Interstitial Leydig Cell Tumorigenesis—Leptin and Adiponectin Signaling in Relation to Aromatase Expression in the Human Testis
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Samples and Ethical Considerations
2.2. Body Fat Measurement
2.3. Light andTtransmission Electron Microscopy Analyses
2.4. Western Blotting
2.5. Immunohistochemistry
2.6. Statistics
3. Results
3.1. Body Mass Index in Patients with Leydig Cell Tumor
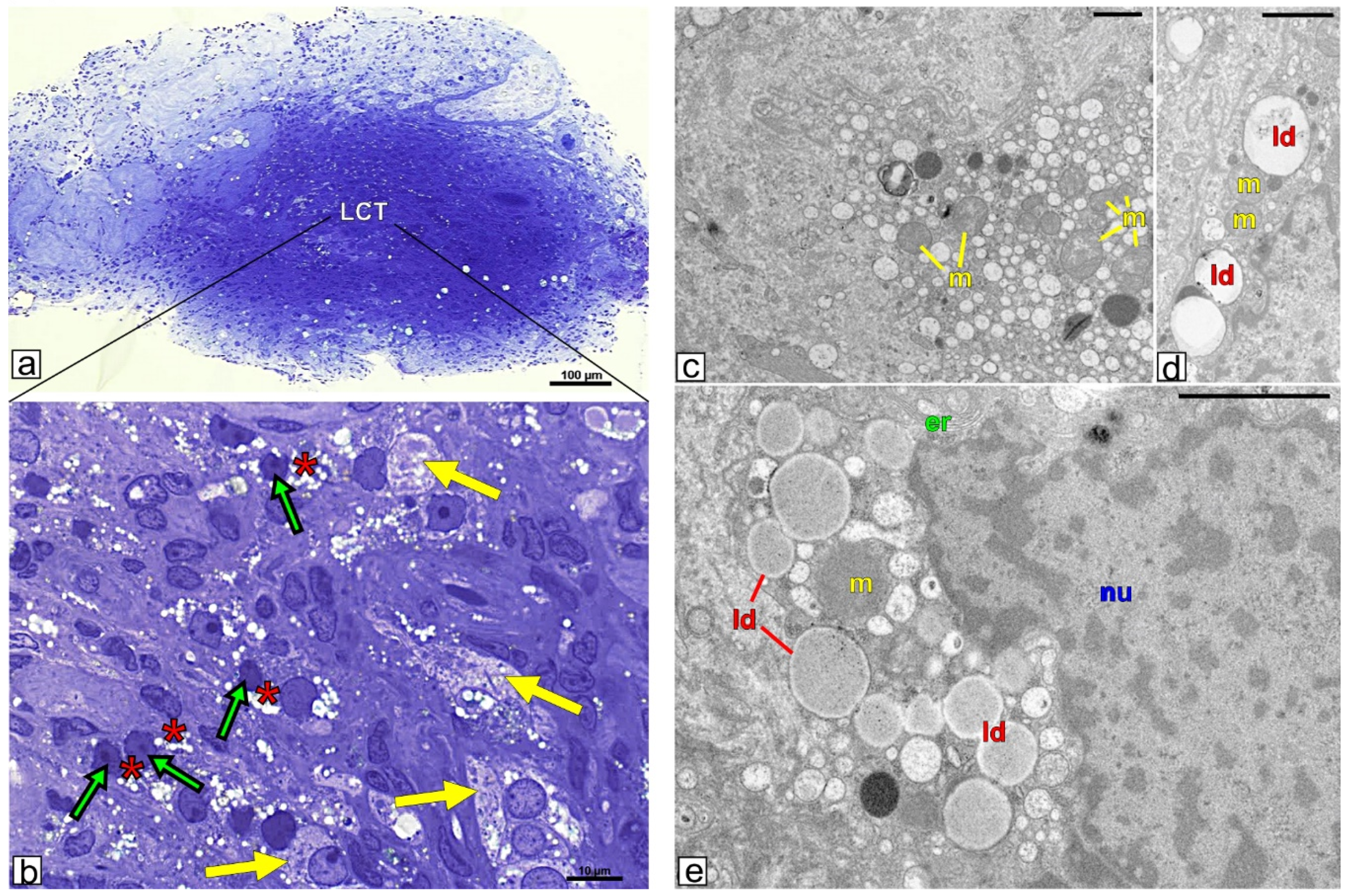
3.2. Topography and Ultrastructure of Leydig Cell Tumor
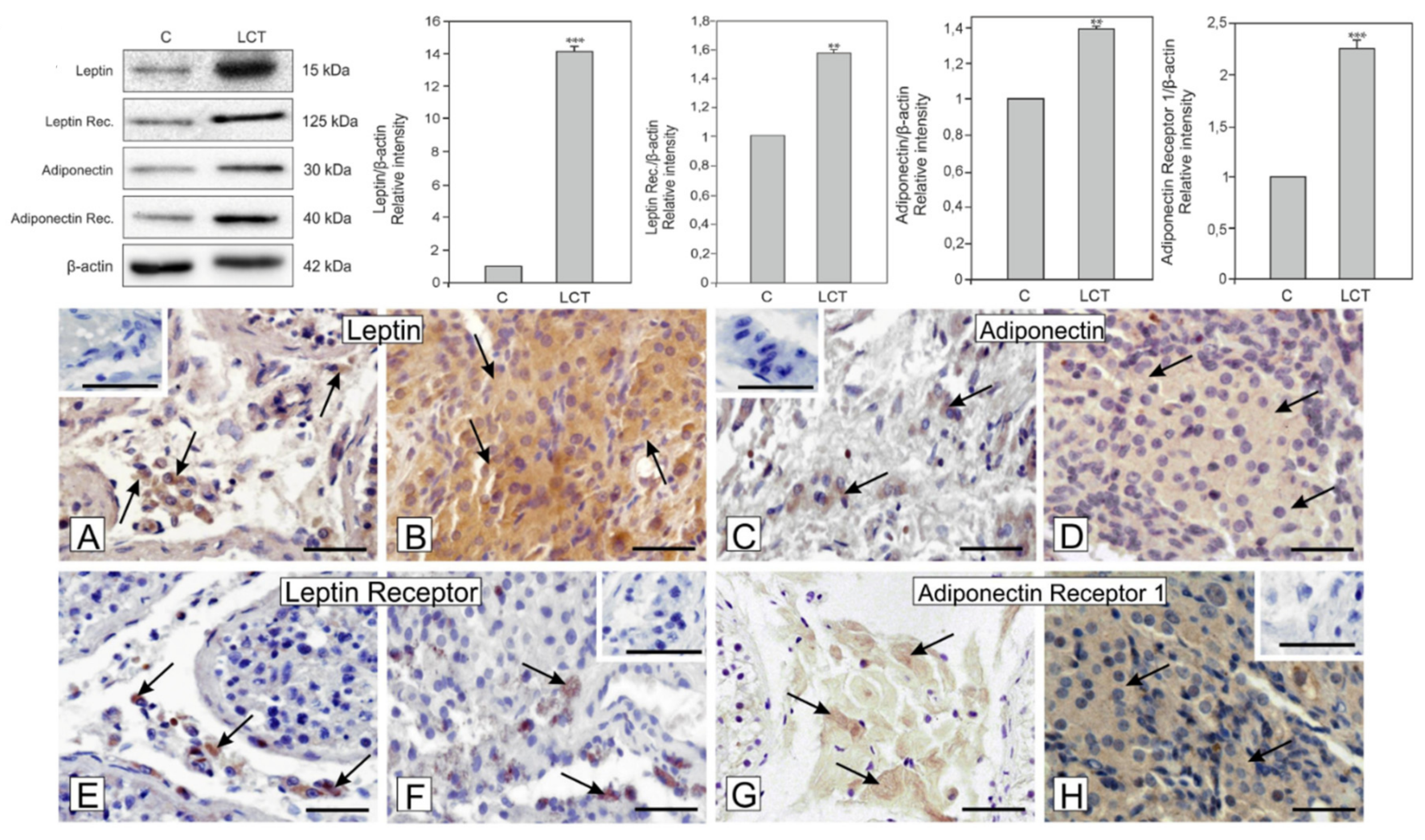
3.3. Expression of Leptin and Adiponectin and Their Receptors in Leydig Cell Tumor
3.4. Localization of Leptin, Adiponectin and Their Receptors in Leydig Cell Tumor
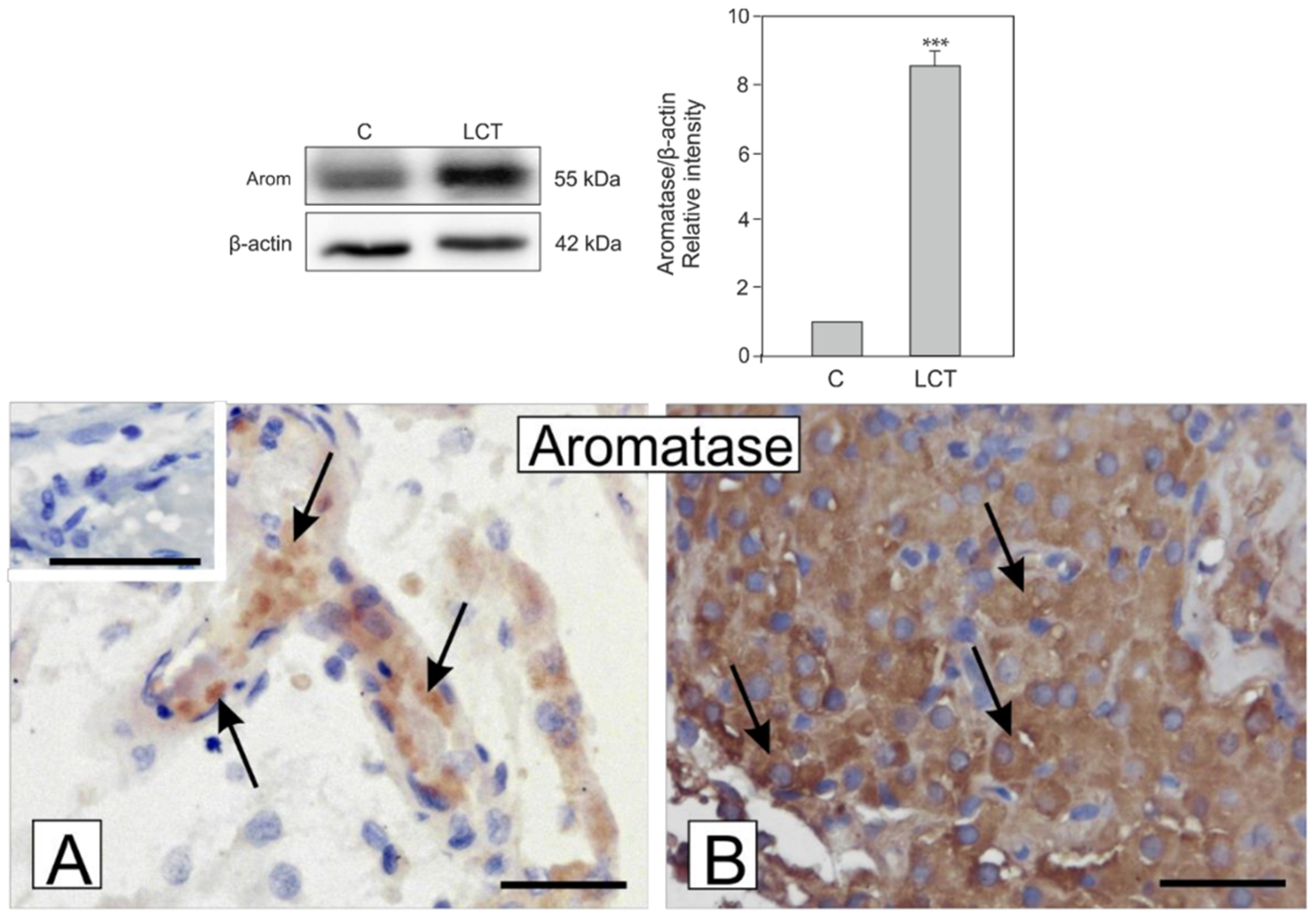
3.5. Expression and Localization of Aromatase in Leydig Cell Tumor
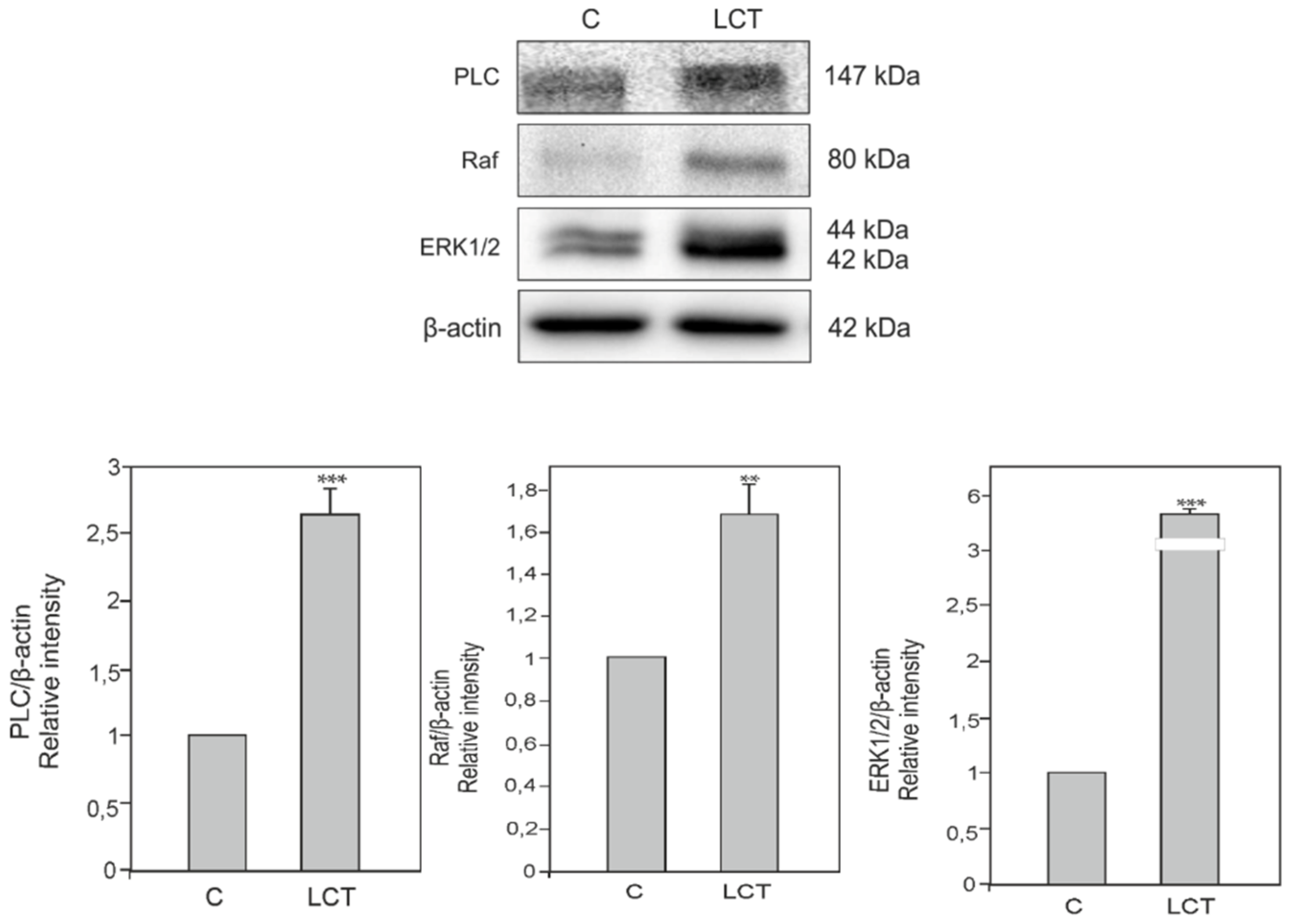
3.6. Expression of Phospholipase C (PLC), Kinase Raf (Raf) and Extracellular signal-regulated kinase (ERK) in Leydig Cell Tumor
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coelho, M.; Oliveira, T.; Fernandes, R. State of the art paper Biochemistry of adipose tissue: An endocrine organ. Arch. Med. Sci. 2013, 9, 191–200. [Google Scholar] [CrossRef]
- Zhang, Y.; Proença, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Friedman, J.M.; Halaas, J.L. Leptin and the regulation of body weight in mammals. Nature 1998, 395, 763–770. [Google Scholar] [CrossRef]
- Rosenbaum, M.; Goldsmith, R.; Bloomfield, D.; Magnano, A.; Weimer, L.; Heymsfield, S.; Gallagher, D.; Mayer, L.; Murphy, E.; Leibel, R.L. Low-dose leptin reverses skeletal muscle, autonomic, and neuroendocrine adaptations to maintenance of reduced weight. J. Clin. Investig. 2005, 115, 3579–3586. [Google Scholar] [CrossRef]
- Berg, A.H.; Combs, T.P.; Du, X.; Brownlee, M.; Scherer, P.E. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat. Med. 2001, 7, 947–953. [Google Scholar] [CrossRef]
- Qi, Y.; Takahashi, N.; Hileman, S.M.; Patel, H.R.; Berg, A.H.; Pajvani, U.B.; Scherer, E.P.; Ahima, R.S. Adiponectin acts in the brain to decrease body weight. Nat. Med. 2004, 10, 524–529. [Google Scholar] [CrossRef]
- Stern, J.H.; Rutkowski, J.M.; Scherer, P.E. Adiponectin, Leptin, and Fatty Acids in the Maintenance of Metabolic Homeostasis through Adipose Tissue Crosstalk. Cell Metab. 2016, 23, 770–784. [Google Scholar] [CrossRef]
- Brunner, L.; Nick, H.-P.; Cumin, F.; Chiesi, M.; Baum, H.-P.; Whitebread, S.; Stricker-Krongrad, A.; Levens, N. Leptin is a physiologically important regulator of food intake. Int. J. Obes. 1997, 21, 1152–1160. [Google Scholar] [CrossRef]
