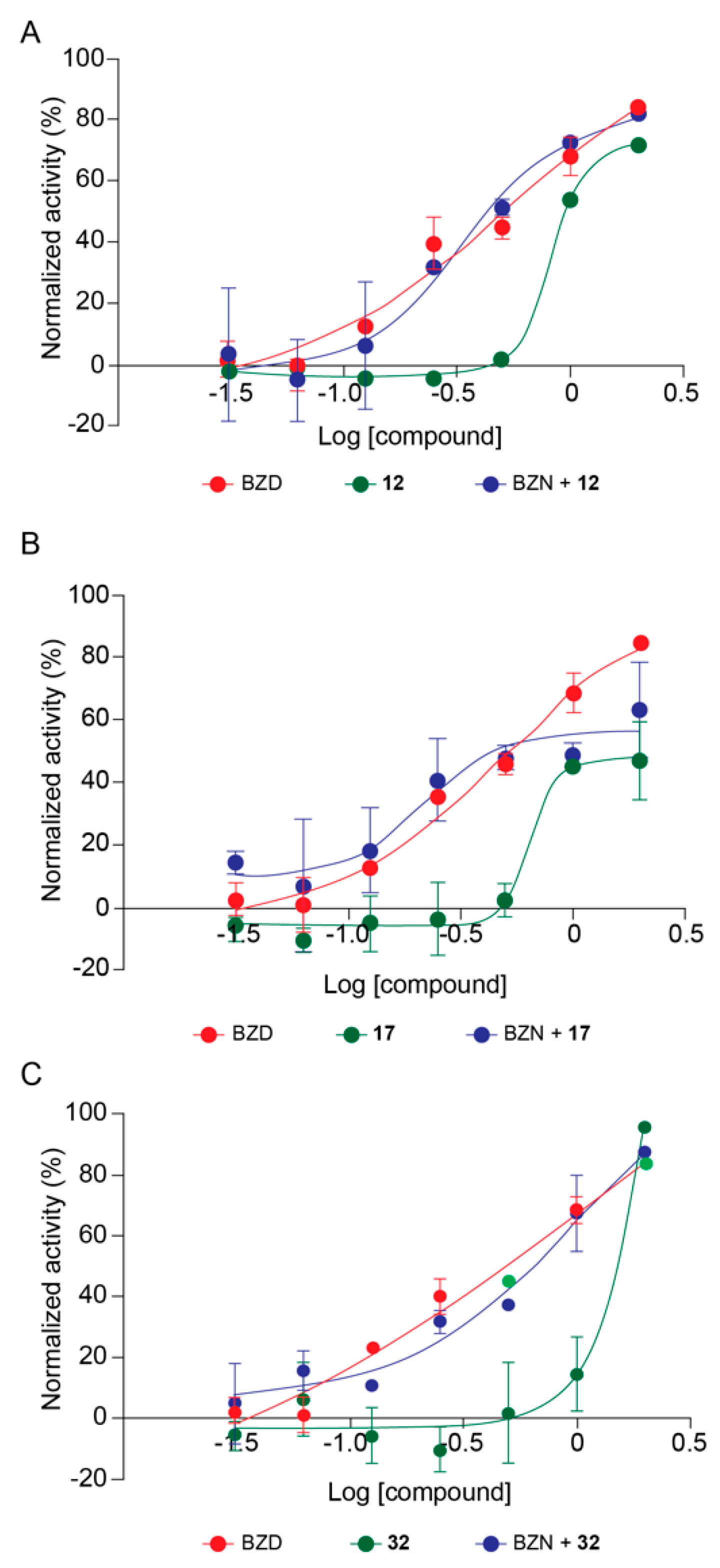
2.5. Combinatory Trypanocidal Effect of SIRTi and Benznidazole
3. Discussion
4. Materials and Methods
4.1. TcSir2rp1 and TcSir2rp3 Heterologous Expression and Purification
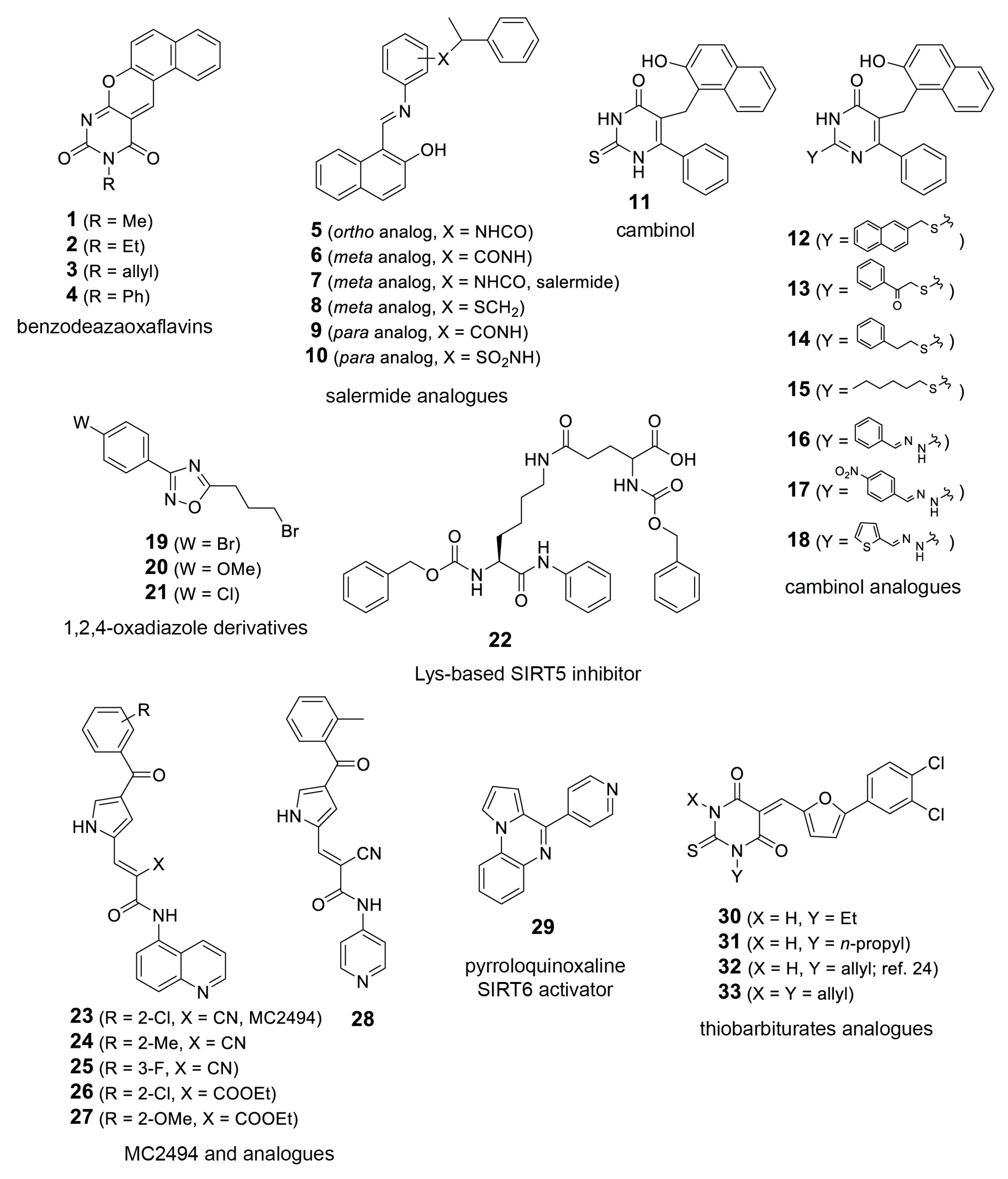
4.2. SIRTi Library
4.3. Deacetylase Activity Assay
4.4. Molecular Modeling
4.4.1. Homology Modeling of T. cruzi Sirtuins
4.4.2. Conformation Preparation of T cruzi Sirtuins Complexes
4.4.3. Molecular Docking Simulations
Docking Assessment
Molecular Docking of the SIRT Inhibitors
4.5. Parasites
4.6. High Content Assays (HCA)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernatchez, J.A.; Chen, E.; Hull, M.V.; McNamara, C.W.; McKerrow, J.H.; Siqueira-Neto, J.L. High-throughput screening of the ReFRAME library identifies potential drug repurposing candidates for Trypanosoma cruzi. Trends Parasitol. 2019. [Google Scholar] [CrossRef]
- Zulantay, I.; Apt, W.; Ramos, D.; Godoy, L.; Valencia, C.; Molina, M.; Sepulveda, E.; Thieme, P.; Martinez, G.; Corral, G. The Epidemiological Relevance of Family Study in Chagas Disease. PLoS Negl. Trop. Dis. 2013, 7, e1959. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, J.; Davies, C.; Simonazzi, A.; Real, J.P.; Palma, S. Current drug therapy and pharmaceutical challenges for Chagas disease. Acta Trop. 2016, 156, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Coura, J.R. Current prospects of specific treatment of Chagas’ disease. Boletín chileno de parasitología 1996, 51, 69–75. [Google Scholar]
- Moretti, N.S.; Schenkman, S. Chromatin modifications in trypanosomes due to stress. Cell. Microbiol. 2013, 15, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Moretti, N.S.; Cestari, I.; Anupama, A.; Stuart, K.; Schenkman, S. Comparative Proteomic Analysis of Lysine Acetylation in Trypanosomes. J. Proteome Res. 2017, 17, 374–385. [Google Scholar] [CrossRef]
- Hyndman, K.A.; Knepper, M.A. Dynamic regulation of lysine acetylation: The balance between acetyltransferase and deacetylase activities. Am. J. Physiol. Physiol. 2017, 313, F842–F846. [Google Scholar] [CrossRef]
- Brachmann, C.B.; Pillus, L.; Sherman, J.M.; Devine, S.E.; Cameron, E.E.; Boeke, J.D. The SIR2 gene family, conserved from bacteria to humans, functions in silencing, cell cycle progression, and chromosome stability. Genes Dev. 1995, 9, 2888–2902. [Google Scholar] [CrossRef]