- Tatemoto, K.; Hosoya, M.; Habata, Y.; Fujii, R.; Kakegawa, T.; Zou, M.-X.; Kawamata, Y.; Fukusumi, S.; Hinuma, S.; Kitada, C.; et al. Isolation and Characterization of a Novel Endogenous Peptide Ligand for the Human APJ Receptor. Biochem. Biophys. Res. Commun. 1998, 251, 471–476. [Google Scholar] [CrossRef]
- Tena-Sempere, M.; Manna, P.; Zhang, F.; Pinilla, L.; Gonzalez, L.; Dieguez, C.; Huhtaniemi, I.; Aguilar, E.; Thomas, G.; Baker, S.; et al. Molecular mechanisms of leptin action in adult rat testis: Potential targets for leptin-induced inhibition of steroidogenesis and pattern of leptin receptor messenger ribonucleic acid expression. J. Endocrinol. 2001, 170, 413–423. [Google Scholar] [CrossRef]
- Leisegang, K.; Bouic, P.J.; Menkveld, R.; Henkel, R.R.; Bouic, P.J.; Menkveld, R.; Henkel, R.R. Obesity is associated with increased seminal insulin and leptin alongside reduced fertility parameters in a controlled male cohort. Reproduct. Biol. Endocrinol. 2014, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Hess, R. Estrogen in the adult male reproductive tract: A review. Reprod. Boil. Endocrinol. 2003, 1, 52. [Google Scholar] [CrossRef] [PubMed]
- Carreau, S.; Wolczynski, S.; Galeraud-Denis, I. Aromatase, oestrogens and human male reproduction. Philos. Trans. R. Soc. B: Boil. Sci. 2010, 365, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Carpino, A.; Rago, V.; Pezzi, V.; Carani, C.; Andὸ, S. Detection of aromatase and estrogen receptors (ERα, ERβ1, ERβ2) in human Leydig cell tumor. Eur. J. Endocrinol. 2007, 157, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Gancarczyk, M.; Paziewska-Hejmej, A.; Carreau, S.; Tabarowski, Z.; Bilinska, B. Dose- and photoperiod-dependent effects of 17β-estradiol and the anti-estrogen ICI 182,780 on testicular structure, acceleration of spermatogenesis, and aromatase immunoexpression in immature bank voles. Acta Histochem. 2004, 106, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Ehrlich, S.; Toth, T.L.; Wright, D.L.; Calafat, A.M.; Trisini, A.T.; Ye, X.; Hauser, R. Semen quality and sperm DNA damage in relation to urinary bisphenol A among men from an infertility clinic. Reprod. Toxicol. 2010, 30, 532–539. [Google Scholar] [CrossRef]
- Toppari, J.; Virtanen, H.E.; Main, K.M.; Skakkebaek, N.E. Cryptorchidism and hypospadias as a sign of testicular dysgenesis syndrome (TDS): Environmental connection. Birth Defects Res. Part A Clin. Mol. Teratol. 2010, 88, 910–919. [Google Scholar] [CrossRef]
- Panza, S.; Malivindi, R.; Chemi, F.; Rago, V.; Giordano, C.; Barone, I.; Bonofiglio, D.; Gelsomino, L.; Giordano, F.; Andò, S.; et al. Glucocorticoid Receptor as a Potential Target to Decrease Aromatase Expression and Inhibit Leydig Tumor Growth. Am. J. Pathol. 2016, 186, 1328–1339. [Google Scholar] [CrossRef]
- Maris, P.; Campana, A.; Barone, I.; Giordano, C.; Morelli, C.; Malivindi, R.; Sisci, D.; Aquila, S.; Rago, V.; Bonofiglio, D.; et al. Androgens Inhibit Aromatase Expression Through DAX-1: Insights Into the Molecular Link Between Hormone Balance and Leydig Cancer Development. Endocrinology 2015, 156, 1251–1262. [Google Scholar] [CrossRef][Green Version]
- Sirianni, R.; Chimento, A.; Malivindi, R.; Mazzitelli, I.; Andò, S.; Pezzi, V. Insulin-Like Growth Factor-I, Regulating Aromatase Expression through Steroidogenic Factor 1, Supports Estrogen-Dependent Tumor Leydig Cell Proliferation. Cancer Res. 2007, 67, 8368–8377. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Shelton, L.M. Cancer as a metabolic disease. Nutr. Metab. 2010, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Gheorghisan-Galateanu, A.-A. Leydig cell tumors of the testis: A case report. BMC Res. Notes 2014, 7, 656. [Google Scholar] [CrossRef] [PubMed]
- Due, W.; Dieckmann, K.-P.; Loy, V.; Stein, H. Immunohistological determination of oestrogen receptor, progesterone receptor, and intermediate filaments in Leydig cell tumours, Leydig cell hyperplasia, and normal Leydig cells of the human testis. J. Pathol. 1989, 157, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.; Goel, A.; Sharma, P.; Kumar, S.; Vijay, M.; Dutta, A. Leydig cell tumor with mediastinal and lung metastasis. Saudi J. Kidney Dis. Transplant. 2011, 22, 354–356. [Google Scholar]
- Vasilakaki, T.; Michalis, L.; Skafida, E.; Arkoumani, E.; Delliou, E.; Grammatoglou, X.; Kontovourkis, P.; Papamichail, V.; Stamatiou, K. An unusual case of unilateral malignant leydig cell tumour of the testis. Case Rep. Oncol. 2011, 4, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Richmond, I.; Banerjee, S.S.; Eyden, B.P.; Sissons, M.C. Sarcomatoid Leydig cell tumor of the testis. Histopathology 1995, 27, 578–580. [Google Scholar] [CrossRef]
- Hibi, H.; Yamashita, K.; Sumitomo, M.; Asada, Y. Leydig cell tumor of the testis, presenting with azoospermia. Reprod. Med.Biol. 2017, 16, 392–395. [Google Scholar] [CrossRef]
- Kotula-Balak, M.; Gorowska-Wojtowicz, E.; Milon, A.; Pawlicki, P.; Tworzydlo, W.; Płachno, B.J.; Krakowska, I.; Hejmej, A.; Wolski, J.K.; Bilinska, B. Towards understanding leydigioma: Do G protein-coupled estrogen receptor and peroxisome proliferator–activated receptor regulate lipid metabolism and steroidogenesis in Leydig cell tumors? Protoplasma 2020, 1–15. [Google Scholar] [CrossRef]