- Carafa, V.; Rotili, D.; Forgione, M.; Cuomo, F.; Serretiello, E.; Hailu, G.S.; Jarho, E.; Lahtela-Kakkonen, M.; Mai, A.; Altucci, L. Sirtuin functions and modulation: From chemistry to the clinic. Clin. Epigenetics 2016, 8, 61. [Google Scholar] [CrossRef]
- Religa, A.A.; Waters, A.P. Sirtuins of parasitic protozoa: In search of function(s). Mol. Biochem. Parasitol. 2012, 185, 71–88. [Google Scholar] [CrossRef]
- Alsford, S.; Kawahara, T.; Isamah, C.; Horn, D. A sirtuin in the African trypanosome is involved in both DNA repair and telomeric gene silencing but is not required for antigenic variation. Mol. Microbiol. 2007, 63, 724–736. [Google Scholar] [CrossRef] [PubMed]
- Moretti, N.S.; Augusto, L.D.S.; Clemente, T.M.; Antunes, R.P.P.; Yoshida, N.; Torrecilhas, A.C.; Cano, M.I.N.; Schenkman, S. Characterization of Trypanosoma cruzi Sirtuins as Possible Drug Targets for Chagas Disease. Antimicrob. Agents Chemother. 2015, 59, 4669–4679. [Google Scholar] [CrossRef] [PubMed]
- Ritagliati, C.; Alonso, V.L.; Manarin, R.; Cribb, P.; Serra, E.C. Overexpression of Cytoplasmic TcSIR2RP1 and Mitochondrial TcSIR2RP3 Impacts on Trypanosoma cruzi Growth and Cell Invasion. PLOS Neglected Trop. Dis. 2015, 9, e0003725. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Rosa, B.A.; Nare, B.; Powell, K.; Valente, S.; Rotili, D.; Mai, A.; Marshall, G.; Mitreva, M. Targeting Lysine Deacetylases (KDACs) in Parasites. PLOS Neglected Trop. Dis. 2015, 9, e0004026. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.; Silva, C.V.; Bastos, T.M.; Guimarães, E.T.; Figueira, C.P.; Smirlis, D.; Azevedo, W.F., Jr. Anti-Trypanosoma cruzi activity of nicotinamide. Acta Trop. 2012, 122, 224–229. [Google Scholar] [CrossRef]
- Zheng, W. Sirtuins as emerging anti-parasitic targets. Eur. J. Med. Chem. 2013, 59, 132–140. [Google Scholar] [CrossRef]
- Wilkinson, S.R.; Taylor, M.C.; Horn, D.; Kelly, J.M.; Cheeseman, I.H. A mechanism for cross-resistance to nifurtimox and benznidazole in trypanosomes. Proc. Natl. Acad. Sci. USA 2008, 105, 5022–5027. [Google Scholar] [CrossRef]