- Gorowska-Wojtowicz, E.; Duliban, M.; Kudrycka, M.; Dutka, P.; Pawlicki, P.; Milon, A.; Zarzycka, M.; Placha, W.; Kotula-Balak, M.; Ptak, A.; et al. Leydig cell tumorigenesis—Implication of G-coupled estrogen receptor/peroxisome proliferator-activated receptor interaction and xenoestrogen exposure. In vivo and in vitro appraisal. Tissue Cell 2019, 61, 51–60. [Google Scholar] [CrossRef]
- Schlegel, P.N.; Li, P.S. Microdissection TESE: Sperm retrieval in non-obstructive azoospermia. Hum. Reprod. Updat. 1998, 4. [Google Scholar] [CrossRef]
- Bilinska, B.; Hejmej, A.; Kotula-Balak, M. Preparation of Testicular Samples for Histology and Immunohistochemistry. In Advanced Structural Safety Studies; Springer Science and Business Media LLC: Berlin, Germany, 2018; Volume 1748, pp. 17–36. [Google Scholar]
- Smolen, A.J. Image analytic techniques for quantification of immunocytochemical staining in the nervous system. In Methods in Neurosciences; Academic Press: San Diego, CA, USA, 1990; pp. 208–229. [Google Scholar]
- Brown, L.M.; Gent, L.; Davis, K.; Clegg, D.J. Metabolic impact of sex hormones on obesity. Brain Res. 2010, 1350, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Pelleymounter, M.A.; Baker, M.B.; McCaleb, M. Does estradiol mediate leptin’s effects on adiposity and body weight? Am. J. Physiol. Content 1999, 276, E955–E963. [Google Scholar] [CrossRef]
- Fungfuang, W.; Terada, M.; Komatsu, N.; Moon, C.; Saito, T.R. Effects of estrogen on food intake, serum leptin levels and leptin mRNA expression in adipose tissue of female rats. Lab. Anim. Res. 2013, 29, 168–173. [Google Scholar] [CrossRef]
- Saad, M.F.; Damani, S.; Gingerich, R.L.; Riad-Gabriel, M.G.; Khan, A.; Boyadjian, R.; Jinagouda, S.D.; el-Tawil, K.; Rude, R.K.; Kamdar, V. Sexual dimorphism in plasma leptin concentration. J. Clin. Endocrinol. Metab. 1997, 82, 579–584. [Google Scholar] [CrossRef]
- Tanaka, M.; Nakaya, S.; Kumai, T.; Watanabe, M.; Tateishi, T.; Shimizu, H.; Kobayashi, S. Effects of estrogen on serum leptin levels and leptin mRNA expression in adipose tissue in rats. Horm. Res. 2001, 56, 98–104. [Google Scholar] [CrossRef]
- Kunnari, A.; Santaniemi, M.; Jokela, M.; Karjalainen, A.H.; Heikkinen, J.; Ukkola, O.; Kesäniemi, Y.A. Estrogen replacement therapy decreases plasma adiponectin but not resistin in postmenopausal women. Metabolism 2008, 57, 1509–1515. [Google Scholar] [CrossRef]
- Lizcano, F.; Guzmán, G. Estrogen Deficiency and the Origin of Obesity during Menopause. BioMed Res. Int. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Wang, Q.-P.; Yang, L.; Li, X.-P.; Xie, H.; Liao, E.-Y.; Wang, M.; Luo, X.-H. Effects of 17β-estradiol on adiponectin regulation of the expression of osteoprotegerin and receptor activator of nuclear factor-κB ligand. Bone 2012, 51, 515–523. [Google Scholar] [CrossRef]
- Ishikawa, T.; Fujioka, H.; Ishimura, T.; Takenaka, A.; Fujisawa, M. Expression of leptin and leptin receptor in the testis of fertile and infertile patients. Andrologia 2007, 39, 22–27. [Google Scholar] [CrossRef]
- Herrid, M.; O’Shea, T.; McFarlane, J.R. Ontogeny of leptin and its receptor expression in mouse testis during the postnatal period. Mol. Reprod. Dev. 2008, 75, 874–880. [Google Scholar] [CrossRef]
- Kawwass, J.F.; Summer, R.; Kallen, C. Direct effects of leptin and adiponectin on peripheral reproductive tissues: A critical review. Mol. Hum. Reprod. 2015, 21, 617–632. [Google Scholar] [CrossRef] [PubMed]
- Milon, A.; Pawlicki, P.; Rak, A.; Mlyczynska, E.; Płachno, B.J.; Tworzydlo, W.; Gorowska-Wojtowicz, E.; Bilinska, B.; Kotula-Balak, M. Telocytes are localized to testis of the bank vole (Myodes glareolus) and are affected by lighting conditions and G-coupled membrane estrogen receptor (GPER) signaling. Gen. Comp. Endocrinol. 2018, 271, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Vansaun, M.N. Molecular pathways: Adiponectin and leptin signaling in cancer. Clin. Cancer Res. 2013, 19, 1926–1932. [Google Scholar] [CrossRef]
- Caminos, J.E.; Nogueiras, R.; Gaytán, F.; Pineda, R.; González, C.R.; Barreiro, M.L.; Castaño, J.P.; Malagón, M.M.; Pinilla, L.; Toppari, J.; et al. Novel expression and direct effects of adiponectin in the rat testis. Endocrinology 2008, 149, 3390–3402. [Google Scholar] [CrossRef]
- Martin, L.J. Implications of adiponectin in linking metabolism to testicular function. Endocrine 2014, 46, 16–28. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef]
- Schech, A.; Yu, S.; Goloubeva, O.; McLenithan, J.; Sabnis, G. A nude mouse model of obesity to study the mechanisms of resistance to aromatase inhibitors. Endocr. Relat. Cancer 2015, 22, 645–656. [Google Scholar] [CrossRef]
- Hoffmann, M.; Gogola, J.; Ptak, A. Adiponectin Reverses the Proliferative Effects of Estradiol and IGF-1 in Human Epithelial Ovarian Cancer Cells by Downregulating the Expression of Their Receptors. Horm. Cancer 2018, 9, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Shuang, L.; Jidong, W.; Hongjuan, P.; Zhenwei, Y. Effects of apelin on proliferation and apoptosis in rat ovarian granulosa cells. Clin. Exp. Obstet. Gynecol. 2016, 43, 409–413. [Google Scholar]
- Szyszka, M.; Tyczewska, M.; Milecka, P.; Jopek, K.; Celichowski, P.; Malendowicz, L.K.; Rucinski, M. Effects of leptin on leptin receptor isoform expression and proliferative activity in human normal prostate and prostate cancer cell lines. Oncol. Rep. 2017, 39, 182–192. [Google Scholar] [CrossRef]