- Tao, R.; Vassilopoulos, A.; Parisiadou, L.; Yan, Y.; Gius, D.R. Regulation of MnSOD Enzymatic Activity by Sirt3 Connects the Mitochondrial Acetylome Signaling Networks to Aging and Carcinogenesis. Antioxidants Redox Signal. 2014, 20, 1646–1654. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, H.; Chen, X.; Zou, Y.; Li, J.; Wang, L.; Wu, M.; Zang, J.; Yu, Y.; Zhuang, W.; et al. A small molecule activator of SIRT3 promotes deacetylation and activation of manganese superoxide dismutase. Free. Radic. Boil. Med. 2017, 112, 287–297. [Google Scholar] [CrossRef]
- Dali-Youcef, N.; Lagouge, M.; Froelich, S.; Koehl, C.; Schoonjans, K.; Auwerx, J. Sirtuins: The ‘magnificent seven’, function, metabolism and longevity. Ann. Med. 2007, 39, 335–345. [Google Scholar] [CrossRef]
- Rotili, D.; Tarantino, D.; Carafa, V.; Paolini, C.; Schemies, J.; Jung, M.; Botta, G.; Di Maro, S.; Novellino, E.; Steinkühler, C.; et al. Benzodeazaoxaflavins as Sirtuin Inhibitors with Antiproliferative Properties in Cancer Stem Cells. J. Med. Chem. 2012, 55, 8193–8197. [Google Scholar] [CrossRef][Green Version]
- Rotili, D.; Tarantino, D.; Nebbioso, A.; Paolini, C.; Huidobro, C.; Lara, E.; Mellini, P.; Lenoci, A.; Pezzi, R.; Botta, G.; et al. Discovery of Salermide-Related Sirtuin Inhibitors: Binding Mode Studies and Antiproliferative Effects in Cancer Cells Including Cancer Stem Cells. J. Med. Chem. 2012, 55, 10937–10947. [Google Scholar] [CrossRef]
- Heltweg, B. Antitumor Activity of a Small-Molecule Inhibitor of Human Silent Information Regulator 2 Enzymes. Cancer Res. 2006, 66, 4368–4377. [Google Scholar] [CrossRef] [PubMed]
- Moniot, S.; Forgione, M.; Lucidi, A.; Hailu, G.S.; Nebbioso, A.; Carafa, V.; Baratta, F.; Altucci, L.; Giacché, N.; Passeri, D.; et al. Development of 1,2,4-Oxadiazoles as Potent and Selective Inhibitors of the Human Deacetylase Sirtuin 2: Structure–Activity Relationship, X-ray Crystal Structure, and Anticancer Activity. J. Med. Chem. 2017, 60, 2344–2360. [Google Scholar] [CrossRef] [PubMed]
- Polletta, L.; Vernucci, E.; Carnevale, I.; Arcangeli, T.; Rotili, D.; Palmerio, S.; Steegborn, C.; Nowak, T.; Schutkowski, M.; Pellegrini, L.; et al. SIRT5 regulation of ammonia-induced autophagy and mitophagy. Autophagy 2015, 11, 253–270. [Google Scholar] [CrossRef] [PubMed]