- Mendis-Handagama, C.; Ariyaratne, H.S. Differentiation of the adult Leydig cell population in the postnatal testis. Boil. Reprod. 2001, 65, 660–671. [Google Scholar] [CrossRef]
- Shi, L.; Song, R.; Yao, X.; Ren, Y. Effects of selenium on the proliferation, apoptosis and testosterone production of sheep Leydig cells in vitro. Theriogenology 2017, 93, 24–32. [Google Scholar] [CrossRef]
- Zumoff, B.; Strain, G.W.; Kream, J.; O’Connor, J.; Levin, J.; Fukushima, D.K. Obese young men have elevated plasma estrogen levels but obese premenopausal women do not. Metabolism 1981, 30, 1011–1014. [Google Scholar] [CrossRef]
- Xu, X.; Sun, M.; Ye, J.; Luo, D.; Su, X.; Zheng, D.; Feng, L.; Gao, L.; Yu, C.; Guan, Q. The Effect of Aromatase on the Reproductive Function of Obese Males. Horm. Metab. Res. 2017, 49, 572–579. [Google Scholar] [CrossRef]
- Simpson, E.R.; Misso, M.; Hewitt, K.N.; Hill, R.A.; Boon, W.C.; Jones, M.E.; Kovacic, A.; Zhou, J.; Clyne, C.D. Estrogen—The good, the bad, and the unexpected. Endocr. Rev. 2005, 26, 322–330. [Google Scholar] [CrossRef]
- Haverfield, J.T.; Ham, S.; Brown, K.A.; Simpson, E.R.; Meachem, S.J. Teasing out the role of aromatase in the healthy and diseased testis. Spermatogenesis 2011, 1, 240–249. [Google Scholar] [CrossRef][Green Version]
- Melichar, B.; Kalábová, H.; Krcmová, L.; Urbánek, L.; Hyspler, R.; Solichová, D.; Melicharová, K.; Pecka, M.; Zadák, Z. Effect of aromatase inhibitors on lipid metabolism, inflammatory response and antioxidant balance in patients with breast carcinoma. Anticancer. Res. 2009, 29, 3337–3346. [Google Scholar]
- Rago, V.; Romeo, F.; Giordano, F.; Maggiolini, M.; Carpino, A. Identification of the estrogen receptor GPER in neoplastic and non-neoplastic human testes. Reprod. Boil. Endocrinol. 2011, 9, 135. [Google Scholar] [CrossRef]
- O’Neil, J.S.; Burow, M.E.; Green, A.E.; McLachlan, J.A.; Henson, M.C. Effects of estrogen on leptin gene promoter activation in MCF-7 breast cancer and JEG-3 choriocarcinoma cells: Selective regulation via estrogen receptors alpha and beta. Mol. Cell. Endocrinol. 2001, 176, 67–75. [Google Scholar] [CrossRef]
- Gambino, Y.P.; Maymó, J.L.; Pérez, A.P.; Dueñas-Díez, J.-L.; Sánchez-Margalet, V.; Calvo, J.C.; Varone, C. 17Beta-Estradiol Enhances Leptin Expression in Human Placental Cells Through Genomic and Nongenomic Actions1. Boil. Reprod. 2010, 83, 42–51. [Google Scholar] [CrossRef]
- Yi, K.W.; Shin, J.H.; Seo, H.S.; Lee, J.K.; Oh, M.J.; Kim, T.; Saw, H.S.; Kim, S.H.; Hur, J.Y. Role of estrogen receptor-alpha and -beta in regulating leptin expression in 3T3-L1 adipocytes. Obesity 2008, 16, 2393–2399. [Google Scholar] [CrossRef] [PubMed]
- Frühbeck, G. Intracellular signalling pathways activated by leptin. Biochem. J. 2005, 393, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Swick, A.G.; Romsos, D.R. Leptin constrains phospholipase C-protein kinase C-induced insulin secretion via a phosphatidylinositol 3-kinase-dependent pathway. Exp. Boil. Med. 2003, 228, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Deepa, S.S.; Etzler, J.C.; Ryu, J.; Mao, X.; Fang, Q.; Liu, D.D.; Torres, J.M.; Jia, W.; Lechleiter, J.D.; et al. Adiponectin activates AMP-activated protein kinase in muscle cells via APPL1/LKB1-dependent and phospholipase C/Ca2+/Ca2+/calmodulin-dependent protein kinase kinase-dependent pathways. J. Biol. Chem. 2009, 284, 22426–224350. [Google Scholar] [CrossRef]
- Zhang, W.; Shu, C.; Li, Q.; Li, M.; Li, X. Adiponectin affects vascular smooth muscle cell proliferation and apoptosis through modulation of the mitofusin-2-mediated Ras-Raf-Erk1/2 signaling pathway. Mol. Med. Rep. 2015, 12, 4703–4707. [Google Scholar] [CrossRef]
- Arias-Alvarez, M.; García-García, R.M.; Torres-Rovira, L.; González-Bulnes, A.; Rebollar, P.G.; Lorenzo, P.L. Influence of leptin on in vitro maturation and steroidogenic secretion of cumulus-oocyte complexes through JAK2/STAT3 and MEK 1/2 pathways in the rabbit model. Reproduction 2010, 139, 523–532. [Google Scholar] [CrossRef]
- Gomes, E.T.; Costa, J.A.S.; Silva, D.M.F.; Al Shebli, W.; Azevedo, M.L.; Monteiro, P.L.J.; Araújo Silva, R.A.J.; Santos Filho, A.S.; Guerra, M.M.P.; Bartolomeu, C.C. Effects of adiponectin during in vitro maturation of goat oocytes: MEK 1/2 pathway and gene expression pattern. Reprod. Domest. Anim. 2018, 53, 1323–1329. [Google Scholar] [CrossRef]
- Donadeu, F.X.; Esteves, C.L.; Doyle, L.K.; Walker, C.A.; Schauer, S.N.; Diaz, C.A. Phospholipase Cβ3 mediates LH-induced granulosa cell differentiation. Endocrinology 2011, 152, 2857–2869. [Google Scholar] [CrossRef]
- Kadooka, K.; Sato, M.; Matsumoto, T.; Kuhara, S.; Katakura, Y.; Fujimura, T. Pig testis extract augments adiponectin expression and secretion through the peroxisome proliferator-activated receptor signaling pathway in 3T3-L1 adipocytes. Cytotechnology 2018, 70, 983–992. [Google Scholar] [CrossRef]
- Frühbeck, G.; Catalán, V.; Rodríguez, A.; Gómez-Ambrosi, J. Adiponectin- leptin ratio: A promising index to estimate adipose tissue dysfunction. Relation with obesity-associated cardiometabolic risk. Adipocyte 2018, 7, 57–62. [Google Scholar] [CrossRef]
- Awede, B.; Adovoekpe, D.; Adehan, G.; Macfarlane, N.G.; Azonbakin, S.; Dossou, E.; Amoussou-Guénou, M.; Djrolo, F. Adiponectin, in contrast to leptin, is not associated with body mass index, waist circumference and HOMA-IR in subjects of a west-African population. Physiol. Rep. 2018, 6, e13718. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Sengupta, P.; Biswas, A. Adiponectin in male reproduction and infertility. Asian Pac. J. Reprod. 2019, 8, 244. [Google Scholar] [CrossRef]