- Carafa, V.; Nebbioso, A.; Cuomo, F.; Rotili, D.; Cobellis, G.; Bontempo, P.; Baldi, A.; Spugnini, E.P.; Citro, G.; Chambery, A.; et al. RIP1–HAT1–SIRT Complex Identification and Targeting in Treatment and Prevention of Cancer. Clin. Cancer Res. 2018, 24, 2886–2900. [Google Scholar] [CrossRef]
- Maurer, B.; Rumpf, T.; Scharfe, M.; Stolfa, D.A.; Schmitt, M.L.; He, W.; Verdin, E.; Sippl, W.; Jung, M. Inhibitors of the NAD+-Dependent Protein Desuccinylase and Demalonylase Sirt5. ACS Med. Chem. Lett. 2012, 3, 1050–1053. [Google Scholar] [CrossRef]
- You, W.; Rotili, D.; Li, T.-M.; Kambach, C.; Meleshin, M.; Schutkowski, M.; Chua, K.F.; Mai, A.; Steegborn, C. Structural Basis of Sirtuin 6 Activation by Synthetic Small Molecules. Angew. Chem. Int. Ed. 2016, 56, 1007–1011. [Google Scholar] [CrossRef]
- Ragno, R.; Frasca, S.; Manetti, F.; Brizzi, A.; Massa, S. HIV-Reverse Transcriptase Inhibition: Inclusion of Ligand-Induced Fit by Cross-Docking Studies. J. Med. Chem. 2005, 48, 200–212. [Google Scholar] [CrossRef]
- Gaspar, L.; Coron, R.P.; Kong-Thoo-Lin, P.; Costa, D.; Pérez-Cabezas, B.; Tavares, J.; Roura-Ferrer, M.; Ramos, I.; Ronin, C.; Major, L.; et al. Inhibitors of Trypanosoma cruzi Sir2 related protein 1 as potential drugs against Chagas disease. PLOS Neglected Trop. Dis. 2018, 12, e0006180. [Google Scholar] [CrossRef]
- Bastos, T.M.; Russo, H.M.; Moretti, N.S.; Schenkman, S.; Marcourt, L.; Gupta, M.P.; Wolfender, J.-L.; Queiroz, E.F.; Soares, M. Chemical Constituents of Anacardium occidentale as Inhibitors of Trypanosoma cruzi Sirtuins. Mol. 2019, 24, 1299. [Google Scholar] [CrossRef] [PubMed]
- The UniProt Consortium UniProt: The universal protein knowledgebase. Nucleic Acids Res. 2018, 46, 2699. [CrossRef] [PubMed]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; Beer, T.A.P.D.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [PubMed]
- Ronin, C.; Costa, D.; Tavares, J.; Faria, J.; Ciesielski, F.; Ciapetti, P.; Smith, T.K.; MacDougall, J.; Da Silva, A.C.; Pemberton, I. The crystal structure of the Leishmania infantum Silent Information Regulator 2 related protein 1: Implications to protein function and drug design. PLoS ONE 2018, 13, e0193602. [Google Scholar] [CrossRef]
- Zhao, K.; Chai, X.; Marmorstein, R. Structure and Substrate Binding Properties of cobB, a Sir2 Homolog Protein Deacetylase from Escherichia coli. J. Mol. Boil. 2004, 337, 731–741. [Google Scholar] [CrossRef]
- Bas, D.C.; Rogers, D.; Jensen, J.H. Very fast prediction and rationalization of pKa values for protein-ligand complexes. Proteins: Struct. Funct. Bioinform. 2008, 73, 765–783. [Google Scholar] [CrossRef]
- Dolinsky, T.J.; Czodrowski, P.; Li, H.; Nielsen, J.E.; Jensen, J.H.; Klebe, G.; Baker, N.A. PDB2PQR: Expanding and upgrading automated preparation of biomolecular structures for molecular simulations. Nucleic Acids Res. 2007, 35, W522–W525. [Google Scholar] [CrossRef]
- Zhao, X.; Allison, D.; Condon, B.; Zhang, F.; Gheyi, T.; Zhang, A.; Ashok, S.; Russell, M.; MacEwan, I.; Qian, Y.; et al. The 2.5 Å Crystal Structure of the SIRT1 Catalytic Domain Bound to Nicotinamide Adenine Dinucleotide (NAD+) and an Indole (EX527 Analogue) Reveals a Novel Mechanism of Histone Deacetylase Inhibition. J. Med. Chem. 2013, 56, 963–969. [Google Scholar] [CrossRef]
- Min, J.; Landry, J.; Sternglanz, R.; Xu, R.-M. Crystal Structure of a SIR2 Homolog–NAD Complex. Cell 2001, 105, 269–279. [Google Scholar] [CrossRef]
- Hoff, K.G.; Avalos, J.L.; Sens, K.; Wolberger, C. Insights into the Sirtuin Mechanism from Ternary Complexes Containing NAD+ and Acetylated Peptide. Struct. 2006, 14, 1231–1240. [Google Scholar] [CrossRef]
- Papamokos, G.; Tziatzos, G.; Papageorgiou, D.G.; Georgatos, S.; Politou, A.S.; Kaxiras, E. Structural Role of RKS Motifs in Chromatin Interactions: A Molecular Dynamics Study of HP1 Bound to a Variably Modified Histone Tail. Biophys. J. 2012, 102, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and testing of a general amber force field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, W.; Kollman, P.A.; Case, D.A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 2006, 25, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Jakalian, A.; Jack, D.B.; Bayly, C.I. Fast, efficient generation of high-quality atomic charges. AM1-BCC model: II. Parameterization and validation. J. Comput. Chem. 2002, 23, 1623–1641. [Google Scholar] [CrossRef]
- Izadi, S.; Anandakrishnan, R.; Onufriev, A.V. Building Water Models: A Different Approach. J. Phys. Chem. Lett. 2014, 5, 3863–3871. [Google Scholar] [CrossRef]
- Maier, J.A.; Martinez, C.; Kasavajhala, K.; Wickstrom, L.; Hauser, K.; Simmerling, C.L. ff14SB: Improving the Accuracy of Protein Side Chain and Backbone Parameters from ff99SB. J. Chem. Theory Comput. 2015, 11, 3696–3713. [Google Scholar] [CrossRef]