| Antibody | Host Species | Vendor | Dilution |
|---|---|---|---|
| adiponectin | goat | Santa Cruz Biotechnology cat no. sc-26496 | 1:50 (IHC) 1:500 (WB) |
| adiponectin receptor 1 | goat | Santa Cruz Biotechnology cat no. sc-46749 | 1:50 (IHC) 1:500 (WB) |
| cytochrome P450 aromatase | mouse | Bio-Rad cat no. MCA2077S | 1:50 (IHC) 1:500 (WB) |
| leptin | mouse | Sigma–Aldrich cat. no. L3160 | 1:50 (IHC) 1:500 (WB) |
| leptin receptor | mouse | Santa Cruz Biotechnology cat. no. sc-8391 | 1:50 (IHC) 1:500 (WB) |
| phospholipase C (PLC) | mouse | Abcam cat. no. ab243181 | 1:500 (WB) |
| kinase Raf | rabbit | Cell Signaling Technology cat. no. 9422 | 1:500 (WB) |
| extracellular signal-regulated kinase (ERK1/2) | rabbit | Cell Signaling Technology cat. no. 9102 | 1:1000 (WB) |
| β-actin | mouse | Sigma–Aldrich cat. no. A2228 | 1:3000 (WB) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duliban, M.; Gorowska-Wojtowicz, E.; Tworzydlo, W.; Rak, A.; Brzoskwinia, M.; Krakowska, I.; Wolski, J.K.; Kotula-Balak, M.; Płachno, B.J.; Bilinska, B. Interstitial Leydig Cell Tumorigenesis—Leptin and Adiponectin Signaling in Relation to Aromatase Expression in the Human Testis. Int. J. Mol. Sci. 2020, 21, 3649. https://doi.org/10.3390/ijms21103649
Duliban M, Gorowska-Wojtowicz E, Tworzydlo W, Rak A, Brzoskwinia M, Krakowska I, Wolski JK, Kotula-Balak M, Płachno BJ, Bilinska B. Interstitial Leydig Cell Tumorigenesis—Leptin and Adiponectin Signaling in Relation to Aromatase Expression in the Human Testis. International Journal of Molecular Sciences. 2020; 21(10):3649. https://doi.org/10.3390/ijms21103649
Chicago/Turabian StyleDuliban, Michal, Ewelina Gorowska-Wojtowicz, Waclaw Tworzydlo, Agnieszka Rak, Malgorzata Brzoskwinia, Izabella Krakowska, Jan K. Wolski, Malgorzata Kotula-Balak, Bartosz J. Płachno, and Barbara Bilinska. 2020. "Interstitial Leydig Cell Tumorigenesis—Leptin and Adiponectin Signaling in Relation to Aromatase Expression in the Human Testis" International Journal of Molecular Sciences 21, no. 10: 3649. https://doi.org/10.3390/ijms21103649
APA StyleDuliban, M., Gorowska-Wojtowicz, E., Tworzydlo, W., Rak, A., Brzoskwinia, M., Krakowska, I., Wolski, J. K., Kotula-Balak, M., Płachno, B. J., & Bilinska, B. (2020). Interstitial Leydig Cell Tumorigenesis—Leptin and Adiponectin Signaling in Relation to Aromatase Expression in the Human Testis. International Journal of Molecular Sciences, 21(10), 3649. https://doi.org/10.3390/ijms21103649