- David, A.; Case, R.C.; Walker, T.E.; Cheatham, C., III; Simmerling, A.; Roitberg, K.M.; Merz, R.; Luo, T.; Darden, J.; Wang, R.E.; et al. AMBER 2018 Reference Manual Covers Amber18 and AmberTools18. 2018. Available online: https://ambermd.org/doc12/Amber18.pdf (accessed on 28 April 2020).
- Eastman, P.; Swails, J.; Chodera, J.D.; McGibbon, R.T.; Zhao, Y.; Beauchamp, K.A.; Wang, L.-P.; Simmonett, A.; Harrigan, M.; Stern, C.D.; et al. OpenMM 7: Rapid development of high performance algorithms for molecular dynamics. PLoS Comput. Boil. 2017, 13, e1005659. [Google Scholar] [CrossRef]
- Korb, O.; Monecke, P.; Hessler, G.; Stutzle, T.; Exner, T.E. pharmACOphore: Multiple flexible ligand alignment based on ant colony optimization. J. Chem. Inf. Model. 2010, 50, 1669–1681. [Google Scholar] [CrossRef]
- Korb, O.; Möller, H.M.; Exner, T.E. NMR-Guided Molecular Docking of a Protein-Peptide Complex Based on Ant Colony Optimization. ChemMedChem 2010, 5, 1001–1006. [Google Scholar] [CrossRef]
- Koes, D.R.; Baumgartner, M.; Camacho, C.J. Lessons Learned in Empirical Scoring with smina from the CSAR 2011 Benchmarking Exercise. J. Chem. Inf. Model. 2013, 53, 1893–1904. [Google Scholar] [CrossRef]
- Ragno, R.; Ballante, F.; Pirolli, A.; Wickersham, R.B.; Patsilinakos, A.; Hesse, S.; Perspicace, E.; Kirsch, G. Vascular endothelial growth factor receptor-2 (VEGFR-2) inhibitors: Development and validation of predictive 3-D QSAR models through extensive ligand- and structure-based approaches. J. Comput. Mol. Des. 2015, 29, 757–776. [Google Scholar] [CrossRef] [PubMed]
- Siqueira-Neto, J.; Moon, S.; Jang, J.; Yang, G.; Lee, C.; Moon, H.K.; Chatelain, E.; Genovesio, A.; Cechetto, J.; Freitas-Junior, L.H. An Image-Based High-Content Screening Assay for Compounds Targeting Intracellular Leishmania donovani Amastigotes in Human Macrophages. PLoS Negl. Trop. Dis. 2012, 6, e1671. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.H.; Alcântara, L.M.; Chatelain, E.; Freitas-Junior, L.; Moraes, C.B. Drug Discovery for Chagas Disease: Impact of Different Host Cell Lines on Assay Performance and Hit Compound Selection. Trop. Med. Infect. Dis. 2019, 4, 82. [Google Scholar] [CrossRef] [PubMed]

| Compounds | TcSir2rp1 | TcSir2rp3 |
|---|---|---|
| IC50 ± SD (µM) a | IC50 ± SD (µM) a | |
| 8 | ND | 18.8 ± 5.34 |
| 12 | ND | 14.3 ± 6.06 |
| 13 | ND | 18.5 ± 1.97 |
| 15 | 32.4 ± 7.88 | ND |
| 17 | 25.5 ± 4.73 | ND |
| 31 | ND | 16.3 ± 9.54 |
| 32 | ND | 16.4 ± 6.70 |
| Compound | EC50 Value (µM) | CC50 (µM) | SI |
|---|---|---|---|
| BZN | 1.96 | >200 | 102 |
| 27a | 5.73 | 46.87 | 8.18 |
| 17b | 8.5 | 19.6 | 2.31 |
| 15b | 19.35 | 36.4 | 1.89 |
| 12c | 35.4 | 48.8 | 1.38 |
| 32c | 39.8 | 77 | 1.93 |
| 8c | 40.6 | 76.8 | 1.89 |
| 30c | 42.7 | >100 | 1.93 |
| 13c | 45.1 | >100 | 2.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matutino Bastos, T.; Botelho Pereira Soares, M.; Haddad Franco, C.; Alcântara, L.; Antonini, L.; Sabatino, M.; Mautone, N.; Holanda Freitas-Junior, L.; Moraes, C.B.; Ragno, R.; et al. Identification of Inhibitors to Trypanosoma cruzi Sirtuins Based on Compounds Developed to Human Enzymes. Int. J. Mol. Sci. 2020, 21, 3659. https://doi.org/10.3390/ijms21103659
Matutino Bastos T, Botelho Pereira Soares M, Haddad Franco C, Alcântara L, Antonini L, Sabatino M, Mautone N, Holanda Freitas-Junior L, Moraes CB, Ragno R, et al. Identification of Inhibitors to Trypanosoma cruzi Sirtuins Based on Compounds Developed to Human Enzymes. International Journal of Molecular Sciences. 2020; 21(10):3659. https://doi.org/10.3390/ijms21103659
Chicago/Turabian StyleMatutino Bastos, Tanira, Milena Botelho Pereira Soares, Caio Haddad Franco, Laura Alcântara, Lorenzo Antonini, Manuela Sabatino, Nicola Mautone, Lucio Holanda Freitas-Junior, Carolina Borsoi Moraes, Rino Ragno, and et al. 2020. "Identification of Inhibitors to Trypanosoma cruzi Sirtuins Based on Compounds Developed to Human Enzymes" International Journal of Molecular Sciences 21, no. 10: 3659. https://doi.org/10.3390/ijms21103659
APA StyleMatutino Bastos, T., Botelho Pereira Soares, M., Haddad Franco, C., Alcântara, L., Antonini, L., Sabatino, M., Mautone, N., Holanda Freitas-Junior, L., Moraes, C. B., Ragno, R., Rotili, D., Schenkman, S., Mai, A., & Silvio Moretti, N. (2020). Identification of Inhibitors to Trypanosoma cruzi Sirtuins Based on Compounds Developed to Human Enzymes. International Journal of Molecular Sciences, 21(10), 3659. https://doi.org/10.3390/ijms21103659









